HCO3- modified N-heterocyclic carbene silver organic porous polymer catalyzes the conversion of low concentration CO2 into oxazolidinone compounds
Abstract
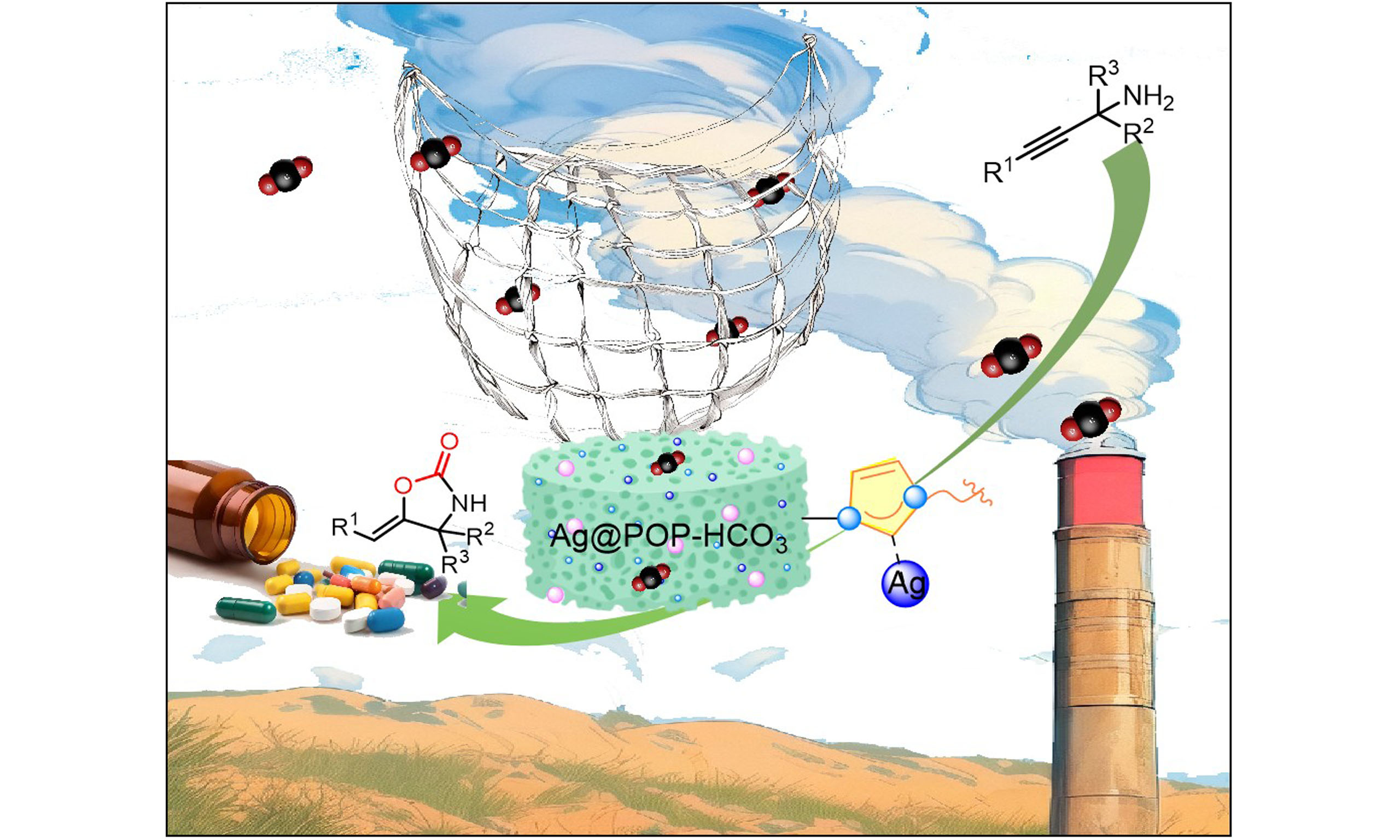
Silver metalized organic porous polymer (Ag@POP-HCO3) was prepared by copolymerizing the HCO3- modified N-heterocyclic carbene monomer loading silver with divinylbenzene. The in-situ conversion of low concentration CO2 from coal-fired flue gas or air into oxazolidinone compounds was achieved through carboxylation cyclization reaction catalyzed by Ag@POP-HCO3 under ambient conditions, without the addition of any cocatalyst. Additionally, both SO2 and NO2 did not interfere with the reaction at normal concentration presented in flue gas. The Ag@POP-HCO3 can effectively catalyze the gram reaction, and its catalytic activity is not significantly reduced after being recycled. The introduction of HCO3- increased the specific surface area and microporous volumes of the catalyst, enhancing its ability to adsorb CO2. Furthermore, N-heterocyclic carbene and HCO3- collaborated to expedite the activation of CO2, while the coordination of silver serves to activate the substrate. The proposed approach avoids cost issues of traditional carbon capture, utilization and storage technology and promotes green chemical process development.
Keywords
INTRODUCTION
The issue of climate change is becoming increasingly severe due to rising global carbon emissions[1]. Carbon capture, utilization and storage (CCUS) technology is considered a key solution to address this issue[2,3]. The process of capturing, utilizing, or storing carbon within the framework of CCUS technology typically involves multiple steps. While this method is crucial for addressing climate concerns, it also presents its own set of challenges, particularly in terms of financial investment and energy usage. For example, the amine-based carbon capture method has become a commonly utilized approach in CCUS technology. This process requires 0.5 to 1 kilogram of amine solution and 1.4 tons of steam to capture a single ton of CO2[4]. Furthermore, the logistics of transporting captured carbon through pipelines also entail significant financial considerations. The costs associated with this transportation infrastructure can range from $50,000 to $700,000 per mile[4]. In addition, the process of carbon storage requires extensive geological exploration and engineering work, leading to potentially high costs. Furthermore, the conversion of CO2 into high-value organic chemicals shows promise for carbon utilization[5,6], but it necessitates the use of pure CO2 obtained after carbon capture and transportation. The aforementioned challenges may currently be influencing the commercial viability of the CCUS technology[7]. In-situ CO2 conversion, as proposed in our previous report[8-10] and by other scientists[11-13], offers an approach to achieve simultaneous capture and utilization of CO2. This approach has the potential to facilitate the transition from multi-step to one-step CCUS technology, addressing the current challenge the CCUS technology faces.
Oxazolidinones are often used as the core structure of drugs because of their extensive biological activities. The use of CO2 to synthesize such compounds thus opens up an entirely new source of drugs, helping to reduce the dependence on petrochemicals in traditional pharmaceutical processes, while also contributing to environmental protection and the fight against climate change. However, researchers studying the in-situ catalytic conversion of CO2 into high-value organic chemicals are confronted with a significant challenge due to the strong chemical inertia of CO2 under normal conditions[14]. This is primarily because: (1) the majority of studies on the catalytic conversion of CO2 to high-value organic chemicals rely on the use of homogeneous catalysts for converting high-concentration CO2 and require the participation of alkali or cocatalyst[15-17]. However, these approaches are associated with difficulties in recycling, poor stability, and environmental pollution; (2) the low concentration of CO2 in flue gas further complicates the process of catalytic conversion for capturing into high-value organic chemicals[18,19]. Therefore, it is imperative and groundbreaking to achieve in-situ catalytic conversion of CO2 in waste gas without relying on homogeneous catalyst, or base and cocatalyst. This could be achieved through designing a specialized heterogeneous catalyst with a large specific surface area and unique active site[20,21].
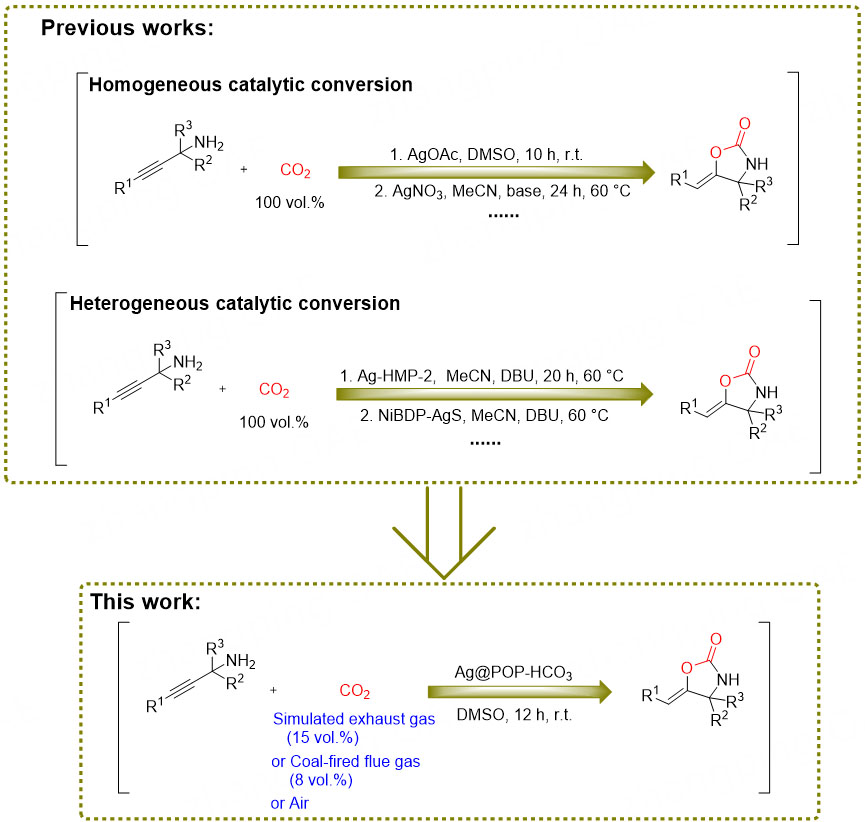
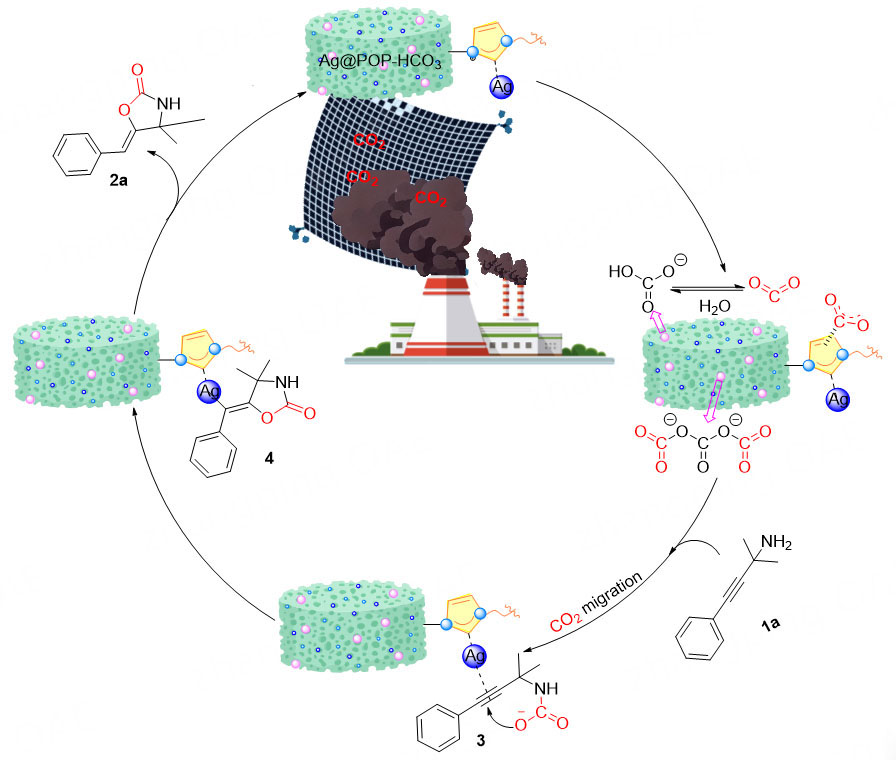
In recent years, the role of bicarbonate (HCO3-) in CO2 reduction has become increasingly important in the field of chemistry, biology and environmental science[22,23]. Therefore, incorporating it into heterogeneous catalysts may enhance the catalytic conversion ability of CO2 and is expected to replace the base and cocatalyst in the reaction system. Additionally, metal pre-coordination can be uniformly dispersed on the carrier surface to provide more active sites[24]. Our previous studies have shown that N-heterocyclic carbene organic porous polymers (POPs) exhibit superior ability for CO2 capture and catalytic conversion[8,25]. Therefore, in this study, silver metalized organic porous polymers (Ag@POP-HCO3) were designed by copolymerizing N-heterocyclic carbene monomer modified by HCO3- and pre-coordinated silver, with divinylbenzene (DVB). The polymer relies on the established abundant pores and HCO3- co-enrichment of CO2. Additionally, N-heterocyclic carbene and HCO3- accelerate the activation of CO2, while the coordination silver serves to activate the substrate. This allows for the in-situ conversion of CO2 in flue gas or air to oxazolidinone compounds through carboxylation cyclization reaction with no cocatalyst at room temperature and ambient pressure. The catalyst is unaffected by atmospheric pollutants (NO2, SO2), and exhibits good selectivity. This approach mitigates the cost and energy issues associated with traditional CCUS technology, thereby facilitating the development of environmentally friendly chemical processes. The comparison between this work and previous reports[26-29] is shown in Figure 1 and Supplementary Table 1.
EXPERIMENTAL
Materials and reagents
1,2-Dibromo-methyl benzene, n-vinylimidazole, DVB, azodiisobutyronitrile (AIBN), bis(triphenylphosphine)palladium(II) dichloride [Pd(PPh3)2Cl2], palladium(II) acetate [Pd(OAc)2], cuprous iodide (CuI), iodobenzene, 2-methyl-3-butyn-2-amine, etc. were purchased from Innochem in Beijing, China. Potassium carbonate (KHCO3) and silver nitrate (AgNO3) were procured from Xilong Science Co., LTD. Solvents such as toluene, ethyl acetate (EA), dimethyl sulfoxide (DMSO), tetrahydrofuran (THF) and methanol (MeOH), sulfuric acid (H2SO4) are purchased from local suppliers.
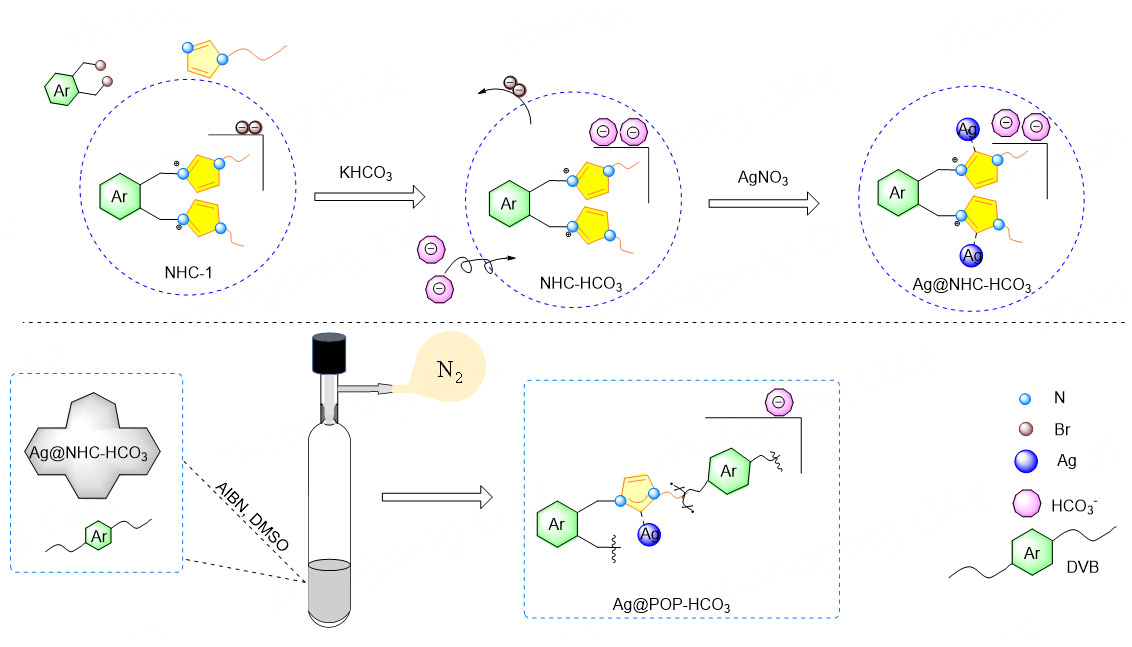
Synthetic method of Ag@POP-HCO3
The monomer (NHC-1) was synthesized by quaternary ammonium reaction of 20.0 mmol 1-vinylimidazole and 10.0 mmol halogenated aromatic hydrocarbons in 20 ml toluene at 90 °C for 24 h[24]. Secondly, NHC-HCO3 was synthesized by mixing 2 mmol NHC-1 and 4.2 mmol KHCO3 in MeOH at room temperature for 48 h to achieve anion conversion, stirring (400 rpm) at room temperature under shade for 12 h for metal pre-coordination synthesis Ag@NHC-HCO3. Finally, the polymer Ag@POP-HCO3 was obtained by polymerization in 3 mL DMSO under high pressure of 100 °C for 24 h with 0.10 mmol Ag@NHC-HCO3 as the polymerization unit, 2.0 mmol DVB as the crosslinker, and 33 mg AIBN as the initiator [Scheme 1].
Characterization methods
The functional groups and structures of the catalysts were investigated by various analytical techniques. These included 1H/13C nuclear magnetic resonance (NMR) spectroscopy (Bruker SBAVance III 400 M, Germany), solid-state 13C NMR (Bruker Avance Neo 400WB 400M, Germany) and Fourier infrared spectroscopy (FT-IR, Thermo Scientific Nicolet iS20, United States). Additionally, the elemental composition of the nanoparticles was further analyzed by X-ray photoelectron spectroscopy (XPS, Thermo Fisher Scientific K-Alpha, United States). The morphology and pore distribution of the catalyst were observed and determined by a scanning electron microscope (SEM, ZEISS sigma300, Germany), a transmission electron microscope (TEM, FEI Tecnai F20, United States), and a specific surface area and pore volume analyzer (BET, Kubo-X1000, China). Finally, the active center type of the catalyst was analyzed by in situ Raman (Horiba HR Evolution, Japan) and the thermal stability was studied by thermogravimetric analysis (TGA, NETZSCH STA2500, Germany).
The evaluating method of catalyst performance
The carboxylation cyclization reactions between propargyl amines and CO2 were employed to evaluate the performance of the catalysts proposed by this work [Scheme 2]. The brand of pharmaceutical reagent used in this system is listed in Supplementary Materials. In particular, an appropriate amount of catalyst was carefully weighed and added to a 10 mL branched glass reaction tube. Subsequently, 0.2 mmol of the reaction substrate and a specific quantity of solvent were introduced into the tube. The tube was then connected to CO2 atmosphere (0-100 vol.% CO2 /N2) provided by a balloon. Finally, the reaction tube containing the reaction mixture was then placed in a dark environment and subjected to magnetic stirring (400 rpm) for a certain time at a certain reaction temperature. After the reaction, the yield was determined using high performance liquid chromatography (HPLC Dionex UltiMate 3000) with biphenyl as the internal standard. The structure of the reaction products was elucidated by analyzing 1H/13C NMR spectra obtained using a Bruker AVANCE-III 400 spectrometer.
RESULTS AND DISCUSSION
Catalytic performance evaluation
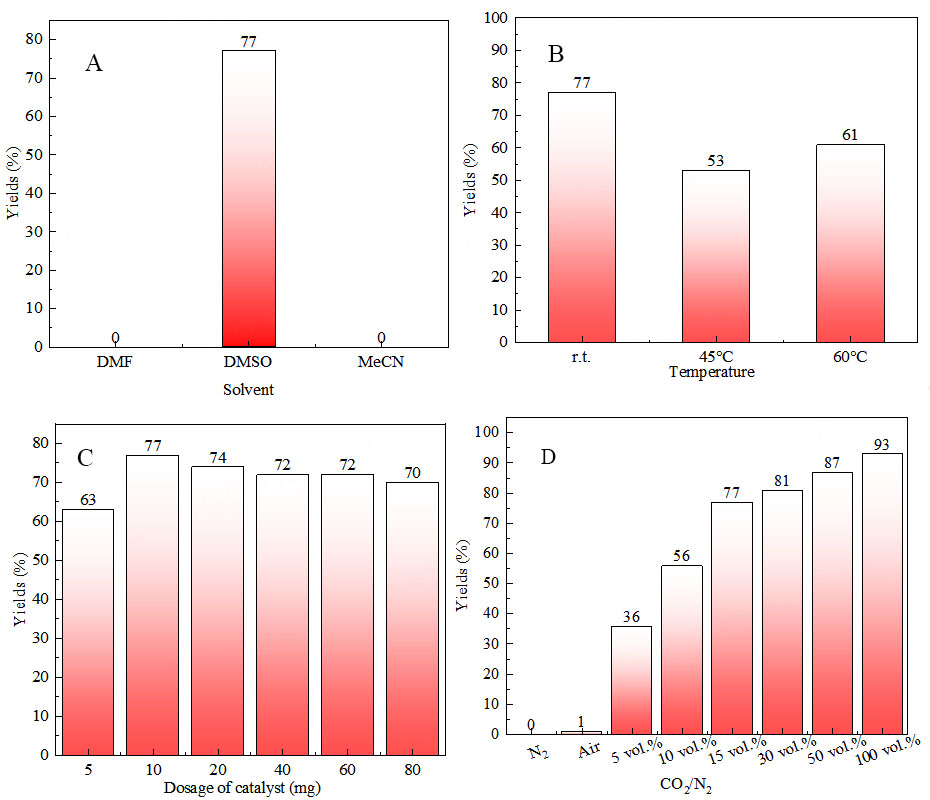
The polymer material Ag@POP-HCO3 was employed as a catalyst to catalyze the model carboxylation cyclization reaction between 2-methyl-4-phenylbut-3-yn-2-amine and CO2. The reaction conditions, including the choice of solvent, reaction temperature, catalyst dosage, CO2 concentration and reaction time, were optimized using a univariate method. The optimization conditions resulted in the following: when utilizing 15 vol.% CO2/N2 simulated gas as the carbon source and 0.2 mmol propargyl amines as the
Figure 2. Under the ambient pressure of low concentration CO2 and other different conditions, the catalyst reacted with 0.2 mmol propargyl amines for 12 h and the yields are as follows. (A) Solvent type (1 mL); (B) DMSO (1 mL), temperature; (C) DMSO (1 mL), dosage of catalyst at r.t.; (D) DMSO (1 mL), 10 mg catalyst catalyzes the conversion of different concentrations of CO2 at r.t. DMSO: Dimethyl sulfoxide.
The higher concentration of CO2 in the carbon source gas leads to a greater yield within the same reaction time (12 h) [Figure 2D]. With the increase of CO2 concentration to 50 vol.% CO2/N2 and 100 vol.% CO2/N2, the yield can reach 87% and 93%, respectively. When using air as the carbon source, only a 1% yield of oxazolidinone compounds was obtained. However, no oxazolidinone was produced in the absence of CO2, such as under a pure N2 atmosphere. These findings suggested that the carboxylation cyclization reaction between propargyl amines and CO2 was dependent on the nucleophilic addition and cyclization steps of CO2.
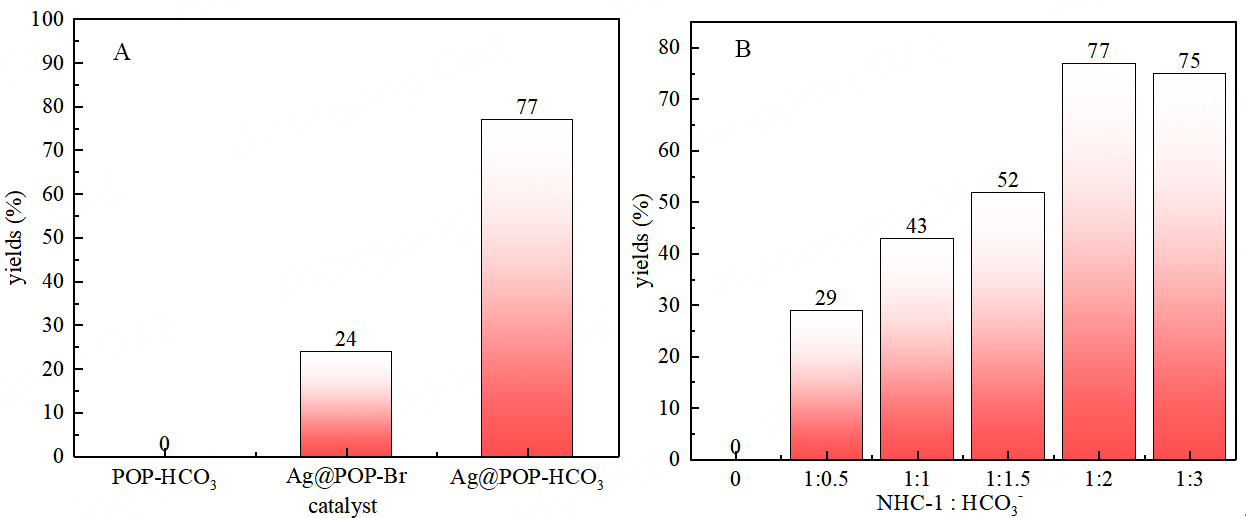
Under the optimal reaction conditions, POP-HCO3 and Ag@POP-Br (these synthesis methods are shown in Supplementary Schemes 1 and 2) were also utilized as catalysts to facilitate the conversion of low concentration CO2 through the model reaction, respectively. Both materials are components of the structure of Ag@POP-HCO3. The results showed that POP-HCO3 did not yield any target products, while Ag@POP-Br resulted in a relatively low yield of only 24% [Figure 3A]. However, Ag@POP-HCO3 outperformed both catalysts with a resulting yield of 77%. These findings indicate that both silver and HCO3- active sites in the polymer materials are essential for catalyzing the carboxylation cyclization reactions.
Figure 3. Under the environment pressure of 15 vol % CO2, DMSO (1 mL), the catalytic reaction yield of 10 mg catalyst with different structures stirred for 12 h are as follows. (A) Catalyst screening; (B) Influence of bicarbonate content. DMSO: Dimethyl sulfoxide.
To investigate the impact of HCO3- on the catalytic performance, different Ag@POP-HCO3 materials with varying HCO3- content were synthesized by adjusting the reaction ratios of NHC and KHCO3 to establish the anionic environment of NHC-1 in the Ag@POP-HCO3 synthesis process. The catalytic performance for the model reaction was then evaluated [Figure 3B]. When the reaction ratios of NHC-1 and KHCO3 ranged from 1:0 to 1:2, the yield increased up to 77% due to rising HCO3- content in the Ag@POP-HCO3 material. However, there was no significant increase in yield when changing the ratio of NHC and KHCO3 to 1:3. This is because the theoretical reaction ratio of NHC and KHCO3 is 1:2; increasing it to 1:3 did not raise the HCO3- content in the Ag@POP-HCO3. These findings further confirm that HCO3- is an effective active site in the Ag@POP-HCO3 catalyst.
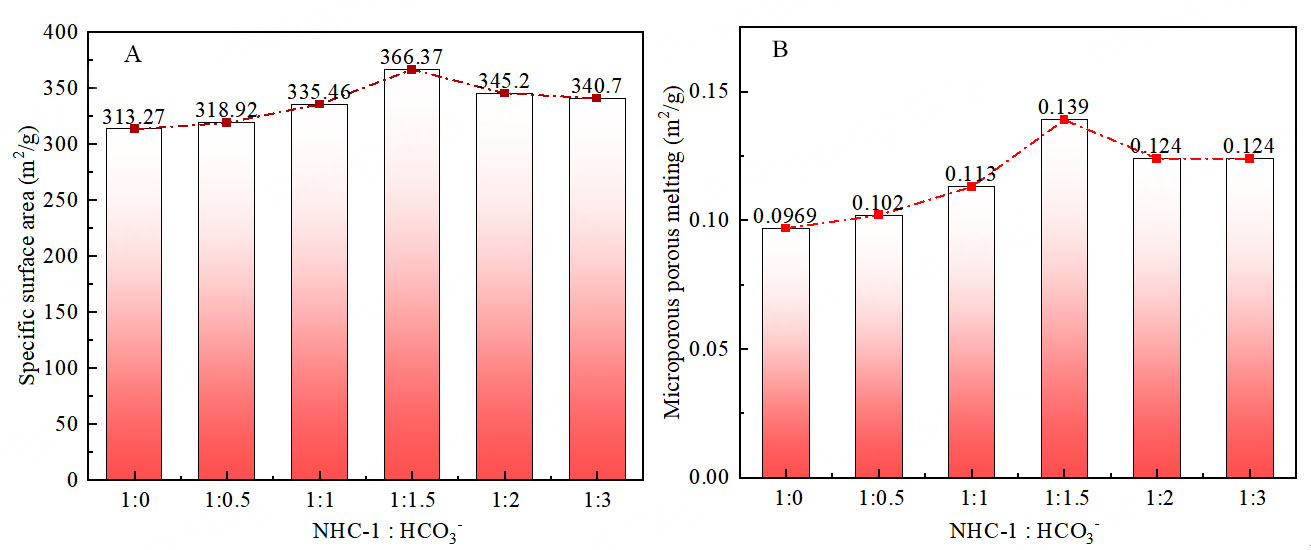
In order to further verify the experimental results, the specific surface area and microporous fusion of NHC-1 and HCO3- proportional polymers were determined by N2 adsorption and desorption curves [Figure 4, Supplementary Figures 1 and 2]. The higher the HCO3- content, the larger the specific surface area and microporous melting of the catalyst. When NHC-1: HCO3- = 1:1.5, the specific surface area and micropore porosity are the largest. The specific surface area is 366.37m2/g, and the micropore volume is 0.139 m3/g. However, due to the small number of active sites of HCO3- compared with the NHC-1: HCO3- = 1:2 polymer, the catalytic conversion rate of HCO3- to oxazolidinone was only 52%. When NHC-1: HCO3- = 1:3, the specific surface area and micropore volume of the catalyst remain basically unchanged. Furthermore, increasing the ratio of NHC-1 to HCO3- to 1:3 does not increase the HCO3- content in Ag@POP-HCO3. Based on the above experimental results, it is proved that HCO3- is an important influencing factor of the catalytic system.
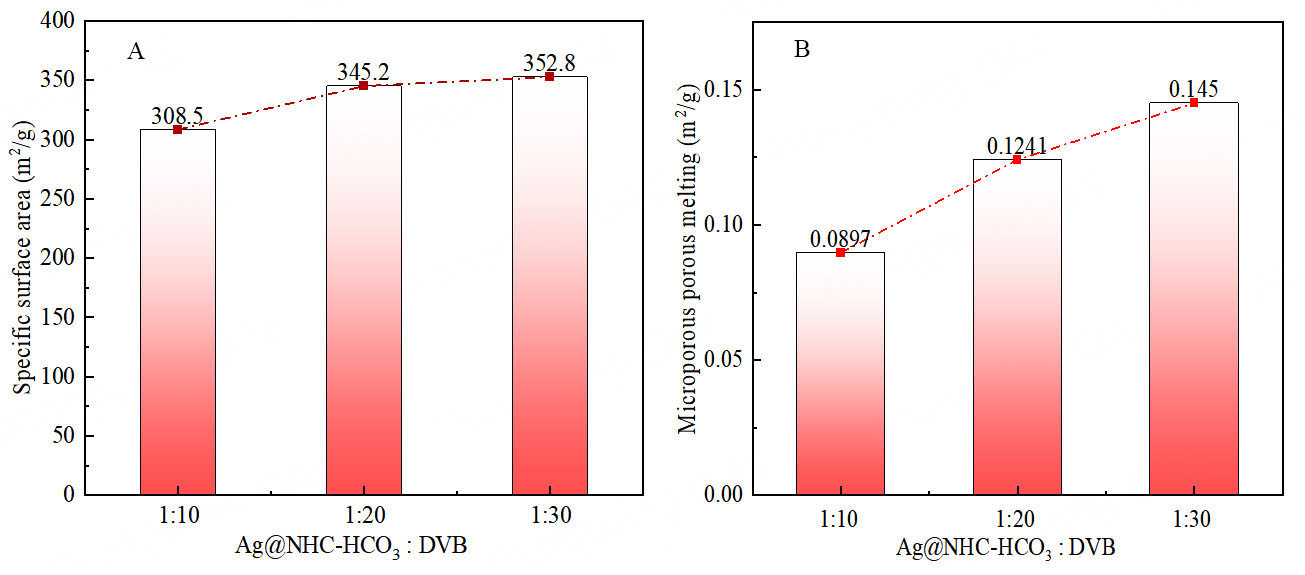
Figure 4. Specific surface area and microporous fusion of polymers with different NHC-1 and HCO3- ratios. (A) Specific surface area; (B) Microporous porous melting.
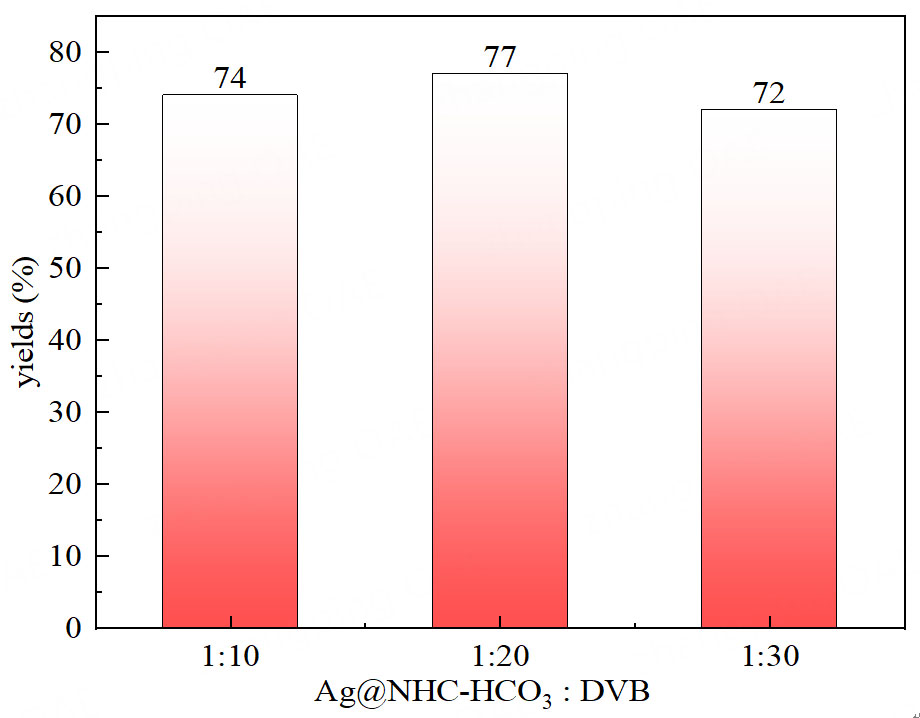
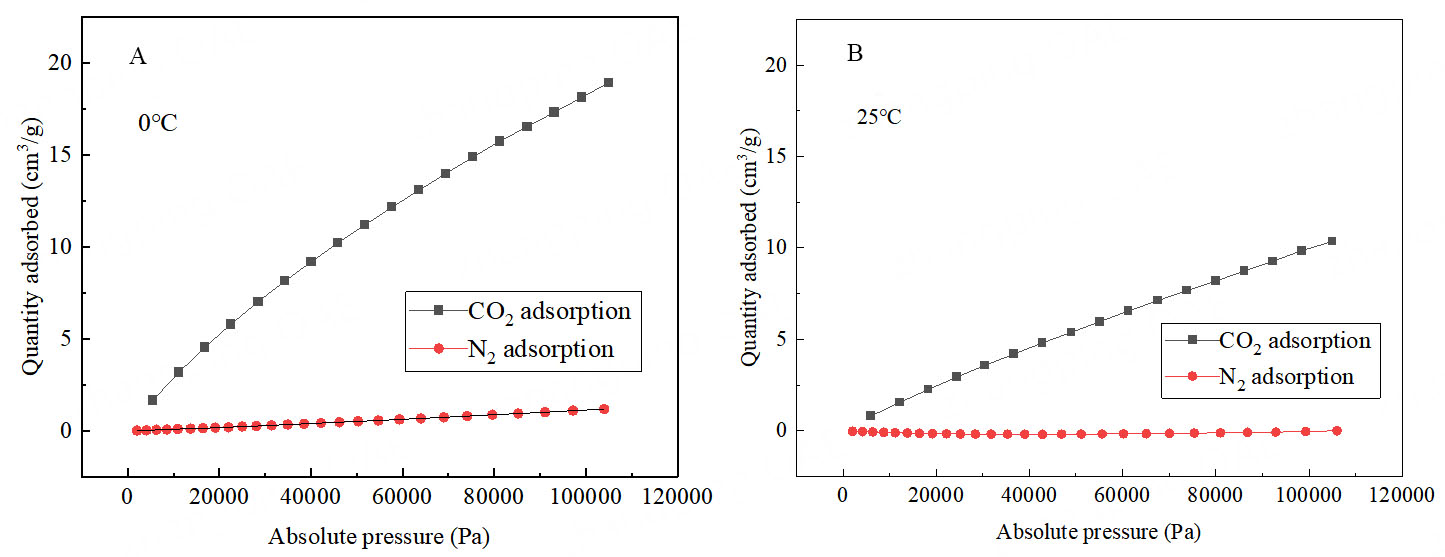
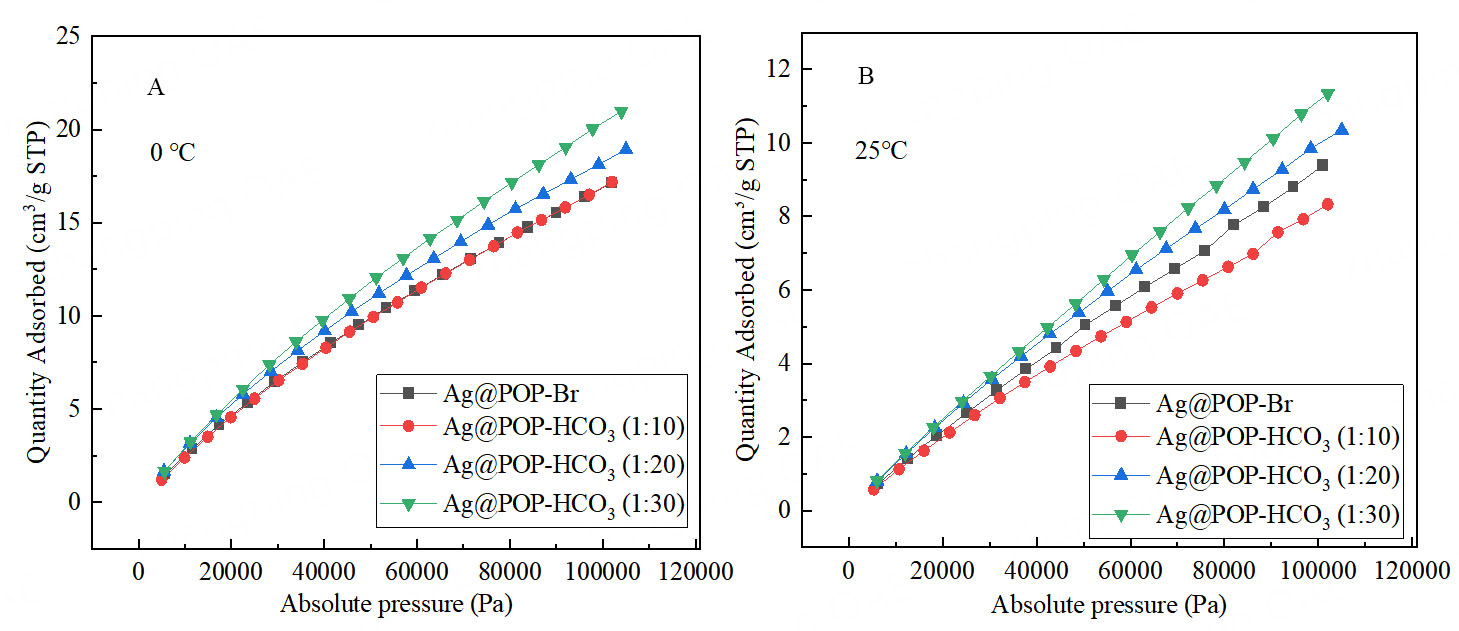
Increasing the usage of DVB in the polymerization process for the materials will increase both the specific surface area and micropore volume, potentially benefiting the catalytic performance[30,31]. However, it will decrease both the silver and HCO3- active sites, negatively affecting the catalytic performance. Therefore, an optimum polymerization ratio needs to be sought to balance both sides of the conflict. Experimental data revealed that the best catalytic conversion of low concentration CO2 to oxazolidinone occurs when Ag@NHC-HCO3: DVB = 1:20 [Figure 5]. According to Figure 6, Supplementary Figures 3 and 4, the presence of DVB in the polymer benefits the increase in specific surface area and micropore volume of the polymer. As shown in Figure 7, Ag@POP-HCO3 has significant selectivity for CO2 adsorption at both 0 °C and 25 °C. Comparison of the CO2 adsorption isotherms of Ag@POP-Br and Ag@POP-HCO3 (1:20) showed that introduction of HCO3- anions into the catalyst led to enhancement of CO2 adsorption capacity. Additionally, there was a positive correlation between adsorption performance for CO2 and the increase in DVB usage [Figure 8]. Although the maximum CO2 capacity belonged to the polymer material Ag@POP-HCO3 (1:30), optimal catalytic performance occurred with a polymerization ratio of 1:20 for Ag@POP-HCO3, indicating that an ideal equilibrium state was achieved in terms of distribution of active center, pore structure and adsorption capacity of the catalyst when using a ratio of 1:20.
Figure 6. Specific surface area and microporous fusion of polymers with different NHC-1 and HCO3- ratios. (A) Specific surface area; (B) Microporous porous melting.
Figure 7. (A) Comparison of adsorption isotherms of carbon dioxide and N2 at 0 °C of Ag@POP-HCO3; (B) Comparison of adsorption isotherms of carbon dioxide and N2 at 25 °C of Ag@POP-HCO3.
Mechanistic considerations
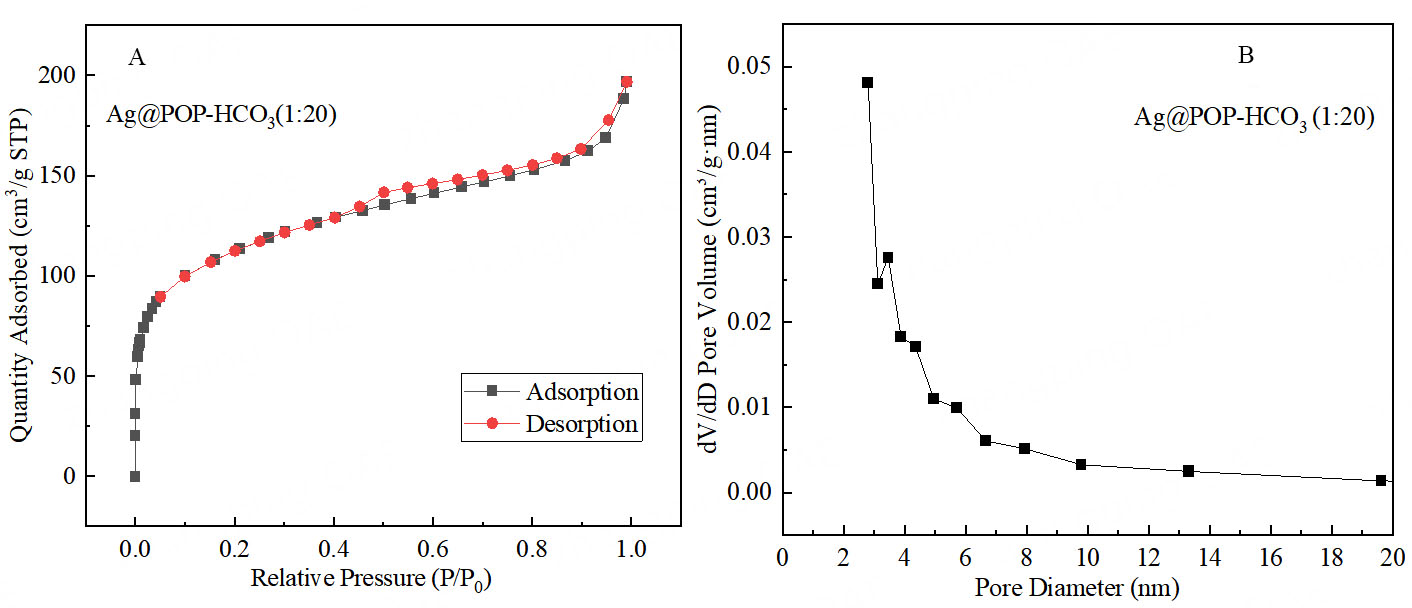
Following the successful construction and optimization of the internal structure, the micro-structure of the optimized catalyst Ag@POP-HCO3 (1:20) was characterized and verified. The specific surface area of Ag@POP-HCO3 (1:20) material is 345.2 m2/g and the micropore volume is 0.1241 cm3/g. The adsorption and desorption curve of Ag@POP-HCO3 exhibits type IV isotherm features [Figure 9]. The analysis of the pore size distribution curve indicated a pore size of approximately 3 nm for the polymer material, demonstrating characteristics of mesoporous materials.
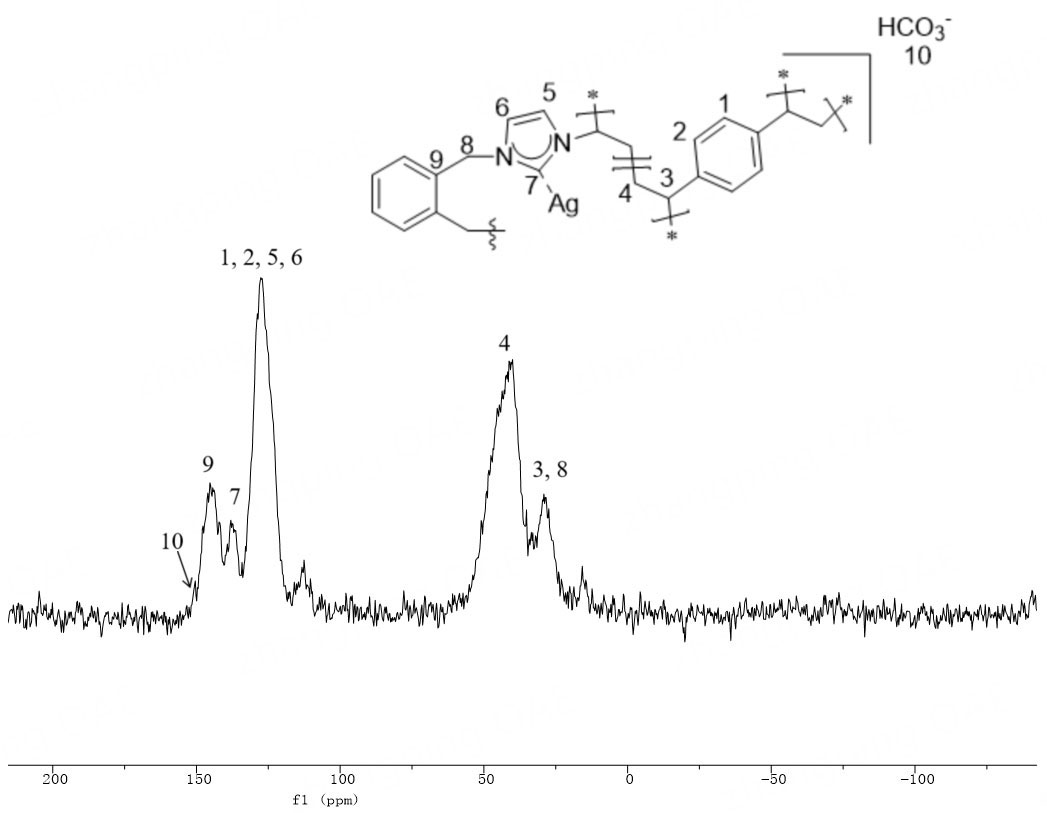
The 13C NMR spectrum of NHC-HCO3 revealed an increase in HCO3- peaks, indicating the successful replacement of original Br- in NHC-1 by HCO3- through the reaction of NHC-1 with KHCO3. The solid-state 13C NMR spectrum of Ag@POP-HCO3 [Figure 10] shows vibration peaks at 29 ppm caused by methylene units (C3, C8) attached to the benzene ring, wide overlapping peaks at 40-60 ppm attributed to polyethylene (C4) vibration, vibration peaks at 128 ppm, 138 ppm, and 145 ppm associated with C signals from imidazole and aromatic rings, and a spike at 150 ppm due to HCO3-[32].
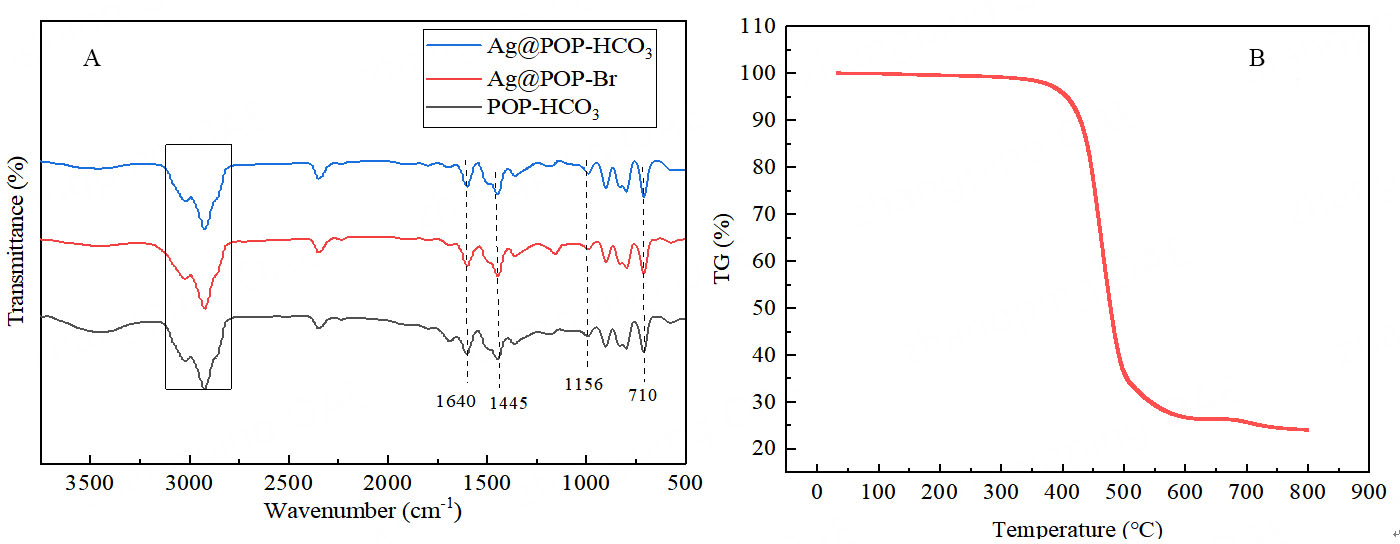
The FT-IR spectra of Ag@POP-HCO3, Ag@POP-Br and POP-HCO3 polymers are exhibited in Figure 11A. The peaks observed at 3,100-2,850 cm-1 can be attributed to the stretching vibrations of aliphatic C-H bonds, while a bending vibration peak of around 710 cm-1 is caused by aromatic C-H. These findings suggest DVB unit presence in the structure of the polymers[33,34]. Peaks observed at 1,600 cm and 1,156 cm are separately induced by the vibration of the aromatic ring and imidazole ring skeleton (C-C)[35], while peaks at 1,640 cm and 1,445 cm arise from C=N and C-N bonds[36,37], indicating that imidazole framework of Ag@POP-HCO3 remains undamaged after KHCO3 modification and silver loading.
Figure 11. (A) FT-IR spectra of Ag@POP-HCO3, Ag@POP-Br and POP-HCO3; (B) Thermogravimetric analysis diagram of Ag@POP-HCO3.
TGA [Figure 11B] in N2 atmosphere shows that the catalyst Ag@POP-HCO3 has excellent thermal stability. It did not exhibit significant mass loss until it reached a temperature above 400. However, a rapid increase of mass loss was observed between 400 °C and 500 °C, indicating substantial polymer decomposition. Further mass loss above 500 °C was attributed to imidazole ring decomposition[8,9]. However, as the temperature increases, the polymer will undergo carbonization, eventually resulting in a non-volatile carbon skeleton and metal residues.
The SEM image reveals that the polymer Ag@POP-HCO3 is formed through the accumulation and superposition of particles, resulting in irregular pore structures conducive to the enrichment of CO2 in waste gas [Figure 12A]. TEM images demonstrate that the silver particles are uniformly dispersed within the polymer [Figure 12B and Supplementary Figure 5]. This can be attributed to the benefits of the method of metal pre-coordination, where the metal coordinated NHC-HCO3 to form a metal complex monomer (Ag@NHC-HCO3), and then copolymerizes with the crosslinking agent, ensuring the dispersion of metallic silver and preventing cluster phenomena[10].
Figure 12. (A) 100 nm SEM image of catalyst before cycling; (B) TEM image of catalyst before cycling. SEM: Scanning electron microscope; TEM: transmission electron microscope.
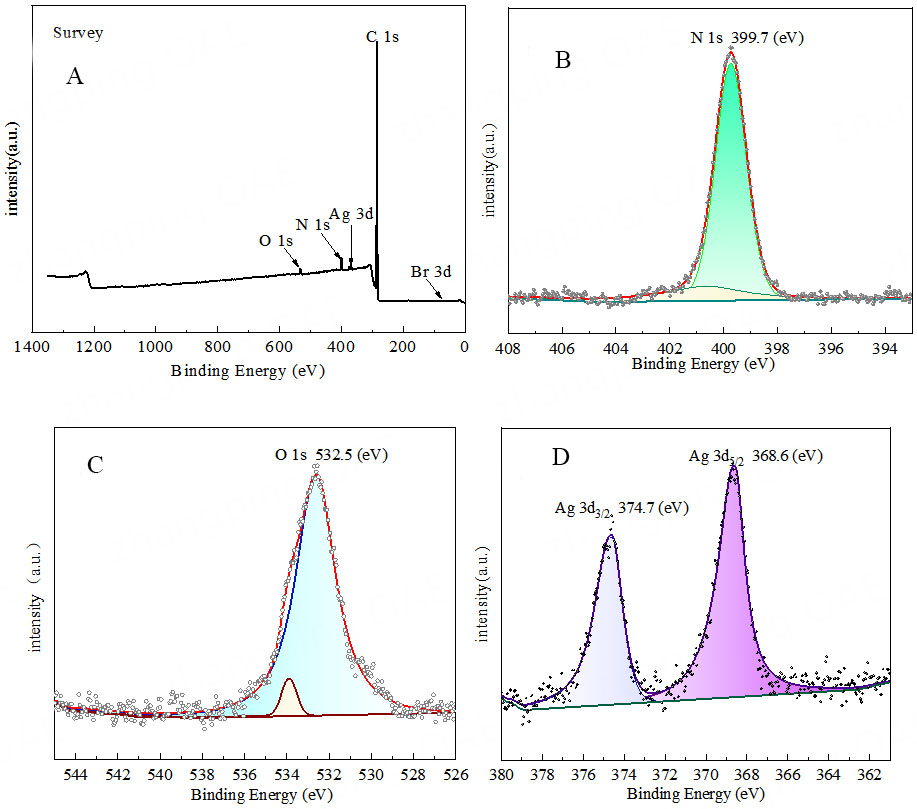
The XPS diagram [Figure 13 and Supplementary Figure 6] clearly indicates the binding energy of N 1s at
Figure 13. High resolution XPS spectrum. (A) Ag@POP-HCO3; (B) N 1s; (C) O 1s; (D) Ag 3d3/2, Ag 3d5/2. XPS: X-ray photoelectron spectroscopy.
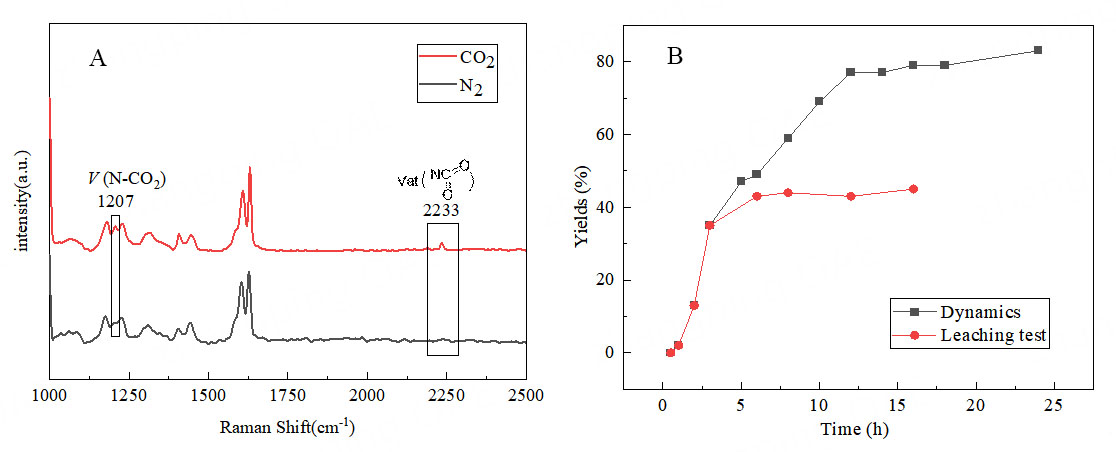
In situ Raman spectra of Ag@POP-HCO3 under CO2 and N2 atmosphere were depicted in Figure 14A. Peaks at 1,207 cm-1 and 2,233 cm-1 are clearly observed in the spectrum of Ag@POP-HCO3 under CO2, but not under N2 atmosphere. The peak at 1,207 cm-1 corresponds to the tensile vibration of CO2 adsorbed on N, while the peak at 2,233 cm-1 is derived from the antisymmetric stretching of the adsorbed CO2 on the N-heterocyclic carbene[42]. These findings demonstrate that N-heterocyclic carbene in Ag@POP-HCO3 can effectively activate CO2. In addition, elemental analysis and atomic absorption were performed on the catalyst, and the content of N and silver was measured to be 0.9% and 0.046 wt%, respectively [Supplementary Table 2].
Figure 14. (A) In situ Raman spectra of Ag@POP-HCO3 under CO2 and N2 atmosphere; (B) Reaction kinetics and leaching experiments.
The reaction kinetics were investigated using a 15 vol.% CO2/N2 gas mixture as the carbon source [Figure 14B]. It was observed that after 12 h, the yield almost reached a plateau. Further extending the reaction time to 24 h resulted in only a 6% increase in yield. Additionally, when the catalyst was filtered out at 3 h and no longer participated in the subsequent CO2 reaction, the reaction almost stopped. This confirms that this catalytic system operates through a heterophase catalytic mechanism.
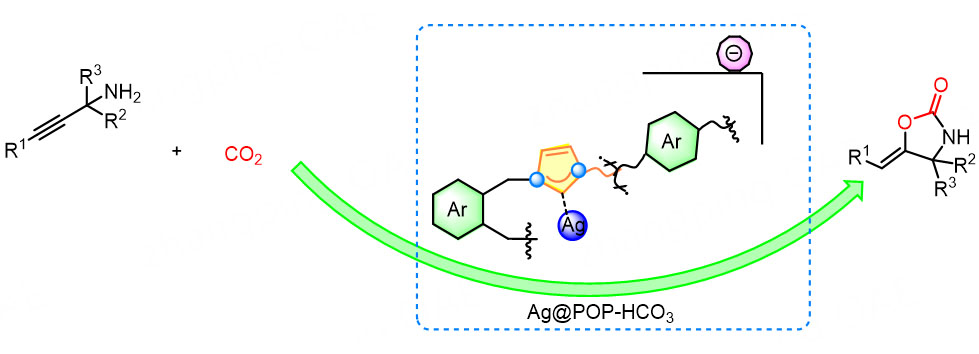
Based on aforementioned experimental results, we propose a possible mechanism for Ag@POP-HCO3 catalyzing the carboxylation cyclization of propargyl amines with low concentration CO2 [Scheme 3]. Firstly, CO2 is co-enriched by the pores and HCO3- anions of Ag@POP-HCO3 catalyst. During the enrichment process, CO2 is initially adsorbed by the pores and then captured by HCO3- anions. The solvent DMSO exhibits strong water absorption, which allows for species transformation of HCO3- and CO2, thereby enhancing the capability of the catalyst Ag@POP-HCO3 to enrich CO2. Subsequently, the enriched CO2 is activated by carbene N. The Ag in the catalyst interacts with the C≡C bond of propargyl amine through empty orbitals and π electrons to active propargyl amine. The activated CO2 and propargyl amines are converted into carbamate intermediates 3. Carbamate intermediate 3 is converted to vinyl-silver intermediate 4 by intramolecular cyclization. Finally, oxazolidinone 2a was produced by in-situ demetallization, and the catalyst Ag@POP-HCO3 was regenerated.
Application research of catalyst
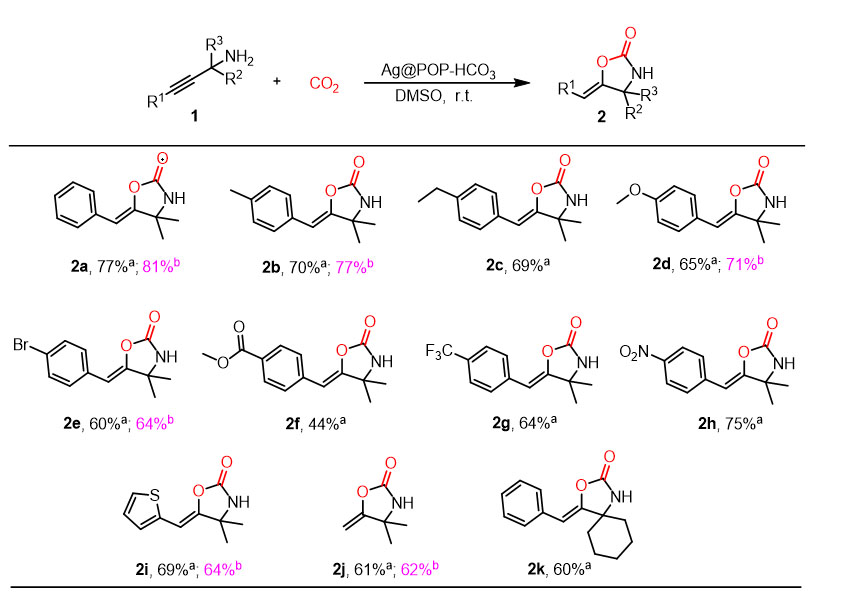
The carboxylation cyclization reactions were conducted under optimized reaction conditions using 15 vol.% CO2/N2 as a carbon source and various substituents of propargyl amines (For the synthesis procedure, see Supplementary Schemes 3 and 4)[27,43] as substrate [Figure 15]. In addition to the model substrate (1a), benzene substituents, including electron-donating substituents (1b-1d), electron-withdrawing groups (1f-1h), all successfully participated in the carboxylation cyclization reaction in considerable yields. Additionally, successful completion of carboxylative cyclization reaction was also observed when the propargyl amines skeleton contained a heterocyclic structure (1i) or a terminal alkyne structure (1j). The study also investigated changes in substituents at R2 and R3 positions (1k), resulting in a 60% yield.
Figure 15. Suitability studies of substrates. Reaction conditions: (a) Propargyl amines 1 (0.2 mmol), catalyst (10 mg), 15 vol.% CO2/N2, DMSO (1 mL), 12 h, r.t.; (b) Propargyl amines 1 (0.2 mmol), catalyst (10 mg), coal-fired flue gas, DMSO (1 mL), 36 h, r.t.
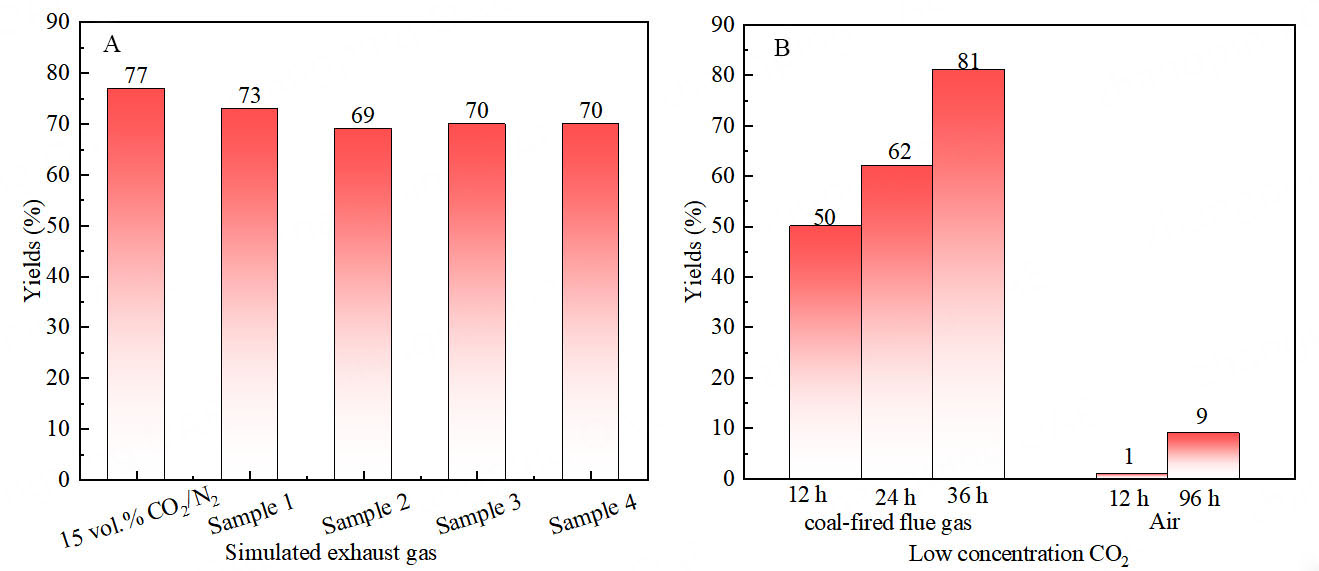
In order to access the potential applications of the Ag@POP-HCO3 catalytic system in actual flue gases, the impact of co-existing SO2 and NO2 on the model reaction was investigated using simulated flue gases as a carbon source [Table 1]. The results indicated that, with both SO2 content of up to 428 mg/m3 and NO2 content of up to 221 mg/m3, there was no significant negative impact on the reaction, which maintained a relatively stable yield [Figure 16A]. It is worth noting that SO2 and NO2 content in actual flue gases are usually less than 400 mg/m³ and 200 mg/m³, respectively. Therefore, it can be concluded that the catalytic system proposed in this work is also available for actual flue gases.
Figure 16. (A) The influence of SO2 and NO2 contents on the reaction; (B) Ag@POP-HCO3 for fixing CO2 in coal-fired flue gas and air.
The components of simulated exhaust gases
| Simulated exhaust gas | Content of components |
| Sample 1 | 15% CO2, 71 mg/m3 SO2, 51 mg/m3 NO2, 1.0% O2, 0.05% CO, N2 balance |
| Sample 2 | 15% CO2, 134 mg/m3 SO2, 107 mg/m3 NO2, 1.0% O2, 0.05% CO, N2 balance |
| Sample 3 | 15% CO2, 294 mg/m3 SO2, 221 mg/m3 NO2, 1.0% O2, 0.05% CO, N2 balance |
| Sample 4 | 15% CO2, 428 mg/m3 SO2, 1.0% O2, 0.05% CO, N2 balance |
In practical application, a coal-fired flue gas (containing 8 vol.% CO2) collected from a coal-fired power plant in Guilin was treated with Ag@POP-HCO3 catalyst. This resulted in the successful conversion of CO2 to oxazolidinone (2a), with a remarkable yield of 50%. When reaction time was extended to 24 h and 36 h, the yield reached 62% and 81%, respectively [Figure 16B]. When benzene substituents with electron-donating substituents (1b, 1d), halogen substituents (1e) and propargyl amine scaffolded with the heterocyclic structure (1i) or a terminal alkyne structure (1j), they could react with coal-fired flue gas, and form the corresponding oxazolidinones in 62%-77% yields [Figure 15]. Furthermore, the catalyst was used to catalyze the carboxylation cyclization reaction of propargyl amines with CO2 in real air. Initially, the yield was only 1% during the 12 h reaction time; however, after extending the reaction time to 96 h, the yield increased to 9% [Figure 16B]. This experimental data strongly demonstrate that Ag@POP-HCO3 catalyst exhibits high selectivity and catalytic activity for CO2, even in actual flue gas environments containing pollutants such as SO2 and NO2.
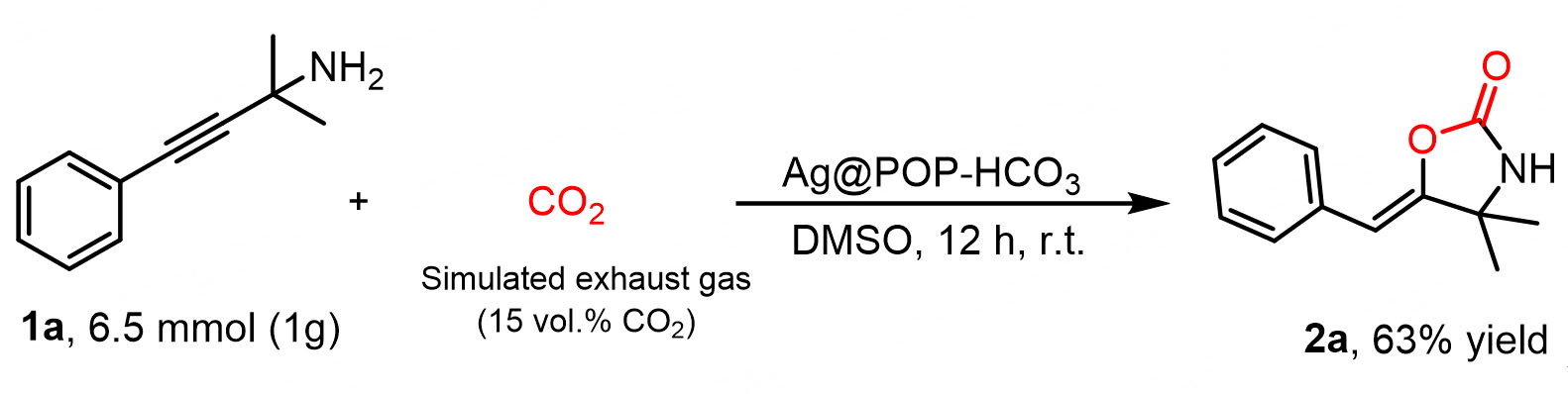
The model reaction of carboxylation cyclization was conducted on a gram scale [Scheme 4] with a yield of 63% within 12 h reaction time. The Ag@POP-HCO3 catalyst recovered from the previous reaction was utilized to react propargyl amines with CO2 in simulated flue gas under optimal reaction conditions. It was observed that the catalyst could be recycled [Supplementary Figure 7]. These findings further provide strong evidence for the practicality of the Ag@POP-HCO3 catalyst.
CONCLUSION
In summary, we have successfully synthesized an organic porous polymer material named Ag@POP-HCO3 for catalyzing the in-situ conversion of low concentration CO2 from coal-fired flue gas or air into oxazolidinone compounds through carboxylation cyclization reaction under ambient conditions. The main results obtained are as follows:
This process does not require the addition of any cocatalyst. The abundant pore structure and large specific surface area of the material provide a basis for achieving excellent adsorption capacity of CO2.
The introduction of HCO3- to N-heterocyclic carbene further enhances the adsorption capacity. Silver loaded by a pre-coordinated method ensures that silver particles are uniformly dispersed within the polymer. Furthermore, the catalyst exhibits excellent thermal stability below 400 °C.
Ag@POP-HCO3 catalyst exhibits exceptional performance in the carboxylation cyclization of low concentration with various propargyl amines. The catalyst demonstrates broad substrate adaptability and is effective for actual flue gas, unaffected by co-existing both SO2 content of up to 428 mg/m3 and NO2 content of up to 221 mg/m3. It is exciting that the catalyst effectively converts low concentrations of CO2 in coal-fired flue gas and real air to oxazolidinone.
The proposed catalytic approach has the potential to be scaled up and developed into a one-step CCUS technology, which can effectively address the cost issues associated with traditional CCUS technology. This advancement also promotes the development of green chemical processes.
DECLARATIONS
Authors’ contributions
Conceptualization, methodology, supervision, funding acquisition, writing - review and editing: Liang, Y.
Conceptualization, investigation, data curation, writing - original draft: Yang, J.
Investigation, data curation: Chen, W.
Conceptualization, methodology: Chen, P.
Conceptualization, discussion: Fang, P.
Conceptualization, supervision, funding acquisition, writing - review and editing: Pan, Y.
Availability of data and materials
The experimental data and associated test results are published as Supplementary Materials in the journal.
Financial support and sponsorship
We thank the financial support from the National Natural Science Foundation of China (Nos. 52170107, 52360014, 41965006), Guangxi Natural Science Foundation of China (No.2024GXNSFDA010013) and the Innovation Project of GUET Graduate Education (2023YCXS182).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Shakoor, A.; Ashraf, F.; Shakoor, S.; Mustafa, A.; Rehman, A.; Altaf, M. M. Biogeochemical transformation of greenhouse gas emissions from terrestrial to atmospheric environment and potential feedback to climate forcing. Environ. Sci. Pollut. Res. 2020, 27, 38513-36.
2. Nocito, F.; Dibenedetto, A. Atmospheric CO2 mitigation technologies: carbon capture utilization and storage. Curr. Opin. Green. Sustain. Chem. 2020, 21, 34-43.
3. Gadikota, G.; Gaffney, A. M.; Santos, R. M.; Duan, L. Editorial: emerging technologies and associated scientific advancements for CCUS deployment. Front. Energy. Res. 2021, 9, 649126.
4. Chatterjee, S.; Parsapur, R. K.; Huang, K. Limitations of ammonia as a hydrogen energy carrier for the transportation sector. ACS. Energy. Lett. 2021, 6, 4390-4.
5. Xiong, T.; Zhou, X.; Zhang, M.; Tang, H.; Pan, Y.; Liang, Y. Electrochemical-mediated fixation of CO 2 : three-component synthesis of carbamate compounds from CO 2 , amines and N -alkenylsulfonamides. Green. Chem. 2021, 23, 4328-32.
6. Zhou, X.; Tang, H.; Cui, F.; Liang, Y.; Li, S.; Pan, Y. Electrocatalytic three-component reactions: synthesis of tellurium-containing oxazolidinone for anticancer agents. Green. Chem. 2023, 25, 5024-9.
7. Wang, P.; Shi, B.; Li, N.; et al. CCUS development in China and forecast its contribution to emission reduction. Sci. Rep. 2023, 13, 17811.
8. Rao, Z. X.; Chen, P. B.; Xu, J.; et al. Direct conversion of CO2 in lime kiln waste gas catalyzed by a copper-based N-heterocyclic carbene porous polymer. ChemSusChem 2023, 16, e202300170.
9. Liang, Y.; Wang, Q.; Shen, X. X.; et al. A six-membered N-heterocyclic polyionic liquids with palladium nanoparticles as a heterogeneous catalyst for the multicomponent one-pot reaction of carbon dioxide. J. Colloid. Interface. Sci. 2025, 678, 754-65.
10. Chen, P. B.; Yang, J. W.; Rao, Z. X.; et al. Efficient in-situ conversion of low-concentration carbon dioxide in exhaust gas using silver nanoparticles in N-heterocyclic carbene polymer. J. Colloid. Interface. Sci. 2023, 652, 866-77.
11. Wen, J.; Chen, S.; Liu, Y.; et al. Triphenylimidazolium-incorporated, benzbisimidazole-linked porous organic polymers as efficient catalyst for CO2 conversion. Micropor. Mesopor. Mater. 2022, 339, 111999.
12. Du, C.; Lan, X.; An, G.; Li, Q.; Bai, G. Direct surface modification of graphitic C3N 4 with porous organic polymer and silver nanoparticles for promoting CO2 conversion. ACS. Sustainable. Chem. Eng. 2020, 8, 7051-8.
13. Zhang, X.; Chen, K.; Zhou, Z.; He, L. Reduced graphene oxide supported ag nanoparticles: an efficient catalyst for CO2 conversion at ambient conditions. ChemCatChem 2020, 12, 4825-30.
14. Hong, J.; Li, M.; Zhang, J.; Sun, B.; Mo, F. C-H bond carboxylation with carbon dioxide. ChemSusChem 2019, 12, 6-39.
15. Chen, X. C.; Yao, Y. Q.; Zhao, K. C.; Liu, L.; Lu, Y.; Liu, Y. Cooperative catalysis of Ru(III)-porphyrin in CO2 -involved synthesis of oxazolidinones. Chem. Asian. J. 2021, 16, 2504-10.
16. Mei, C.; Zhao, Y.; Chen, Q.; Cao, C.; Pang, G.; Shi, Y. Synthesis of oxazolidinones and derivatives through three-component fixation of carbon dioxide. ChemCatChem 2018, 10, 3057-68.
17. Wu, J.; Ma, S.; Cui, J.; Yang, Z.; Zhang, J. Nitrogen-rich porous organic polymers with supported ag nanoparticles for efficient CO2 conversion. Nanomaterials 2022, 12, 3088.
18. Kumar, A.; Sharma, P.; Sharma, N.; Kumar, Y.; Mahajan, D. Catalyst free N-formylation of aromatic and aliphatic amines exploiting reductive formylation of CO2 using NaBH4. RSC. Adv. 2021, 11, 25777-87.
19. Zhang, Q.; Shi, P.; Zeng, A. Carboxylate-assisted carboxylation of thiophene with CO2 in the solvent-free carbonate medium. Catalysts 2022, 12, 369.
20. Tang, X.; Liu, J.; Chen, P.; et al. Effective N-formylation of amines with CO2 in anaerobic fermentation gas catalyzed by triply synergistic effect of ionic porous organic polymer. ChemCatChem 2023, 15, e202201351.
21. Liang, Y.; Xia, Q.; Yang, J.; Meng, X.; Hu, X.; Pan, Y. Efficient in-situ conversion of low-concentration carbon dioxide in coal-fired flue gas using silver nanoparticles in amino-functionalism poly-ionic liquids. Chem. Eng. J. 2024, 498, 155305.
22. Lux, S.; Baldauf-Sommerbauer, G.; Siebenhofer, M. Hydrogenation of inorganic metal carbonates: a review on its potential for carbon dioxide utilization and emission reduction. ChemSusChem 2018, 11, 3357-75.
23. Chen, P.; Tang, X.; Meng, X.; Tang, H.; Pan, Y.; Liang, Y. Transition metal-free catalytic formylation of carbon dioxide and amide with novel poly(ionic liquid)s. Green. Synth. Catal. 2022, 3, 162-7.
24. Li, W. H.; Li, C. Y.; Xiong, H. Y.; et al. Constructing mononuclear palladium catalysts by precoordination/solvothermal polymerization: recyclable catalyst for regioselective oxidative heck reactions. Angew. Chem. Int. Ed. 2019, 58, 2448-53.
25. Chen, P.; Xiong, T.; Pan, Y.; Liang, Y. Recent progress on N-heterocyclic Carbene catalysts in chemical fixation of CO2. Asian. J. Org. Chem. 2022, 11, e202100738.
26. Yoshida, S.; Fukui, K.; Kikuchi, S.; Yamada, T. Silver-catalyzed preparation of oxazolidinones from carbon dioxide and propargylic amines. Chemistry. Letters. 2009, 38, 786-7.
27. Ishida, T.; Kobayashi, R.; Yamada, T. Novel method of tetramic acid synthesis: silver-catalyzed carbon dioxide incorporation into propargylic amine and intramolecular rearrangement. Org. Lett. 2014, 16, 2430-3.
28. Ghosh, S.; Molla, R. A.; Kayal, U.; Bhaumik, A.; Islam, S. M. Ag NPs decorated on a COF in the presence of DBU as an efficient catalytic system for the synthesis of tetramic acids via CO2 fixation into propargylic amines at atmospheric pressure. Dalton. Trans. 2019, 48, 4657-66.
29. Yang, H.; Zhang, X.; Zhang, G.; Fei, H. An alkaline-resistant Ag(I)-anchored pyrazolate-based metal-organic framework for chemical fixation of CO2. . Chem. Commun. 2018, 54, 4469-72.
30. Wei, Z. Q.; Wang, Z.; Hong, R. Y.; Wang, Y. F. Monodisperse plum-like sulfonated PGMA-DVB microspheres as a new ion exchange resin. J. of. Applied. Polymer. Sci. 2017, 134, app.44994.
31. Cong, H.; Xing, J.; Ding, X.; Zhang, S.; Shen, Y.; Yu, B. Preparation of porous sulfonated poly(styrene-divinylbenzene) microspheres and its application in hydrophilic and chiral separation. Talanta 2020, 210, 120586.
32. Zhao, T.; Long, G.; Liang, H.; Xiong, W.; Hu, X. One-pot synthesis of cyclic carbonates from olefins and CO2 catalyzed by silica-supported imidazolium hydrogen carbonate ionic liquids. Microporous. Mesoporous. Mater. 2023, 356, 112576.
33. Yang, W.; Li, H.; Wu, Q.; et al. Functionalized core–shell polystyrene sphere-supported alkaline imidazolium ionic liquid: an efficient and recyclable catalyst for knoevenagel condensation. ACS. Sustainable. Chem. Eng. 2020, 8, 18126-37.
34. Fu, J.; He, Z.; Hu, X.; et al. Synthesis of amphiphilic AIE fluorescent nanoparticles via CO2 involved multicomponent reaction and its biological imaging potential. Dyes. and. Pigments. 2023, 210, 110990.
35. Li, D.; Li, J.; Mao, D.; Wen, H.; Zhou, Y.; Wang, J. Direct synthesis of sulfonic group tethered mesoporous poly(ionic liquid) for catalyzing deoximation reactions. Mater. Chem. Phys. 2017, 189, 118-26.
36. An, R.; Chen, S.; Zhang, R.; et al. Synthesis of propylene glycol methyl ether catalyzed by imidazole polymer catalyst: performance evaluation, kinetics study, and process simulation. . Chem. Eng. J. 2021, 405, 126636.
37. Zhang, W.; Ma, F.; Ma, L.; Zhou, Y.; Wang, J. Imidazolium-functionalized ionic hypercrosslinked porous polymers for efficient synthesis of cyclic carbonates from simulated flue gas. ChemSusChem 2020, 13, 341-50.
38. Wang, X.; Zhang, L.; Guo, Z.; Shi, Y.; Zhou, Y.; Wang, J. Synergistic catalysis of one-pot cascade reactions by acidic and basic binary porous polymers. Appl. Surf. Sci. 2019, 478, 221-9.
39. Ramos, G. J. A.; Sanches, E. A.; Bruns, F.; et al. Vacancy ordered γ-Fe2O3 nanoparticles functionalized with nanohydroxyapatite: XRD, FTIR, TEM, XPS and mössbauer studies. Appl. Surf. Sci. 2016, 389, 721-34.
40. Bai, X.; Zhang, Y.; Yan, F.; Lan, X. Thiadiazol-based conjugated organic polymer anchoring ag nanoparticles for efficient conversion of CO2 into oxazolidinones from propargylic amines. Appl. Surf. Sci. 2022, 604, 154566.
41. Wang, X.; Li, W.; Wang, J.; et al. A dual-functional urea-linked conjugated porous polymer anchoring silver nanoparticles for highly efficient CO2 conversion under mild conditions. Dalton. Trans. 2020, 49, 13052-9.
42. Li, Z.; Yang, Y.; Yin, Z.; et al. Interface-enhanced catalytic selectivity on the C2 Products of CO2 electroreduction. ACS. Catal. 2021, 11, 2473-82.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].