Recent advances in MXenes-based photocatalysts for CO2 reduction: a review on fundamentals and future directions
Abstract
CO2 photoreduction is a sustainable strategy for converting CO2 into high-value solar fuels. However, its complex reaction pathways, kinetics, and thermodynamic challenges limit its application. MXenes, as an emerging two-dimensional material, have become a focus in photocatalysis due to their unique structural characteristics and highly tunable electrical and optical properties. As for the MXenes-based photocatalysts towards CO2 reduction, significant advances have been achieved in this field. This review starts with an introduction to the key thermodynamic and kinetic factors limiting CO2 photoreduction performance. The structural, electronic and optical properties of MXenes will be discussed to highlight their potential applications in CO2 photoreduction. Then, the latest research progress in MXenes-based photocatalysts for CO2 reduction will be summarized from the perspective of the role of MXenes in MXenes-based photocatalysts. Finally, the review concludes with a summary and future perspectives, outlining potential research directions in this rapidly evolving field. This work can serve as a guide for the development of MXenes-based catalysts for photocatalytic CO2 reduction, benefiting researchers working in this potentially game-changing field.
Keywords
INTRODUCTION
Global warming and energy shortages are significant challenges currently facing human society[1]. Carbon dioxide (CO2), as one of the primary greenhouse gases, has become a focal point of research for its reduction and resource utilization[2]. Photocatalytic CO2 reduction (PCR) technology, which mimics the process of natural photosynthesis in plants to convert CO2 into valuable chemical fuels or chemicals, offers a promising approach to address these challenges[3-8]. However, existing photocatalysts, such as TiO2[9],
In 2004, the successful exfoliation of graphene overturned the previous view in the physics community that two-dimensional (2D) materials could not exist at finite temperatures, opening the door to research on 2D materials[21]. Due to their unique structure and electronic properties, 2D materials have shown great potential in the field of photocatalysis. Compared to traditional bulk materials, 2D materials possess higher specific surface areas, superior electronic transport properties, and tunable band structures, enabling more efficient light absorption, and separation and transport of photogenerated carriers for photocatalytic reactions[22-25]. Currently, 2D materials such as graphene[26,27], transition metal dichalcogenides (TMDs)[25,28], and black phosphorus[29,30] have been extensively studied to construct composite photocatalysts that exhibit superior performance in PCR reactions. Current research mainly focuses on utilizing the metallic conductivity of 2D materials to construct Schottky junctions with semiconductors to suppress the recombination of photogenerated electron-hole pairs in the photocatalysts, while this does not effectively resolve the issue of low photocatalytic activity in PCR. The activation of CO2 molecules by 2D materials still requires further exploration and optimization, which is crucial for achieving a breakthrough in PCR performance.
MXenes, an emerging and growing family of 2D materials in recent years, possess unique metallic and metal-like properties[31]. Compared with other 2D materials, MXenes exhibit superior properties such as high conductivity, tunable band gaps, rich surface chemistry and exposed transition metal sites[32,33]. The diversity of MXenes, including Ti3C2Tx, Nb2CTx, Mo3C2Tx, and others, allows for precise control over their band gaps and electronic properties through the combination of different metal elements and variations in surface functional groups[34,35]. Additionally, MXenes exhibit unique localized surface plasmon resonance (LSPR) effects[36,37], strong support effects[38], and strong adsorption capabilities[39,40], demonstrating great potential in CO2 photoactivation.
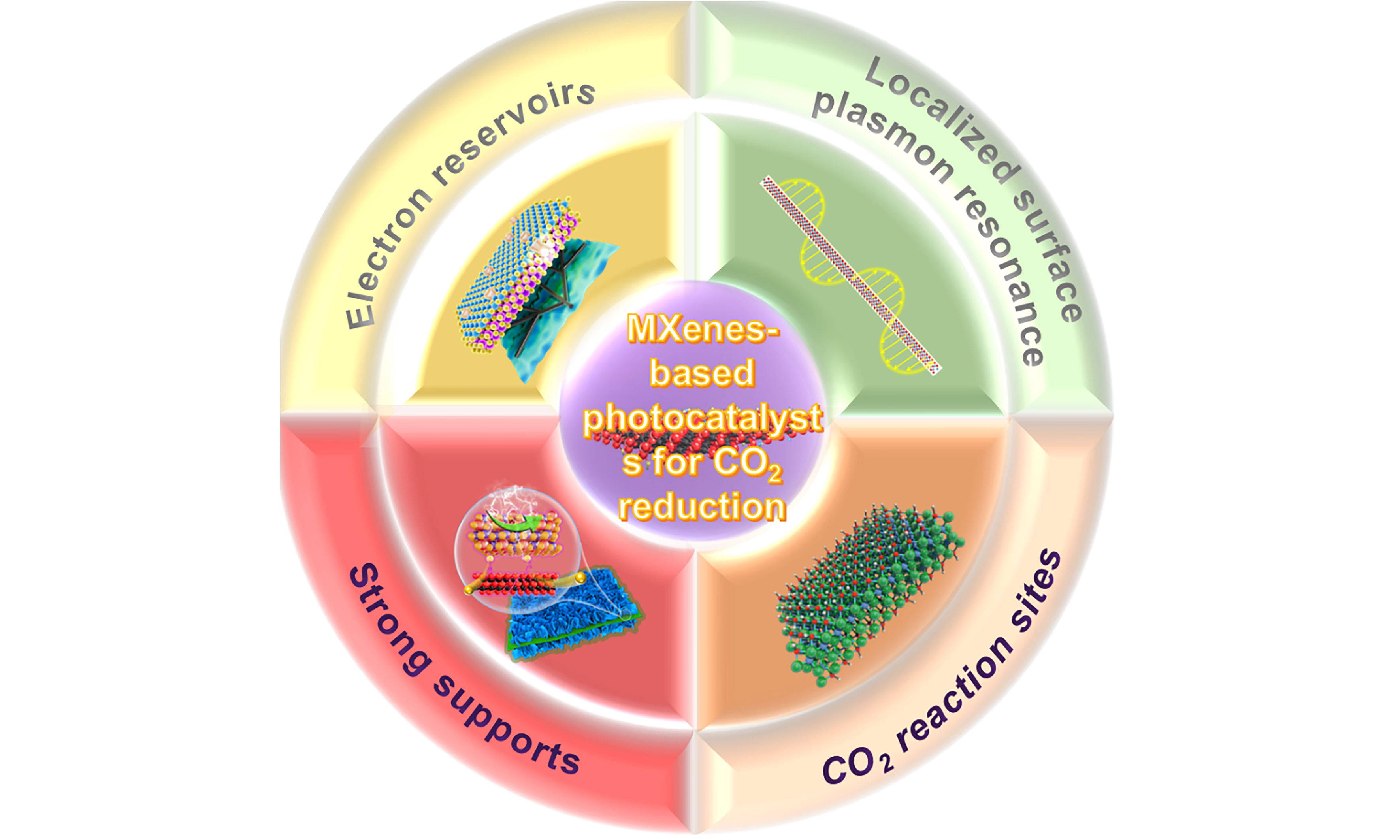
In this review, we will summarize the recent advances of MXenes-based composites in PCR to solar fuels, focusing on the roles of MXenes in PCR reactions. First, the key factors limiting CO2 photoreduction will be introduced from both thermodynamic and kinetic perspectives. Then, the high adjustment in structural, electronic and optical properties of MXenes will be discussed, highlighting their potential applications in PCR. Subsequently, the role of MXenes in MXenes-based photocatalysts for PCR and the latest research progress will be summarized, with a particular focus on the electron reservoir effects, LSPR, strong support effects, and the CO2 reaction sites of MXenes. Finally, the future development prospects of MXenes for selective photoreduction of CO2 into solar fuels will be prospected. This review aims to provide the latest theoretical insights and practical references for the design and development of highly efficient MXenes-based photocatalysts for CO2 reduction.
FUNDAMENTALS OF PCR
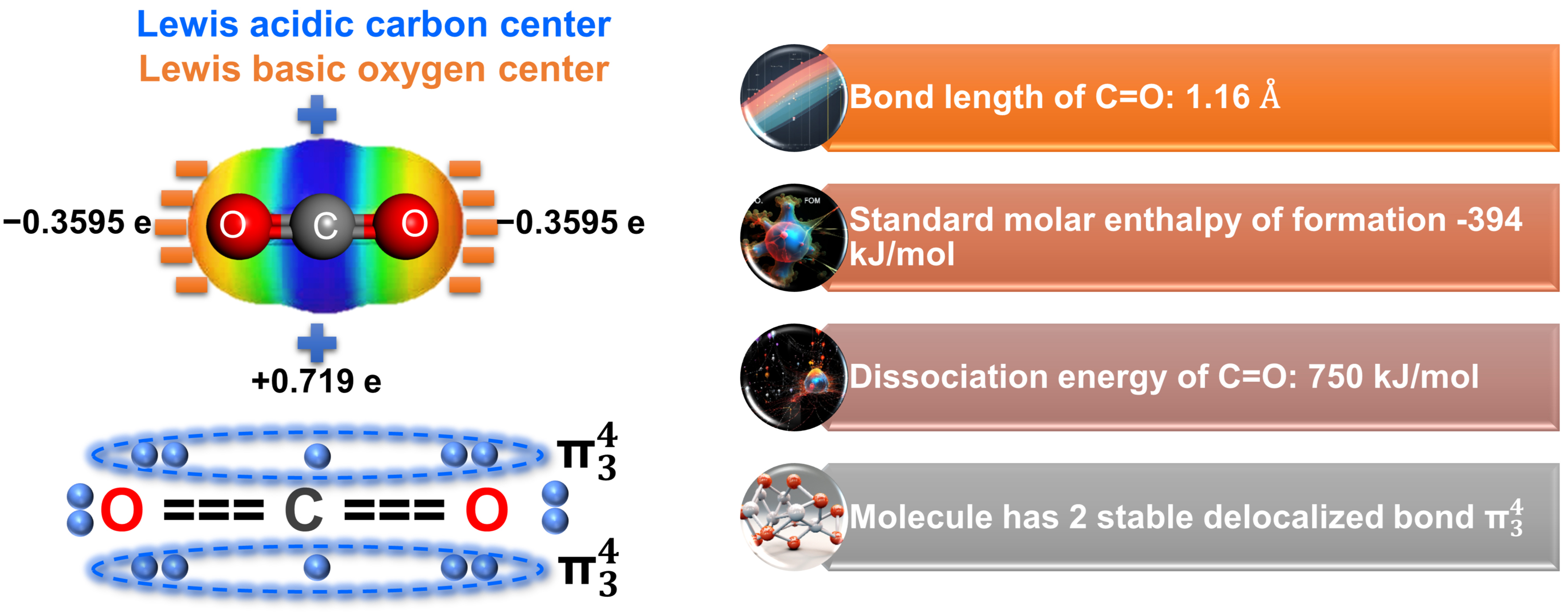
Understanding the thermodynamics of PCR is essential for the design of efficient photocatalysts with high selectivity for target products. The thermodynamic characteristics of CO2 molecules provide an initial insight into the challenges of PCR. CO2 is a linear molecule composed of one carbon atom and two oxygen atoms, which act as Lewis acid and Lewis bases, respectively, to participate in chemical
Fundamental mechanism of photocatalysis
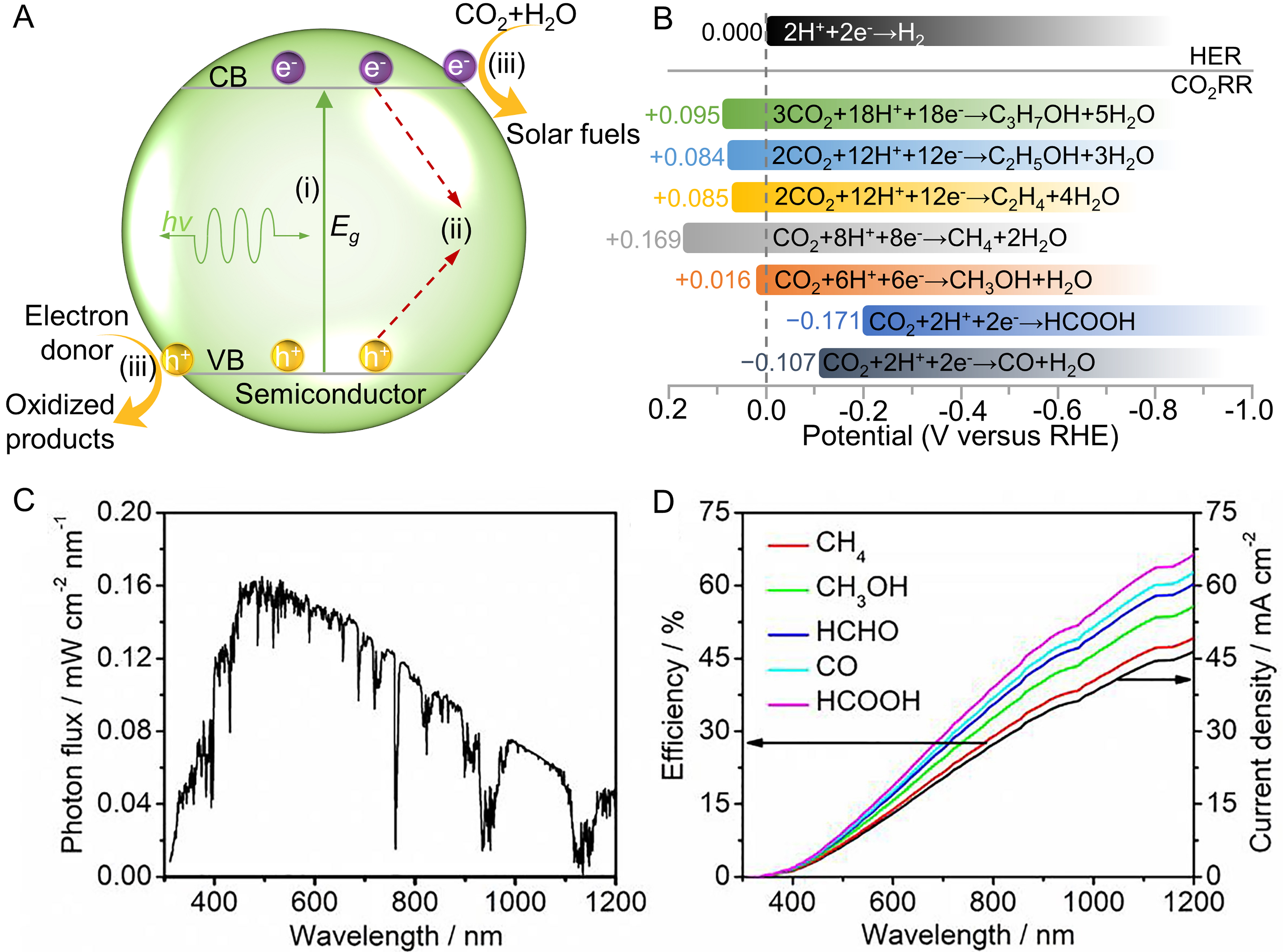
The photocatalytic process represented by semiconductor photocatalysts[45] mainly involves the following stages [Figure 2A]:
Figure 2. (A) Schematic of photocatalytic CO2 reduction; (B) The potentials for the CO2 reduction and H2 reduction under standard conditions. The left is the thermodynamic equilibrium, and the colors highlight the overpotential for the specific products; (C) AM 1.5G solar spectrum; (D) SFE (left) and theoretical photocurrent density (right) under one sun illumination. (C and D) Reproduced with permission[64]. Copyright Elsevier. SFE: Solar-to-fuel conversion efficiency; AM: air mass; HER: hydrogen evolution reaction; RHE: reversible hydrogen electrode.
(i) Generation of photo-induced carriers: When photons with energy greater than the band gap are absorbed by the semiconductor catalysts, electrons are excited from the valence band (VB) to the conduction band (CB), generating electron-hole pairs. The efficiency of this step can be engineered by tuning the band gap of the semiconductor, such as through doping with metal[46] or non-metal atoms[47], or by constructing heterojunctions with different semiconductors[48]. Additionally, quantum dot (QD) sensitization[49] or plasmonic enhancement using metal nanoparticles (e.g., Au and Ag)[50] can further improve the light absorption of semiconductors, leading to more efficient generation of photo-induced charge carriers.
(ii) Transport/Separation of photo-induced carriers: After the photogenerated carriers reach the surface of the catalyst and undergo separation, electrons and holes move to the reduction and oxidation reaction sites, respectively. This separation efficiency can be significantly improved by engineering the charge carrier dynamics. Cocatalysts can be introduced as electron sinks or hole traps to reduce the recombination of charge carriers[51]. Nanostructuring the photocatalyst [e.g., nanoparticles, nanowires (NWs), or nanosheets] can shorten the charge migration distances and improve electron-hole separation[52]. Additionally, constructing heterojunctions between different semiconductors[53] or the use of carbon-based supports (such as graphene or carbon nanotubes)[54] can provide charge transfer pathways to facilitate charge transport.
(iii) Redox reactions: The electrons reach the active sites to drive the reduction of CO2 on the catalyst surface, while the holes participate in oxidation reactions with water or a sacrificial agent. The activity of the redox reactions can be enhanced by optimizing the surface area and porosity of the catalyst, which increases the active sites for the reactions[55]. Additionally, surface functionalization with specific groups (e.g., -OH,
Thermodynamics and kinetics of CO2 photoreduction
The PCR process involves several key steps, including CO2 adsorption, photogeneration of electron-hole pairs, separation and transport of electron-hole pairs, CO2 activation, formation and desorption of intermediates, and the production of final products. The thermodynamics of CO2 photoreduction determine whether the reaction can occur, while the kinetics govern the rate and efficiency of the process.
Due to the high stability of the CO2 molecule, a high kinetic overpotential of -1.44 V [vs. reversible hydrogen electrode (RHE)] is required to bend the linear CO2 into its active species CO2•-, making the efficient occurrence of PCR challenging[63]. Notably, multi-electron and proton-coupled CO2 reduction reactions can significantly lower the reduction potential of CO2. Depending on the number of transferred electrons and involved protons as shown in Figure 2B, CO2 reduction products include carbon monoxide (CO), formic acid (HCOOH), formaldehyde (HCHO), methanol (CH3OH), methane (CH4), and other hydrocarbons[63]. The results of CO2 reduction potential in Figure 2B indicate that obtaining target products from CO2 photoreduction primarily requires meeting the corresponding reduction potentials. Additionally, the CO2 reduction process involves complex reaction pathways, such as C–O bond cleavage, C–H bond formation, and radical dimerization reactions[17]. These pathways are further complicated by competing reactions, such as HER and oxygen reduction reaction (ORR), which exacerbate the difficulty of achieving efficient CO2 photoreduction. Therefore, adopting effective strategies for adsorption and activation of CO2 by photocatalysts is essential to reduce the activation energy and overpotential of PCR reaction.
The AM1.5G solar spectrum is commonly used to evaluate the photocatalytic performance of catalysts under real sunlight as shown in Figure 2C[64]. The AM1.5G spectrum covers the ultraviolet (UV), visible, and near-infrared regions, with higher light intensity in the visible and near-infrared areas [Figure 2C]. Therefore, the match between the catalyst light absorption range and this spectrum is crucial. Photocatalysts with an appropriate band gap can effectively absorb visible light in the 400-700 nm range, thereby enhancing photocatalytic efficiency. Optimizing the band gap and spectral response range of catalysts is important to improve CO2 photoreduction efficiency[64].
Currently, the primary challenge in realizing the industrial application of photocatalysis lies in the relatively low solar-to-fuel conversion efficiency (SFE), which represents the ratio of the converted chemical energy to the incident solar energy[26]. The SFE is linked to the photocurrent density, which can be theoretically determined from the semiconductor light absorption range as shown in the right axis of Figure 2D. Unlike other photocatalytic reactions, such as HER, the diversity of PCR products imposes more stringent requirements on the band gap of the photocatalyst, as shown in the left axis of Figure 2D. The semiconductor with 2.0 eV bandgap exhibits theoretical maximum SFEs of 15.4%, 17.5%, 18.7%, 19.7%, and 20.8% for CH4, CH3OH, HCHO, CO, and HCOOH, respectively (left axis of Figure 2D)[64]. An ideal SFE of at least 10% is the threshold for commercialization[26], requiring light absorption wavelengths of at least
Reaction pathway of CO2 photoreduction
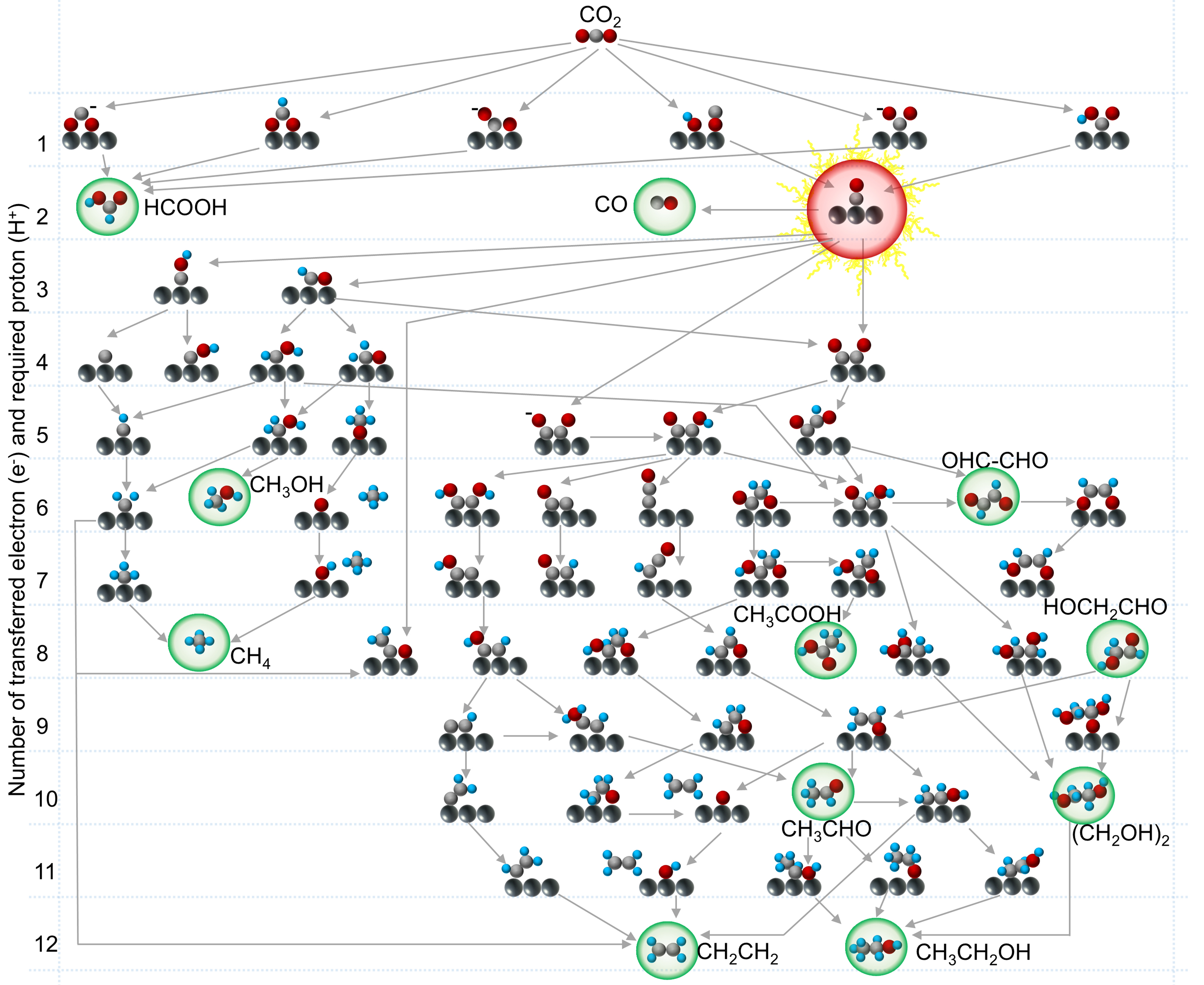
The reaction mechanism plays the role of “guiding light” in achieving efficient CO2 photoreduction due to the complexity of the reaction process and the diversity of products. On this basis, a reaction pathway map for the reduction of CO2 to various hydrocarbon products has been illustrated[17,43,65,66], as shown in Figure 3. It should be noted that the complexity of CO2 reduction pathways in Figure 3 does not encompass all possible mechanisms. With the rapid advancements in understanding CO2 reduction, new reaction pathways, different mechanisms, and related strategies are continually being discovered, showcasing the scientific allure of CO2 photoreduction.
As illustrated in Figure 3, the deeply reduced products of CO2 are significantly influenced by the reaction pathway, which involves synergistic proton-electron transfer, stepwise proton and electron transfer, multi-electron transfer, and even purely chemical reactions. Typically, the CO2 reduction begins with CO2 adsorption on the catalytic sites, followed by proton and electron acceptance to form various intermediates. The initial adsorption configuration of CO2 determines the reaction pathway for CO2 reduction[67-70]. CO and HCOOH are currently the most accessible products from CO2 over various photocatalysts, requiring only two electrons and two protons [Figure 3]. Various hydrocarbon compounds except CO and HCOOH can be obtained by further reduction of CO* intermediates [Figure 3]. This highlights that the key step for achieving high-value products from CO2 is the stabilization and further hydrogenation of CO* intermediates. To date, many reviews have summarized relevant strategies to address this issue, mainly focusing on optimizing the adsorption configuration of CO* on the catalyst surface and improving the ability of catalysts to stabilize CO*[71]. Different hydrocarbon products can be obtained from CO* after the reduction with the same number of electrons and protons. Additionally, the reduction of CO* involves complex reaction pathways, making it more challenging to obtain a single hydrocarbon product. Therefore, while pursuing the high rate of CO2 reduction reactions, we should also focus on improving the selectivity of CO2 reduction to avoid the separation and purification of complex products to obtain the target product.
OVERVIEW AND FUNDAMENTALS OF MXENES
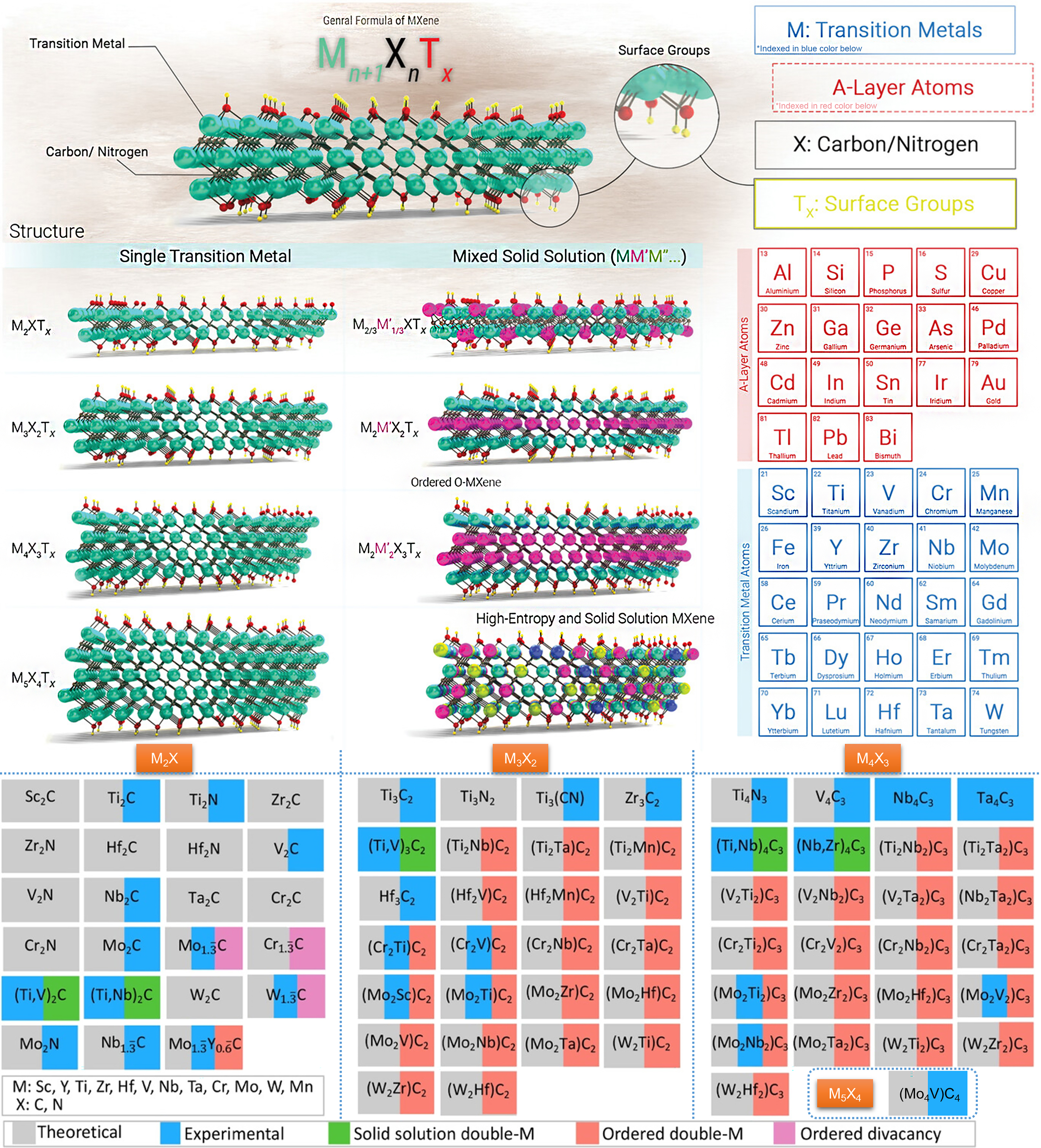
In 2011, MXenes, novel 2D multilayer materials, are first obtained by etching a MAX phase at room temperature by Naguib et al.[72]. To date, the family of MXenes has expanded significantly, with some being successfully synthesized in the laboratory and others predicted theoretically, as shown in Figure 4[73,74]. Due to their exceptional physicochemical properties, MXenes have attracted interest from researchers in exploring their applications in areas such as catalysis, sensing, and energy storage.
Structural characteristics
MXenes are generally expressed as Mn+1Xn (n = 1-4), where M denotes transition metals such as Ti, V, Sc, Y, Zr, Nb, Hf, Ta, Mo, etc., and X represents C and/or N[75]. Currently, Mn+1Xn structures include M2X, M3X2, M4X3, and M5C4[76]. The M distribution in the structure varies with the number of layers for single-metal MXenes, whereas for multi-metal MXenes, the distribution can be classified into disordered solid solutions and ordered distributions both in-plane and out-of-plane[73]. When MXenes are prepared via solution-based etching and molten salt etching methods, their structure becomes Mn+1XnTx, where Tx denotes surface termination groups such as -OH, -F, -O, -NH2, -F, -Cl, -Br, -S, -Se, or -Te[76]. To date, over 100 types of MXenes (excluding surface termination groups) have been theoretically predicted, while only more than 30 distinct MXenes have been experimentally prepared[73,76]. Currently, the exploration of MXenes in CO2 photoreduction applications is primarily focused on Ti3C2 MXene. As shown in Table 1, the results indicate that Ti3C2 can effectively enhance the CO2 reduction activity of semiconductor photocatalysts and improve the selectivity for target products such as CO, CH4, CH3COOH, and HCOOH, demonstrating its potential as an excellent cocatalyst for CO2 photoreduction[77-86]. Additionally, research on other types of MXenes designed for CO2 photoreduction photocatalysts remain limited, and mainly includes TiNbC[87], V4C3[88], V2C[89], Nb2C[90], and Ti3CN[91] [Table 1]. This indicates that there is still great potential for exploration in the synthesis, electronic and physicochemical properties of MXenes, along with their application in the field of PCR. It is well-known that metals can effectively activate CO2 and exhibit excellent deep hydrogenation capabilities[92]. Given the diverse metal compositions and structural configurations of MXenes, there is immense potential for their use in PCR and selective conversion of CO2 to high-value target hydrocarbons.
Summary of typical examples of MXenes-based semiconductor photocatalysts for CO2 reduction
| MXene | Semiconductor | Main products | Production rate (mol·g-1·h-1) | Selectivity (%) | Ref. |
| Ti3C2 | CuInS2 | CH3COOH | 250.0 | 97.5 | [77] |
| Ti3C2 | COF-367 | HCOOH | 1,880.0 | 100.0 | [78] |
| Ti3C2 | C3N5 | HCOOH | 3,510.0 | 100.0 | [79] |
| Ti3C2 | TiO2 | HCOOH | 767.0 | 77.4 | [80] |
| Ti3C2 | C3N4/bentonite | CH4 | 955.0 | 72.4 | [81] |
| Ti3C2 | Co-Co LDH | CO | 12,500.0 | - | [82] |
| Ti3C2 | g-C3N4 | CO | 306.8 | 80.2 | [83] |
| Ti3C2 | Cobalt atom | CO | 6,060.0 | 72.9 | [84] |
| Ti3C2 | CeO2/TiO2 | CH4 | 0.22 | 70.7 | [85] |
| Ti3C2 | Cs2AgBiBr6 | CO | 11.1 | 70.9 | [86] |
| TiNbC | TiO2 | CH4, CO, H2 | 43.0 | 92.1 | [87] |
| V4C3 | None | CO | 95.7 | 96.0 | [88] |
| V2C | g-C3N4 | CO | 9,289.0 | 99.4 | [89] |
| Nb2C | g-C3N4/CsPbBr3 | CO | 53.1 | - | [90] |
| Ti3CN | Ru-TiO2 | CO | 67.4 | 67.4 | [91] |
Electronic structure and highly adjustable properties
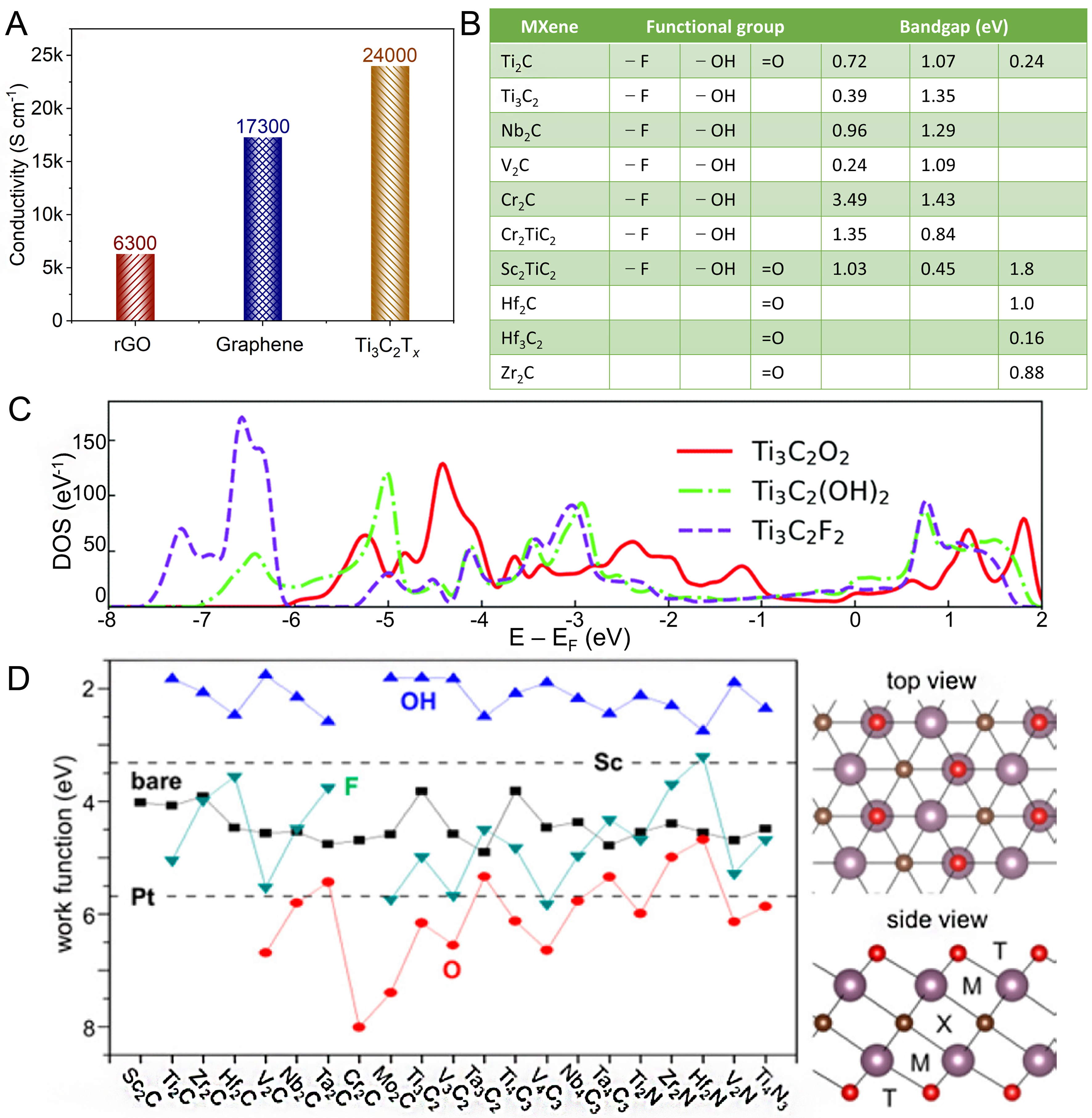
The electronic structure of MXenes can be precisely tuned through surface chemistry, which determines their role in the PCR. It has been shown that bare Ti3C2 exhibits metallic-like electronic behavior. The conductivity of modified Ti3C2Tx can reach up to 24,000·S·cm-1, which is significantly superior to that of reduced graphene oxide (rGO) (6,300 S·cm-1) and graphene (17,300 S·cm-1) [Figure 5A][93,94]. When the surface of Ti3C2 is modified with -F and -OH functional groups, the band gap of Ti3C2 is transformed from metallic to semiconducting[95]. With variations in the type and structure of the M metal in MXenes and changes in surface terminations, the bandgap of some MXenes is gradually opened, exhibiting semiconductor-like behavior [Figure 5B and C] and significant changes in their work function
Figure 5. Electronic properties of MXenes. (A) Comparison of electrical conductivity of rGO, graphene, and Ti3C2Tx; (B) Bandgap width of some Mxenes; (C) DOS of Ti3C2, Ti3C2O2, Ti3C2(OH)2, and Ti3C2F2; (D) Dependence of the work function of MXenes on their M metal and surface terminations. (C and D) Reproduced with permission[76]. Copyright the American Association for the Advancement of Science. DOS: Density of States.
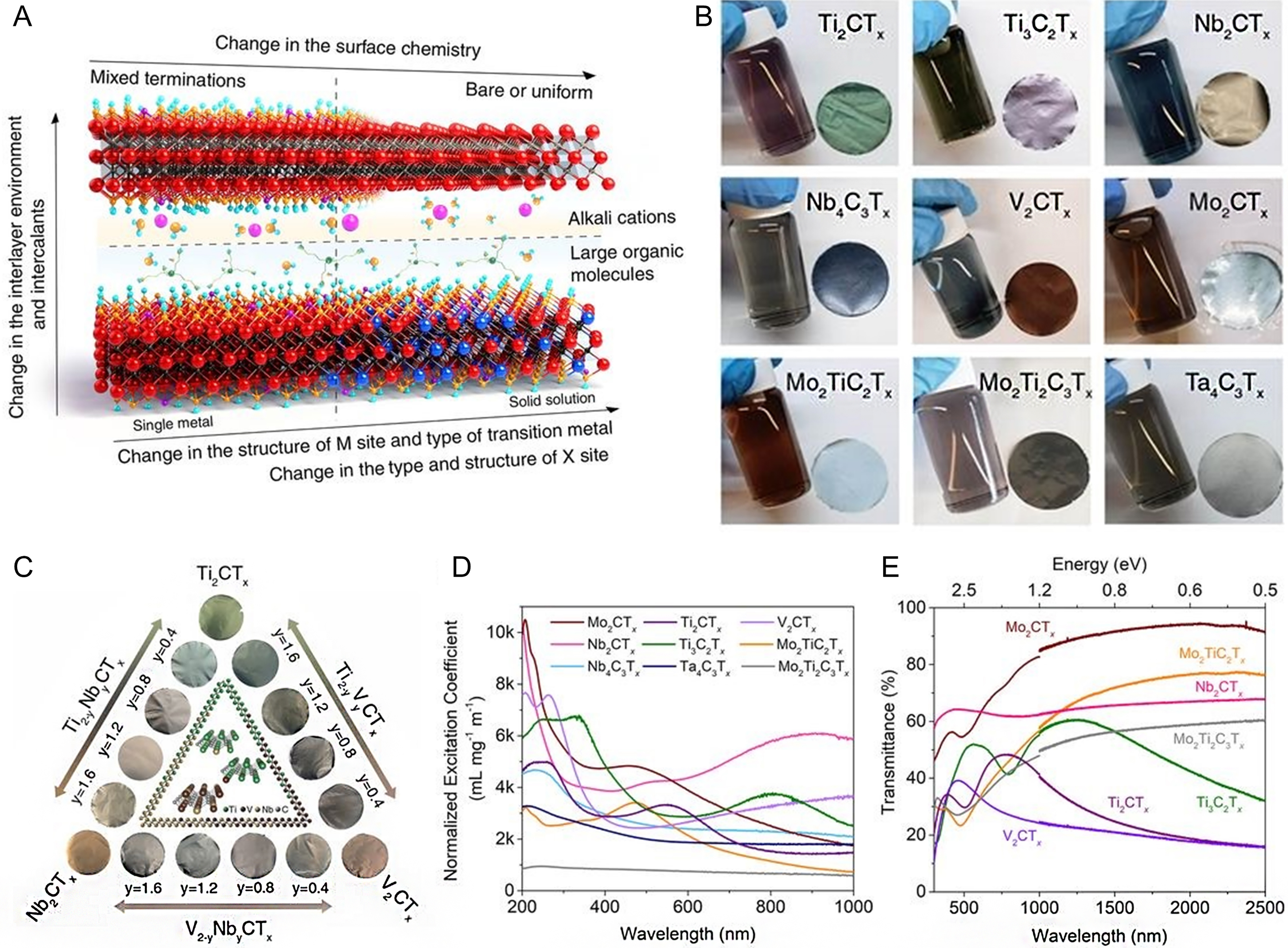
As shown in Figure 6A, the rich surface functional groups (such as -OH, -F, -O, -NH2, -F, -Cl, -Br, -S, -Se, or -Te[76]) on MXenes endow their surface properties with significant tunability, which can be achieved by modifying the etching methods. These surface terminations play a crucial role in modulating the hydrophilic or hydrophobic nature of MXenes[97], which is of particular importance for their performance in PCR reactions in aqueous environments [Figure 6A]. For instance, surface termination with -OH or -O groups generally increases the hydrophilicity of MXenes, promoting better interaction with water and enhancing CO2 adsorption and reduction[97,98]. On the other hand, the presence of hydrophobic groups such as -F can reduce the interaction with water, potentially decreasing CO2 adsorption but offering better stability under certain reaction conditions[97,98]. The feasibility of tuning the surface properties by modifying the surface terminations of MXenes provides fine control over their behavior in photocatalytic processes, particularly in CO2 photoreduction, where water interaction and CO2 adsorption are key factors. Therefore, the surface characteristics of MXenes not only influence their photocatalytic activity but also their long-term stability and performance in practical applications. Such tunable properties make MXenes promising candidates for advanced photocatalytic systems aimed at CO2 reduction.
Figure 6. Optical properties of MXenes. (A) Schematic illustration of highly adjustable properties of Mxenes; (B) The color of colloidal solutions of various MXenes and their corresponding freestanding films; (C) Digital photographs of three M’2-yM”yCTx solid solution systems; (D) UV-vis-NIR optical extinction properties of aqueous dispersions of various 2D transition metal carbides; (E) UV-vis-NIR transmittance spectra from 300 to 2,500 nm for MXenes thin films. (A-E) Reproduced with permission[76]. Copyright the American Association for the Advancement of Science. UV-vis-NIR: ultraviolet-visible-near-infrared.
Optical properties
The optical properties of MXenes are closely linked to their electronic band structure. The density of states for MXenes reveals a high density at the Fermi level, leading to unique optical characteristics [Figure 5C][76]. Different MXenes exhibit varying optical absorption behaviors [Figure 6B-D]. MXenes exhibit strong light absorption for interband transition in the UV range [Figure 6D][99]. Notably, the high concentration of free electrons in MXenes endows them with unique local surface plasmon resonance (LSPR) absorption properties[100-102]. They exhibit both transversal and longitudinal plasmonic modes in the visible and infrared ranges, where transverse plasmon modes are independent of flake size but depend on the type and structure of the M sites, and surface terminations [Figure 6A and D][103]. Consequently, different MXenes reveal distinct plasmonic absorption characteristics and display different colors in transmission and reflection
APPLICATION OF MXENES IN ENHANCING CO2 SELECTIVE PHOTOREDUCTION
MXenes have been widely used to fabricate photocatalysts for CO2 reduction due to their excellent electrical conductivity, broad absorption in the visible and near-infrared regions, and tunable properties. The previous reviews are mainly focused on the research progress of MXenes in photocatalysis and energy fields[71,106-108]. In this section, focusing on the roles of MXenes in composite photocatalysts, we specifically summarize the latest advances of MXenes-based photocatalysts in the field of PCR.
Role of “electron reservoir”
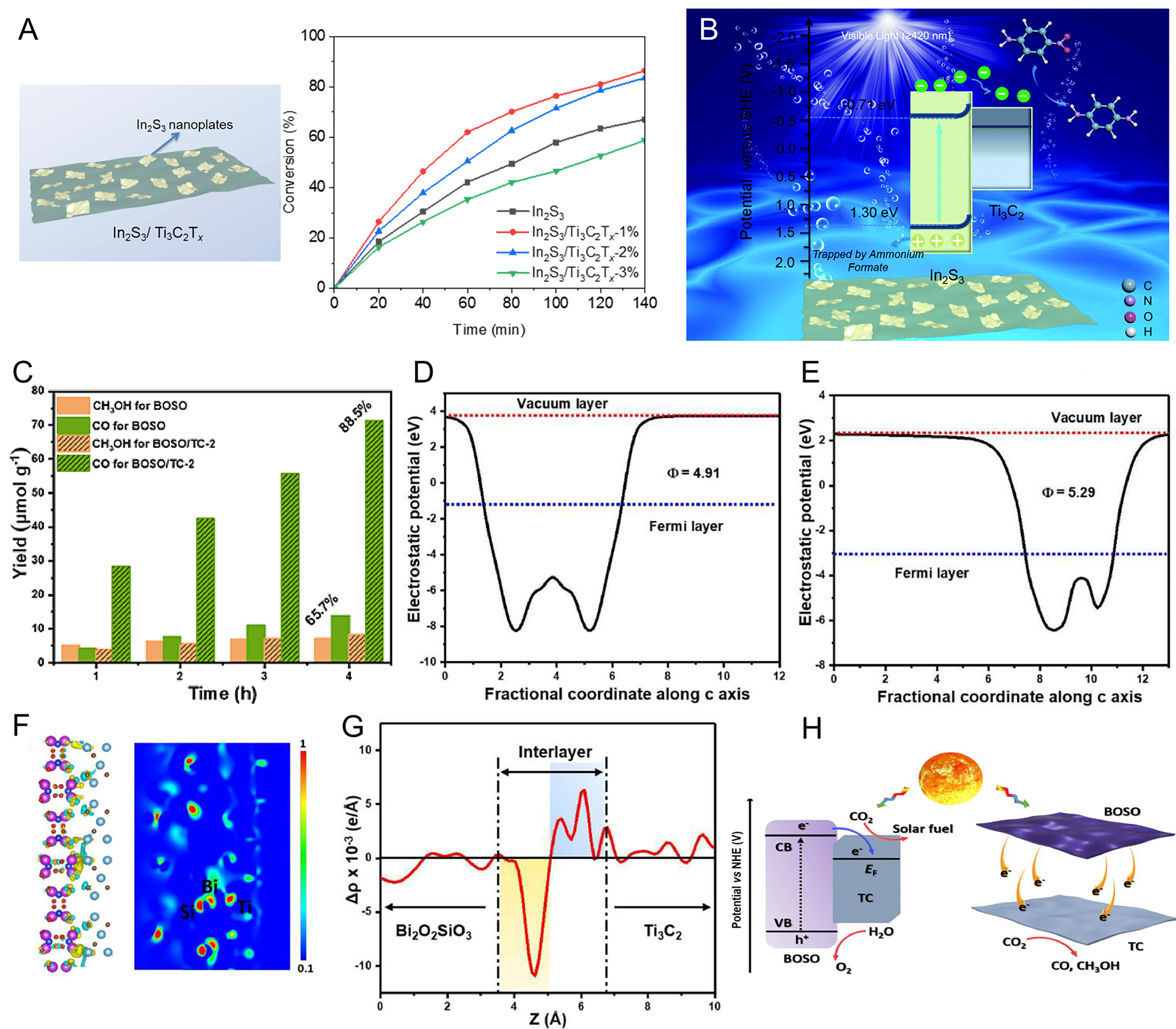
Utilizing cocatalysts with high work functions and favorable energy structure is an effective strategy to enhance the separation of electron-hole pairs in photocatalysts. As shown in Figure 7A and B, Ti3C2Tx Mxene, acting as an electron reservoir, forms Schottky heterojunctions with In2S3 semiconductors to promote efficient separation of photogenerated electrons and holes over In2S3 semiconductors, thereby enhancing the photocatalytic activity[51]. Notably, Ti3C2Tx, with the highest conductivity among various MXenes, has been demonstrated to effectively separate photogenerated electrons from photocatalysts, thereby reducing CO2 to CO, CH4, methanol, and other products[64]. For example, when Bi2O2SiO3 is in-situ grown on the surface of Ti3C2Tx[109], it exhibits significantly enhanced activity in PCR. The optimal Bi2O2SiO3/Ti3C2Tx composite achieves an enhanced CO and CH3OH generation rate that is 3.8-fold greater that of pristine Bi2O2SiO3, with a selectivity for CO approaching 90% [Figure 7C]. In this work, the selectivity for CO is calculated using: (CO evolution rates)/(CO evolution rates + CH3OH evolution rates) × 100%. The enhanced performance is attributed to the significantly higher work function of Ti3C2Tx compared to Bi2O2SiO3 [Figure 7D and E], indicating that Ti3C2Tx can accept photoelectrons generated by Bi2O2SiO3, thereby effectively promoting the separation and transfer of photogenerated carriers [Figure 7F-H].
Figure 7. (A) Schematic illustration of Ti3C2Tx MXene acting as an electron reservoir to enhance the photocatalytic activity of 2D In2S3 by forming Schottky heterojunctions; (B) Schematic illustration of Ti3C2Tx MXene enhancing photocatalytic activity of In2S3 through Schottky heterojunctions; (C) CO2 photoreduction performances and corresponding selectivity over pure Bi2O2SiO3 and Bi2O2SiO3/Ti3C2; Calculated work functions of (D) Bi2O2SiO3 and (E) Ti3C2; (F) Charge difference distribution coupled ELF and (G) calculated planar averaged electron density difference Δρ(z) of the Bi2O2SiO3/Ti3C2; (H) Relative energy-level diagram and photogenerated electron transfer process at the interface of the hybrids. (C-H) Reproduced with permission[109]. Copyright Elsevier. ELF: Electronic location function.
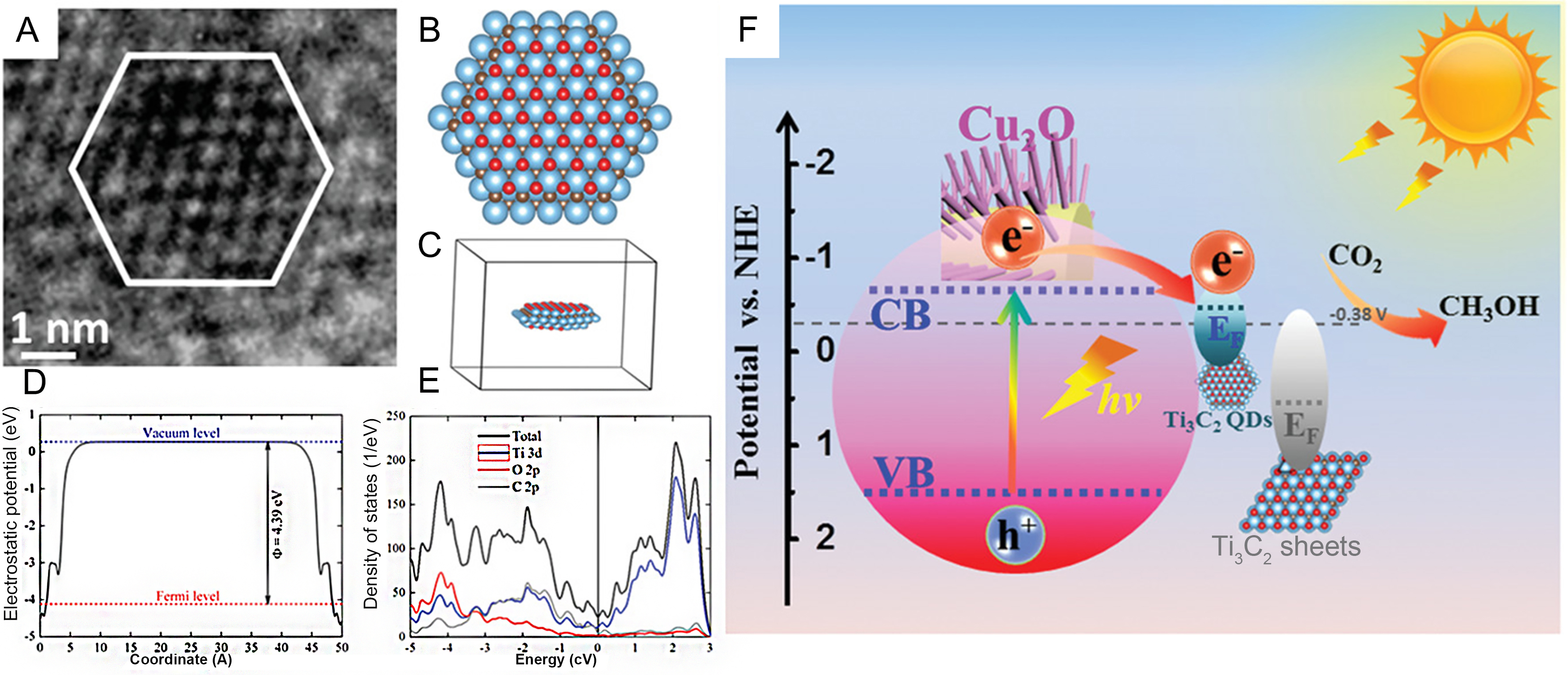
Recently, Ti3C2Tx QDs have been successfully synthesized via hydrothermal shearing, exhibiting unique fluorescence properties compared to Ti3C2Tx sheets[110,111]. They [Figure 8A-C] have been decorated onto
Figure 8. (A) High-resolution TEM image of Ti3C2Tx QDs; (B-E) The top view, side view of the structure, calculated Femi level, and DOS of O-terminated Ti3C2TxQD model; (F) Energy-level diagram of Ti3C2Tx QDs/Cu2O NWs and Ti3C2Tx sheets/Cu2O NWs heterostructures. (A-F) Reproduced with permission[110]. Copyright John Wiley and Sons. DOS: Density of States; TEM: transmission electron microscopy; QDs: quantum dots; NWs: nanowires.
In addition to utilizing the “electron reservoir” effect of metallic-like MXenes to promote the separation of photogenerated electron-hole pairs in photocatalysts, recent studies have reported that semiconductor-type MXenes can significantly enhance photocatalytic reduction performance, such as Nb2CTx[112-114], Mo2CTx[16], and Mo2TiC2Tx[115]. For example, the separation of photogenerated carriers in ZnCdS is promoted through strong electronic coupling with semiconductor Mo2TiC2Tx, resulting in a photocatalytic hydrogen production rate of up to 17.73 mmol·g-1·h-1[116]. This suggests the potential for further tuning the electronic structure of MXenes either as direct catalysts or by constructing heterojunctions with other semiconductors through energy-level alignment to improve PCR performance.
LSPR
Utilizing the LSPR effect of plasmonic metals to enhance photocatalytic activity has been proven to be an effective strategy. The mechanisms for photocatalytic enhancement from LSPR of classical plasmonic metals primarily involve hot electron injection, field enhancement, resonant energy transfer, the scattering of incident light, and thermal effects[117]. MXenes, as a non-noble plasmonic material, have been demonstrated to significantly enhance PCR performance through LSPR-induced hot electron injection, electric field enhancement, and thermal effects. In this section, we have summarized the latest advancements in utilizing the LSPR effect of MXenes to enhance PCR performance of catalysts.
Hot electron injection
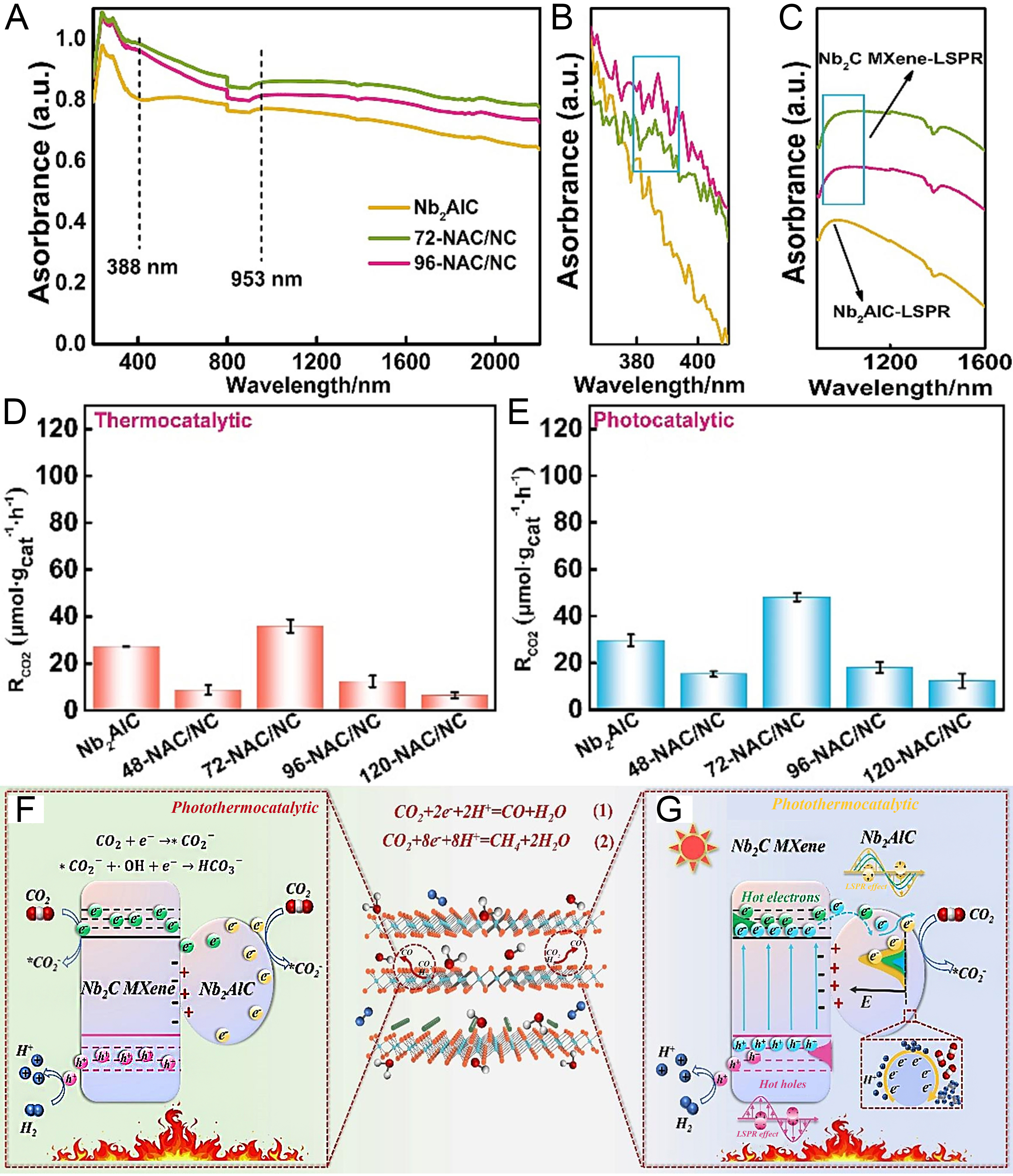
It has been reported that Ti3C2Tx as a single catalyst exhibits excellent HER performance through the direct LSPR-induced hot electron injection effect[118]. Additionally, as a cocatalyst, it enhances the performance of semiconductors for photocatalytic CO2-to-CO reduction via hot electron injection[119]. Recently, semiconductor-type Nb2C, in conjunction with the precursor Nb2AlC, has achieved efficient photocatalytic CO2 hydrogenation through the construction of heterojunctions[120]. As shown in Figure 9A and B, a weak absorption peak at 388 nm is attributed to the transverse surface plasmon resonance effect of Nb2AlC and Nb2C. In contrast, an absorption peak attributed to the longitudinal surface plasmon resonance effect over Nb2AlC and Nb2C can be seen at 953 nm [Figure 9C]. The photocatalytic performance of the Nb2AlC-Nb2C composite is better than the thermocatalytic performance [Figure 9D and E]. Under the pure light conditions, the CO2-to-CO photoactivity for the Nb2AlC-Nb2C composite is 48.2 μmol·g-1·h-1, which is better than pure Nb2AlC and pure Nb2C[120]. Electron spin resonance (ESR) results of free radicals captured by
Figure 9. (A-C) UV-vis-NIR DRS map of Nb2AlC/Nb2C MXene heterojunction; (D) Thermocatalytic and (E) photocatalytic performance of samples; (F and G) Mechanism of the thermally coupled photocatalytic reduction of CO2 by Nb2AlC/Nb2C MXene heterojunction. (A-G) Reproduced with permission[120]. Copyright Elsevier. UV-vis-NIR: Ultraviolet-visible-near-infrared; DRS: diffuse reflectance spectroscopy; LSPR: localized surface plasmon resonance.
Electric field enhancement effect
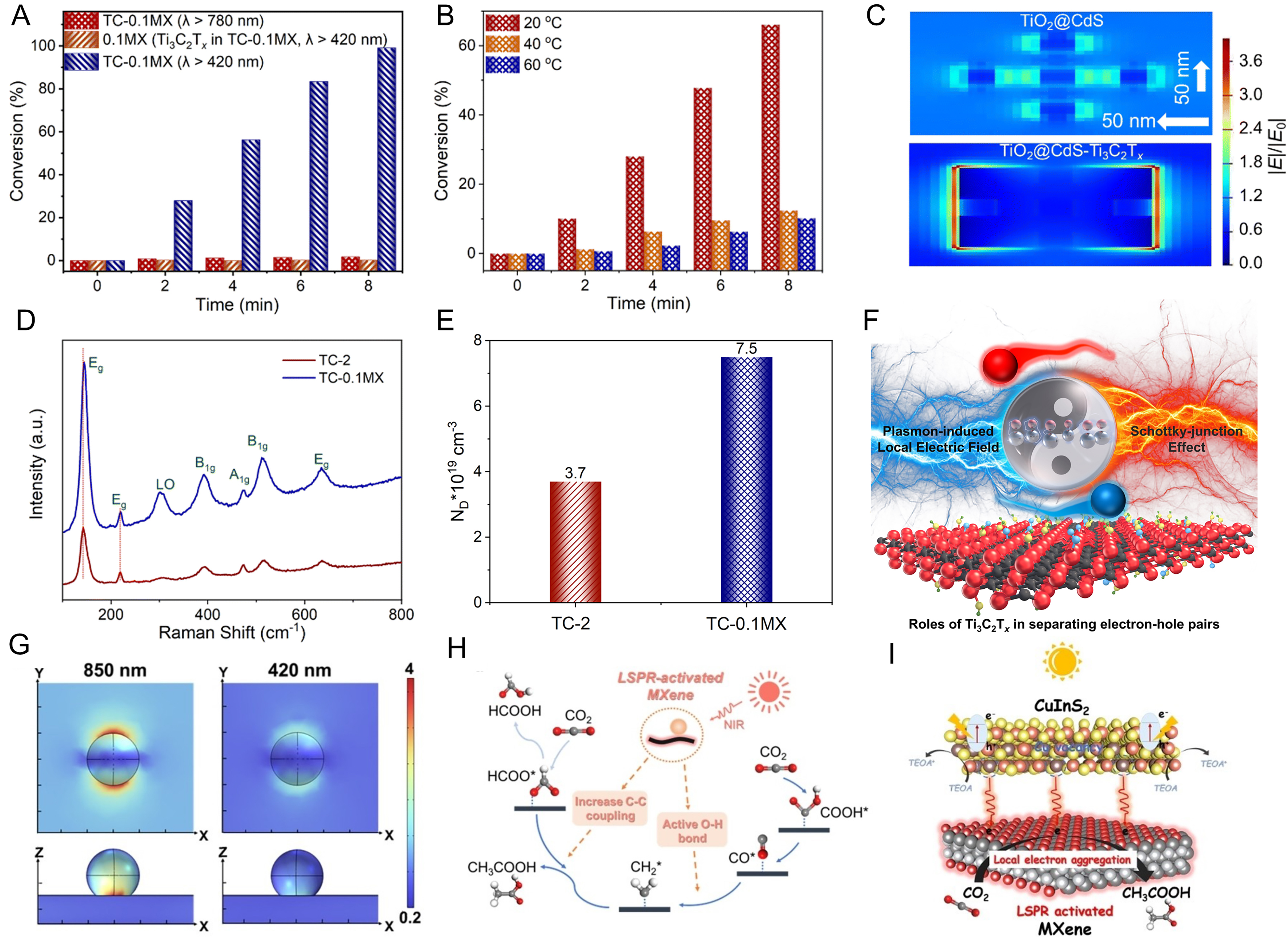
It has been found that the LSPR-induced field enhancement effect of Ti3C2Tx can significantly increase the concentration of photogenerated carriers in CdS[37]. In this system, the contributions of hot electron injection [Figure 10A] and thermal effects [Figure 10B] induced by the LSPR of Ti3C2Tx to the improved photocatalytic activity are excluded, while field enhancement induced by Ti3C2Tx is further confirmed through finite-difference time-domain (FDTD) simulations [Figure 10C] and surface Raman enhancement [Figure 10D]. Compared to TiO2@CdS, the introduction of Ti3C2Tx significantly enhances the carrier concentration in TiO2@CdS [Figure 10E], suggesting that the LSPR of Ti3C2Tx-induced field enhancement is beneficial for generating more photogenerated electrons to participate in the photocatalytic reduction reaction [Figure 10F]. In addition, it is shown that LSPR-activated Ti3C2Tx enables the selective photoreduction of CO2 to acetate[77]. Experiments and simulation results reveal that local electric field enhancement induced by LSPR of Ti3C2Tx promotes the formation of crucial intermediate coupling processes under near-infrared light [Figure 10G-I].
Figure 10. (A) Photocatalytic activities for the selective reduction of 4-NA; (B) Photoactivities of TC-0.1MX for the selective reduction of 4-NA under visible-NIR light irradiation (λ > 420 nm) at different temperatures; (C) Local electric field distributions of TiO2@CdS and TiO2@CdS-Ti3C2Tx composites are calculated by the FDTD method; (D) Raman spectra of TC-2 and TC-MX composites; (E) The photogenerated carrier concentration of TC-2, TC-0.1MX under visible-NIR light irradiation (λ > 420 nm); (F) Schematic illustration of the role of Ti3C2Tx MXene in photogenerated electron-hole separation. (A-F) Reproduced with permission[37]. Copyright Royal Society of Chemistry; (G) LSPR-induced enhancement of the electric field intensity from COMSOL simulations for CuInS2/MXene at excitation wavelengths of 420 and 850 nm; (H) Possible reaction pathway from CO2 to CH3COOH on LSPR-activated VCu CuInS2/Mxene; (I) Schematic illustration of proposed reaction mechanism. (G-I) Reproduced with permission[77]. Copyright John Wiley and Sons. FDTD: finite-difference time-domain; LSPR: localized surface plasmon resonance; 4-NA: 4-nitroaniline.
Thermal effect
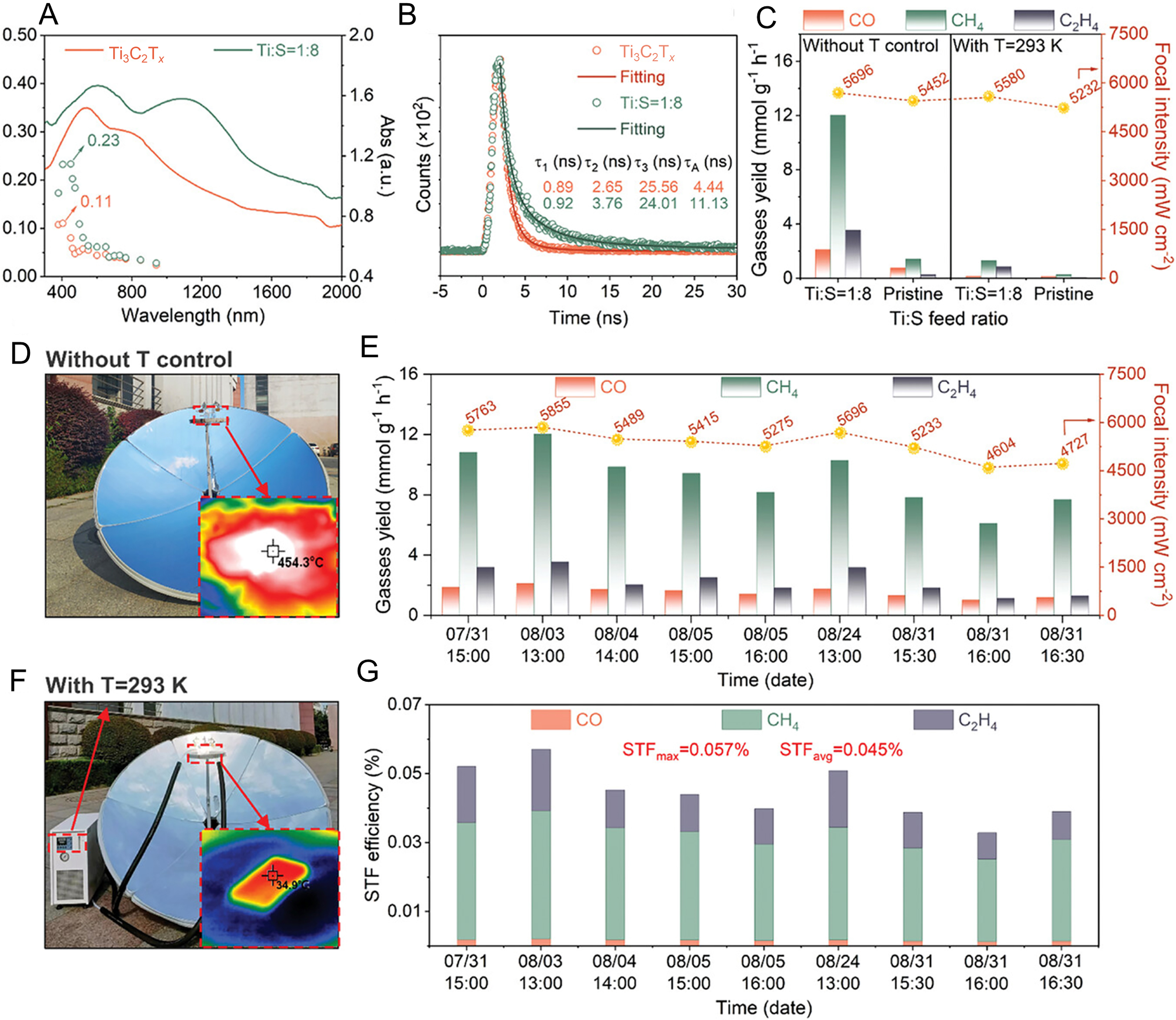
The LSPR effect of MXenes endows them with strong light absorption in the visible and near-infrared light ranges. Even in some cases, the light absorption energy of MXenes cannot directly excite the semiconductor to produce photocarriers, but it can promote charge separation and transfer at the interface through thermal effects[121]. Thus far, several MXenes, such as Ti3C2Tx[122,123,124], V4C3Tx[88], Mo2CTx[125], and Nb2CTx[120,126], have been shown to achieve efficient photoreduction of CO2 through thermal effects induced by LSPR[122]. For example, multilayer Ti3C2Tx has been demonstrated to efficiently drive the photoreduction of CO2 to CO, CH4, and C2H4 through LSPR excited by visible to near-infrared light [Figure 11A][122]. The electron lifetime of moderately sulfurized Ti3C2Tx (S-Ti3C2Tx) is 3-fold enhancement compared to that of pure Ti3C2Tx, which is favorable for the reaction kinetics of S-Ti3C2Tx[Figure 11B]. When the reaction system temperature is maintained at a constant 20 °C [Figure 11C], the CO2 photoreduction activity is significantly inhibited, indicating that the photothermal effect induced by the LSPR effect of Ti3C2Tx can effectively enhance CO2 photoreduction. Under natural sunlight irradiation, the S-Ti3C2Tx catalyst exhibits a maximum CH4 production rate (12.03 mmol·g-1·h-1) and CO production rate (2.11 mmol·g-1·h-1), which are enhanced by 8.6-fold compared to the pure Ti3C2Tx[Figure 11D-11G]. Interestingly, the high-value C2H4 production rate reaches 3.55 mmol·g-1·h-1 with a selectivity of 29.76% [Figure 11E]. More importantly, S-Ti3C2Tx maintains high photocatalytic activity for CO2 reduction even after 30 days of cycling [Figure 11E and G], demonstrating their good stability and promising potential for practical applications.
Figure 11. (A) Wavelength-dependent AQYs vs. the corresponding UV-vis DRS spectra of the pristine Ti3C2Tx and S-Ti3C2Tx (Ti:S = 1:8) catalysts; (B) Time-resolved transient PL emission decay of the pristine Ti3C2Tx and the S-Ti3C2Tx (Ti:S = 1:8); (C) The comparison of gas yields under different focused sunlight without or with temperature control (T = 293 K) using the pristine and the S-Ti3C2Tx (Ti:S = 1:8); (D) The photo of solar energy stove reactor without T control; (E) The comparison of gas yields under different focused incident sunlight (different dates) using the S-Ti3C2Tx (Ti:S = 1:8); (F) The photo of solar energy stove reactor with temperature control at T = 293 K under the focused sunlight; (G) The comparison of STF is measured at different dates using the S-Ti3C2Tx (Ti:S = 1:8). (A-G) Reproduced with permission[122]. Copyright John Wiley and Sons. AQYs: Action quantum yields; PL: photoluminescence; DRS: diffuse reflectance spectroscopy; STF: solar-to-fuel efficiency.
Support effect
Supports play a crucial role in enhancing the photoactivity of catalysts. It not only disperses and stabilizes the photocatalyst but also interacts with the loaded catalytic components, thereby influencing the catalytic activity, selectivity, and stability of the active catalyst. In this section, we summarize the latest advancements in the role of MXenes as a support in enhancing PCR, particularly focusing on the novel mechanisms of strong support interactions induced by MXenes and their influence on catalytic activity.
Catalyst support
Owing to the 2D lamellar structure and excellent conductivity of MXenes, it has been widely employed as catalytic support in electrocatalytic CO2 reduction[127-129]. Recently, MXenes have been used as photocatalytic supports to load semiconductors[130-133], covalent organic frameworks (COFs)[78], single atoms[84,134,135], or metal clusters[136] in PCR. For example, a Ti3C2Tx/Bi2WO6 2D/2D heterojunction has been successfully fabricated by in-situ growing Bi2WO6 ultrathin nanosheets on the surface of Ti3C2Tx nanosheets, resulting in a 4.6-fold enhancement in PCR to target products (CH4 and CH3OH)[130]. The supporting role of Ti3C2Tx facilitates the dispersion of Bi2WO6, leading to the formation of nanosheet arrays during growth through the steric hindrance effect. This increases the specific surface area of Bi2WO6-Ti3C2Txcomposites, which facilitates reactant mass transfer and provides accessible active sites for CO2 reduction. In addition, the heterostructure of Ti3C2Tx loaded with crystalline COF-367 achieves efficient photocatalytic conversion of CO2 to HCOOH[78]. The selective conversion of CO2 to HCOOH in the Ti3C2Tx/COF-367 heterostructure relies on Ti3C2Tx facilitating efficient electron transfer, which drives the reduction of NAD+ to NADH. NADH then acts as a crucial coenzyme, enabling the bioenzyme FDH to selectively convert CO2 to HCOOH. In the absence of NADH, the PCR primarily produces CO and CH4, highlighting the key role of NADH in directing the reaction towards HCOOH. Intriguingly, isolated cobalt single atoms are bonded with carbon/oxygen atoms and anchored on the 2D Ti3C2Tx support, leading to visible-light-driven conversion of CO2 to CO[84]. This work successfully loads homogeneous single-atom catalysts onto a heterogeneous MXenes support, providing a potential solution for the recovery of homogeneous photocatalysts.
Electronic structure control
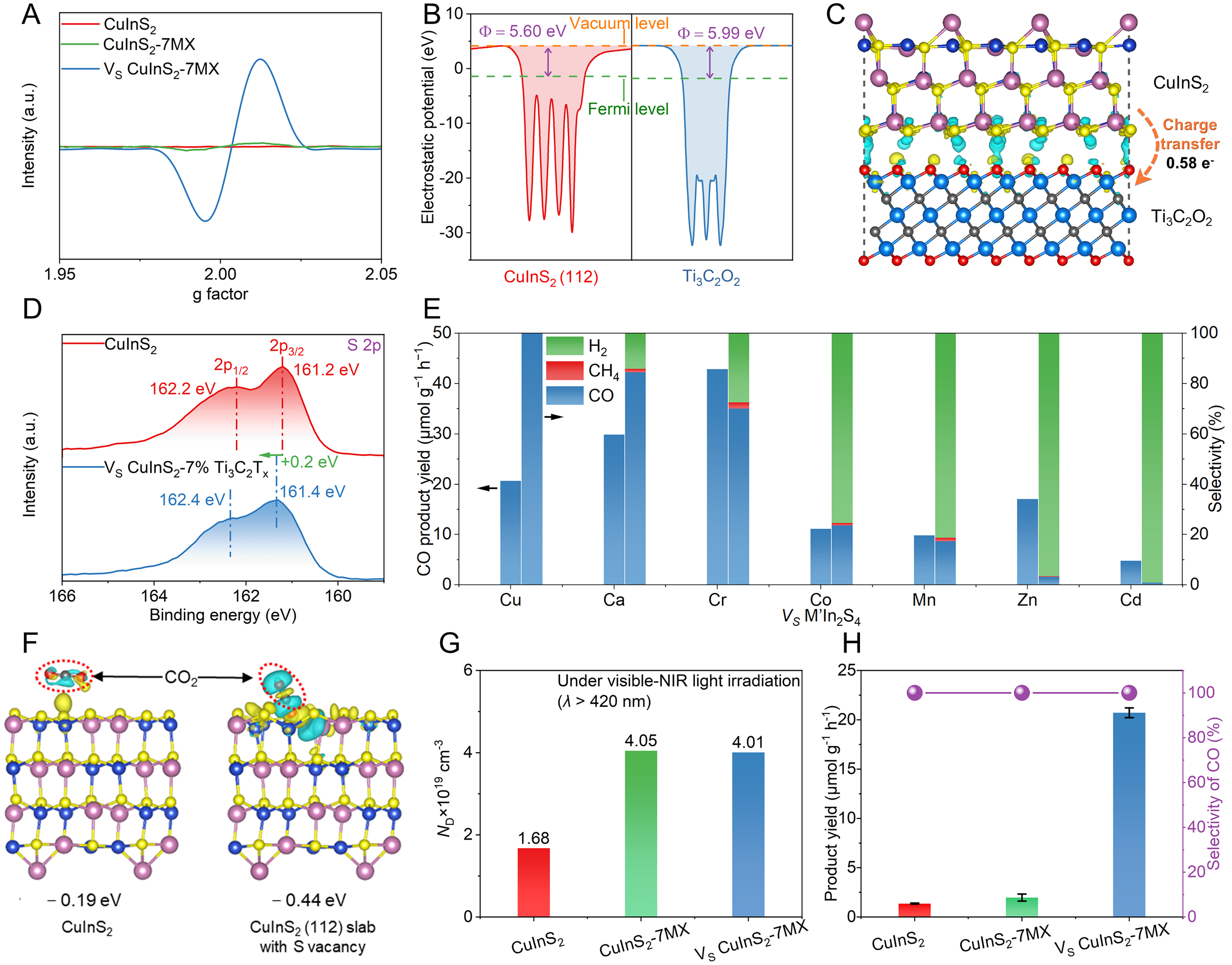
Classical strong metal-support interaction (SMSI) refers to the strong interaction between metallic active catalysts and reducible oxide supports, which is beneficial for enhancing thermal catalytic activity and regulating product selectivity[137,138]. The discovery of many novel SMSI has extended the spectrum of supports to MoC2[139], tellurium nanosheets[140], g-C3N4[141], Ti3C2Tx[142], and others[143]. Recently, the strong support interaction induced by Ti3C2Tx has been shown to modulate the electronic structure of sulfides and enhance their PCR performance[38]. Interestingly, EPR results indicate that the introduction of Ti3C2Tx can lead to the formation of sulfur vacancies (VS) for CuInS2-Ti3C2Tx[Figure 12A]. Experimental and simulation results indicate that the high work function of Ti3C2Tx endows it with strong electron-withdrawing capability [Figure 12B], causing the precursor S2- of CuInS2 to lose electrons and escape during growth
Figure 12. (A) ESR spectra over pure CuInS2, CuInS2-7MX, and VS CuInS2-7MX; (B) Calculated electrostatic potentials of CuInS2 (112) slab and Ti3C2O2 (Φ represents the work function); (C) Differences in charge density of CuInS2 and Ti3C2O2; (D) S 2p XPS spectra of pure CuInS2 (top) and VS CuInS2-Ti3C2Tx (bottom); (E) CO2 photoreduction performances and corresponding selectivity over M’In2S4-7MX (M’ = Cu, Ca, Cr, Mn, Co, Zn, and Cd); (F) The binding energies and the difference of charge density between CO2 and (left) perfect CuInS2 (112) slab or (right) CuInS2 (112) slab with VS; (G) The photo-induced ND, and (H) photocatalytic CO2-to-CO reduction performance over pure CuInS2, CuInS2-7MX, and VS CuInS2-7MX under visible-NIR light irradiation (λ > 420 nm). (A-H) Reproduced with permission[38]. Copyright Elsevier. ESR: Electron spin resonance; XPS: X-ray photoelectron spectroscopy; ND: carrier concentration.
CO2 reaction sites
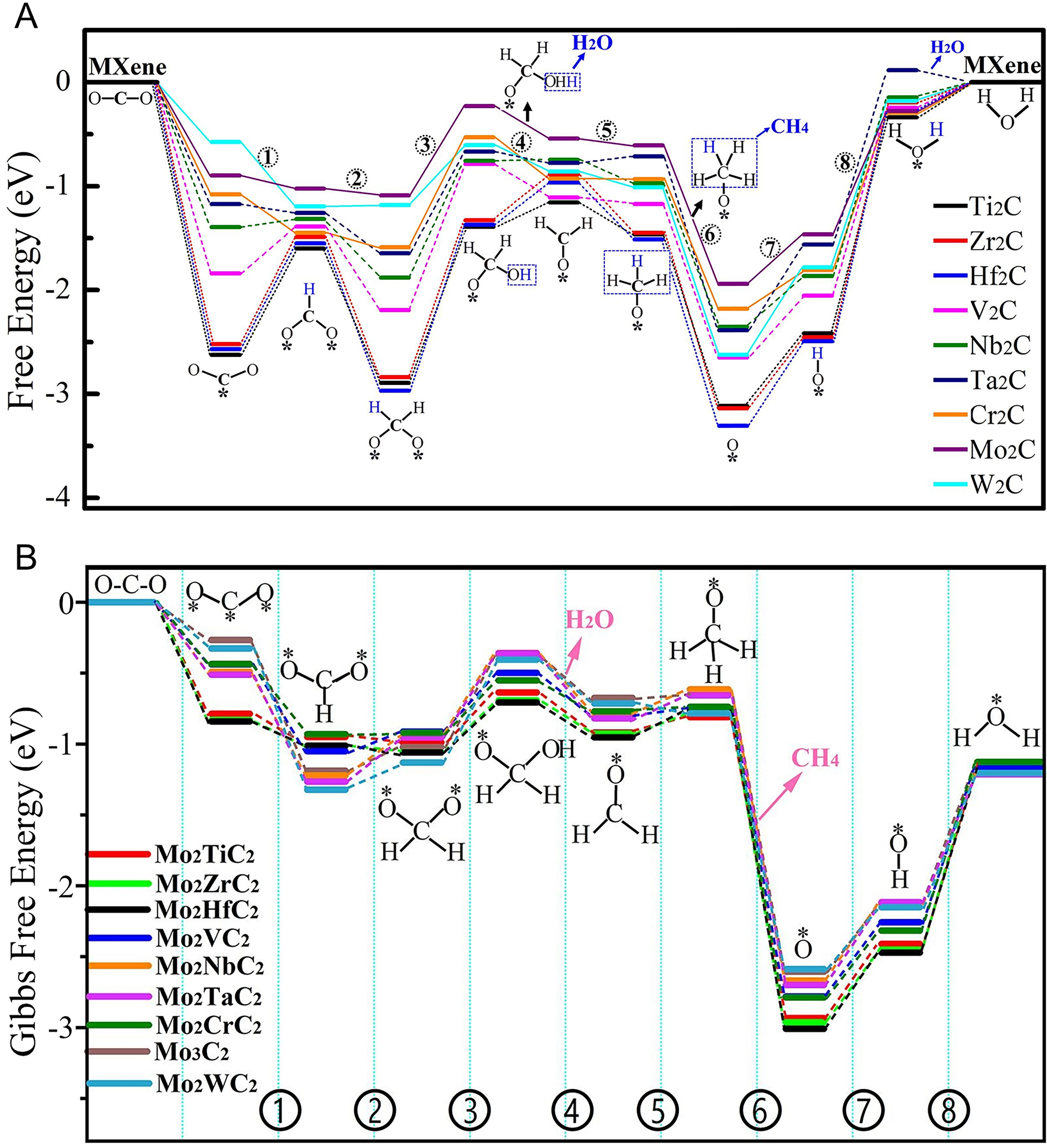
Constructing effective active sites is the fundamental solution to addressing the suite of limiting factors of CO2 photoreduction. Previous studies have focused on utilizing MXenes as a cocatalyst to enhance PCR activity, while research on using MXenes as direct photocatalysts to drive CO2 reduction is quite limited. Therefore, elucidating the intrinsic behavior of CO2 reduction by MXenes is crucial for guiding the design of MXenes-based composite photocatalysts. Recently, extensive theoretical studies have shown that more than 20 types of MXenes exhibit spontaneous adsorption behavior towards CO2. It is noteworthy that the Gibbs free energy for CO adsorption on MXenes is negative [Table 2, Figure 13A and B], indicating that MXenes thermodynamically favor the stability and hydrogenation of the CO* intermediate. This result highlights the potential of designing MXene-based photocatalysts for the deep hydrogenation of CO2 into high-value-added solar fuels.
Figure 13. (A) Gibbs free energy path of the CO2 reduction into CH4 catalyzed by M2C-type MXenes. (A) Reproduced with permission[145]. Copyright Elsevier; (B) The Gibbs free energy diagram of the most feasible pathway for catalyzing CO2 into CH4 and H2O by Mo2MC2-type MXenes. (B) Reproduced with permission[146]. Copyright Elsevier.
Gibbs free energies of the intermediate for CO2 reduction into CH4 by M3C2-type MXenes[144]
| group IV | group V | group VI | ||||||
| Species/M3C2 | Ti3C2 | Zr3C2 | Hf3C2 | V3C2 | Nb3C2 | Ta3C2 | Cr3C2 | Mo3C2 |
| *CO2 | -0.59 | 0.17 | 0.18 | 0.29 | 0.35 | 0.25 | 0.15 | |
| **CO2 | -3.01 | -3.19 | -3.05 | -1.47 | -1.60 | -2.30 | -1.29 | -2.11 |
| **OCHO• | -2.04 | -2.25 | -2.89 | -1.40 | -1.71 | -1.58 | -1.61 | -1.74 |
| **HOCO• | -2.06 | -2.49 | -2.79 | -1.41 | -1.54 | -1.92 | -1.74 | -1.91 |
| **•OCH2O• | -3.51 | -4.08 | -4.31 | -1.93 | -2.22 | -2.86 | -1.60 | -1.64 |
| **HCOOH | -1.01 | -2.19 | -2.47 | -0.15 | -0.12 | -0.32 | 0.01 | -0.78 |
| **CO | -1.18 | -1.11 | -1.54 | -1.45 | -1.39 | -1.80 | -2.00 | -2.27 |
| **HOCH2O• | -2.47 | -2.82 | -3.11 | -1.59 | -1.88 | -2.56 | -1.85 | -2.15 |
| **HOCH2OH | -1.09 | -1.07 | -3.56 | -0.61 | -0.68 | -1.10 | -0.69 | -0.90 |
| **H2CO | -2.43 | -3.21 | -3.31 | -1.81 | -2.16 | -2.39 | -1.78 | -1.86 |
| **CH2OH• | -1.58 | -2.05 | -1.89 | -1.26 | -1.36 | -1.94 | -1.51 | -1.64 |
| **CH3O• | -2.93 | -3.12 | -3.33 | -2.20 | -2.36 | -2.98 | -2.12 | -2.53 |
| **CH2 | -1.81 | -1.88 | -1.21 | -0.97 | -1.47 | -2.33 | -1.65 | -2.11 |
| **CH3OH | 0.06 | 0.07 | 0.01 | 0.33 | 0.08 | 0.22 | 0.33 | 0.17 |
| **O | -4.80 | -5.24 | -5.23 | -3.45 | -3.95 | -4.27 | -3.53 | -3.57 |
| **CH3 | -2.04 | -2.37 | -2.86 | -2.26 | -2.47 | -3.27 | -2.52 | -2.98 |
| **OH• | -4.59 | -4.65 | -4.80 | -3.70 | -3.90 | -4.35 | -3.73 | -3.91 |
| **CH4 | -1.18 | -0.70 | -0.94 | -2.15 | -0.77 | -0.82 | -0.55 | -0.70 |
| **H2O | -3.03 | -2.96 | -3.04 | -2.46 | -2.37 | -2.64 | -2.55 | -2.88 |
Additionally, Mo-based MXenes show unique selectivity for the methanation of CO2 [Table 2, Figure 13A and B][144-146]. DFT calculations have simulated the methanation characteristics of eight types of M3C2 MXenes (M = Ti, Zr, Hf, V, Nb, Ta, Cr, and Mo), all exhibiting spontaneous CO2 chemical adsorption behavior [Table 2][144]. Among them, Mo3C2 stands out as the most promising catalyst for the methanation of CO2[144]. Additionally, the Gibbs free energy of CO2 adsorption on Mo3C2 is significantly higher than that of H2O, indicating its effective suppression of the competitive reaction (HER)[144]. Among nine types of M2C-type MXenes, Mo2C demonstrates the thermodynamically optimal behavior for the methanation of CO2
CONCLUSIONS AND FUTURE PERSPECTIVES
In conclusion, this review first provides a comprehensive overview on the fundamentals of PCR and MXenes. Then, recent advances in MXenes-based photocatalysts for CO2 reduction are summarized according to the roles of MXenes in enhancing the activity of the photocatalysts. On the one hand, the functions of MXenes in facilitating the CO2 reduction reaction have been demonstrated by the available reports; on the other hand, the sufficient understanding of the underlying mechanisms of MXenes in the system still needs more in-depth exploration. For instance, it is widely recognized that the construction of Schottky junctions by combining the metallic MXenes with semiconductors can enhance CO2 reduction performance of MXenes-based photocatalysts by promoting charge separation and transfer. In most reports in this regard, the MXenes are often treated as an alternative to other metallic cocatalysts to serve as electron reservoirs. Noteworthily, MXenes themselves possess unique electronic and surface properties, such as LSPR effects, the strong support effect, and distinctive CO2 activation capabilities. Therefore, further upgrading the performance of MXenes-based photocatalysts for CO2 reduction is expected to be realized by fully exploiting the advantages of MXenes according to the following directions for future research.
Firstly, the utilization of the strong support effects induced by Ti3C2Tx MXene to regulate the electronic structure of photocatalysts has been preliminarily validated[64]. When designing MXenes-based photocatalysts, it is crucial to consider and, most importantly, harness the strong support effects induced by MXenes on the electronic structure of active catalysts. These effects can influence the charge separation of the photocatalyst, the activation of CO2, and the reaction barriers for intermediates. Moreover, the potential strong support effects induced by MXenes other than Ti3C2Txare worthy of further investigation when used as photocatalytic supports.
Secondly, tuning the LSPR absorption characteristics of traditional plasmonic metals can alter the photoactivity of CO2 reduction and the selectivity of the products[147]. Many MXenes exhibit unique LSPR absorption characteristics, but the mechanisms underlying their LSPR generation and their roles in CO2 reduction require further investigation. This will provide valuable insights into utilizing the LSPR of different MXenes to enhance the photoactivity of CO2 reduction and tune the product distribution. Additionally, to regulate the LSPR characteristics of MXenes, we should not only focus on the adjustment of surface functional groups but also consider the introduction of heteroatoms that are beneficial for CO2 photoreduction.
Currently, metallic Ti3C2Tx MXene is primarily used as a cocatalyst to enhance the photoactivity of active catalysts. Effective strategies for opening the bandgap of Ti3C2Tx to transform it into a semiconductor for direct CO2 photoreduction still require further investigation. In addition, exploring the synthesis of semiconductor-type MXenes based on theoretically predicted band structures holds promise to obtain high-performance CO2 reduction photocatalysts.
Finally, theoretical studies indicate that the active sites of MXenes offer several advantages for the selective reduction of CO2, including spontaneous physicochemical adsorption and activation capabilities, pathways for electron or proton transfer to intermediates, regulation of CO2 reaction pathways and selectivity, and reduction of the energy barriers for CO2 reduction reactions. Designing efficient photocatalysts focusing on MXenes as active sites for CO2 photoreduction will be a key focus of future research.
It is confirmed that MXenes hold great promise for the photocatalytic conversion of CO2 into deeply hydrogenated solar fuels due to their rich and tunable electronic and physicochemical properties. More attention should be paid to fully leveraging the advantages of MXenes and thoroughly understanding their fundamental properties to design efficient CO2 reduction catalysts. Additionally, MXenes offer significant cost and yield advantages; some MXenes, such as Ti3C2Tx, Nb2CTx, Ti2CTx, Mo2CTx, and Ti3CNTx, can now be produced at the kilogram scale[148], making them commercially viable for the preparation of MXenes-based photocatalyst or using as direct photocatalysts. Focusing on the aforementioned directions as research priorities, it is expected to further understand and explore the potential of MXenes in constructing high-performance catalysts for CO2 photoreduction.
DECLARATIONS
Authors’ contributions
Prepared and revised the manuscript: Xie, G.; Zhang, S.; Zhan, R.
Revised the manuscript: Xie, X.; Zhang, N.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (52272295, 52071137, 51977071, 51802040, and 21802020), the Science and Technology Innovation Program of Hunan Province (2021RC3066 and 2021RC3067) and Natural Science Foundation of Hunan Province (2020JJ3004 and 2020JJ4192). Zhang, N. and Xie, X. also acknowledge the financial support of the Fundamental Research Funds for the Central Universities.
Conflict of interests
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
2. Johnson, E. J.; Sugerman, E. R.; Morwitz, V. G.; Johar, G. V.; Morris, M. W. Widespread misestimates of greenhouse gas emissions suggest low carbon competence. Nat. Clim. Chang. 2024, 14, 707-14.
3. Ma, Y.; Zhang, Y.; Xie, G.; et al. Isolated Cu sites in CdS hollow nanocubes with doping-location-dependent performance for photocatalytic CO2 reduction. ACS. Catal. 2024, 14, 1468-79.
4. Albero, J.; Peng, Y.; García, H. Photocatalytic CO2 reduction to C2+ products. ACS. Catal. 2020, 10, 5734-49.
5. Ouyang, H.; Peng, M.; Song, K.; Wang, S.; Gao, H.; Tan, B. Synthesis of metal-phenanthroline-modified hypercrosslinked polymer for enhanced CO2 capture and conversion via chemical and photocatalytic methods under ambient conditions. Chem. Synth. 2024, 4, 50.
6. Liu, W. R.; Yu, S.; Liu, Z.; et al. Hierarchical hollow TiO2@Bi2WO6 with light-driven excited Bi(3-x)+ sites for efficient photothermal catalytic CO2 reduction. Inorg. Chem. 2024, 63, 6714-22.
7. Yin, Y.; Kang, X.; Han, B. Two-dimensional materials: synthesis and applications in the electro-reduction of carbon dioxide. Chem. Synth. 2022, 2, 19.
8. He, Y.; Lei, Q.; Li, C.; Han, Y.; Shi, Z.; Feng, S. Defect engineering of photocatalysts for solar-driven conversion of CO2 into valuable fuels. Materials. Today. 2021, 50, 358-84.
9. Guo, Q.; Zhou, C.; Ma, Z.; Yang, X. Fundamentals of TiO2 photocatalysis: concepts, mechanisms, and challenges. Adv. Mater. 2019, 31, e1901997.
10. Xu, Q.; Xia, Z.; Zhang, J.; et al. Recent advances in solar-driven CO2 reduction over g-C3N4 -based photocatalysts. Carbon. Energy. 2023, 5, e205.
11. Kshirsagar, S. D.; Shelake, S. P.; Biswas, B.; et al. Emerging ZnO semiconductors for photocatalytic CO2 reduction to methanol. Small 2024, 20, e2407318.
12. Meng, A.; Zhang, L.; Cheng, B.; Yu, J. Dual cocatalysts in TiO2 photocatalysis. Adv. Mater. 2019, 31, e1807660.
13. Fu, J.; Yu, J.; Jiang, C.; Cheng, B. g-C3N4-based heterostructured photocatalysts. Adv. Energy. Mater. 2018, 8, 1701503.
14. Pawar, A. U.; Pal, U.; Zheng, J. Y.; Kim, C. W.; Kang, Y. S. Thermodynamically controlled photo-electrochemical CO2 reduction at Cu/rGO/PVP/Nafion multi-layered dark cathode for selective production of formaldehyde and acetaldehyde. Appl. Catal. B. Environ. 2022, 303, 120921.
15. Zhang, T.; Huang, Z.; Xie, G.; et al. A non-equivalent Ni doped La-MOF for enhanced photocatalytic CO2 reduction through oxygen vacancy regulation and electronic structure optimization. Inorg. Chem. Front. 2024, 11, 8890-901.
16. Miao, R.; Bao, Y.; Yang, G.; et al. Lowering schottky barrier in MoC-based cocatalyst via heteroatoms tunning for enhanced photocatalytic hydrogen evolution. Chem. Eng. J. 2024, 497, 154909.
17. Wu, W.; Xu, L.; Lu, Q.; et al. Addressing the carbonate issue: electrocatalysts for acidic CO2 reduction reaction. Adv. Mater. 2025, 37, e2312894.
18. You, S.; Xiao, J.; Liang, S.; et al. Doping engineering of Cu-based catalysts for electrocatalytic CO2 reduction to multi-carbon products. Energy. Environ. Sci. 2024, 17, 5795-818.
19. Chang, F.; Xiao, M.; Miao, R.; et al. Copper-based catalysts for electrochemical carbon dioxide reduction to multicarbon products. Electrochem. Energy. Rev. 2022, 5, 139.
20. Lai, W.; Qiao, Y.; Zhang, J.; Lin, Z.; Huang, H. Design strategies for markedly enhancing energy efficiency in the electrocatalytic CO2 reduction reaction. Energy. Environ. Sci. 2022, 15, 3603-29.
21. Zhao, M.; Casiraghi, C.; Parvez, K. Electrochemical exfoliation of 2D materials beyond graphene. Chem. Soc. Rev. 2024, 53, 3036-64.
22. Yang, J.; Zeng, X.; Tebyetekerwa, M.; et al. Engineering 2D photocatalysts for solar hydrogen peroxide production. Adv. Energy. Mater. 2024, 14, 2400740.
23. Li, Q.; Wu, K.; Zhu, H.; Yang, Y.; He, S.; Lian, T. Charge transfer from Quantum-Confined 0D, 1D, and 2D nanocrystals. Chem. Rev. 2024, 124, 5695-763.
24. Zeng, W.; Ye, X.; Dong, Y.; et al. MXene for photocatalysis and photothermal conversion: synthesis, physicochemical properties, and applications. Coord. Chem. Rev. 2024, 508, 215753.
25. Yang, R.; Fan, Y.; Zhang, Y.; et al. 2D transition metal dichalcogenides for photocatalysis. Angew. Chem. Int. Ed. 2023, 135, e202218016.
26. Cox, C. R.; Lee, J. Z.; Nocera, D. G.; Buonassisi, T. Ten-percent solar-to-fuel conversion with nonprecious materials. Proc. Natl. Acad. Sci. U. S. A. 2014, 111, 14057-61.
27. Ullah, N.; Xie, M.; Hussain, S.; et al. Simultaneous synthesis of bimetallic@3D graphene electrocatalyst for HER and OER. Front. Mater. Sci. 2021, 15, 305-15.
28. Pedersen, P. D.; Vegge, T.; Bligaard, T.; Hansen, H. A. Trends in CO2 reduction on transition metal dichalcogenide edges. ACS. Catal. 2023, 13, 2341-50.
29. He, M.; Tian, Z.; Lin, H.; Wang, G. Dual-atom P-Co-Dy charge-transfer bridge on black phosphorus for enhanced photocatalytic CO2 reduction. Small 2024, 20, e2404162.
30. Niu, R.; Liu, Q.; Huang, B.; et al. Black phosphorus/Bi19Br3S27 van der Waals heterojunctions ensure the supply of activated hydrogen for effective CO2 photoreduction. Appl. Catal. B. Environ. 2022, 317, 121727.
31. Li, X.; Huang, Z.; Shuck, C. E.; Liang, G.; Gogotsi, Y.; Zhi, C. MXene chemistry, electrochemistry and energy storage applications. Nat. Rev. Chem. 2022, 6, 389-404.
32. Yin, X.; Zheng, W.; Tang, H.; et al. Unraveling cation intercalation mechanism in MXene for enhanced supercapacitor performance. Researchsquare 2024. Available online: https://www.researchsquare.com/article/rs-4161663/v1. (accessed 10 April 2025).
33. Zhang, P.; Wang, X.; Zhang, Y.; et al. Burgeoning Silicon/MXene nanocomposites for lithium ion batteries: a review. Adv. Funct. Mater. 2024, 34, 2402307.
34. Han, M.; Maleski, K.; Shuck, C. E.; et al. Tailoring electronic and optical properties of MXenes through forming solid solutions. J. Am. Chem. Soc. 2020, 142, 19110-8.
35. Kim, H.; Wang, Z.; Alshareef, H. N. MXetronics: electronic and photonic applications of MXenes. Nano. Energy. 2019, 60, 179-97.
36. Xu, T.; Tan, S.; Li, S.; et al. Synergistic densification in hybrid organic-inorganic MXenes for optimized photothermal conversion. Adv. Funct. Mater. 2024, 34, 2400424.
37. Xie, G.; Han, C.; Song, F.; et al. A study on the role of plasmonic Ti3C2Tx MXene in enhancing photoredox catalysis. Nanoscale 2022, 14, 18010-21.
38. Xie, G.; Liao, L.; Wang, J.; et al. Strong support effect induced by MXene for the synthesis of metal sulfides nanosheet arrays with sulfur vacancies towards selective CO2-to-CO photoreduction. Science. Bulletin. 2024, 69, 3247-59.
39. Guo, J.; Ma, G.; Liu, G.; Dai, C.; Lin, Z. Ti2CTx MXene cathode host for enhanced zinc‐bromine battery performance. Adv. Energy. Mater. 2024, 14, 2304516.
40. Zhao, Q.; Zhou, W.; Zhang, M.; et al. Edge-enriched Mo2TiC2Tx/MoS2 heterostructure with coupling interface for selective NO2 monitoring. Adv. Funct. Mater. 2022, 32, 2203528.
41. Raveendran, P.; Wallen, S. L. Cooperative C–H···O hydrogen bonding in CO2-Lewis base complexes: implications for solvation in supercritical CO2. J. Am. Chem. Soc. 2002, 124, 12590-9.
42. Murphy, L. J.; Robertson, K. N.; Kemp, R. A.; Tuononen, H. M.; Clyburne, J. A. C. Structurally simple complexes of CO2. Chem. Commun. 2015, 51, 3942-56.
43. Yu, H.; Wu, H.; Chow, Y. L.; Wang, J.; Zhang, J. Revolutionizing electrochemical CO2 reduction to deeply reduced products on non-Cu-based electrocatalysts. Energy. Environ. Sci. 2024, 17, 5336-64.
44. Yang, X.; Jiang, D.; Cheng, X.; et al. Adsorption properties of seaweed-based biochar with the greenhouse gases (CO2, CH4, N2O) through density functional theory (DFT). Biomass. Bioenergy. 2022, 163, 106519.
45. Jia, G.; Zhang, Y.; Yu, J. C.; Guo, Z. Asymmetric atomic dual-sites for photocatalytic CO2 reduction. Adv. Mater. 2024, 36, e2403153.
46. Kumaravel, V.; Mathew, S.; Bartlett, J.; Pillai, S. C. Photocatalytic hydrogen production using metal doped TiO2: a review of recent advances. Appl. Catal. B. Environ. 2019, 244, 1021-64.
47. Starukh, H.; Praus, P. Doping of graphitic carbon nitride with non-metal elements and its applications in photocatalysis. Catalysts 2020, 10, 1119.
48. Liao, L.; Xie, G.; Yu, C.; et al. Active site-exposed Bi2WO6@BiOCl heterostructures for photocatalytic hydrogenation of nitroaromatic compounds. Nanoscale 2024, 16, 19704-14.
49. Sun, P.; Xing, Z.; Li, Z.; Zhou, W. Recent advances in quantum dots photocatalysts. Chem. Eng. J. 2023, 458, 141399.
50. Li, S.; Miao, P.; Zhang, Y.; et al. Recent advances in plasmonic nanostructures for enhanced photocatalysis and electrocatalysis. Adv. Mater. 2021, 33, e2000086.
51. Zhu, Y.; Xie, G.; Li, G.; et al. Facial synthesis of two-dimensional In2S3/Ti3C2Tx heterostructures with boosted photoactivity for the hydrogenation of nitroaromatic compounds. Mater. Chem. Front. 2021, 5, 6883-90.
52. Xu, C.; Ravi, A. P.; Aymonier, C.; Luque, R.; Marre, S. Nanostructured materials for photocatalysis. Chem. Soc. Rev. 2019, 48, 3868-902.
53. Wang, Z.; Lin, Z.; Shen, S.; Zhong, W.; Cao, S. Advances in designing heterojunction photocatalytic materials. Chin. J. Catal. 2021, 42, 710-30.
54. Li, X.; Yu, J.; Wageh, S.; Al-Ghamdi, A. A.; Xie, J. Graphene in photocatalysis: a review. Small 2016, 12, 6640-96.
55. Chen, H.; Liang, X.; Liu, Y.; Ai, X.; Asefa, T.; Zou, X. Active site engineering in porous electrocatalysts. Adv. Mater. 2020, 32, e2002435.
56. Wang, N.; Cheng, L.; Liao, Y.; Xiang, Q. Effect of functional group modifications on the photocatalytic performance of g-C3N4. Small 2023, 19, e2300109.
57. Bai, S.; Zhang, N.; Gao, C.; Xiong, Y. Defect engineering in photocatalytic materials. Nano. Energy. 2018, 53, 296-336.
58. Khan, I.; Khan, S.; Wu, S. Y.; et al. Synergistic functionality of dopants and defects in Co-phthalocyanine/B-CN Z-scheme photocatalysts for promoting photocatalytic CO2 reduction reactions. Small 2023, 19, e2208179.
59. Wang, J.; Xie, G.; Yu, C.; et al. Stabilizing Ti3C2Tx in a water medium under multiple environmental conditions by scavenging oxidative free radicals. Chem. Mater. 2022, 34, 9517-26.
60. Murali, G.; Reddy, M. J. K.; Park, Y. H.; et al. A review on MXene synthesis, stability, and photocatalytic applications. ACS. Nano. 2022, 16, 13370-429.
61. Ali, S.; Iqbal, R.; Wahid, F.; et al. Cobalt coordinated two-dimensional covalent organic framework a sustainable and robust electrocatalyst for selective CO2 electrochemical conversion to formic acid. Fuel. Process. Technol. 2022, 237, 107451.
62. Wang, J.; Li, G.; Xie, G.; et al. Solvation structure design for stabilizing MXene in transition metal ion solutions. SusMat 2024, 4, e202.
63. Liu, X.; Chen, T.; Xue, Y.; et al. Nanoarchitectonics of MXene/semiconductor heterojunctions toward artificial photosynthesis via photocatalytic CO2 reduction. Coord. Chem. Rev. 2022, 459, 214440.
64. Li, D.; Yang, K.; Lian, J.; Yan, J.; Liu, S. Powering the world with solar fuels from photoelectrochemical CO2 reduction: basic principles and recent advances. Adv. Energy. Mater. 2022, 12, 2201070.
65. Zhang, C.; Lin, Z.; Jiao, L.; Jiang, H. L. Metal-organic frameworks for electrocatalytic CO2 reduction: from catalytic site design to microenvironment modulation. Angew. Chem. Int. Ed. Engl. 2024, 63, e202414506.
66. Wang, Y.; Liu, J.; Zheng, G. Designing copper-based catalysts for efficient carbon dioxide electroreduction. Adv. Mater. 2021, 33, e2005798.
67. Deng, S.; Wang, R.; Feng, X.; et al. Dual lewis acid-base sites regulate silver-copper bimetallic oxide nanowires for highly selective photoreduction of carbon dioxide to methane. Angew. Chem. Int. Ed. 2023, 135, e202309625.
68. Li, X.; Sun, Y.; Xu, J.; et al. Selective visible-light-driven photocatalytic CO2 reduction to CH4 mediated by atomically thin CuIn5S8 layers. Nat. Energy. 2019, 4, 690-9.
69. Li, J.; Huang, H.; Xue, W.; et al. Self-adaptive dual-metal-site pairs in metal-organic frameworks for selective CO2 photoreduction to CH4. Nat. Catal. 2021, 4, 719-29.
70. Huang, H.; Song, H.; Kou, J.; Lu, C.; Ye, J. Atomic-level insights into surface engineering of semiconductors for photocatalytic CO2 reduction. J. Energy. Chem. 2022, 67, 309-41.
71. Liao, L.; Xie, G.; Xie, X.; Zhang, N. Advances in modulating the activity and selectivity of photocatalytic CO2 reduction to multicarbon products. J. Phys. Chem. C. 2023, 127, 2766-81.
72. Naguib, M.; Kurtoglu, M.; Presser, V.; et al. Two-dimensional nanocrystals: two-dimensional nanocrystals produced by exfoliation of Ti3AlC2 (Adv. Mater. 37/2011). Adv. Mater. 2011, 23, 4207-4207.
73. Seidi, F.; Arabi, S. A.; Dadashi, F. M.; et al. MXenes antibacterial properties and applications: a review and perspective. Small 2023, 19, e2206716.
75. Anasori, B.; Lukatskaya, M. R.; Gogotsi, Y. 2D metal carbides and nitrides (MXenes) for energy storage. Nat. Rev. Mater. 2017, 2, BFnatrevmats201698.
76. VahidMohammadi, A.; Rosen, J.; Gogotsi, Y. The world of two-dimensional carbides and nitrides (MXenes). Science 2021, 372, eabf1581.
77. Liao, C.; Zhou, H.; Zhang, S.; Wang, F.; Liu, Y.; Guo, L. Copper vacancy and LSPR-activated mxene synergistically enabling selective photoreduction CO2 to acetate. ChemSusChem 2024, 17, e202301927.
78. Qu, J.; Yang, T.; Zhang, P.; et al. Artificial photosynthesis platform of 2D/2D MXene/crystalline covalent organic frameworks heterostructure for efficient photoenzymatic CO2 reduction. Appl. Catal. B. Environ. Energy. 2024, 348, 123827.
79. Yang, F.; Zhang, P.; Qu, J.; et al. Highly efficient photoenzymatic CO2 reduction dominated by 2D/2D MXene/C3N5 heterostructured artificial photosynthesis platform. J. Colloid. Interface. Sci. 2025, 678, 1121-31.
80. Wei, P.; Zhang, Y.; Dong, J.; et al. Engineering MXene-based photocatalyst for efficient NADH regeneration and photoenzymatic CO2 reduction without electron mediator. Appl. Catal. B. Environ. Energy. 2024, 357, 124257.
81. Tahir, M.; Tahir, B. 2D/2D/2D O-C3N4/Bt/Ti3C2Tx heterojunction with novel MXene/clay multi-electron mediator for stimulating photo-induced CO2 reforming to CO and CH4. Chem. Eng. J. 2020, 400, 125868.
82. Chen, W.; Han, B.; Xie, Y.; Liang, S.; Deng, H.; Lin, Z. Ultrathin Co-Co LDHs nanosheets assembled vertically on MXene: 3D nanoarrays for boosted visible-light-driven CO2 reduction. Chem. Eng. J. 2020, 391, 123519.
83. Tahir, M.; Tahir, B. In-situ growth of TiO2 imbedded Ti3C2TA nanosheets to construct PCN/Ti3C2TA MXenes 2D/3D heterojunction for efficient solar driven photocatalytic CO2 reduction towards CO and CH4 production. J. Colloid. Interface. Sci. 2021, 591, 20-37.
84. Chen, Y.; Qi, M.; Li, Y.; et al. Activating two-dimensional Ti3C2Tx-MXene with single-atom cobalt for efficient CO2 photoreduction. Cell. Rep. Phys. Sci. 2021, 2, 100371.
85. Wu, Y.; Xu, W.; Tang, W.; et al. In-situ annealed “M-scheme” MXene-based photocatalyst for enhanced photoelectric performance and highly selective CO2 photoreduction. Nano. Energy. 2021, 90, 106532.
86. Zhang, Z.; Wang, B.; Zhao, H.; et al. Self-assembled lead-free double perovskite-MXene heterostructure with efficient charge separation for photocatalytic CO2 reduction. Appl. Catal. B. Environ. 2022, 312, 121358.
87. Syuy, A. V.; Shtarev, D. S.; Kozlova, E. A.; et al. Photocatalytic activity of TiNbC-modified TiO2 during hydrogen evolution and CO2 reduction. Appl. Sci. 2023, 13, 9410.
88. Liu, Y.; Tan, G.; Feng, S.; et al. Localized surface plasmon resonance effect of V4C3-MXene for enhancing photothermal reduction of CO2 in full solar spectrum. Sep. Purif. Technol. 2023, 326, 124726.
89. Tahir, M. Vanadium carbide (V2CTx) MXene-supported exfoliated g-C3N4 with the role of hole scavenger as a rapid electron transfer channel for enhancing photocatalytic CO2 reduction to CO and CH4. Energy. Fuels. 2023, 37, 10615-30.
90. Zhang, S.; Wang, Y.; Mersal, G. A. M.; et al. Enhanced photocatalytic CO2 reduction via MXene synergism: constructing an efficient heterojunction structure of g-C3N4/Nb2C/CsPbBr3. Adv. Compos. Hybrid. Mater. 2024, 7, 1026.
91. Tang, Q.; Xiong, P.; Wang, H.; Wu, Z. Boosted CO2 photoreduction performance on Ru-Ti3CN MXene-TiO2 photocatalyst synthesized by non-HF Lewis acidic etching method. J. Colloid. Interface. Sci. 2022, 619, 179-87.
92. Vasileff, A.; Xu, C.; Jiao, Y.; Zheng, Y.; Qiao, S. Surface and interface engineering in copper-based bimetallic materials for selective CO2 electroreduction. Chem 2018, 4, 1809-31.
93. An, Y.; Tian, Y.; Shen, H.; Man, Q.; Xiong, S.; Feng, J. Two-dimensional MXenes for flexible energy storage devices. Energy. Environ. Sci. 2023, 16, 4191-250.
94. Cao, W.; Nie, J.; Cao, Y.; et al. A review of how to improve Ti3C2Tx MXene stability. Chem. Eng. J. 2024, 496, 154097.
95. Patil, A. M.; Jadhav, A. A.; Chodankar, N. R.; et al. Recent progress of MXene synthesis, properties, microelectrode fabrication techniques for microsupercapacitors and microbatteries energy storage devices and integration: a comprehensive review. Coord. Chem. Rev. 2024, 517, 216020.
96. Wang, Y.; Wang, Y.; Jian, M.; Jiang, Q.; Li, X. MXene key composites: a new arena for gas sensors. Nanomicro. Lett. 2024, 16, 209.
97. Sherryna, A.; Tahir, M. Role of surface morphology and terminating groups in titanium carbide MXenes (Ti3C2Tx) cocatalysts with engineering aspects for modulating solar hydrogen production: a critical review. Chem. Eng. J. 2022, 433, 134573.
98. Natu, V.; Barsoum, M. W. MXene surface terminations: a perspective. J. Phys. Chem. C. 2023, 127, 20197-206.
99. Song, W.; Chen, J.; Li, Z.; Fang, X. Self-powered MXene/GaN van der Waals heterojunction ultraviolet photodiodes with superhigh efficiency and stable current outputs. Adv. Mater. 2021, 33, e2101059.
100. Zhang, D.; Shah, D.; Boltasseva, A.; Gogotsi, Y. MXenes for photonics. ACS. Photonics. 2022, 9, 1108-16.
101. Lioi, D. B.; Stevenson, P. R.; Seymour, B. T.; et al. Simultaneous ultrafast transmission and reflection of nanometer-thick Ti3C2Tx MXene films in the visible and near-infrared: implications for energy storage, electromagnetic shielding, and laser systems. ACS. Appl. Nano. Mater. 2020, 3, 9604-9.
102. Mauchamp, V.; Bugnet, M.; Bellido, E. P.; et al. Enhanced and tunable surface plasmons in two-dimensional Ti3C2 stacks: electronic structure versus boundary effects. Phys. Rev. B. 2014, 89, 235428.
103. El-Demellawi, J. K.; Lopatin, S.; Yin, J.; Mohammed, O. F.; Alshareef, H. N. Tunable multipolar surface plasmons in 2D Ti3C2Tx MXene flakes. ACS. Nano. 2018, 12, 8485-93.
104. Zhang, C.; Anasori, B.; Seral-ascaso, A.; et al. Transparent, flexible, and conductive 2D titanium carbide (MXene) films with high volumetric capacitance. Adv. Mater. 2017, 29, 1702678.
105. Ying, G.; Kota, S.; Dillon, A. D.; Fafarman, A. T.; Barsoum, M. W. Conductive transparent V2CTx (MXene) films. FlatChem 2018, 8, 25-30.
106. Li, G.; Wyatt, B. C.; Song, F.; et al. 2D titanium carbide (MXene) based films: expanding the frontier of functional film materials. Adv. Funct. Mater. 2021, 31, 2105043.
107. Xie, X.; Zhang, N. Positioning MXenes in the photocatalysis landscape: competitiveness, challenges, and future perspectives. Adv. Funct. Mater. 2020, 30, 2002528.
108. Li, G.; Lian, S.; Wang, J.; Xie, G.; Zhang, N.; Xie, X. Surface chemistry engineering and the applications of MXenes. J. Materiomics. 2023, 9, 1160-84.
109. Wang, K.; Wang, Q.; Zhang, K.; Wang, G.; Wang, H. Selective solar-driven CO2 reduction mediated by 2D/2D Bi2O2SiO3/MXene nanosheets heterojunction. J. Mater. Sci. Technol. 2022, 124, 202-8.
110. Zeng, Z.; Yan, Y.; Chen, J.; Zan, P.; Tian, Q.; Chen, P. Boosting the photocatalytic ability of Cu2O nanowires for CO2 conversion by MXene quantum dots. Adv. Funct. Mater. 2019, 29, 1806500.
111. He, F.; Zhu, B.; Cheng, B.; Yu, J.; Ho, W.; Macyk, W. 2D/2D/0D TiO2/C3N4/Ti3C2 MXene composite S-scheme photocatalyst with enhanced CO2 reduction activity. Appl. Catal. B. Environ. 2020, 272, 119006.
112. Wang, Y.; Hu, X.; Song, H.; et al. Oxygen vacancies in actiniae-like Nb2O5/Nb2C MXene heterojunction boosting visible light photocatalytic NO removal. Appl. Catal. B. Environ. 2021, 299, 120677.
113. Chen, Y.; Wang, Z.; Zhang, Y.; et al. S-scheme and schottky junction synchronous regulation boost hierarchical
114. Xu, W.; Li, X.; Peng, C.; et al. One-pot synthesis of Ru/Nb2O5@Nb2C ternary photocatalysts for water splitting by harnessing hydrothermal redox reactions. Appl. Catal. B. Environ. 2022, 303, 120910.
115. Tang, C.; Zheng, X.; Chen, X.; Fu, Y.; Lü, Q. Defect-rich MoS2/CoS2 on Mo2TiC2Tx MXene as an efficient catalyst for hydrogen evolution reaction in acidic media. FlatChem 2023, 42, 100581.
116. Fan, Y.; Hao, X.; Yi, N.; Jin, Z. Strong electronic coupling of Mo2TiC2 MXene/ZnCdS ohmic junction for boosting photocatalytic hydrogen evolution. Appl. Catal. B. Environ. Energy. 2024, 357, 124313.
117. Zhang, N.; Han, C.; Fu, X.; Xu, Y. Function-oriented engineering of metal-based nanohybrids for photoredox catalysis: exerting plasmonic effect and beyond. Chem 2018, 4, 1832-61.
118. Wu, X.; Wang, J.; Wang, Z.; et al. Boosting the electrocatalysis of MXenes by plasmon-induced thermalization and hot-electron injection. Angew. Chem. Int. Ed. Engl. 2021, 60, 9416-20.
119. Gao, Y.; Wang, Y.; Sun, R.; Luo, Y.; Xin, L.; Wang, D. Interfacial hot electron injection in Cu2O/MXene-g-C3N4 p-n heterojunction for efficient photocatalytic CO2 reduction. Colloids. Surf. A. Physicochem. Eng. Aspects. 2024, 684, 133236.
120. Liu, T.; Tan, G.; Feng, S.; et al. Dual localized surface plasmon resonance effect enhances Nb2AlC/Nb2C MXene thermally coupled photocatalytic reduction of CO2 hydrogenation activity. J. Colloid. Interface. Sci. 2023, 652, 599-611.
121. Ullah, S.; Ullah, N.; Shah, S. S.; et al. Theoretical and experimental progress in photothermal catalysis for sustainable energy and environmental protection: key problems and strategies towards commercialization. Renew. Sustain. Energy. 2024, 201, 114615.
122. Chen, Y.; Lin, X.; Li, W.; et al. Harnessing photo-to-thermal conversion in sulfur-vulcanized mxene for high-efficiency solar-to-carbon-fuel synthesis. Adv. Funct. Mater. 2024, 34, 2400121.
123. Meng, Y.; Yue, F.; Zhang, S.; et al. Zr-MOF/MXene composite for enhanced photothermal catalytic CO2 reduction in atmospheric and industrial flue gas streams. Carbon. Capture. Sci. Technol. 2024, 13, 100274.
124. Yue, F.; Meng, Y.; Zhang, S.; et al. Efficient solar-driven: photothermal catalytic reduction of atmospheric CO2 at the gas-solid interface by CuTCPP/MXene/TiO2. J. Colloid. Interface. Sci. 2025, 677, 758-70.
125. Guo, C.; Jiang, E.; Chen, Q.; et al. Photo-to-thermal conversion harnessing low-energy photons renders efficient solar CO2 reduction. ACS. Appl. Mater. Interfaces. 2024, 16, 36247-54.
126. Wu, Z.; Li, C.; Li, Z.; et al. Niobium and titanium carbides (MXenes) as superior photothermal supports for CO2 photocatalysis. ACS. Nano. 2021, 15, 5696-705.
127. Handoko, A. D.; Chen, H.; Lum, Y.; Zhang, Q.; Anasori, B.; Seh, Z. W. Two-dimensional titanium and molybdenum carbide MXenes as electrocatalysts for CO2 reduction. iScience 2020, 23, 101181.
128. Liu, A.; Liang, X.; Ren, X.; et al. Recent progress in MXene-based materials: potential high-performance electrocatalysts. Adv. Funct. Mater. 2020, 30, 2003437.
129. Hao, Y.; Hu, F.; Zhu, S.; et al. MXene-regulated metal-oxide interfaces with modified intermediate configurations realizing nearly 100% CO2 electrocatalytic conversion. Angew. Chem. Int. Ed. 2023, 135, e202304179.
130. Cao, S.; Shen, B.; Tong, T.; Fu, J.; Yu, J. 2D/2D heterojunction of ultrathin MXene/Bi2WO6 nanosheets for improved photocatalytic CO2 reduction. Adv. Funct. Mater. 2018, 28, 1800136.
131. Yang, C.; Tan, Q.; Li, Q.; et al. 2D/2D Ti3C2 MXene/g-C3N4 nanosheets heterojunction for high efficient CO2 reduction photocatalyst: dual effects of urea. Appl. Catal. B. Environ. 2020, 268, 118738.
132. Hu, J.; Ding, J.; Zhong, Q. Ultrathin 2D Ti3C2 MXene Co-catalyst anchored on porous g-C3N4 for enhanced photocatalytic CO2 reduction under visible-light irradiation. J. Colloid. Interface. Sci. 2021, 582, 647-57.
133. Tang, Q.; Sun, Z.; Deng, S.; Wang, H.; Wu, Z. Decorating g-C3N4 with alkalinized Ti3C2 MXene for promoted photocatalytic CO2 reduction performance. J. Colloid. Interface. Sci. 2020, 564, 406-17.
134. Feng, W.; Zhu, P.; Li, S.; et al. Engineering fast Ti electron channels to single-atom Fe for enhanced CO2 photoreduction. J. Mater. Chem. A. 2024, 12, 14437-45.
135. Li, H.; Song, Q.; Wan, S.; et al. Atomic interface engineering of single-atom Pt/TiO2-Ti3C2 for boosting photocatalytic CO2 reduction. Small 2023, 19, e2301711.
136. Cai, Y. S.; Chen, J. Q.; Su, P.; et al. Atomically precise metal nanoclusters combine with MXene towards solar CO2 conversion. Chem. Sci. 2024, 15, 13495-505.
137. Luo, Z.; Zhao, G.; Pan, H.; Sun, W. Strong metal-support interaction in heterogeneous catalysts. Adv. Energy. Mater. 2022, 12, 2201395.
138. Yin, P.; Yan, Q.; Liang, H. Strong metal-support interactions through sulfur-anchoring of metal catalysts on carbon supports. Angew. Chem. Int. Ed. Engl. 2023, 135, e202302819.
139. Dong, J.; Fu, Q.; Jiang, Z.; Mei, B.; Bao, X. Carbide-supported au catalysts for water-gas shift reactions: a new territory for the strong metal-support interaction effect. J. Am. Chem. Soc. 2018, 140, 13808-16.
140. Shi, L.; Ren, X.; Wang, Q.; et al. Stabilizing atomically dispersed catalytic sites on tellurium nanosheets with strong metal-support interaction boosts photocatalysis. Small 2020, 16, 2002356.
141. Sun, N.; Zhu, Y.; Li, M.; et al. Thermal coupled photocatalysis over Pt/g-C3N4 for selectively reducing CO2 to CH4 via cooperation of the electronic metal-support interaction effect and the oxidation state of Pt. Appl. Catal. B. Environ. 2021, 298, 120565.
142. Zhang, Y.; Zhao, Q.; Danil, B.; Xiao, W.; Yang, X. Oxygen-vacancy-induced formation of Pt-based intermetallics on MXene with strong metal-support interactions for efficient oxygen reduction reaction. Adv. Mater. 2024, 36, e2400198.
143. Qi, Y.; Zhang, B.; Zhang, G.; et al. Efficient overall water splitting of a suspended photocatalyst boosted by metal-support interaction. Joule 2024, 8, 193-203.
144. Li, N.; Chen, X.; Ong, W.; et al. Understanding of electrochemical mechanisms for CO2 capture and conversion into hydrocarbon fuels in transition-metal carbides (MXenes). ACS. Nano. 2017, 11, 10825-33.
145. Guo, Z.; Li, Y.; Sa, B.; et al. M2C-type MXenes: promising catalysts for CO2 capture and reduction. Appl. Surf. Sci. 2020, 521, 146436.
146. Li, Y.; Chen, Y.; Guo, Z.; et al. Breaking the linear scaling relations in MXene catalysts for efficient CO2 reduction. Chem. Eng. J. 2022, 429, 132171.
147. Zhang, H.; Itoi, T.; Konishi, T.; Izumi, Y. Dual photocatalytic roles of light: charge separation at the band gap and heat via localized surface plasmon resonance to convert CO2 into CO over silver-zirconium oxide. J. Am. Chem. Soc. 2019, 141, 6292-301.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].