When minimally invasive pancreatoduodenectomy becomes risky in elderly patients: BMI and aCCI define the crossover point for surgical safety
Abstract
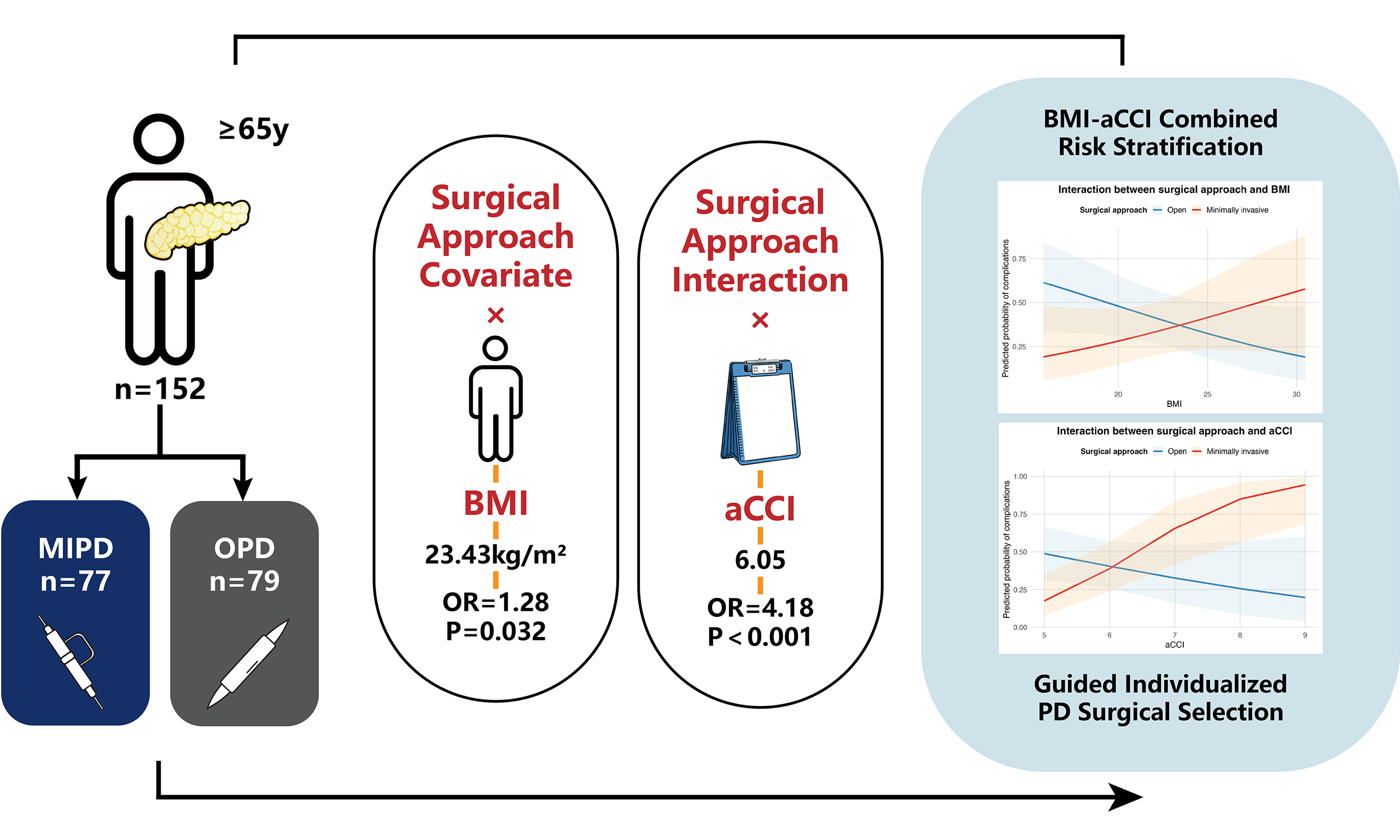
Aim: To assess whether the surgical approach - open pancreatoduodenectomy (OPD) vs. minimally invasive pancreatoduodenectomy (MIPD) - affects short-term postoperative complications in elderly patients, and to determine whether body mass index (BMI) and age-adjusted Charlson Comorbidity Index (aCCI) modify this effect.
Methods: This retrospective cohort study included 156 elderly patients (≥ 65 years) undergoing pancreatoduodenectomy (PD) between 2020 and 2025. Multivariable logistic regression with interaction terms evaluated effect modification by BMI and aCCI on 30-day postoperative complications. Predicted probability-based scenario analyses were used for risk stratification. Exploratory computed tomography (CT) analyses were performed in a representative subgroup (n = 80).
Results: Overall complication rates were comparable between OPD and MIPD, and surgical approach alone was not an independent predictor of complications. Significant interactions were identified between surgical approach and BMI [odds ratio (OR) = 1.28, P = 0.032] and aCCI (OR = 4.18, P < 0.001). Scenario analyses showed that MIPD was associated with lower predicted complication risk in patients with aCCI ≤ 6 and BMI < 23.43 kg/m2, whereas OPD was safer in patients with aCCI > 6. CT analysis demonstrated fewer complications after MIPD in patients with low subcutaneous adipose tissue.
Conclusion: A combined BMI–aCCI–based risk stratification framework supports individualized surgical approach selection in elderly patients undergoing PD.
Keywords
INTRODUCTION
Pancreatoduodenectomy (PD) is the standard surgical procedure for treating space-occupying lesions of the pancreatic head, ampulla of Vater, and distal bile duct[1]. The surgical resection involves removal of the pancreatic head, duodenum, distal stomach, and gallbladder, followed by complex gastrointestinal reconstruction, including pancreaticojejunostomy, choledochojejunostomy, and gastrojejunal anastomoses. Because of its resection and reconstruction of multiple vital organs, PD challenges patients’ physiological condition. It is consistently regarded as one of the most challenging surgical procedures in abdominal surgery. Thanks to advances in anesthesia and surgical techniques, along with improved perioperative management, the safety of PD has been strongly enhanced. In some high-volume pancreatic surgery centers, perioperative mortality has decreased to below 5%[2]. However, PD remains one of the most technically demanding procedures in general surgery and continues to impose substantial physiological stress on patients. This challenge is particularly evident in elderly patients. Due to their reduced physiological reserve, age-related organ dysfunction and a substantial comorbidity burden, the rate of perioperative complication for the elderly is markedly higher than that for younger patients[3].
In recent years, advances in minimally invasive surgical techniques have led to the widespread global adoption of minimally invasive pancreatoduodenectomy (MIPD), including laparoscopic and robotic approaches[4]. Previous studies have shown that, in the general population, MIPD offers advantages over open surgery by reducing intraoperative blood loss, shortening postoperative recovery, and improving cosmetic outcomes[5,6]. Furthermore, robotic surgery has partially overcome the technical limitations of laparoscopic PD by providing three-dimensional visualization and enhanced instrument dexterity[7,8]. However, the applicability and safety of minimally invasive surgery in elderly patients remain controversial. On the one hand, several studies suggest that elderly patients can safely undergo minimally invasive surgery[9]; on the other hand, some reports indicate that elderly or high-risk individuals - particularly those aged over 80 years - may have higher rates of postoperative complications and short-term mortality after minimally invasive procedures[10]. Therefore, in elderly patients, careful and individual risk assessment is essential for the decision of the most appropriate surgical approach.
Beyond physical age, physiological status and underlying comorbidities also influence postoperative outcomes in elderly patients. The Charlson Comorbidity Index (CCI), which reflects overall chronic disease burden, is a well-established predictor for postoperative complications and short-term mortality in elderly surgical patients[11]. The age-adjusted CCI (aCCI) further refines these criteria for evaluating the health status of elderly patients[12]. Additionally, body mass index (BMI) is closely associated with perioperative risk[13]. Patients with high BMI often have substantial intra-abdominal adiposity, which can limit operative visibility, increase intraoperative blood loss, and complicate anastomotic procedures[14]. Conversely, patients with low BMI or malnutrition are more susceptible to delayed wound healing and postoperative infections[15]. Therefore, the selection of surgical approach in elderly patients undergoing PD should rely on a comprehensive assessment, including physiological reserve, nutritional status, and comorbidity burden, instead of age alone serving as a contraindication.
This study aims to integrate multidimensional geriatric patient data to comprehensively evaluate the effects of surgical approach [open pancreatoduodenectomy (OPD) vs. MIPD] and the modifying roles of BMI and the aCCI on short-term postoperative complications in elderly patients. By systematically analyzing the interactions among these factors, our study seeks to generate evidence to guide surgical approach selection in elderly patients undergoing PD and to support personalized surgical planning. These findings have important clinical implications for optimizing surgical decision-making, enhancing perioperative safety, reducing postoperative complications, and improving outcomes in elderly patients. The study will also contribute to advancing precision and individualized approaches in geriatric pancreatic surgery.
METHODS
Study design
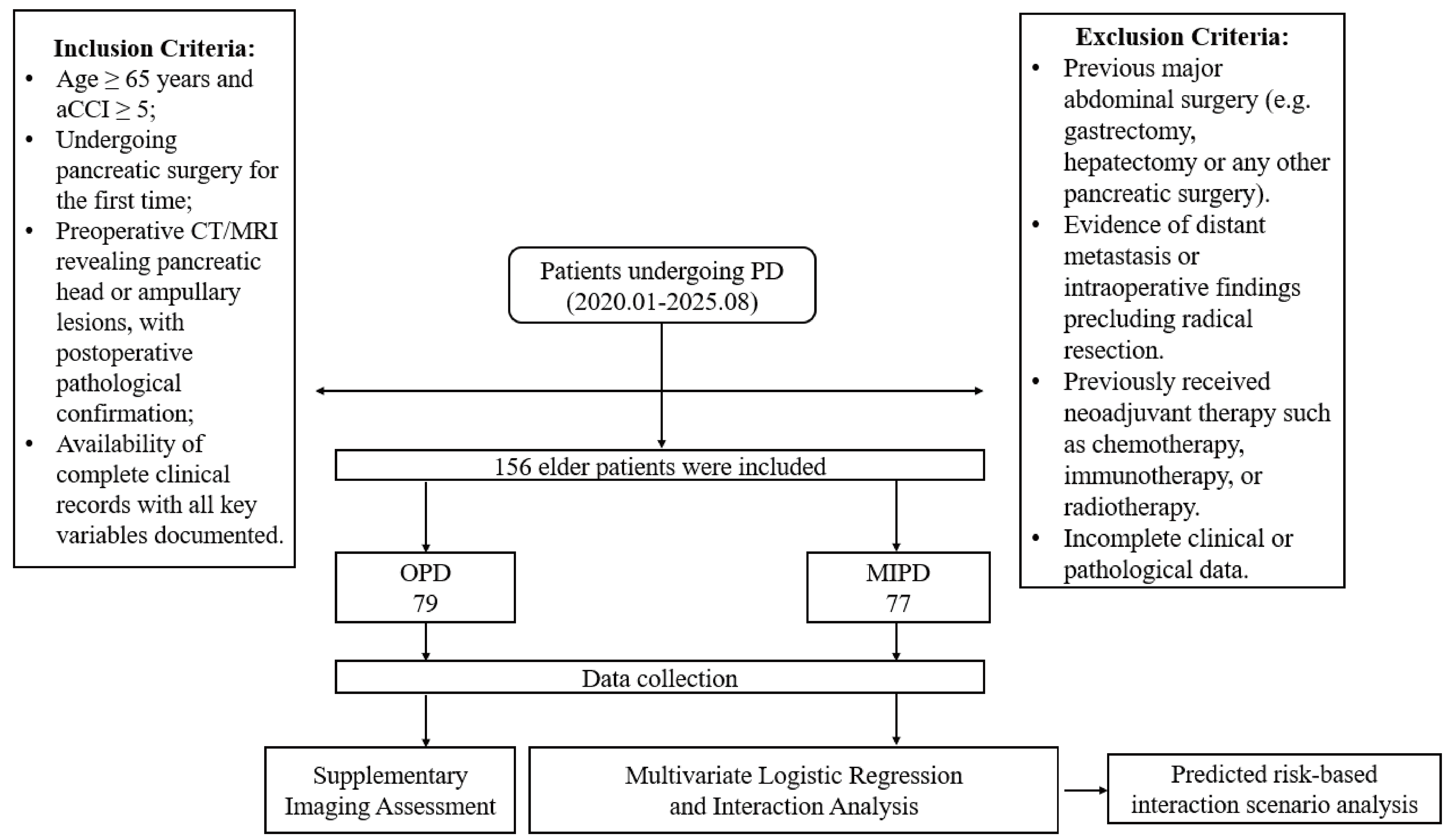
This study was designed as a single-center retrospective cohort study conducted at the Pancreatic Disease Diagnosis and Treatment Center in the First Affiliated Hospital of Harbin Medical University, which is the largest pancreatic surgery unit in Northeast China, performing more than 300 pancreatic surgeries annually including approximately 70 PDs. A total of 156 elderly patients who underwent PD between January 2020 and August 2025 were included. The study was approved by the Ethics Review Committee of the First Affiliated Hospital of Harbin Medical University, and all personal identifiers were removed to ensure patient confidentiality (approval number: 202552).
Patient selection and grouping
(1) Inclusion Criteria
Patients were eligible if they met all the following conditions:
• Age ≥ 65 years and aCCI ≥ 5.
• Undergoing pancreatic surgery for the first time.
• Preoperative computed tomography (CT)/magnetic resonance imaging (MRI) revealing pancreatic head or ampullary lesions, with postoperative pathological confirmation.
• Availability of complete clinical records with all key variables documented.
(2) Exclusion Criteria
Patients were excluded if any of the following conditions were present:
• Previous major abdominal surgery (e.g., gastrectomy, hepatectomy, or any other pancreatic surgery).
• Evidence of distant metastasis or intraoperative findings precluding radical resection.
• Previously received neoadjuvant therapy such as chemotherapy, immunotherapy, or radiotherapy.
• Incomplete clinical or pathological data.
(3) Surgical Technique Grouping
Patients were categorized according to the surgical approach:
• OPD: Conventional OPD and conversion from minimally invasive to open surgery during the operation.
• MIPD: Laparoscopic pancreatoduodenectomy (LPD) or robotic pancreatoduodenectomy (RPD).
RPD was defined as a procedure performed with the da Vinci robotic system (IV), encompassing both the resection and reconstruction phases. All surgeries were conducted by the pancreatic surgery team in our center, which has over two decades of experience in high-volume pancreatic surgery (> 50 PD cases annually), ensuring technical consistency and procedural standardization.
Data collection and variable definition
Data source
All data were obtained from the electronic medical record system of the First Affiliated Hospital of Harbin Medical University, including preoperative assessments, intraoperative variables, pathology results, 30-day postoperative outcomes and 90-day postoperative outcomes. Data entry was performed independently by two investigators, and discrepancies were adjudicated by a third reviewer.
Primary outcome measures
The primary outcome measure was short-term postoperative complications, defined as complications classified as Grade II or higher according to the Clavien-Dindo classification[16] and requiring clinical intervention were used as inclusion criteria, such as prolonged or escalated antibiotic therapy, placement of a chest tube for drainage, insertion of a jejunal feeding tube, establishment of a drainage tube for continuous negative pressure lavage, or secondary surgery. These events included postoperative hemorrhage, delayed gastric emptying, Grade B or C pancreatic fistula [2016 International Study Group for Pancreatic Surgery (ISGPS) criteria][17], pneumonia, and pleural effusion. If any of the above complications occur, the patient is considered to have developed a complication. Complications were analyzed as a binary variable (0 = no complication; 1 = complication).
Covariates and intraoperative variables
Key covariates included:
Patient demographic information: gender, American Society of Anesthesiologists (ASA) physical status classification, BMI, and aCCI.
BMI was calculated as weight divided by height squared (kg/m2); ASA classification followed ASA guidelines[18]; and aCCI was computed according to the aCCI scoring system[12].
Preoperative biochemical variables included hemoglobin (Hb), albumin, total bilirubin (TBIL), gamma-glutamyl transferase (GGT), alkaline phosphatase (AKP), and carbohydrate antigen 19-9 (CA19-9).
Intraoperative variables included operative time (hours), estimated blood loss (mL) and Fistula Risk Score (FRS)[19].
Statistical analysis
Analytical tools
Baseline statistical analyses were performed using GraphPad Prism (version 10.1.2). Multivariable logistic regression was conducted in R (version 4.5.1) using the tidyverse, ggplot2, dplyr, and ggeffects packages. A standardized analytical workflow was used to ensure reproducibility.
Descriptive analysis
Normally distributed continuous variables are reported as mean ± standard deviation. Between-group comparisons were performed using independent-sample t tests. Non-normally distributed continuous variables are expressed as median (interquartile range) and compared using the Mann–Whitney U test. Categorical variables were compared using the chi-square (χ2) test, and Fisher’s exact test was applied when appropriate.
Multivariate logistic regression
Multivariable models were constructed with postoperative complications (presence vs. absence) as the dependent variable.
Interaction and threshold analysis
To assess whether patient characteristics modified the association between surgical approach and complication risk, interaction terms (“surgical approach × covariate”) were sequentially added to the regression models. For significant interactions (P < 0.05), the ggeffects package was used to estimate predicted complication probabilities, generate interaction plots, and calculate crossover points - defined as the threshold at which the risks of open and minimally invasive surgery were equal - using linear interpolation. This analytical strategy enabled identification of thresholds for patient-characteristic–surgical-approach interactions, thereby revealing the risk advantage of specific surgical approaches below these thresholds.
Predicted risk–based interaction scenario analysis
To translate statistically significant interaction effects into clinically interpretable estimates, we performed a predicted probability-based scenario analysis. Using the final multivariable logistic regression model incorporating interaction terms between surgical approach and BMI as well as aCCI, we calculated predicted probabilities of postoperative complications under predefined clinical scenarios.
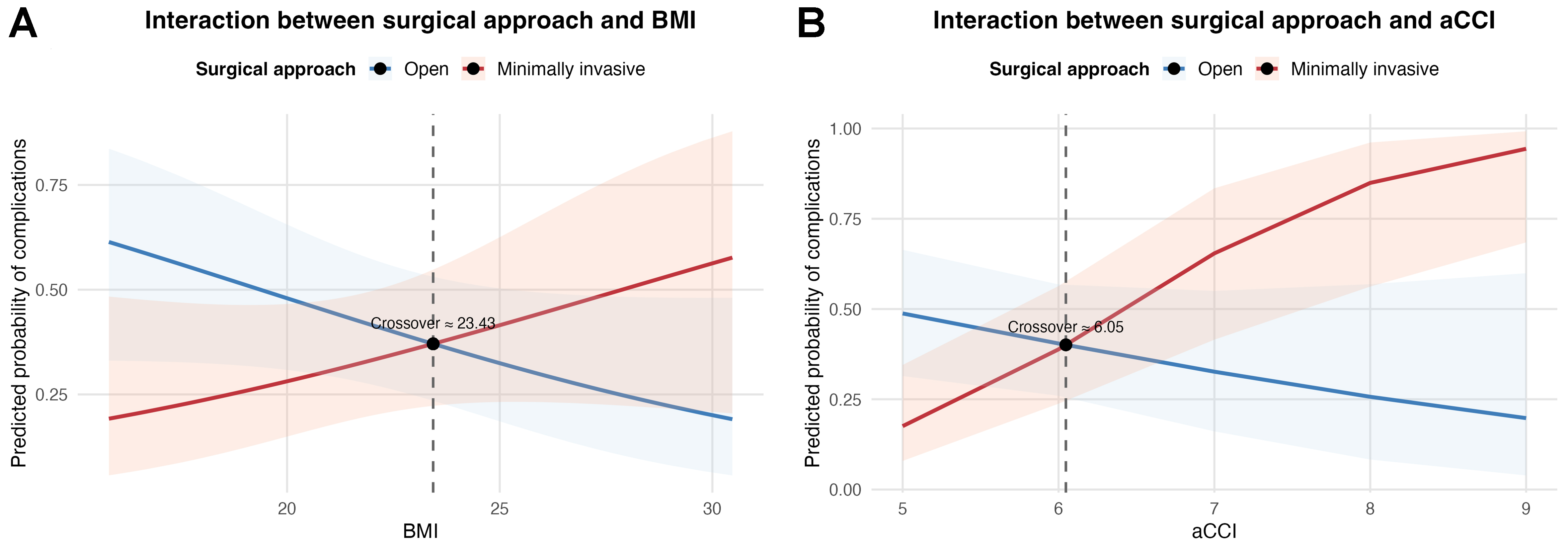
Crossover points for BMI and aCCI were determined from interaction plots as the values at which predicted risks for open and MIPD were equivalent. Based on these thresholds (BMI = 23.43 kg/m2 and aCCI = 6.05), four representative interaction scenarios were constructed by combining low vs. high BMI and low vs. high aCCI values.
For each scenario, predicted complication risks and corresponding 95% confidence intervals (CIs) were estimated for both surgical approaches, with other covariates held constant at their mean or most frequent values. Differences in absolute risk were calculated to facilitate comparison and support clinical interpretation.
Supplementary imaging assessment
To further investigate the potential mechanisms underlying the observed interactions between BMI, surgical approach, and postoperative complications, we conducted supplemental quantitative imaging analyses on a subset of 2022-2025 cohort patients who demonstrated the representative interaction patterns described above (OPD: n = 31; MIPD: n = 49; total n = 80). All included patients had preoperative triphasic contrast-enhanced CT scans (non-contrast, arterial, and portal venous phases) with a slice thickness of
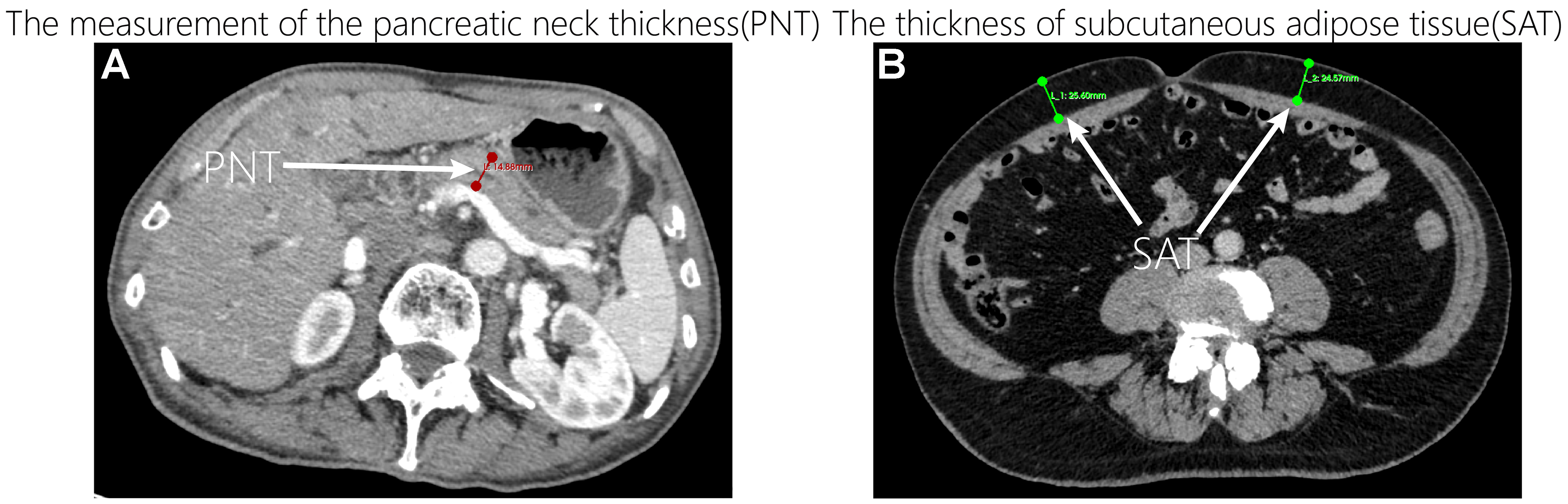
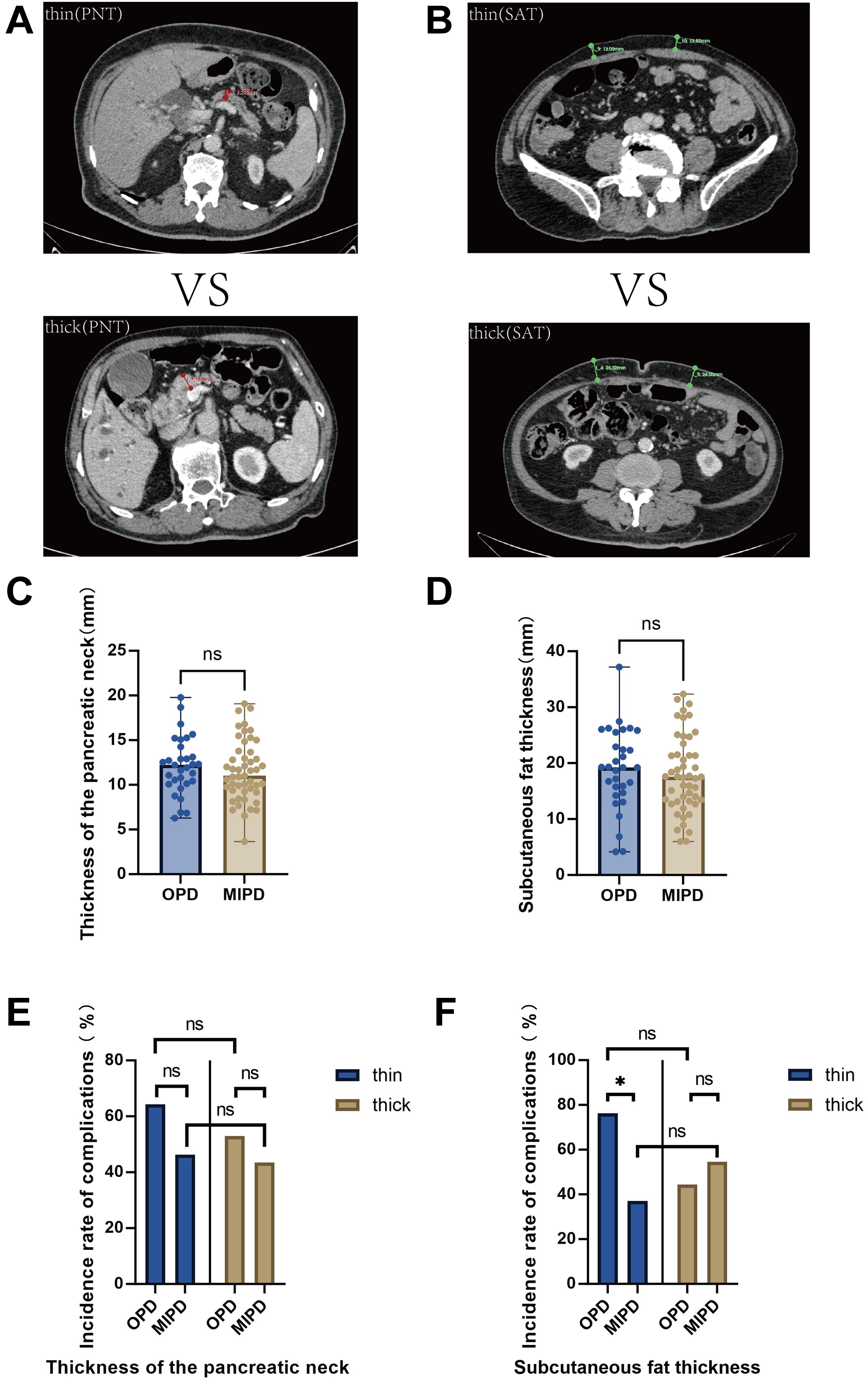
Subcutaneous adipose tissue thickness
At the L3-L4 intervertebral disc level, subcutaneous adipose thickness was measured perpendicular to the skin at the maximal point on both the left and right anterior abdominal walls along the midclavicular line. The mean of the two measurements (in millimeters) was recorded as the patient’s subcutaneous adipose tissue thickness (SAT). The L3-L4 level has been widely validated as a reliable anatomical surrogate for overall subcutaneous fat volume[20].
Pancreatic neck thickness
We selected three consecutive axial slices of the pancreatic neck for analysis. The anterior-posterior thickness of the pancreatic neck should be measured on each slice using a linear tool perpendicular to the gland’s longitudinal axis. It is imperative that measurements are taken above the confluence of the portal vein and superior mesenteric vein during the portal venous phase in order to ensure anatomical landmark consistency. The mean of three measurements is calculated to obtain the patient-specific pancreatic neck thickness (PNT). Greater PNT values are positively correlated with the occurrence of clinically relevant postoperative pancreatic fistula[21].
Subsequently, median values for each parameter were determined based on the overall subcutaneous fat thickness and PNT of 80 patients. Patients were then classified into eight subgroups according to surgical approach (OPD vs. MIPD) and dichotomized thickness (thin vs. thick) for each imaging parameter, namely PNT and subcutaneous fat thickness. Finally, the chi-square test was employed to compare the incidence of short-term postoperative complications among patients within these eight subgroups, thereby identifying key factors potentially influencing short-term postoperative complications in conjunction with surgical approach.
Significance and reproducibility
All analyses were two-tailed, and statistical significance was set at P < 0.05. To promote transparency and reproducibility, statistical scripts and modeling code are available upon reasonable request [Figures 1 and 2].
Figure 1. Research flowchart. aCCI: Age-adjusted Charlson Comorbidity Index; CT: computed tomography; MRI: magnetic resonance imaging; PD: pancreatoduodenectomy; OPD: open pancreatoduodenectomy; MIPD: minimally invasive pancreatoduodenectomy.
RESULTS
Patient demographic information
A total of 156 elderly patients undergoing PD were included in the analysis, comprising 79 (49.3%) in the OPD group and 77 (50.7%) in the MIPD group. Within the MIPD group, there were no significant differences between LPD and RPD patients in demographic information or surgical indicators [Supplementary Tables 1 and 2]. Four patients initially undergoing MIPD required conversion to open surgery and were ultimately analyzed in the OPD group, resulting in a conversion rate of 4.9%. Among these cases, two conversions were necessitated by close tumor–vascular involvement, and two were due to severe intra-abdominal adhesions. There were no significant differences between the OPD and MIPD groups with respect to age, sex distribution, ASA classification, or preoperative laboratory parameters (all P > 0.05). Compared with the OPD group, the MIPD group had a significantly longer operative time (P < 0.05), whereas intraoperative blood loss, pancreatic texture, main pancreatic duct diameter, and the FRS score did not differ significantly between groups (all P > 0.05). The 30-day postoperative complication rates were 44.3% in the OPD group and 39% in the MIPD group, with no statistically significant difference (P = 0.499). Likewise, 90-day mortality, readmission rates, and the incidence of postoperative pancreatic insufficiency showed no significant differences between the two groups (all P > 0.05) [Tables 1 and 2].
Comparison of demographic information between two groups
| Variables | OPD | MIPD | Statistical value | P value |
| Sex | χ 2 = 0.711 | 0.40 | ||
| Male | 44 | 48 | ||
| Female | 35 | 29 | ||
| Age (year) | 70 (67, 72) | 70 (67, 73) | U = 2,739 | 0.28 |
| Median (IQR) | ||||
| BMI | 21.97 (19.98, 25.23) | 21.02 (19.89, 22.64) | U = 2,532 | 0.07 |
| Median (IQR) | ||||
| ASA | χ 2 = 1.989 | 0.16 | ||
| II | 5 | 10 | ||
| III | 74 | 67 | ||
| Hb | 125.4 ± 17.09 | 124.4 ± 14.73 | t = 0.3940 | 0.69 |
| Mean (SD) | ||||
| Albumin | 36.20 (33.40, 39.60) | 37.40 (34.25, 39.85) | U = 2,668 | 0.19 |
| Median (IQR) | ||||
| TBIL | 50.30 (15.40, 139.60) | 71.90 (17.05, 157.80) | U = 2,790 | 0.37 |
| Median (IQR) | ||||
| GGT | 359.5 (58.50, 932.6) | 364.2 (87.08, 771.8) | U = 2,986 | 0.85 |
| Median (IQR) | ||||
| AKP | 302.1 (100.6, 527.5) | 305.8 (114.8, 504.7) | U = 3,021 | 0.94 |
| Median (IQR) | ||||
| CA19-9 | 136.7 (35.04, 425.6) | 99.10 (18.03, 222.8) | U = 2,642 | 0.16 |
| Median (IQR) | ||||
| aCCI Score | χ 2 = 0.008 | 0.927 | ||
| 5-6 | 60 | 58 | ||
| ≥ 7 | 19 | 19 |
Comparison of surgical indicators between two groups
| Variables | OPD | MIPD | Statistical value | P value |
| Operative time (hour) | 4.62 (4.0, 5.0) | 6.5 (5.4, 8.0) | U = 800.5 | < 0.0001 |
| Median (IQR) | ||||
| Intraoperative blood loss (mL) | 100 (100, 200) | 100 (50, 200) | U = 2,718 | 0.237 |
| Median (IQR) | ||||
| Wirsung diameter | 3 (2, 4) | 3 (2, 4) | U = 2,919 | 0.654 |
| Median (IQR) | ||||
| FRS score | 3.09 ± 1.98 | 3.20 ± 1.54 | t = 0.374 | 0.709 |
| Mean (SD) | ||||
| Pancreatic texture | χ2 = 0.020 | 0.964 | ||
| Soft | 29 | 28 | ||
| Hard | 50 | 49 | ||
| 30-day Complications | 44% (35/79) | 39% (30/77) | χ2 = 0.458 | 0.499 |
| Postoperative pancreatic fistula | 0.118 | |||
| B | 12 | 12 | ||
| C | 1 | 6 | ||
| Postoperative hemorrhage | 5% (4/79) | 8% (6/77) | χ2 = 0.484 | 0.487 |
| Postoperative gastroparesis | 15% (12/79) | 6% (5/77) | χ2 = 3.037 | 0.081 |
| Postoperative pneumonia | 24% (19/79) | 18% (14/77) | χ2 = 0.805 | 0.369 |
| Postoperative pleural effusion | 14% (11/79) | 18% (14/77) | χ2 = 0.525 | 0.469 |
| 90-day readmission rate after surgery | 4% (3/79) | 10.4% (8/77) | χ2 = 2.59 | 0.108 |
| 90-day postoperative mortality rate | 4% (3/79) | 5.2% (4/77) | χ2 = 0.178 | 0.673 |
| Incidence of 90-day postoperative pancreatic endocrine dysfunction | 8.9% (7/79) | 6.5% (5/77) | χ2 = 0.308 | 0.579 |
| Incidence of 90-day postoperative pancreatic exocrine dysfunction | 17.7% (14/79) | 10.4% (8/77) | χ2 = 2.71 | 0.099 |
Multivariate and interaction analysis of surgical approach and complication risk
A multivariate logistic regression model was developed using postoperative complications (presence vs. absence) as the dependent variable. Independent variables included surgical approach, BMI, sex, ASA classification, Hb, albumin, TBIL, GGT, AKP, and CA19-9. Surgical approach (open vs. minimally invasive) was not identified as an independent predictor of 30-day postoperative complications [odds ratio (OR) = 0.692, 95%CI: 0.349-1.36; P = 0.286]. To determine whether specific patient characteristics modified the association between surgical approach and postoperative complications, an expanded model including interaction terms (“surgical approach × covariate”) was applied. Significant interactions were identified for BMI × surgical approach (OR = 1.28, 95%CI: 1.02-1.61, P = 0.032) and aCCI × surgical approach (OR = 4.18, 95%CI: 1.92-9.09, P < 0.001). No significant interactions were detected for other covariates, including gender, ASA classification, Hb, albumin, TBIL, GGT, AKP, FRS score and CA19-9 (all P > 0.05) [Table 3].
Interaction effects between surgical approach and key covariates
| Variable | OR | CI_lower | CI_upper | P value |
| aCCI | 4.18 | 1.92 | 9.09 | < 0.001 |
| BMI | 1.28 | 1.02 | 1.61 | 0.032 |
| TBIL | 1.00 | 0.998 | 1.01 | 0.179 |
| Hb | 0.991 | 0.949 | 1.04 | 0.692 |
| Albumin | 1.05 | 0.909 | 1.22 | 0.484 |
| Gender | 0.964 | 0.249 | 3.74 | 0.958 |
| GGT | 1.00 | 0.999 | 1.00 | 0.610 |
| CA19-9 | 1.00 | 0.999 | 1.00 | 0.408 |
| AKP | 1.00 | 0.999 | 1.00 | 0.313 |
| FRS | 1.17 | 0.789 | 1.73 | 0.436 |
| ASA | 0.00 | 0.00 | ∞ | 0.988 |
At the same time, we also used the same method to analyze the relationship between the above covariates and 90-day complications and found that the interaction between all covariates and the surgical method was not statistically significant (P > 0.05) [Supplementary Table 3].
Interaction analysis
For covariates that showed significant interaction with surgical approach (BMI and aCCI), we calculated the predicted probability of 30-day postoperative complications across different covariate levels to clarify how these variables modify the effect of surgical approach [Figure 3].
Figure 3. Interaction analysis results. (A) Effect of the interaction between BMI and surgical approach on short-term postoperative complications; (B) Effect of the interaction between aCCI and surgical approach on short-term postoperative complications. BMI: Body mass index; aCCI: age-adjusted Charlson Comorbidity Index.
Interaction between BMI and surgical approach
• When BMI was below 23.43 kg/m2, MIPD patients exhibited a low incidence of complications requiring clinical intervention. Consequently, they required a shorter course of postoperative antibiotics, less frequent jejunal feeding tube retention, and fewer additional procedures, including continuous negative-pressure drainage irrigation, local puncture interventions, and secondary surgeries.
• When BMI exceeded 23.43 kg/m2, patients undergoing OPD showed the opposite pattern, with a lower likelihood of complications and reduced need for postoperative therapeutic interventions.
• The predicted complication risk curves for the two surgical approaches intersected at a BMI of approximately 23.43 kg/m2.
Interaction between aCCI and surgical approach
• When aCCI exceeded 6, patients undergoing MIPD had a higher likelihood of developing short-term postoperative complications that required clinical intervention.
• When aCCI is below 7, MIPD patients have a lower probability of experiencing short-term postoperative complications requiring clinical intervention compared to OPD patients.
Risk stratification based on combined BMI and aCCI scenarios
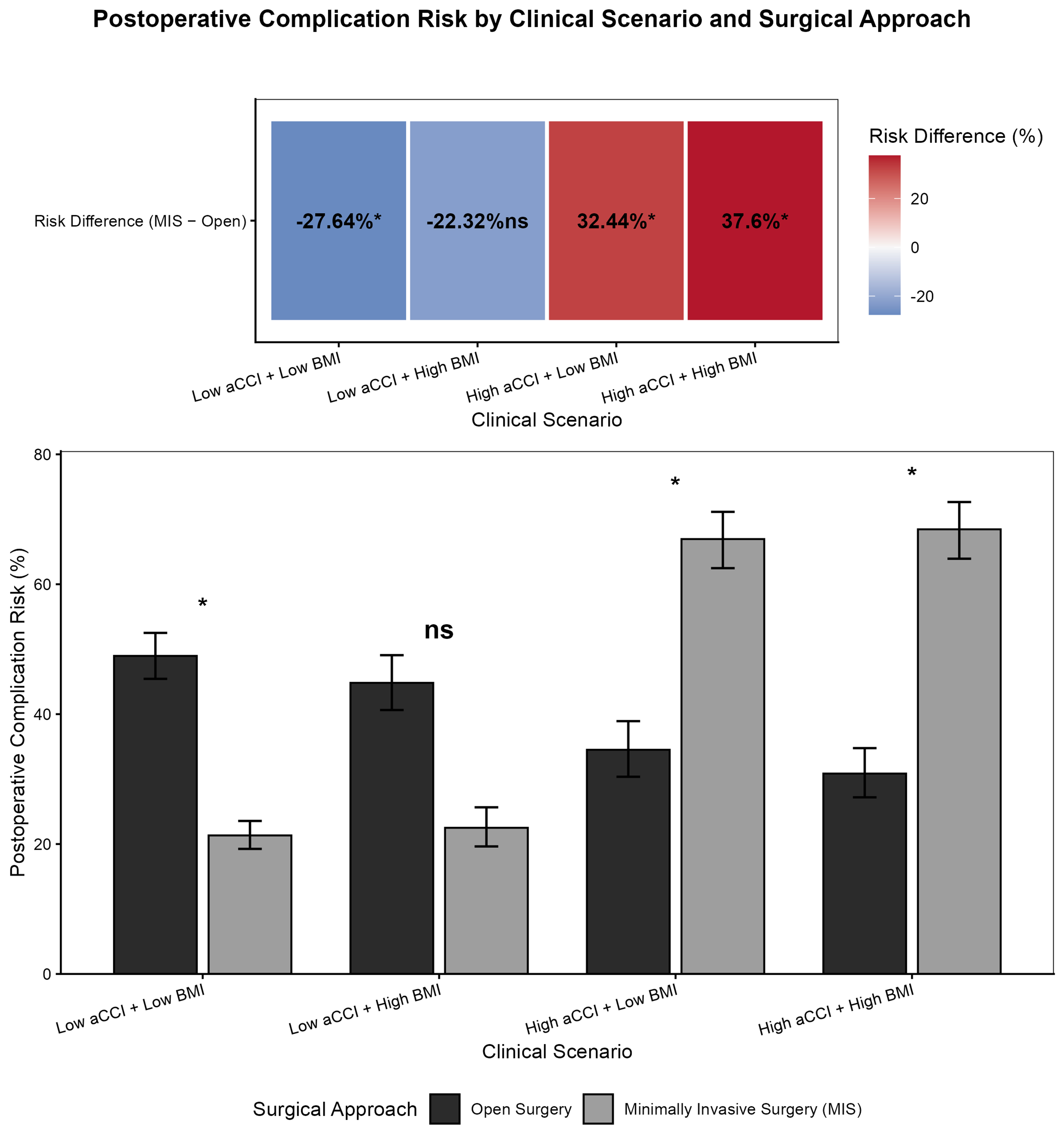
To further elucidate the clinical implications of the observed interaction effects, we estimated model-based predicted probabilities of postoperative complications across four representative clinical scenarios defined by BMI and aCCI thresholds.
In patients with low aCCI (≤ 6) and low BMI (< 23.43 kg/m2), MIPD was associated with a substantially lower predicted complication risk compared with open surgery (21.3% vs. 49.0%; absolute risk difference -27.7%). This difference was statistically significant based on Wald tests performed on the model-derived logit scale (P = 0.010).
A similar risk reduction favoring minimally invasive surgery was observed among patients with low aCCI and high BMI (22.5% vs. 44.8%; absolute risk difference -22.3%). However, this difference did not reach statistical significance (P = 0.095).
In contrast, among patients with high aCCI (> 6), the direction of risk was reversed. In both low- and high-BMI subgroups, OPD was associated with markedly lower predicted complication risks than MIPD (34.5% vs. 67.0% and 30.9% vs. 68.5%, respectively), corresponding to absolute risk differences exceeding 30%. These differences were statistically significant (P = 0.031 and P = 0.014, respectively).
These findings indicate that the effect of surgical approach is strongly modified by comorbidity burden, with minimally invasive surgery conferring benefit primarily in patients with lower aCCI scores [Figure 4].
Figure 4. Postoperative complication risk in PD by clinical scenario. “ns” indicates P > 0.05, with no statistically significant difference; “*” indicates P < 0.05, with statistically significant difference. PD: Pancreatoduodenectomy; aCCI: age-adjusted Charlson Comorbidity Index; BMI: body mass index.
Supplementary imaging assessment
Based on imaging data obtained from 80 patients, we measured a median PNT of 11.64 mm and a median subcutaneous fat thickness of 17.65 mm. Following the earlier described grouping, patients were categorized into eight groups according to surgical approach, PNT and subcutaneous fat thickness. We then applied chi-square tests sequentially to compare differences between groups. The results revealed that: Patients in the low-SAT group demonstrated a lower incidence of complications in MIPD procedures (χ2 = 5.584, P = 0.018) [Figure 5].
Figure 5. Results of the supplementary imaging analysis. (A) Comparison of CT images of a narrow pancreatic neck and a thick pancreatic neck; (B) Comparison of CT images between thin subcutaneous fat and thick subcutaneous fat; (C) Comparison of differences in PNT between OPD and MIPD; (D) Comparison of differences in subcutaneous fat thickness between OPD and MIPD; (E) Comparison of incidence rate of complications between subgroups based on PNT; (F) Comparison of incidence rate of complications between subgroups based on subcutaneous fat thickness. Chi-square tests were applied; “ns” indicates P > 0.05, with no statistically significant difference; “*” indicates P < 0.05, with statistically significant difference. CT: Computed tomography; PNT: pancreatic neck thickness; OPD: open pancreatoduodenectomy; MIPD: minimally invasive pancreatoduodenectomy; SAT: subcutaneous adipose tissue thickness.
DISCUSSION
This retrospective study included elderly patients (≥ 65 years) undergoing PD between January 2020 and August 2025, examining the interaction between surgical approach, BMI, and aCCI on short-term complications. A total of 156 patients were analyzed (79 OPD; 77 MIPD). Operative time was longer in the MIPD group, whereas baseline characteristics were comparable. The 30-day complication rates were comparable (44% vs. 39%, P = 0.499), and surgical approach was not independently associated with 30-day complications. Multivariable analysis with interaction terms showed that BMI and aCCI significantly modified the association between surgical approach and 30-day complication risk. Significant interactions were observed for BMI (OR = 1.28, 95%CI: 1.02-1.61) and aCCI (OR = 4.18, 95%CI: 1.92-9.09; both P < 0.05), indicating effect modification by physiological reserve and comorbidity burden. A predicted probability analysis using combined BMI and aCCI thresholds was conducted. In patients with low comorbidity burden (aCCI ≤ 6), MIPD showed lower predicted risk, particularly at lower BMI. In contrast, in patients with high comorbidity burden (aCCI > 6), OPD showed lower predicted risk across BMI strata. These findings indicate that perioperative safety is context-dependent. A supplementary CT-based analysis in a subset (n = 80) explored anatomical factors. In the MIPD group, lower SAT was associated with fewer complications (P < 0.05). PNT did not differ significantly between subgroups. Overall, surgical strategy should be individualized based on combined assessment of BMI and aCCI. This BMI–aCCI interaction framework offers a clinically actionable risk stratification tool and challenges the assumption of uniform benefit from MIPD.
With advances in surgical techniques, anesthesia, and perioperative management, the safety of PD in elderly patients has improved substantially. Large multicenter studies, including national registry data from Japan, have demonstrated acceptable perioperative mortality and complication rates even among patients aged over 80 years undergoing PD in high-volume centers[22]. Similarly, previous reports indicate that carefully selected elderly patients can achieve short-term outcomes comparable to those of younger patients[23]. These data support the feasibility of PD in elderly populations when appropriate patient selection and surgical strategies are applied. However, they also highlight that chronological age alone is insufficient to guide surgical decision-making, emphasizing the need to identify patient-specific risk modifiers.
Our interaction and scenario-based risk stratification analyses revealed that the relative safety of MIPD vs. OPD is strongly influenced by BMI and comorbidity burden. In patients with lower BMI and preserved physiological reserve (aCCI ≤ 6), MIPD was associated with lower predicted complication risks. In contrast, a clear risk inversion was observed in patients with higher comorbidity burden (aCCI > 6), in whom OPD consistently demonstrated lower predicted complication risks across BMI strata. These findings indicate that the advantage of surgical approach is not static but depends on the combined physiological and metabolic profile of the patient, rather than on BMI or aCCI in isolation.
The diminishing benefit of MIPD in elderly patients with higher BMI can be partially explained by technical and anatomical constraints. Excessive subcutaneous and visceral adiposity restricts the already limited operative workspace created by pneumoperitoneum and fixed trocar placement. In our CT-based subgroup analysis, patients with lower SAT undergoing MIPD experienced significantly fewer postoperative complications, supporting the hypothesis that reduced anterior abdominal fat improves operative exposure and visualization. Moreover, abundant mesenteric and retroperitoneal fat obscures tissue planes, increases glare under laparoscopic illumination, and prolongs dissection of critical vascular structures, including the superior mesenteric vein, pancreatic uncinate process, and portal–mesenteric axis. These challenges are further amplified by longer operative times and sustained pneumoperitoneum, which may exacerbate cardiopulmonary stress in frail elderly patients with limited physiological reserve[24]. Unlike colorectal or bariatric surgery - where minimally invasive approaches often benefit obese patients - PD involves deeply fixed retroperitoneal anatomy and technically demanding anastomoses, leaving minimal margin for error[25]. Within this context, our findings suggest that, particularly in Asian populations with lower BMI thresholds, open surgery may provide superior safety when BMI exceeds approximately 23.43 kg/m2.
Comorbidity burden, as quantified by aCCI, emerged as an even more influential modifier of surgical risk. The aCCI is a well-established predictor of postoperative morbidity and mortality in elderly surgical patients[11]. Prior studies have shown that patients with aCCI > 6 are particularly vulnerable to perioperative stress. Prolonged pneumoperitoneum and operative duration during minimally invasive procedures may further compromise cardiopulmonary function in this high-risk population[26]. Consistent with these observations, our scenario-based analyses demonstrated that OPD conferred a lower predicted complication risk than MIPD in patients with aCCI > 6, regardless of BMI. These results reinforce the notion that physiological reserve and chronic disease burden may outweigh the theoretical benefits of minimally invasive approaches in frail elderly patients. For instance, in diabetic pancreatic ductal adenocarcinoma (PDAC) patients, diabetes-driven cholesterol synthesis and neutrophil extracellular trap (NET) formation in the tumor microenvironment may further amplify perioperative immune dysregulation and complication risks[27], underscoring the need for preoperative metabolic assessments alongside BMI and aCCI.
Nutritional status represents another important dimension of surgical risk in elderly patients. Previous studies have shown that the Geriatric Nutritional Risk Index (GNRI) provides additional prognostic value for postoperative outcomes following PD, with lower GNRI scores associated with higher rates of major complications and pulmonary infections[28]. Given the close relationship between GNRI, BMI, and serum albumin, incorporating nutritional indices alongside aCCI may further refine perioperative risk stratification. Although GNRI was not directly analyzed in our primary models, it may serve as a complementary tool to enhance individualized surgical planning in future studies.
Taken together, our findings support a structured, clinically actionable risk-stratification framework integrating BMI and aCCI to guide surgical approach selection in elderly patients undergoing PD. Minimally invasive approaches appear most suitable for physiologically robust elderly patients with lower BMI and comorbidity burden, whereas open surgery offers greater perioperative safety in patients with higher BMI or substantial comorbidities. Incorporating preoperative CT-based body composition assessment may further enhance risk evaluation by providing mechanistic insight into anatomical and technical constraints. Beyond PD, this combined risk-stratification paradigm may inform decision-making for other high-risk surgical procedures in elderly populations, advancing a shift from standardized treatment strategies toward precision, patient-centered surgical care.
This study has several limitations that should be acknowledged. First, its retrospective single-center design introduces risks of selection and information bias. Surgical approach selection was influenced by patient condition, anatomical complexity, and surgeon discretion, which could not be fully controlled despite multivariable adjustment. Variability in surgeon experience with minimally invasive techniques represents an unmeasured confounder that may have influenced operative time and outcomes. Second, the relatively small sample size (n = 156) limited statistical power, particularly for interaction analyses, and may have affected the stability of BMI and aCCI threshold estimates. This was compounded by incomplete imaging data, as CT-based body composition measurements were available only in a subset, reducing the precision of subgroup analyses. Third, generalizability may be limited. The study population was derived from a single high-volume center in Northeast China, and BMI thresholds may not be directly applicable to Western populations, where body composition and perioperative management differ[29]. Finally, the study focused primarily on short-term (30-day) complications. Although selected 90-day outcomes were examined, long-term complications and overall prognosis were not systematically evaluated. Key endpoints, including long-term survival, functional recovery, and quality of life, were not assessed. Accordingly, the predictive value of the BMI-aCCI interaction framework for long-term outcomes is limited. Its applicability to long-term trajectories requires validation in studies with comprehensive longitudinal follow-up.
Future studies should prioritize prospective, multicenter validation of the combined BMI–aCCI risk stratification framework to enhance statistical power and external validity. Incorporating long-term oncologic and functional outcomes, as well as standardized geriatric assessment tools, will be essential to comprehensively evaluate surgical benefit in elderly patients. Advanced imaging, artificial intelligence–based predictive models, and emerging minimally invasive technologies such as RPD may further refine personalized surgical decision-making[30]. Together, these efforts may facilitate a shift from standardized surgical approaches toward precision, patient-centered care in geriatric pancreatic surgery.
DECLARATIONS
Authors’ contributions
Made substantial contributions to the conception and design of the study and performed data analysis and interpretation: Miao Z, Li G, Chen H, Sun B
Data analysis and interpretation: Miao Z, Zhang Z, Zhong Z, Wang Z, Zhu M, Bai K, Wu J, Bai R, Song Y, Zhang D
Conceptual design of study and review of data and manuscript: Li G
Data acquisition, analysis, and interpretation: Miao Z
Availability of data and materials
All the data and materials used in our study are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This study was supported by the National Natural Science Foundation of China [Nos. 82400751 (Li G); 82270665, 82470672 (Sun B)]; the Science Fund for Excellent Young Scholars of the First Affiliated Hospital of Harbin Medical University [No. HYD2024YQ01 (Li G)]; the Natural Science Foundation of Heilongjiang Province [Nos. PL2024H037 (Li G); JD2023SJ42 (Sun B)]; and the Noncommunicable Chronic Diseases–National Science and Technology Major Project (Nos. 2024ZD0525500/2024ZD0525503).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of the First Affiliated Hospital of Harbin Medical University (approval number: 202552). This was a retrospective observational study focusing on postoperative complications. Written informed consent from individual participants was formally waived by the above Ethics Committee, as the study utilized de-identified existing clinical data from the hospital’s medical records, with no identifiable information linking to specific subjects.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Li GQ, Zhang T, Yang WG, et al. Gut microbiota patterns associated with somatostatin in patients undergoing pancreaticoduodenectomy: a prospective study. Cell Death Discov 2020;6:94.
2. Cao Y, Gu HY, Huang ZD, et al. Impact of enhanced recovery after surgery on postoperative recovery for pancreaticoduodenectomy: pooled analysis of observational study. Front Oncol 2019;9:687.
3. Li X, Lu Y, Chen C, et al. Development and validation of a patient-specific model to predict postoperative SIRS in older patients: a two-center study. Front Public Health 2023;11:1145013.
4. Caban M, Małecka-Wojciesko E. Gaps and opportunities in the diagnosis and treatment of pancreatic cancer. Cancers 2023;15:5577.
5. Cutolo C, Izzo F, Valeriani M, Belli A. Is robotic pancreaticoduodenectomy (PD) a valid alternative to open pancreaticoduodenectomy? Mini-invasive Surg 2025;9:40.
6. Shannon A, Bath NM, Ejaz A. A narrative review of the history and recent advances in minimally invasive pancreatic resection. Mini-invasive Surg 2023;7:32.
7. Liu Y, Yun H, Zhang W, et al. Robotic versus laparoscopic total gastrectomy for gastric cancer: a systematic review and meta-analysis of perioperative and oncologic outcomes. Int J Surg 2025;111:6397-411.
8. Zhang L, Shi F, Hu C, et al. Comparison of robotic versus laparoscopic lateral lymph node dissection for advanced lower rectal cancer: a retrospective study at two institutions. Surg Endosc 2023;37:4403-13.
9. Abdelmaseeh M, Mhmndar A, Siddiqa A, et al. Minimally invasive approaches for high-risk and elderly patients with acute cholecystitis: a systematic review of techniques and outcomes. Cureus 2025;17:e78271.
10. Ballarin R, Esposito G, Guerrini GP, et al. Minimally invasive pancreaticoduodenectomy in elderly versus younger patients: a meta-analysis. Cancers 2024;16:323.
11. Wang J, Xu L, Huang S, Hui Q, Shi X, Zhang Q. Low muscle mass and Charlson comorbidity index are risk factors for short-term postoperative prognosis of elderly patients with gastrointestinal tumor: a cross-sectional study. BMC Geriatr 2021;21:730.
12. Bhattacharjee HK, Kaviyarasan MP, Singh KJ, et al. Age adjusted Charlson comorbidity index (a-CCI) AS a tool to predict 30-day post-operative outcome in general surgery patients. ANZ J Surg 2023;93:132-8.
13. Yamashita Y, Baudo M, Sicouri S, et al. Effect of elevated body mass index on outcomes of transcatheter aortic valve replacement for severe aortic stenosis. Anatol J Cardiol 2024;28:565-74.
14. Sun Y, Xu Z, Tang Y, et al. Unveiling the impact of body mass index on surgical difficulty and oncological prognosis in low rectal cancer: post-hoc analysis of the LASRE trial. Int J Colorectal Dis 2025;40:185.
15. Kowshik V, Velkumary S, Sethi P, Feula JM, Subhashri S, Abirami M. Association of handgrip strength and endurance with body composition in head and neck cancer patients. J Family Med Prim Care 2021;10:910-6.
16. Wang HB, Xiong GB, Zhu F, et al. [Clavien-Dindo classification and influencing factors analysis of complications after laparoscopic pancreaticoduodenectomy]. Zhonghua Wai Ke Za Zhi 2018;56:828-32.
17. Pulvirenti A, Ramera M, Bassi C. Modifications in the International Study Group for Pancreatic Surgery (ISGPS) definition of postoperative pancreatic fistula. Transl Gastroenterol Hepatol 2017;2:107.
18. Hendrix JM, Garmon EH. American Society of Anesthesiologists Physical Status Classification System. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025.
19. Lee B, Yoon YS, Kang CM, et al. Fistula risk score-adjusted comparison of postoperative pancreatic fistula following laparoscopic vs open pancreatoduodenectomy. J Hepatobiliary Pancreat Sci 2021;28:1089-97.
20. Salam B, Nowak S, Theis M, et al. Associations between cardiac adipose tissue and abdominal visceral fat and muscle based on computed tomography area and density. Sci Rep 2025;15:20192.
21. Sok C, Sandhu S, Shah H, et al. Simple preoperative imaging measurements predict postoperative pancreatic fistula after pancreatoduodenectomy. Ann Surg Oncol 2024;31:1898-905.
22. Omichi K, Hasegawa K, Kumamaru H, et al. Association between age and short-term outcomes of gastroenterological surgeries in older patients: an analysis using the National Clinical Database in Japan. Langenbecks Arch Surg 2021;406:2827-36.
23. Ikenaga N, Nakata K, Abe T, et al. Risks and benefits of pancreaticoduodenectomy in patients aged 80 years and over. Langenbecks Arch Surg 2023;408:108.
24. Cheng H, Clymer JW, Chen BPH, et al. Prolonged operative duration is associated with complications: a systematic review and meta-analysis. J Surg Res 2018;229:134-44.
25. Cullinane C, Fullard A, Croghan SM, Elliott JA, Fleming CA. Effect of obesity on perioperative outcomes following gastrointestinal surgery: meta-analysis. BJS Open 2023;7:zrad026.
26. Qi F, Wang B, Fei F, et al. Impact of pneumoperitoneum pressure on cardiac output in laparoscopic surgery. BMC Anesthesiol 2025;25:317.
27. Li G, Zhang C, Lu T, et al. Spatial multi-omics analyses reveal diabetes promotes pancreatic cancer progression by stimulating cholesterol-induced neutrophil extracellular trap formation. Cancer Res 2026.
28. Liu X, Xue K, Zhang Y, Tian B. Geriatric nutritional risk index predicts postoperative outcomes in elderly patients with pancreatoduodenectomy: a propensity score-matched analysis. Gland Surg 2025;14:807-17.
29. Caleyachetty R, Barber TM, Mohammed NI, et al. Ethnicity-specific BMI cutoffs for obesity based on type 2 diabetes risk in England: a population-based cohort study. Lancet Diabetes Endocrinol 2021;9:419-26.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].