Combined ablation and resection for multiple colorectal liver metastases: the laparoscopic approach versus the open approach
Abstract
Aim: Combined ablation and resection (CARe) is a recognized approach for managing multiple colorectal liver metastases (CRLM). Perioperative and long-term results of laparoscopic versus open CARe were evaluated in this study.
Methods: This retrospective multicenter cohort study included CRLM patients who underwent CARe at three hospitals between January 2018 and December 2023. Laparoscopic and open approaches were compared for perioperative and oncological results, with propensity score matching (PSM) applied to reduce baseline imbalance-related bias.
Results: Of the 107 CARe procedures, 65 (60.7%) were performed laparoscopically and 42 (39.3%) via open approach. After PSM, patients who underwent laparoscopic CARe had significantly lower intraoperative blood loss (55 vs. 100 mL, P = 0.028) and shorter postoperative hospital stays (6 vs. 8 days, P = 0.005) compared to those who underwent open surgery. However, the complication rates were comparable (P = 0.415). No differences in median recurrence-free survival (14.9 vs. 13.5 months, P = 0.781) or overall survival (70.9 vs. 48.7 months, P = 0.183) were observed. Notably, laparoscopic CARe group had a higher repeat resection rate following intrahepatic recurrence (52.94% vs. 20.00%, P = 0.036).
Conclusion: Laparoscopic CARe is a safe and effective approach for multiple CRLM, offering reduced perioperative morbidity and enhanced feasibility of repeat hepatic resection compared with the open approach.
Keywords
INTRODUCTION
Colorectal cancer (CRC) is the third most common cancer and the second leading cause of cancer deaths worldwide[1]. About half of CRC patients develop liver metastases, either at the time of initial diagnosis or subsequently[2,3]. Colorectal liver metastases (CRLM) represent the primary cause of death in CRC patients, underscoring the critical importance of effective hepatic disease control. Currently, liver resection remains the primary curative-intent treatment for CRLM, offering the best opportunity for long-term survival[4]. Following radical resection, the 5-year overall survival (OS) rate for patients with CRLM can reach up to 50%, a figure comparable to that of patients with stage III primary CRC and significantly higher than the survival outcomes observed in non-surgical management[5,6]. Despite this survival benefit, more than 80% of patients with CRLM are deemed ineligible for upfront surgical resection at diagnosis due to factors such as excessive tumor burden, bilobar distribution, or proximity to major vascular structures[7].
Owing to advances in surgical techniques and the development of effective systemic antitumor therapies, the traditional constraints of tumor burden, such as the number, size, and bilobar distribution of liver metastases, are no longer absolute contraindications to curative-intent surgery for CRLM[8]. However, patients with borderline resectable CRLM, particularly those with bilobar disease or multiple small deep-seated lesions, still pose significant technical challenges. In such cases, achieving complete tumor clearance through resection alone may require extensive hepatectomy, risking an insufficient future liver remnant (FLR) and increasing the likelihood of postoperative liver failure. As a parenchymal-sparing strategy, combined ablation and resection (CARe) offers a more tailored, tissue-preserving approach: it resects accessible lesions while ablating deeper or strategically difficult nodules ≤ 20 mm in diameter - a size range that has achieved excellent local control with ablation[9,10]. By avoiding major or extended hepatectomies, CARe preserves functional hepatic reserve. Growing evidence from recent studies indicates that CARe achieves oncologic outcomes comparable to those of resection alone, while demonstrating superior short-term postoperative results[11-19]. Given its safety, feasibility, and favorable outcomes, CARe has been increasingly adopted as a dependable substitute for resection alone in the treatment of multiple CRLM.
Laparoscopic resection has become the standard of care for CRLM in recent years[20]. As a minimally invasive approach, laparoscopic resection offers several advantages over open surgery. Evidence indicates that laparoscopic liver resection is associated with reduced postoperative pain, lower complication rates, faster recovery, and shorter hospitalization duration[21-24]. These benefits are attributed to reduced surgical trauma, less blood loss, and better preservation of abdominal wall integrity. Importantly, laparoscopic resection significantly reduces the formation of intra-abdominal adhesions, which not only lowers the risk of postoperative bowel obstruction but also facilitates safer and less technically challenging reoperations in the event of tumor recurrence. This favorable adhesion profile may therefore improve the feasibility of repeat hepatic interventions, supporting a long-term, multimodal treatment strategy for patients with CRLM.
As a novel surgical approach for CRLM, laparoscopic CARe integrates the advantages of parenchymal preservation and minimally invasive techniques. By enabling precise ablation of deep-seated or centrally located small lesions while resecting larger or peripherally situated tumors, this approach maximizes curative intent while minimizing unnecessary loss of healthy liver tissue. The technique has been increasingly adopted in clinical practice[25,26]. In this multicenter retrospective study, the laparoscopic and open approaches were compared to evaluate its application in terms of perioperative and oncological outcomes.
METHODS
Study design and patients
In this multicenter retrospective cohort study, we included all patients with multiple CRLM who underwent CARe between January 2018 and December 2023 across three participating centers. Patients underwent either laparoscopic or open surgery and were accordingly grouped for analysis; all diagnoses of CRLM were confirmed by pathology. Patients who had other coexisting cancers or lacked essential primary clinical data were excluded. This study was approved by the Institutional Ethics Review Committee of the First Affiliated Hospital of University of Science and Technology of China (2025-KY-116).
Surgical procedure
A comprehensive preoperative assessment was performed for all patients, encompassing routine laboratory analyses to evaluate liver function and tumor markers, as well as imaging examinations including computed tomography (CT) and magnetic resonance imaging (MRI). The multidisciplinary team determined the optimal surgical strategy through a personalized approach, taking into account the patient’s physical status and tumor features.
CARe was performed in two primary clinical scenarios: (1) for metastases smaller than 20 mm that were deeply located within the liver parenchyma or situated near major vascular or biliary structures, where surgical resection would necessitate removal of a disproportionately large volume of healthy liver tissue; (2) for bilobar disease involving lesions close to critical intrahepatic anatomy (e.g., hepatic veins, portal pedicles, or major bile ducts), where anatomic resection would significantly reduce the FLR volume and increase the risk of postoperative liver insufficiency. Because no universal criteria exist for selecting lesions for ablation during combined therapy, this decision was guided by intraoperative judgment and surgeon experience. In our practice, nodules that directly abut major portal pedicles or hepatic veins were generally not considered suitable for thermal ablation due to (1) the heat-sink effect from adjacent high-flow vessels, which may compromise ablation efficacy; and (2) the potential for thermal injury to nearby bile ducts. However, for lesions located in close proximity - but not in direct contact - with these critical structures, ablation was selectively performed only after real-time intraoperative ultrasound confirmed that a complete ablation zone with a ≥ 5mm margin could be achieved without endangering adjacent biliary or vascular anatomy. Detailed procedural descriptions have been reported in prior publications[25,27,28].
A J-shaped incision was made during open surgery, whereas 5 trocars were placed for laparoscopic surgery. The decision to perform ablation before or after resection was made intraoperatively by the operating surgeon, based on anatomical constraints and technical feasibility. Either microwave ablation or radiofrequency ablation was used, as these are the two most commonly employed thermal ablation techniques. Intraoperative ultrasound was utilized to guide needle placement and monitor the ablation zone in real time. To ensure adequate oncologic margins, ablation was performed under real-time ultrasound guidance. Lesions ≤ 15 mm were usually treated with a single electrode pass. For nodules > 15 mm, or when a circumferential 5 mm margin could not be obtained from one trajectory, the electrode was re-inserted from 2-3 different angles to ensure complete coverage. Microwave or radio-frequency energy was applied until hyperechoic changes extended ≥ 5 mm beyond the visible tumor[9].
Definition
Postoperative complications were classified using the Clavien-Dindo grading system[29]. Synchronous metastases were considered to be lesions identified concurrently with the primary cancer, regardless of whether they are detected at the time of initial diagnosis or during the surgical resection of the primary tumor[30]. OS was measured from the date of liver surgery until death from any cause. Recurrence-free survival (RFS) was defined as the interval from surgery to the first occurrence of disease recurrence - confirmed by imaging - or death, whichever came first. Recurrences were categorized by timing: early (6 months), intermediate (6-12 months), and late (≥ 12 months)[31].
Outcome endpoints and follow-up
The primary outcome was 30-day complication rates. Secondary outcomes included OS, RFS, recurrence patterns, repeat liver resection rate, operative duration, estimated blood loss, and length of postoperative hospital stay.
The last follow-up assessment was completed in January 2025. Follow-up evaluations consisted of serum carcinoembryonic antigen (CEA) and carbohydrate antigen 19-9 (CA19-9) measurements, along with contrast-enhanced CT or MRI. These were conducted every 3 months during the first two postoperative years, and every 6 months subsequently.
Statistical analysis
To minimize selection bias, 1:1 propensity score matching (PSM) was performed using nearest neighbor matching with a caliper width of 0.2. The propensity model incorporated the following parameters: age, sex, body mass index (BMI), primary tumor location, lymph node involvement, metastatic type, lesions distribution, number of liver metastases, largest tumor diameter, receipt of neoadjuvant therapy, major hepatectomy, concomitant colorectal resection, and preoperative serum levels of CEA and CA19-9. After Benjamini-Hochberg false discovery rate (FDR) correction (q = 0.05), none of the 14 baseline covariates differed significantly between groups, confirming adequate balance achieved by PSM [Supplementary Table 1].
Continuous variables are presented as mean ± standard deviation (SD) or median (interquartile range, IQR) and were compared using the Student’s t-test or the Mann-Whitney U test, as appropriate. Categorical variables are expressed as n (%) and analyzed using the chi-square test or Fisher’s exact test, depending on group size and expected cell frequencies. Survival outcomes, including RFS and OS, were estimated using the Kaplan-Meier method with the log-rank test for comparison. All data were analyzed using R software (version 4.4.3, http://www.R-project.org). P < 0.05 was considered statistically significant.
RESULTS
Baseline characteristics
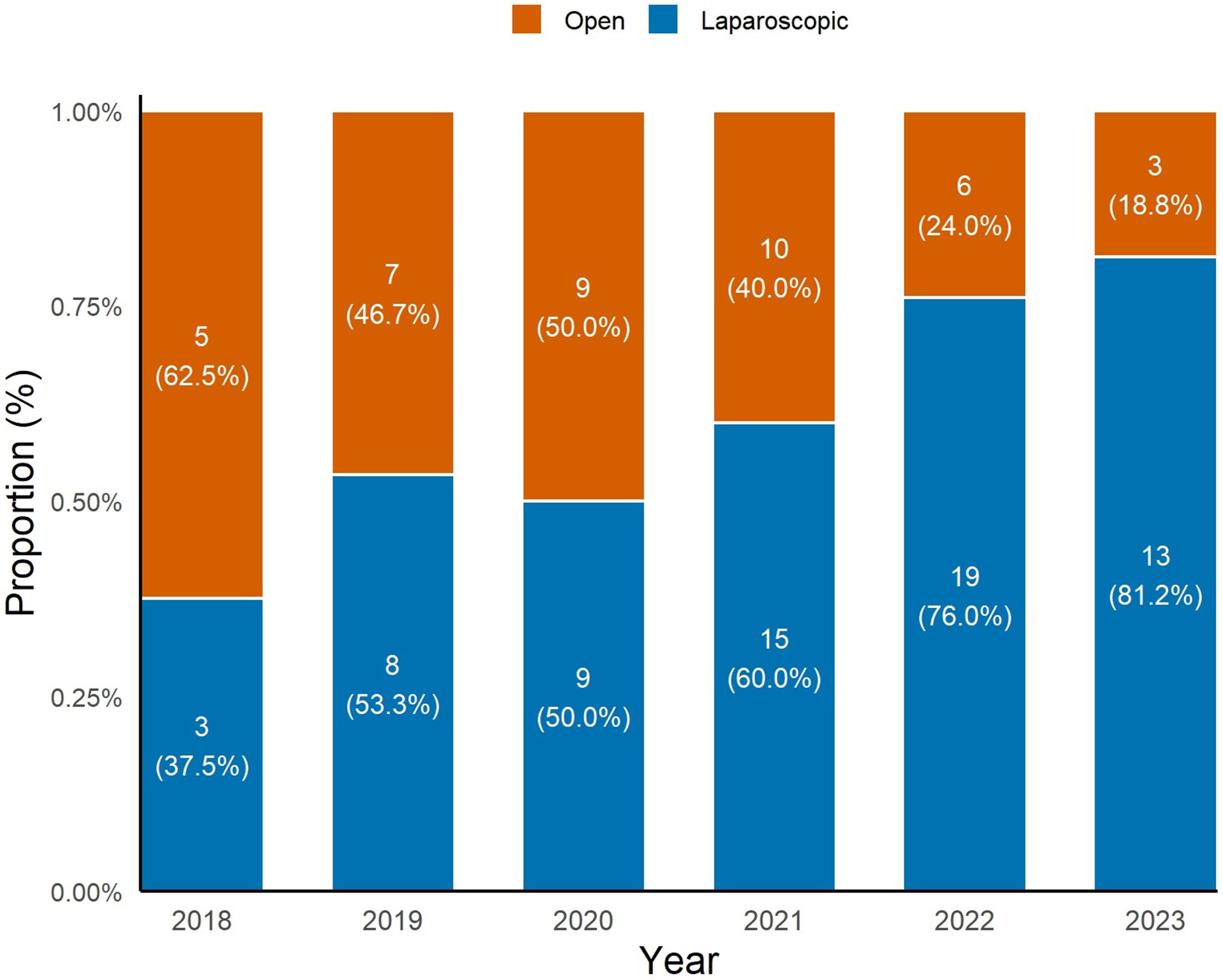
Among the 107 patients who underwent CARe, 65 (60.7%) underwent laparoscopic CARe, and 42 (39.3%) underwent open CARe. The annual proportion of patients undergoing laparoscopic CARe has risen steadily, reaching over 80% receiving minimally invasive approaches in 2023 [Figure 1]. Assessment of the whole cohort revealed that the laparoscopic group had tumors with smaller diameters (P = 0.017), more rectal primaries (P = 0.049), less synchronous colorectal resection (P = 0.039), and higher neoadjuvant therapy administration (P = 0.041). No statistically significant differences were found in the other variables (P > 0.05). After matching, 36 patient pairs were successfully matched. The demographic and clinical data before and after matching are presented in Table 1.
Figure 1. The trend in laparoscopic approach over time for combined ablation and resection in the treatment of colorectal liver metastases.
Demographics and baseline characteristics before and after propensity score matching
| Variables | Before PSM | After PSM | ||||
| Open (n = 42) | Laparoscopic (n = 65) | P | Open (n = 36) | Laparoscopic (n = 36) | P | |
| Age, years | 60.71 ± 9.92 | 57.28 ± 10.33 | 0.091 | 60.08 ± 10.05 | 60.64 ± 9.75 | 0.813 |
| Sex | - | 0.051 | - | 0.743 | ||
| Female | 7 (16.67) | 22 (33.85) | - | 6 (16.67) | 5 (13.89) | - |
| Male | 35 (83.33) | 43 (66.15) | - | 30 (83.33) | 31 (86.11) | - |
| BMI (kg/m2) | 24.56 ± 2.32 | 24.50 ± 3.44 | 0.913 | 24.67 ± 2.28 | 24.56 ± 3.27 | 0.864 |
| Primary tumor location | - | 0.049 | - | 1.000 | ||
| Rectum | 12 (28.57) | 31 (47.69) | - | 12 (33.33) | 12 (33.33) | - |
| Colon | 30 (71.43) | 34 (52.31) | - | 24 (66.67) | 24 (66.67) | - |
| Lymph nodal metastasis | - | 0.466 | - | 0.114 | ||
| No | 9 (21.43) | 18 (27.69) | - | 7 (19.44) | 13 (36.11) | - |
| Yes | 33 (78.57) | 47 (72.31) | - | 29 (80.56) | 23 (63.89) | - |
| Type of metastases | - | 0.732 | - | 0.800 | ||
| Metachronous | 11 (26.19) | 19 (29.23) | - | 11 (30.56) | 12 (33.33) | - |
| Synchronous | 31 (73.81) | 46 (70.77) | - | 25 (69.44) | 24 (66.67) | - |
| Distribution | - | 0.381 | - | 1.000 | ||
| Unilobar | 18 (43.90) | 23 (35.38) | - | 14 (38.89) | 14 (38.89) | - |
| Bilobar | 23 (56.10) | 42 (64.62) | - | 22 (61.11) | 22 (61.11) | - |
| Max diameter (cm) | 2.60 (2.02, 3.45) | 2.00 (1.50, 2.90) | 0.017 | 2.50 (2.00, 3.08) | 2.05 (1.50, 3.50) | 0.273 |
| Number of lesions | 3.50 (3.00, 5.00) | 3.00 (2.00, 5.00) | 0.598 | 3.50 (3.00, 5.00) | 3.00 (2.00, 5.00) | 0.290 |
| Number of resections | 2.00 (1.00, 3.00) | 2.00 (1.00, 3.00) | 0.277 | 2.00 (1.00, 3.00) | 1.00 (1.00, 3.00) | 0.187 |
| Number of ablations | 1.00 (1.00, 2.00) | 1.00 (1.00, 2.00) | 0.545 | 1.00 (1.00, 2.25) | 1.00 (1.00, 2.00) | 0.813 |
| Neoadjuvant therapy | - | 0.041 | - | 0.334 | ||
| No | 19 (45.24) | 17 (26.15) | - | 16 (44.44) | 12 (33.33) | - |
| Yes | 23 (54.76) | 48 (73.85) | - | 20 (55.56) | 24 (66.67) | - |
| Major hepatectomy | - | 0.364 | - | 1.000 | ||
| No | 39 (92.86) | 63 (96.92) | - | 34 (94.44) | 34 (94.44) | - |
| Yes | 3 (7.14) | 2 (3.08) | - | 2 (5.56) | 2 (5.56) | - |
| Simultaneous resection | - | 0.039 | - | 0.224 | ||
| No | 20 (47.62) | 44 (67.69) | - | 20 (55.56) | 25 (69.44) | - |
| Yes | 22 (52.38) | 21 (32.31) | - | 16 (44.44) | 11 (30.56) | - |
| CEA (ng/mL) | 8.98 (3.45, 19.36) | 6.82 (3.41, 24.07) | 0.553 | 9.57 (4.48, 20.32) | 11.65 (4.08, 29.08) | 0.474 |
| CA19-9 (U/mL) | 16.76 (8.91, 61.27) | 13.90 (6.55, 35.27) | 0.300 | 16.76 (8.39, 59.21) | 15.82 (9.87, 32.10) | 0.906 |
Before matching, the laparoscopic group included 65 patients with a total of 289 metastases, of which 155 were resected and 134 were ablated. The open surgery group included 42 patients with 168 metastases, of which 97 were resected and 71 were ablated. After PSM, the laparoscopic group comprised 36 patients with 158 metastases, including 89 resections and 69 ablations. The open group included 36 patients with 145 lesions, with 82 undergoing resection and 63 undergoing ablation.
Perioperative outcomes
There were no grade 4 or higher 30-day complications and no 90-day deaths in either group. The overall complication rates were similar between the two groups, both in the entire (P = 0.336) and matched cohort (P = 0.415). In contrast, the estimated blood loss was significantly reduced in the laparoscopic group, both before (60 vs. 125 mL, P = 0.001) and after matching (55 vs. 100 mL, P = 0.028). Furthermore, patients in the laparoscopic group had a significantly shorter length of postoperative hospital stay compared to those in the open group, in both the overall (7 vs. 8.5 days, P = 0.002) and matched cohorts (6 vs. 8 days, P = 0.005) [Table 2].
Perioperative outcomes between laparoscopic and open groups before and after propensity score matching
| Variables | Before PSM | After PSM | ||||
| Open (n = 42) | Laparoscopic (n = 65) | P | Open (n = 36) | Laparoscopic (n = 36) | P | |
| Operation time (min) | 222.50 (201.25, 315.00) | 235.00 (180.00, 320.00) | 0.667 | 222.50 (198.75, 292.50) | 210.00 (147.50, 291.25) | 0.292 |
| Blood loss (mL) | 125.00 (50.00, 200.00) | 60.00 (50.00, 100.00) | 0.001 | 100.00 (50.00, 270.00) | 55.00 (50.00, 162.50) | 0.028 |
| Blood transfusion | - | 0.614 | - | 1.000 | ||
| No | 39 (92.86) | 63 (96.92) | - | 33 (91.67) | 34 (94.44) | - |
| Yes | 3 (7.14) | 2 (3.08) | - | 3 (8.33) | 2 (5.56) | - |
| Complications (Clavien-Dindo) | - | 0.336 | - | 0.415 | ||
| 1 | 14 (33.33) | 22 (33.85) | - | 12 (33.33) | 11 (30.56) | - |
| 2 | 4 (9.52) | 5 (7.69) | - | 3 (8.33) | 2 (5.56) | - |
| 3 | 3 (7.14) | 1 (1.54) | - | 2 (5.56) | 1 (2.78) | - |
| Length of stay (days) | 8.50 (7.00, 12.75) | 7.00 (5.00, 9.00) | 0.002 | 8.00 (7.00, 12.00) | 6.00 (5.00, 8.00) | 0.005 |
Survival analysis before and after PSM
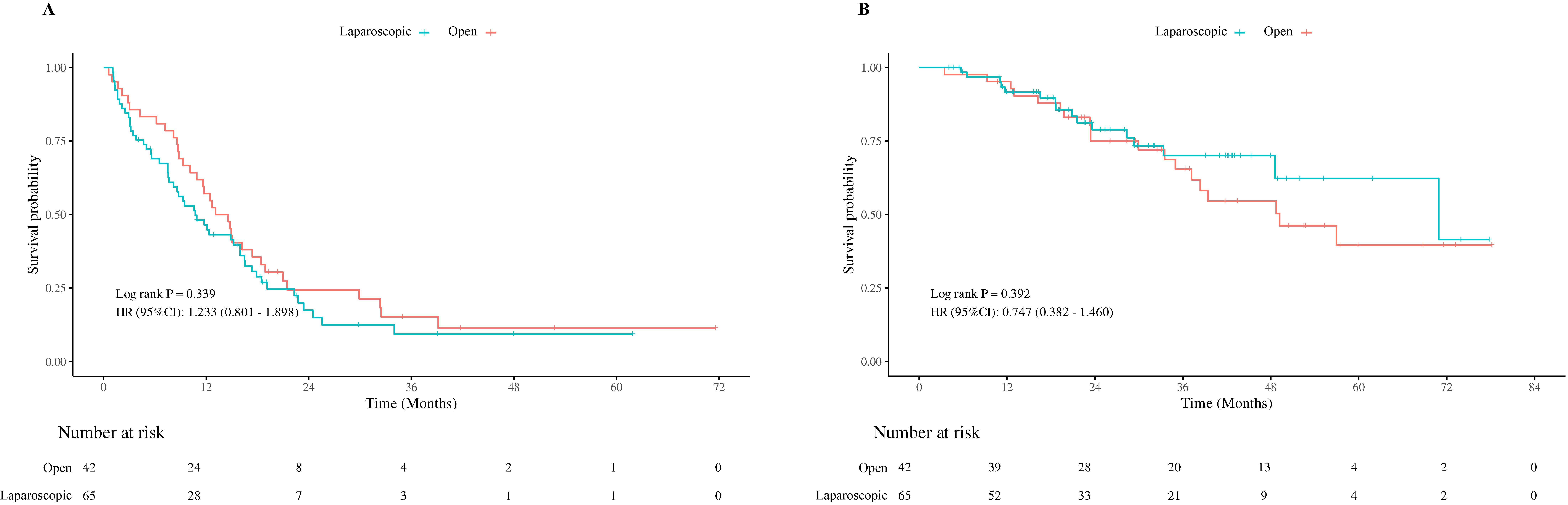
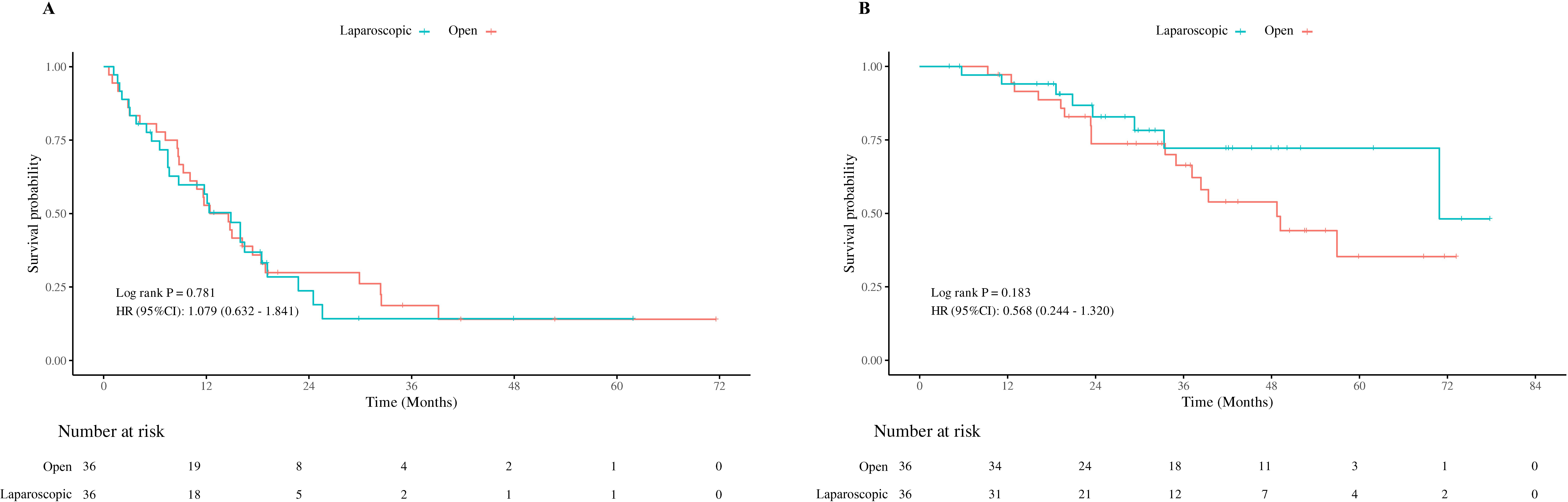
The median follow-up time for all the patients was 31.9 months. The median RFS was 10.7 months [95% confidence interval (CI): 7.6-16.0] for the laparoscopic group and 13.8 months (95%CI: 10.1-17.4) for the open group (P = 0.339). The median OS was 70.9 months [95%CI: 48.6 - not available (NA)] for the laparoscopic group and 49.2 months (95%CI: 35.0 - NA) for the open group (P = 0.392) [Figure 2]. After PSM, the median RFS was 14.9 months (95%CI: 7.5-18.5) for the laparoscopic group and 13.5 months (95%CI: 8.8-18.4) for the open group (P = 0.781). The median OS was 70.9 months (95%CI: 33.33 - NA) for the laparoscopic group and 48.7 months (95%CI: 35.0 - NA) for the open group (P = 0.183) [Figure 3].
Figure 2. Recurrence-free survival, overall survival for patients underwent combined ablation and resection before propensity score matching. (A) Recurrence-free survival (P = 0.339); (B) Overall survival (P = 0.392).
Recurrence patterns and repeat liver resection
At the time of the last follow-up, disease recurrence was observed in 87 (81.31%) patients, including 9 (8.41%) cases of local recurrence at ablation sites. Regarding the pattern of recurrence, 53 patients (60.92%) had intrahepatic recurrence, 20 (22.99%) developed isolated extrahepatic disease, and 14 (16.09%) experienced both intra- and extrahepatic recurrence. In terms of timing, recurrence occurred early in 28 patients (32.18%), intermediately in 25 (28.74%), and late in 34 (39.08%). For patients with intrahepatic recurrence, 20 (37.74%) underwent repeat liver resection, and 33 (62.26%) underwent other therapies.
Before and after matching, no statistically significant differences in the recurrence rate, recurrence site, or timing of recurrence were observed between the two approaches. However, after PSM, the laparoscopic group underwent repeat liver resection for intrahepatic recurrence more frequently (52.94% vs. 20.00%, P = 0.036) [Table 3].
Recurrence patterns and repeat liver resection before and after propensity score matching
| Variables | Before PSM | After PSM | ||||
| Open (n = 42) | Laparoscopic (n = 65) | P | Open (n = 36) | Laparoscopic (n = 36) | P | |
| Recurrence | - | 0.666 | - | 0.405 | ||
| No | 7 (16.67) | 13 (20.00) | - | 7 (19.44) | 10 (27.78) | - |
| Yes | 35 (83.33) | 52 (80.00) | - | 29 (80.56) | 26 (72.22) | - |
| Local recurrence | - | 1.00 | - | 1.00 | ||
| No | 38 (90.48) | 60 (92.31) | - | 32 (88.89) | 33 (91.67) | - |
| Yes | 4 (9.52) | 5 (7.69) | - | 4 (11.11) | 3 (8.33) | - |
| Recurrence sitea | - | 0.945 | - | 0.301 | ||
| Intrahepatic | 21 (60.00) | 32 (61.54) | - | 20 (68.97) | 17 (65.38) | - |
| Extrahepatic | 9 (25.71) | 11 (21.15) | - | 7 (24.14) | 3 (11.54) | - |
| Intra- and extrahepatic | 5 (14.29) | 9 (17.31) | - | 2 (6.90) | 6 (23.08) | - |
| Recurrence timinga | - | 0.372 | - | 0.689 | ||
| Early | 8 (22.86) | 20 (38.46) | - | 8 (27.59) | 9 (34.62) | - |
| Intermediate | 11 (31.43) | 14 (26.92) | - | 10 (34.48) | 6 (23.08) | - |
| Late | 16 (45.71) | 18 (34.62) | - | 11 (37.93) | 11 (42.31) | - |
| Repeat liver resectionb | - | 0.090 | - | 0.036 | ||
| Yes | 5 (23.81) | 15 (46.88) | - | 4 (20.00) | 9 (52.94) | - |
| No | 16 (76.19) | 17 (53.12) | - | 16 (80.00) | 8 (47.06) | - |
DISCUSSION
The findings of this study indicate that, in the management of multiple CRLM, both laparoscopic and open CARe yield comparable postoperative morbidity and long-term oncological outcomes. However, the laparoscopic approach was associated with reduced blood loss, shortened postoperative hospitalization, and a higher rate of repeat liver resection following intrahepatic recurrence. This is the first direct comparison of perioperative and long-term outcomes between these two surgical approaches using PSM.
For patients with resectable or initially unresectable multiple CRLM, CARe represents a feasible and safe strategy to achieve curative-intent treatment. As demonstrated by Liu et al., patients undergoing CARe showed comparable postoperative morbidity, disease-free survival, intrahepatic RFS, and OS to those treated with resection alone for initially unresectable CRLM[12]. In patients with resectable CRLM, no differences were observed in disease-free survival, intrahepatic RFS, or OS between the two approaches. However, the combined treatment group demonstrated a lower rate of major hepatectomy (5.2% vs. 21.9%, P = 0.001), a reduced incidence of postoperative hepatic insufficiency (0.0% vs. 5.2%, P = 0.023), and a shorter postoperative hospital stay (7 vs. 8 days, P = 0.019)[13]. Similarly, de Graaff et al. observed no significant differences in postoperative morbidity or OS between the groups, but reported a lower incidence of liver failure with CARe (0.6% vs. 1.9%, P = 0.017) compared to resection alone[16]. Consistent with these findings, another study demonstrated a reduced rate of postoperative complications with CARe (13% vs. 22%, P < 0.01), without an increase in mortality[15]. Karanicolas et al. further reported favorable perioperative outcomes in the CARe group, including reduced intraoperative blood loss and shorter postoperative hospitalization[17]. Although inter-study variations exist, the collective evidence indicates that CARe is associated with improved surgical outcomes and does not compromise oncological results in terms of recurrence or survival. Consequently, CARe has emerged as a safe and viable alternative to resection alone for CRLM management.
It is important to distinguish our research question from the ongoing resection-versus-ablation debate. The COLLISION trial - an international, randomised, phase-3 non-inferiority study - recently showed that thermal ablation of ≤ 3 cm resectable CRLM yields non-inferior OS and local control while causing significantly fewer adverse events than surgical resection[32]. These data confirm that ablation is oncologically safe for small lesions and thus legitimize its adjunctive use within the CARe strategy we examined. In the present study, ablation was selectively applied to nodules ≤ 20 mm in diameter that were either deeply seated or adjacent to critical intrahepatic structures, on the basis of prior evidence demonstrating excellent local control for this size threshold[9,10]. We acknowledge that the upper size limit for safe and effective ablation remains contentious within the CARe strategy; therefore, future work from our group will explore whether lesions > 30 mm can be incorporated into the CARe paradigm without compromising oncological adequacy. Nevertheless, the COLLISION trial also underscores the need for individualised decision-making. In the present study, both laparoscopic and open CARe retain surgical resection as the primary modality, with ablation applied only to additional lesions not amenable to safe resection; our comparison therefore focuses on the optimal surgical approach (laparoscopic versus open) within a resection-centred CARe paradigm, rather than on the choice between resection and ablation.
Laparoscopic liver resection is increasingly used for CRLM, offering perioperative benefits over open surgery and supported by high-level evidence from randomized controlled trials. The first randomized controlled trial (OSLO-COMET)[21] comparing laparoscopic and open resection for CRLM revealed that laparoscopic surgery was associated with lower complication rates and shorter length of postoperative hospital stays. Follow-up data revealed no difference in oncological results between the laparoscopic and open groups[22]. The LapOpHuva[23] randomized controlled trial reached the same conclusion. However, in these two trials, patients with CRLM had a low tumor burden and underwent minor hepatectomy. Consequently, it is uncertain whether laparoscopic surgery remains superior to open surgery in the context of major hepatectomy. Laparoscopic and open hemihepatectomy were compared in the randomized controlled trial (ORANGE II PLUS)[24]. There was no significant difference in the postoperative complication rates (14.5% vs. 16.9%, P = 0.58) between the two groups. However, the laparoscopic hemihepatectomy group demonstrated faster postoperative functional recovery and higher quality of life scores. Notably, to facilitate patient recruitment, the enrollment criteria in the ORANGE II PLUS trial were not restricted to a specific disease. Although most patients had CRLM, the ability to explore oncological outcomes is limited.
Laparoscopic CARe has been performed in the management of CRLM[25], but few relevant studies exist. Our recent findings indicate that, for patients with multiple CRLM, laparoscopic CARe achieves perioperative outcomes, recurrence patterns, RFS, and OS comparable to those of laparoscopic liver resection alone[27]. Serenari et al.[33] explored predictors of textbook outcomes[34] following CARe for CRLM. In their multivariate analysis, a minimally invasive approach emerged as a significant independent predictor of achieving textbook outcomes. Vandeputte et al. conducted a retrospective analysis of the surgical outcomes following CARe in 54 patients[26]. 32 (59.3%) of whom underwent minimally invasive surgery. Compared with the open group, the minimally invasive group had a lower blood loss volume (90 vs. 200 mL, P = 0.005), fewer patients who underwent the Pringle manoeuvre (25.0% vs. 72.7%, P = 0.001), less complications (9.4% vs. 40.9%, P = 0.034), and shorter length of postoperative hospital stay (4 vs. 8 days, P < 0.001). However, the baseline characteristics were not evenly distributed between the two groups in this study. The Southampton difficulty score for liver resection, number of lesions and extent of bilobar involvement were significantly greater in the open CARe group[35]. Therefore, these results should be interpreted with caution. Similarly, heterogeneity in preoperative baseline characteristics between the two groups was also found in our study. We performed PSM in this study to account for differences in the impact of baseline characteristics on result reliability between groups.
The most important finding of this study was that, compared with open CARe, laparoscopic CARe was associated with higher repeat resection rate following intrahepatic recurrence. This increased likelihood of repeat intervention may be attributed to better preservation of liver functional reserve and reduced postoperative inflammatory response in the laparoscopic group. Minimally invasive techniques minimize surgical trauma, thereby preserving hepatic parenchymal integrity and creating more favorable conditions for subsequent procedures. Furthermore, the lower incidence of intra-abdominal adhesions facilitates easier surgical dissection during re-resection, reduces operative risks, and expands the pool of patients eligible for curative-intent surgery upon recurrence. Previous studies revealed that repeated resection is the optimal strategy for improving oncological outcomes for patients with recurrent CRLM[31,36-38]. Takeda et al. reported that the cure rates following initial, second, and third resections for CRLM were similar (23.0% vs. 30% vs. 22.5%, respectively)[36]. Moreover, multivariate analysis revealed that repeat resection was an independent predictor of curative resection. In our study, although OS difference was not statistically significant, a numerically longer survival was observed in the laparoscopic group. This may be attributed to comparable recurrence rates between the two groups, coupled with a greater tendency toward repeat liver resection in the laparoscopic group following intrahepatic recurrence.
This study had several limitations. First, this is a retrospective study with a relatively small sample size, making selection bias inevitable. Although PSM was carefully performed to mitigate imbalances, residual bias may still persist. Therefore, prospective, large-scale, randomized controlled trials are warranted to deliver high-level evidence. Second, there are currently no universally accepted criteria for selecting ablation over resection for liver lesions. Treatment decisions were based solely on surgeon preference and clinical judgment, which may have introduced additional selection bias in the allocation of therapeutic modalities. Third, the follow-up duration was relatively short for evaluating long-term survival outcomes. As such, interpretations of overall and RFS should be made cautiously. Given that late recurrences can occur in patients with CRLM, extended follow-up is essential to fully assess the oncological impact of both surgical approaches.
In conclusion, laparoscopic CARe is associated with reduced blood loss, shorter hospital stays, and a higher likelihood of repeat resection for recurrent disease compared to open surgery. Although long-term oncological survival outcomes are comparable, these perioperative advantages support laparoscopic CARe as a feasible and safe surgical option for patients with multiple CRLM.
DECLARATIONS
Authors’ contributions
Conceptualization: Song H, Li Z, Meng F, Wang J, Han B
Methodology: Song H, Li Z, Meng F, Wang J
Software: Song H, Ling X
Validation: Song H, Li Z, Meng F, Chen L, Zhang S, Wang Y, Zhang F, Song R, Wu Z
Formal analysis: Song H, Ling X, Chen L
Investigation: Li Z, Meng F, Ling X, Chen L, Zhang S, Wang Y, Zhang F, Song R, Wu Z
Resources: Han B, Wu Z
Data curation: Li Z, Meng F, Ling X, Chen L, Zhang S, Wang Y, Zhang F, Song R
Writing - original draft: Song H, Li Z, Meng F
Writing - review and editing: Song H, Li Z, Meng F, Wang J, Han B
Visualization: Song H, Ling X
Supervision: Wang J, Han B
Project administration: Wang J, Han B
Funding acquisition: Wang J
Availability of data and materials
The datasets are available from the corresponding authors upon reasonable request and with Ethics Committee approval.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (No. 82170618), Anhui University Outstanding Youth Research Project (2024AH020016), Research Institute of Pioneer Medicine and Frontier Technology, Hefei Comprehensive National Science Center (2023IHM01020), and Key Research and Development Program of Anhui Province (202204295107020019).
Conflicts of interest
Wang J is a Junior Chief Editor of the Junior Editorial Board of Mini-invasive Surgery. Wang J was not involved in any stage of the editorial process for this manuscript, including reviewer selection, manuscript handling, or decision-making. The other authors declare that they have no conflicts of interest.
Ethical approval and consent to participate
The study protocol was approved by the Institutional Ethics Review Committee of the First Affiliated Hospital of University of Science and Technology of China (approval No. 2025-KY-116). All procedures were conducted in accordance with the Declaration of Helsinki. Because this is a retrospective analysis of anonymized data, the requirement for written informed consent was waived by the Ethics Committee.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Bray F, Laversanne M, Sung H, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-63.
2. Cervantes A, Adam R, Roselló S, et al.; ESMO Guidelines Committee. Electronic address: [email protected]. Metastatic colorectal cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023;34:10-32.
3. Siegel RL, Wagle NS, Cercek A, Smith RA, Jemal A. Colorectal cancer statistics, 2023. CA Cancer J Clin. 2023;73:233-54.
4. Creasy JM, Sadot E, Koerkamp BG, et al. Actual 10-year survival after hepatic resection of colorectal liver metastases: what factors preclude cure? Surgery. 2018;163:1238-44.
5. Ratti F, Cipriani F, Fiorentini G, et al. Evolution of surgical treatment of colorectal liver metastases in the real world: single center experience in 1212 cases. Cancers. 2021:13.
6. Bond MJG, Bolhuis K, Loosveld OJL, et al.; Dutch Colorectal Cancer Group. First-line systemic treatment for initially unresectable colorectal liver metastases: post hoc analysis of the CAIRO5 randomized clinical trial. JAMA Oncol. 2025;11:36-45.
7. Adam R, Delvart V, Pascal G, et al. Rescue surgery for unresectable colorectal liver metastases downstaged by chemotherapy: a model to predict long-term survival. Ann Surg. 2004;240:644-57; discussion 657.
8. Allard MA, Adam R, Giuliante F, et al. Long-term outcomes of patients with 10 or more colorectal liver metastases. Br J Cancer. 2017;117:604-11.
9. Solbiati L, Ahmed M, Cova L, Ierace T, Brioschi M, Goldberg SN. Small liver colorectal metastases treated with percutaneous radiofrequency ablation: local response rate and long-term survival with up to 10-year follow-up. Radiology. 2012;265:958-68.
10. Karagkounis G, McIntyre SM, Wang T, et al. Rates and patterns of recurrence after microwave ablation of colorectal liver metastases: a per lesion analysis of 416 tumors in the era of 2.45 GHz generators. Ann Surg Oncol. 2023;30:6571-8.
11. Imai K, Allard MA, Castro Benitez C, et al. Long-term outcomes of radiofrequency ablation combined with hepatectomy compared with hepatectomy alone for colorectal liver metastases. Br J Surg. 2017;104:570-9.
12. Liu M, Wang K, Wang Y, et al. Short- and long-term outcomes of hepatectomy combined with intraoperative radiofrequency ablation for patients with multiple primarily unresectable colorectal liver metastases: a propensity matching analysis. HPB. 2021;23:1586-94.
13. Liu M, Wang Y, Wang K, et al. Combined ablation and resection (CARe) for resectable colorectal cancer liver metastases - a propensity score matching study. Eur J Surg Oncol. 2023;49:106931.
14. Elfrink AKE, Nieuwenhuizen S, van den Tol MP, et al.; Dutch Hepato Biliary Audit Group. Hospital variation in combined liver resection and thermal ablation for colorectal liver metastases and impact on short-term postoperative outcomes: a nationwide population-based study. HPB. 2021;23:827-39.
15. Xourafas D, Pawlik TM, Ejaz A, et al. Impact of concomitant ablation on the perioperative outcomes of patients with colorectal liver metastases undergoing hepatectomy: a propensity score matched nationwide analysis. HPB. 2019;21:1079-86.
16. Graaff MR, Klaase JM, den Dulk M, et al; Dutch Hepato Biliary Audit Group, Collaborators. Trends and overall survival after combined liver resection and thermal ablation of colorectal liver metastases: a nationwide population-based propensity score-matched study. HPB. 2024;26:34-43.
17. Karanicolas PJ, Jarnagin WR, Gonen M, et al. Long-term outcomes following tumor ablation for treatment of bilateral colorectal liver metastases. JAMA Surg. 2013;148:597-601.
18. Dai Y, Zhang Y, He W, et al. Long-term outcome for colorectal liver metastases: combining hepatectomy with intraoperative ultrasound guided open microwave ablation versus hepatectomy alone. Int J Hyperthermia. 2021;38:372-81.
19. Amygdalos I, Hitpass L, Schmidt F, et al. Survival after combined resection and ablation is not inferior to that after resection alone, in patients with four or more colorectal liver metastases. Langenbecks Arch Surg. 2023;408:343.
20. Sawa Y, Kawaguchi Y, Miyata A, et al. Gradual expansion of the indications for minimally invasive liver resection to include highly complex procedures may improve postoperative outcomes. Mini-invasive Surg. 2024;8:30.
21. Fretland ÅA, Dagenborg VJ, Bjørnelv GMW, et al. Laparoscopic versus open resection for colorectal liver metastases: the OSLO-COMET randomized controlled trial. Ann Surg. 2018;267:199-207.
22. Aghayan DL, Kazaryan AM, Dagenborg VJ, et al.; OSLO-COMET Survival Study Collaborators. Long-term oncologic outcomes after laparoscopic versus open resection for colorectal liver metastases: a randomized trial. Ann Intern Med. 2021;174:175-82.
23. Robles-Campos R, Lopez-Lopez V, Brusadin R, et al. Open versus minimally invasive liver surgery for colorectal liver metastases (LapOpHuva): a prospective randomized controlled trial. Surg Endosc. 2019;33:3926-36.
24. Fichtinger RS, Aldrighetti LA, Abu Hilal M, et al.; ORANGE II PLUS Collaborative. Laparoscopic versus open hemihepatectomy: the ORANGE II PLUS multicenter randomized controlled trial. J Clin Oncol. 2024;42:1799-809.
25. Zhang D, Chen Z, Zhang L, et al. Application of laparoscopic hepatectomy combined with intraoperative microwave ablation in colorectal cancer liver metastasis. J Vis Exp. 2023.
26. Vandeputte M, Saveyn T, Lutin B, De Meyere C, Parmentier I, D’Hondt M. Combined ablation and resection for colorectal liver metastases in the minimally invasive surgical era. Surg Laparosc Endosc Percutan Tech. 2023;33:121-8.
27. Song H, Li Z, Zhang S, et al. Laparoscopic resection combined with ablation for multiple colorectal liver metastases: a multicentre propensity-matched analysis. Surg Endosc. 2025;39:6514-24.
28. Sasaki K, Margonis GA, Andreatos N, et al. Combined resection and RFA in colorectal liver metastases: stratification of long-term outcomes. J Surg Res. 2016;206:182-9.
29. Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205-13.
30. Siriwardena AK, Serrablo A, Fretland ÅA, et al. Multisocietal European consensus on the terminology, diagnosis, and management of patients with synchronous colorectal cancer and liver metastases: an E-AHPBA consensus in partnership with ESSO, ESCP, ESGAR, and CIRSE. Br J Surg. 2023;110:1161-70.
31. Boyev A, Tzeng CD, Maki H, et al. Local therapy improves survival for early recurrence after resection of colorectal liver metastases. Ann Surg Oncol. 2024;31:2547-56.
32. van der Lei S, Puijk RS, Dijkstra M, et al. Thermal ablation versus surgical resection of small-size colorectal liver metastases (COLLISION): an international, randomised, controlled, phase 3 non-inferiority trial. Lancet Oncol. 2025;26:187-99.
33. Serenari M, Ratti F, Stocco A, et al. Achievement of textbook outcome after hepatectomy combined with thermal ablation for colorectal liver metastases. Surg Endosc. 2024;38:2611-21.
34. Görgec B, Benedetti Cacciaguerra A, Pawlik TM, et al. An international expert Delphi consensus on defining Textbook Outcome in Liver Surgery (TOLS). Ann Surg. 2023;277:821-8.
35. Halls MC, Berardi G, Cipriani F, et al. Development and validation of a difficulty score to predict intraoperative complications during laparoscopic liver resection. Br J Surg. 2018;105:1182-91.
36. Takeda Y, Mise Y, Ito H, et al. Repeat resection for advanced colorectal liver metastases-does it have the potential for cure? World J Surg. 2022;46:2253-61.
37. Dupré A, Jones RP, Diaz-Nieto R, Fenwick SW, Poston GJ, Malik HZ. Curative-intent treatment of recurrent colorectal liver metastases: a comparison between ablation and resection. Eur J Surg Oncol. 2017;43:1901-7.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].