Toward CO2-neutral biodiesel production: industrial challenges and advances in CaO-based catalysts
Abstract
Biodiesel is recognized as a promising and sustainable alternative to petroleum-derived diesel fuel. The use of biodiesel as fuel can be considered as a closed carbon cycle. In fact, CO2 emission under combustion is balanced by the fixed CO2 consumed by growing biomass. Furthermore, biodiesel combustion generates lower emissions of sulfur oxides and solid particles compared with hydrocarbon-derived diesel, reinforcing the environmental advantages. A wide range of renewable feedstocks, including vegetable oils, waste cooking oils, and animal fats, can be utilized for biodiesel synthesis; however, the efficiency of the process strongly depends on the design of catalytic system applied. Homogeneous and enzymatic catalysts demonstrated significant success; however, in industry, heterogeneous catalysts were preferred due to their easy separation, high stability, and recycling potential. Calcium oxide (CaO) and CaO-based materials were considered as appropriate catalysts due to strong basicity, low cost, and abundance in nature. Nevertheless, the catalytic performance is limited by many issues, e.g., precursor type, loading, surface area, and stability under reuse. Understanding the structure-activity relationship of CaO-based catalysts is therefore essential for optimizing the design of catalysts. This review analyzes the performance of CaO-based catalysts for biodiesel production considering key development stages - source sustainability, catalyst loading, specific surface area, and reusability - to critically examine the impact of parameters on catalytic efficiency and potential industrial implementation. The review highlights the nonlinearity and interdependence of key catalyst parameters affecting biodiesel yield in transesterification. All the reviewed studies were summarized in tables at the end of the relevant review chapter.
Keywords
INTRODUCTION
The progressive accumulation of carbon dioxide (CO2) in the atmosphere, primarily increasing as a result of the combustion of fossil fuels, remains one of the major drivers of global climate change[1-3]. Transfer to renewable and carbon-neutral fuels is therefore essential to mitigate greenhouse gas emissions and achieve global decarbonization targets[4,5]. In this context, biodiesel represents a particularly compelling alternative due to nearly balanced carbon cycle [Figure 1]. CO2 released under biodiesel combustion is approximately equivalent to the amount fixed by plants due to photosynthesis of the oil-bearing biomass, resulting in no net increase in atmospheric carbon[6]. The closed-loop carbon exchange contrasts fundamentally with the linear carbon release associated with fossil-derived hydrocarbons. The oxidation of geologically deposited carbon results in cumulative atmospheric enrichment of CO2[7]. Thus, biodiesel utilization supports the broader objective of maintaining a sustainable carbon balance within the global energy system.
Biodiesel consists primarily of fatty acid methyl esters (FAMEs) obtained via transesterification of triglycerides with methanol[8,9]. Biodiesel is now commercially produced and utilized in many countries either as a pure fuel (B100) or in blends with fossil diesel (B5-B20) due to high biodegradability[10-12], lower emission of sulfur oxides and dust[6], and favorable combustion properties[13]. Continuous governmental support, environmental regulations, and advances in feedstock valorization further reinforce the role of biofuels in the global transition toward carbon-neutral energy paradigm[14-16].
A wide variety of feedstocks can be employed for biodiesel production, including edible and non-edible vegetable oils, waste cooking oils, animal fats, and microalgal lipids[17-19]. The feedstock selection depends largely on local availability and economic issues, yet the choice of oil significantly impacts the process efficiency and the quality of the final product[17]. While the chemical pathway of transesterification is straightforward, the industrial realization is challenging due to side reactions, impurities in the feedstock, and limitations of catalytic issues[20]. Therefore, optimizing the catalytic route is essential to ensure high conversion rates, process stability, and cost-efficiency[21].
Catalysts used for biodiesel synthesis are generally classified into three rows: homogeneous, heterogeneous, and enzymatic[22]. Homogeneous alkaline catalysts, e.g., sodium hydroxide or potassium methoxide, provide high biodiesel yields due to high activity and short reaction time[23]. However, the use of homogeneous catalysts is limited by foam formation with free fatty acids, challenges in catalyst recovery and wastewater generation after purification[24,25]. Although acidic catalysts successfully esterified high acid number feedstocks, require longer reaction time and higher temperature[26]. Enzymatic catalysts based on lipases provided high selectivity, operating under mild conditions, yet the high cost and susceptibility to deactivation limit large-scale implementation[27].
Heterogeneous catalysts are considered as more appropriate for industrial biodiesel manufacturing[28-30]. The solid nature enables simple separation from a reaction mixture, regeneration, and potential reuse, minimizing further processing costs[31]. Moreover, tuning physicochemical properties - basicity, surface area, and pore architecture provide flexible optimization of catalytic performance[32]. CaO-derived catalysts demonstrated promising potential due to the strong basicity, low cost, wide availability from natural and waste-derived sources[33-35]. Despite the advantages, pure CaO suffers from several limitations that restrict reusability and long-term stability. Common challenges include leaching of active Ca2+ species into reaction medium, surface poisoning by glycerol and free fatty acids, and structural sintering during repeated use[36-38]. The physicochemical characteristics of CaO are strongly dependent on precursor type, calcination temperature, and post-treatment methods, resulting in significant variations in catalytic activity reported in the literature. To address these issues, many modifications were developed: doping with alkali or rare-earth metals, immobilization on porous supports (e.g., SiO2, Al2O3, zeolites, biochar), formation of mixed oxides, and the design of nanostructured or core-shell morphologies[32,39-42]. The developed approaches aimed to enhance the dispersion of active sites, increase specific surface area, and inhibit the catalyst leaching.
A series of recent reviews considered CaO-based catalysts for biodiesel production, varying in scope, depth, and focus. Biomass-derived CaO catalysts and/or industrial wastes were summarized elsewhere[31-33,35,43-49], and various approaches to catalyst modification were discussed[35,39,50-52]. Deactivation processes, regeneration conditions, and reuse opportunities were previously highlighted[34,52-55]; the other studies were devoted to the use of CaO nanocatalysts[56]. A comparative analysis of the issues considered in related reviews is presented in Table 1.
Comparison of some related reviews on CaO catalysts for biodiesel production
| Main idea | Issues to be addressed | Ref | |||
| Source of CaO | Effect of surface area | Dependence of reuse cycles on design | Integrated analysis (parameter - biodiesel yield) | ||
| The catalytic performances of recently reported CaO as heterogeneous solid-base catalytic systems | √ | √ | × | × | [32] |
| The sources, production methods and characteristics of CaO, data on improving catalytic activity and durability through modification | √ | √ | √ | × | [33] |
| Current advancements and suggested pathways for future development to improve the industrial viability of waste-derived CaO catalysts | √ | √ | √ | × | [34] |
| Modification approaches for waste-shell derived CaO-based catalysts | √ | √ | √ | × | [35] |
| Achievements in biodiesel production using calcium oxide catalysts; impact on the synthesis of calcium oxide catalysts | × | √ | √ | × | [39] |
| The use of calcium oxide waste-based catalysts for industrial and environmental applications | √ | × | × | × | [43] |
| The role of the catalysts derived from waste shells, animal bones, biomass ashes and carbon-based catalysts in transesterification | √ | × | × | × | [44] |
| Critical study of the parameters of CaO catalyst for catalytic efficiency and guaranteed industrial application | √ | √ | √ | √ | This work |
In the field of biodiesel synthesis and heterogeneous catalysis, current research is focused not on the feasibility of CaO-based systems, but on the improvement of the performance under industrial conditions. Catalyst durability is considered as a crucial parameter for technological and economic viability[57,58]. Although numerous studies optimized strength, surface area, precursor origin, calcination strategy, and loading, the effect of these values is often interdependent and nonlinear[34,38,59] Thus, the potential of CaO-based catalysts is in positive trend, and researchers can achieve outstanding results through rational design, structural modification, and regeneration methods[60-63]. Despite the progress, the gap between laboratory performance and long-term industrial applicability remains high, highlighting the need to improve catalyst durability under high activity[23]. In this context, the present review provides a systematic analysis of CaO and CaO-based catalysts for biodiesel synthesis, focusing on physicochemical properties and catalytic activity. The discussion was systematized according to the key stages of the development in production of CaO catalysts: (i) sustainability and origins of the CaO source, (ii) catalyst loading, (iii) specific surface area (BET), and (iv) reusability and structural stability. Each chapter is considered through recent literature data to elucidate how these parameters define catalytic efficiency and industrial perspectives. This structured approach aims to bridge the gap between laboratory-scale observations and industrial feasibility, guiding the design of robust and sustainable CaO-based catalysts for future biodiesel technologies.
THE REACTION MECHANISM IN THE PRESENCE OF CaO-BASED CATALYSTS
The mechanism of CaO-catalyzed transesterification and the role of modification strategies
Biodiesel is produced by transesterification of triglycerides of various origins (vegetable, animal, or waste fats) in the presence of alcohols, primarily methanol, and the catalyst. The catalytic activity of CaO is high due to the presence of oxygen anions (O2-) and calcium cations (Ca2+) on the surface, forming strong basic and weak Lewis acidic sites, respectively[34]. The sites activate an alcohol, providing transesterification reaction[64]. Before the reaction, the catalyst surface must be free of carbonates and hydroxides; therefore, thermal activation at 600-900 °C is desirable to obtain pure and highly basic CaO[32]. The transesterification mechanism for biodiesel production using CaO-based catalyst is shown in Scheme 1. In the first step, CaO base sites eliminate a proton from methanol, forming a surface methoxide ion (CH3O-) bounded to a calcium ion[65]. The methoxide then attacks the carbonyl carbon of a triglyceride molecule, forming a tetrahedral intermediate. The decomposition of the tetrahedral intermediate results in FAME and diglyceride anion. A diglyceride anion is stabilized by the transfer of a proton from the hydroxyl group in a catalyst, regenerating the active CaO site. This sequence is repeated until all three ester bonds of the triglyceride have reacted, resulting in the formation of three moles of methyl esters and one mole of glycerol. The process involves alternating steps of methanol activation, nucleophilic attack and catalyst regeneration. Several other studies were reported complementary in mechanistic insights of CaO-catalyzed biodiesel production[32,35,52,66,67].
Various modification strategies - including metal doping, support integration, surface functionalization, and the construction of composite or magnetic systems - have been employed to regulate the catalytic behavior of CaO-based materials[33,38,68,69]. For example, support integration and dopant addition have been shown to reduce Ca2+ leaching and enhance catalyst reusability[32,68,70]. Modifications with alkali and alkaline metals can alter basic strength and surface properties, improving catalytic performance and stability[71]. Structural optimization through nano-structured catalysts further enhances activity and durability of heterogeneous systems[72]. Consequently, catalyst modification serves as a regulatory tool that governs both reaction performance and long-term durability, highlighting its importance in the rational design of advanced CaO-based catalysts.
The dynamic evolution mechanism of active sites during the reaction process
Interestingly, the active phase of the CaO catalyst changes with the conversion rate[55]. When the transesterification of oil is carried out in a batch mode at low conversion rates, the reaction is catalyzed either by Ca-OCH3, obtained by the reaction of CaO with methanol, or by the original CaO surface. After the conversion rate is high enough to produce a significant amount of glycerol, CaO is converted to calcium glyceroxide Ca[O(OH)2C3H52. Then, CH3O-Ca-O(OH)2C3H5 is deposited on calcium glyceroxide and acts as a solid base catalyst. This species is known to be partially soluble in a reaction mixture (biodiesel/methanol/glycerol)[73], and its formation reflects a combination of surface reconstruction and moderate leaching of calcium compounds. The CaO → Ca(OH)2 transformation decreases the availability of strong basic sites required for methanol activation, while the formation of calcium diglyceroxide leads to the removal of Ca2+ from the solid lattice. Although calcium diglyceroxide can act as a homogeneous cocatalyst[74], its partial solubility contributes to catalyst weight loss and explains the observed decrease in solid phase basicity after reuse. Overall, these transformations provide a mechanistic explanation for the decrease in catalytic activity, directly linking phase evolution to active-site depletion and leaching-driven deactivation.
The synergistic action pathway of acid-base dual-functional catalysts
The activity of the bifunctional CaO catalyst in oil transesterification depended on the acidic and basic nature of a catalyst[75-80]. A general description of the oil transesterification mechanism using a bifunctional catalyst is shown in Scheme 2.
The acidic site of the positively charged bifunctional catalyst adsorbed fatty acids, while the basic site was negatively charged active site and adsorbed methanol[75]. This dual functionality enables a synergistic reaction pathway in which esterification and transesterification occur simultaneously rather than as independent processes. The presence of acid-base synergy improves tolerance toward feedstocks with elevated free fatty acid content and reduces soap formation[81,82]. Moreover, the cooperative interaction between acidic and basic sites enhances reaction kinetics and broadens the operational window of CaO-based catalysts, making them more suitable for practical biodiesel production.
STAGES OF DEVELOPMENT IN THE DESIGN OF AN EFFICIENT CAO-BASED CATALYST
Source of CaO-catalyst
One of the key steps in the development of CaO-based catalysts for biodiesel production is the study of feedstock sustainability. Catalysts with distinct physicochemical properties can be produced from various feedstocks due to the diversity of matrix and the presence of associated elements. Therefore, the choice of feedstock directly impacts the activity, stability, and recovery potential of a catalyst. This development is a valuable step in ensuring a stable supply of materials with reproducible chemical composition and sustaining environmental impact over a long period of time. This chapter systematizes the literature data on the proposed sustainable CaO sources derived from biomass and industrial wastes. Biodiesel synthesis using catalysts obtained from the selected sources were discussed, including reaction conditions, achieved conversion rates, product yields, the resource and environmental advantages of employing renewable and secondary feedstocks. The achievements and results discussed in this chapter were summarized in Table 2.
Sources of CaO precursors, calcination temperatures and biodiesel yield
| Catalyst type | Source of CaO | Calcination temperature,°C | Calcination time, h | Biodiesel yield, % | Ref. |
| CaO | White bivalve clam shell | 900 | 4 | 94.25 | [83] |
| CaO | Mussel shell | 1050 | 2 | 94.1 | [84] |
| CaO | Cockle shell (Anadara granosa) | 900 | 2 | 97.48 | [85] |
| CaO | Clam shell (Mereterix mereterix) | 900 | 3.5 | 89.91 | [86] |
| CaO | Razor clam shell surf clam shell | 900 | 2 | 98 98 | [87] |
| CaO | Oyster shell, Pyramidella shell | 900 | 2 | 93.92 94.81 | [88] |
| CaO | Scallop shell | 900-1,000 | 2 | 90 | [89] |
| CaO | Scallop seashells (Noble Pectin) | 700 | 3 | 97 | [90] |
| CaO | Scallop shell | 1,000 | 2 | 86 | [91] |
| CaO | Scallop shell | 1000 | 3 | 81.7-97.5 | [92] |
| CaO MgO-CaO | Limestone scallop shell MgCO3-scallop shell | 900 | NR | 93 83 96 | [93] |
| CaO | Donax deltoides shell | 900 | 3 | 96.5[b] | [94] |
| CaO | Bivalve mollusk shell (Margaritifera falcata) | 850 | 2.5 | 98.2[b] | [95] |
| CaO | Obtuse horn shells | 800 | NR | 86.75[c] | [96] |
| CaO | Crab shell eggshell | 900 | 2 | 83.10 97.75 | [97] |
| CaO | Crab shell | 800 | 4 | 96.6 | [98] |
| CaO | Mollusk shell crab shell | 800 700 | NR | 95 97 | [99] |
| CaO ZnO-CaO TiO2-CaO | Snail shell | 900 500-600 500-600 | 3 | 90 90 95 | [100] |
| CaO-TiO2 | Cockle and sea snail shells | 900 | 4 | 93.19 | [101] |
| CaO | Calcium nitrate, snail shells | 700 | 3 | 93 96 | [102] |
| CaO | River snail shell ash | 800 | 4 | 98.19 | [103] |
| KF-CaO | Snail shell | 850 | 4 | 98 | [104] |
| CaO | Pomacea sp. shell | 900 | 2 | 95.61 | [105] |
| CaO | Snail shell | 900 | 4 | 97.4 | [106] |
| CaO | Shell (Turbonilla striatula) | 900 | 4 | 93.3 | [107] |
| CaO-Fe3O4 | Snail shell | 900 | 4 | 97.7 | [108] |
| CaO | Chicken eggshell | 900 | 2 | 95.0 | [109] |
| CaO | Chicken eggshell | 900 | 4 | 97.98 | [110] |
| CaO CaO-NP | Chicken eggshell calcium nitrate | 800 | 3 | 83 85 | [111] |
| CaO | Chicken eggshell | 900 | 3 | 99.07 | [112] |
| CaO-NC | Eggshell | 900 | 3 | 94 | [113] |
| CaO | Chicken eggshell | 900 | 2 | 91 | [114] |
| CaO-NC | Eggshell | 950 | 3 | 88 | [115] |
| CaO | Eggshell-coconut pith | 700 | 3 | 72 | [116] |
| CaO | Eggshell | 900 | 4 | 97.84[c] | [117] |
| CaO ZnO-CaO CuO-CaO | Chicken eggshell | 900 | 4 | 68 93.2 90.6 | [118] |
| CaO-MgO-NC | Chicken eggshell | 850-900 | 4 | 98.37[b] | [119] |
| CaO-PVA | Eggshell | 900 | 4 | 95 | [120] |
| CaO | Eggshell | 900 | 2.5 | 93.48[c] | [121] |
| CaO | Chicken eggshell | 900 | 3 | 83.2 | [122] |
| CaO | Eggshell | 900 | 3.5 | 91.6[b] | [123] |
| CaO | Eggshell | 900 | 3 | 97 | [124] |
| CaO | Eggshell golden apple snail shell meretrix venus shell | 800 | 4 | 95 | [125] |
| MEC-CaO NEC-CaO | Eggshell | 900 | 4 | 94.7 96.7 | [126] |
| CaO | Nano eggshell | 900 | 4 | 98.1 | [127] |
| CaO | Eggshell | 800 | 4 | 96.7 | [128] |
| CaO | Chicken eggshell | 900 | 3 | 93 | [129] |
| CaO | Commercial eggshell | 850 | 3 | 92.6 91.4 | [130] |
| CaO | Eggshell | 900 | 2 | 100 | [131] |
| CaO | Eggshell | 900 | 4 | 99.7[c] | [132] |
| CaO | Waste coral | 900 | 2.5 | 100 | [133] |
| CaO-MgO | Skeletons of Clypeaster reticulatus (Sand Dollar) | 800 | 6 | 90 | [134] |
| CaO | Chicken bone | 800 | 5 | 96.31 | [135] |
| CaO-PVA | fish bone | 900 | 4 | 80.4 | [136] |
| CaO | Chicken manure | 850 | 4 | 91 | [137] |
| CaO | Biomass gasification bottom ash | 800 | 4 | 96 | [138] |
| CaO | Palm kernel shell biochar | 800 | 2 | 5 | [139] |
| CaO | Palm kernel shell biochar | 800 | 2 | 99 | [140] |
| CaO | Carbide slag | 850 | NR | 93.83 | [144] |
| NaOH/Na2CO3/NaNO3-CaO | Carbide slag | 850 | NR | 95[a] | [171] |
| CaO | Lime mud | 800 | NR | 94.35[c] | [173] |
| Ba-CaO | Waste plaster of paris | 900 | 3 | 91.83 | [174] |
| MgO/CaO | Waste from a phosphoric acid plant | 900 | 2 | 93.32 | [175] |
Biomass waste
CaO was synthesized from natural white bivalve clam shells (WBCS) via calcination-hydration-dehydration treatment, yielding the catalyst with increased surface area and basic strength with rod-like particles ranging in size from 53.9 to 62.6 nm, bound into aggregates[83]. Biodiesel conversion of 94.25% was observed by transesterification of waste frying oil using 7 wt% catalyst loading, a methanol-to-oil molar ratio of 12:1, at
The Anadara granosa shell waste was converted to CaO catalyst under calcination at 900 °C for 2 h[85]. Under optimized conditions, 97.48% ± 0.24% biodiesel yield was obtained from palm oil. Biodiesel yield of over 89% and conversion of over 97% from waste frying oil were obtained using powdered calcined clam shell (Mereterix mereterix) as a catalyst[86]. Long-term calcination of the clam shell from 2.5 to 3.5 h resulted in increased catalyst activity and shorter transesterification reaction time. However, the improvement of the yield was not observed. CaO was obtained from the razor and surf clam shells by crushing, sieving through
Calcined waste scallop shell (Patinopecten yessoensis) was used as a basic heterogeneous solid catalyst for the transesterification of tripalmitin as a model triglyceride for desired biodiesel production[89]. The scallop shell was pulverized and calcined at 900-1,000 °C for 4 h. The temperature under calcination of the scallop shell did not impact the overall reaction rate constant. Interestingly, compared to the calcined commercial source (CaCO3), lower overall reaction rate was observed in the presence of CaO from scallop shell due to the difference in CaO amount. CaO catalysts derived from scallop shells of noble pectin were active in biodiesel production, providing 97% yield for both pure sunflower oil and waste cooking oil[90].The calcination temperature of 700 °C was sufficient to ensure the decomposition of CaCO3 in the shells to CaO. Transesterification of sunflower oil proceeded according to pseudo-first-order kinetics with an activation energy (Ea) of 133.57 kJ/mol and a pre-exponential factor (A) of 4.025·1018 min-1. In another case, a catalyst obtained by calcining scallop shell at 1,000 °C provided biodiesel fuel in 86% yield from waste cooking oil under optimized conditions[91]. The authors demonstrated the morphology of the catalysts with significant transformations at a higher calcination temperature, exceeding 1,000 °C: the particles sintered, were larger and smoother, resulting in decreased surface area of the catalyst and decreased yields of biodiesel.
The specific surface area of CaO obtained from scallop shell was increased by dry nano-grinding using methanol[92]. The analysis of the catalyst after nano-grinding demonstrated the formation of three various calcium-based phases on the oxide surface: calcium oxide, calcium methoxide and calcium hydroxide. The highest catalytic activity in the methanolysis was observed for the catalyst samples subjected to partial surface conversion to Ca(OH)2. The crucial issue for the catalytic activity of the scallop shell catalyst was the presence of NaCl impurity[93]. The shell-based catalyst with lower basicity consisted of fairly large particles. For calcined limestone, all the observed particles were very small and formed in micron-sized agglomerates with a significant number of gaps. On the other hand, calcined scallop shell consisted of large micron-sized particles with no gaps, which probably acted as catalytic pores. The catalytic activity of calcined scallop shell was increased by mixing the shell source with basic MgCO3 before calcination, which resulted in a decrease in the crystal size of the lime phase and the particle size consisting of the CaO matrix. In particular, the CaO catalyst was successfully obtained by calcining Donax deltoides (DDS) bivalve sea shells[94], freshwater bivalve waste (outer shell of Margaritifera falcata)[95], waste obtuse horn shells[96].
The crab shell CaO catalyst converted triglycerides from sunflower oil to biodiesel in 83.10 ± 0.27 wt% yield, which was lower than for eggshell-derived catalysts 97.75 ± 0.02 wt%[97]. The lower catalytic activity of calcined crab waste was explained by the lower surface Ca content. In a recent work, crab shell waste calcined at 800 °C for 4 h catalyzed the transesterification of Waste Fish Oil in biodiesel (96.6% yield)[98]. Biodiesel was obtained in 95% and 97% yields, respectively, using waste mollusk and crab shells collected from a seafood restaurant[99].
CaO catalyst obtained by calcining snail shells provided biodiesel from waste cooking oil in 80% yield[100]. Further modification of the CaO from the shells with ZnO and TiO2 oxides followed by calcination increased the biodiesel yield to 90% and 95%, respectively. In a similar case, an increase in biodiesel production was also observed using CaO obtained from waste cockle and sea snail in the presence of the catalyst impregnated with TiO2 (from 94.58% for CaO to 98.78% for CaO-TiO2)[101]. According to the Hammett indicator method, CaO nanoparticles from snail shell demonstrated stronger basicity (base strength in the range of 9.8 < H < 18.4) than CaO nanoparticles prepared from calcium nitrate (base strength in the range of 9.8 < H < 17.2)[102]. Differences between the catalysts were also observed in the average crystallite size, 44 and
Eggshells calcined at 850 °C were used for transesterification of karanja oil to produce biodiesel in 95% yield and high conversion (97.43%)[109]. In another work, CaO derived from eggshells was used to produce biodiesel from Terminalia bellerica oil with 97.98% FAME yield[110]. Optimization was carried out to maximize the biodiesel yield using Response Surface Methodology (RSM) with three independent values: molar ratio, amount of catalyst and reaction temperature. CaO from eggshell was comparatively characterized with CaO nanoparticles (CaO-NPs) prepared from Ca(NO3)2[111]. XRD data demonstrated that CaO was present in both catalysts and the average crystalline size obtained was 42 and 50 nm for CaO-NPs and eggshell CaO, respectively. Eggshell CaO demonstrated the best results in the catalyst loading amount study: 6 wt% loading of eggshell CaO catalyst resulted in 83% biodiesel yield, compared to 85% biodiesel yield at 12 wt% loading of CaO-NPs.
Modification in the preparation of CaO catalyst from chicken eggshell (after the standard calcination at
CaO derived from eggshells and doped with transition metals was used in the transesterification of eucalyptus oil[118]. According to SEM analysis, zinc doping of chicken eggshell waste changed the surface structure of the catalyst at 900 °C from an irregular micromorphology of rod and dumbbell-like structures to honeycomb-like porous surface, which provided a larger surface area and a higher resulting biodiesel yield (from 68% for CaO to 93.2% for Zn-CaO). CaO-MgO obtained from eggshell and magnesium compounds was used in transesterification of waste edible oil[119]. The process optimization was performed using RSM with central composite design (parameters: reaction time, temperature, catalyst dosage and methanol/oil ratio) to obtain the maximum biodiesel yield of 98.37%.
CaO catalyst prepared from eggshell and solid coconut waste (in a ratio of 5:1) mixed with polyvinyl acetate (PVA) was loaded into a PBR[120]. The mixture of solid coconut waste and eggshell was calcined at 900 °C for 4 h and, then, added to an aqueous solution of PVA. The mixture was stirred and poured into Petri dishes and dried in an air dryer at 65 °C for 24 h. The X-ray diffraction pattern of the catalyst demonstrated that the polymeric catalytic membrane formed crystalline structure. The highest biodiesel yield 95% was obtained at 61 °C with 2.29 wt% catalyst loading and methanol to solid ratio of 12:1. A laboratory scale continuous flow jacketed reactive distillation (RD) unit was developed for the transesterification of waste frying oil using a heterogeneous eggshell catalyst[121]. Eggshells were washed, dried, chopped to small pieces and, then, calcined in a muffle furnace under static air conditions at 900 °C for 2.5 h. According to XRD analysis, the prepared catalyst was identified by characteristic peaks for CaO and low intensity peaks for Ca(OH)2. The micromorphology of the catalyst was regular with rod-shaped particles with a size range of 1.29 to 2.0 mm width. The maximum conversion of methyl ester 93.48% was obtained at reagent flow rate of 0.2 mL min-1, methanol/oil ratio of 4:1 and catalyst bed height of 150 mm. To intensify the biodiesel production process, the eggshell catalyst was first incorporated into a continuous centrifugal contactor separator (CCCS)[122]. The CCCS consisted of an outer static housing and an inner rotating centrifuge. The reactor was designed for in situ separation of biodiesel from the glycerol layer by centrifugal forces (rather than in another separation vessel) and resulted in an increased mass transfer rate in the annular mixing zone in the presence of a strong shear force acting on the liquids by a rotating centrifuge. The eggshell catalyst was prepared by washing with distilled water, drying at 100 °C, grinding with a mechanical mill, further sieving (0.3-0.5 mm) and calcining at 900 °C for 3 h. Optimized 83.2% FAME yield was obtained with a productivity of 638 kgFAME/(m3reactor·h) at oil flow rate of 9 mL/min, methanol/oil molar ratio of 11:1, and weight hourly space time (defined as catalyst weight over the oil mass flow rate) for 0.05 h[122]. In particular, eggshell catalysts demonstrated successful implementation in the transesterification of oils in a series of other studies[123-132]. Coral waste calcined at
Waste chicken bones containing calcium carbonate were successfully converted into CaO catalyst[135]. Waste chicken bones were washed with water, boiled in sodium hydroxide solution for 1 h and rinsed with water again to remove proteins. After cleaning, the waste bones were dried in an oven at 100 °C for 2 h, pulverized and calcined at 800 °C in a muffle furnace for 5 h. The maximum yield of the obtained biodiesel was 96.31% ± 0.72% under the optimal conditions of 3.0 wt% catalyst, 3:1 methanol/oil molar ratio of 3:1 and at 80 ± 5 °C for 3 h. CaO catalyst for transesterification of palm oil was synthesized from fish bone waste by calcination at 900 °C for 4 h and was supported on polyvinyl alcohol (PVA)[136]. The maximum yield of FAME was 80.40% at 10 wt% catalyst amount, 20:1 methanol to oil molar ratio at 65 °C.
Calcium compounds (CaCO3, hydroxyapatite according to XRD analysis) were found in chicken manure[137]. The collected fresh chicken manure was dried in the sun for several days, then grinded, calcined in air for 4 h at 550 to 950 °C and grinded again. The optimized temperature 850 °C resulting in complete conversion of CaCO3 to CaO. According to Energy-Dispersive X-Ray (EDX) analysis, up to 74% by weight of calcium,
A significant amount of calcium carbonate (precursor of CaO catalyst) was observed in bottom ash from woody biomass gasification[138]. Interestingly, the char (or unburned carbon) in the raw bottom ash was completely removed after calcination. The particle size of the catalysts and their regularity increased with increasing calcination temperature. In the XRD patterns, the intensity of the characteristic peaks for CaO increased with increasing calcination temperature. For the bottom ash calcined at 600 °C, the main peaks observed were still the characteristic peaks of CaCO3. Complete conversion of CaCO3 to CaO was observed when the bottom ash was calcined at 800 °C and above. Under optimized reaction conditions (methanol-to-oil molar ratio of 20:1, 5 wt% catalyst, and 65 °C), 90% yield of biodiesel was obtained from palm oil.
Industrial waste
Palm kernel shells (PKS) are the most difficult fraction of the solid waste to decompose that are produced in oil palm mills. The PKS solid residues were thermally treated in a gasifier at 800 °C for 2 h under atmospheric pressure to produce palm kernel shells biochars (PKSB)[139]. The extensive characterization of the biochars demonstrated a high calcium content, predominantly in the form of calcium carbonate. After calcination of the biochars, calcium carbonate was converted to quicklime (CaO). As a result, FAME were obtained in 99% yield in the presence of the calcined PKSB at 9:1 methanol-to-sunflower oil ratio at 60 °C. Later, the same research group carried out optimization the production of FAME[140].
The CaO catalyst for biodiesel production was successfully prepared by calcining large-scale industrial waste calcium carbide residue (CCR)[141-145]. CCR is a byproduct of acetylene manufacturing via hydrolysis of calcium carbide[146,147]. Since calcium carbide is widely used in industry and demonstrated promising potential for application in chemistry[148-159], the amount of calcium carbide slag generated is constantly increased. Although CCR is used in many fields[160-169], the amount of slag accumulated is several times higher than the actual consumption. The principal advantage of CCR as a CaO source is the decrease of carbon dioxide emission at the catalyst preparation stage compared to carbonate sources[145,170]. Calcined CCR successfully catalyzed the transesterification of soybean oil and waste cooking oil with 99% and 96% yields, respectively. In another example, CCR calcined at 850 °C was successfully used in the transesterification of peanut oil to obtain 93.83% biodiesel yield[144]. Calcium hydroxide and calcium carbonate, as the main components of CCR, were converted to CaO by calcination, resulting in a catalyst with large surface area, simple functional groups, developed porous morphology and basic strength 9.8 < H < 15.0. NaOH added to calcined CCR decreased the activation temperature of CCR[171].
Lime mud (LM) is a large-ton waste of paper mills, which is formed by the causticization reaction in the alkali recycling process of paper making[172]. The dominant component of LM is calcium carbonate, which can be easily converted to calcium oxide by calcination. The optimum calcination temperature for the conversion to CaO was 800 °C[173]. Higher calcination temperatures indicated catalyst sintering and catalyst grain shrinkage as determined by N2 adsorption. Under optimized conditions (methanol to oil molar ratio 12:1, reaction temperature 64 °C, and 6% catalyst loading), the transesterification conversion exceeded 90% for 2 h.
Barium-doped catalyst was synthesized from waste plaster of Paris (PoP) using hydrogen peroxide treatment and modification with calcium and barium, followed by calcination at 800-900 °C[174]. During the transesterification of Catharanthus roseus seed at 58 °C for 70 min, with a methanol-to-oil molar ratio of 15:1 and 4 wt% catalyst loading, 91.83% biodiesel yield was achieved, enabling the combination of waste valorization with high catalytic efficiency and environmental sustainability.
Industrial waste from a phosphoric acid plant in Yazd, Iran, was used as a source of CaO to produce MgO/CaO composite catalysts[175]. The daily waste generated at this plant consisted of 2.98% Fe2O3, 92.32% CaO and 3.93% P2O5. At the optimal catalyst calcination temperature (1,100 °C), the biodiesel yield from waste cooking oil was 93.32%.
Current research demonstrates that both biogenic carbonate materials (e.g., shells, bones, eggshells) and hydroxide-containing industrial byproducts (e.g., CCR, LM) can be used as sustainable and efficient sources of CaO catalysts for biodiesel production. While biogenic carbonates are abundant and easily accessible, their use accompanied higher CO2 emission as a result of calcination and logistical challenges in large-scale collection and preprocessing. In contrast, hydroxide-based sources provide lower carbon footprints and often require lower activation temperatures, but their availability may be locally limited. Despite remarkable laboratory-scale performance - with yields frequently exceeding 95% under optimized conditions - several critical issues are still challenging. Long-term catalyst stability, resistance to deactivation by feedstock impurities, and reproducibility of performance from variable raw material qualities are rarely assessed under continuous or industrial-scale conditions. Moreover, the interplay between source composition, activation protocol, and final CaO microstructure requires deeper mechanistic insights to guide rational catalyst design. Further research is expected to converge on integrated waste valorization strategies that not only recover CaO from diverse residues but also tailor its physicochemical properties via controlled calcination, doping, and surface functionalization. Combining tailored CaO catalysts with process intensification technologies - including microwave-assisted transesterification, reactive distillation, and centrifugal contactors - opens significant promise for scaling biodiesel production with minimized environmental impact.
Catalyst loading for transesterification
Catalyst loading is important operating parameter for the modification of the efficiency of the transesterification. The availability of active basic sites, the degree of triglyceride conversion, and the overall reaction kinetics are originated from a catalyst. Low catalyst loading results in incomplete transesterification and low biodiesel yield. In contrast, high loading may increase the viscosity of the reaction medium, inhibited mass transfer, promote foam formation, and make separation of biodiesel from the catalyst challenging. Using a minimum amount of a catalyst is beneficial from both an economic and operational point of view, since it reduces material costs and simplifies processing. This section presents a critical review of the literature, considering CaO-based catalysts in transesterification at loadings lower than 2 wt%. The focus was on the effect of catalyst dosage in this low range on biodiesel yield and reaction efficiency. A comparative analysis was carried out to assess whether a minimum catalyst loading can provide high efficiency while maintaining simplicity and cost-effectiveness of the process. The achievements and results discussed in this chapter were summarized in Table 3.
Impact of catalyst loading on biodiesel yield in the presence of CaO-based catalysts
| Catalyst type | Source of CaO | Catalyst loading, wt% | Reaction condition: alcohol/oil ratio, temperature (°C), time (h) | Biodiesel yield, % | Ref. |
| CaO/MgO | Ca(OAc)2 | 2 | 18:1, 64.5, 3.5 | 92[c] | [201] |
| CaO@γ-Fe2O3 | CaCl2 | 2 | 15:1, 70, 3 | 98.8 | [202] |
| CaO/ZnO | Ca(OH)2 | 2 | 10:1, 60, 3 | 99 | [203] |
| Ag-CaO | Eggshell | 2 | 3:1, 60, 2 | 99.3 | [204] |
| CaO | Commercial | 2 | 9:1, 60, NR | 84.5 | [205] |
| W-Mo/CaO | Eggshell | 2 | 15:1, 70, 2 | 96.2 | [206] |
| W-Zr/CaO | Eggshell | 2 | 15:1, 80, 1 | 94.1 | [207] |
| CaO | Eggshell | 2 | 12:1, 60, 3 | 100[c] | [208] |
| CaO | Eggshell | 2 | 8:1, 65, 2.5 | 90 | [210] |
| CaO | Eggshell | 2 | 13:1, 60, 1 | 96 | [211] |
| CaO | Eggshell | 2 | 10.5:1, 60, 2 | 99 | [209] |
| CaO | Snail shell | 2 | 6.03:1, 60, 8 | 87.28 | [212] |
| CaO | Snail shell | 2 | 12:1, 60, 1.5 | 93 | [213] |
| CaO/MgO | Dolomite | 1 | 6:1, 60, 4 | 98.81 | [183] |
| CaO | CaCO3 | 0.85[d] | 12:1, 65, 1 | 93 | [66] |
| CaO | Eggshell | 1.7 | 10:1, 70, 3.6 | 86.41 | [199] |
| CaO | Ca(NO3)2·4H2O | 1.6 | 9:1, 65, 1.17 | 96.2 | [198] |
| CaO | Eggshell | 1.5 | 10:1, 65, 2 | 93 | [193] |
| CaO | Commercial | 1.5 | 9:1, 70, 2.5 | 93[c] | [194] |
| Co-CaO | Eggshell | 1.5 | NR, 60, NR | 98 | [196] |
| CaO | Oyster shell | 1.5 | 9:1, 60, 6 | 96 | [195] |
| CaO | Snail shell | 1.4 | 6:1, 55, 1 | 96.73 | [60] |
| CaO/ZnO | Ca(OAc)2 | 0.2 | 12:1, 60, 2 | 95[a] | [178] |
| K2O/CaO | Commercial | 2 | 9:1, 60, 2 | 91.22 | [200] |
| K2O/CaO | Commercial | 1 | 9:1, 60, 2 | 93.3 | [200] |
| Ba-CaO | shell (Turbonilla striatula) | 1 | 6:1, 65, 3 | 98[c] | [184] |
| CaO | CaCO3 | 1 | 6:1, 60, 2.5 | 88 | [186] |
| CaO | Ca(NO3)2ּ·4H2O | 1 | 7:1, 65, 4 | 99 | [187] |
| CaO | Ca(NO3)2ּ·4H2O | 1 | 8:1, 50, 1.5 | 96 | [188] |
| CaO | Carbide slag | 1 | 9:1, 65, 0.5 | 91.3 | [189] |
| CaO | Eggshell | 1 | 8:1, NR, 0.083 | 95[c] | [192] |
| CaO | Eggshell | 1 | 9:1, 60, 1 | 79.4-83.1 | [185] |
| Au-CaO | Eggshell | 1 | 12:1, 70, 3 | 94.69 | [190] |
| CaO-mayenite | Hydrocalumite (from CaCl2) | 1 | 12:1, 60, 3 | 98 | [191] |
| CaO | Snail shell | 0.866 0.892 | 12.7:1, 56.56, 2 12.4:1, 61.6, 2.42 | 96.93 98.83 | [182] |
| CaO | Snail shell | 0.8 | 12:1, 65, 6 | 95.2 | [181] |
| CaO | Commercial | 0.67[d] | 7:1, 60, 3 | 90 | [180] |
| CaO | Eggshell | 1.5[d] | 12:1, 65, 2 | 98[c] | [197] |
| CaO | Shell | 0.4 | 35:1, 65, 2 | 93 | [179] |
| CaO | Egg, oyster and clam shell | 0.3-0.6 | 60:1[e], 60, 0.5 | 99[a] | [176] |
| CaO | Calcium acetate Calcium carbonate Calcium hydroxide Calcium nitrate Calcium oxalate Commercial | 0.06[d] | 6:1, 60, 1 | 68[c] 38[c] 82[c] < 1[c] 60[c] 3[c] | [177] |
Catalyst loading (X): X < 1 wt%
Calcium oxide (CaO) catalysts derived from waste shells of egg, oyster and clam were prepared and used in the methanolysis of soybean oil at 0.6 wt% catalyst loading[176]. Five h of sonication of eggshells reduced the CaO particle size by 34%, which resulted in 56% increase in the activity. Two cycles of hydration-dehydration applied to the material obtained by calcination of oyster shells provided CaO catalyst with 27 m2g-1 surface and the transesterification rate 2.5 times higher than that obtained with the untreated sample. The authors demonstrated that additionally treated catalysts can be successfully used in loading of 0.03 wt%. CaO catalysts prepared by calcination of various calcium compounds (acetate, carbonate, hydroxide, nitrate, and oxalate) were used in a model reaction of tributyrin transesterification with methanol at 0.06 wt% loading (16.4 g of reactant to 0.010 g catalyst)[177]. The shapes of the catalyst particles and their surface states varied depending on their precursors. The calcium acetate catalyst (CA-800) consisted of bundles of rods with many voids, the calcium carbonate catalyst (CC-800) of small aggregated blocks, and the calcium hydroxide catalyst (CH-800) of aggregated NPs. In contrast, the SEM image of the calcium nitrate catalyst (CN-800) showed a clean surface of large particles. The small microscale particles of the oxalate catalyst (CO-800) had small grooves, and the control CaO catalyst (commercial) demonstrated an uneven surface. Tributyrin was obtained with the best conversion (82%) in the presence of CaO catalyst obtained by calcining calcium hydroxide.
The impregnation of CaO on zinc oxide provided FAME in more than 90% yield under lower catalyst loading: 0.2 wt% based on CaO (1.3 wt% catalyst with 16 wt% CaO was used) for 2 h[178]. It was assumed that the filling of the mesoporous network with ZnO was a key parameter for the stabilization of CaO to prevent leaching of the active phase in a reaction medium. However, the reuse studies and measurements of calcium content in the solutions were not discussed in the research, which would be valuable for a complete understanding of the recycling of the catalyst.
Transesterification of ricebran oil using 0.4 wt% Chicoreus brunneus shell derived CaO nanocatalyst by calcination at 1,100 °C resulted in FAME in 93% yield after 120 min[179]. Pre-activation of commercially available CaO with methanol at 25 °C for 1.5 h followed by catalyst use in the transesterification of rapeseed oil resulted in biodiesel production in 90% yield at catalyst loading of 0.1 g per 15 g of oil (loading of about 0.67 wt%)[180]. The combination of two steps in the preparation of a CaO catalyst from golden apple cherry snail shells - deproteinization (removal of protein) and subsequent calcination resulted in biodiesel in 95.2% yield at 0.8 wt% catalyst loading under optimized conditions[181]. The catalyst provided biodiesel from soybean oil in 93% yield after 1 h of reaction at 0.85 wt% CaO loading (calculated as weight of catalyst/weight of used oil, with an average density of soybean oil of 0.92 g/mL)[66]. Transesterification of used cooking oil resulted in FAME in 99% yield after 2 h; however, a part of the CaO catalyst was converted to calcium foam via reaction with free fatty acids incorporated into WCO during the initial transesterification step (the acid value of WCO was 5.1 mg-KOH/g). The biodiesel yield from Scum Oil and Hydnocarpus wightiana oil was 96.929% and 98.93% at 0.866 wt% and 0.892 wt% loading of CaO obtained from snail shell, respectively[182].
Catalyst loading (X): 1 ≤ X ≤ 1.5 wt%
1 wt% loading of CaO-based dolomite catalyst (CaO/MgO) was used to produce biodiesel from canola oil in 98.81% yield[183]. Conversion of WCO to biodiesel over 98% was obtained using 1 wt% loading CaO obtained from Turbonilla striatula shell waste and doped with 1.0 wt% of Ba[184]. According to XRD data, increasing the amount of barium additive (1.5%) in the catalyst resulted in decrease in catalytic activity, which was associated with the coverage of the basic sites. The decisive factor for the catalytic activity of the catalyst was in high basicity, compared to the other prepared catalyst samples. WCO and bleached palm oil (BPO) were successfully converted into biodiesel using 1 wt% eggshell derived CaO subjected to additional hydrothermal treatments[185]. Waste chicken eggshells were calcined at 850 °C and, then, hydrothermally treated at 120 and 140 °C to obtain two sets of CaO catalysts. Hydrothermal treatment of the catalysts resulted in the decreasing of crystallite size from 47 to 30 and 28 nm at 120 and 140 °C. The biodiesel yields from WCO were 82.7 and 83.1%, and from BPO were 79.4 and 79.6% in the presence of CaO catalysts aged at 120 and 140 °C, respectively. Among the other studies, high biodiesel yields in the range of 88% to 99% were also observed at
The maximum conversion of jatropha oil was 96.73% using 1.4 wt% catalyst loading from calcined snail shell waste[60]. Both a decrease and an increase in the catalyst loading to 0.2 wt% and 2.6 wt% resulted in a decrease in the biodiesel yield to 85.47% and 96.41%, respectively. Biodiesel was successfully synthesized from waste chicken fat in 93% yield using 1.5 wt% eggshell-derived CaO catalyst loading[193]. In another case, treatment of CaO with an ammonium carbonate solution and calcination at 900 °C provided a superbase catalyst with base strength higher than 26.5[194]. Under optimized reaction conditions and catalyst loading of 1.5 wt%, the conversion of Jatropha curcas oil reached 93%. For decalcification of crude synthesized biodiesel, the best results were obtained using citric acid. Under the optimum reaction conditions (1.5% catalyst loading CaO derived from waste oyster shells, methanol to oil ratio 9:1, 60 °C and 6 h) determined using Taguchi-grey (GRA) multi-response analysis, biodiesel was obtained from waste cooking oil in 96% yield[195]. 1.5 wt% loading of CaO catalyst obtained by calcining eggshell at 900 °C and modified with cobalt nitrate hexahydrate by coprecipitation method was used in transesterification of lipids from microalgae biomass with 98% conversion[196]. Unfortunately, the reaction conditions (methanol to lipid ratio and reaction time) were not specified. An acid-treated quail eggshell catalyst at 1.5 wt% loading (0.03 g catalyst to 2 g oil) maintained high conversions higher than 98% even after repeated use for five times in the transesterification of palm oil[197]. The authors suggested that additional two-hour acid treatment of the shell removed the dense cuticular layer and enhanced the catalytic activity by increasing the proportion of the porous front layer with a large number of basic sites and accelerating the diffusion of large oil molecules in the micron-sized pores.
Catalyst loading (X): 1.5 < X ≤ 2 wt%
After Ca(NO3) solution combustion using crude glycerin (biodiesel production byproduct) as the combustion fuel the synthesized CaO catalyst provided 96.2% biodiesel yield from Butea monosperma oil at 1.6 wt% catalyst loading[198]. The CaO nanocatalyst obtained from hen eggs provided biodiesel from microalgae in 86.41% yield at 1.7 wt% optimal catalyst loading[199]. The impact of transesterification reaction parameters on the yield of biodiesel from microalgae was analyzed using RSM based on the full factorial central composite design. The CaO catalyst obtained by impregnation with potassium acetate was applied in the transesterification reaction of bitter almond oil (BAO) and waste fish oil (WFO)[200]. The highest conversion of BAO to methyl esters was 91.22 wt% using 2.0 wt% catalyst. A decrease in the possible loading to 1 wt% in catalyst amount was realized, using WFO in the transesterification, yielding 93.30 wt% methyl esters. The supported CaO/MgO catalyst with 16.5 wt% CaO content resulted in 92% conversion of rapeseed oil at 2 wt% catalyst loading in transesterification[201]. 2 wt% of the core-shell catalyst CaO@γ-Fe2O3 provided biodiesel from soybean oil in 98.8% yield[202]. A decrease in the amount of catalyst resulted in a decrease in biodiesel yield, and an increase did not change the yield. The catalyst provided biodiesel from any feedstock: biodiesel from palm and castor oils was obtained in 95.8% and 90.95% yields, respectively, with the same catalyst loading. The CaO/ZnO catalyst was prepared by ball milling of CaO and ZnO powders with addition of water and in the absence of water, followed by calcination[203]. After calcination at 700 °C, the catalyst prepared by ball milling with the addition of water was more active than the catalyst prepared in the absence of water, which was due to various basicity. The catalyst prepared in this manner at 2 wt% loading provided FAME in 99% yields for 3 h of the transesterification of sunflower oil with methanol in a batch reactor at
Waste cooking oil was converted to biodiesel in 96.2% yield using 2 wt% of solid bifunctional tungsten molybdenum supported calcium mixed oxide catalyst (W-Mo/CaO), which was prepared by simple wet impregnation[206]. Later, the same research group carried out simultaneous transesterification and esterification of unrefined palm-derived waste oil using 2 wt% loading of bifunctional WO3-Zr2O3/CaO catalyst, obtaining biodiesel in 94.1% yield[207]. The other related studies highlighted the use of 2 wt% CaO catalyst loading from eggshells[208-211] and snail shells[212,213] for biodiesel production.
Literature data indicate that CaO-based catalysts are highly efficient even at low loadings (< 1 wt%). In the range of 1-1.5 wt%, a practical optimum can be achieved, combining high biodiesel yields (typically > 90%) with minimized side reactions and process simplicity. Higher loadings (up to 2 wt%) significantly improve performance and may cause foam formation and other drawbacks. The most practical operating range is between 0.8 and 1.5 wt%, where CaO catalysts combine high conversion efficiency with economic and technological feasibility. Going forward, systematic correlations between catalyst structure, base strength, and minimum efficient loading will be crucial for the rational development of next-generation CaO catalysts.
Specific surface area of catalyst (according to Brunauer-Emmett-Teller (BET) method)
The catalytic efficiency of a CaO-based catalyst strongly depends on specific surface area (SBET). This value determines the accessibility of active basic sites and the rate of triglyceride-methanol interactions under transesterification. Standard CaO prepared by direct calcination of limestone or waste shells demonstrates relatively low BET surface areas, up to 15 m2g-1, due to sintering and particle agglomeration at high calcination temperatures[74,214-217]. Limited surface areas restrict the dispersion of active sites and decrease catalytic activity. Recent studies highlighted when BET surface area of CaO-based catalysts higher than
Impact of surface area on biodiesel yield for CaO-based catalysts
| Catalyst type | Source of CaO | SBET, m2g-1 | Preparation or modification technique | Biodiesel yield, % | Ref. |
| CaO | Shell | 40 | Grinding | 95.84 | [223] |
| Mo-CaO | Commercial | 44.2 | Wet impregnation and calcination | 99 | [222] |
| K2O-CaO | Eggshell | 46.3 | Calcination, hydration-dehydration-incorporation and calcination | 87.9 | [228] |
| CaO-CeO2 | Ca(NO3)2·4H2O | 47.6 | Gel-formation via a co-precipitation and calcination | 97.6 | [229] |
| CaO-biochar | Eggshells | 50.22 | Calcination, doping and annealing | 99 | [231] |
| CaO | Chicken eggshell Ostrich eggshell | 54.6 71.0 | Grinding and calcination | 94 96 | [235] |
| CaO | Ca(NO3)2·4H2O | 67.781 | Sol-gel method and calcination | 97.61[b] | [224] |
| CaO | Mud clam shell | 68.57 | Grinding, calcination, hydrothermal treatment, calcination | 96.7 | [225] |
| CaO-BrC8H17 | Commercial | 68.6 | Mixing and vacuum drying | 99.5 | [226] |
| CaO | Shell | 69.738 | Grinding, reprecipitation in magnetic field and calcination | 96.7 | [227] |
| CaO-SiO2 | Eggshell | 79.8 | Grinding, impregnation and calcination | 96 | [232] |
| CaO | CaCl2 | 80.69 | Precipitation and calcination | 97 | [233] |
| CaO-Al2O3 | Ca(OAc)2 | 82.74 | Mixed, boiling and calcination | 98.64 | [234] |
| CaO-SiO2 | Commercial | 83.048 | Mixing and calcination | 93.77 | [41] |
| CaO-zeolite | Eggshell | 83.6 | Grinding, wet impregnation and calcination | 93.7 | [230] |
| CaO | Mussel shell Cockle shell Scallop shell | 89.91 59.87 74.96 | Calcination and crushed | 95.90 94.13 95.44 | [236] |
| CaO | Commercial | 90 | NR | 100[c] | [237] |
| CaO-MOF | Ca(OAc)2 | 90.01 | Impregnation and calcination | 95.09 | [238] |
| CaO | Seashell | 90.61 | Calcination, hydration-dehydration, calcination | 98.54 | [239] |
| CaO | Razor shell | 92.63 | Calcination, hydro aeration-dehydration, calcination | 98.8 | [240] |
| CaO | Cow teeth | 124.77 | Calcination, hydration-dehydration, calcination | 98.7 | [241] |
| CaO | CaCl2 | 97.63 | Sonication assisted reverse emulsion-based synthesis | 89.89[c] | [242] |
| Ce-CaO-SiO2 | Eggshell | 108.35 | Impregnation, calcination, doping and calcination | 95.29 | [243] |
| KF/CaO | Commercial | 109 | Impregnation and calcination | 96.8 93.7 | [244] [245] |
| KF/CaO-MgO | CaCl2 | 113.8 | Coprecipitation, calcination, isochoric impregnation, calcination | 95 | [246] |
| KOH/CaO | Ca(NO3)2·4H2O | 113.7 | Sol-gel method and calcination | 99 | [247] |
| CaO-zeolite/Fe3O4 | Eggshell | 135.31 | Calcination, wet impregnation and mixed | 96.91 91 | [248] [249] |
| CaO-zeolite | Ca(OAc)2 | 148 | Wet impregnation, drying and calcination | 93 | [250] |
| CaO-zeolite | Ca(NO3)2·4H2O | 219 | Wet impregnation and calcination | 91.1 | [251] |
| CaO-zeolite-CTAB | Eggshell | 225 | Grinding, calcination, wet impregnation, drying and calcination | 89 | [252] |
| CaO-Na-zeolite | Crab shell | 370 | Grinding, calcination, wet impregnation, drying and calcination | 95 | [253] |
| CaO/C | Ca(OAc)2 | 369.66 | Precursor synthesis and calcination | 97.37 | [259] |
| CaO/C | Ca(NO3)2·4H2O | 232.2 | Wet impregnation and calcination | 98.2 | [260] |
| CaO/C(magnetic) | Ca(NO3)2·4H2O | 274.18 | Wet impregnation, drying and calcination | 93.2 | [261] |
| CaO/Graphene oxide | Ca(NO3)2·4H2O | 437 | Wet impregnation, evaporation and calcination | 98.3 | [262] |
| CaO/C | Ca(NO3)2·4H2O | 596 | Wet impregnation, evaporation and calcination | 98 | [263] |
| CaO/C | Ca(NO3)2·4H2O | 777.7 | Wet impregnation, drying and calcination | 98.4 | [264] |
| CaO/Al2O3 | CaCl2 | 319 | Impregnation and calcination | 96.68 | [254] |
| Ce-CaO-MCM-41 | Ca(NO3)2·4H2O | 321 | Sono-dispersion, drying and calcination | 96.8[c] | [255] |
| Zr-CaO-MCM-41 | Ca(NO3)2·4H2O | 350 | Impregnation, ultrasound dispersion, drying and calcination | 88.5[c] | [256] |
| CaO-NaY zeolite-Fe3O4 | Commercial | 359 | Mixed, grinding, ultrasonicated, evaporation, drying and calcination | 95.37 | [257] |
| CaO-CeO2-SBA-15 | Ca(NO3)2·4H2O | 590 | Wet impregnation, drying and calcination | 87.1 | [258] |
40 ≤ SBET < 90
According to the literature data, moderate increase of CaO surface area at the 40-90 m2g-1 range can significantly improve catalytic efficiency in biodiesel synthesis. For example, molybdenum impregnation of commercially available CaO followed by calcination at 700 °C increased surface area more than eleven-fold, from a few m2g-1 to 44.2 m2g-1, resulting in biodiesel yields exceeding 99%[222]. Interestingly, further Mo loading (5 wt%) decreased the surface area due to pore blockage, illustrating the delicate balance of textural properties and active site accessibility. CaO derived from seashells with SBET 40 m2g-1 was used to produce biodiesel in 95.84% yield from WCO using a solar-heated parabolic trough collector and a parabolic trough collector as an intensifier[223]. Sol-gel-derived nano-CaO with 67.781 m2g-1 surface area provided biodiesel in 97.61% yield from soybean oil[224], while clam shell-derived CaO (68.57 m2g-1) achieved 96.7% yield (compared to 93% for commercially available CaO)[225]. Similar BET specific surface area were obtained in the related studies: 68.6 m2g-1 for chemically modified CaO by chemical addition of bromooctane to the catalyst surface[226] and 69.738 m2g-1 for CaCO3 reprecipitated in magnetic field from shells and, then, calcined[227]. In both cases, under optimized conditions, the biodiesel yield was high and reached 99.5% and 96.7%, respectively.
Hydration-dehydration treatment represents another efficient approach. A simple modification of shell-derived CaO increased surface area to 55.1 m2g-1, while subsequent doping with KI maintained enhanced catalytic activity despite a gradual decrease of SBET with increasing KI content (from 46.3 to
Mixed oxide and hybrid composites also benefit from higher surface areas. A CaO-CeO2 catalyst with a 1:1 molar ratio with surface area of 47.6 m2g-1 - almost nine times higher than pure CaO - provided 97.6% FAME yield, due to the combination of strong alkalinity and improved dispersion of basic sites[229]. A biochar-based catalyst loaded with 30 wt% CaO with BET surface area of 83.048 m2g-1 was successfully involved in the transesterification of palm oil, providing biodiesel in 93.77% yield[41]. The catalyst was prepared by pyrolysis and KOH activation of dried municipal sludge, subsequent loading with CaO, and calcination at 700 °C. Comparable surface area values of 83.6 m2g-1 were obtained for a composite catalyst from eggshell and zeolite prepared by wet impregnation followed by calcination[230]. Lower SBET values (50.2 m2g-1) for biochar doped with CaO nanocatalyst were reported in the other study; however, the conversion of algal oil to biodiesel in 99% yield was observed[231]. The eggshell CaO catalyst [Figure 2A] supported on SiO2 [Figure 2B] demonstrated a high surface area of 79.8 m2g-1 compared to pure CaO (2.8 m2g-1) and was uniformly distributed over the silica surface [Figure 2C][232]. Under optimal reaction conditions, the biodiesel yield from waste cooking oil was 96%. The low CaO/SiO2 surface area after the reaction was due to the covering of the catalyst surface with oil (as shown in Figure 2D).
Figure 2. SEM images of (A) CaO; (B) SiO2; (C) CaO/SiO2 (fresh); (D) CaO/SiO2 (used). Reproduced from ref.[232] under a Creative Commons Attribution 3.0 Unported License (CC BY 3.0).
Precipitation and calcination-derived nano-CaO from calcium chloride with surface area of 80.7 m2g-1 produced biodiesel in 97% yield under mild conditions[233], while CaO-Al2O3 composites with 82.74 m2g-1 value, prepared by mixing calcium acetate solution with aluminum oxide, boiling to remove water and calcining at 718 °C, provided 98.64% biodiesel from palm oil[234].
Natural precursors demonstrated intrinsic variations. For instance, ostrich eggshell-derived CaO demonstrated a larger SBET (71.0 m2g-1) than that for obtained from chicken eggshells (54.6 m2g-1)[235]. The biodiesel was obtained in 94% and 96% high yield in both cases. In the related studies, mussel, cockle, and scallop shells calcined at 1000 °C with different surface areas of 89.91, 59.87, and 74.96 m2g-1, respectively, provided biodiesel in 95.90%, 94.13%, and 95.44% yields from palm oil[236].
90 ≤ SBET ≤ 150
Nanocrystalline CaO with a crystallite size of 20 nm and specific surface area of 90 m2g-1 demonstrated excellent catalytic activity, resulting in complete conversion of soybean oil for 12 h, which was challenging using commercially available CaO with a crystallite size of 43 nm and SBET of 1 m2g-1 (conversion was 2%)[237]. Comparable values were obtained for impregnated CaO on a magnetic metal-organic framework
Using the reverse emulsion method with ultrasonic treatment increased the specific surface area of CaO by 1.53 times (to 97.63 m2g-1), resulting in 18.81% increase in oil conversion (to 89.89%) compared to commercially available CaO-catalyst under the same operating conditions[242]. An interesting approach involved synthesis of a catalyst from waste glass and eggshells doped with CeO2 was developed to form highly active catalytic sites for transesterification[243]. The resulting catalyst with BET specific surface area of
Magnetic CaO-zeolite(ZSM-5)/Fe3O4 catalyst with SBET 135.31 m2g-1 was used in the transesterification of waste cooking oil in standard and magnetic reactors with 93.64% and 96.91% biodiesel yields, respectively[248]. After preparing the required CaO (calcination from chicken eggshell) and zeolite, CaO particles were deposited on the desilicated zeolite surface using the wet impregnation method, resulting in a CaO-zeolite catalyst, which was mixed with silicon-coated Fe3O4 to form the desired magnetic catalyst. Later, the same research group evaluated the effect of varying the CaO and Fe3O4 ratio during catalyst synthesis on its catalytic performance[249]. In a related study, high BET surface area of 148 m2g-1 was achieved with 35% CaO loading (from Ca(OAc)2) on ZSM-5 zeolite using wet impregnation and subsequent calcination, resulting in biodiesel in 93% yield from refined sunflower oil[250].
SBET > 150
CaO-based catalysts supported on ammonium and hydrogen forms of ZSM-5 zeolites with various Si:Al ratios by impregnation were successfully used in the transesterification of rapeseed oil with methanol[251]. The most active catalyst was 10% CaO/ZSM-5 (Si:Al = 50) with surface area of 219 m2g-1, providing FAME in 91.1% yield at 220 °C. Similar SBET values of 225 m2g-1 were determined for CaO-ZSM-5 obtained by desilication using a surfactant CTAB[252]. The high mesoporosity of the zeolite support obtained by this method resulted in high dispersion of CaO particles within the pores. The CaO/ZSM-5-CTAB catalyst demonstrated performance with 89% biodiesel yield due to strong acidic and basic properties combined with large surface area. Interestingly, in the related study, activated CaO from waste crab shells, wet-impregnated onto Na-ZSM-5 (15% CaO), yielded a catalyst with an SBET of 370 m2g-1, which provided biodiesel in 95% yield under the transesterification of neem oil with methanol at 75 °C[253]. Among other developments, advances were accomplished in CaO impregnation on Al2O3 supports (319 m2g-1)[254], a series of studies on CaO impregnation of Ce- and Zr-doped mesoporous silica (MCM-41) supports (321 and 350 m2g1, respectively)[255,256], CaO impregnation of magnetic NaY zeolite (359 m2g-1)[257], and Ca-doped Ce-incorporated SBA-15 mesoporous catalyst (590 m2g-1)[258]. An efficient approach to increase the BET specific surface area of CaO-based catalyst was in the use of carbon as a support. CaO anchored in carbon nanosheets increased the microwave absorption capacity of the catalyst and achieved SBET of 369.66 m2g-1[[259]. The maximum oil conversion reached 97.37% at 60 °C for 30 min in 400 W microwave field.
In another case, powdered and calcined date pits were used as a carbon source for CaO support, resulting in the catalyst surface area of 232.2 m2g-1 and 98.2% biodiesel yield[260]. Magnetically activated carbon was derived from almond shells and modified with CaO with high surface area (274.18 m2g1), basicity
The review of CaO-based catalysts with increased BET surface areas demonstrated that surface increase higher ≈40 m2g-1 generally boosted biodiesel yield, exceeding 95%. However, no direct correlation between SBET and conversion was identified: catalysts with moderate values (50-80 m2g-1) frequently reach yields comparable to catalysts with very high surface areas (> 300 m2g1). Thus, while sufficient porosity improves accessibility of active sites, the overall performance is also strongly impacted by basicity, dopants, and the stability of catalyst structure. In future, research on CaO-based catalysts will likely focus on fine-tuning pore architecture, ensuring long-term keeping of surface area under reaction conditions, and developing synthesis routes that combine high SBET with cost efficiency and scalability.
Catalyst reusability
Catalyst reusability is a crucial value in assessing the practical applicability and cost-effectiveness of CaO-based catalysts in biodiesel production. Numerous studies have demonstrated that the the stability of CaO-based catalysts tends to decrease with increasing cycle number due to active site leaching, surface poisoning, or structural degradation, which limits the use of a catalyst in industrial applications. To address this issue, various reusable catalyst set ups were actively developed. This section presents a review on CaO-based catalysts successfully reused for at least six cycles. Particular attention was paid to catalyst modification, support incorporation, regeneration methods, and performance decreasing observed after reuse. The achievements and results discussed in this chapter were summarized in Table 5.
Reusability of CaO-based catalysts: number of reuse cycles and yield decrease
| Catalyst type | Source of CaO | Reuse, number of cycles | Catalyst recovery/regeneration between cycles | Biodiesel yield, % 1st cycle → last cycle | Ref. |
| Sr-CaO | Ca(NO3)2·4H2O | 6 | Filtration, washing (methanol), drying at 120 °C and calcination at 600 °C | 99.2 → 49.4 | [265] |
| CaO | Eggshell | 6 | Filtration, washing (methanol) and calcination at 600 °C | 94.52 → 90[c] | [267] |
| CaO | Snail shell | 7 | Centrifugation, washing (distilled water), drying at | 96.1 → 82 | [268] |
| CaO-fiber mats | Commercial | 7 | Without any treatment | 5.63 → 7.79[a] | [273] |
| Sr-CaO | Ca(NO3)2·4H2O | 7 | Filtration and used without any treatment | 98.31 → 92.25 | [266] |
| MgO-CaO ZnO-CaO | Ca(NO3)2·4H2O | 6 | Centrifugation, washing (hexane, methanol), drying and calcination at 800 or 900 °C | 83 → 77[a] 81 → 73[a] | [269] |
| ZnO-CaO | Eggshell | 6 | Filtration, washing (hexane) and calcination at 900 °C | 94.5 → 70 | [59] |
| CaO-SiO2 | Eggshell | 10 | Extraction, washing (methanol) and calcination at 800 °C | 90 → 52.5 | [284] |
| CaO | Freshwater mussel shell | 7 | Filtration and used without any treatment | 97.5 → 90 | [67] |
| CaO | Waste filter press cake | 7 | NR | 96 → 90 | [270] |
| CaO | CaSO4 | 7 | Centrifugation, washing (hexane), drying at 65 °C (overnight), and calcination at 500 °C | 95 → 65 | [271] |
| CaO | Commercial | 8 | Centrifugation | 94 → 81 | [272] |
| CaO | Ostrich eggshell | 8 | Washing (methanol and tetrahydrofuran) and drying | 92.7 → 80 | [274] |
| CaO | Snail shell | 8 | Centrifugation, washing (distilled water), drying 100 °C and calcination at 900 °C | 98 → 91 | [275] |
| CaO-SiO2 | Goat bones Cockle shell | 8 | Separation, washing (hexane) and drying at 60 °C | 94 → 38[a] 85 → 25[a] | [276] |
| K+-CaO | Eggshell | 8 | Filtration, washing (hexane), drying at 105 °C and calcination at 900 °C | 98.48 → 68.52 | [277] |
| СeO2-CaO | Ca(NO3)2·4H2O | 9 | Centrifugation, washing (with a mixed solution of methanol and heptane), and drying | 98 → 80 | [278] |
| KF-CaO | Clam shell | 9 | Filtration and calcination | 95.77 → 90 | [279] |
| CaO | Commercial | 10 | Centrifugation, washing (methanol) and drying at 50 °C | 97.4 → 86.7 | [280] |
| nano-CaO | Commercial | 10 | Centrifugation | 98 → 90 | [281] |
| CaO-Fe3O4 | CaCl2 | 10 | Magnetic separation, washing (isopropanol) and drying | 99 → 70 | [283] |
| Au-CaO | Calcite Mussel shell Eggshell CaO Dolomite | 10 | Filtration, washing (methanol) and drying | 96.5 → 89.5[a] 96 → 89[a] 96 → 89.5[a] 95 → 85[a] 94 → 86[a] | [285] |
| porous CaO | Ca(NO3)2·4H2O | 10 | Centrifugation, washing (methanol and tetrahydrofuranׄ) and drying at 50 °C | 97.2 → 71.8 | [286] |
| CaO-MgO-Al2O3 | Limestone | 10 | Washing (methanol), centrifugation and drying | 97.1 → 90 | [287] |
| CaO | Mud crab shell | 11 | Washing (methanol) and calcination at 900 °C | 98.8 → 96.5[f] | [288] |
| CaO | Eggshell | 11 | Filtration, washing (methanol) and calcination at | 95.05 → 90 | [289] |
| CaO-SiO2 | Eggshell | 12 | Filtration, washing (tetrahydrofuran) and drying at | 78 → 65 | [292] |
| Na2CaSiO4-CaO | Ca(NO3)2·4H2O | 12 | NR | 98.2 → 57.3 | [291] |
| CaO | Eggshell | 13 | NR | 95 → NR | [290] |
| CaO-SiO2 | Eggshell | 14 | NR | 97.8 → 95 | [293] |
| KF/CaO-Fe3O4 | Unspecified | 14 | Magnetic extraction, washing (methanol) and drying at 105 °C | 95 → 85 | [294] |
| NaCl/CaO | Commercial | 15 | Washing (acetone) and drying at 60 °C | 97.1 → 90 | [295] |
| CaO | Commercial | 20 | Filtration | 95 → 85 | [282] |
| CaO-fly ash | Eggshell | 16 | NR | 96.97 → NR | [296] |
| CaO/α-Fe fibers | Ca(NO3)2·4H2O | 20 | Magnetic extraction and washing | 95.7 → 85.2 | [297] |
From 6 to 9 reuse cycles
The calcium-strontium oxide nanocatalyst demonstrated moderate stability over 6 cycles[265]. Conversion efficiency was approximately constant in 3 cycles and then decreased to 49.4% in the 6th reaction cycle. The decrease in activity was due to leaching and agglomeration, which decrease the number of active sites on the surface of the catalyst. In another study, a Sr doped CaO catalyst provided conversion of more than 92.25% at the 6th cycle[266]. Calcium oxide obtained by simple calcination of shells (eggshells, snail shells or freshwater mussel shell) was successfully reused in 6-7 reaction cycles[67,267,268]. Mixed CaO-MgO and CaO-ZnO catalysts were reused up to four cycles, maintaining 80% biodiesel yields[269]. Interestingly, by the 6th reaction cycle, conversion for the mixed catalyst was higher than 20% compared to pure CaO. In another case, for a catalyst obtained from eggshells and mixed with ZnO, the conversion of palm oil waste in the 6th cycle was 70%[59]. CaO NPs obtained from waste filter press cake at a sugar manufacturing facility, hydrated with a surfactant, maintained stable catalytic performance over 7 reaction cycles, providing biodiesel in 90% yield[270]. Green CaO NPs were efficiently recycled for up to 5 consecutive cycles with insignificant changes in biodiesel yield[271]. However, biodiesel yield decreased to 65% in the 7th reaction cycle due to precipitation of organic matter present in a transesterification reaction mixture. Poisoning of the active sites of CaO NPs resulted in shrinkage of the particle surface area and pore capacity, decreasing the catalytic activity. A commercial sample of CaO, pre-degassed at 973 K, demonstrated catalytic activity during 8 cycles of transesterification of sunflower oil to biodiesel, with a yield decrease from 94% to 81%[272].
An intriguing approach for CaO modification involved physical or chemical (via dopamine bonds) immobilization on polymer support of polyacrylonitrile (PAN) fibers produced by electrohydrodynamic processing (also known as electrospinning or electrospraying)[273]. The synthesized PAN fibers were homogeneous and uniform, and CaO catalyst particles were successfully retained within or bound to the fiber surface. CaO particles mechanically entrapped in PAN fibers could be reused for 7 reaction cycles, while the chemically immobilized catalyst failed in reuse studies. Unfortunately, the biodiesel fuel yield by cycles were not provided, which would provide better understanding of the stability of the proposed catalyst compared to an assessment of the kinematic viscosity of the samples.
Different shell and bone-derived CaO catalysts with various catalytic activity over 8 reaction cycles: from ostrich eggshells with a decrease in yield from 92.7% to 80%[274], from snail shells from 98% to 91%[275], silica-supported CaO from waste goat bones and cockle shells with yield from 94% to ca. 38% and from 85% to ca. 25%, respectively[276], and potassium-impregnated CaO from eggshells with a decrease in biodiesel yield from 98.48% to 68.52%[277]. Notable results for catalyst reuse were obtained for various CeO2-doped CaO catalysts [Figure 3][278]. The CeO2-CaO catalyst obtained by wet impregnation demonstrated superior stability compared to pure CaO, although the FAME yield ultimately decreased by 50% after the 6th cycle. For pure CaO, the biodiesel yield rapidly decreased, reaching less than 30% by the 4th cycle.
Figure 3. The reusability studies of the CaO, CeO2@CaO and CeO2-CaO catalysts. Reproduced from ref.[278] under a Creative Commons Attribution 3.0 Unported License (CC BY 3.0).
On the other hand, the core-shell CeO2@CaO catalyst sample obtained by the hydrothermal method demonstrated the most stable catalytic activity, maintaining the FAME yield higher than 80% even after 9 cycles. The KF-impregnated CaO catalyst from clam shells demonstrated more stable catalytic activity over 9 cycles of reuse, with the decrease in biodiesel yield from 95.77% to 90%[279].
10 and more reuse cycles
Commercially available CaO was successfully used in microwave-assisted transesterification of non-edible Ceiba pentandra seed oil during 10 reaction runs, decreasing biodiesel yield from 97.4 to 86.7%[280]. A similar decrease in biodiesel yield to 90% after 10 reaction cycles was observed for commercially available CaO nanocatalysts with BET surface areas of 22.25 and 89.52 m2g-1, respectively; however, the catalysts were not subjected to additional preparation steps from cycle to cycle[281]. Interestingly, in another study, commercially available CaO pre-activated by methanol with 2.03% water in the first use demonstrated excellent reuse results, with biodiesel yield decreasing from 95 to 85% by the 20th cycle[282]. The nanometer magnetic solid base catalyst prepared by loading CaO on Fe3O4 with Na2CO3 and NaOH as precipitators, respectively, demonstrated longer lifetime than pure CaO[283]. After 5 runs, the FAME yield for the mixed CaO-Fe3O4 catalyst was over 90%, and after 10 runs, FAME conversion reached over 70%. However, for CaO under the same conditions, FAME conversion dropped to 22% after just 5 runs, and after the 8th reaction cycle, a complete loss of catalytic activity was observed. Furthermore, after the reaction, a CaO catalyst was virtually invisible in a reactor, as large amount of CaO dissolved in methanol or formed colloidal suspensions with methanol in the upper liquid and was discharged with the reaction products. CaO obtained from eggshell waste and supported on silica from rice husk demonstrated superior catalytic performance in biodiesel production compared to non-modified CaO[284]. The catalyst was used in 10 reaction cycles, with a slight decrease in biodiesel yield from 90% to 85%, 80%, and 77.5% at the 5th, 6th, and 7th cycles. A significant decrease in catalytic activity was observed at the 8th and subsequent cycles, when biodiesel yield dropped to 52.5%. The decrease in biodiesel yield from the 5th to 10th cycles was presumably due to the leaching of the Ca compound under reaction conditions.
In related studies, 10 cycles of catalyst runs were found for Au NPs-doped CaO from various precursors[285]. Porous CaO was synthesized using fast mineralization process induced by sodium poly(styrenesulfonate) followed by calcination[286], and CaO derived from limestone and mixed with MgO and Al2O3[287]. CaO catalyst from waste mud crab shells calcined at 900 °C provided 11 cycles of reuse in the production of biodiesel from palm oil with purity higher than 96.5%[288]. Similar catalyst recycling was later observed for CaO from eggshells calcined at 900 °C in the transesterification of waste frying oil[289]. The biodiesel yield decreased slightly by the 11th reaction cycle, from 95.05% to 90%. Interestingly, unlike other research studies, the comparative commercially available CaO sample demonstrated comparable catalytic activity in the 1st reaction cycle, with 95.66% FAME yield. Unfortunately, no information was available on the recycling of the commercially available catalyst or a comparison of the data with eggshell-derived CaO. Interestingly, in the other study, the eggshell-based catalyst demonstrated efficient performance over 13 reuse cycles without a quantitative decrease in biodiesel yield[290].
A silicate-strength strategy was developed to synthesize a solid base catalyst[291]. The catalyst precursor was calcined at 1,050 °C to form two main catalytic phases, Na2CaSiO4 and CaO, which demonstrated a synergistic effect, ensuring high catalyst activity and good reusability [Figure 4].
Figure 4. Structure and catalytic centers of Na2CaSiO4-CaO catalyst. Reproduced from ref.[291] under a Creative Commons Attribution 4.0 International License (CC BY 4.0).
Biodiesel yield was 94.3% and 57.3% even after 6 and 12 consecutive reuse cycles. Despite slightly lower biodiesel yields in the 1st cycle (ca. 78%) for the CaO-SiO2 catalyst synthesized via the biomimetic silicification approach, using eggshell and Na2SiO3 as raw materials, compared to other studies, the proposed catalyst demonstrated minor deactivation by the 12th cycle, providing biodiesel in 65% yield[292]. In another case, SiO2 NPs obtained from rice husks were chemically bonded to the surface of CaO, resulting in 97.8% yield of FAMEs from WCO, significantly exceeding 83.5% yield for nonmodified CaO under the same reaction conditions[293]. Modification of CaO resulted in well-dispersed CaO with a relatively small particle size and high surface area. Even after 14 cycles of catalyst reuse, the biodiesel yield remained stable at 95%.
The KF/CaO-Fe3O4 nanomagnetic catalyst prepared by a simple impregnation maintained constant catalytic activity even after 14 repeated runs (activity decreased by 10%)[294]. Notably, catalyst recovery by the 14th run was over 90%. It was found that the initial oil transesterification rate gradually decreased with each reaction cycle due to the loss of active components under reaction conditions and separation process. Commercially available CaO was modified with various sodium salts (Na2WO2, Na2CO2, Na2SO2, and NaCl), and NaCl/CaO and demonstrated the best catalytic activity for the production of glycerol-free biodiesel using 3 components: rapeseed oil, dimethyl carbonate (DMC), and methanol as reactants[295]. The biodiesel yield was 97.1% after
According to the literature data on CaO-based catalyst reusability, the performance of both pure and modified catalysts remained highly variable and dependent strongly on the conditions of regeneration and reuse. Commercially available CaO and shell-derived catalysts maintain high activity for several cycles, yet in many cases demonstrating rapid deactivation after the fifth or sixth reuse due to leaching of Ca species or surface contamination by organic residues. Surprisingly, complicated composites and doped catalysts designed to enhance stability demonstrate moderate results; complicated catalysts demonstrated faster loss of active sites or partial pore blockage, resulting in lower yields compared with simpler CaO preparations.
Catalyst deactivation during repeated transesterification cycles is mainly attributed to several mechanisms, including Ca2+ leaching into a reaction mixture as calcium diglyceroxide; surface poisoning by free fatty acids and formation of soap; formation of carbonate or hydroxide species due to CO2 and H2O consumption. Accordingly, various regeneration strategies have been proposed, including solvent washing to remove surface deposits, thermal treatment or recalcination to restore basic sites, and structural stabilization via supports or promoters to suppress leaching and sintering, highlighting the need for tailored regeneration approaches for long-term catalyst operation. A recent study demonstrated significant differences in CaO-Fe3O4 catalyst recovery depending on the chosen regeneration method[298].
Overall, maintaining catalytic activity over multiple cycles requires not only structural stabilization, but also reproducible and gentle regeneration procedures that prevent irreversible surface changes and calcium leaching. These findings indicate, that catalyst reusability is governed by the interplay between deactivation mechanisms, catalyst composition, and regeneration conditions, rather than by catalyst complexity alone.
INTEGRATED ANALYSIS OF DEVELOPMENT AND IDENTIFICATION OF INTERRELATIONS
Calcium sources can be classified in carbonate-based (e.g., mollusk shells, eggshells, limestone, calcium-containing ash) and hydroxide-based (e.g., carbide sludge). Calcination of carbonate materials proceeds via the release of 0.44 ton of CO2 per 1 ton of CaCO3 decomposed, which corresponds to ≈0.79 ton of CO2 per 1 ton of CaO produced, thereby increasing the carbon footprint of the process (Equation 1).
In contrast, hydroxide-based sources yield ≈0.32 ton of H2O per 1 kg of CaO produced with no direct CO2 release from the calcination process (Equation 2).
As shown in Figure 5, no clear correlation of catalyst loading and biodiesel yield could be notified for CaO-based catalysts. Data points are widely scattered, indicating a nonlinear effect of catalyst loading on transesterification efficiency and strong dependence on other values: feedstock composition, alcohol-to-oil molar ratio, reaction temperature, and mixing conditions. In many studies, FAME yields higher than 90% were achieved over a wide range of catalyst loadings (0.5-3 wt%), suggesting that catalytic activity was determined primarily by intrinsic catalyst properties (e.g., base site density, surface area, and structural stability) rather than by the absolute amount of a catalyst.
Figure 5. Correlation of catalyst loading (wt%) and biodiesel yield (%) for CaO-based catalysts reported in literature.
As shown in Figure 6, no clear correlation was found for the BET surface area of CaO-based catalysts and biodiesel yield. Although a higher surface area is generally expected to increase the number and accessibility of active basic sites, the data reviewed demonstrate a wide range of FAME yields for catalysts with similar SBET values. The effect of surface area cannot be considered independently of catalyst loading and porosity. Catalysts with very high SBET often require lower loadings, which can lead to mass transfer limitations during transesterification. Conversely, catalysts with a lower surface area but higher basicity or stronger active sites can still demonstrate superior performance.
As shown in Figure 7, the decrease in biodiesel yield with increasing recycle rate demonstrates a generally negative trend: most CaO-based catalysts exhibited a 10%-30% activity loss after 6-10 reaction cycles. Some catalysts maintained stable performance (yield decrease < 5%), which typically corresponds to modified or supported catalysts with less Ca2+ leaching and improved structural integrity. The data suggest that, although the initial catalytic activity of CaO-based materials may be high, long-term stability remains principal limitation. The sharp deactivation observed in some cases (loss of up to 60% after 8 cycles) is generally attributed to the combined effects of Ca2+ leaching, surface carbonization, and pore clogging by reaction intermediates. In contrast, catalysts stabilized by doping (e.g., CeO2, KF) or immobilization on inert supports (SiO2, Fe3O4, carbon) demonstrate longer recycling and more gradual decline in activity.
A comprehensive analysis of CaO-based catalysts activity for biodiesel production revealed that the impact of catalyst loading, surface area, and reusability is nonlinear and depends on a variety of experimental conditions. Consequently, significant potential to achieve outstanding results remains in the development of highly efficient catalysts for biodiesel production.
INDUSTRIAL APPLICATION CHALLENGES AND SCALING ISSUES FOR CaO-BASED CATALYSTS
Despite the extensive laboratory studies demonstrating the high catalytic activity of CaO-based catalysts for biodiesel production, their industrial implementation remains limited by a number of unresolved engineering and technological challenges. One of the most critical issues is the limited stability and reproducibility of CaO-based catalysts under real-world operating conditions. Some studies report significant leaching of active Ca2+ species, leading to rapid catalyst deactivation over multiple cycles and contamination of the biodiesel product, complicating catalyst reuse and quality control in large-scale operations[32,68,299]. The low stability of CaO is due to the high sensitivity to moisture and atmospheric CO2, leading to Ca(OH)2 and CaCO3 formation, blocking the main active sites and reducing catalytic activity[300-304]. This inherent susceptibility imposes selected requirements on catalyst handling, storage, and pretreatment, which may be impractical or uneconomical at industrial scale.
From a reactor engineering perspective, CaO-catalyzed transesterification is limited by mass transfer resistance due to the three-phase nature of the reaction system (solid catalyst, oil, and alcohol)[305,306]. Insufficient dispersion of methanol in the oil and limited accessibility of active sites often result in reduced apparent reaction rates under continuous or scaled-up conditions, even when catalysts demonstrate high intrinsic activity in laboratory batch experiments. These limitations highlight the need to optimize reactor configurations and improve mixing strategies to ensure efficient continuous processing.
Besides technical challenges, process economics and operational durability play a crucial role in determining industrial feasibility. Although CaO is an inexpensive and abundant source, the overall process cost is highly dependent on catalyst stability, energy consumption for high-temperature regeneration, wastewater generation under product purification, and feedstock variability. On the other hand, the solid nature allows the catalyst to be easily separated from a reaction mixture, regenerated and potentially reused, minimizing further processing costs[31]. Overall, these challenges demonstrate that the successful industrialization of CaO-based catalysts requires a systematic approach integrating catalyst stabilization, reactor design, mass transfer optimization, and feasibility assessment.
Although CaO-based catalysts are frequently described as sustainable due to their low cost and the availability of waste-derived sources, a rigorous assessment of their full life-cycle environmental and economic impacts remains limited. A comprehensive life cycle assessment (LCA) of CaO catalysts requires detailed, process-specific data on precursor pretreatment, calcination energy demand, CO2 emissions, catalyst lifetime, regeneration efficiency, and end-of-life management, which are often unavailable, fragmented, or highly system-dependent. In particular, the energy-intensive calcination step and the variability in catalyst durability across different CaO sources can significantly impact the overall carbon footprint and economic feasibility. Therefore, while qualitative comparisons suggest that waste-derived CaO catalysts may offer advantages in terms of resource efficiency and waste valorization, a quantitative evaluation of their environmental and economic performance should be addressed in dedicated techno-economic and LCA studies. Such analyses are essential for aligning CaO-based catalyst development with carbon neutrality goals and industrial sustainability criteria, but fall beyond the scope of the present review.
CONCLUSION
This review systematically examined the performance of CaO-based catalysts for biodiesel production in the context of carbon-neutral and low-emission fuel technologies. Biodiesel represents a nearly closed CO2 cycle, as the carbon released under combustion is balanced by the CO2 consumed by biomass. Replacing fossil-derived diesel with biodiesel therefore mitigates net CO2 emissions and contributes to decreasing the overall carbon footprint of transportation fuels. Consequently, improving catalyst durability, efficiency, and recyclability directly supports the development of sustainable, low-carbon biodiesel production systems.
Several forward-looking research directions can be identified, including data-driven catalyst design to rationalize complex composition-structure-performance relationships, process intensification and reactor integration for stable long-term operation, catalyst stabilization and regeneration to suppress Ca2+ leaching and carbonation while maximizing reuse cycles, and systematic techno-economic and life-cycle assessments aligned with carbon-neutral and low-emission fuel targets.
DECLARATIONS
Authors’ contributions
Investigation, visualization, formal analysis, writing - original draft: Potorochenko, A. N.
Conceptualization, writing - original draft, writing - review & editing: Rodygin, K. S. T.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
The authors thank Saint Petersburg State University for the financial support (Pure ID: 116857908).
Conflicts of interest
Both authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Nunes, L. J. R. The rising threat of atmospheric CO2: a review on the causes, impacts, and mitigation strategies. Environments 2023, 10, 66.
2. Chenxiao, Y.; Xiaoguang, Y.; Jiantao, M.; Sijin, L. A systematic review of urban road traffic CO2 emission models. Carbon. Footprints. 2025, 4, 17.
3. Rothenberg, G. A realistic look at CO2 emissions, climate change and the role of sustainable chemistry. Sustain. Chem. Climate. Action. 2023, 2, 100012.
4. Rui, W.; Yuzhong, Z.; Shuang, Z.; Xinlu, W. Open-data-based city-scale gridded carbon dioxide emission inventory: supporting urban carbon monitoring in Chengdu, China. Carbon. Footprints. 2024, 3, 14.
5. Li, G.; Yao, J. A review of algae-based carbon capture, utilization, and storage (Algae-Based CCUS). Gases 2024, 4, 468-503.
6. Wan Osman, W. N. A.; Rosli, M. H.; Mazli, W. N. A.; Samsuri, S. Comparative review of biodiesel production and purification. Carbon. Capture. Sci. Technol. 2024, 13, 100264.
7. Suhara, A.; Karyadi, .; Herawan, S. G.; et al. Biodiesel sustainability: review of progress and challenges of biodiesel as sustainable biofuel. Clean. Technol. 2024, 6, 886-906.
8. Naseef, H. H.; Tulaimat, R. H. Transesterification and esterification for biodiesel production: a comprehensive review of catalysts and palm oil feedstocks. Energy. Convers. Manag. X. 2025, 26, 100931.
9. Yusuf, B. O.; Oladepo, S. A.; Ganiyu, S. A. Efficient and sustainable biodiesel production via transesterification: catalysts and operating conditions. Catalysts 2024, 14, 581.
10. Gaide, I.; Grigas, A.; Makareviciene, V.; Sendzikiene, E. Life cycle assessment and biodegradability of biodiesel produced using different alcohols and heterogeneous catalysts. Green. Chem. Lett. Rev. 2024, 17, 2394503.
11. Nogales-Delgado, S. Biodiesel production and life cycle assessment: status and prospects. Energies 2025, 18, 3338.
12. Christopher, Selvam. D.; Raja, T.; Nagappan, B.; et al. The role of biodiesel in marine decarbonization: Technological innovations and ocean engineering challenges. Results. Eng. 2025, 25, 103974.
13. K, N. K.; Akash, Deep. B. N.; Birur, Jayanna. V.; et al. Influence of ceramic thermal barrier coating on diesel engine performance using scum oil biodiesel at different compression ratios. Sci. Rep. 2025, 15, 31094.
14. Shanthini, V.; Chitra, D.; Moorthy, G. Biodiesel: a comprehensive review of properties, catalyst types, and feedstock sources. Results. Chem. 2025, 18, 102678.
15. Zheng, F.; Cho, H. M. Study on biodiesel production: feedstock evolution, catalyst selection, and influencing factors analysis. Energies 2025, 18, 2533.
16. Debnath, D.; Whistance, J.; Thompson, W. The U.S. biodiesel outlook: a pathway toward sustainability. Sustainability 2025, 17, 689.
17. Mhetras, N.; Gokhale, D. Sustainable biodiesel production: importance of feedstock resources and production methods. RSC. Adv. 2025, 15, 26739-54.
18. Abdelrahman, A. A.; Abo, El-khair. M. A. Advanced biodiesel production: feedstocks, technologies, catalysts, challenges, and environmental impacts. J. Environ. Chem. Eng. 2025, 13, 114966.
19. Boichenko, S.; Yakovlieva, A.; Zubenko, S.; Shkilniuk, I. Physical, chemical, and performance properties of biodiesel fuels: a comparative study of lipid-based feedstocks. Energies 2025, 18, 4274.
20. Anil, N.; Rao, P. K.; Sarkar, A.; et al. Advancements in sustainable biodiesel production: A comprehensive review of bio-waste derived catalysts. Energy. Convers. Manag. 2024, 318, 118884.
21. El-Araby, R. Biofuel production: exploring renewable energy solutions for a greener future. Biotechnol. Biofuels. Bioprod. 2024, 17, 129.
22. Mandari, V.; Devarai, S. K. Biodiesel production using homogeneous, heterogeneous, and enzyme catalysts via transesterification and esterification reactions: a critical review. Bioenergy. Res. 2022, 15, 935-61.
23. Wang, B.; Wang, B.; Shukla, S. K.; Wang, R. Enabling catalysts for biodiesel production via transesterification. Catalysts 2023, 13, 740.
24. Wang, H.; Zhou, H.; Yan, Q.; Wu, X.; Zhang, H. Superparamagnetic nanospheres with efficient bifunctional acidic sites enable sustainable production of biodiesel from budget non-edible oils. Energy. Convers. Manag. 2023, 297, 117758.
25. Mohiddin, M. N. B.; Tan, Y. H.; Seow, Y. X.; et al. Evaluation on feedstock, technologies, catalyst and reactor for sustainable biodiesel production: a review. J. Ind. Eng. Chem. 2021, 98, 60-81.
26. Nabgan, W.; Jalil, A. A.; Nabgan, B.; et al. Sustainable biodiesel generation through catalytic transesterification of waste sources: a literature review and bibliometric survey. RSC. Adv. 2022, 12, 1604-27.
27. Kibar, M. E.; Hilal, L.; Çapa, B. T.; Bahçıvanlar, B.; Abdeljelil, B. B. Assessment of homogeneous and heterogeneous catalysts in transesterification reaction: a mini review. ChemBioEng. Reviews. 2023, 10, 412-22.
28. Kawashima, A.; Matsubara, K.; Honda, K. Development of heterogeneous base catalysts for biodiesel production. Bioresour. Technol. 2008, 99, 3439-43.
29. He, L.; Zhang, L. L.; Zhou, H.; et al. Boosting photocatalytic upcycling of liquid biomass into biodiesel via microenvironment modulation. Adv. Energy. Mater. 2024, 15, 2403168.
30. He, L.; Chen, L.; Nie, Y.; et al. A practical approach for enhanced biodiesel production using organic modified montmorillonites as efficient heterogeneous hybrid catalysts. Green. Chem. 2024, 26, 5954-65.
31. Changmai, B.; Vanlalveni, C.; Ingle, A. P.; Bhagat, R.; Rokhum, S. L. Widely used catalysts in biodiesel production: a review. RSC. Adv. 2020, 10, 41625-79.
32. Basumatary, S. F.; Brahma, S.; Hoque, M.; et al. Advances in CaO-based catalysts for sustainable biodiesel synthesis. Green. Energy. Resour. 2023, 1, 100032.
33. Zul, N. A.; Ganesan, S.; Hamidon, T. S.; Oh, W.; Hussin, M. H. A review on the utilization of calcium oxide as a base catalyst in biodiesel production. J. Environ. Chem. Eng. 2021, 9, 105741.
34. Macheli, L.; Malefane, M. E.; Jewell, L. L. Waste-derived calcium oxide catalysts in biodiesel production: exploring various waste sources, deactivation challenges, and improvement strategies. Bioresour. Technol. Rep. 2025, 29, 102021.
35. Ooi, H. K.; Koh, X. N.; Ong, H. C.; et al. Progress on modified calcium oxide derived waste-shell catalysts for biodiesel production. Catalysts 2021, 11, 194.
36. Liu, X.; Xing, S.; Yang, L.; et al. Highly active and durable Ca-based solid base catalyst for biodiesel production. Fuel 2021, 302, 121094.
37. Granados, M. L.; Alonso, D. M.; Sádaba, I.; Mariscal, R.; Ocón, P. Leaching and homogeneous contribution in liquid phase reaction catalysed by solids: the case of triglycerides methanolysis using CaO. Appl. Catal. B. Environ. 2009, 89, 265-72.
38. Ghosh, N.; Khan, A. M.; Halder, G. Advances in calcium oxide mediated catalytic biodiesel production: a paradigm shift in complying with UN's SDG7. Appl. Energy. 2025, 391, 125954.
39. Mazaheri, H.; Ong, H. C.; Amini, Z.; et al. An overview of biodiesel production via calcium oxide based catalysts: current state and perspective. Energies 2021, 14, 3950.
40. Sulaiman, N. F.; Ramly, N. I.; Abd, Mubin. M. H.; Lee, S. L. Transition metal oxide (NiO, CuO, ZnO)-doped calcium oxide catalysts derived from eggshells for the transesterification of refined waste cooking oil. RSC. Adv. 2021, 11, 21781-95.
41. Wang, Y.; Li, D.; Zhao, D.; et al. Calcium-loaded municipal sludge-biochar as an efficient and stable catalyst for biodiesel production from vegetable oil. ACS. Omega. 2020, 5, 17471-8.
42. Arenas-quevedo, M. G.; Manríquez, M. E.; Wang, J. A.; et al. ZnO-doped CaO binary core-shell catalysts for biodiesel production via mexican palm oil transesterification. Inorganics 2024, 12, 51.
43. Teo, C. K.; Chia, P. W.; Nordin, N.; et al. Calcium oxide waste-based catalysts for biodiesel production and depollution: a review. Environ. Chem. Lett. 2024, 22, 1741-58.
44. Parida, S.; Singh, M.; Pradhan, S. Biomass wastes: a potential catalyst source for biodiesel production. Bioresour. Technol. Rep. 2022, 18, 101081.
45. Chandra, Kishore. S.; Perumal, S.; Atchudan, R.; et al. A review of biomass-derived heterogeneous catalysts for biodiesel production. Catalysts 2022, 12, 1501.
46. Shan, R.; Zhao, C.; Lv, P.; Yuan, H.; Yao, J. Catalytic applications of calcium rich waste materials for biodiesel: current state and perspectives. Energy. Convers. Manag. 2016, 127, 273-83.
47. Ao, S.; Changmai, B.; Vanlalveni, C.; Chhandama, M. V. L.; Wheatley, A. E.; Rokhum, S. L. Biomass waste-derived catalysts for biodiesel production: Recent advances and key challenges. Renew. Energy. 2024, 223, 120031.
48. Amal, R.; Nizamuddin, S. Towards sustainable biodiesel production: efficient utilization of animal fat wastes and shell-derived catalysts. Environ. Sci. Pollut. Res. Int. 2025, 32, 14383-404.
49. Makarevičienė, V.; Gaidė, I.; Sendžikienė, E. Heterogeneous catalysts from food waste for biodiesel synthesis - a comprehensive review. Catalysts 2025, 15, 957.
50. Amal, R.; Usman, M. A review of breakthroughs in biodiesel production with transition and non-transition metal-doped CaO nano-catalysts. Biomass. Bioenergy. 2024, 184, 107158.
51. Melo, V. M. E.; Ferreira, G. F.; Fregolente, L. V. Sustainable catalysts for biodiesel production: the potential of CaO supported on sugarcane bagasse biochar. Renew. Sustain. Energy. Rev. 2024, 189, 114042.
52. Boey, P.; Maniam, G. P.; Hamid, S. A. Performance of calcium oxide as a heterogeneous catalyst in biodiesel production: a review. Chem. Eng. J. 2011, 168, 15-22.
53. Gholami, A.; Pourfayaz, F.; Rodygin, K. Reusable chemical catalysts for sustainable biodiesel production: the role of metallic elements. ChemBioEng. Rev. 2025, 12, e202400033.
54. Oueda, N.; Bonzi-coulibaly, Y. L.; Ouédraogo, I. W. K. Deactivation processes, regeneration conditions and reusability performance of CaO or MgO based catalysts used for biodiesel production - a review. Mater. Sci. Appl. 2017, 08, 94-122.
55. Kouzu, M.; Hidaka, J. Transesterification of vegetable oil into biodiesel catalyzed by CaO: a review. Fuel 2012, 93, 1-12.
56. Tigro, A. A.; Hailegiorgis, S. M.; Reshad, A. S. Heterogeneous alkaline calcium oxide nano-catalyst supported on porous materials for the transesterification reaction: a review. Results. Eng. 2025, 27, 105912.
57. Song, C.; Kazemi, M. Based on magnetically recoverable catalysts: a green strategy to sulfonamides. Mol. Divers. 2025, 29, 5045-74.
58. Rodriguez, Y.; Ponce, S.; Keller, M. H.; et al. Calcium-based catalyst prepared in the presence of biopolymer/biomass for biodiesel production: Elucidating their influence on the catalytic efficiency. Biomass. Bioenergy. 2024, 186, 107268.
59. Rahman, W. U.; Khan, R. I. A.; Ahmad, S.; et al. Valorizing waste palm oil towards biodiesel production using calcareous eggshell based heterogeneous catalyst. Bioresour. Technol. Rep. 2023, 23, 101584.
60. Ulakpa, W. C.; Aseibichin, C.; Chimezie, O. A.; et al. Nano-CaO as a heterogeneous catalyst for biodiesel synthesis by transesterification of Jatropha oil. J. Trace. Elem. Miner. 2024, 9, 100183.
61. Garg, R.; Garg, R.; Eddy, N. O.; Abdulraheem, M. I.; Hazzan, O. O.; Abdi, G. Green synthesis of calcium oxide nanocatalyst and application in transesterification of waste cooking oil. Bioresour. Bioprocess. 2025, 12, 52.
62. Kouider, Elouahed. S.; Asikin-Mijan, N.; Kaddour, O.; et al. Efficient biodiesel production from soybean oil using calcium oxide supported bimetallic molybdenum-zinc catalysts. Int. J. Green. Energy. 2025, 22, 3670-86.
63. Ho, W. W. S.; Ng, H. K.; Gan, S. Biodiesel synthesis from refined palm oil using a calcium oxide impregnated ash-based catalyst: parametric, kinetics, and product characterization studies. Catalysts 2022, 12, 706.
64. Kouzu, M.; Tsunomori, M.; Yamanaka, S.; Hidaka, J. Solid base catalysis of calcium oxide for a reaction to convert vegetable oil into biodiesel. Adv. Powder. Technol. 2010, 21, 488-94.
65. Ahmed, M.; Ahmad, K. A.; Vo, D. N.; et al. Recent trends in sustainable biodiesel production using heterogeneous nanocatalysts: Function of supports, promoters, synthesis techniques, reaction mechanism, and kinetics and thermodynamic studies. Energy. Convers. Manag. 2023, 280, 116821.
66. Kouzu, M.; Kasuno, T.; Tajika, M.; Sugimoto, Y.; Yamanaka, S.; Hidaka, J. Calcium oxide as a solid base catalyst for transesterification of soybean oil and its application to biodiesel production. Fuel 2008, 87, 2798-806.
67. Hu, S.; Wang, Y.; Han, H. Utilization of waste freshwater mussel shell as an economic catalyst for biodiesel production. Biomass. Bioenergy. 2011, 35, 3627-35.
68. Sharifi, M.; Tangestaninejad, S.; Moghadam, M.; et al. Metal-organic frameworks-derived CaO/ZnO composites as stable catalysts for biodiesel production from soybean oil at room temperature. Sci. Rep. 2025, 15, 3610.
69. Ahmad, Khan. R. I.; Ghosh, N.; Khan, A. R.; et al. Unlocking the efficacy of eggshell-derived CaO@TiO2 nanocatalyst for intensified green biodiesel production. J. Ind. Eng. Chem. 2026, 153, 355-69.
70. Weldeslase, M. G.; Benti, N. E.; Desta, M. A.; Mekonnen, Y. S. Maximizing biodiesel production from waste cooking oil with lime-based zinc-doped CaO using response surface methodology. Sci. Rep. 2023, 13, 4430.
71. Olvera, D.; Rodriguez, J. A.; Perez-Silva, I.; Chavez-Esquivel, G.; Tavizón-Pozos, J. A. Catalytic evaluation of Li and K supported on CaO in the transesterification of triolein, triestearin, and tributyrin. Chem. Pap. 2022, 76, 6287-95.
72. Li, X.; Zhang, S.; Jia, X.; Li, W.; Song, J. Catalytic properties and structural optimization of solid transesterification catalysts to enhance the efficiency of biodiesel synthesis. Catalysts 2025, 15, 239.
73. Catarino, M.; Martins, S.; Soares, Dias. A. P.; Costa, Pereira. M. F.; Gomes, J. Calcium diglyceroxide as a catalyst for biodiesel production. J. Environ. Chem. Eng. 2019, 7, 103099.
74. Kouzu, M.; Yamanaka, S.; Hidaka, J.; Tsunomori, M. Heterogeneous catalysis of calcium oxide used for transesterification of soybean oil with refluxing methanol. Appl. Catal. A. Gen. 2009, 355, 94-9.
75. Sharma, S.; Saxena, V.; Baranwal, A.; Chandra, P.; Pandey, L. M. Engineered nanoporous materials mediated heterogeneous catalysts and their implications in biodiesel production. Mater. Sci. Energy. Technol. 2018, 1, 11-21.
76. Wang, S.; Fu, H.; Abulizi, A.; et al. Biodiesel production from sheep fat catalyzed by CaO-SrOx/BCsy acid-base bifunctional catalyst and process optimization. Renew. Energy. 2025, 238, 121881.
77. Lee, H.; Juan, J.; Taufiq-Yap, Y. Preparation and application of binary acid-base CaO-La2O3 catalyst for biodiesel production. Renew. Energy. 2015, 74, 124-32.
78. Enguilo Gonzaga, V.; Romero, R.; Romero, A.; Martínez, S. L.; Natividad, R. Biodiesel production from waste cooking oil catalyzed by a bifunctional catalyst. ACS. Omega. 2021, 6, 24092-105.
79. Zhang, Z.; Meng, P.; Luo, H.; Pei, Z.; Liu, X. Lewis acid-base site-assisted in situ transesterification catalysis to produce biodiesel. Catalysts 2024, 14, 731.
80. Mulyatun, M.; Prameswari, J.; Istadi, I.; Widayat, W. Synthesis method effect on the catalytic performance of acid-base bifunctional catalysts for converting low-quality waste cooking oil to biodiesel. Catal. Lett. 2024, 154, 4837-55.
81. Vishnulal, K.; Ramadesigan, V.; Seethamraju, S. Biodiesel synthesis using spent FCC catalyst and CaO from a mixture of sunflower oil and oleic acid. Bioresour. Technol. Rep. 2024, 25, 101714.
82. Ali, B.; Yusup, S.; Quitain, A. T.; Alnarabiji, M. S.; Kamil, R. N. M.; Kida, T. Synthesis of novel graphene oxide/bentonite bi-functional heterogeneous catalyst for one-pot esterification and transesterification reactions. Energy. Convers. Manag. 2018, 171, 1801-12.
83. Niju, S.; Meera, Sheriffa. Begum. K.; Anantharaman, N. Enhancement of biodiesel synthesis over highly active CaO derived from natural white bivalve clam shell. Arab. J. Chem. 2016, 9, 633-9.
84. Rezaei, R.; Mohadesi, M.; Moradi, G. Optimization of biodiesel production using waste mussel shell catalyst. Fuel 2013, 109, 534-41.
85. Boey, P.; Maniam, G. P.; Hamid, S. A.; Ali, D. M. H. Utilization of waste cockle shell (Anadara granosa) in biodiesel production from palm olein: optimization using response surface methodology. Fuel 2011, 90, 2353-8.
86. Nair, P.; Singh, B.; Upadhyay, S.; Sharma, Y. Synthesis of biodiesel from low FFA waste frying oil using calcium oxide derived from Mereterix mereterix as a heterogeneous catalyst. J. Clean. Prod. 2012, 29-30, 82-90.
87. Buasri, A.; Loryuenyong, V. The new green catalysts derived from waste razor and surf clam shells for biodiesel production in a continuous reactor. Green. Process. Synth. 2015, 4, 389-97.
88. Buasri, A.; Rattanapan, T.; Boonrin, C.; Wechayan, C.; Loryuenyong, V. Oyster and Pyramidella shells as heterogeneous catalysts for the microwave-assisted biodiesel production from Jatropha curcas oil. J. Chem. 2015, 2015, 578625.
89. Maruyama, H.; Seki, H. Esterification of tripalmitin using calcined scallop shell as a heterogeneous basic catalyst. Asia-Pac. J. Chem. Eng. 2022, 18, e2870.
90. Nahas, L.; Dahdah, E.; Aouad, S.; et al. Highly efficient scallop seashell-derived catalyst for biodiesel production from sunflower and waste cooking oils: Reaction kinetics and effect of calcination temperature studies. Renew. Energy. 2023, 202, 1086-95.
91. Sirisomboonchai, S.; Abuduwayiti, M.; Guan, G.; et al. Biodiesel production from waste cooking oil using calcined scallop shell as catalyst. Energy. Convers. Manag. 2015, 95, 242-7.
92. Panjaitan, F. R.; Yamanaka, S.; Kuga, Y. Soybean oil methanolysis over scallop shell-derived CaO prepared via methanol-assisted dry nano-grinding. Adv. Powder. Technol. 2017, 28, 1627-35.
93. Kouzu, M.; Kajita, A.; Fujimori, A. Catalytic activity of calcined scallop shell for rapeseed oil transesterification to produce biodiesel. Fuel 2016, 182, 220-6.
94. Niju, S.; Vishnupriya, G.; Balajii, M. Process optimization of Calophyllum inophyllum-waste cooking oil mixture for biodiesel production using Donax deltoides shells as heterogeneous catalyst. Sustain. Environ. Res. 2019, 29, 18.
95. Madhuvilakku, R.; Mariappan, R.; Jeyapal, S.; Sundar, S.; Piraman, S. Transesterification of palm oil catalyzed by fresh water bivalve mollusk (margaritifera falcata) shell as heterogeneous catalyst. Ind. Eng. Chem. Res. 2013, 52, 17407-13.
96. Lee, S. L.; Wong, Y. C.; Tan, Y. P.; Yew, S. Y. Transesterification of palm oil to biodiesel by using waste obtuse horn shell-derived CaO catalyst. Energy. Convers. Manag. 2015, 93, 282-8.
97. Correia, L. M.; Saboya, R. M.; Campelo, Nde. S.; et al. Characterization of calcium oxide catalysts from natural sources and their application in the transesterification of sunflower oil. Bioresour. Technol. 2014, 151, 207-13.
98. Madhu, D.; Arora, R.; Sahani, S.; Singh, V.; Sharma, Y. C. Synthesis of high-quality biodiesel using feedstock and catalyst derived from fish wastes. J. Agric. Food. Chem. 2017, 65, 2100-9.
99. El-Gendy, N. S. H.; Hamdy, A.; Abu Amr, S. S. An investigation of biodiesel production from wastes of seafood restaurants. Int. J. Biomater. 2014, 2014, 609624.
100. Kedir, W. M.; Wondimu, K. T.; Weldegrum, G. S. Optimization and characterization of biodiesel from waste cooking oil using modified CaO catalyst derived from snail shell. Heliyon 2023, 9, e16475.
101. Abdul Rahim, N.; Rosdzimin Abdul Rahman, M.; Amali Bin Ahmad, K.; Rahim Mat Sarip, A.; Hasbullah, S.; Suhel, A. Analysis of waste cooking oil biodiesel (WCO) synthesis with TiO2 impregnated CaO from waste shells nano-catalyst. Mater. Today. Proc. 2023, S2214785323044681.
102. Gupta, J.; Agarwal, M. Preparation and characterizaton of CaO nanoparticle for biodiesel production. AIP. Conf. Proc. 2016, 1724, 020066.
103. Kaewdaeng, S.; Sintuya, P.; Nirunsin, R. Biodiesel production using calcium oxide from river snail shell ash as catalyst. Energy. Proc. 2017, 138, 937-42.
104. Gupta, J.; Agarwal, M.; Dalai, A. K. Experimental evaluation of the catalytic efficiency of calcium based natural and modified catalyst for biodiesel synthesis. Int. J. Green. Energy. 2017, 14, 878-88.
105. Margaretha, Y. Y.; Prastyo, H. S.; Ayucitra, A.; Ismadji, S. Calcium oxide from Pomacea sp. shell as a catalyst for biodiesel production. Int. J. Energy. Environ. Eng. 2012, 3, 33.
106. Ogunsola, A. D.; Durowoju, M. O.; Alade, A. O.; Jekayinfa, S. O.; Ogunkunle, O. Modeling and optimization of two-step shea butter oil biodiesel synthesis using snail shells as heterogeneous base catalysts. Energy. Adv. 2022, 1, 113-28.
107. Boro, J.; Thakur, A. J.; Deka, D. Solid oxide derived from waste shells of Turbonilla striatula as a renewable catalyst for biodiesel production. Fuel. Process. Technol. 2011, 92, 2061-7.
108. Ao, S.; Greer, H. F.; Alghamdi, L. A.; et al. Snail shell derived magnetic nanocatalysts for biodiesel production: process optimization through response surface methodology, kinetics, and thermodynamic studies. Biomass. Bioenergy. 2024, 191, 107442.
109. Sharma, Y. C.; Singh, B.; Korstad, J. Application of an efficient nonconventional heterogeneous catalyst for biodiesel synthesis from pongamia pinnata oil. Energy. Fuels. 2010, 24, 3223-31.
110. Marwaha, A.; Rosha, P.; Mohapatra, S. K.; Mahla, S. K.; Dhir, A. Biodiesel production from Terminalia bellerica using eggshell-based green catalyst: an optimization study with response surface methodology. Energy. Rep. 2019, 5, 1580-8.
111. Mmusi, K. C.; Odisitse, S.; Nareetsile, F. Comparison of CaO-NPs and chicken eggshell-derived CaO in the production of biodiesel from schinziophyton rautanenii (mongongo) nut oil. J. Chem. 2021, 2021, 1-15.
112. Ashine, F.; Kiflie, Z.; Prabhu, S. V.; et al. Biodiesel production from Argemone mexicana oil using chicken eggshell derived CaO catalyst. Fuel 2023, 332, 126166.
113. Erchamo, Y. S.; Mamo, T. T.; Workneh, G. A.; Mekonnen, Y. S. Improved biodiesel production from waste cooking oil with mixed methanol-ethanol using enhanced eggshell-derived CaO nano-catalyst. Sci. Rep. 2021, 11, 6708.
114. Solomon, Y.; Girma, E.; Ramayya, A. V. Amalgamation and optimization of FAEE biodiesel fuel from croton macrostachyus hochst. ex delile seed oil utilizing solid calcium oxide-chicken eggshell squander as a heterogeneous catalyst. Int. J. Anal. Chem. 2022, 2022, 7984077.
115. Alsaiari, R.; Musa, E.; Rizk, M. Effects of calcination temperature of eggshell-derived CaO as a catalyst for biodiesel production from waste cooking oil. S. Afr. J. Chem. 2023, 77, 30-5.
116. Kumar, H.; Renita, A. A.; Jabasingh, S. A. Biodiesel production from preutilized cooking oil using a renewable heterogeneous eggshell-coconut pith catalyst: process optimization and characterization. Environ. Prog. Sustain. Energy. 2021, 40, e13632.
117. AVSLSai, B.; Subramaniapillai, N.; Khadhar, Mohamed. M. S. B.; Narayanan, A. Optimization of continuous biodiesel production from rubber seed oil (RSO) using calcined eggshells as heterogeneous catalyst. J. Environ. Chem. Eng. 2020, 8, 103603.
118. Rahman, W. U.; Fatima, A.; Anwer, A. H.; et al. Biodiesel synthesis from eucalyptus oil by utilizing waste egg shell derived calcium based metal oxide catalyst. Process. Saf. Environ. Prot. 2019, 122, 313-9.
119. Foroutan, R.; Mohammadi, R.; Esmaeili, H.; Mirzaee, Bektashi. F.; Tamjidi, S. Transesterification of waste edible oils to biodiesel using calcium oxide@magnesium oxide nanocatalyst. Waste. Manag. 2020, 105, 373-83.
120. Talha, N. S.; Sulaiman, S. In situ transesterification of solid coconut waste in a packed bed reactor with CaO/PVA catalyst. Waste. Manag. 2018, 78, 929-37.
121. Niju, S. S.; Begum, K. M. M.; Anantharaman, N. Continuous flow reactive distillation process for biodiesel production using waste egg shells as heterogeneous catalysts. RSC. Adv. 2014, 4, 54109-14.
122. Fayyazi, E.; Ghobadian, B.; van, de. Bovenkamp. H. H.; et al. Optimization of biodiesel production over chicken eggshell-derived CaO catalyst in a continuous centrifugal contactor separator. Ind. Eng. Chem. Res. 2018, 57, 12742-55.
123. Proença, B. D. S. G.; Fioroto, P. O.; Heck, S. C.; et al. Obtention of methyl esters from macauba oil using egg shell catalyst. Chem. Eng. Res. Des. 2021, 169, 288-96.
124. Piker, A.; Tabah, B.; Perkas, N.; Gedanken, A. A green and low-cost room temperature biodiesel production method from waste oil using egg shells as catalyst. Fuel 2016, 182, 34-41.
125. Viriya-Empikul, N.; Krasae, P.; Puttasawat, B.; Yoosuk, B.; Chollacoop, N.; Faungnawakij, K. Waste shells of mollusk and egg as biodiesel production catalysts. Bioresour. Technol. 2010, 101, 3765-7.
126. Kirubakaran, M.; Arul, Mozhi. Selvan. V. Experimental investigation on the effects of micro eggshell and nano-eggshell catalysts on biodiesel optimization from waste chicken fat. Bioresour. Technol. Rep. 2021, 14, 100658.
127. Kirubakaran, M.; Arul, Mozhi. Selvan. V. Biodiesel production from waste chicken oil using nanoeggshell heterogeneous catalyst with isopropyl ether as cosolvent. Environ. Qual. Manag. 2020, 31, 51-64.
128. Khemthong, P.; Luadthong, C.; Nualpaeng, W.; et al. Industrial eggshell wastes as the heterogeneous catalysts for microwave-assisted biodiesel production. Catal. Today. 2012, 190, 112-6.
129. Goli, J.; Sahu, O. Development of heterogeneous alkali catalyst from waste chicken eggshell for biodiesel production. Renew. Energy. 2018, 128, 142-54.
130. Ayoola, A.; Fayomi, O.; Adeeyo, O.; Omodara, J.; Adegbite, O. Impact assessment of biodiesel production using CaO catalyst obtained from two different sources. Cogent. Eng. 2019, 6, 1615198.
131. Navajas, A.; Issariyakul, T.; Arzamendi, G.; Gandía, L.; Dalai, A. Development of eggshell derived catalyst for transesterification of used cooking oil for biodiesel production. Asia. Pac. J. Chem. Eng. 2013, 8, 742-8.
132. Bharadwaj, A. S.; Singh, M.; Niju, S.; Begum, K. M. S.; Anantharaman, N. Biodiesel production from rubber seed oil using calcium oxide derived from eggshell as catalyst - optimization and modeling studies. Green. Process. Synth. 2019, 8, 430-42.
133. Moradi, G.; Mohammadi, F. Utilization of waste coral for biodiesel production via transesterification of soybean oil. Int. J. Environ. Sci. Technol. 2013, 11, 805-12.
134. Teo, S. H.; Islam, A.; Ng, F. L.; Taufiq-Yap, Y. H. Biodiesel synthesis from photoautotrophic cultivated oleoginous microalgae using a sand dollar catalyst. RSC. Adv. 2015, 5, 47140-52.
135. Suwannasom, P.; Tansupo, P.; Ruangviriyachai, C. A bone-based catalyst for biodiesel production from waste cooking oil. Energy. Sources. Part. A. 2016, 38, 3167-73.
136. Sulaiman, S.; Abdul, Jamaludin. N. F.; Kabbashi, N. A. Development of CaO/PVA catalyst from fish bone for biodiesel production. Bull. Chem. React. Eng. Catal. 2019, 14, 153-7.
137. Maneerung, T.; Kawi, S.; Dai, Y.; Wang, C. Sustainable biodiesel production via transesterification of waste cooking oil by using CaO catalysts prepared from chicken manure. Energy. Convers. Manag. 2016, 123, 487-97.
138. Maneerung, T.; Kawi, S.; Wang, C. Biomass gasification bottom ash as a source of CaO catalyst for biodiesel production via transesterification of palm oil. Energy. Convers. Manag. 2015, 92, 234-43.
139. Bazargan, A.; Kostić, M. D.; Stamenković, O. S.; Veljković, V. B.; Mckay, G. A calcium oxide-based catalyst derived from palm kernel shell gasification residues for biodiesel production. Fuel 2015, 150, 519-25.
140. Kostić, M. D.; Bazargan, A.; Stamenković, O. S.; Veljković, V. B.; Mckay, G. Optimization and kinetics of sunflower oil methanolysis catalyzed by calcium oxide-based catalyst derived from palm kernel shell biochar. Fuel 2016, 163, 304-13.
141. Ajala, E.; Ajala, M.; Ajao, A.; Saka, H.; Oladipo, A. Calcium-carbide residue: a precursor for the synthesis of CaO-Al2O3-SiO2-CaSO4 solid acid catalyst for biodiesel production using waste lard. Chem. Eng. J. Adv. 2020, 4, 100033.
142. Ajala, E.; Ehinmowo, A.; Ajala, M.; Ohiro, O.; Aderibigbe, F.; Ajao, A. Optimisation of CaO-Al2O3-SiO2-CaSO4-based catalysts performance for methanolysis of waste lard for biodiesel production using response surface methodology and meta-heuristic algorithms. Fuel. Process. Technol. 2022, 226, 107066.
143. Liu, M.; Huang, Y.; Liu, Q.; et al. Ferric oxide as a support of carbide slag for effective transesterification of triglycerides in soybean oil. Energy. Convers. Manag. 2019, 198, 111785.
144. Liu, M.; Niu, S.; Lu, C.; Cheng, S. An optimization study on transesterification catalyzed by the activated carbide slag through the response surface methodology. Energy. Convers. Manag. 2015, 92, 498-506.
145. Potorochenko, A. N.; Gyrdymova, Y. V.; Rodygin, K. S. Waste-derived catalyst for biodiesel manufacturing in CO2-free manner: preparation, catalytic activity, and reuse studies. ChemCatChem 2025, 17, e202401607.
146. Rodygin, K. S.; Gyrdymova, Y. V.; Ananikov, V. P. Calcium carbide residue - a key inorganic component of the sustainable carbon cycle. Russ. Chem. Rev. 2022, 91, RCR5048.
147. Bawab, J.; El-Hassan, H.; El-Dieb, A.; Khatib, J.; El-Mir, A. Utilization of calcium carbide residue as a concrete component: a comprehensive review. Сase. Stud. Constr. Mater. 2025, 22, e04823.
148. Rodygin, K. S.; Bogachenkov, A. S.; Gyrdymova, Y. V.; Potorochenko, A. N. 13C-Labeling as a method in organic synthesis, catalysis and biochemical applications. Chem. Methods. 2024, 5, e202400045.
149. Gyrdymova, Y. V.; Potorochenko, A. N. Reactions of calcium carbide catalyzed by transition metals: recent advances and prospects. Russ. Chem. Bull. 2025, 74, 1576-91.
150. Potorochenko, A. N.; Rodygin, K. S.; Ananikov, V. P. Assembly of (2×C2+C’2)×n molecular complexity using a sequence of Pt- and Pd-catalyzed transformations with calcium carbide. Eur. J. Org. Chem. 2023, 27, e202301012.
151. Korabelnikova, V. A.; Gyrdymova, Y. V.; Gordeev, E. G.; Potorochenko, A. N.; Rodygin, K. S.; Ananikov, V. P. 3D printing for safe organic synthesis in mixed liquid/gas-phase chemistry. React. Chem. Eng. 2025, 10, 2474-89.
152. Wu, J.; Ma, Y.; Wang, Y.; et al. Copper-catalyzed direct synthesis of 3-methylene-2-arylisoindolin-1-ones with calcium carbide as a surrogate of gaseous acetylene. Green. Chem. 2023, 25, 3425-30.
153. Liu, Z.; Wang, Z.; Liao, H.; Li, Z. One-pot synthesis of 1,2,3-triarylindoles through cascade reactions using calcium carbide, iodoarenes, and aromatic amines. Org. Lett. 2023, 25, 5812-6.
154. Liu, L.; Sun, G.; Zhang, J. Constructing 5-methyl-2,4-diaryl-1H-imidazoles using calcium carbide as alkyne source via A3-coupling cyclization. Adv. Synth. Catal. 2023, 365, 1801-5.
155. Liu, H.; You, X.; Wen, F.; Zhang, Z.; Li, Z. Calcium carbide as a surrogate of acetylene: copper-catalyzed construction of 3-methylene-2-arylisoindolin-1-ones. Asian. J. Org. Chem. 2022, 11, e202200204.
156. Jing, T.; Liu, N.; Xu, C.; Bu, Q. Zinc chloride-promoted coupling reaction between calcium carbide and aryl chlorides. Eur. J. Org. Chem. 2022, 2022, e202200178.
157. An, R.; Cui, W.; Gu, P.; Wei, M. Highly stereoselective synthesis ofN-sulfonyl amidines with calcium carbide as an acetylene source. Org. Chem. Front. 2025, 12, 6588-93.
158. Wang, Z.; Liu, Z.; Wang, B.; Li, Z. Construction of 2-methylindoles using solid calcium carbide as a substitute for gaseous acetylene. Eur. J. Org. Chem. 2024, 27, e202301262.
159. Wang, B.; You, X.; Wang, J.; Li, Z. Highly stereoselective synthesis of 2-Acyl-3-sulfonamidobut-2-enoates using solid calcium carbide as a substitute for gaseous acetylene. Org. Lett. 2024, 26, 6269-73.
160. Potorochenko, A. N.; Rodygin, K. S. Calcium waste as a catalyst in the transesterification for demanding esters: scalability perspective. Beilstein. J. Org. Chem. 2025, 21, 1520-7.
161. Wang, F.; Li, H.; Gao, J.; et al. High efficiency removal of NO using waste calcium carbide slag by facile KOH modification. J. Environ. Sci. 2024, 139, 182-92.
162. Gong, X.; Zhang, T.; Zhang, J.; et al. Recycling and utilization of calcium carbide slag - current status and new opportunities. Renew. Sustain. Energy. Rev. 2022, 159, 112133.
163. Lotsman, K. A.; Rodygin, K. S. Calcium carbide residue - a promising hidden source of hydrogen. Green. Chem. 2023, 25, 3524-32.
164. Samoylenko, D. E.; Rodygin, K. S.; Ananikov, V. P. Sustainable application of calcium carbide residue as a filler for 3D printing materials. Sci. Rep. 2023, 13, 4465.
165. Potorochenko, A. N.; Ovchinnikov, A. A.; Rodygin, K. S. Two times faster glycolysis of poly(ethylene terephthalate) with CaO filler-catalyst. J. Compos. Sci. 2025, 9, 680.
166. Mohammadi Darestani, A.; Arabani, M.; Zanganeh Ranjbar, P.; Payan, M.; Salimi, M. Strength enhancement of calcium carbide residue-stabilized clay with perlite powder and water treatment sludge. Sci. Rep. 2025, 15, 43701.
167. Anandan Govindan, S.; Mudavath, H.; Vaddemani, G. P. R. Evaluating the sustainability of using calcium carbide residue for clay stabilization: integrating mechanical behaviour and life cycle assessment. Environ. Sci. Pollut. Res. 2025, 32, 15772-89.
168. Moazami, D.; Pourabbas, Bilondi. M.; Rahnama, A.; Zaresefat, M.; Moretti, L. Recycled glass powder and calcium carbide residue geopolymer to stabilise silty sand soil: Mechanical performances and statistical analysis. Heliyon 2025, 11, e41738.
169. Wang, Q.; Guo, H.; Yu, T.; Yuan, P.; Deng, L.; Zhang, B. Utilization of calcium carbide residue as solid alkali for preparing fly ash-based geopolymers: dependence of compressive strength and microstructure on calcium carbide residue, water content and curing temperature. Materials 2022, 15, 973.
170. Ma, W.; Zhu, G.; Guan, X.; et al. An innovative approach to prepare calcium oxide from calcium carbide slag based on sequential mineral transformation. ACS. Sustain. Resour. Manag. 2024, 1, 1279-90.
171. Liu, M. Q.; Niu, S. L.; Lu, C. M. The study on the transesterification catalyzed by sodium compound loaded on carbide slag. Appl. Mech. Mater. 2015, 733, 195-8.
172. Wang, T.; Tan, J.; Liu, J.; Tang, Y.; He, Y.; Cui, X. Valorizing and detoxifying alkaline lime mud waste from pulp mills via the production of high activity lime and lye. J. Environ. Chem. Eng. 2025, 13, 118010.
173. Li, H.; Niu, S.; Lu, C.; Liu, M.; Huo, M. Use of lime mud from paper mill as a heterogeneous catalyst for transesterification. Sci. China. Technol. Sci. 2013, 57, 438-44.
174. M, K.; Sundararaman, S.; J, A. K.; Deivasigamani, P.; M, R. Synthesis and characterization of barium doped CaO heterogeneous nanocatalyst for the production of biodiesel from Catharanthus roseus seeds: Kinetics, optimization and performance evaluation. Environ. Res. 2023, 222, 115336.
175. Aghel, B.; Gouran, A.; Shahsavari, P. Optimizing the production of biodiesel from waste cooking oil utilizing industrial waste-derived MgO/CaO catalysts. Chem. Eng. Technol. 2022, 45, 348-54.
176. Risso, R.; Ferraz, P.; Meireles, S.; Fonseca, I.; Vital, J. Highly active Cao catalysts from waste shells of egg, oyster and clam for biodiesel production. Appl. Catal. A. Gen. 2018, 567, 56-64.
177. Cho, Y. B.; Seo, G.; Chang, D. R. Transesterification of tributyrin with methanol over calcium oxide catalysts prepared from various precursors. Fuel. Process. Technol. 2009, 90, 1252-8.
178. Alba-Rubio, A. C.; Santamaría-González, J.; Mérida-Robles, J. M.; et al. Heterogeneous transesterification processes by using CaO supported on zinc oxide as basic catalysts. Catal. Today. 2010, 149, 281-7.
179. Mazaheri, H.; Ong, H. C.; Masjuki, H.; et al. Rice bran oil based biodiesel production using calcium oxide catalyst derived from Chicoreus brunneus shell. Energy 2018, 144, 10-9.
180. Kawashima, A.; Matsubara, K.; Honda, K. Acceleration of catalytic activity of calcium oxide for biodiesel production. Bioresour. Technol. 2009, 100, 696-700.
181. Trisupakitti, S.; Ketwong, C.; Senajuk, W.; Phukapak, C.; Wiriyaumpaiwong, S. Golden apple cherry snail shell as catalyst for heterogeneous transesterification of biodiesel. Braz. J. Chem. Eng. 2018, 35, 1283-91.
182. Krishnamurthy, K.; Sridhara, S.; Ananda, Kumar. C. Optimization and kinetic study of biodiesel production from Hydnocarpus wightiana oil and dairy waste scum using snail shell CaO nano catalyst. Renew. Energy. 2020, 146, 280-96.
183. Correia, L. M.; De, Sousa. Campelo. N.; Novaes, D. S.; et al. Characterization and application of dolomite as catalytic precursor for canola and sunflower oils for biodiesel production. Chem. Eng. J. 2015, 269, 35-43.
184. Boro, J.; Konwar, L. J.; Thakur, A. J.; Deka, D. Ba doped CaO derived from waste shells of T striatula (TS-CaO) as heterogeneous catalyst for biodiesel production. Fuel 2014, 129, 182-7.
185. Fasanya, O. O.; Gbadamasi, S.; Osigbesan, A. A.; et al. Effect of hydrothermal treatment on the properties of calcium oxide from eggshells used as a biodiesel catalyst. Chem. Eng. Technol. 2021, 45, 283-90.
186. Kojima, Y.; Takai, S. Transesterification of vegetable oil with methanol using solid base catalyst of calcium oxide under ultrasonication. Chem. Eng. Proc. 2019, 136, 101-6.
187. Acosta, P. I.; Campedelli, R. R.; Correa, E. L.; et al. Efficient production of biodiesel by using a highly active calcium oxide prepared in presence of pectin as heterogeneous catalyst. Fuel 2020, 271, 117651.
188. Degfie, T. A.; Mamo, T. T.; Mekonnen, Y. S. Optimized biodiesel production from waste cooking oil (WCO) using calcium oxide (CaO) nano-catalyst. Sci. Rep. 2019, 9, 18982.
189. Li, F.; Li, H.; Wang, L.; Cao, Y. Waste carbide slag as a solid base catalyst for effective synthesis of biodiesel via transesterification of soybean oil with methanol. Fuel. Process. Technol. 2015, 131, 421-9.
190. Liu, J.; Liu, M.; Chen, S.; et al. Conversion of Au(III)-polluted waste eggshell into functional CaO/Au nanocatalyst for biodiesel production. Green. Energy. Environ. 2022, 7, 352-9.
191. Campos-Molina, M. J.; Santamaría-González, J.; Mérida-Robles, J.; et al. Base catalysts derived from hydrocalumite for the transesterification of sunflower oil. Energy. Fuels. 2009, 24, 979-84.
192. Zhang, M.; Ramya, G.; Brindhadevi, K.; et al. RETRACTED: microwave assisted biodiesel production from chicken feather meal oil using bio-nano calcium oxide derived from chicken egg shell. Environ. Res. 2022, 205, 112509.
193. Rahmayanti, M.; Fatimah, I.; Yahdiyani, Ikhsani. A.; Nur, Azizah. D. The potential of calcium oxide nanocatalyst from chicken eggshells for biodiesel production using chicken fat waste. Inorg. Chem. Commun. 2024, 165, 112604.
194. Zhu, H.; Wu, Z.; Chen, Y.; et al. Preparation of biodiesel catalyzed by solid super base of calcium oxide and its refining process. Chine. J. Catal. 2006, 27, 391-6.
195. Sochima, E. V.; Aigbodion, V. S.; Von, Kallon. D. V. Correlation between biodiesel parameters and output of CaO nanoparticles from oyster catalyst on waste cooking oil. Biomass. Conv. Bioref. 2023, 14, 23461-72.
196. Das, V.; Tripathi, A. M.; Borah, M. J.; Dunford, N. T.; Deka, D. Cobalt-doped CaO catalyst synthesized and applied for algal biodiesel production. Renew. Energy. 2020, 161, 1110-9.
197. Cho, Y. B.; Seo, G. High activity of acid-treated quail eggshell catalysts in the transesterification of palm oil with methanol. Bioresour. Technol. 2010, 101, 8515-9.
198. Kalanakoppal Venkatesh, Y.; Mahadevaiah, R.; Haraluru Shankaraiah, L.; Ramappa, S.; Sannagoudar Basanagouda, A. Preparation of a CaO nanocatalyst and its application for biodiesel production using Butea monosperma oil: an optimization study. J. Am. Oil. Chem. Soc. 2018, 95, 635-49.
199. Pandit, P. R.; Fulekar, M. Egg shell waste as heterogeneous nanocatalyst for biodiesel production: optimized by response surface methodology. J. Environ. Manag. 2017, 198, 319-29.
200. Fadhil, A. B.; Al-tikrity, E. T.; Khalaf, A. M. Transesterification of non-edible oils over potassium acetate impregnated CaO solid base catalyst. Fuel 2018, 234, 81-93.
201. Yan, S.; Lu, H.; Liang, B. Supported CaO catalysts used in the transesterification of rapeseed oil for the purpose of biodiesel production. Energy. Fuels. 2007, 22, 646-51.
202. Shi, M.; Zhang, P.; Fan, M.; Jiang, P.; Dong, Y. Influence of crystal of Fe2O3 in magnetism and activity of nanoparticle CaO@Fe2O3 for biodiesel production. Fuel 2017, 197, 343-7.
203. Kesica, Z.; Lukic, I.; Zdujic, M.; Liu, H.; Skala, D. Mechanochemically synthesized CaO ZnO catalyst for biodiesel production. Proc. Eng. 2012, 42, 1169-78.
204. El-sherif, A. A.; Hamad, A. M.; Shams-eldin, E.; et al. Power of recycling waste cooking oil into biodiesel via green CaO-based eggshells/Ag heterogeneous nanocatalyst. Renew. Energy. 2023, 202, 1412-23.
205. Poosumas, J.; Ngaosuwan, K.; Quitain, A. T.; Assabumrungrat, S. Role of ultrasonic irradiation on transesterification of palm oil using calcium oxide as a solid base catalyst. Energy. Convers. Manag. 2016, 120, 62-70.
206. Mansir, N.; Hwa, Teo. S.; Lokman, Ibrahim. M.; Yun, Hin. T. Synthesis and application of waste egg shell derived CaO supported W-Mo mixed oxide catalysts for FAME production from waste cooking oil: Effect of stoichiometry. Energy. Convers. Manag. 2017, 151, 216-26.
207. Mansir, N.; Teo, S. H.; Mijan, N.; Taufiq-yap, Y. H. Efficient reaction for biodiesel manufacturing using bi-functional oxide catalyst. Catal. Commun. 2021, 149, 106201.
208. Reyero, I.; Bimbela, F.; Navajas, A.; Arzamendi, G.; Gandía, L. M. Issues concerning the use of renewable Ca-based solids as transesterification catalysts. Fuel 2015, 158, 558-64.
209. Marques Correia, L.; Cecilia, J. A.; Rodríguez-Castellón, E.; Cavalcante, C. L.; Vieira, R. S. Relevance of the physicochemical properties of calcined quail eggshell (CaO) as a catalyst for biodiesel production. J. Chem. 2017, 2017, 1-12.
210. Chavan, S. B.; Kumbhar, R. R.; Madhu, D.; Singh, B.; Sharma, Y. C. Synthesis of biodiesel from Jatropha curcas oil using waste eggshell and study of its fuel properties. RSC. Adv. 2015, 5, 63596-604.
211. Saleem, M.; Jamil, F.; Qamar, O. A.; et al. Enhancing the catalytic activity of eggshell-derived CaO catalyst and its application in biodiesel production from waste chicken fat. Catalysts 2022, 12, 1627.
212. Birla, A.; Singh, B.; Upadhyay, S.; Sharma, Y. Kinetics studies of synthesis of biodiesel from waste frying oil using a heterogeneous catalyst derived from snail shell. Bioresour. Technol. 2012, 106, 95-100.
213. Ouafi, R.; Haldhar, R.; Mehdaoui, I.; et al. Waste snail shells-derived mixed oxide catalyst for efficient transesterification of vegetable oil: Towards sustainable biodiesel production. Mater. Today. Commun. 2024, 39, 109128.
214. Ahmad, W.; Sethupathi, S.; Munusamy, Y.; Kanthasamy, R. Valorization of raw and calcined chicken eggshell for sulfur dioxide and hydrogen sulfide removal at low temperature. Catalysts 2021, 11, 295.
215. Kouzu, M.; Kasuno, T.; Tajika, M.; Yamanaka, S.; Hidaka, J. Active phase of calcium oxide used as solid base catalyst for transesterification of soybean oil with refluxing methanol. Appl. Catal. A. Gen. 2008, 334, 357-65.
216. Nunes, A.; Castilhos, F. Chemical interesterification of soybean oil and methyl acetate to FAME using CaO as catalyst. Fuel 2020, 267, 117264.
217. Roschat, W.; Siritanon, T.; Yoosuk, B.; Promarak, V. Biodiesel production from palm oil using hydrated lime-derived CaO as a low-cost basic heterogeneous catalyst. Energy. Convers. Manag. 2016, 108, 459-67.
218. Kingkam, W.; Maisomboon, J.; Khamenkit, K.; et al. Preparation of CaO@CeO2 solid base catalysts used for biodiesel production. Catalysts 2024, 14, 240.
219. Ahmad, S.; Chaudhary, S.; Pathak, V. V.; Kothari, R.; Tyagi, V. Optimization of direct transesterification of Chlorella pyrenoidosa catalyzed by waste egg shell based heterogenous nano - CaO catalyst. Renew. Energy. 2020, 160, 86-97.
220. Helwani, Z.; Ramli, M.; Saputra, E.; et al. Impregnation of CaO from eggshell waste with magnetite as a solid catalyst (Fe3O4/CaO) for transesterification of palm oil off-grade. Catalysts 2020, 10, 164.
221. Islam, S.; Basumatary, B.; Rokhum, S. L.; Mochahari, P. K.; Basumatary, S. Advancement in utilization of nanomaterials as efficient and recyclable solid catalyst for biodiesel synthesis. Clean. Chem. Eng. 2022, 3, 100043.
222. Kaur, N.; Ali, A. Biodiesel production via ethanolysis of jatropha oil using molybdenum impregnated calcium oxide as solid catalyst. RSC. Adv. 2015, 5, 13285-95.
223. Naveen, S.; Gopinath, K. P.; Malolan, R.; Ramesh, S. J.; Aakriti, K.; Arun, J. Novel solar parabolic trough collector cum reactor for the production of biodiesel from waste cooking oil using calcium oxide catalyst derived from seashells waste. Chem. Eng. Process. 2020, 157, 108145.
224. Bharti, P.; Singh, B.; Dey, R. K. Process optimization of biodiesel production catalyzed by CaO nanocatalyst using response surface methodology. J. Nanostruct. Chem. 2019, 9, 269-80.
225. Ismail, S.; Ahmed, A. S.; Anr, R.; Hamdan, S. Biodiesel production from castor oil by using calcium oxide derived from mud clam shell. J. Renew. Energy. 2016, 2016, 1-8.
226. Tang, Y.; Xu, J.; Zhang, J.; Lu, Y. Biodiesel production from vegetable oil by using modified CaO as solid basic catalysts. J. Clean. Prod. 2013, 42, 198-203.
227. Sronsri, C.; Sittipol, W.; U-Yen, K. Performance of CaO catalyst prepared from magnetic-derived CaCO3 for biodiesel production. Fuel 2021, 304, 121419.
228. Maneechakr, P.; Karnjanakom, S. Systematic production of biodiesel fuel from palm oil over porous K2O@CaO catalyst derived from waste chicken eggshell via RSM/kinetic/thermodynamic studies. J. Environ. Chem. Eng. 2021, 9, 106542.
229. Thitsartarn, W.; Kawi, S. An active and stable CaO-CeO2 catalyst for transesterification of oil to biodiesel. Green. Chem. 2011, 13, 3423.
230. Yusuff, A. S.; Gbadamosi, A. O.; Atray, N. Development of a zeolite supported CaO derived from chicken eggshell as active base catalyst for used cooking oil biodiesel production. Renew. Energy. 2022, 197, 1151-62.
231. Das, V.; Dunford, N.; Deka, D. Waste utilization and biodiesel production from Desmodesmus maximus grown in swine wastewater using waste eggshells as a catalyst. Aquac. Eng. 2022, 99, 102293.
232. Putra, M. D.; Ristianingsih, Y.; Jelita, R.; Irawan, C.; Nata, I. F. Potential waste from palm empty fruit bunches and eggshells as a heterogeneous catalyst for biodiesel production. RSC. Adv. 2017, 7, 55547-54.
233. Tayeb, A. M.; Abdel-hamid, S. M. S.; Osman, R. M.; Farouk, S. M. Optimization of biodiesel production from waste cooking oil using nano calcium oxide catalyst. Chem. Eng. Technol. 2023, 47, 175-83.
234. Zabeti, M.; Daud, W. M. A. W.; Aroua, M. K. Biodiesel production using alumina-supported calcium oxide: an optimization study. Fuel. Process. Technol. 2010, 91, 243-8.
235. Tan, Y. H.; Abdullah, M. O.; Nolasco-Hipolito, C.; Taufiq-Yap, Y. H. Waste ostrich- and chicken-eggshells as heterogeneous base catalyst for biodiesel production from used cooking oil: Catalyst characterization and biodiesel yield performance. Appl. Energy. 2015, 160, 58-70.
236. Buasri, A.; Chaiyut, N.; Loryuenyong, V.; Worawanitchaphong, P.; Trongyong, S. Calcium oxide derived from waste shells of mussel, cockle, and scallop as the heterogeneous catalyst for biodiesel production. Sci. World. J. 2013, 2013, 460923.
237. Venkat Reddy, C. R.; Oshel, R.; Verkade, J. C. Room-Temperature conversion of soybean oil and poultry fat to biodiesel catalyzed by nanocrystalline calcium oxides. Energy. Fuels. 2006, 20, 1310-4.
238. Li, H.; Wang, Y.; Ma, X.; et al. A novel magnetic CaO-based catalyst synthesis and characterization: Enhancing the catalytic activity and stability of CaO for biodiesel production. Chem. Eng. J. 2020, 391, 123549.
239. Anr, R.; Saleh, A. A.; Islam, M. S.; Hamdan, S.; Maleque, M. A. Biodiesel production from crude jatropha oil using a highly active heterogeneous nanocatalyst by optimizing transesterification reaction parameters. Energy. Fuels. 2015, 30, 334-43.
240. Reddy, A. N. R.; Saleh, A. A.; Islam, M. S.; Hamdan, S. Active razor shell cao catalyst synthesis for jatropha methyl ester production via optimized two-step transesterification. J. Chem. 2017, 2017, 1-20.
241. Aworanti, O. A.; Agbede, O. O.; Popoola, A. O.; et al. Synthesized heterogeneous nano-catalyst from cow teeth for fatty acid methyl esters production through transesterification of waste vegetable frying oil and methanol: characterization and optimization studies. Eng. Rep. 2022, 5, e12605.
242. Badnore, A. U.; Jadhav, N. L.; Pinjari, D. V.; Pandit, A. B. Efficacy of newly developed nano-crystalline calcium oxide catalyst for biodiesel production. Chem. Eng. Process. 2018, 133, 312-9.
243. Al-muhtaseb, A. H.; Jamil, F.; Osman, A. I.; et al. State-of-the-art novel catalyst synthesised from waste glassware and eggshells for cleaner fuel production. Fuel 2022, 330, 125526.
244. Wen, L.; Wang, Y.; Lu, D.; Hu, S.; Han, H. Preparation of KF/CaO nanocatalyst and its application in biodiesel production from Chinese tallow seed oil. Fuel 2010, 89, 2267-71.
245. Hu, S.; Wen, L.; Wang, Y.; Zheng, X.; Han, H. Gas-liquid countercurrent integration process for continuous biodiesel production using a microporous solid base KF/CaO as catalyst. Bioresour. Technol. 2012, 123, 413-8.
246. Wang, Y.; Hu, S.; Guan, Y.; Wen, L.; Han, H. Preparation of mesoporous nanosized KF/CaO-MgO catalyst and its application for biodiesel production by transesterification. Catal. Lett. 2009, 131, 574-8.
247. Bazhdan, E.; Tamjidi, S.; Rouhi, P.; Esmaeili, H. Synthesis of KOH@CaO catalyst by sol-gel method for highly efficient biodiesel production from olive oil. Phys. Chem. Res. 2021, 9, 43-55.
248. Lani, N. S.; Ngadi, N.; Mohammed, Inuwa. I.; Anako, Opotu. L.; Zakaria, Z. Y.; Haron, S. A cleaner approach with magnetically assisted reactor setup over CaO-zeolite/Fe3O4 catalyst in biodiesel production: evaluation of catalytic performance, reusability and life cycle assessment studies. J. Clean. Prod. 2023, 419, 138329.
249. Lani, N. S.; Ngadi, N.; Haron, S.; Mohammed, Inuwa. I.; Anako, Opotu. L. The catalytic effect of calcium oxide and magnetite loading on magnetically supported calcium oxide-zeolite catalyst for biodiesel production from used cooking oil. Renew. Energy. 2024, 222, 119846.
250. Dahdah, E.; Estephane, J.; Moussa, Chalouhi. L.; et al. Transesterification of refined sunflower oil to biodiesel using a CaO/ZSM-5 powder catalyst. Chem. Eng. Technol. 2021, 45, 51-7.
251. Szkudlarek, Ł.; Chałupka-Śpiewak, K.; Maniukiewicz, W.; et al. CaO catalysts supported on ZSM-5 zeolite for biodiesel production via transesterification of rapeseed oil. Appl. Catal. O. Open. 2024, 194, 206999.
252. Lani, N. S.; Ngadi, N.; Inuwa, I. M.; Opotu, L. A.; Zakaria, Z. Y.; Widayat, W. Influence of desilication route of ZSM-5 zeolite in mesoporous zeolite supported calcium oxide catalyst for biodiesel production. Microporous. Mesoporous. Mater. 2022, 343, 112153.
253. Shankar, V.; Jambulingam, R. Waste crab shell derived CaO impregnated Na-ZSM-5 as a solid base catalyst for the transesterification of neem oil into biodiesel. Sustain. Environ. Res. 2017, 27, 273-8.
254. Qasemi, Z.; Jafari, D.; Jafari, K.; Esmaeili, H. Heterogeneous aluminum oxide/calcium oxide catalyzed transesterification of Mespilus germanica triglyceride for biodiesel production. Environ. Prog. Sustain. Energy. 2021, 41, e13738.
255. Dehghani, S.; Haghighi, M.; Vardast, N. Structural/texture evolution of CaO/MCM-41 nanocatalyst by doping various amounts of cerium for active and stable catalyst: biodiesel production from waste vegetable cooking oil. Int. J. Energy. Res. 2019, 43, 3779-93.
256. Dehghani, S.; Haghighi, M. Sono-enhanced dispersion of CaO over Zr-Doped MCM-41 bifunctional nanocatalyst with various Si/Zr ratios for conversion of waste cooking oil to biodiesel. Renew. Energy. 2020, 153, 801-12.
257. Firouzjaee, M. H.; Taghizadeh, M. Optimization of process variables for biodiesel production using the nanomagnetic catalyst CaO/NaY-Fe3O4. Chem. Eng. Technol. 2017, 40, 1140-8.
258. Thitsartarn, W.; Maneerung, T.; Kawi, S. Highly active and durable Ca-doped Ce-SBA-15 catalyst for biodiesel production. Energy 2015, 89, 946-56.
259. Li, H.; Wang, Y.; Han, Z.; et al. Nanosheet like CaO/C derived from Ca-BTC for biodiesel production assisted with microwave. Appl. Energy. 2022, 326, 120045.
260. Al-muhtaseb, A. H.; Jamil, F.; Al-haj, L.; et al. Biodiesel production over a catalyst prepared from biomass-derived waste date pits. Biotechnol. Rep. 2018, 20, e00284.
261. Almasi, S.; Ghobadian, B.; Dehghani, Soufi. M.; Kakavandi, B.; Aubin, J. Calcium oxide anchored on magnetic waste-based activated carbon (MAC@CaO): a sustainable green heterogeneous catalyst for bio-based fuel and lubricant production. Biomass. Bioenergy. 2024, 182, 107071.
262. Zu, Y.; Tang, J.; Zhu, W.; et al. Graphite oxide-supported CaO catalysts for transesterification of soybean oil with methanol. Bioresour. Technol. 2011, 102, 8939-44.
263. Zu, Y.; Liu, G.; Wang, Z.; et al. CaO supported on porous carbon as highly efficient heterogeneous catalysts for transesterification of triacetin with methanol. Energy. Fuels. 2010, 24, 3810-6.
264. Sun, H.; Ma, M.; Fan, M.; et al. Controllable preparation of biomass derived mesoporous activated carbon supported nano-CaO catalysts for biodiesel production. Energy 2022, 261, 125369.
265. Shahraini, N.; Entezari, M. H. Biodiesel from waste oil under mild conditions by a combination of calcium-strontium oxide nanocatalyst and ultrasonic waves. Intl. J. Energy. Res. 2022, 46, 13781-800.
266. Li, H.; Niu, S.; Lu, C.; Li, J. Calcium oxide functionalized with strontium as heterogeneous transesterification catalyst for biodiesel production. Fuel 2016, 176, 63-71.
267. Niju, S.; Meera, K.; Begum, S.; Anantharaman, N. Modification of egg shell and its application in biodiesel production. J. Saudi. Chem. Soc. 2014, 18, 702-6.
268. Das, S.; Anal, J. M. H.; Kalita, P.; Saikia, L.; Rokhum, S. L. Process optimization of biodiesel production using waste snail shell as a highly active nanocatalyst. Int. J. Energy. Res. 2023, 2023, 1-26.
269. Taufiq-yap, Y.; Lee, H.; Hussein, M.; Yunus, R. Calcium-based mixed oxide catalysts for methanolysis of Jatropha curcas oil to biodiesel. Biomass. Bioenergy. 2011, 35, 827-34.
270. Aghabarari, B.; Martinez-huerta, M. V. Biodiesel production using calcined waste filter press cake from a sugar manufacturing facility as a highly economic catalyst. J. Am. Oil. Chem. Soc. 2016, 93, 773-9.
271. Rozina,
272. Granados, M. L.; Poves, M. Z.; Alonso, D. M.; et al. Biodiesel from sunflower oil by using activated calcium oxide. Appl. Catal. B. Environ. 2007, 73, 317-26.
273. Wilkanowicz, S. I.; Hollingsworth, N. R.; Saud, K.; Kadiyala, U.; Larson, R. G. Immobilization of calcium oxide onto polyacrylonitrile (PAN) fibers as a heterogeneous catalyst for biodiesel production. Fuel. Process. Technol. 2020, 197, 106214.
274. Chen, G.; Shan, R.; Shi, J.; Yan, B. Ultrasonic-assisted production of biodiesel from transesterification of palm oil over ostrich eggshell-derived CaO catalysts. Bioresour. Technol. 2014, 171, 428-32.
275. Laskar, I. B.; Rajkumari, K.; Gupta, R.; Chatterjee, S.; Paul, B.; Rokhum, S. L. Waste snail shell derived heterogeneous catalyst for biodiesel production by the transesterification of soybean oil. RSC. Adv. 2018, 8, 20131-42.
276. Lani, N. S.; Ngadi, N.; Inuwa, I. M. New route for the synthesis of silica-supported calcium oxide catalyst in biodiesel production. Renew. Energy. 2020, 156, 1266-77.
277. Hossain, M.; Muntaha, N.; Osman, Goni. L. K. M.; et al. Triglyceride conversion of waste frying oil up to 98.46% using low concentration K+/CaO catalysts derived from eggshells. ACS. Omega. 2021, 6, 35679-91.
278. Zhang, N.; Xue, H.; Hu, R. The activity and stability of CeO2@CaO catalysts for the production of biodiesel. RSC. Adv. 2018, 8, 32922-9.
279. Niju, S.; Niyas, M.; Sheriffa, Begum. K. M. M.; Anantharaman, N. KF-impregnated clam shells for biodiesel production and its effect on a diesel engine performance and emission characteristics. Environ. Prog. Sustain. Energy. 2014, 34, 1166-73.
280. Rahul Soosai, M.; Moorthy, I. M. G.; Varalakshmi, P.; Yonas, C. J. Integrated global optimization and process modelling for biodiesel production from non-edible silk-cotton seed oil by microwave-assisted transesterification with heterogeneous calcium oxide catalyst. J. Clean. Prod. 2022, 367, 132946.
281. Zhao, L.; Qiu, Z.; Stagg-williams, S. M. Transesterification of canola oil catalyzed by nanopowder calcium oxide. Fuel. Process. Technol. 2013, 114, 154-62.
282. Liu, X.; He, H.; Wang, Y.; Zhu, S.; Piao, X. Transesterification of soybean oil to biodiesel using CaO as a solid base catalyst. Fuel 2008, 87, 216-21.
283. Liu, C.; Lv, P.; Yuan, Z.; Yan, F.; Luo, W. The nanometer magnetic solid base catalyst for production of biodiesel. Renew. Energy. 2010, 35, 1531-6.
284. Lani, N. S.; Ngadi, N.; Yahya, N. Y.; Rahman, R. A. Synthesis, characterization and performance of silica impregnated calcium oxide as heterogeneous catalyst in biodiesel production. J. Clean. Prod. 2017, 146, 116-24.
285. Bet-moushoul, E.; Farhadi, K.; Mansourpanah, Y.; Nikbakht, A. M.; Molaei, R.; Forough, M. Application of CaO-based/Au nanoparticles as heterogeneous nanocatalysts in biodiesel production. Fuel 2016, 164, 119-27.
286. Shan, R.; Chen, G.; Yan, B.; Shi, J.; Liu, C. Porous CaO-based catalyst derived from PSS-induced mineralization for biodiesel production enhancement. Energy. Convers. Manag. 2015, 106, 405-13.
287. Ngamcharussrivichai, C.; Meechan, W.; Ketcong, A.; Kangwansaichon, K.; Butnark, S. Preparation of heterogeneous catalysts from limestone for transesterification of vegetable oils - Effects of binder addition. J. Ind. Eng. Chem. 2011, 17, 587-95.
288. Boey, P.; Maniam, G. P.; Hamid, S. A. Biodiesel production via transesterification of palm olein using waste mud crab (Scylla serrata) shell as a heterogeneous catalyst. Bioresour. Technol. 2009, 100, 6362-8.
289. Niju, S.; Sheriffa, Begum. K. M. M.; Anantharaman, N. Preparation of biodiesel from waste frying oil using a green and renewable solid catalyst derived from egg shell. Environ. Prog. Sustain. Energy. 2014, 34, 248-54.
290. Wei, Z.; Xu, C.; Li, B. Application of waste eggshell as low-cost solid catalyst for biodiesel production. Bioresour. Technol. 2009, 100, 2883-5.
291. Zhang, S.; Fu, J.; Xing, S.; et al. Sodium silicates modified calcium oxide as a high-performance solid base catalyst for biodiesel production. Catalysts 2023, 13, 775.
292. Chen, G.; Shan, R.; Li, S.; Shi, J. A biomimetic silicification approach to synthesize CaO-SiO2 catalyst for the transesterification of palm oil into biodiesel. Fuel 2015, 153, 48-55.
293. Ozor, P. A.; Aigbodion, V. S.; Sukdeo, N. I. Modified calcium oxide nanoparticles derived from oyster shells for biodiesel production from waste cooking oil. Fuel. Commun. 2023, 14, 100085.
294. Hu, S.; Guan, Y.; Wang, Y.; Han, H. Nano-magnetic catalyst KF/CaO-Fe3O4 for biodiesel production. Appl. Energy. 2011, 88, 2685-90.
295. Tang, Y.; Li, S.; Dong, J.; Meng, M.; Zhang, J. An efficient CaO-based catalyst for rapid production of biodiesel without glycerol as a by-product using a tri-component reaction. J. Am. Oil. Chem. Soc. 2018, 95, 1487-96.
296. Chakraborty, R.; Bepari, S.; Banerjee, A. Transesterification of soybean oil catalyzed by fly ash and egg shell derived solid catalysts. Chem. Eng. J. 2010, 165, 798-805.
297. Lin, L.; Vittayapadung, S.; Li, X.; Jiang, W.; Shen, A. X. Synthesis of magnetic calcium oxide hollow fiber catalyst for the production of biodiesel. Env. Prog. Sustain. Energy. 2012, 32, 1255-61.
298. Potorochenko, A. N.; Ovchinnikov, A. A.; Rodygin, K. S. Centrifugation, filtration or magnetic extraction: evaluation of catalyst recovery technique in biodiesel production. Bioresour. Technol. Rep. 2026, 33, 102471.
299. Ferreira, G. F.; Pinto, L. F. R.; Filho, R. M.; Fregolente, L. V.; Hayward, J.; Bartley, J. K. Ethanol-based transesterification of rapeseed oil with CaO catalyst: process optimization and validation using microalgal lipids. Catal. Lett. 2025, 155, 84.
300. Ochoa-Gómez, J. R.; Gómez-Jiménez-Aberasturi, O.; Maestro-Madurga, B.; et al. Synthesis of glycerol carbonate from glycerol and dimethyl carbonate by transesterification: Catalyst screening and reaction optimization. Appl. Catal. A. Gen. 2009, 366, 315-24.
301. Roschat, W.; Phewphong, S.; Thangthong, A.; et al. Catalytic performance enhancement of CaO by hydration-dehydration process for biodiesel production at room temperature. Energy. Convers. Manag. 2018, 165, 1-7.
302. Kaur, A.; Ali, A. Surface-modified CaO catalyst for the production of glycerol carbonate. ChemistrySelect 2021, 6, 6102-14.
303. Zheng, H.; He, Y.; Zhu, Y.; Liu, L.; Cui, X. Novel procedure of CO2 capture of the CaO sorbent activator on the reaction of one-part alkali-activated slag. RSC. Adv. 2021, 11, 12476-83.
304. Khawaja, A. S.; Zaheer, M. A.; Ahmad, A.; Mirani, A. A.; Ali, Z. Advances in limitations and opportunities of clean biofuel production to promote decarbonization. Fuel 2023, 342, 127662.
305. Vitidsant, R.; Kodama, S.; Sekiguchi, H. Transesterification using ultrasonic spray of triolein containing CaO particles into methanol vapor in a 3-phase reactor. Processes 2021, 9, 181.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data






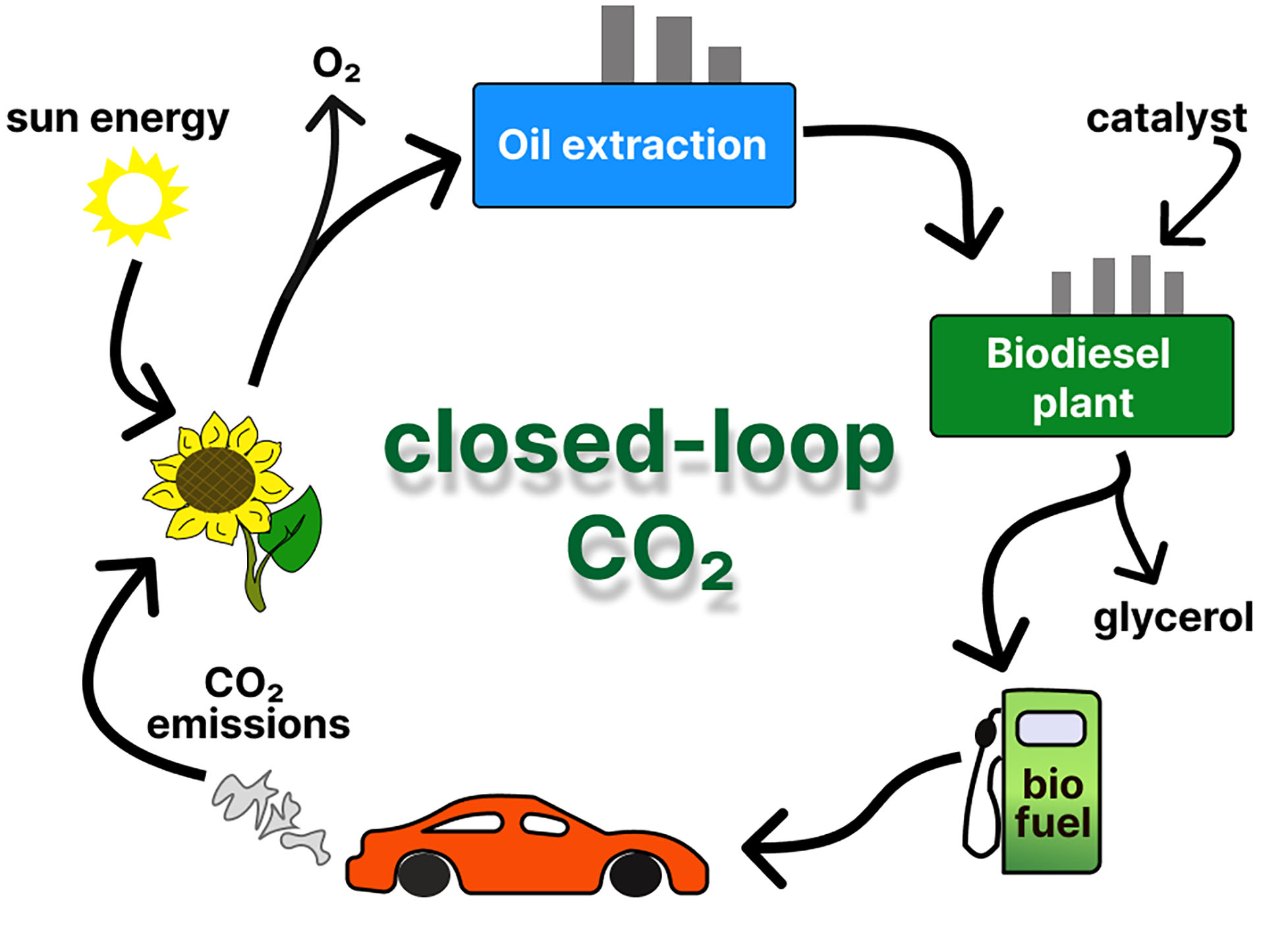
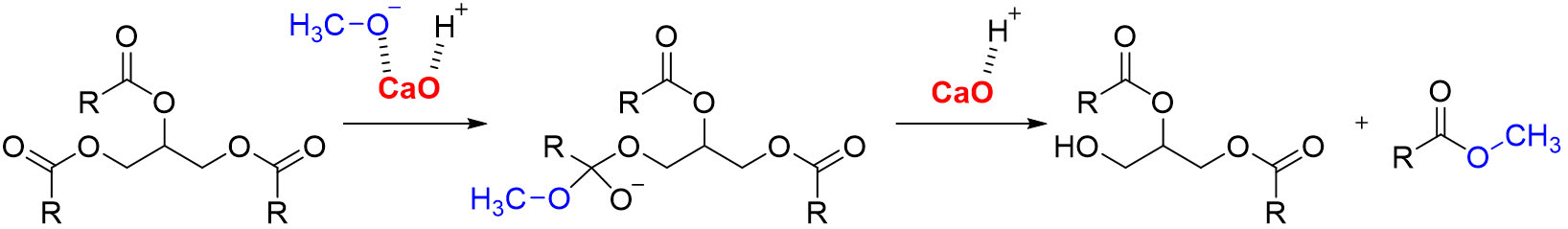
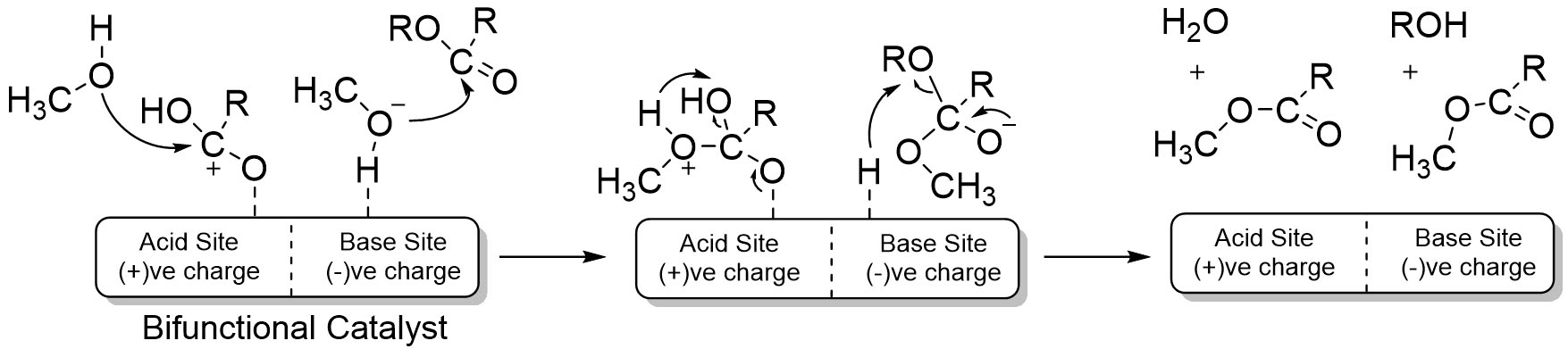

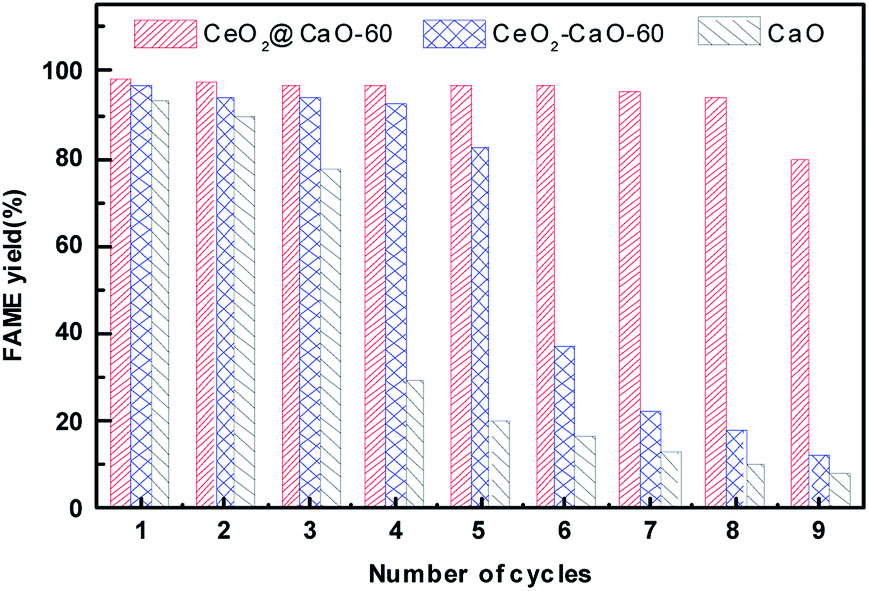
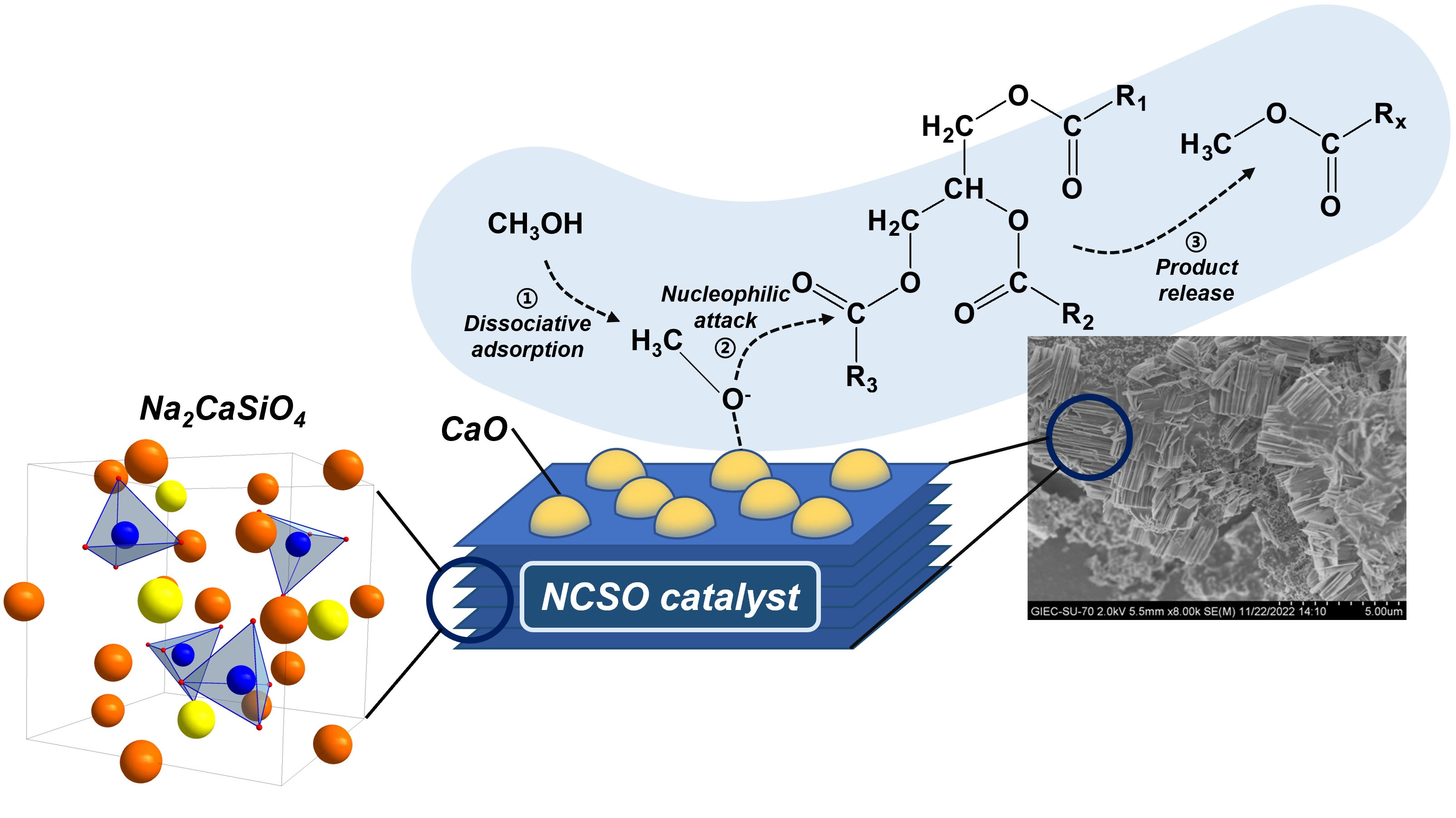
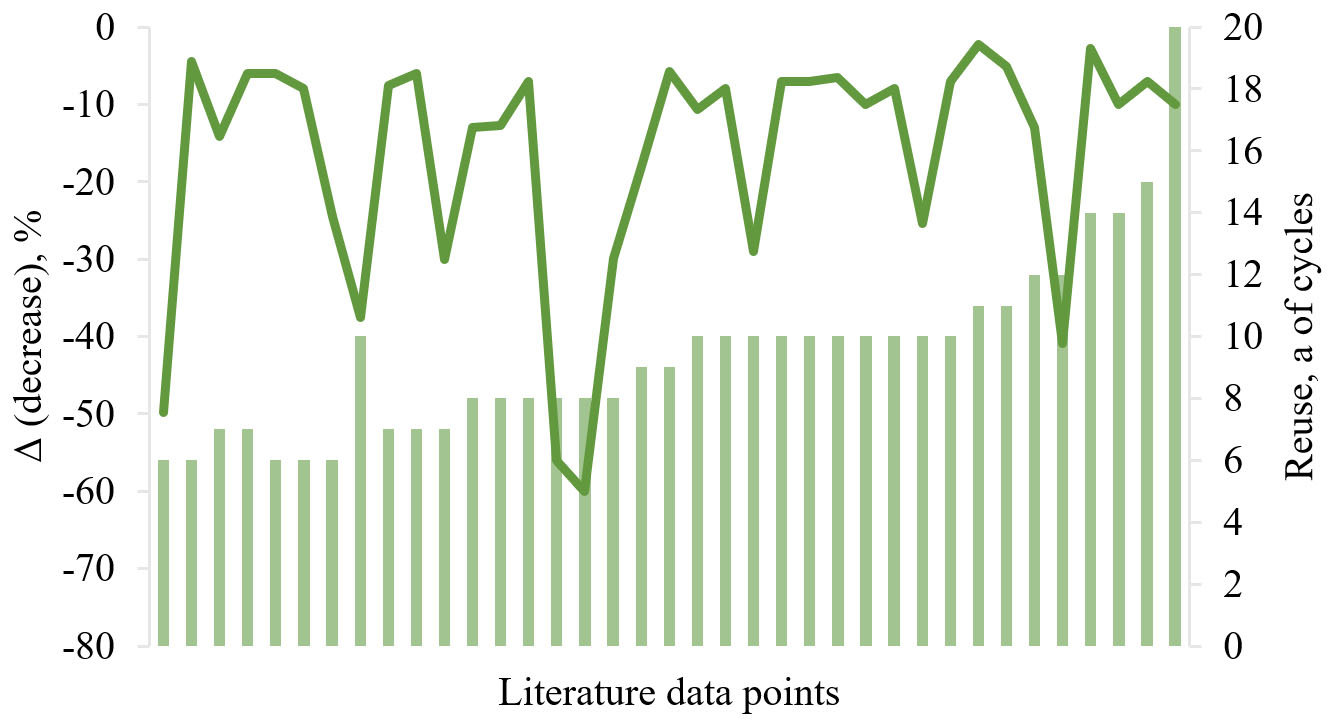












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].