Defective graphene synergizes with NiOx nanosheets to facilitate dual-defect-assisted two-electron oxygen reduction
Abstract
One of the biggest challenges in the electrochemical synthesis of H2O2 is the development of high-performance and economical catalysts. In this work, a two-dimensional composite material consisting of NiOx nanosheets and defective graphene (DG) (NiOx@DG) was prepared and showed excellent electrocatalytic performance toward electrosynthesis of H2O2 from oxygen reduction reaction. Particularly, the NiOx@DG catalysts present superior activity indicators to physical mixing counterparts (NiOx-DG), encompassing a high onset potential of 0.78 V, high efficiency and 2e- selectivity over a wide potential range between 0.20-0.60 V (maximal value of 95%). The high activity of NiOx@DG can be attributed to the dual-defects (oxygen vacancies on NiOx and topological defects on DG)-induced strong electronic metal–support interaction. Such multi-defects collaborative enhancement strategy may provide a promising avenue for the preparation of high-performance catalysts toward application in different reactions.
Keywords
INTRODUCTION
As an environmentally friendly and multipurpose oxidant, H2O2 is crucial to modern industry and is widely used in various fields including bleaching, textiles, chemical synthesis, wastewater treatment and disinfection[1,2]. Due to the inherent properties of H2O2, such as low cost, strong oxidizing tendency, the ability to eradicate pathogenic organisms, and the production of ecologically friendly byproducts of oxygen and water, traditional H2O2 wastewater recovery technologies provide ideal recovery efficiency. At present, the anthraquinone process produces about 95% of H2O2, but it comes with a high initial infrastructure cost, much energy input, a lot of byproducts, and significant safety risks when it comes to storing and transporting high concentrations of H2O2[3]. The electrochemical two-electron oxygen reduction reaction (2e- ORR) technology has recently served as a promising alternative pathway and has attracted enormous attention[4]. The key issue involving this route is the design of catalyst materials which need to convert traditional 4e- ORR to 2e- pathways[5]. Although a great number of catalysts, including precious metals[6-8], non-precious metals[9,10], metal oxides[11], carbon and its composite materials[3,12,13], have been developed and reported, their selectivity and activity are still far from practical utilization. Therefore, it is urgently required to explore new sustainable high-performance catalyst materials.
Currently, metal oxides are widely used in reactions such as OER, but they are not yet mature for 2e- ORR. However, reasonable modification of them can greatly enhance the catalytic performance of non-precious metal oxides, even rivaling that of precious metal catalysts[14]. For example, amorphous materials are one such type. Recently, it has been found that some amorphous nanomaterials, including metal oxides, layered double hydroxides, and spinel compounds, exhibit high activity for electrochemical reactions due to their tunable composition, homogeneity, lattice defect-induced active sites, and unsaturated coordination active sites[15-17]. Moreover, under potentials where electrical conductivity is not dominant, amorphous metal oxides exhibit higher electrocatalytic activity for the oxygen evolution reaction (OER) compared to crystalline metal oxides[18-20]. This is because they are isotropic and single-phase, and unsaturated cation sites with more coordination are available for the reaction[21]. Although early results suggest that there is very little research on amorphous metal oxides as 2e- ORR catalysts, the potential for amorphous materials to become competitive catalysts remains.
Carbon materials possess the advantages of high abundance, low cost, and strong electroreduction activity, along with tunable physicochemical properties and outstanding stability in corrosive acids. Heteroatom doping and intrinsic defects can be used to modify the electrical characteristics of carbon-based materials, which are intimately linked to their electrocatalytic activity and selectivity. For example, doping heteroatoms (N, S, B, P, etc.) into the carbon carriers has been shown to increase catalytic activity by inducing a local electronic structural rearrangement because of their distinct radii and electronegativities[22-26]. Additionally, the electrical neutrality of the conventional planar hexagonal graphite lattice can be broken by structural defects in carbon-based matrices, leading to asymmetric local electron redistribution. This greatly improves the electrocatalytic performance of non-metallic carbon materials[27-30]. Crucially, the neutral binding of the intermediate *OOH is a major determinant of both activity and selectivity. The adsorption energy of *OOH (ΔG*OOH) may be optimized by amorphous catalysts with a large number of defect sites and disordered structures under thermodynamic equilibrium potentials[31]. Therefore, we expect that a combination of oxygen-deficient metal oxides and defective carbon materials may vary the local interfacial electronic properties via the dual-defect engineering to boost the 2e- ORR performance.
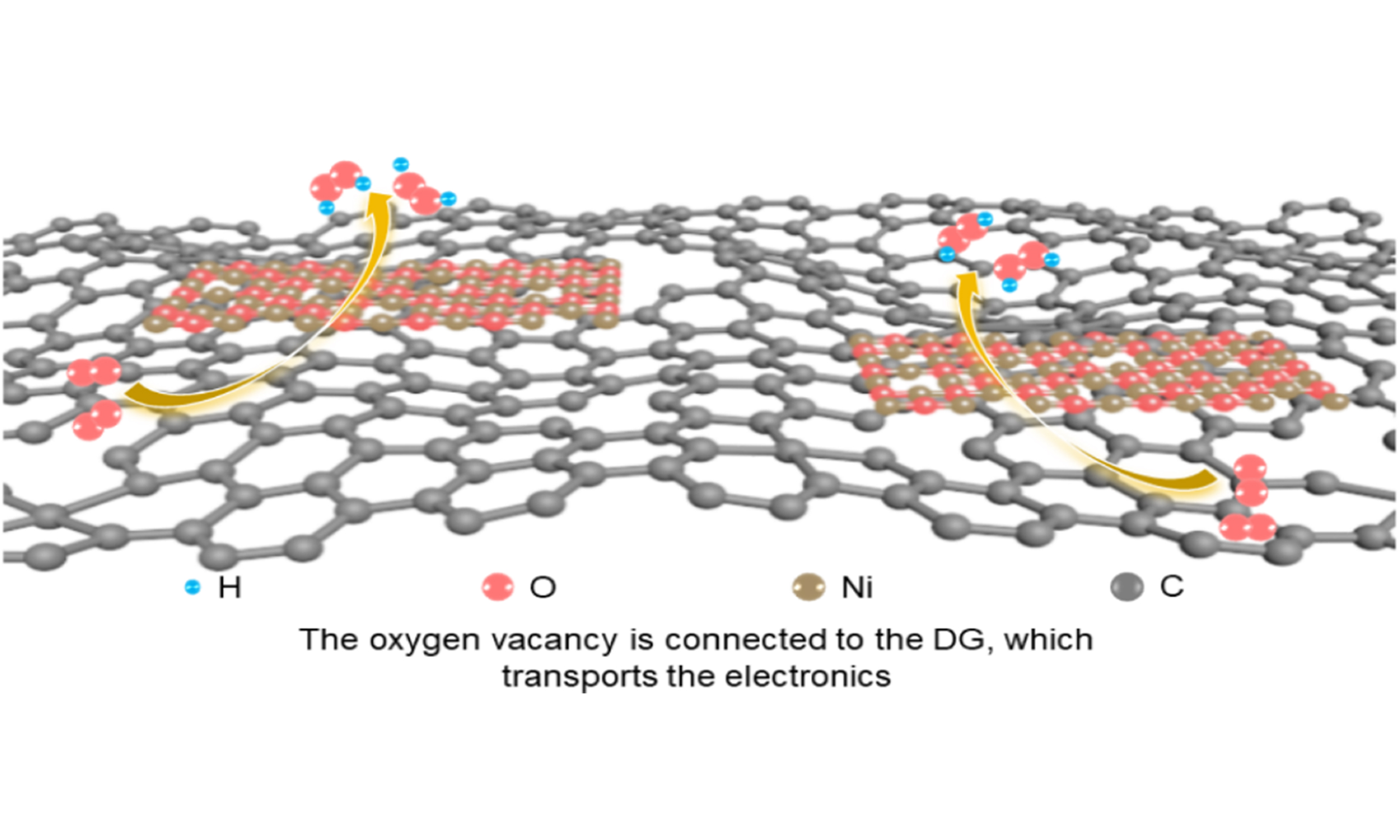
In this work, we report the synthesis of oxygen-deficient NiOx nanosheets (NiOx NS) constructed at low temperatures and hydrothermally combined with defective graphene (DG), forming a synergistic effect between oxygen vacancies and defective topology. During this process, electrons from the defective topology flow through the oxygen vacancies to the Ni center, modulating the charge density at the Ni sites in the reaction system and facilitating the electrochemical synthesis of H2O2. This further highlights the significant potential of defect engineering in carbon-supported oxides for electrochemical conversion applications.
EXPERIMENTAL
Materials and synthesis methods
Every reagent was acquired from commercial sources and used as received without extra purification. Nafion (5%), Potassium nitrate (KNO3, AR) and Melamine (C3H6N6, 99%) were purchased from Aladdin. Potassium hydroxide (KOH, ≥ 85%) was purchased from Titan Technology. Nickel acetylacetone
Preparation of NiOx nanosheet
Dissolve 30 mg of nickel acetylacetonate (C10H14NiO4) and 60 mg of potassium nitrate in a mixed solution of deionized water and ethanol (V ethanol: V water = 5:1). After ultrasonication for one hour, evaporate the solvent to obtain the precursor. Place the precursor in a crucible and put it in a muffle furnace, heating at a rate of 5
Preparation of DG
Uniformly mix 30 mg of G with 30 mg of melamine and place the mixture on a porcelain boat. Under a nitrogen (N2) atmosphere, heat the mixture at a rate of 5 °C·min-1 from room temperature to 700 °C, and hold for 2 h; then, under the same atmosphere, increase the temperature at a rate of 5 °C·min-1 from 700 to 1,150 °C, hold for another 2 h, and allow the sample to cool to room temperature to obtain DG.
Take an appropriate amount of the prepared DG and disperse it in water to make a mixed solution with a concentration of 0.1 mg·mL-1. Sonicate the solution for 8 h to obtain the DG dispersion.
Preparation of Nitrogen-doped graphene (NG)
Uniformly mix 30 mg of G with 30 mg of melamine and place the mixture on a porcelain boat. Under a N2 atmosphere, heat the mixture at a rate of 5 °C·min-1 from room temperature to 700 °C, and hold for 2 h.
Preparation of NiOx@DG
Mix the prepared NiOx dispersion with the DG dispersion in mass ratios of 6:1 and 9:1 for NiOx to DG, respectively, under ultrasonication. Place the mixture in a 50 mL polytetrafluoroethylene (PTFE)-lined autoclave and hydrothermally treat at 60 °C for 12 h. Allow the product to cool down to room temperature after the treatment.
Preparation of NiO nanosheet
The synthesis method is similar to the aforementioned method for NiOx, with the only difference being the absence of KNO3.
Preparation of NiOx NS-550
The synthesis method is similar to the aforementioned method for NiOx, with the only difference being the temperature of 550 °C.
Preparation of NiOx-DG
The synthesis method is similar to the aforementioned method for NiOx@DG, with the only difference being the physical mixing.
Preparation of NiOx@NG, NiOx@G, NiO@DG, NiO@NG and NiO@G
The synthesis method is similar to the aforementioned method for NiOx@DG, with the only difference being the hydrothermal treatment with the corresponding substance.
Materials characterizations
A Zeiss Geminisem 500 (Carl Zeiss AG, Germany) microscope operating at 5 kV was used to acquire scanning electron microscopy (SEM) pictures. Energy dispersive X-ray spectroscopy (EDX) mapping pictures, high-resolution transmission electron microscopy (HRTEM), high angle annular dark field scanning transmission electron microscopy (TEM), and TEM were acquired on the JEOL JEM-2100F (JEOL Ltd., Japan) 300 kV. The Rigaku Ultima IV (JEOL Ltd., Japan) was used to record X-ray diffraction (XRD) patterns. It operated at 40 kV with Cu Kα radiation (λ = 0.15406 nm), a scan rate of 10°/min, and a theta range of 5° to 90°. Raman spectra were obtained using a laser with a wavelength of 532 nm on a Raman spectrometer system (Raman spectrometer model: HORIBA HR Evolution, HORIBA, Ltd., France). The Brunauer-Emmett-Teller (BET) surface area of +the resultant materialthe resultant materialthe resultant material was calculated using a relative pressure of P/P0 = 0.05-0.1. An Al-Ka radiation-powered Thermo Scientific K-Alpha device (Thermo Fisher Scientific, America) operating at 12 kV was used to perform X-ray photoelectron spectroscopy (XPS). The beamline 4B9A at the Beijing Synchrotron Radiation Facility (BSRF) in Beijing, China, has an optical emission end station where the Ni-K-edge X-ray absorption near-edge structure (XANES) spectra of the catalysts were examined.
Electrochemical measurements
Through the use of the CHI760E electrochemical workstation, a three-electrode system is used for all electrochemical testing. An Ag/AgCl electrode (saturated KCl solution), a carbon rod, and a rotating ring disc electrode (RRDE) (glassy carbon disc, 4 mm diameter, 5 mm inner diameter, 7 mm outer diameter, Pt ring) make up a three-electrode system. The platinum wire was used as the counter electrode and the Ag/AgCl electrode was used as the reference electrode. The reversible hydrogen electrode (RHE), ERHE = EAg/AgCl + 0.197 + 0.0592*PH[32], was used to calibrate the reference electrode. Materials 5 mg of the sample, 10 uL of Nafion (5 weight percent), 700 uL of water, and 290 uL of isopropanol were sonicated for 1 h to create catalyst ink. The disc electrode was then covered with 5 uL of the dispersion, which was allowed to dry at room temperature. At the disc electrode, the catalyst’s mass loading was 0.20 mg·cm-2. Before testing, O2 was passed into 0.1 M KOH for 30 min to make sure O2 saturation was achieved. Linear scanning voltammetry (LSV) tests were performed at 1,600 rpm in an O2-saturated 0.1 M KOH electrolyte. Repeat at a scan rate of 10 mV·s-1 over a potential range of 0.1 V vs. RHE to 1.0 V vs. RHE. The ring potential was also held at 1.2 V vs. RHE. Normalize all current densities to the area of the disc electrode.
The hydrogen peroxide yield [H2O2 (%)] and the number of electron transfers (n) are calculated based on[33]
Where Id represents the disk current, Ir denotes the ring current, and N = 0.42 signifies the current collection efficiency of the platinum ring.
RESULTS AND DISCUSSION
Morphology and structure
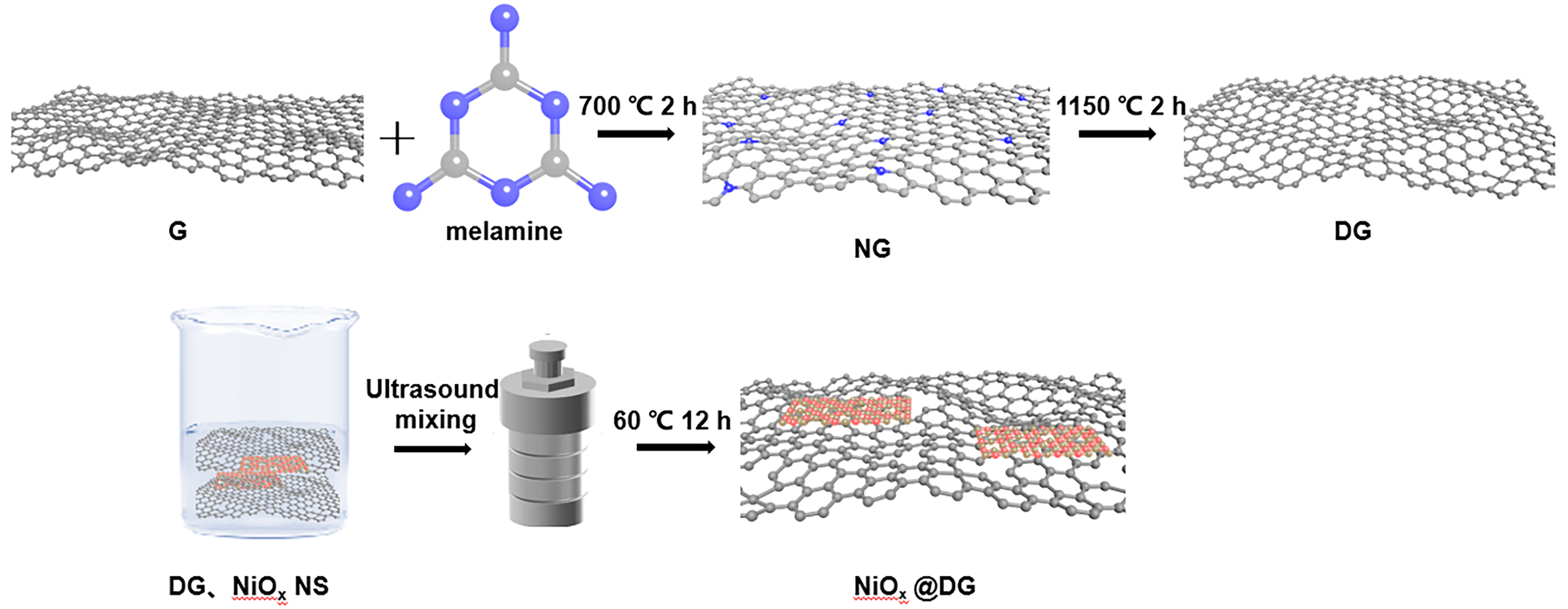
NiOx@DG was synthesized following the procedure depicted in Scheme 1, and detailed information can be found in the Supporting Information. In simple terms, NiOx powder was obtained by heating nickel acetylacetone and KNO3 in a muffle furnace at 350 °C for 90 min. In the XRD result [Supplementary Figure 1], it was concluded from the observation of characteristic peaks that the XRD of bulk NiOx showed the characteristic peaks of NiO. This was because KNO3 can act as an oxidizing base in this process, providing O2- to form metal oxides[34]. Furthermore, it was evident that the characteristic peaks of NiOx bulk are broader and lower compared to those of NiO. This was attributed to the introduction of oxygen vacancies, which disrupt the order of the oxide’s lattice structure. In the SEM images [Supplementary Figure 2A], we can observe its rough surface, and the layered structure can be clearly seen (indicated by the white arrows in the image). However, no distinct particles were observed in the TEM images [Supplementary Figure 2B]. Nevertheless, HRTEM further describes the morphological structure of the NiOx powder and the spatial distribution of the NiO particles within it. The presence of small black particles as NiO is indicated [enclosed by red dashed lines in Supplementary Figure 2C], and the lattice fringe spacing of 0.244 nm corresponds to the interplanar spacing of the (111) plane of NiO [Supplementary Figure 2D-F], a result that was consistent with the XRD analysis. The elemental mapping results [Supplementary Figure 2G] showed that Ni and O elements were uniformly distributed on the surface of the carbon carrier. Subsequently, the NiOx powder was placed in ethanol and sonicated to exfoliate NiOx NS, and the upper clear liquid was taken to remove larger NiO crystal particles and incompletely exfoliated bulk NiOx. This was followed by hydrothermal treatment with DG (for detailed procedures, see Supporting Information) to obtain the final NiOx@DG composite. In addition, control samples, including physical mixtures of NiOx and DG (NiOx-DG) and NiOx@DG-9 composite with a changed mass ratio (NiOx:DG = 9:1), were also prepared.
Scheme 1. The schematic procedure for synthesizing the NiOx@DG; The gray balls represent C, the blue balls represent N, the red balls represent Ni, and the brown balls represent O. DG: Defective graphene; NG: nitrogen-doped graphene.
We characterized the composited sample to confirm the completion of exfoliation. From the XRD image in [Supplementary Figure 3], it can be observed that the characteristic peaks of the oxides disappear after the exfoliation and composite process. This indicated that centrifugation can separate the big crystalline particles and bulk NiOx, leaving behind NiOx NS in the upper clear liquid. In the SEM image [Figure 1A], NiOx NS with a diameter of approximately 100 nm can be observed loaded on DG, and the presence of these nanosheets was also observed in the TEM image [Figure 1B]. The average thickness of NiOx@DG was measured to be about 2 nm using atomic force microscopy (AFM) [Figure 1C and D]. HRTEM and corresponding inverse fast Fourier transform (IFFT) images were used to investigate the presence of crystalline structures of NiO [Figure 1E and F and Supplementary Figure 4]. In specific areas (enclosed by red boxes), irregular atomic arrangements were observed, indicating the presence of characteristic features of defective structures, which was caused by the overflow of lattice oxygen, consistent with the XRD results. Line scan analysis and elemental mapping spectra revealed the uniform distribution of C, O and Ni over the composite catalysts [Figure 1G and H and Supplementary Figure 5].
Figure 1. (A) SEM image, (B) TEM image, (C) AFM image of NiOx@DG and (D), Height profile along the line of NiOx@DG; (E) HRTEM image and (F) The enlarged HRTEM image within the red dashed frame marks the local area of disordered atoms; (G and H) SEM and line-scan element mapping for NiOx@DG. SEM: Scanning electron microscopy; TEM: transmission electron microscopy; AFM: atomic force microscopy; DG: defective graphene; HRTEM: high-resolution transmission electron microscopy.
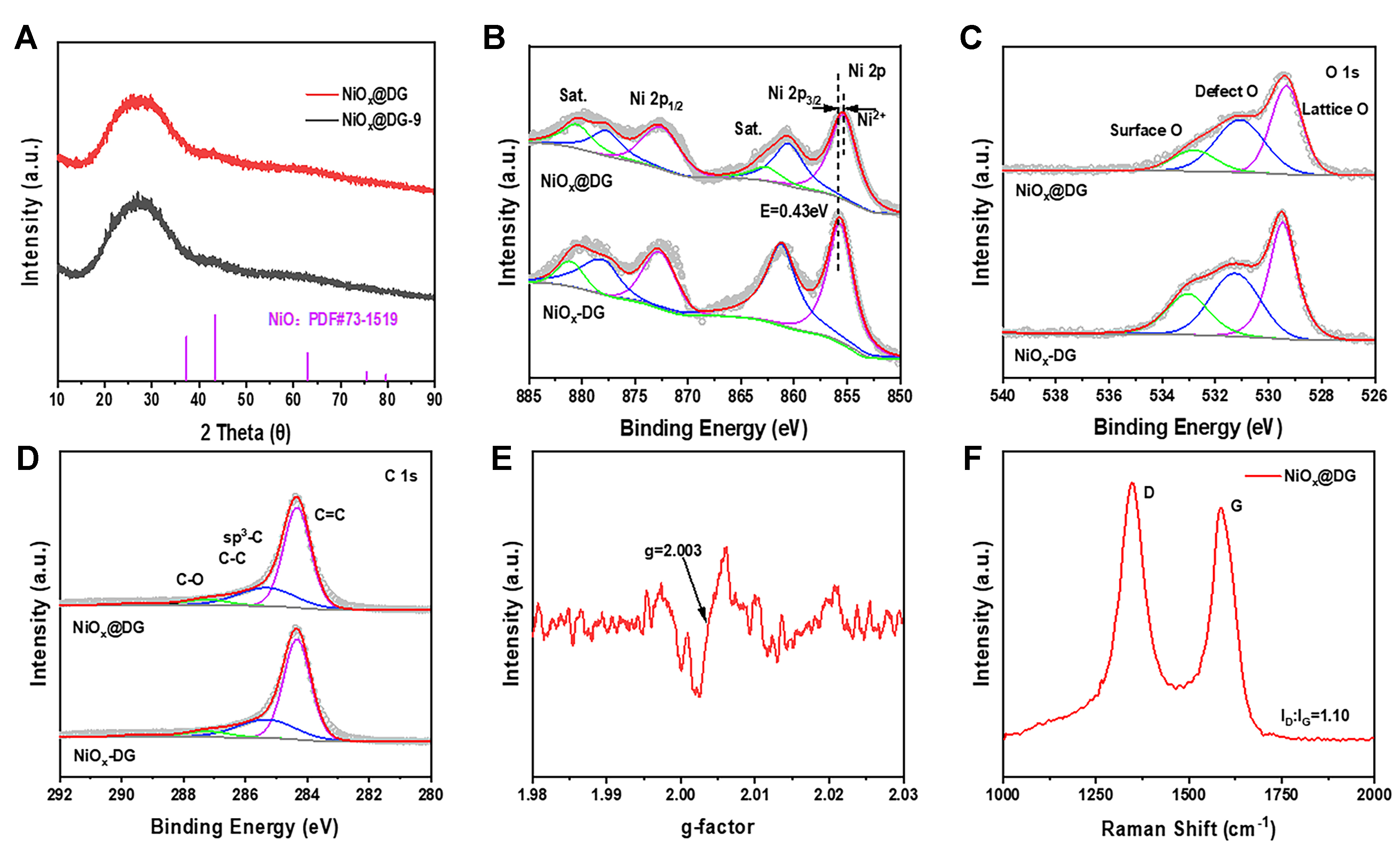
The powder XRD (PXRD) patterns [Figure 2A] verified that no clear typical diffraction peaks of metal oxides were present in NiOx@DG and NiOx@DG-9, with the exception of a broad peak at about 26° corresponding to the (002) plane of graphitic carbon. Subsequently, the chemical properties of the samples were thoroughly investigated using XPS. The high-resolution XPS spectra of Ni 2p for NiOx@DG and NiOx-DG are shown in Figure 2B and Supplementary Figure 6. These spectra can be well-deconvoluted into several sub-peaks that correspond to Ni2+ (nearly 855.7 eV) and two associated satellite peaks. Compared to NiOx-DG, there is a noticeable negative shift of around 0.45 eV in the Ni2+ peak of NiOx@DG, indicating that Ni acts as an electron acceptor to receive electrons transferred from DG. However, there is no significant shift in the Ni 2p XPS spectra between NiOx NS-350 and NiOx-DG [Supplementary Figure 7], implying that simple physical mixing cannot alter the electronic structure of the Ni centers. The Ni 2p XPS spectrum of NiOx@DG-9 also shows two peaks [Supplementary Figure 8], the oxidation peak of Ni (2+) at a binding energy of ~ 855.7 eV, and the associated satellite peak, which is consistent with the results of Ni oxides observed in XRD and HRTEM. A larger negative shift in binding energy of 0.8 eV can be observed, suggesting that higher loading of NiOx NS in NiOx@DG-9 is beneficial for more oxygen vacancies to communicate with DG, thereby gaining more electrons. In the high-resolution XPS spectrum of O 1s [Figure 2C], three peaks can be observed, which were assigned to lattice oxygen at 529.93 eV, surface adsorbed oxygen at 532.63 eV, and lattice oxygen near oxygen vacancies at 531.4 eV. The Od/Ot (Od represents oxygen vacancy content and Ot indicates the total O content) ratio of NiOx@DG was 20%, lower than that of NiOx-DG (36.5%), which may be attributed to the increased surface adsorbed oxygen during the hydrothermal treatment to synthesize NiOx@DG composite[35]. Figure 2D showed the high-resolution XPS spectra of C 1s, which can be deconvoluted into three subpeaks, corresponding to C=C (sp2 carbon,
Figure 2. (A) PXRD patterns; (B) Ni 2p XPS spectra; (C) N 1s XPS spectra and (D) C 1s XPS spectra of NiOx@DG and NiOx-DG; (E) EPR spectra of NiOx@DG; (F) Raman spectroscopy of NiOx@DG. PXRD: Powder X-ray diffraction; XPS: X-ray photoelectron spectroscopy; DG: defective graphene.
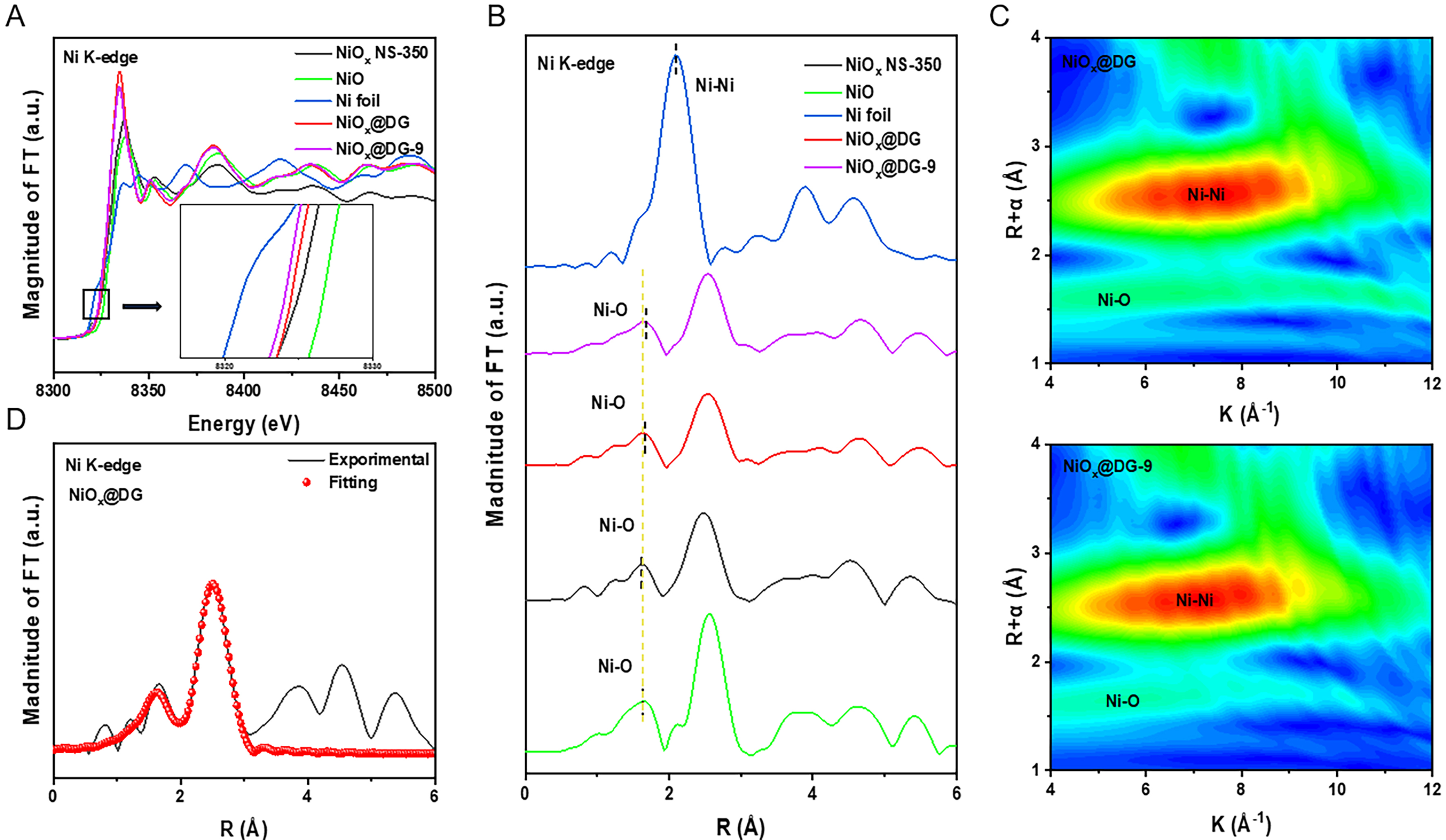
Extended X-ray absorption fine structure (EXAFS) and XANES studies were performed to further determine the local coordination environment of Ni in the catalyst. The Ni-K edge XANES spectra for the NiOx@DG and reference materials are shown in Figure 3A. The absorption edges of these samples were located between the absorption edge of Ni foil and NiO. The Ni valence state was between 0 and +2, which was consistent with the XPS data. The Fourier-transformed EXAFS curves for NiOx@DG, NiOx@DG-9, and NiOx NS-350 also showed the presence of Ni-O, with a peak in the R-space at approximately 1.68 Å, confirming that the Ni species primarily exist in the form of NiO clusters [Figure 3B]. The intensity of the peak is related to the number of atoms with the same average interatomic distance and the distribution of Ni centers[39]. The introduction of oxygen vacancies led to the reduction of central Ni species, resulting in a decrease in coordination number and an increase in bond length compared to NiO, which was consistent with other literature reports[40]. There is no Ni-Ni scattering channel at ~ 2.2 Å that corresponds to the crystal nickel metal structure. In the meantime, the Ni-Oxo backward scattering path of NiO is closely matched by another prominent peak at about 2.6 Å[41]. The wavelet transform (WT)-EXAFS method can be used to distinguish backscattered atoms in R space and K space. Figure 3C and Supplementary Figure 11 demonstrated that the WT contour plots of the various reference samples show Ni-O at ~ 5.0 Å-1; the intensity of Ni-O is strongest at ~ 7.3 Å-1 in WT contour plots of different prepared samples, which corresponds to the Ni-Ni coordination structure of NiO and further indicates the presence of NiOx. Finally, in order to accurately determine the local coordination environment of the Ni species, quantitative EXAFS fitting was carried out. The fitting results in Figure 3D and Supplementary Table 1 show that the Ni-O coordination number in NiOx@DG is approximately 4.46, less than the Ni-O coordination number of bulk NiO (6.0). This result confirms the existence of oxygen vacancies.
Figure 3. (A) Normalized Ni K-edge XANES spectra of NiOx@DG and reference materials; (B) FT of Ni K-edge EXAFS data of NiOx@DG and reference materials. The boxed area is enlarged in Figure 3A’s inset; (C) Ni K-edge WT-EXAFS contour plots for NiOx@DG and reference materials; (D) Ni K-edge EXAFS fitting curves for NiOx@DG. XANES: X-ray absorption near-edge structure; DG: defective graphene; FT: fourier transform; EXAFS: extended X-ray absorption fine structure; WT: wavelet transform.
Performance testing
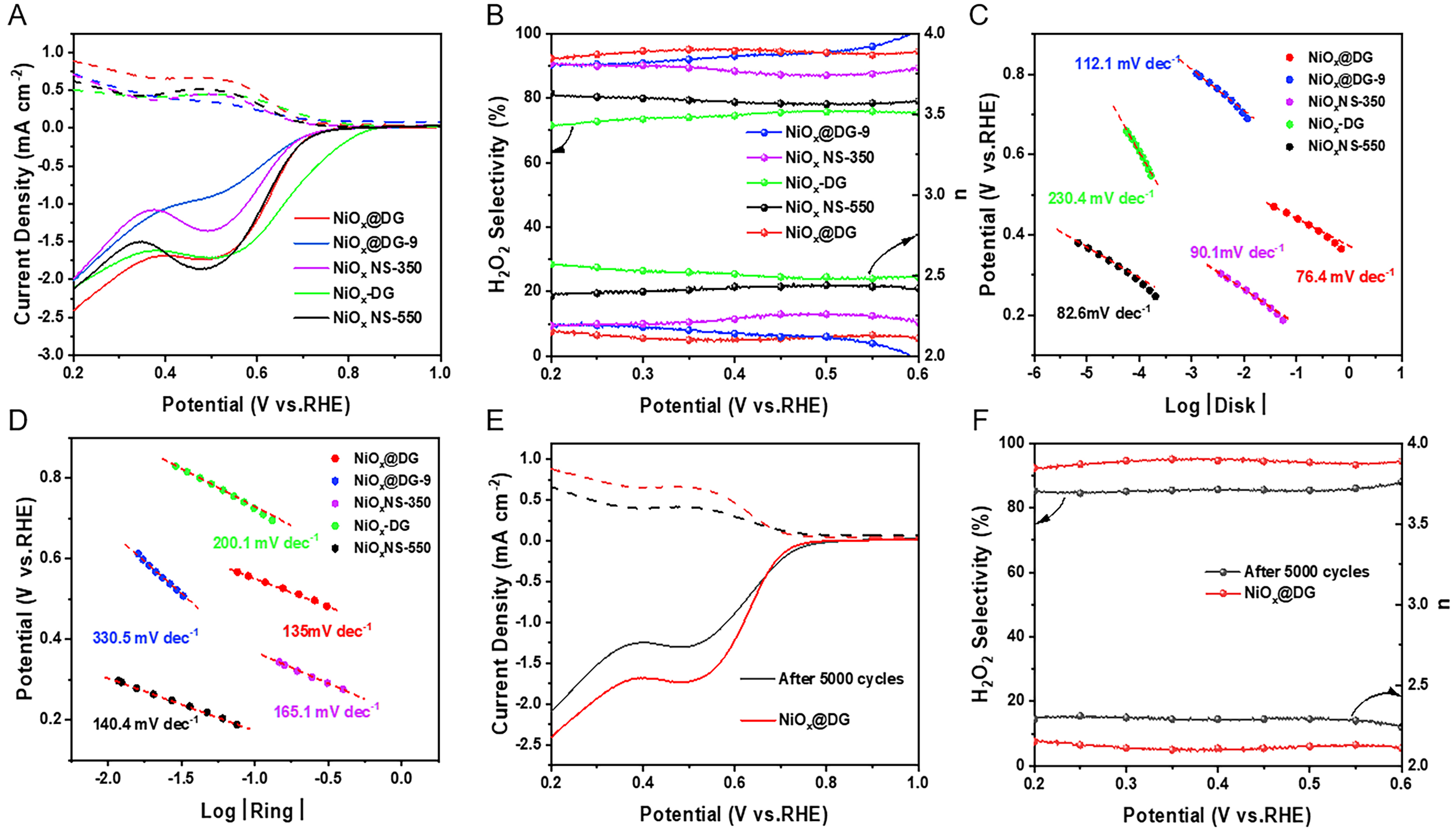
RRDE on a three-electrode system was used to assess the ORR performance of the catalysts in 0.1 M KOH solution. We first studied the effect of catalyst loading amount on ORR activity. As shown in Supplementary Figure 12, the optimal catalytic performance can be achieved when the loading amount is below
Figure 4. (A) Polarization curves of NiOx@DG, NiOx@DG-9, NiOx-DG, NiOx NS-350 and NiOx NS-550 on RRDE at 1,600 rpm in O2-saturated 0.1 M KOH; (B) The calculated H2O2 selectivity and Transfer electron n of catalysts; (C and D) Tafel slope of NiOx@DG, NiOx@DG-9, NiOx-DG, NiOx NS-350 and NiOx NS-550; (E and F) Stability test of NiOx@DG catalyst after 5,000 cycles. DG: Defective graphene; RRDE: rotating ring disc electrode; n: number; RHE: reversible hydrogen electrode.
To further confirm the synergistic effect of oxygen vacancies in NiOx and topological defects in DG for the electrosynthesis of hydrogen peroxide, two single-defect samples (NiOx@G and NiO@DG) were prepared for comparison. As illustrated in Supplementary Figures 15 and 16, both NiOx@G and NiO@DG manifested significantly reduced H2O2 selectivity compared with NiOx@DG composites, emphasizing the critical role of synergy between interfacial oxygen vacancies and carbon defects toward 2e- ORR. Noteworthily, the ORR pathway on the NiO@DG sample is 4e- process, while NiOx@G still preserves certain electrosynthesis performance of H2O2. In addition, comparing the ORR performance between NiO@DG and NiO@G
Doping heteroatoms in carbon materials is also well-accepted to efficiently regulate the atomic and electronic configurations besides the intrinsic defect engineering. Hence, we further studied whether doping N2 atoms in G can also boost the H2O2 electroproduction performance, similar to the effect of topological defects. As shown in Supplementary Figures 18 and 19, N dopants in G cannot lead to activity enhancement in 2e- ORR as compared to NiO@G and NiOx@G, which indicates its uselessness in such composite system toward ORR process. To directly uncover the influence of the G substrate itself on ORR processes, a series of G-based carbon materials were prepared with similar methods at different temperatures (800-1,150 °C, denoted as NG-800, NG-900, NG-1,000 and DG-1,150 °C) were prepared. The defect structure and elemental state were first characterized by Raman and XPS spectra, showing that N2 content decreased while the defect density rose as the pyrolysis temperature increased, consistent with previous reports that removal of N2 dopants in carbon lattice can induce the rearrangement of surrounding carbon atoms to form more topological defects [Supplementary Figure 20A and B]. The ORR activities of these carbon samples were then evaluated on RRDE and the results were presented in Supplementary Figure 20C and D. Apparently, the ORR processes on all the carbon materials without coupled NiO species are close to 4e- transfer pathway, indicating that neither N dopants nor carbon defects can directly boost the desorption of generated H2O2 to prevent the cleavage of O-O bonds. This can further confirm our speculation that, in NiOx@DG composites, Ni atoms beside oxygen vacancies act as the real catalytic centers for O2 adsorption and activation, while the interfacial carbon defects contribute to optimizing the electronic structure of active Ni atom to achieve the final excellent 2e- ORR activity.
Density functional theory (DFT) calculations were further conducted to gain a deep understanding on the activity mechanism. As pentagon defects in carbon lattice have been demonstrated to show excellent electrocatalytic performance in various reactions, here we choose G lattice possessing pentagon defects as the defective carbon model. Accordingly, three composite models were constructed: NiO with one oxygen vacancy coupled DG (NiOx@DG) [Supplementary Figure 21A], NiO with one oxygen vacancy coupled perfect G (NiOx@G) [Supplementary Figure 21B], and NiO lattice without oxygen vacancies coupled DG (NiO@DG) [Supplementary Figure 21C]. The models with adsorbed *OOH intermediates were also presented in Supplementary Figure 21. It can be seen that, only in NiOx@DG model, the *OOH is laterally adsorbed on the NiOx layer, which may facilitate the elongation of the O–O bond[16], and lead to the consequent desorption process. The adsorption energy of the key intermediate *OOH is generally used as a key descriptor for predicting the 2e- ORR.[15] As shown in Supplementary Figure 21D, NiOx@DG model has the lowest energy barrier (1.24 eV) under alkaline conditions as compared to the other two models, indicating its energetically favorable 2e- ORR process due to the enhanced intermediate desorption capacity.
CONCLUSION
In summary, we prepared oxygen-deficient NiOx using a low-temperature molten salt method and conducted a composite process with DG (NiOx@DG). This not only enhanced the conductivity but also connected the DG to the Ni centers through oxygen vacancies, increasing the electron cloud density of Ni and improving the 2e- ORR activity of the catalyst. The oxygen vacancies acted as a bridge for electron transport, facilitating the transfer from the topological defects of the DG to the Ni centers. The catalyst achieved a 95% selectivity for hydrogen peroxide under alkaline conditions, with a Tafel slope of
DECLARATIONS
Acknowledgements
The authors would like to thank Shi-yanjia Lab (www.shiyanjia.com) for material characterizations and thank facility support of the 4B9A beamline of Beijing Synchrotron Radiation Facility (BSRF).
Authors’ contributions
Conceptualization, investigation, methodology, data curation, formal analysis, DFT calculations, writing - original draft: Huang, R.; Wu, H.; Fang, Q.; Chen, Y.; Guo, P.; Liu, X.; Huang, M.; Zhang, J.; Du, A.
Conceptualization, funding acquisition, resources, supervision, project administration, writing - review and editing: Wang, L.; Wang, X.
Availability of data and materials
The authors confirm that the data supporting the findings of this study are available within its Supplementary Materials.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool Kimi (version Enterprise API, released online in China in August 2024) was used solely for language editing. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (NSFC, Nos. 22275165, 22101254, and 52403118).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Jiang, Y.; Ni, P.; Chen, C.; et al. Selective electrochemical H2O2 production through two-electron oxygen electrochemistry. Adv. Energy. Mater. 2018, 8, 1801909.
2. Sun, Y.; Han, L.; Strasser, P. A comparative perspective of electrochemical and photochemical approaches for catalytic H2O2 production. Chem. Soc. Rev. 2020, 49, 6605-31.
3. Wu, Z.; Wang, T.; Zou, J.; Li, Y.; Zhang, C. Amorphous nickel oxides supported on carbon nanosheets as high-performance catalysts for electrochemical synthesis of hydrogen peroxide. ACS. Catal. 2022, 12, 5911-20.
4. Tian, Q.; Jing, L.; Wang, W.; et al. Utilizing carbonaceous catalysts for H2O2 electrosynthesis via the two-electron oxygen reduction reaction. Energy. Lab. 2025, 3, 240019.
5. Liu, W.; Zhang, C.; Zhang, J.; et al. Tuning the atomic configuration of Co-N-C electrocatalyst enables highly-selective H2O2 production in acidic media. Appl. Catal. B. Environ. 2022, 310, 121312.
6. Jirkovský, J. S.; Panas, I.; Ahlberg, E.; Halasa, M.; Romani, S.; Schiffrin, D. J. Single atom hot-spots at Au-Pd nanoalloys for electrocatalytic H2O2 production. J. Am. Chem. Soc. 2011, 133, 19432-41.
7. Siahrostami, S.; Verdaguer-Casadevall, A.; Karamad, M.; et al. Enabling direct H2O2 production through rational electrocatalyst design. Nat. Mater. 2013, 12, 1137-43.
8. Ko, Y.; Choi, K.; Yang, B.; et al. A catalyst design for selective electrochemical reactions: direct production of hydrogen peroxide in advanced electrochemical oxidation. J. Mater. Chem. A. 2020, 8, 9859-70.
9. Xie, J.; Zhong, L.; Yang, X.; et al. Phosphorous and selenium tuning Co-based non-precious catalysts for electrosynthesis of H2O2 in acidic media. Chin. Chem. Lett. 2024, 35, 108472.
10. Jung, E.; Shin, H.; Lee, B. H.; et al. Atomic-level tuning of Co-N-C catalyst for high-performance electrochemical H2O2 production. Nat. Mater. 2020, 19, 436-42.
11. Yan, L.; Cheng, X.; Wang, Y.; et al. Exsolved Co3O4 with tunable oxygen vacancies for electrocatalytic H2O2 production. Mater. Today. Energy. 2022, 24, 100931.
12. Sun, Y.; Sinev, I.; Ju, W.; et al. Efficient electrochemical hydrogen peroxide production from molecular oxygen on nitrogen-doped mesoporous carbon catalysts. ACS. Catal. 2018, 8, 2844-56.
13. Liu, Y.; Quan, X.; Fan, X.; Wang, H.; Chen, S. High-yield electrosynthesis of hydrogen peroxide from oxygen reduction by hierarchically porous carbon. Angew. Chem. 2015, 127, 6941-5.
14. Wang, M.; Zhang, N.; Feng, Y.; Hu, Z.; Shao, Q.; Huang, X. Partially pyrolyzed binary metal-organic framework nanosheets for efficient electrochemical hydrogen peroxide synthesis. Angew. Chem. Int. Ed. Engl. 2020, 59, 14373-7.
15. Cai, P.; Huang, J.; Chen, J.; Wen, Z. Oxygen-containing amorphous cobalt sulfide porous nanocubes as high-activity electrocatalysts for the oxygen evolution reaction in an alkaline/neutral medium. Angew. Chem. Int. Ed. Engl. 2017, 56, 4858-61.
16. Indra, A.; Menezes, P. W.; Zaharieva, I.; et al. Active mixed-valent MnOx water oxidation catalysts through partial oxidation (corrosion) of nanostructured MnO particles. Angew. Chem. Int. Ed. Engl. 2013, 52, 13206-10.
17. Yin, S.; Tu, W.; Sheng, Y.; et al. A highly efficient oxygen evolution catalyst consisting of interconnected nickel-iron-layered double hydroxide and carbon nanodomains. Adv. Mater. 2018, 30, 1705106.
18. Zhang, C.; Zhang, X.; Daly, K.; Berlinguette, C. P.; Trudel, S. Water oxidation catalysis: tuning the electrocatalytic properties of amorphous lanthanum cobaltite through calcium doping. ACS. Catal. 2017, 7, 6385-91.
19. Smith, R. D.; Prévot, M. S.; Fagan, R. D.; et al. Photochemical route for accessing amorphous metal oxide materials for water oxidation catalysis. Science 2013, 340, 60-3.
20. Pan, L.; Wang, Q.; Li, Y.; Zhang, C. Amorphous cobalt-cerium binary metal oxides as high performance electrocatalyst for oxygen evolution reaction. J. Catal. 2020, 384, 14-21.
21. Li, X.; Cai, W.; Li, D.; Xu, J.; Tao, H.; Liu, B. Amorphous alloys for electrocatalysis: the significant role of the amorphous alloy structure. Nano. Res. 2023, 16, 4277-88.
22. Xia, Y.; Zhao, X.; Xia, C.; et al. Highly active and selective oxygen reduction to H2O2 on boron-doped carbon for high production rates. Nat. Commun. 2021, 12, 24329.
23. Jiao, Y.; Zheng, Y.; Jaroniec, M.; Qiao, S. Z. Origin of the electrocatalytic oxygen reduction activity of graphene-based catalysts: a roadmap to achieve the best performance. J. Am. Chem. Soc. 2014, 136, 4394-403.
24. Chen, G.; Liu, J.; Li, Q.; et al. A direct H2O2 production based on hollow porous carbon sphere-sulfur nanocrystal composites by confinement effect as oxygen reduction electrocatalysts. Nano. Res. 2019, 12, 2614-22.
25. Zhang, J.; Zhang, J.; He, F.; et al. Defect and doping Co-engineered non-metal nanocarbon ORR electrocatalyst. Nanomicro. Lett. 2021, 13, 65.
26. Melchionna, M.; Fornasiero, P.; Prato, M. The rise of hydrogen peroxide as the main product by metal-free catalysis in oxygen reductions. Adv. Mater. 2019, 31, 1802920.
27. Chen, S.; Chen, Z.; Siahrostami, S.; et al. Defective carbon-based materials for the electrochemical synthesis of hydrogen peroxide. ACS. Sustainable. Chem. Eng. 2018, 6, 311-7.
28. Zhang, L.; Xu, Q.; Niu, J.; Xia, Z. Role of lattice defects in catalytic activities of graphene clusters for fuel cells. Phys. Chem. Chem. Phys. 2015, 17, 16733-43.
29. Zhu, J.; Huang, Y.; Mei, W.; et al. Effects of intrinsic pentagon defects on electrochemical reactivity of carbon nanomaterials. Angew. Chem. Int. Ed. 2019, 58, 3859-64.
30. Wang, W.; Shang, L.; Chang, G.; et al. Intrinsic carbon-defect-driven electrocatalytic reduction of carbon dioxide. Adv. Mater. 2019, 31, e1808276.
31. Huang, J.; Fu, C.; Chen, J.; Senthilkumar, N.; Peng, X.; Wen, Z. The enhancement of selectivity and activity for two-electron oxygen reduction reaction by tuned oxygen defects on amorphous hydroxide catalysts. CCS. Chem. 2022, 4, 566-83.
32. Shen, S.; Jiang, J.; Guo, P.; et al. Effect of Cr doping on the photoelectrochemical performance of hematite nanorod photoanodes. Nano. Energy. 2012, 1, 732-41.
33. Paulus, U. A.; Schmidt, T. J.; Gasteiger, H. A.; Behm, R. J. Oxygen reduction on a high-surface area Pt:Vulcan carbon catalyst: a thin-film rotating ring-disk electrode study. J. Electroanal. Chem. 2001, 495, 134-45.
34. FLOOD, H.; FORLAND, T. The acidic and basic properties of oxides. Acta. Chem. Scand. 1947, 1, 592-604.
35. Deming, C. P.; Mercado, R.; Gadiraju, V.; Sweeney, S. W.; Khan, M.; Chen, S. Graphene quantum dots-supported palladium nanoparticles for efficient electrocatalytic reduction of oxygen in alkaline media. ACS. Sustainable. Chem. Eng. 2015, 3, 3315-23.
36. Liu, Q.; Zhu, Y.; He, Z.; Jin, S.; Chen, Y. A facile top-down approach for constructing perovskite oxide nanostructure with abundant oxygen defects as highly efficient water oxidation electrocatalyst. Int. J. Hydrogen. Energy. 2020, 45, 22808-16.
37. Kim, C.; Park, S. O.; Kwak, S. K.; Xia, Z.; Kim, G.; Dai, L. Concurrent oxygen reduction and water oxidation at high ionic strength for scalable electrosynthesis of hydrogen peroxide. Nat. Commun. 2023, 14, 41397.
38. Wei, Z.; Wang, H.; Zhang, C.; Xu, K.; Lu, X.; Lu, T. Reversed charge transfer and enhanced hydrogen spillover in platinum nanoclusters anchored on titanium oxide with rich oxygen vacancies boost hydrogen evolution reaction. Angew. Chem. Int. Ed. 2021, 60, 16622-7.
39. Huang, H.; Huang, A.; Liu, D.; et al. Tailoring oxygen reduction reaction kinetics on perovskite oxides via oxygen vacancies for low-temperature and knittable Zinc-air batteries. Adv. Mater. 2023, 35, e2303109.
40. Shao, Z.; Zhu, Q.; Sun, Y.; et al. Phase-reconfiguration-induced NiS/NiFe2O4 composite for performance-enhanced Zinc-air batteries. Adv. Mater. 2022, 34, e2110172.
41. Lepre, E.; Heske, J.; Nowakowski, M.; et al. Ni-based electrocatalysts for unconventional CO2 reduction reaction to formic acid. Nano. Energy. 2022, 97, 107191.
42. Choi, C. H.; Kim, M.; Kwon, H. C.; et al. Tuning selectivity of electrochemical reactions by atomically dispersed platinum catalyst. Nat. Commun. 2016, 7, 10922.
43. Kim, H. W.; Ross, M. B.; Kornienko, N.; et al. Efficient hydrogen peroxide generation using reduced graphene oxide-based oxygen reduction electrocatalysts. Nat. Catal. 2018, 1, 282-90.
44. Sa, Y. J.; Kim, J. H.; Joo, S. H. Active edge-site-rich carbon nanocatalysts with enhanced electron transfer for efficient electrochemical hydrogen peroxide production. Angew. Chem. Int. Ed. 2019, 58, 1100-5.
45. Lei, X.; Tang, Q.; Zheng, Y.; et al. High-entropy single-atom activated carbon catalysts for sustainable oxygen electrocatalysis. Nat. Sustain. 2023, 6, 816-26.
46. Tian, Q.; Jing, L.; Du, H.; et al. Mesoporous carbon spheres with programmable interiors as efficient nanoreactors for H2O2 electrosynthesis. Nat. Commun. 2024, 15, 983.
47. Zhi, Q.; Jiang, R.; Yang, X.; et al. Dithiine-linked metalphthalocyanine framework with undulated layers for highly efficient and stable H2O2 electroproduction. Nat. Commun. 2024, 15, 678.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].