Artificial intelligence in the management of acute kidney injury after cardiac surgery
Abstract
Acute kidney injury (AKI) is a common and serious complication after cardiac surgery, affecting 10%-40% of patients. It worsens patient outcomes and consumes significant healthcare resources. Its pathophysiology is complex and involves ischemia-reperfusion injury, inflammatory responses, and endothelial dysfunction. Artificial intelligence (AI) offers considerable potential to improve the management of this condition. AI models can integrate multimodal data, including preoperative clinical profiles, intraoperative hemodynamics, and postoperative laboratory values, thereby enabling early prediction of AKI. By identifying distinct clinical subtypes, AI may support personalized therapeutic strategies. Furthermore, it may improve prognostic assessments, allowing more precise risk stratification for both cardiac and renal outcomes. However, current applications face challenges, including inconsistent data quality, limited model interpretability, and high implementation costs. Existing models are also constrained by the range of variables they incorporate. Future technological advances may enable the analysis of a broader array of variables, potentially revealing novel biomarkers and clinically useful combinations of indicators. Such progress could advance precision medicine in this field, ultimately improving patient care and optimizing clinical workflows.
Keywords
INTRODUCTION
Epidemiology and burden
According to the Kidney Disease: Improving Global Outcomes (KDIGO) criteria, postoperative acute kidney injury (PO-AKI) is defined as an increase in serum creatinine (Cr) of at least 0.3 mg/dL or 50% from baseline within 48 h after surgery, or a urine output of less than 0.5 mL/kg/h for at least 6 h[1-3]. Cardiac surgery-associated acute kidney injury (CSA-AKI), also referred to as acute kidney injury (AKI) after cardiac surgery, is a common and serious complication of procedures such as coronary artery bypass grafting (CABG) and valve surgery[4-6]. It poses a major threat to patient recovery and survival[7-9] and places a substantial burden on healthcare systems[10,11]. The reported incidence ranges from 10% to 40%[12-15], and 2% to 5% of patients require renal replacement therapy (RRT)[16,17]. Importantly, many of these patients subsequently develop chronic kidney disease (CKD) or end-stage renal disease (ESRD)[14,18]. CSA-AKI is independently associated with a marked increase in both short- and long-term morbidity and mortality[19]. For example, patients who develop AKI have been shown to have a threefold higher 30-day postoperative mortality rate than those who do not, along with hospital stays that are, on average, 5 days longer[5] and substantially higher healthcare costs, reaching approximately $40,000 per patient[20,21].
Pathophysiology
The pathophysiology of CSA-AKI is complex[12,22]. Cardiopulmonary bypass (CPB) is a key initiating factor in renal injury because it disrupts renal hemodynamic balance, causes ischemia and hypoxia in the renal medulla, and triggers oxidative stress[23]. These effects are accompanied by rapid activation of the inflammatory cascade, with the release of cytokines such as tumor necrosis factor-alpha (TNF-α) and interleukin (IL)-6. At the same time, renal tubular epithelial cells undergo apoptosis in response to ischemia and hypoxia. Together, these processes contribute to ischemia-reperfusion injury. In addition, blood contact with the artificial surfaces of the bypass circuit activates the coagulation system, leading to the formation of numerous microthrombi that clog the renal microvasculature and impair renal perfusion. This process, combined with an imbalance in vasoactive mediators such as endothelin-1, exacerbates renal vasoconstriction and promotes microembolism and endothelial injury. In patients with postoperative low cardiac output, activation of the renin-angiotensin-aldosterone system (RAAS) and the sympathetic nervous system further increases renal vascular resistance and promotes sodium and water retention, creating a neurohormonal imbalance that further compromises renal function[24,25].
Perioperative risk factors and the potential role of artificial intelligence
Risk factors for CSA-AKI are often classified into three stages: preoperative, intraoperative, and postoperative[12,22]. Preoperative[26] factors include advanced age (> 70 years), diabetes, CKD, and low left ventricular ejection fraction (LVEF < 35%). Intraoperative factors include prolonged CPB time and contrast exposure[27,28]. Postoperative factors are more heterogeneous. They may include infectious shock, vasopressor requirement, and exposure to nephrotoxic medications such as vancomycin and non-steroidal anti-inflammatory drugs (NSAIDs)[12]. One study[29], for example, found that severe hyperglycemia within 24 h of intensive care unit (ICU) admission after cardiac surgery increased the risks of AKI within 7 days, as well as ICU mortality, and in-hospital mortality.
The severity of this complication and its complex origins underscore the urgent need for improved management strategies. This is where artificial intelligence (AI) may play an important role[30,31]. AI has the capacity to integrate diverse data streams, including preoperative patient characteristics, real-time intraoperative hemodynamics, and high-granularity postoperative ICU monitoring data[32]. This makes it a powerful tool for developing predictive models that provide early warning of CSA-AKI. Furthermore, the integration of AI with cluster analysis can facilitate the identification of patient phenotypes, which may guide individualized intervention strategies, such as goal-directed fluid therapy and the targeted use of anti-inflammatory agents. Continued advances in AI, coupled with large-scale multicenter clinical studies, may further optimize perioperative renal protection and lead to new breakthroughs in the prevention and treatment of this condition.
Literature search strategy
For this narrative review, we systematically searched PubMed, Embase, Web of Science, and the Cochrane Library for articles published through March 2026. We used a combination of Medical Subject Headings terms and keywords, including “acute kidney injury,” “AKI,” “cardiac surgery-associated AKI,” “CSA-AKI,” “cardiac surgical procedures,” “cardiopulmonary bypass,” and “heart surgery,” together with terms such as “artificial intelligence,” “machine learning,” “deep learning,” “predictive model,” “neural network,” “random forest,” and “gradient boosting.” Our search included both pediatric and adult populations.
We included studies that evaluated AI applications in the management of postoperative CSA-AKI, including risk prediction, subtype classification, diagnostic assistance, prognostic assessment, and clinical decision support. All included studies were published in peer-reviewed journals and reported original data with clear model-performance metrics. We excluded editorials, commentaries, conference abstracts, case reports, and non-English articles. Studies not focused on cardiac surgery or AI were also excluded. We manually screened the reference lists of included articles to identify additional relevant studies.
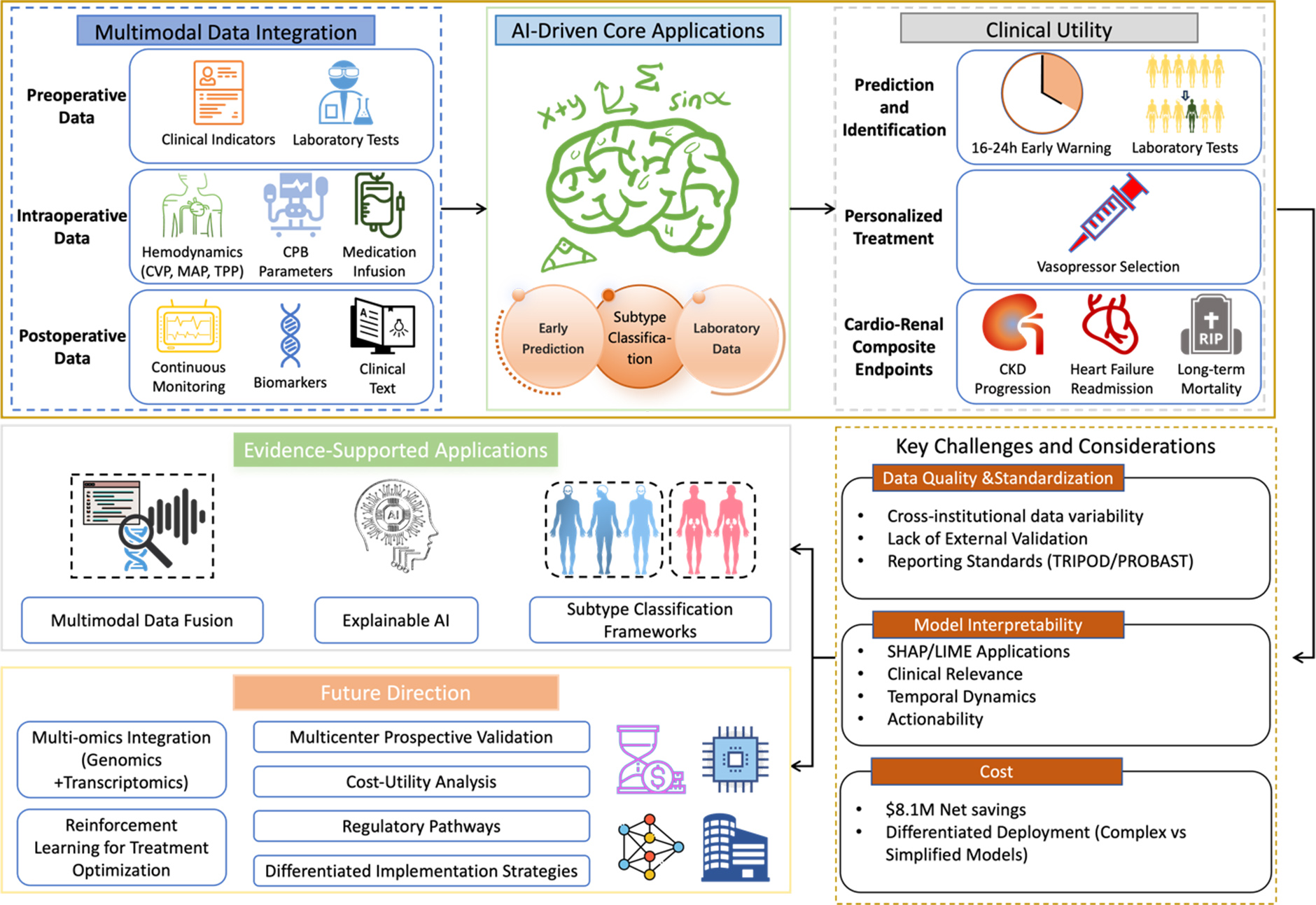
Our search identified many potentially relevant articles. After screening titles, abstracts, and full texts, we selected the primary studies that met all eligibility criteria. Their key features are summarized in Table 1. The remaining articles informed the background, theoretical framework, and discussion sections of this review. The overall framework of the review is illustrated in Figure 1.
Figure 1. Artificial intelligence in cardiac surgery-associated acute kidney injury management: a comprehensive framework. Figure 1 illustrates the multidimensional framework of AI applications in CSA-AKI management. The framework is organized into six core layers: (1) multimodal data integration, encompassing preoperative, intraoperative, and postoperative data sources; (2) AI-driven core applications, including early prediction, subtype classification, and prognostic assessment, with representative studies and performance metrics; (3) clinical utility, highlighting key outcomes such as early warning, personalized treatment, and cardio-renal composite endpoints; (4) key challenges and considerations for clinical translation, including data quality, model interpretability, and implementation costs; (5) evidence-supported applications; and (6) future research directions. AI: Artificial intelligence; CSA-AKI: cardiac surgery-associated acute kidney injury; CVP: central venous pressure; MAP: mean arterial pressure; TPP: tissue perfusion pressure; CPB: cardiopulmonary bypass; CKD: chronic kidney disease; TRIPOD: Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis; PROBAST: Prediction model Risk Of Bias ASsessment Tool; SHAP: SHapley Additive exPlanations; LIME: local interpretable model-agnostic explanations.
Representative studies on artificial intelligence in cardiac surgery-associated acute kidney injury management
| Number | Author | Title | Category | AI application domains |
| 1 | Tseng et al.[33] (2020) | Prediction of the development of acute kidney injury following cardiac surgery by machine learning | Predictive models | Early prediction (RF+XGBoost, AUC 0.839) |
| 2 | Kalisnik et al.[34] (2022) | Artificial intelligence-based early detection of acute kidney injury after cardiac surgery | Predictive models | Early detection within 12 h (AUC 0.88, accuracy 82.1%) |
| 3 | Penny-Dimri et al.[35] (2021) | Machine learning algorithms for predicting and risk profiling of cardiac surgery-associated acute kidney injury | Predictive models | Risk stratification for RRT (GBM AUC 0.85) |
| 4 | Thongprayoon et al.[36] (2022) | Explainable preoperative automated machine learning prediction model for cardiac surgery-associated acute kidney injury | Predictive models | Preoperative autoML with SHAP/LIME explainability |
| 5 | Li et al.[37] (2023) | Development and validation of a machine learning predictive model for cardiac surgery-associated acute kidney injury | Predictive models | CatBoost model, NT-proBNP as key predictor |
| 6 | Gao et al.[38] (2023) | An explainable machine learning model to predict acute kidney injury after cardiac surgery | Predictive models | XGBoost with SHAP, eGFR and ICU Cr as top features |
| 7 | Jiang et al.[39] (2023) | Interpretable machine learning models for early prediction of acute kidney injury after cardiac surgery | Predictive models | LR for any AKI (AUC 0.812), GBC for severe AKI (AUC 0.86) |
| 8 | Song et al.[40] (2024) | Machine learning-based prediction of off-pump coronary artery bypass grafting-associated acute kidney injury | Predictive models | GBDT for OPCABG-AKI (AUC 0.861), insulin use as top predictor |
| 9 | Zhong et al.[41] (2025) | Causal deep learning for real-time detection of cardiac surgery-associated acute kidney injury: derivation and validation in seven time-series cohorts | Predictive models | Causal deep learning (REACT) across 7 cohorts, 16.35 h earlier detection |
| 10 | Baloglu et al.[42] (2026) | Performance of supervised machine learning models for cardiac surgery-associated acute kidney injury in children: multicenter retrospective cohort study | Predictive models | 4-center pediatric study, model performance variability (AUC 0.64-0.83) |
| 11 | Ryan et al.[43] (2023) | Machine learning for dynamic and early prediction of acute kidney injury after cardiac surgery | Predictive models | Ensemble ML on MIMIC-IV, 89% cases predicted before clinical detection |
| 12 | Lee et al.[44] (2018) | Derivation and validation of machine learning approaches to predict acute kidney injury after cardiac surgery | Predictive models | GBM best (AUC 0.78), online risk calculator |
| 13 | Luo et al.[45] (2023) | Machine learning-based prediction of acute kidney injury following pediatric cardiac surgery | Predictive models | XGBoost in 3 centers, AUC 0.912 (internal)/0.889 (external) |
| 14 | Shao et al.[46] (2023) | Development, external validation, and visualization of machine learning models for predicting occurrence of acute kidney injury after cardiac surgery | Predictive models | RF best in external validation, SHAP visualization |
| 15 | Sun et al.[47] (2025) | Explainable machine learning models for early prediction of acute kidney injury after cardiac surgery | Predictive models | Ensemble model AUC 0.856, SVM external validation AUC 0.847 |
| 16 | Chen et al.[48] (2025) | Artificial intelligence-driven prediction of acute kidney injury following acute type a aortic dissection surgery in a chinese population | Predictive models | LightGBM for ATAAD, AUC 0.874, web application |
| 17 | Ranucci et al.[49] (2024) | The multifactorial dynamic perfusion index: a predictive tool of cardiac surgery associated acute kidney injury | Predictive models | MDPI integrating preoperative risk and CPB quality, AUC 0.769 |
| 18 | Thongprayoon et al.[36] (2022) | Explainable preoperative automated machine learning prediction model for cardiac surgery-associated acute kidney injury | Explainable AI studies | SHAP and LIME for patient-specific predictions |
| 19 | Li et al.[37] (2023) | Development and validation of a machine learning predictive model for cardiac surgery-associated acute kidney injury | Explainable AI studies | SHAP identified positive/negative associations with clinical variables |
| 20 | Tseng et al.[33] (2020) | Prediction of the development of acute kidney injury following cardiac surgery by machine learning | Explainable AI studies | SHAP summary and dependence plots for RF model |
| 21 | Zeng et al.[50] (2024) | An interpretable machine learning model to predict off-pump coronary artery bypass grafting-associated acute kidney injury | Explainable AI studies | SHAP-based feature importance, intraoperative urine volume top |
| 22 | Ejmalian et al.[51] (2022) | Prediction of acute kidney injury after cardiac surgery using interpretable machine learning | Explainable AI studies | LIME and Shapley methods, confirmed Cr, CPB time, BS, Alb as top features |
| 23 | Han et al.[52] (2025) | Care guided by tissue oxygenation and haemodynamic monitoring in off-pump coronary artery bypass grafting (Bottomline-CS) | Multimodal data integration | Tissue oxygenation + hemodynamic monitoring for early warning |
| 24 | Li et al.[53] (2025) | Integration of machine learning and large language models for screening and identifying key risk factors of acute kidney injury after cardiac surgery | Multimodal data integration | LLM-simulated expert judgment enhanced risk factor selection |
| 25 | Wang et al.[54] (2025) | Leveraging large language models for preoperative prevention of cardiopulmonary bypass-associated acute kidney injury | Multimodal data integration | LLM-extracted semantics from clinical text, multimodal fusion AUC 0.9201 |
| 26 | Karway et al.[55] (2023) | Development and external validation of multimodal postoperative acute kidney injury risk machine learning models | Multimodal data integration | Structured data + CUI features from notes, AUC 0.82 |
| 27 | Han et al.[56] (2024) | Machine learning with clinical and intraoperative biosignal data for predicting cardiac surgery-associated acute kidney injury | Multimodal data integration | High-resolution intraoperative biosignals improved AUC from 0.767 to 0.840 |
| 28 | Milam et al.[57] (2023) | Derivation and validation of clinical phenotypes of the cardiopulmonary bypass-induced inflammatory response | Subtype classification | K-means identified 3 phenotypes; α phenotype had highest inflammatory/kidney biomarkers |
| 29 | Zhao et al.[58] (2024) | Development and validation of LCMM prediction algorithms to estimate recovery pattern of postoperative AKI in type A aortic dissection | Subtype classification | LCMM identified early recovery (51.8%) and late/no recovery (48.2%) |
| 30 | Giles et al.[59] (2024) | Prediction of acute kidney injury after cardiac surgery with combined arterial and venous intrarenal doppler | Subtype classification | Hierarchical clustering identified 3 intrarenal Doppler subtypes; high-risk subtype had higher AKI risk |
| 31 | Li et al.[60] (2020) | A novel machine learning algorithm, Bayesian networks model, to predict the high-risk patients with cardiac surgery-associated acute kidney injury | Subtype classification | Bayesian networks revealed different variable relationships for AKI and severe AKI |
| 32 | Fan et al.[61] (2023) | Clinical characteristics and risk factors of cardiac surgery associated-acute kidney injury progressed to chronic kidney disease in adults | Prognostic assessment | 19.1% progressed to CKD; risk prediction model AUC 0.859 |
| 33 | Chen et al.[62] (2023) | A novel predictive model for poor in-hospital outcomes in patients with acute kidney injury after cardiac surgery | Prognostic assessment | IL-16, IL-8, ΔSCr predicted RRT/death, AUC 0.947 |
| 34 | Huang et al.[63] (2023) | Development and validation of clinical prediction models for acute kidney injury recovery at hospital discharge in critically ill adults | Prognostic assessment | Cardiac surgery subgroup showed acceptable recovery prediction (AUC 0.71) |
| 35 | Chen et al.[64] (2020) | Novel blood cytokine-based model for predicting severe acute kidney injury and poor outcomes after cardiac surgery | Prognostic assessment | Cytokine model (IFN-γ, IL-16, MIP-1α) C-statistic 0.86-0.87 |
AI-DRIVEN CSA-AKI EARLY PREDICTION: FROM DATA INTEGRATION TO CLINICAL TRANSLATION
The disease burden of CSA-AKI and the urgency of early prediction
AKI after cardiac surgery poses a significant risk to patients during the perioperative period. Studies have shown that it is independently associated with higher in-hospital mortality, with the highest odds of death observed in patients who require acute dialysis[20]. More importantly, a substantial proportion of these patients may progress to CKD. A systematic review found that PO-AKI is associated with an increased risk of long-term mortality, particularly in patients with persistent renal impairment compared with those whose kidney function recovers to baseline levels[22,65]. Patients with AKI have longer hospital stays, incur higher medical costs, and require long-term nephrology follow-up, thereby placing a substantial burden on healthcare systems[17]. Early identification of high-risk patients, followed by timely intervention, has been shown to reduce the incidence of AKI by 40%, mitigate progression to ESRD, and lower healthcare expenditures. This is important from both individualized-care and socioeconomic perspectives[66].
Traditional risk scores have long been used to predict CSA-AKI. Common examples include the Cleveland Clinic Score (CCS) and the European System for Cardiac Operative Risk Evaluation (EuroSCORE)[67,68]. However, these scores typically rely on relatively static preoperative and intraoperative data. They do not incorporate real-time postoperative biomarkers or other dynamic postoperative variables. This limits their ability to accurately predict early-onset CSA-AKI, thereby delaying treatment adjustments and ultimately affecting patient outcomes[34].
In contrast, a machine learning (ML)-based model developed by Penny-Dimri et al. using preoperative and intraoperative data outperformed traditional risk scores in predicting the need for postoperative RRT[35]. Their Gradient Boosting Machine (GBM) algorithm achieved an area under the curve (AUC) of 0.85 (0.01) on the receiver operating characteristic (ROC) curve, outperforming the CCS, which achieved an AUC of 0.81 (0.004). In 2020, Tseng et al.[33] used ML to integrate a wide range of data, including demographic characteristics, preoperative laboratory results, medications, and intraoperative hemodynamic time-series data. They tested several algorithms, including random forest (RF) and extreme gradient boosting (XGBoost). The RF model, which incorporated intraoperative variables, achieved the highest AUC. Kalisnik et al.[34] applied AI specifically to the early detection of CSA-AKI. By tracking serial changes in Cr and estimated glomerular filtration rate (eGFR), their “Detect-A(K)I” model detected CSA-AKI within 12 h after surgery with high accuracy, sensitivity, and specificity.
Despite these promising results, several methodological limitations warrant consideration. Many studies selectively report performance metrics, focusing heavily on AUC while giving limited attention to calibration, clinical utility, and real-world feasibility. For example, the GBM model developed by Penny-Dimri et al. lacked calibration assessment and decision-curve analysis[35]. The XGBoost model developed by Tseng et al. was derived from a single-center cohort and lacked external validation[33]. The “Detect-A(K)I” model, although highly accurate, depends on serial Cr measurements that may not be feasible in all clinical settings and also requires validation in more diverse populations[34].
More recent multicenter studies have begun to address these issues by reporting a more comprehensive set of performance metrics. For instance, Baloglu et al. evaluated 40 ML models across four pediatric cardiac surgery centers[42]. They reported not only AUC but also calibration and decision-curve analyses, showing that a model with good discrimination may still be poorly calibrated. Similarly, Zhong et al. developed a causal deep learning (DL) model, called REACT, using seven international cohorts comprising more than 63,000 patients[41]. They provided calibration plots and decision-curve analysis, demonstrating substantial net benefit across a range of risk thresholds and an average lead time of more than 16 h before clinical detection. These studies highlight the need for rigorous external validation and comprehensive performance assessment before any model is deployed clinically.
Integration and characterization of multimodal data for cardiac surgery
Multimodal data refers to information collected in multiple forms or modalities, often from different sources or sensors[69]. In the context of cardiac surgery, these data may include physiological time-series data, medical imaging, laboratory results, clinical notes, bioelectrical signals[70], and medication infusion records [Table 2]. The integration and analysis of these datasets enable researchers to obtain more comprehensive information, thereby providing high-dimensional feature support for AI modeling[88].
Multimodal data types and storage formats in cardiac surgery perioperative period
| Phase | Category | Data type | Examples | Storage format | References |
| Preoperative | Lab tests | Cardiac function markers | Serum creatinine (Cr), N-terminal pro-B-type natriuretic peptide (NT-proBNP) | Electronic health record (EHR) system (structured database) | [71-73] |
| Imaging data | Cardiac structure analysis | Left ventricular ejection fraction (LVEF), coronary CT angiography (CTA) | Picture archiving and communication system (PACS) (DICOM format) | [74-78] | |
| Intraoperative | Hemodynamic monitoring | Real-time waveform data | Invasive blood pressure (IBP, 100 Hz Sampling), central venous pressure (CVP) | Anesthesia information management system (AIMS) (time-series database) | [79,80] |
| Medication infusion | Vasoactive drug delivery | Epinephrine infusion rate (μg/kg/min), heparin concentration monitoring | Smart infusion pump system (temporal log) | [81,82] | |
| Postoperative | Continuous monitoring | Organ function dynamics | Pulse contour cardiac output (PiCCO), mixed venous oxygen saturation (SvO2) | ICU central monitoring system (High-frequency streaming data) | [52,79,83-85] |
| Biomarkers | Tissue injury evaluation | Troponin I (cTnI), neutrophil gelatinase-associated lipocalin (NGAL) | Laboratory information system (LIS) (numerical reporting system) | [73,86,87] |
Several studies support the value of this approach in CSA-AKI. For example, Han et al. developed an early warning system that fused intraoperative tissue oxygen monitoring data with hemodynamic parameters, helping maintain stable tissue oxygen levels and reducing postoperative complications[52]. Li et al. integrated structured electronic health record (EHR) data with a large language model (LLM), to simulate expert clinical judgment. This approach identified 18 key risk factors for PO-AKI and improved model interpretability[53]. Karway et al. demonstrated that combining structured data, such as demographics, vital signs, and laboratory results, with concept unique identifiers (CUIs), extracted from unstructured clinical notes improved AKI prediction in a multicenter cohort, increasing the AUC from 0.79 to 0.82[55]. Adding high-frequency intraoperative biosignals, such as perfusion pressure waveforms, progressively improved model performance, with the AUC rising from 0.767 with preoperative data alone to 0.840 when postoperative data were included[56].
Looking ahead, integrating multi-omics data, including genomics, transcriptomics, and proteomics, with real-time physiological monitoring could enable truly dynamic and personalized risk stratification. Preliminary studies have already identified associations between specific genetic polymorphisms such as those in IL-6 and Apolipoprotein E (APOE), and the risk of CSA-AKI[89,90]. However, the clinical utility of this approach in dynamic prediction models has not yet been established and requires prospective validation in large, multicenter cohorts. Future work should focus on hypothesis-driven studies that test specific integrative frameworks, rather than exploratory multi-omics analyses that lack a clear mechanistic basis.
CLASSIFICATION OF CSA-AKI SUBTYPES: FROM RESOLVING HETEROGENEITY TO PRECISE INTERVENTION
AKI after cardiac surgery: challenges posed by heterogeneity
AKI after cardiac surgery is a highly heterogeneous clinical syndrome. Despite the shared exposure to cardiac surgery, this heterogeneity is evident in its complex pathophysiology, which involves interacting mechanisms such as ischemic injury, inflammation, and nephrotoxin exposure. It is also reflected in its diverse clinical presentations, which vary widely in severity, disease trajectory, and effects on other organs[14,91-94]. In clinical practice, the effectiveness of traditional one-size-fits-all approaches, such as standard fluid protocols or simple avoidance of nephrotoxins, is limited because these strategies ignore this heterogeneity. This likely explains why several randomized controlled trials (RCT) of such interventions have failed to meet their primary endpoints[95-97]. For instance, one RCT of perioperative spironolactone not only failed to reduce the incidence of KDIGO-defined AKI but also was associated with a significantly increased AKI risk, with no benefit in secondary outcomes such as the need for RRT, ICU length of stay, or mortality. Therefore, establishing a personalized therapeutic framework based on multidimensional omics data and real-time physiological monitoring will be critical for overcoming current therapeutic limitations[94,98].
Subtype classification
In medicine, personalized therapy is defined as the development of targeted intervention strategies informed by multidimensional features. These features may include the patient's genome, pathophysiological mechanisms, clinical phenotype, and biomarkers[99]. A clinical phenotype refers to the observable characteristics of a disease. By contrast, a subtype is a more specific subgroup of patients defined by shared features such as etiology, biomarkers, treatment response, or prognosis[100]. The goal of subtyping is to identify groups with similar underlying disease mechanisms, thereby paving the way for targeted therapeutic interventions[101]. Current approaches to subtyping CSA-AKI can be organized into three dimensions: etiology and pathophysiology, biomarker profiles, and clinical course with recovery patterns [Table 3].
Classification and characteristics of AKI subtypes after cardiac surgery
| Dimension | Subtype | Characteristics | Related factors/biomarkers |
| Etiology & pathophysiology | Ischemia-reperfusion injury[22,102,103] | Renal tubular epithelial cell apoptosis, elevated oxidative stress biomarkers | Intraoperative hypoperfusion, prolonged CPB; NGAL, KIM-1 |
| Inflammation-driven[100,104,105] | Systemic inflammatory response syndrome (SIRS), elevated inflammatory cytokines | Sepsis/postoperative infection; IL-6, IL-8, TNF-α | |
| Endothelial dysfunction[106-108] | Increased vascular permeability, elevated ANG-2/ANG-1 ratio | Risk of postoperative multiorgan failure | |
| Biomarker features | Tubular stress[109-111] | Cell cycle arrest | Urinary [TIMP-2]·[IGFBP7] > 0.3 |
| Tubular injury[112-115] | Acute tubular necrosis, linked to persistent AKI and CKD progression | Elevated urinary NGAL or KIM-1 | |
| Clinical course & recovery | Risk factor subtype[58,61,116,117] | Distinct risk factors for persistent severe AKI (septic shock, catecholamine use, comorbidities) | Cardiac surgery-specific risk factors |
From an etiologic and pathophysiologic standpoint, one subtype is driven by ischemia-reperfusion injury. This subtype is linked to intraoperative hypoperfusion and prolonged CPB and is characterized by renal tubular epithelial cell apoptosis and elevated oxidative stress markers, such as neutrophil gelatinase-associated lipocalin (NGAL) and kidney injury molecule-1 (KIM-1)[103,118]. Animal models have shown that sepsis or postoperative infection can trigger an inflammation-driven phenotype consistent with systemic inflammatory response syndrome (SIRS). This phenotype is characterized by marked elevations in cytokines such as IL-6, IL-8, and TNF-α[119]. An endothelial dysfunction subtype has also been described, featuring increased vascular permeability and a higher angiopoietin (ANG)-2/ANG-1 ratio, which correlated with an elevated risk of postoperative multiorgan failure[106,120]. In terms of biomarker profiles[113], one subtype reflects tubular stress and is identified by a urinary product of tissue inhibitor of metalloproteinases-2 and insulin-like growth factor-binding protein 7 ([TIMP-2]·[IGFBP7]) greater than 0.3. This finding indicates cell cycle arrest and is commonly observed in the early postoperative period[109,114,121-124]. Another subtype is a tubular injury phenotype, marked by elevated urinary NGAL or KIM-1 levels and reflecting acute tubular necrosis as well as an increased risk of persistent AKI and progression to CKD[112,115]. Finally, subtypes can also be defined by clinical course and recovery patterns. For example, the risk factors for persistent severe AKI after cardiac surgery, such as septic shock, catecholamine use, and comorbidities, are distinct from those observed in non-cardiac surgery populations[125].
Based on the aforementioned subtype classification system, the precise identification of patient populations with distinct pathophysiological characteristics is key to advancing personalized treatment. AI models provide an efficient quantitative tool for subtype-driven therapeutic decision-making by integrating multidimensional biomarker and clinical data. The inflammation-endothelial injury classification system for sepsis-associated AKI (SA-AKI), proposed by Bhatraju et al., may offer a useful framework for CSA-AKI[107]. Both conditions share key biomarker patterns and demonstrate heterogeneity in treatment response. For example, the elevated ANG-2/ANG-1 ratio and IL-8 levels observed in some CSA-AKI subtypes are also characteristic of the sepsis AKI-SP2 subtype, suggesting that the classification by Bhatraju et al. could be adapted for this population. In terms of treatment, vasopressin shows efficacy in the sepsis AKI-SP1 subtype, while its use in cardiac surgery patients with vasoplegia similarly improves renal perfusion[107,126]. The three-variable model by Bhatraju et al., incorporating ANG-2/ANG-1, IL-8, and soluble TNF receptor-1 (sTNFR-1), retains discriminative power in CSA-AKI. By enabling dynamic biomarker monitoring, AI can help identify these subtypes early, optimize the timing of interventions, and leverage these pathophysiological links to guide more precise treatment.
However, despite these promising mechanistic insights and potential avenues for personalized therapy, the direct clinical applicability of such subtype-classification frameworks in cardiac surgery populations remains to be rigorously established. Several important caveats warrant consideration. First, most studies to date, including those by Milam et al.[57] and Zhao et al.[58], have derived subtypes from retrospective, often single-center cohorts without prospective validation. The generalizability of these phenotypes across diverse surgical settings and patient populations is therefore uncertain. Second, the inherent overlap among pathophysiological subtypes poses a significant challenge for real-time classification. For instance, a single patient may simultaneously exhibit features of both ischemia-reperfusion injury and inflammation-driven injury, complicating assignment to a discrete therapeutic category. Third, AKI is a dynamic process, and subtype membership may evolve over the course of illness, necessitating serial assessments rather than reliance on a single baseline classification.
PROGNOSTIC EVALUATION OF CARDIAC AND RENAL FUNCTION: FROM MULTIDIMENSIONAL INDICATORS TO PRECISE STRATIFICATION
Prognostic evaluation after CSA-AKI now extends well beyond the immediate postoperative period and focuses on long-term, multi-organ outcomes. Although 40% to 60% of patients achieve complete renal recovery according to KDIGO criteria[9], survivors remain at significantly higher risks of CKD, ESRD, heart failure, and premature death. The concept of cardiorenal syndrome (CRS), as defined by the Acute Dialysis Quality Initiative (ADQI), consensus, captures the bidirectional pathophysiologic interactions between the heart and kidneys[127,128]. This framework provides a rationale for including both cardiac and renal markers in prognostic models. Modern prognostic models have moved beyond static outcome measures toward a more dynamic, multidimensional approach. Key domains now include the timing of renal recovery, which is an independent prognostic factor, with delayed recovery linked to progressively worse long-term kidney outcomes[116].
The inclusion of perioperative hemodynamic parameters such as central venous pressure (CVP)[129], oxygen delivery (DO2)[52], tissue perfusion pressure (TPP)[81], and intrarenal Doppler indices[59] has improved AKI risk prediction. Tracking temporal trajectories of biomarkers such as inflammatory cytokines provides better prognostic information than a single static measurement[62,64]. Molecular subphenotyping has identified distinct AKI subgroups that respond differently to targeted therapies, highlighting the potential for predictive enrichment and more personalized treatment[107]. AKI survivors require prolonged surveillance for multi-organ complications, including cardiovascular events, stroke, dementia, sepsis, and malignancy[130]. Importantly, pre-existing CKD independently predicts both ischemic and bleeding events after complex percutaneous coronary intervention and doubles the risk of contrast-induced AKI[131]. This underscores that cardiac and renal outcomes are inseparable and that composite endpoints provide a more complete picture of a patient's overall prognosis[92].
CHALLENGES AND FUTURE RESEARCH DIRECTIONS FOR AI IN CSA-AKI MANAGEMENT
Current challenges in AI implementation
AI models rely on large, high-quality datasets, but real-world data are often inconsistent. Variability in data collection standards and equipment accuracy across institutions can compromise data quality and reliability. Certain complex models such as DL networks can be highly accurate, but their internal decision-making processes are often opaque. This lack of interpretability, the so-called “black box” problem, can erode clinician trust and limit clinical adoption. Finally, integrating AI into clinical workflows requires substantial investments in personnel, infrastructure, and funding. These include both one-time costs, such as hardware upgrades and system integration, and recurring costs, such as data governance, model maintenance, and personnel training[132-134]. These challenges, including data quality, interpretability, generalizability, and cost, are not unique to this field but are particularly relevant in this context. They currently limit the broader adoption and further development of AI for managing CSA-AKI.
Future research priorities
Enhancing model interpretability
Among these barriers, the lack of interpretability is perhaps the most significant. Recent advances in explainable AI (XAI) offer tools to address this. For instance, Thongprayoon et al.[36] applied SHapley Additive exPlanations (SHAP) and local interpretable model-agnostic explanations (LIME) to an automated ML (autoML) model for preoperative prediction of CSA-AKI, thereby providing patient-specific explanations for the key risk factors identified. Li et al.[37] used SHAP to show that preoperative blood urea nitrogen, prothrombin time, and age were positively associated with CSA-AKI, whereas platelet count and albumin were negatively associated. These findings align well with established clinical knowledge. Similarly, Tseng et al.[33] used SHAP dependence plots to show how intraoperative urine output and red blood cell transfusion influenced their model's predictions. Ejmalian et al.[51] found that combining SHAP with LIME provided robust interpretability, confirming that Cr, CPB time, and albumin were among the most important predictors. Based on these studies, we propose that effective XAI for CSA-AKI should meet several criteria. First, it should demonstrate clinical relevance, with identified features aligning with known pathophysiology. Second, it should demonstrate decision consistency, meaning that model outputs correlate with expert clinical judgment. Third, it should capture temporal dynamics by explaining how time-varying parameters, such as hemodynamic variables, contribute to changing risk. Fourth, it should offer actionability, with explanations suggesting potential interventions, such as adjusting fluids or titrating vasopressors. Future research should focus on integrating these tools directly into clinical workflows to build trust and enable real-time decision support.
Multicenter validation and generalizability
Most AI research in this area is still based on single-center studies or small-scale trials[135]. Future work must prioritize prospective, multicenter trials with predefined protocols, external validation in diverse populations, and comprehensive performance metrics, including calibration and decision-curve analysis. Strict adherence to established guidelines, such as Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis (TRIPOD)[136] and the Prediction model Risk Of Bias ASsessment Tool (PROBAST)[137] will be essential for ensuring transparency and reproducibility.
Multi-omics integration
Currently, AI models for predicting and managing CSA-AKI have largely focused on proteomic and metabolomic data[138]. Future research should integrate other omics layers, such as genomics and transcriptomics, to enrich model inputs and potentially improve predictive accuracy. Analyzing these multi-omics datasets could reveal complex biological pathways and provide deeper insights into AKI pathophysiology, paving the way for interventions targeted to specific disease mechanisms.
Standardization, regulatory pathways, and implementation
To move these tools from research into clinical practice, an international consensus on standardized definitions for CSA-AKI and cardiorenal composite endpoints is urgently needed. This would enable meaningful cross-study comparisons and support regulatory approval. Early engagement with regulatory agencies can help define acceptable performance thresholds and ensure compliance with relevant medical device regulations. Implementation science frameworks should be used to systematically identify barriers to and facilitators of AI-tool adoption across the full range of healthcare settings, from large tertiary cardiac centers to smaller community hospitals. Formal economic evaluations, including cost-utility analyses based on quality-adjusted life years (QALYs) are also needed to guide reimbursement decisions and health policy. Preliminary evidence is promising, with one cost-sensitive model estimating net savings of around eight million dollars through optimized resource allocation and a reduction in complications[139].
Differentiated implementation strategies will be key to ensuring equitable access. High-resource settings might deploy complex multimodal models such as REACT[41], whereas resource-limited environments could use simpler, preoperative models such as autoML tools[36]. This tailored approach can help ensure that the benefits of AI-enhanced perioperative care are available to all patients.
CONCLUSIONS
In summary, AI has shown considerable promise in managing AKI after cardiac surgery, with preliminary advances in prediction, treatment guidance, and prognostic assessment. By integrating multimodal data, these models can detect early signs of CSA-AKI, thereby enabling timely intervention. They can also help tailor treatment plans through more precise subtype classification and improve prognostic accuracy for both cardiac and renal outcomes, thereby guiding postoperative care. However, significant challenges remain. These include inconsistent data quality across institutions, the opacity of many AI models, which limits clinical trust, high implementation costs that restrict access, and the failure to incorporate many variables likely to be critical to the pathophysiology of this condition. Future advancements hinge on optimizing AI algorithms, establishing standardized, high-quality databases, and fostering interdisciplinary collaboration. Priorities include developing interpretable AI systems, integrating broader clinical variables to identify novel biomarkers or combinations, and refining data governance frameworks. These efforts aim to advance precision medicine for AKI after cardiac surgery, ultimately transforming perioperative care and improving patient outcomes in this patient population.
DECLARATIONS
Acknowledgments
We would like to express our sincere thanks to Parnia Ghanad and Maryam Maleki Goli for their professional language editing assistance, which greatly improved the clarity and quality of this manuscript.
Authors’ contributions
Wrote the original draft: Yang S, Shen H
Contributed to the discussion and analysis: Yang J, Wang Y, Zhang J, Xing L, Zhou P, Chen P, Ni H, Yu Y
Acquired the funding and supervised the study: Zhang Z
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
Zhang Z received funding from the Prevention and control of Emerging and Major Infectious Diseases-National Science and Technology Major Project (Nos. 2025ZD01902500 and 2025ZD01902501), the China National Key Research and Development Program (No. 2023YFC3603104), National Natural Science Foundation of China (Nos. 82272180 and 82472243), the Fundamental Research Funds for the Central Universities (No. 226-2025-00024), the Huadong Medicine Joint Funds of the Zhejiang Provincial Natural Science Foundation of China under Grant No. LHDMD24H150001, the Key Research & Development Project of Zhejiang Province (No. 2024C03240), a collaborative scientific project co-established by the Science and Technology Department of the National Administration of Traditional Chinese Medicine and the Zhejiang Provincial Administration of Traditional Chinese Medicine (No. GZY-ZJ-KJ-24082), General Health Science and Technology Program of Zhejiang Province (No. 2024KY1099), the Project of Zhejiang University Longquan Innovation Center (No. ZJDXLQCXZCJBGS2024016), the Beijing Municipal Natural Science Foundation (No. 7252298), Wu Jieping Medical Foundation Special Research Grant (No. 320.6750.2024-23-07), and the Zhejiang Provincial Science and Technology Program for Disease Control and Prevention (No. 2026JKZ042).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Khwaja A. KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract 2012;120:c179-84.
2. Ostermann M, Bellomo R, Burdmann EA, et al. ; Conference Participants. Controversies in acute kidney injury: conclusions from a Kidney Disease: Improving Global Outcomes (KDIGO) Conference. Kidney Int 2020;98:294-309.
3. Kellum JA, Lameire N; KDIGO AKI Guideline Work Group. Diagnosis, evaluation, and management of acute kidney injury: a KDIGO summary (Part 1). Crit Care 2013;17:204.
4. Brown JR, Baker RA, Shore-Lesserson L, et al. The Society of Thoracic Surgeons/Society of Cardiovascular Anesthesiologists/ American Society for Extracorporeal Technology clinical practice guidelines for the prevention of adult cardiac surgery-associated acute kidney injury. Anesth Analg 2023;136:176-84.
5. Hu J, Chen R, Liu S, Yu X, Zou J, Ding X. Global incidence and outcomes of adult patients with acute kidney injury after cardiac surgery: a systematic review and meta-analysis. J Cardiothorac Vasc Anesth 2016;30:82-9.
6. Vandenberghe W, Gevaert S, Kellum JA, et al. Acute kidney injury in cardiorenal syndrome type 1 patients: a systematic review and meta-analysis. Cardiorenal Med 2016;6:116-28.
7. Vives M, Wijeysundera D, Marczin N, Monedero P, Rao V. Cardiac surgery-associated acute kidney injury. Interact Cardiovasc Thorac Surg 2014;18:637-45.
8. Elmistekawy E, McDonald B, Hudson C, et al. Clinical impact of mild acute kidney injury after cardiac surgery. Ann Thorac Surg 2014;98:815-22.
9. Cheng Y, Nie S, Zhao X, et al. Incidence, risk factors and outcome of postoperative acute kidney injury in China. Nephrol Dial Transplant 2024;39:967-77.
10. Mehaffey JH, Hawkins RB, Byler M, et al. ; Virginia Cardiac Services Quality Initiative. Cost of individual complications following coronary artery bypass grafting. J Thorac Cardiovasc Surg 2018;155:875-882.e1.
11. Dasta JF, Kane-Gill SL, Durtschi AJ, Pathak DS, Kellum JA. Costs and outcomes of acute kidney injury (AKI) following cardiac surgery. Nephrol Dial Transplant 2008;23:1970-4.
12. Cheruku SR, Raphael J, Neyra JA, Fox AA. Acute kidney injury after cardiac surgery: prediction, prevention, and management. Anesthesiology 2023;139:880-98.
13. Fuhrman DY, Kellum JA. Epidemiology and pathophysiology of cardiac surgery-associated acute kidney injury. Curr Opin Anaesthesiol 2017;30:60-5.
14. Thiele RH, Isbell JM, Rosner MH. AKI associated with cardiac surgery. Clin J Am Soc Nephrol 2015;10:500-14.
16. O’Neal JB, Shaw AD, Billings FT 4th. Acute kidney injury following cardiac surgery: current understanding and future directions. Crit Care 2016;20:187.
17. James MT, Bhatt M, Pannu N, Tonelli M. Long-term outcomes of acute kidney injury and strategies for improved care. Nat Rev Nephrol 2020;16:193-205.
18. Ishani A, Nelson D, Clothier B, et al. The magnitude of acute serum creatinine increase after cardiac surgery and the risk of chronic kidney disease, progression of kidney disease, and death. Arch Intern Med 2011;171:226-33.
19. Gallagher M, Cass A, Bellomo R, et al. ; POST-RENAL Study Investigators and the ANZICS Clinical Trials Group. Long-term survival and dialysis dependency following acute kidney injury in intensive care: extended follow-up of a randomized controlled trial. PLoS Med 2014;11:e1001601.
20. Lau D, Pannu N, James MT, et al. Costs and consequences of acute kidney injury after cardiac surgery: a cohort study. J Thorac Cardiovasc Surg 2021;162:880-7.
21. Schurle A, Koyner JL. CSA-AKI: incidence, epidemiology, clinical outcomes, and economic impact. J Clin Med 2021;10:5746.
22. Wang Y, Bellomo R. Cardiac surgery-associated acute kidney injury: risk factors, pathophysiology and treatment. Nat Rev Nephrol 2017;13:697-711.
24. Bellomo R, Auriemma S, Fabbri A, et al. The pathophysiology of cardiac surgery-associated acute kidney injury (CSA-AKI). Int J Artif Organs 2008;31:166-78.
25. Massoth C, Zarbock A, Meersch M. Acute kidney injury in cardiac surgery. Crit Care Clin 2021;37:267-78.
26. Karkouti K, Wijeysundera DN, Yau TM, et al. Acute kidney injury after cardiac surgery: focus on modifiable risk factors. Circulation 2009;119:495-502.
27. Duca D, Iqbal S, Rahme E, Goldberg P, de Varennes B. Renal failure after cardiac surgery: timing of cardiac catheterization and other perioperative risk factors. Ann Thorac Surg 2007;84:1264-71.
28. Davenport MS, Khalatbari S, Cohan RH, Dillman JR, Myles JD, Ellis JH. Contrast material-induced nephrotoxicity and intravenous low-osmolality iodinated contrast material: risk stratification by using estimated glomerular filtration rate. Radiology 2013;268:719-28.
29. Ruan J, Huang W, Jiang J, et al. Association between hyperglycemia at ICU admission and postoperative acute kidney injury in patients undergoing cardiac surgery: analysis of the MIMIC-IV database. J Intensive Med 2024;4:526-36.
30. Corte T, Van Hoecke S, De Waele J. Artificial intelligence in infection management in the ICU. Crit Care 2022;26:79.
31. Haug CJ, Drazen JM. Artificial intelligence and machine learning in clinical medicine, 2023. N Engl J Med 2023;388:1201-8.
33. Tseng PY, Chen YT, Wang CH, et al. Prediction of the development of acute kidney injury following cardiac surgery by machine learning. Crit Care 2020;24:478.
34. Kalisnik JM, Bauer A, Vogt FA, et al. Artificial intelligence-based early detection of acute kidney injury after cardiac surgery. Eur J Cardiothorac Surg 2022:62.
35. Penny-Dimri JC, Bergmeir C, Reid CM, Williams-Spence J, Cochrane AD, Smith JA. Machine learning algorithms for predicting and risk profiling of cardiac surgery-associated acute kidney injury. Semin Thorac Cardiovasc Surg 2021;33:735-45.
36. Thongprayoon C, Pattharanitima P, Kattah AG, et al. Explainable preoperative automated machine learning prediction model for cardiac surgery-associated acute kidney injury. J Clin Med 2022:11.
37. Li Q, Lv H, Chen Y, Shen J, Shi J, Zhou C. Development and validation of a machine learning predictive model for cardiac surgery-associated acute kidney injury. J Clin Med 2023:12.
38. Gao Y, Wang C, Dong W, et al. An explainable machine learning model to predict acute kidney injury after cardiac surgery: a retrospective cohort study. Clin Epidemiol 2023;15:1145-57.
39. Jiang J, Liu X, Cheng Z, Liu Q, Xing W. Interpretable machine learning models for early prediction of acute kidney injury after cardiac surgery. BMC Nephrol 2023;24:326.
40. Song Y, Zhai W, Ma S, et al. Machine learning-based prediction of off-pump coronary artery bypass grafting-associated acute kidney injury. J Thorac Dis 2024;16:4535-42.
41. Zhong Q, Cheng Y, Li Z, et al. Causal deep learning for real-time detection of cardiac surgery-associated acute kidney injury: derivation and validation in seven time-series cohorts. Lancet Digit Health ;2025:100901.
42. Baloglu O, Akbasli IT, Morca A, et al. Performance of supervised machine learning models for cardiac surgery-associated acute kidney injury in children: multicenter retrospective cohort study, 2019-2022. Pediatr Crit Care Med 2026;27:3-13.
43. Ryan CT, Zeng Z, Chatterjee S, et al. Machine learning for dynamic and early prediction of acute kidney injury after cardiac surgery. J Thorac Cardiovasc Surg 2023;166:e551-64.
44. Lee HC, Yoon HK, Nam K, et al. Derivation and validation of machine learning approaches to predict acute kidney injury after cardiac surgery. J Clin Med 2018:7.
45. Luo XQ, Kang YX, Duan SB, et al. Machine learning-based prediction of acute kidney injury following pediatric cardiac surgery: model development and validation study. J Med Internet Res 2023;25:e41142.
46. Shao J, Liu F, Ji S, et al. Development, external validation, and visualization of machine learning models for predicting occurrence of acute kidney injury after cardiac surgery. Rev Cardiovasc Med 2023;24:229.
47. Sun Q, Ma M, Du X, Wan X, Cao C, Liang X. Explainable machine learning models for early prediction of acute kidney injury after cardiac surgery. Int Urol Nephrol 2026;58:1499-509.
48. Chen Z, Lu X, Liu M, et al. Artificial intelligence-driven prediction of acute kidney injury following acute type a aortic dissection surgery in a Chinese population. J Cardiothorac Vasc Anesth 2025;39:2729-38.
49. Ranucci M, Di Dedda U, Cotza M, Zamalloa Moreano K. The multifactorial dynamic perfusion index: a predictive tool of cardiac surgery associated acute kidney injury. Perfusion 2024;39:201-9.
50. Zeng Z, Tian X, Li L, Diao Y, Zhang T. An interpretable machine learning model to predict off-pump coronary artery bypass grafting-associated acute kidney injury. Adv Clin Exp Med 2024;33:473-81.
51. Ejmalian A, Aghaei A, Nabavi S, et al. Prediction of acute kidney injury after cardiac surgery using interpretable machine learning. Anesth Pain Med 2022;12:e127140.
52. Han J, Zhai W, Wu Z, et al. ; Bottomline-CS investigation group. Care guided by tissue oxygenation and haemodynamic monitoring in off-pump coronary artery bypass grafting (Bottomline-CS): assessor blind, single centre, randomised controlled trial. BMJ 2025;388:e082104.
53. Li Z, Wang L, Zhang X, Wu A, Liu T. Integration of machine learning and large language models for screening and identifying key risk factors of acute kidney injury after cardiac surgery. Front Med 2025;12:1618222.
54. Wang K, Lin L, Zheng R, Nan S, Lu X, Duan H. Leveraging large language models for preoperative prevention of cardiopulmonary bypass-associated acute kidney injury. Renal Fail 2025;47:2509786.
55. Karway GK, Koyner JL, Caskey J, et al. Development and external validation of multimodal postoperative acute kidney injury risk machine learning models. JAMIA Open 2023;6:ooad109.
56. Han C, Soh S, Kim HI, Song JW, Yoon D. Machine learning with clinical and intraoperative biosignal data for predicting cardiac surgery-associated acute kidney injury. Stud Health Technol Inform 2024;316:286-90.
57. Milam AJ, Liang C, Mi J, et al. Derivation and validation of clinical phenotypes of the cardiopulmonary bypass-induced inflammatory response. Anesth Analg 2023;136:507-17.
58. Zhao W, Wang YP, Tang X, et al. Development and validation of LCMM prediction algorithms to estimate recovery pattern of postoperative AKI in type A aortic dissection: a retrospective study. Front Cardiovasc Med 2024;11:1364332.
59. Giles C, Huard K, Denault A, Beaubien-Souligny W. Prediction of acute kidney injury after cardiac surgery with combined arterial and venous intrarenal doppler. Can J Kidney Health Dis 2024;11:20543581241309976.
60. Li Y, Xu J, Wang Y, et al. A novel machine learning algorithm, Bayesian networks model, to predict the high-risk patients with cardiac surgery-associated acute kidney injury. Clin Cardiol 2020;43:752-61.
61. Fan X, Shao Z, Gao S, et al. Clinical characteristics and risk factors of cardiac surgery associated-acute kidney injury progressed to chronic kidney disease in adults: a retrospective, observational cohort study. Front Cardiovasc Med 2023;10:1108538.
62. Chen Z, Li J, Sun Y, et al. A novel predictive model for poor in-hospital outcomes in patients with acute kidney injury after cardiac surgery. J Thorac Cardiovasc Surg 2023;165:1180-1191.e7.
63. Huang CY, Güiza F, De Vlieger G, et al. Development and validation of clinical prediction models for acute kidney injury recovery at hospital discharge in critically ill adults. J Clin Monit Comput 2023;37:113-25.
64. Chen Z, Chen L, Yao G, Yang W, Yang K, Xiong C. Novel blood cytokine-based model for predicting severe acute kidney injury and poor outcomes after cardiac surgery. J Am Heart Assoc 2020;9:e018004.
65. Corredor C, Thomson R, Al-Subaie N. Long-term consequences of acute kidney injury after cardiac surgery: a systematic review and meta-analysis. J Cardiothorac Vasc Anesth 2016;30:69-75.
66. Hoste EAJ, Kellum JA, Selby NM, et al. Global epidemiology and outcomes of acute kidney injury. Nat Rev Nephrol 2018;14:607-25.
67. Guida P, Mastro F, Scrascia G, Whitlock R, Paparella D. Performance of the European System for Cardiac Operative Risk Evaluation II: a meta-analysis of 22 studies involving 145,592 cardiac surgery procedures. J Thorac Cardiovasc Surg 2014;148:3049-57.e1.
68. Vives M, Candela A, Monedero P, et al. ; Spanish Perioperative Cardiac Surgery Research Group. Improving the performance of the Cleveland Clinic Score for predicting acute kidney injury after cardiac surgery: a prospective multicenter cohort study. Minerva Anestesiol 2024;90:245-53.
69. Acosta JN, Falcone GJ, Rajpurkar P, Topol EJ. Multimodal biomedical AI. Nat Med 2022;28:1773-84.
70. Jalagam MK, Mittal VK. Studies on biomedical signal processing: a review. In: International Conference on Signal Processing & Communication Engineering Systems: Spaces-2021; 2021 Jun 21-22; Andhra Pradesh, India. Melville: AIP Publishing; 2024.
71. Patel UD, Garg AX, Krumholz HM, et al. ; Translational Research Investigating Biomarker Endpoints in Acute Kidney Injury (TRIBE-AKI) Consortium. Preoperative serum brain natriuretic peptide and risk of acute kidney injury after cardiac surgery. Circulation 2012;125:1347-55.
72. Wang J, Dong Y, Zhao B, Liu K. Preoperative NT-proBNP and LVEF for the prediction of acute kidney injury after noncardiac surgery: a single-centre retrospective study. BMC Anesthesiol 2022;22:196.
73. Perez SC, Manghelli JL, Khiabani AJ, et al. Biomarkers of postoperative cardiac surgery-associated acute kidney injury: narrowing the field. JTCVS Open 2025;25:264-74.
74. Robinson S, Ring L, Oxborough D, et al. The assessment of left ventricular diastolic function: guidance and recommendations from the British Society of Echocardiography. Echo Res Pract 2024;11:16.
75. Kim C, Park CH, Lee BY, et al. 2024 consensus statement on coronary stenosis and plaque evaluation in CT angiography from the Asian Society of Cardiovascular Imaging-Practical Tutorial (ASCI-PT). Korean J Radiol ;25:331-42.
76. Chinese Society of Cardiology, Chinese Medical Association; Chinese Geriatrics Society; Editorial Board of Chinese Journal of Cardiology. Expert consensus on the application of coronary CTA in risk stratification, diagnosis and treatment of chronic coronary syndrome. Zhonghua Xin Xue Guan Bing Za Zhi 2025;53:16-27. (in Chinese).
77. Tsai PC, Lee TH, Kuo KC, et al. Histopathology images predict multi-omics aberrations and prognoses in colorectal cancer patients. Nat Commun 2023;14:2102.
78. Pinker K, Chin J, Melsaether AN, Morris EA, Moy L. Precision medicine and radiogenomics in breast cancer: new approaches toward diagnosis and treatment. Radiology 2018;287:732-47.
79. Della Rocca G, Costa MG, Coccia C, et al. Cardiac output monitoring: aortic transpulmonary thermodilution and pulse contour analysis agree with standard thermodilution methods in patients undergoing lung transplantation. Can J Anaesth 2003;50:707-11.
80. Miles TJ, Guinn MT, Tan X, et al. Tissue perfusion pressure: a novel hemodynamic measure to assess risk of acute kidney injury after cardiac surgery. J Thorac Cardiovasc Surg 2026;171:455-462.e3.
81. Tamura T, Yokoyama T. The TEG6s-Derived R ratio accurately reflects the anti-Xa level after cardiac surgery: a proof-of-concept study. Perfusion ;2025:2676591251388353.
82. Abin AA, Molla A, Ejmalian A, et al. Anesthetic management recommendations using a machine learning algorithm to reduce the risk of acute kidney injury after cardiac surgeries. Anesth Pain Med 2024;14:e143853.
83. Badas JC, Guembe J, Novials C, et al. Usefulness of the recruitment-to-inflation ratio and pulmonary compliance for assessing pulmonary recruitability in cardiac surgery patients: a prospective study. J Cardiothorac Vasc Anesth 2025;39:2970-7.
84. Ferreira FM, Araujo DD, Dantas GM, Cunha LCC, Zeferino SP, Galas FB. Goal-directed therapy with continuous SvcO2 monitoring in pediatric cardiac surgery: the PediaSat single-center randomized trial. Braz J Anesthesiol 2025;75:844614.
85. Lankadeva YR, Cochrane AD, Marino B, et al. Strategies that improve renal medullary oxygenation during experimental cardiopulmonary bypass may mitigate postoperative acute kidney injury. Kidney Int 2019;95:1338-46.
86. Kiss N, Papp M, Turan C, et al. Combination of urinary biomarkers can predict cardiac surgery-associated acute kidney injury: a systematic review and meta-analysis. Ann Intensive Care 2025;15:45.
87. Parikh CR, Devarajan P, Zappitelli M, et al. ; TRIBE-AKI Consortium. Postoperative biomarkers predict acute kidney injury and poor outcomes after pediatric cardiac surgery. J Am Soc Nephrol 2011;22:1737-47.
88. Liu J, Cen X, Yi C, et al. Challenges in AI-driven biomedical multimodal data fusion and analysis. Genom Proteom Bioinform 2025;23:qzaf011.
89. Jouan J, Golmard L, Benhamouda N, et al. Gene polymorphisms and cytokine plasma levels as predictive factors of complications after cardiopulmonary bypass. J Thorac Cardiovasc Surg 2012;144:467-73, 473.e1.
90. Averdunk L, Bernhagen J, Fehnle K, et al. the macrophage migration inhibitory factor (MIF) promoter polymorphisms (rs3063368, rs755622) predict acute kidney injury and death after cardiac surgery. J Clin Med 2020:9.
91. Basu RK, Gist KM. Prevention of acute kidney injury after cardiac surgery: when fixing broken hearts, is breaking kidneys avoidable? Clin J Am Soc Nephrol 2021;16:1459-61.
92. Pickkers P, Darmon M, Hoste E, et al. Acute kidney injury in the critically ill: an updated review on pathophysiology and management. Intensive Care Med 2021;47:835-50.
93. Barasch J, Zager R, Bonventre JV. Acute kidney injury: a problem of definition. Lancet 2017;389:779-81.
94. Kellum JA, Prowle JR. Paradigms of acute kidney injury in the intensive care setting. Nat Rev Nephrol 2018;14:217-30.
95. Smith OM, Wald R, Adhikari NK, Pope K, Weir MA, Bagshaw SM. ; Canadian Critical Care Trials Group. Standard versus accelerated initiation of renal replacement therapy in acute kidney injury (STARRT-AKI): study protocol for a randomized controlled trial. Trials 2013;14:320.
96. Barba-Navarro R, Tapia-Silva M, Garza-Garcia C, et al. The effect of spironolactone on acute kidney injury after cardiac surgery: a randomized, placebo-controlled trial. Am J Kidney Dis 2017;69:192-9.
97. Miralles Bagán J, Parrilla Quiles L, Paniagua Iglesias P, et al. The potential role of albumin in reducing cardiac surgery-associated acute kidney injury: a randomized controlled trial. J Cardiothorac Vasc Anesth 2025;39:453-60.
98. Stanski NL, Rodrigues CE, Strader M, Murray PT, Endre ZH, Bagshaw SM. Precision management of acute kidney injury in the intensive care unit: current state of the art. Intensive Care Med 2023;49:1049-61.
99. Vaara ST, Forni LG, Joannidis M. Subphenotypes of acute kidney injury in adults. Curr Opin Crit Care 2022;28:599-604.
100. Zarbock A, Nadim MK, Pickkers P, et al. Sepsis-associated acute kidney injury: consensus report of the 28th Acute Disease Quality Initiative workgroup. Nat Rev Nephrol 2023;19:401-17.
101. Vaara ST, Bhatraju PK, Stanski NL, et al. Subphenotypes in acute kidney injury: a narrative review. Crit Care 2022;26:251.
102. Rosner MH, Okusa MD. Acute kidney injury associated with cardiac surgery. Clin J Am Soc Nephrol 2006;1:19-32.
103. Mishra J, Dent C, Tarabishi R, et al. Neutrophil gelatinase-associated lipocalin (NGAL) as a biomarker for acute renal injury after cardiac surgery. Lancet 2005;365:1231-8.
104. Zhang WR, Garg AX, Coca SG, et al. ; TRIBE-AKI Consortium. Plasma IL-6 and IL-10 concentrations predict AKI and long-term mortality in adults after cardiac surgery. J Am Soc Nephrol 2015;26:3123-32.
105. Greenberg JH, Whitlock R, Zhang WR, et al. ; TRIBE-AKI Consortium. Interleukin-6 and interleukin-10 as acute kidney injury biomarkers in pediatric cardiac surgery. Pediatr Nephrol 2015;30:1519-27.
106. Jongman RM, van Klarenbosch J, Molema G, Zijlstra JG, de Vries AJ, van Meurs M. Angiopoietin/Tie2 dysbalance is associated with acute kidney injury after cardiac surgery assisted by cardiopulmonary bypass. PLoS ONE 2015;10:e0136205.
107. Bhatraju PK, Zelnick LR, Herting J, et al. Identification of acute kidney injury subphenotypes with differing molecular signatures and responses to vasopressin therapy. Am J Respir Crit Care Med 2019;199:863-72.
108. Robinson-Cohen C, Katz R, Price BL, et al. Association of markers of endothelial dysregulation Ang1 and Ang2 with acute kidney injury in critically ill patients. Crit Care 2016;20:207.
109. Kashani K, Al-Khafaji A, Ardiles T, et al. Discovery and validation of cell cycle arrest biomarkers in human acute kidney injury. Crit Care 2013;17:R25.
110. Meersch M, Schmidt C, Van Aken H, et al. Urinary TIMP-2 and IGFBP7 as early biomarkers of acute kidney injury and renal recovery following cardiac surgery. PLoS ONE 2014;9:e93460.
111. Kimmel M, Schanz M, Alscher MD. Risk prediction of acute kidney injury by [TIMP-2]•[IGFBP7]. Drugs Today 2017;53:349-56.
112. Parikh CR, Coca SG, Thiessen-Philbrook H, et al. ; TRIBE-AKI Consortium. Postoperative biomarkers predict acute kidney injury and poor outcomes after adult cardiac surgery. J Am Soc Nephrol 2011;22:1748-57.
113. Koyner JL, Garg AX, Coca SG, et al. ; TRIBE-AKI Consortium. Biomarkers predict progression of acute kidney injury after cardiac surgery. J Am Soc Nephrol 2012;23:905-14.
114. Haase M, Devarajan P, Haase-Fielitz A, et al. The outcome of neutrophil gelatinase-associated lipocalin-positive subclinical acute kidney injury: a multicenter pooled analysis of prospective studies. J Am Coll Cardiol 2011;57:1752-61.
115. Coca SG, Garg AX, Thiessen-Philbrook H, et al. ; TRIBE-AKI Consortium. Urinary biomarkers of AKI and mortality 3 years after cardiac surgery. J Am Soc Nephrol 2014;25:1063-71.
116. Siew ED, Abdel-Kader K, Perkins AM, et al. Timing of recovery from moderate to severe AKI and the risk for future loss of kidney function. Am J Kidney Dis 2020;75:204-13.
117. Kellum JA, Sileanu FE, Bihorac A, Hoste EA, Chawla LS. Recovery after acute kidney injury. Am J Respir Crit Care Med 2017;195:784-91.
118. Liu L, Hu Z. When to start renal replacement therapy in acute kidney injury: what are we waiting for? J Intensive Med 2024;4:341-6.
119. Singbartl K, Bishop JV, Wen X, et al. Differential effects of kidney-lung cross-talk during acute kidney injury and bacterial pneumonia. Kidney Int 2011;80:633-44.
120. Meurs M, Kümpers P, Ligtenberg JJ, Meertens JH, Molema G, Zijlstra JG. Bench-to-bedside review: angiopoietin signalling in critical illness - a future target? Crit Care 2009;13:207.
121. Schanz M, Wasser C, Allgaeuer S, et al. Urinary [TIMP-2]·[IGFBP7]-guided randomized controlled intervention trial to prevent acute kidney injury in the emergency department. Nephrol Dial Transplant 2019;34:1902-9.
122. Murray PT, Mehta RL, Shaw A, et al. ; ADQI 10 workgroup. Potential use of biomarkers in acute kidney injury: report and summary of recommendations from the 10th Acute Dialysis Quality Initiative consensus conference. Kidney Int 2014;85:513-21.
123. Bihorac A, Chawla LS, Shaw AD, et al. Validation of cell-cycle arrest biomarkers for acute kidney injury using clinical adjudication. Am J Respir Crit Care Med 2014;189:932-9.
124. Zaouter C, Potvin J, Bats ML, Beauvieux MC, Remy A, Ouattara A. A combined approach for the early recognition of acute kidney injury after adult cardiac surgery. Anaesth Crit Care Pain Med 2018;37:335-41.
125. Gómez H, Chen X, Minturn JS, et al. Persistent severe acute kidney injury is a major modifiable determinant of outcome during critical illness. Intensive Care Med 2025;51:542-55.
126. Morelli A, Ertmer C, Rehberg S, et al. Phenylephrine versus norepinephrine for initial hemodynamic support of patients with septic shock: a randomized, controlled trial. Crit Care 2008;12:R143.
127. Ronco C, McCullough P, Anker SD, et al. ; Acute Dialysis Quality Initiative (ADQI) Consensus Group. Cardio-renal syndromes: report from the consensus conference of the acute dialysis quality initiative. Eur Heart J 2010;31:703-11.
128. House AA, Anand I, Bellomo R, et al. ; Acute Dialysis Quality Initiative Consensus Group. Definition and classification of Cardio-Renal Syndromes: workgroup statements from the 7th ADQI Consensus Conference. Nephrol Dial Transplant 2010;25:1416-20.
129. Damman K, van Deursen VM, Navis G, Voors AA, van Veldhuisen DJ, Hillege HL. Increased central venous pressure is associated with impaired renal function and mortality in a broad spectrum of patients with cardiovascular disease. J Am Coll Cardiol 2009;53:582-8.
130. Ostermann M, Lumlertgul N, Jeong R, See E, Joannidis M, James M. Acute kidney injury. Lancet 2025;405:241-56.
131. Di Muro FM, Sartori S, Vogel B, et al. Clinical outcomes after complex and high-risk percutaneous coronary intervention according to baseline chronic kidney disease. Clin Res Cardiol 2025;114:1049-58.
132. Burns ML, Chen SY, Tsai CA, et al. Generative AI costs in large healthcare systems, an example in revenue cycle. NPJ Digit Med 2025;8:579.
133. Lee JT, Shen TK, Liu VT, et al. Costing methods for artificial intelligence: systematic review and recommended cost inventory for in health technology assessment. medRxiv 2025.
135. Ozrazgat-Baslanti T, Loftus TJ, Ren Y, Ruppert MM, Bihorac A. Advances in artificial intelligence and deep learning systems in ICU-related acute kidney injury. Curr Opin Crit Care 2021;27:560-72.
136. Collins GS, Reitsma JB, Altman DG, Moons KG. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD statement. BMJ 2015;350:g7594.
137. Kaul T, Damen JAA, Wynants L, et al. Assessing the quality of prediction models in health care using the Prediction model Risk Of Bias ASsessment Tool (PROBAST): an evaluation of its use and practical application. J Clin Epidemiol 2025;181:111732.
138. Dubin RF, Rhee EP. Proteomics and metabolomics in kidney disease, including insights into etiology, treatment, and prevention. Clin J Am Soc Nephrol 2020;15:404-11.
139. Al-Absi DT, Simsekler MCE, Anwar S, Omar MA. Value-driven healthcare: cost-benefit ML approach to AKI management in cardiac surgery. In: 2024 IEEE International Conference on Technology Management, Operations and Decisions (ICTMOD); 2024 Nov 4-6; Sharjah, United Arab Emirates. New York: IEEE; 2024. pp. 1-7.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].