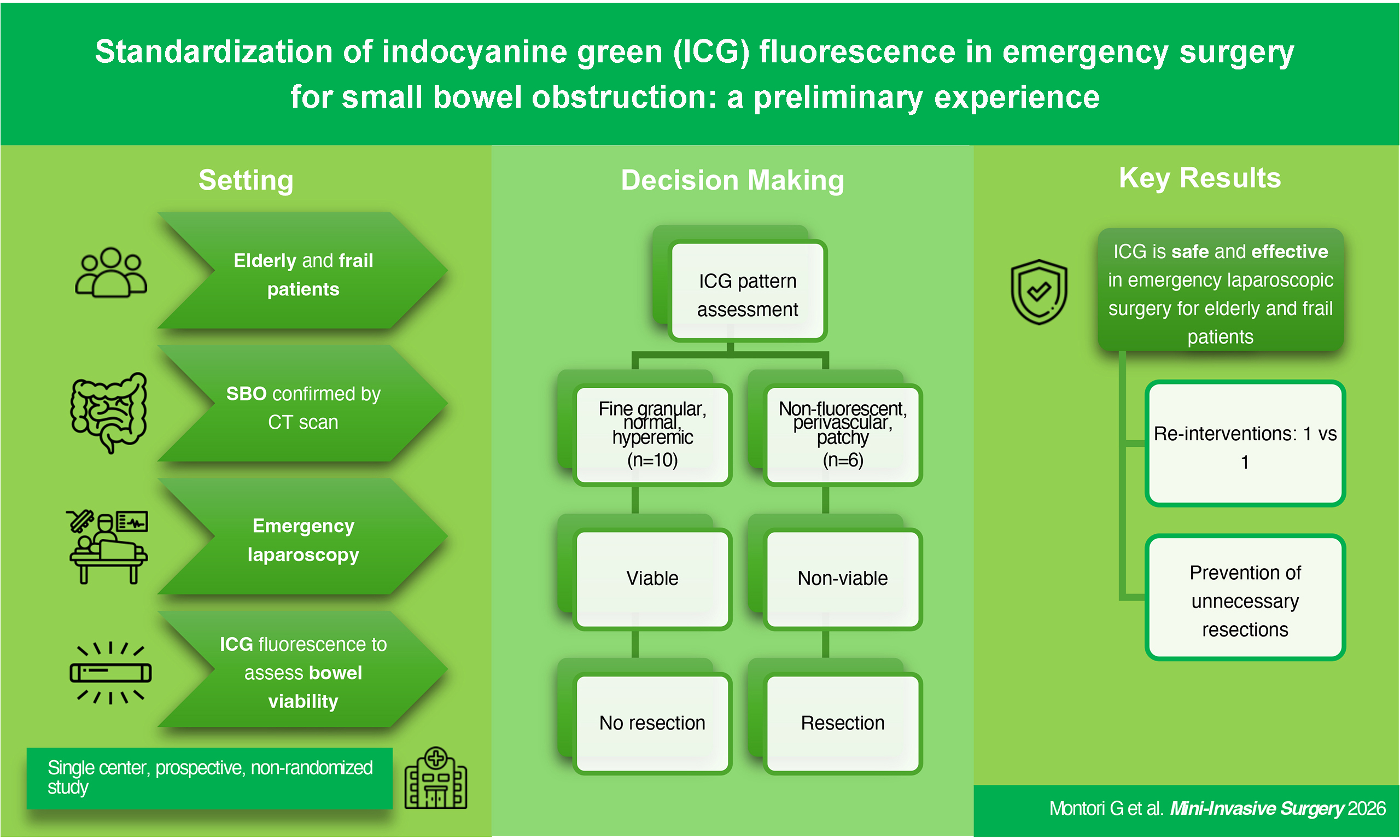
Standardization of indocyanine green (ICG) fluorescence in emergency surgery for small bowel obstruction: a preliminary experience
Abstract
Aim: Indocyanine green (ICG) fluorescence is not routinely used in acute small bowel obstruction (SBO), and standardized methods for its use in assessing intestinal perfusion, particularly via laparoscopy, remain lacking. Therefore, we aimed to explore its role in acute SBO in elderly and frail patients undergoing operative management.
Methods: In this single-center, prospective, non-randomized study, we included a case series of patients who underwent emergency laparoscopic surgery for SBO from January 2024 to January 2025 at an Italian community hospital. Patients included were those with acute SBO confirmed by computed tomography scan, who provided signed informed consent, had no allergy to ICG, and were ≥ 18 years old. In borderline cases of intestinal ischemia, we standardized the application of ICG fluorescence to help determine the need for intestinal resection: ICG dosage, timing of injection, timing of fluorescence imaging capture and assessment of the intestinal segment. The postoperative course and the need for re-intervention were evaluated.
Results: ICG fluorescence was applied in 16 patients. Half of the cases were male, the median age was 72 years old, 63% presented an American Society of Anesthesiologists (ASA) Physical Status score of 3-4, and the median Charlson Comorbidity Index was 3. Conversion rate was 37%. Main causes of SBO were strangulated hernia, adhesive bands, adhesions and vascular ischemia. In 6 patients, ICG fluorescence showed a non-fluorescent or perivascular pattern, prompting an intestinal resection. Two patients needed re-intervention. Postoperative complications occurred in 43% of the patients.
Conclusion: SBO often affects elderly and frail patients. ICG fluorescence serves as a useful and feasible tool in emergency laparoscopic surgery. It facilitates the surgical assessment in borderline cases of intestinal ischemia, thereby preventing unnecessary bowel resections.
Keywords
INTRODUCTION
Indocyanine green (ICG) is a liquid dye that can be injected into the human bloodstream with minimal adverse effects and becomes fluorescent when excited with a specific light wavelength[1]. Moreover, due to the wide availability of ICG and the technical simplicity of its enhanced fluorescence, this tool has been increasingly applied in several areas of general surgery, and, to date, extensive research has been conducted on this topic. However, most of these studies deal with the intraoperative use of ICG in elective and oncologic surgery, and they are focused on the lymph node mapping in oncologic surgery, the study of the biliary tract during cholecystectomy, or the evaluation of anastomosis perfusion after gastrointestinal resections[1,2]. Instead, only few studies have assessed the feasibility and benefits of its implementation in emergency surgery, especially in cases of small bowel obstruction (SBO)[3]. Furthermore, there is a lack of standardized protocols and randomized clinical trials in both open and minimally invasive surgery, with this deficiency being particularly evident in the latter[4]. A few case reports[2,5,6] have investigated the role of ICG in acute mesenteric ischemia; however, these represent a limited number of cases, managed exclusively via open surgery. A recent systematic review explores the role of ICG in acute care settings, especially in acute cholecystitis, ischemic bowel, and trauma[4]. Nevertheless, although the included studies are case reports, the surgical technique (open or minimally invasive) is not specified, and in just 21.6% of the cases a more conservative approach (defined as no resection or resection less extensive than predicted) could be performed; conversely, in 15% of cases the use of ICG led to a more extensive resection than anticipated[4]. These findings suggest that ICG fluorescence plays a key role in the surgical decision-making process, thereby leading to better outcomes.
In several studies, ICG has been employed for the evaluation of intestinal blood flow in patients undergoing open surgery for strangulated bowel obstruction, and a correlation with the pathological findings of the resected intestinal segment and with the postoperative course has been observed. In a few studies, a classification of intestinal patterns of fluorescence has been applied to assess bowel perfusion and guide the decision regarding intestinal resection, ultimately optimizing emergency surgical management[7].
Given the increasing adoption of the laparoscopic approach for SBO, we aimed to standardize the use of ICG fluorescence in emergency laparoscopic surgery. In this case series, we report our initial experience at an Italian community hospital using this tool in emergency laparoscopic surgery to assess the extent of bowel ischemia and determine the need for resection in patients with SBO.
METHODS
Between January 2024 and January 2025, patients aged ≥ 18 years, with a diagnosis of SBO confirmed by computed tomography (CT) scan at admission to the Emergency Room of Vittorio Veneto Hospital and subsequently undergoing emergency surgery, were included in this single-center, prospective, non-randomized study. Informed consent for the use of ICG was obtained preoperatively, and patients with a known allergy to ICG were excluded. We included all patients hospitalized for acute SBO undergoing operative management (OM) during the study period [Figure 1]. ASA score, as well as patient characteristics, were collected.
Figure 1. Inclusion and exclusion criteria. ASBO: Acute small bowel obstruction; CT: computed tomography; ED: emergency department; ICG: indocyanine green.
All patients initially underwent a laparoscopic approach, with induction of 10-mmHg pneumoperitoneum via an open access technique to reduce the risk of bowel injury. Conversion to open surgery was performed if required.
Intraoperatively, the viability of the compromised intestinal segment was first assessed clinically (evaluating peristalsis, intestinal wall status, signs of necrosis, perforation and mesentery status), and ICG fluorescence was employed in borderline cases - according to the Bulkley classification[8] - to determine the need for intestinal resection.
In all patients, following adhesiolysis, the abdominal cavity was irrigated with warm water via a catheter; the bowel was then kept in warm water for 10 min to facilitate reperfusion, and ICG control was subsequently performed using the Karl Storz Image1 S equipment for the ICG fluorescence-guided laparoscopy.
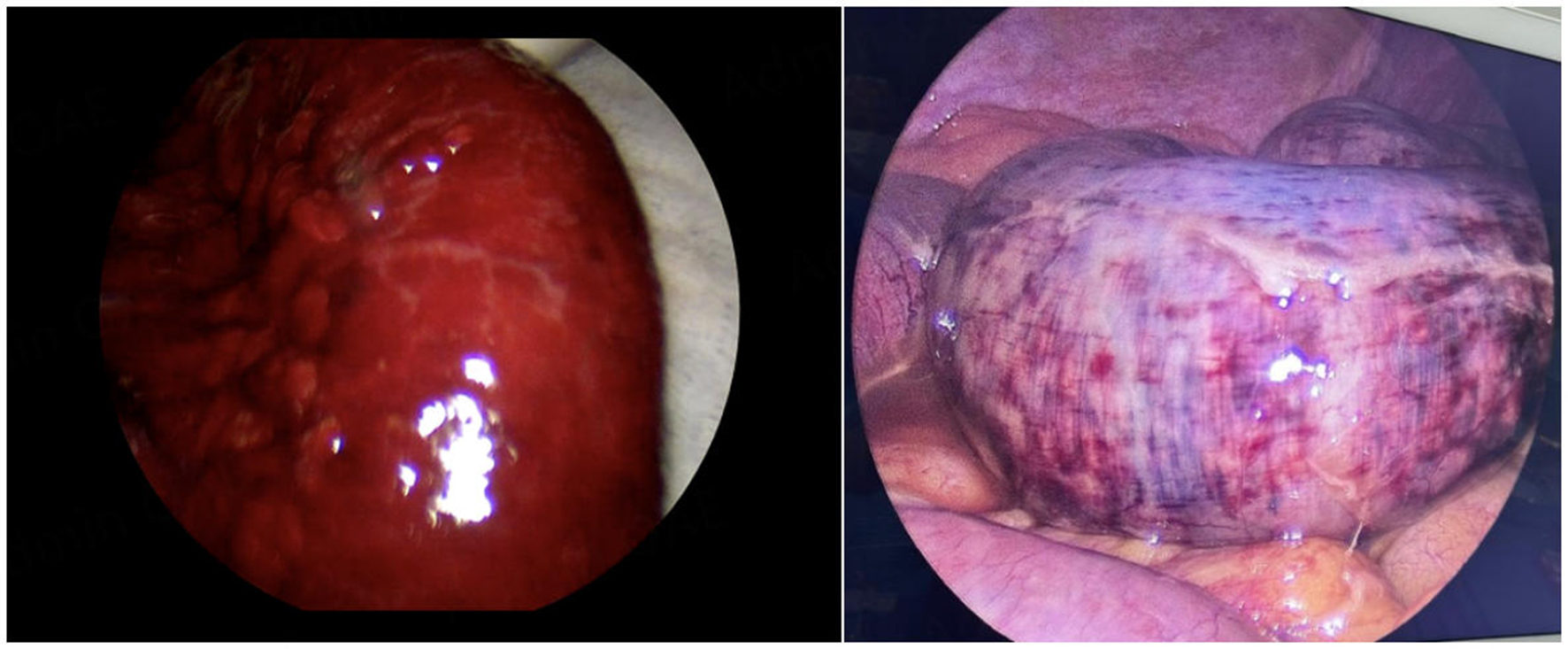
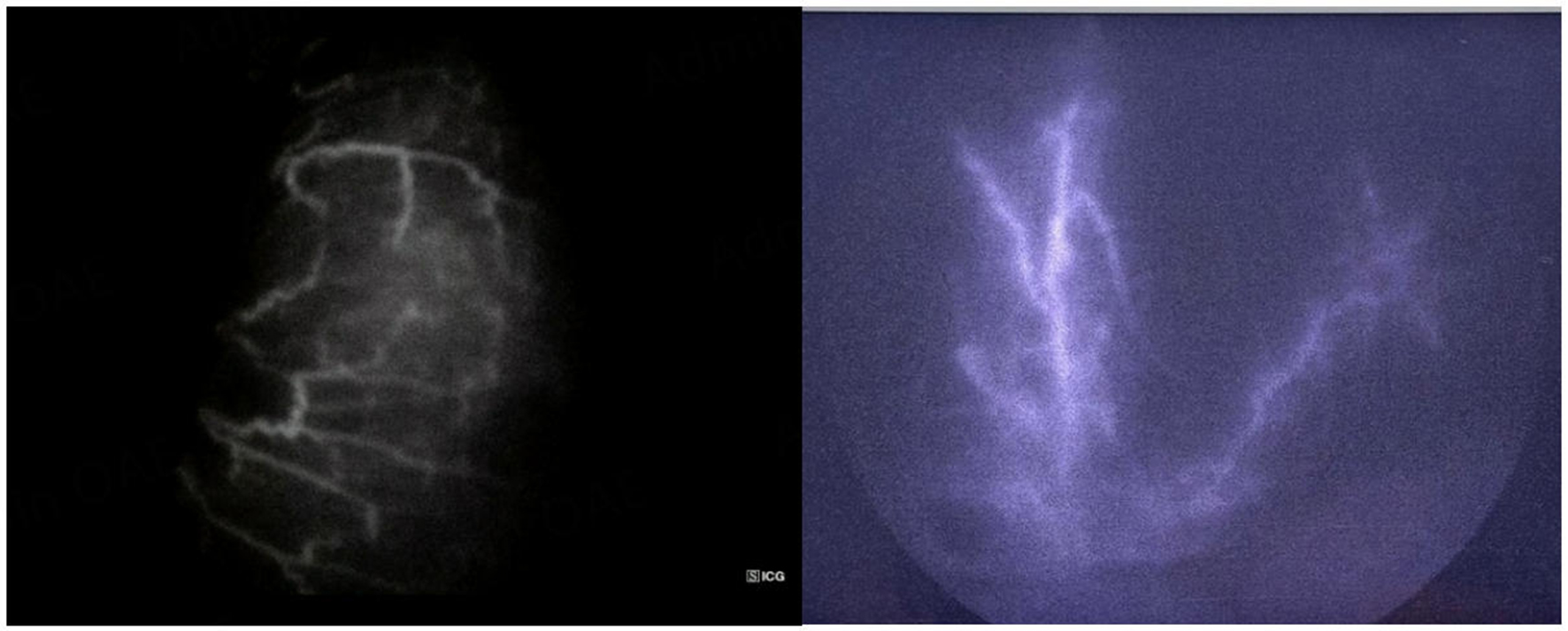
The ICG protocol was standardized in terms of dosage (0.1 mg/kg), timing of injection through a peripheral vein (after 10 min of liberation of the bowel obstruction), timing of fluorescence imaging capture (30-40 s after injection) and evaluation of the ischemic intestinal segments compared with the non-ischemic segments (based on the fluorescence patterns according to Bulkley et al.[8]: hyperemic, normal, fine granular, patchy, perivascular, non-fluorescent). A non-fluorescent, perivascular or patchy pattern was considered non-viable, and intestinal resection was consistently performed [Figures 2 and 3]. On the contrary, a fine granular, normal or hyperemic pattern was considered as a viable intestinal segment.
In case of viability of the intestinal segment after ICG, no resection was performed. In contrast, in the case of a risky pattern or a non-fluorescent pattern, a segmental resection was carried out.
Data about patient characteristics [age, sex, ASA score, Charlson Comorbidity Index (CCI), previous abdominal surgery], time of symptom onset, postoperative course, complications [according to the Clavien-Dindo (CD) classification] and need for a re-intervention were collected for all patients (bowel resected and no-bowel resected), and are reported in Tables 1 and 2.
Characteristics of patients in whom ICG was used to evaluate ischemic bowel segments
| Case (n) | Sex (M/F) | Age (years) | ASA (1, 2, 3, 4) | Previous abdominal surgery (yes: +, no: -) | CCI | Cause of SBO | Time of symptoms to surgery (h) |
| 1 | F | 71 | 4 | + | 3 | Strangulated hernia | 72 |
| 2 | M | 74 | 3 | + | 4 | Adherence | 24 |
| 3 | F | 80 | 3 | + | 4 | Strangulated hernia | 6 |
| 4 | F | 51 | 3 | + | 1 | Strangulated hernia | 24 |
| 5 | F | 94 | 4 | + | 4 | Bridle | 5 |
| 6 | F | 72 | 3 | + | 3 | Bridle | 3 |
| 7 | M | 60 | 3 | + | 2 | Strangulated hernia | 6 |
| 8 | F | 71 | 2 | + | 3 | Strangulated hernia | 4 |
| 9 | M | 74 | 2 | + | 3 | Bridle | 10 |
| 10 | M | 80 | 3 | + | 5 | Adherence | NA |
| 11 | F | 72 | 2 | - | 3 | Strangulated hernia | NA |
| 12 | M | 56 | 2 | - | 1 | Inflammatory intestinal disease | 72 |
| 13 | M | 79 | 4 | - | 8 | Vascular ischemia | 48 |
| 14 | M | 35 | 1 | - | 0 | Inflammatory intestinal disease | 72 |
| 15 | M | 70 | 3 | - | 3 | Bridle | NA |
| 16 | F | 76 | 2 | + | 3 | Bridle | NA |
Summary of the operative and postoperative course of the cases
| Case (n) | Conversion to open surgery (yes: +, no: -) | Pattern of fluorescence | Intestinal resection (yes: +, no: -) | Post-operative complications | CD | Need of reintervention (yes: +, no: -) | Mortality (yes: +, no: -) |
| 1 | + | Non-fluorescent | + | Wound infection | 2 | - | - |
| 2 | - | Perivascular | + | Recurrence SBO | 3b | + | - |
| 3 | - | Normal | - | - | - | - | - |
| 4 | - | Normal | - | - | - | - | - |
| 5 | - | Fine granular | - | MOF | 5 | - | + |
| 6 | + | Non-fluorescent | + | - | - | - | - |
| 7 | + | Fine granular | - | Recurrence SBO | 3b | + | - |
| 8 | - | Non-fluorescent | + | - | - | - | - |
| 9 | - | Fine granular | - | - | - | - | - |
| 10 | + | Perivascular | + | Pulmonary infection with respiratory failure and cardiac arrest | 5 | - | + |
| 11 | - | Fine granular | - | - | - | - | - |
| 12 | - | Normal | - | Slow intestinal recovery needing parenteral nutrition | 2 | - | - |
| 13 | + | Perivascular | + | Atrial fibrillation | 2 | - | - |
| 14 | - | Normal | - | - | - | - | - |
| 15 | - | Normal | - | - | - | - | - |
| 16 | + | Fine granular | - | - | - | - | - |
Statistical analysis was performed on continuous variables by calculating the mean, median, range, and percentages.
RESULTS
During the study period, 23 patients were admitted with a diagnosis of SBO. Non-OM was successfully performed in 7 patients, while OM was required for 16 patients. In those undergoing OM, ICG fluorescence was used to assess the perfusion of the affected intestinal segment responsible for SBO and to guide the decision regarding intestinal resection. The characteristics of each patient are reported in Table 1.
Half of the patients were male (50%). The median age was 72 years (range 35-94). Sixty-three percent of patients presented with ASA 3-4, and the median CCI was 3. The main causes of SBO were strangulated hernia (38%), followed by adhesive bands (31%), adhesions (12%), suspected inflammatory bowel disease (IBD) (13%), and vascular ischemia (6%). Among patients requiring OM, the conversion rate was 37% due to massive adhesions, hemodynamic instability, or the need for intestinal resection.
In 6 cases (38%), the ICG fluorescence showed a non-fluorescent or perivascular pattern, prompting resection of the affected segment and anastomosis. These intraoperative findings were consistent with preoperative CT scans suggestive of intestinal ischemia. Subsequently, fluorescence imaging was performed again to assess the viability of the suture line.
In one case (patient no. 5), the intraoperative ICG pattern appears to have been misinterpreted: despite CT findings and intraoperative macroscopic assessment suggesting ischemia, the pattern of the involved segment was ultimately classified as fine granular. The patient subsequently succumbed to multiple organ failure (MOF), which could likely be attributed to misinterpretation of the intraoperative ICG pattern, as well as advanced age and compromised clinical condition.
In contrast, the remaining 10 patients exhibited a fine granular or normal pattern 10 min after release of the obstruction, thereby avoiding resection. The median surgery time was 108 min (range 40-210).
In the non-resection group, only one patient required re-intervention (10%): in this case, during the initial surgery, ICG fluorescence had displayed a borderline pattern, which was initially interpreted as viable, and thus resection was avoided. Postoperatively, a worsening in the clinical course was observed; therefore, a re-intervention with intestinal resection was performed. Conversely, in the bowel resection group, only one patient needed re-intervention because of recurrence of intestinal obstruction caused by adhesions.
Table 3 presents the correlation between preoperative radiologic findings and intraoperative ICG assessment. Radiologic evidence of ischemia (definitive or suspected) was observed in 9 out of 16 patients (56%). CT findings were confirmed intraoperatively in 6 patients, all of whom underwent targeted bowel resection. In one patient (n. 5), CT findings were not corroborated by intraoperative fluorescence; the patient died, suggesting a probable misinterpretation of the intraoperative data. In contrast, in case n. 9, CT suggested ischemia, but the fluorescence pattern was fine granular; similarly, in case n. 12, CT raised suspicion of ischemia, but the intraoperative ICG was normal: consequently, resection was avoided in both patients. In one case (n. 10), due to an acute kidney injury (AKI), a contrast-enhanced CT scan could not be performed; while the preoperative data were incomplete, the fluorescence pattern confirmed ischemia, and the involved segment was accordingly resected.
Correlations between preoperative radiologic findings with intraoperative perfusion assessment using ICG
| Case (n) | Radiologic exam | CT contraindication | Radiologic signs for ischemia | Intraoperative signs for ischemia | Reasons for not indicating contrast CT |
| 1 | Contrast CT | - | pos | pos | - |
| 2 | Contrast CT | - | Suspected | pos | - |
| 3 | US | - | - | neg | Incarcerated inguinal hernia |
| 4 | Plain CT | Yes | neg | neg | AKI + CKD |
| 5 | Contrast CT | - | Suspected | pos | - |
| 6 | Contrast CT | - | pos | pos | - |
| 7 | Contrast X ray | No | - | neg | Post-operative complication |
| 8 | Contrast CT | - | Suspected | pos | - |
| 9 | Contrast CT | - | pos | neg | - |
| 10 | Plain CT | Yes | neg | pos | AKI |
| 11 | Contrast CT | - | neg | neg | - |
| 12 | Contrast CT | - | Suspected | neg | - |
| 13 | Contrast CT | - | pos | pos | - |
| 14 | Contrast CT | - | neg | neg | - |
| 15 | Contrast CT | - | neg | neg | - |
| 16 | Contrast CT | - | neg | neg | - |
Postoperative complications were observed in 43% of the cases (7/16), 42.8% (3/7 patients) of which were a CD 1-2 (wound infection, atrial fibrillation, slow intestinal transit needing parenteral nutrition), 28.6% (2/7 patients) were a CD 3b (2 patients that needed re-intervention) and 28.6% (2/7 patients) CD 5. The mortality rate was 12.5% (2/16). The summary of the operative and postoperative course of each case is shown in Table 2.
DISCUSSION
SBO is a frequent condition (15%-20% of admissions in the USA). In 30%-40% of the cases, OM is required to resolve the cause of obstruction[9,10]. Moreover, SBO accounts for 60% of recurrent bowel obstruction, contributing to increased readmission rates and the need for both operative and non-OM[10]. Despite the efforts made to clarify the pathophysiology of bowel obstructions, particularly of adhesive etiology, preoperative diagnosis remains difficult and management is associated with high healthcare costs.
OM can be performed via open or minimally invasive approaches. Considering that surgery itself induces adhesion formation (requiring new surgery in 10%-30% of cases), a minimally invasive approach can reduce the risk of adhesion formation[10,11]. However, current literature reports that the laparoscopic approach can be safe and effective only in selected patients: absence of peritonitis, absence of hemodynamic instability, first episode, absence of severe intestinal distension, low peritoneal adhesion index (score < 3 of abdominal quadrants), single band adhesion, previous appendectomy/cholecystectomy/gynecological surgery, pre-operative CT with imaging of identifiable transition point and an anticipated single obstructing band, CT images of completely collapsed distal small bowel loops[10,12]. Furthermore, the surgical team’s expertise in minimally invasive surgery and their proficiency in managing laparoscopic emergencies play a pivotal role[10,12,13]. Laparoscopic approach in emergency surgery has been increasingly adopted over the past twenty years; nevertheless, as reported by the World Society of Emergency Surgery in 2023, laparoscopy is employed in less than 20% of cases of major emergency situations[13]. Moreover, the above-mentioned scientific society stated that “the strongest deterrent to the use of Mini Invasive Surgery in emergency surgery was the patient’s poor physiological condition”[13]. Additional factors discouraging laparoscopy in emergency settings were the deterioration of clinical conditions, unclear anatomical visualization, bowel perforation and bleeding[13]. These issues are particularly critical in frail patients, who are debilitated by age, pre-existing comorbidities and the acute surgical disease[13].
In light of this, we aimed to explore the role of the laparoscopic approach in the emergency setting, particularly in elderly and frail patients. We combined this approach with low pneumoperitoneum and ICG fluorescence to optimize outcomes and avoid unnecessary bowel resection in acute SBO managed operatively.
OM in SBO is associated with increased hospitalization costs, as well as a higher risk of postoperative complications and mortality (3% for each admission for SBO). Furthermore, in cases of persistent bowel obstruction for more than 24 h, the risk of resection due to ischemia or necrosis doubles[14]. In light of this, the laparoscopic approach for SBO is increasingly adopted to reduce morbidity (pneumonia, wound infections), hospital stay, mortality, and to avoid unnecessary laparotomies[9,13,15,16]. Current international guidelines indicate that the laparoscopic approach is safe and feasible in experienced hands in this kind of setting, particularly in patients with SBO caused by a single adhesive band. The benefits of the laparoscopic approach include a shorter length of hospital stay, reduced postoperative pain, and lower SBO recurrence rates.
These advantages are particularly evident in elderly patients with comorbidities, for whom the minimally invasive approach clearly improves postoperative outcomes[11,17]. In our case series, the median age is 72 years old; the cohort only included one young patient with SBO due to suspected IBD (not confirmed in laparoscopy, suggesting a functional obstruction), and two other patients younger than 60. Most of the patients were older and had a median ASA score higher than 3. Younger patients were also included, as this is a preliminary prospective study aiming to evaluate the role of ICG fluorescence in a comprehensive cohort of all patients with SBO undergoing OM. It has been extensively demonstrated that, in this kind of patient, surgical distress can represent an important burden in terms of morbidity and mortality[18].
As reported in the literature, given the rapid aging of the population worldwide, SBO admissions to the emergency rooms involve not only elderly, but also frail patients. The advantages of laparoscopic surgery are well established, and these patients could benefit significantly from such an approach, considering its association with faster recovery, reduced post-operative pain and lower morbidity[19,20].
The Frailty and Emergency Surgery in the Elderly (FRAILESEL) study, an Italian multicenter prospective cohort study comparing open and laparoscopic approaches in emergency abdominal surgery for patients aged > 65 years, demonstrated that morbidity and mortality rates were statistically lower with laparoscopy (36.2% open vs. 22.1% laparoscopy, P < 0.001, and 11.2% open vs. 2.2% laparoscopy, P < 0.001, respectively)[11].
The implementation of ICG fluorescence could represent a potentially advantageous evolution of this strategy. In fact, the combination of minimally invasive surgery and ICG fluorescence in frail and elderly patients may concretely optimize surgical management and maximize outcomes, while also reducing costs, complications, and mortality.
In both simple and complex intestinal obstructions, distinguishing between ischemic and viable segments remains difficult. ICG fluorescence can provide a clear perfusion map, guiding the surgeon in deciding whether to resect or preserve the affected segment. This approach may reduce the risk of postoperative necrosis and its severe complications, such as anastomotic dehiscence, thereby improving postoperative outcomes, particularly in this high-risk population.
Extensive expertise acquired through years of elective minimally invasive surgery allowed us to implement this approach to SBO cases requiring emergency surgical management. In our clinical practice, even the most complex and frail SBO patients are initially approached laparoscopically. We have achieved excellent intraoperative and postoperative outcomes by utilizing a low-pressure pneumoperitoneum (< 10 mmHg): this technique improves tolerance of the procedure, as supported by current literature[21]. Building on this evidence, pneumoperitoneum was induced via an open access technique to avoid intestinal injury, with intra-abdominal pressure maintained between 8 and 10 mmHg. It is well established that prolonged exposure to high intra-abdominal pressure can adversely affect cardiovascular and pulmonary physiology; consequently, reducing pressure may offer clinical benefits, although robust evidence regarding the efficacy of this measure is still lacking[21]. Furthermore, in frail patients undergoing emergency surgery, the hemodynamic reserve is often compromised compared to elective surgery, even in the absence of evident hemodynamic instability. Therefore, low-pressure pneumoperitoneum facilitates anesthesiologic management during surgery and improves postoperative pain control[21]. In our series, the laparoscopic approach was maintained only if intra-abdominal conditions were favorable; otherwise, conversion to open surgery was performed. Our conversion rate was 37%, falling within the reported literature range of 10% to 50%[19]: given the limited sample size and the predominantly elderly population, this rate can be considered in line with the literature. The observed mortality rate (12.5%) aligns with published data[11,22]. The postoperative complications rate (43%), although higher than that reported in other studies[23], is comparable to the results of the FRAILESEL study (32%)[11]; this finding may be explained by the advanced age of most of the included patients.
We implemented ICG-enhanced fluorescence in these settings as a supportive tool to differentiate macroscopically irreversible from reversible bowel ischemia, thereby avoiding unnecessary intestinal resection. We strongly believe that this tool could represent a game-changer in improving postoperative outcomes for frail patients.
Bowel resection is required in cases of intestinal perforation, necrosis or irreversible ischemia. It has been demonstrated that clinical evaluation alone leads in around 46% of the cases to unnecessary resection[8]. The integration of ICG-enhanced fluorescence with laparoscopic surgery represents a valuable tool to ameliorate and refine diagnosis, thereby guiding both experienced and junior surgeons in the decision-making process regarding intestinal resection. Nakashima et al. utilized ICG in SBO cases to objectively assess intestinal perfusion of ischemic sections, proposing a classification based on fluorescence patterns to evaluate the extent of ischemia[7]. Applying the same criteria to our series, we performed a resection of intestinal segments presenting as non-fluorescent or showing perivascular fluorescence. Only in one case (Case #7 on Table 2) a fine granular pattern was identified [Figures 2 and 3], leading to a decision to preserve the bowel segment; this conservative approach was preferred due to the patient’s history of Parkinson’s disease; however, the patient required a re-intervention 2 days later, and a resection of the necrotic intestinal segment (11% of the cases) was necessary. The adoption of ICG in acute SBO in our practice is recent; therefore, as reported in the literature, a lack of solid experience may lead to pattern misinterpretation[3]. In addition, the patient’s comorbidities influenced the initial decision toward a conservative approach, unfortunately resulting in a delayed resection. Similar findings have been reported in other studies, with re-intervention rates ranging between 16% and 29% in patients initially managed conservatively after ICG assessment[24]. As suggested by other authors, the discordance between ICG pattern and actual perfusion (documented by histological or endoscopic findings) is likely due to the fact that intraoperative ICG primarily assesses serosal perfusion, thereby underestimating the true extent of mucosal ischemia[25]. In our cases, we delayed ICG injection for ten minutes after relieving the intestinal obstruction; this interval was observed to allow the resolution of vasospasm, to ensure a more adequate perfusion assessment, and to help restore systemic hemodynamic stability. Furthermore, other authors have investigated the implementation of ICG fluorescence in laparoscopic surgery as an alternative to direct manual palpation in open surgery, concluding that it can represent a useful surrogate for tactile feedback, ultimately improving surgical outcomes[26].
To date, definitive guidelines or standardized protocols for the implementation of ICG fluorescence in emergency SBO surgery are lacking, and studies on this topic remain scarce. As detailed in the methods section of this study, our approach aligns with established literature regarding ICG dosage, comparative assessment of ischemic vs. viable intestinal segments and pattern classification[27]. In our view, our results demonstrate a correlation between intraoperative fluorescence findings and patient outcomes, particularly considering that only one patient in the non-resection group required re-intervention.
We strongly believe that the combination of ICG fluorescence, low-pressure pneumoperitoneum, and minimally invasive surgery in emergency settings, especially in elderly and frail patients, can significantly reduce morbidity and mortality rates. Furthermore, the advantage of this ICG-guided protocol in SBO is that it can be employed in both laparoscopic and open surgical approaches. This tool is cost-effective, readily available, easy to apply, safe, and is already widely adopted in elective and routine surgery. It can be easily extended to emergency surgery, thereby making a pivotal difference, particularly for frail patients. Regarding the utilization of ICG fluorescence, the ARtificial Intelligence in Emergency and trauma Surgery (ARIES)-World Society of Emergency Surgery (WSES) project investigated the current knowledge and practices among surgeons: according to survey responses, approximately 45% of the respondents (87 of 192) have access to a fluorescence imaging system for both elective and emergency procedures; however, less than 1% affirmed that the ICG application influenced their intraoperative decision-making[28]. This finding is likely explained by the lack of high-level evidence in this specific field and the urgent need for standardized protocols in emergency settings.
This study is subject to several limitations. First, it is a single-center, prospective, non-randomized study with a limited sample size. The decision to employ ICG fluorescence was based on intraoperative findings to guide the surgical decision-making process regarding the need for intestinal resection. Furthermore, the patient population was heterogeneous in terms of age, including not only elderly patients. Finally, the study includes prospectively collected data from both ICG and non-ICG managed cases.
In conclusion, SBO is highly prevalent in elderly patients, particularly affecting frail and comorbid individuals. ICG fluorescence, low-pressure pneumoperitoneum, and minimally invasive surgery in acute abdominal settings and in emergency surgery represent a safe and valuable strategy to facilitate the surgical assessment in borderline cases of intestinal ischemia, thereby preventing unnecessary resections and improving the postoperative patient outcomes. Future randomized trials are essential to establish a standardized protocol for ICG employment in emergency surgery, especially in cases of bowel ischemia. A prospective multicentric study targeting geriatric patients would help clarify the role of ICG fluorescence in this specific population.
DECLARATIONS
Authors’ contributions
Made substantial contributions to the conception and design of the study: Agresta F
Wrote the main manuscript text: Montori G, Serrano E
Performed data analysis: Serrano E
Revised the tables and figures: Butera V
Revised the English language: Tolot M
Revised and approved the manuscript: Procida G, Dal Borgo A, Licitra EG, Mazzarolo G
Availability of data and materials
The data that support the findings of this study are securely stored in a dedicated repository at ULSS2 “Marca Trevigiana”, Italy. They are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
None.
Conflicts of interest
Agresta F is a Section Editor of the journal Mini-invasive Surgery. He was not involved in any aspect of the editorial process for this manuscript, including reviewer selection, manuscript handling, or decision-making. All other authors declare no conflicts of interest.
Ethical approval and consent to participate
This study was conducted in accordance with the local regulations of Vittorio Veneto Hospital. As a retrospective analysis of data collected during routine clinical care without any additional interventions, formal ethics committee approval was not required. All patients had previously provided written informed consent for the clinical procedures and for the use of indocyanine green.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Boni L, Macina S, David G, Cassinotti E, Fingerhut A. ICG-enhanced fluorescence-guided laparoscopic surgery. 2nd ed. Tuttlingen (DE): EndoPress; 2019. Available from: https://air.unimi.it/retrieve/dfa8b9a3-43cc-748b-e053-3a05fe0a3a96/ICG-Enhanced%20Fluorescence-Guided%20Laparoscopic%20Surgery%20%E2%80%93%202nd%20edition.pdf. [Last accessed on 12 Mar 2026].
2. Kalayarasan R, Chandrasekar M, Sai Krishna P, Shanmugam D. Indocyanine green fluorescence in gastrointestinal surgery: appraisal of current evidence. World J Gastrointest Surg. 2023;15:2693-708.
3. Perini D, Martellucci J. Don’t forget emergency surgery! Lessons to learn from elective indocyanine green-guided gastrointestinal interventions. World J Gastrointest Surg. 2024;16:270-5.
4. Fransvea P, Fico V, Puccioni C, et al. Application of fluorescence-guided surgery in the acute care setting: a systematic literature review. Langenbecks Arch Surg. 2023;408:375.
5. Furusawa K, Yoshimitsu M, Matsukawa H, Oi K, Yunoki K, Tamura A. Precise diagnosis of acute mesenteric ischemia using indocyanine green imaging prevents small bowel resection: a case report. Int J Surg Case Rep. 2022;97:107463.
6. Аlexander K, Ismail M, Alexander M, et al. Use of ICG imaging to confirm bowel viability after upper mesenteric stenting in patient with acute mesenteric ischemia: case report. Int J Surg Case Rep. 2019;61:322-6.
7. Nakashima K, Ryu S, Okamoto A, et al. Usefulness of blood flow evaluation with indocyanine green fluorescence imaging during laparoscopic surgery for strangulated bowel obstruction: a cohort study. Asian J Surg. 2022;45:867-73.
8. Bulkley GB, Zuidema GD, Hamilton SR, O’Mara CS, Klacsmann PG, Horn SD. Intraoperative determination of small intestinal viability following ischemic injury: a prospective, controlled trial of two adjuvant methods (Doppler and fluorescein) compared with standard clinical judgment. Ann Surg. 1981;193:628-37.
9. Coco D, Leanza S, Fiume I. Small bowel obstruction: a prognostic score index for surgery - a review. Prz Gastroenterol. 2022;17:177-82.
10. Ghimire P, Maharjan S. Adhesive small bowel obstruction: a review. JNMA J Nepal Med Assoc. 2023;61:390-6.
11. Costa G, Fransvea P, Podda M, et al. The use of emergency laparoscopy for acute abdomen in the elderly: the FRAILESEL Italian Multicenter Prospective Cohort Study. Updat Surg. 2020;72:513-25.
12. Podda M, Khan M, Di Saverio S. Adhesive small bowel obstruction and the six w’s: who, how, why, when, what, and where to diagnose and operate? Scand J Surg. 2021;110:159-69.
13. Sermonesi G, Tian BWCA, Vallicelli C, et al. Cesena guidelines: WSES consensus statement on laparoscopic-first approach to general surgery emergencies and abdominal trauma. World J Emerg Surg. 2023;18:57.
14. Karampinis I, Keese M, Jakob J, et al. Indocyanine green tissue angiography can reduce extended bowel resections in acute mesenteric ischemia. J Gastrointest Surg. 2018;22:2117-24.
15. Suh SW, Choi YS. Laparoscopy for small bowel obstruction caused by single adhesive band. JSLS. 2016;20:e2016.00048.
16. Vettoretto N, Carrara A, Corradi A, et al.; Italian Association of Hospital Surgeons (Associazione dei Chirurghi Ospedalieri Italiani-ACOI). Laparoscopic adhesiolysis: consensus conference guidelines. Colorectal Dis. 2012;14:e208-15.
17. Ten Broek RPG, Krielen P, Di Saverio S, et al. Bologna guidelines for diagnosis and management of adhesive small bowel obstruction (ASBO): 2017 update of the evidence-based guidelines from the world society of emergency surgery ASBO working group. World J Emerg Surg. 2018;13:24.
18. Shinall MC Jr, Arya S, Youk A, et al. Association of preoperative patient frailty and operative stress with postoperative mortality. JAMA Surg. 2020;155:e194620.
19. Agresta F, Podda M, Campanile FC, Bergamini C, Anania G. Emergency laparoscopic surgery in the elderly and frail patient. Cham (CH): Springer Nature; 2021.
20. Jeppesen M, Tolstrup MB, Gögenur I. Chronic pain, quality of life, and functional impairment after surgery due to small bowel obstruction. World J Surg. 2016;40:2091-7.
21. Ortenzi M, Montori G, Sartori A, et al. Low-pressure versus standard-pressure pneumoperitoneum in laparoscopic cholecystectomy: a systematic review and meta-analysis of randomized controlled trials. Surg Endosc. 2022;36:7092-113.
22. Rami Reddy SR, Cappell MS. A systematic review of the clinical presentation, diagnosis, and treatment of small bowel obstruction. Curr Gastroenterol Rep. 2017;19:28.
23. Tolstrup MB, Watt SK, Gögenur I. Morbidity and mortality rates after emergency abdominal surgery: an analysis of 4346 patients scheduled for emergency laparotomy or laparoscopy. Langenbecks Arch Surg. 2017;402:615-23.
24. Christofi A, Traska T, Dimitroulis D. Near-infrared indocyanine green angiography in recognizing bowel ischemia in emergency surgery: game changer or overrated? Innov Surg Sci. 2024;9:113-21.
25. Seeliger B, Agnus V, Mascagni P, et al. Simultaneous computer-assisted assessment of mucosal and serosal perfusion in a model of segmental colonic ischemia. Surg Endosc. 2020;34:4818-27.
26. Ryu S, Hara K, Goto K, et al. Fluorescence angiography vs. direct palpation for bowel viability evaluation with strangulated bowel obstruction. Langenbecks Arch Surg. 2022;407:797-803.
27. Guerra F, Coletta D, Greco PA, Eugeni E, Patriti A. The use of indocyanine green fluorescence to define bowel microcirculation during laparoscopic surgery for acute small bowel obstruction. Colorectal Dis. 2021;23:2189-94.
28. De Simone B, Abu-Zidan FM, Saeidi S, et al.; ICG Fluorescence Guided Emergency Surgery Survey Consortium. Knowledge, attitudes and practices of using indocyanine green (ICG) fluorescence in emergency surgery: an international web-based survey in the ARtificial Intelligence in Emergency and trauma Surgery (ARIES)-WSES project. Updates Surg. 2024;76:1969-81.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].