Research on the learning curve and simulation of proximal anastomosis in minimally invasive coronary artery bypass grafting
Abstract
Aim: This study examines the learning curve for vein-to-aorta anastomosis in minimally invasive coronary artery bypass grafting (MICS CABG) and develops a virtual reality (VR) simulation model for this procedure, with validation to confirm its clinical relevance.
Methods: We analyzed 132 consecutive off-pump MICS CABG procedures with multi-vessel grafting performed by a single surgeon (January 2017-January 2020). Proximal anastomosis time was plotted against case sequence, and the learning curve was quantified using cumulative summation analysis. A VR simulation platform was developed to reproduce key clinical challenges, using hierarchical geometric modeling and real-time physics simulation of tissue-instrument interactions with haptic feedback. Cardiac surgery experts evaluated the VR simulator via a structured questionnaire.
Results: The learning curve for proximal anastomosis followed a three-phase progression, with proficiency achieved after 52 cases (model fit R2 (the coefficient of determination) = 0.994). The VR simulation accurately replicated key surgical scenarios, focusing on bimanual suturing mechanics and needle-vessel interactions. Expert validation yielded an overall validity index of 0.78 (exceeding the 0.7 threshold), confirming good construct validity and clinical relevance of the simulator.
Conclusion: Proficiency in MICS CABG proximal anastomosis is achieved after 52 cases, marking the inflection point of the learning curve. Our physics-validated VR simulator, supported by expert evaluation, has potential to accelerate surgical skill acquisition and shorten the learning curve.
Keywords
INTRODUCTION
Minimally invasive coronary artery bypass grafting (MICS CABG) is a significant option for treating coronary artery disease, offering minimal invasiveness and complete revascularization, along with reduced trauma, less intraoperative bleeding, and faster recovery[1-6]. However, compared with conventional sternotomy, the intercostal incision in MICS CABG involves switching surgical fields, and exposure for vein-to-aorta (proximal) anastomosis—particularly challenging—remains a critical technical difficulty and a key determinant of successful multi-vessel grafting[7-12]. This study analyzed data from 132 consecutive multi-vessel MICS CABG procedures performed by a single surgeon at our institution from January 2017 to January 2020. Using the cumulative summation (CUSUM) method, we quantified the learning curve for proximal anastomosis. Additionally, we developed a virtual reality (VR)-based simulation training system for proximal anastomosis, utilizing intraoperative imaging and measurement data from real surgeries, to shorten the learning curve.
METHODS
Patient population
This retrospective cohort study enrolled 132 patients undergoing MICS CABG at Beijing Anzhen Hospital from January 2017 to January 2020. Inclusion criteria required angiographically confirmed multi-vessel coronary artery disease (≥ 2 affected vessels) meeting indications for off-pump CABG (OPCABG). Exclusion criteria comprised: (1) need for concomitant cardiac procedures (valvular surgery, ventricular aneurysm repair, or aortic surgery); (2) preoperative chest CT examinations showing any degree of aortic calcification (mild, moderate, or severe); (3) acute myocardial infarction or heart failure; or (4) redo cardiac surgery. Following a thorough surgical consultation, all participants provided written informed consent after an extensive discussion of the available therapeutic options. The institutional ethics committee of Beijing Anzhen Hospital granted ethical approval for this study, including explicit authorization for the publication of research outcomes.
Clinical data collection
To evaluate the learning curve of proximal anastomosis in MICS CABG, operation times were meticulously recorded. The proximal anastomosis time was defined as the duration from aortic exposure to the completion of both the proximal anastomosis and hemostasis. The start time begins with aortic exposure for the proximal anastomosis, while the end time is marked by completion of hemostasis for the vein-to-aorta anastomosis.
Additionally, clinical characteristics and perioperative details, including demographics, clinical outcomes, and complications, were extracted from patients' medical records. Based on the inflection point of the learning curve, patients were classified into two groups: the initial learning group (Initial Group) and the proficient phase group (Proficient Group). Perioperative outcomes were subsequently analyzed. The pre-operative and pre-discharge Barthel Index, an ordinal scale, was used to measure the performance of daily living activities[13].
Surgical approach for proximal anastomosis
Using the THORATRAK MICS (minimally invasive cardiac surgery) Retractor System through the 5th left intercostal space (ICS), we exposed the operative field by incising the pericardium down to the ascending aortic reflection. The pericardium was cut 2-3 cm below the left phrenic nerve to establish a connection between the pericardial and pleural spaces. To enhance exposure, we placed three pericardial suspension sutures on the right ascending aorta, which were exteriorized via the 2nd ICS, along with one suture on the left side.
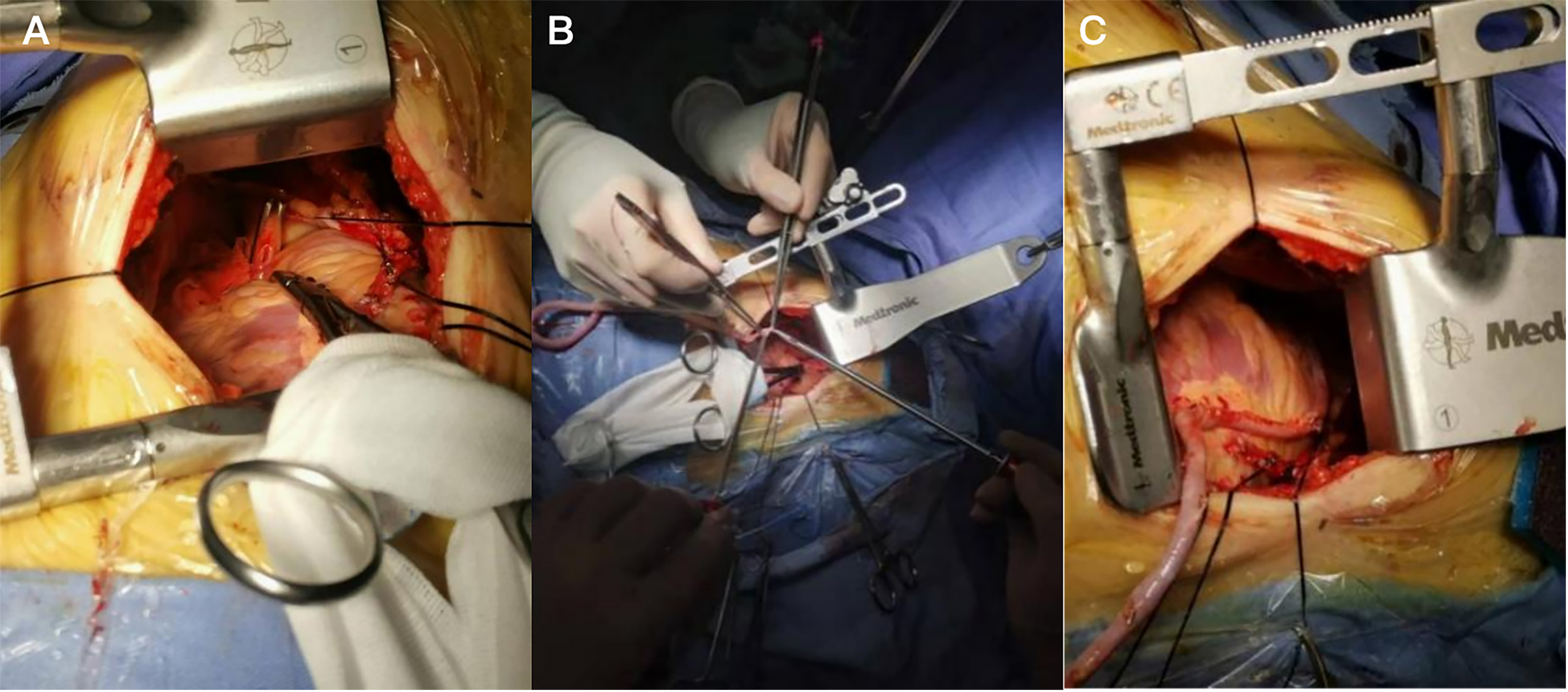
After dissecting the aortopulmonary septum, a specialized side-biting clamp (Cardiomedical GmbH) partially occluded the ascending aorta at 100-120 mmHg [Figure 1A]. A 3-min test clamp was applied, and stability (central venous pressure, hemodynamics, electrocardiogram) was assessed during this period. The anastomosis was then completed using ValveGate™ PRO instruments [Figure 1B], followed by clamp release with careful hemostasis and de-airing [Figure 1C].
Statistical analysis
Statistical analyses were conducted using SPSS 25.0. Categorical variables are presented as frequencies or percentages, while continuous variables with a normal distribution are expressed as mean ± standard deviation (χ ± s). Independent two-sample t-tests were applied to continuous variables, and chi-square tests were used for categorical variables.
A P-value < 0.05 was considered statistically significant. For CUSUM analysis of learning curve, patients were sequentially ordered by surgical case number (x-axis), with proximal anastomosis time (min) as the y-axis to generate a scatter plot of operative time versus case sequence. The CUSUM was calculated as: CUSUM (1) = Proximal anastomosis time (Case 1) - Mean proximal anastomosis time; CUSUM (n) = Proximal anastomosis time (Case n) - Mean proximal anastomosis time + CUSUM (n - 1). The mean operative time for proximal anastomosis, used in the CUSUM calculation, was obtained from a cohort of 132 cases included in this study. This iterative process continued until the final case resulted in a CUSUM of 0. The CUSUM-case sequence scatter plot was analyzed using SPSS 25.0 for regression-curve fitting (P < 0.05 for successful fitting). Goodness-of-fit was evaluated using R2, where values closer to 1 indicate a better fit, leading to the derivation of the optimal regression equation. The inflection point of the CUSUM curve was identified to demarcate the learning phase[7].
VR modeling and simulation
This study developed a VR surgical simulation system specifically for proximal anastomosis in MICS CABG. The design of the VR simulator was informed by real-world clinical learning curve data, with a focus on replicating the technical difficulties that can extend operative time during both the early learning phase—such as bimanual coordination during deep anastomosis and precise control of suturing tension—and the plateau phase, which involves managing complex vessel anatomies. The system processes medical images and surgical videos to extract essential elements and parameters from 30 MICS CABG cases, facilitating geometric and physical modeling of organs and instruments for a parameter-driven VR simulation.
Preoperative CT scans were segmented using the medical image processing software ITK-SNAP[14] to reconstruct triangular models depicting the overall morphology of the heart, aorta, and coronary tree. These models were geometrically refined to enhance realism. To simulate cardiac pulsation and the associated vascular motion, a spatiotemporal dataset of anatomical landmarks was created using MRI sequences that encompassed the entire cardiac cycle[15]. A control mesh encompassing the entire heart was established using the end-diastolic phase morphology as the reference configuration[16]. Displacement vector fields between adjacent frames were computed and mapped onto the vertices of the control mesh, giving each vertex a temporal displacement parameter set that describes local motion patterns. Geometric models were associated with their nearest control mesh vertices via radial basis function, enabling the geometric model to undergo physiologically accurate periodic motion. The hierarchical modeling results, including motion constraints, were imported into the Unity3D VR engine to edit the surgical simulation scene.
For physical modeling and simulation, the Cosserat rod model[17] was employed to characterize the physical properties of both vessels and sutures. Within Unity3D's inherent dynamic simulation framework, these physical properties were translated into geometric constraints. Real-time deformation was solved using the eXtended Position-Based Dynamics (XPBD) algorithm[18] to achieve dynamic responses to interactive inputs. The system integrates Geomagic Touch, a 6-DOF (6 degrees of freedom) force feedback device, to capture real-time user surgical maneuvers and translate them into the virtual environment. The interaction outcomes with the simulated objects are relayed to the user through visual-haptic fusion, creating a high-fidelity surgical simulation.
VR validation
To validate the effectiveness and reliability of the VR system for MICS CABG, we designed and implemented a comprehensive evaluation framework. This framework focused on assessing both the surface validity and content validity of the XPBD-based soft-tissue physics simulation and the force-haptic feedback system.
We invited ten cardiac surgery experts from our institution to evaluate the simulation system by completing a questionnaire based on relevant literature[19]. The panel consisted of three attending surgeons specialized in MICS CABG, three attending doctors in OPCABG, and four residents who had completed CABG training. Their assessment addressed several key dimensions: ease of use, realism, effectiveness, learning curve shortening, and future potential. A five-point Likert scale (1 = strongly disagree, 5 = strongly agree) was used to score each dimension.
RESULTS
Learning curve results
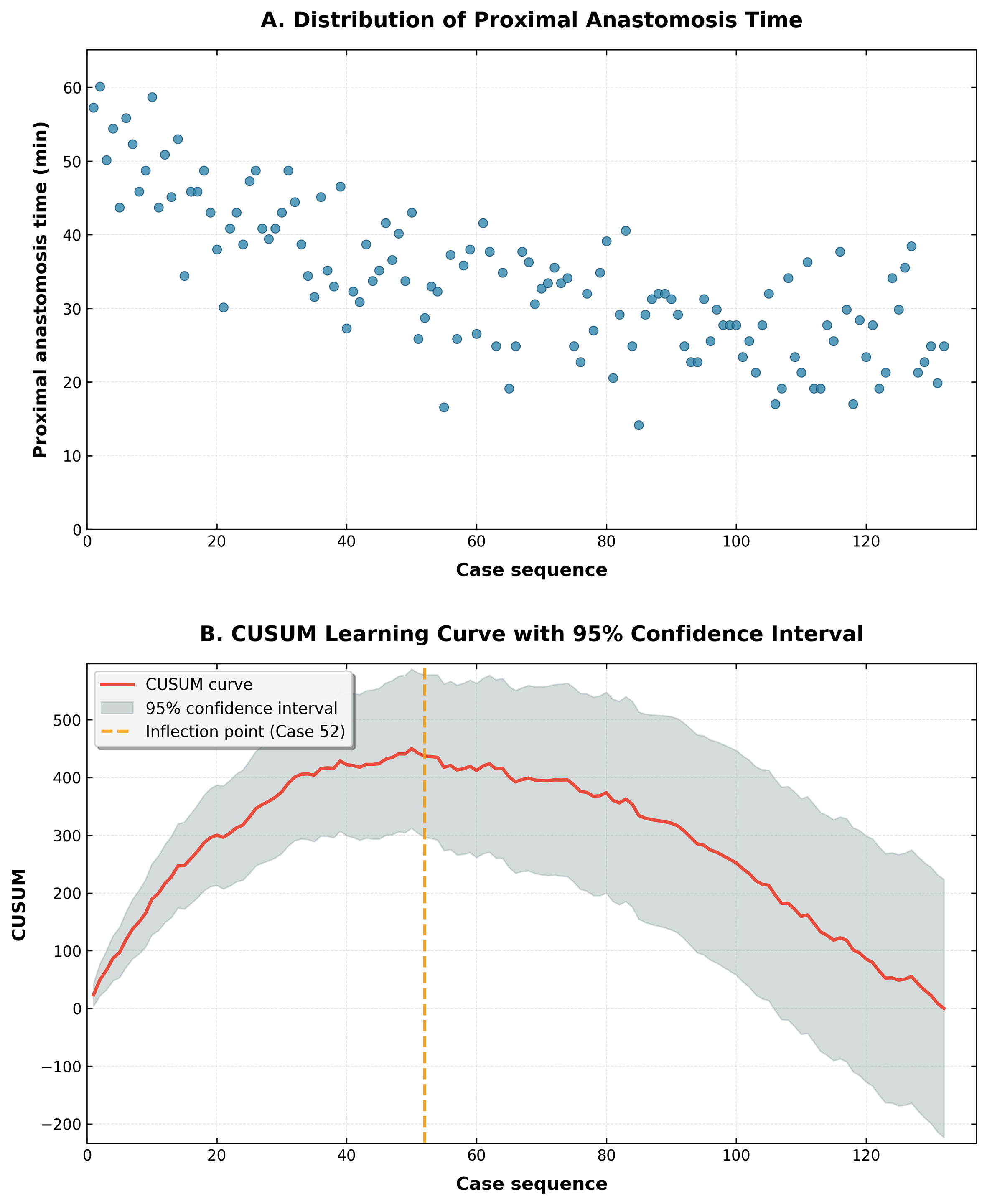
Analysis of 132 cases revealed a characteristic learning curve for proximal anastomosis when plotting operative duration against case sequence [Figure 2A]. Surgical time showed a progressive decrease before stabilizing as experience accumulated. CUSUM quantification demonstrated that this learning pattern followed a cubic function [Figure 2B], exhibiting an excellent model fit (P = 0.000, R2 = 0.994), given by
Figure 2. Learning curve of proximal anastomosis in MICS CABG. (A) Scatter plot of proximal operative time versus case sequence; (B) CUSUM learning curve with 95% confidence interval (inflection point at n = 52). CUSUM: Cumulative summation.
Comparative analysis indicated no significant differences in preoperative characteristics between the pre- and post-inflection groups [Table 1]. However, perioperative metrics [Table 2] showed that the pre-inflection group required longer total operative times (mean difference 42.45 ± 8.15 min, P < 0.01) and longer proximal anastomosis durations (28.33 ± 6.44 min, P < 0.05). Additionally, this group had a higher mean number of grafts (3.39 ± 0.60 vs. 3.07 ± 0.77, P < 0.05) and longer ventilator support (18.40 ± 13.49 vs. 14.49 ± 4.59 h, P < 0.05). Despite these differences, both groups maintained equivalent 30-day outcomes, with zero mortality, myocardial infarction, or cerebrovascular events, confirming the procedure's safety throughout the learning period.
Baseline demographic, clinical, and Echocardiography data of the Initial and Proficient groups based on the inflection point of learning curve [χ ± s, N (%)]
| Characteristics | Initial group | Proficient group | P value |
| Age (years) | 59.57 ± 9.26 | 59.77 ± 9.34 | 0.906 |
| Male, n (%) | 46 (90.2) | 73 (90.1) | 0.989 |
| BMI | 24.90 ± 2.87 | 25.44 ± 2.69 | 0.277 |
| Smoking history, n (%) | 25 (49.0) | 45 (55.6) | 0.509 |
| Hypertension, n (%) | 31 (60.8) | 48 (59.3) | 0.862 |
| Diabetes, n (%) | 22 (43.1) | 35 (43.2) | 0.993 |
| Hyperlipidemia, n (%) | 3 (5.9) | 11 (13.6) | 0.162 |
| Cerebrovascular disease, n (%) | 7 (13.7) | 14 (17.3) | 0.586 |
| History of myocardial infarction, n (%) | 8 (15.7) | 10 (12.3) | 0.586 |
| History of PCI, n (%) | 3 (5.9) | 16 (19.8) | 0.050 |
| LVESD (cm) | 32.49 ± 4.56 | 31.94 ± 3.78 | 0.453 |
| LVEDD (cm) | 49.24 ± 4.00 | 48.31 ± 4.01 | 0.198 |
| EF % | 61.76 ± 5.72 | 61.67 ± 5.38 | 0.921 |
| Preoperative barthel index | 97.65 ± 4.73 | 96.98 ± 4.73 | 0.548 |
Clinical results of the Initial and Proficient groups based on the inflection point of learning curve [χ ± s, N (%)]
| Characteristics | Initial group | Proficient group | P value |
| Operation time (min) | 346.19 ± 57.07 | 253.46 ± 44.76 | 0.000 |
| Proximal anastomosis time (min) | 42.45 ± 8.15 | 28.33 ± 6.44 | 0.000 |
| Mean number of grafts | 3.39 ± 0.60 | 3.07 ± 0.77 | 0.014 |
| Ventilation duration (h) | 18.40 ± 13.49 | 14.49 ± 4.59 | 0.018 |
| ICU duration (h) | 21.26 ± 14.63 | 18.07 ± 5.12 | 0.075 |
| Length of hospital stay (day) | 13.76 ± 2.78 | 13.77 ± 3.15 | 0.999 |
| Predischarge barthel index | 61.86 ± 12.12 | 63.02 ± 9.28 | 0.432 |
| 30-day mortality, n (%) | 0 (0) | 0 (0) | - |
| Myocardial infarction, n (%) | 0 (0) | 0 (0) | - |
| Stroke, n (%) | 0 (0) | 0 (0) | - |
| Transfusion, n (%) | 2 (3.9) | 0 (0) | 0.073 |
VR modeling results
Proximal anastomosis virtual simulation
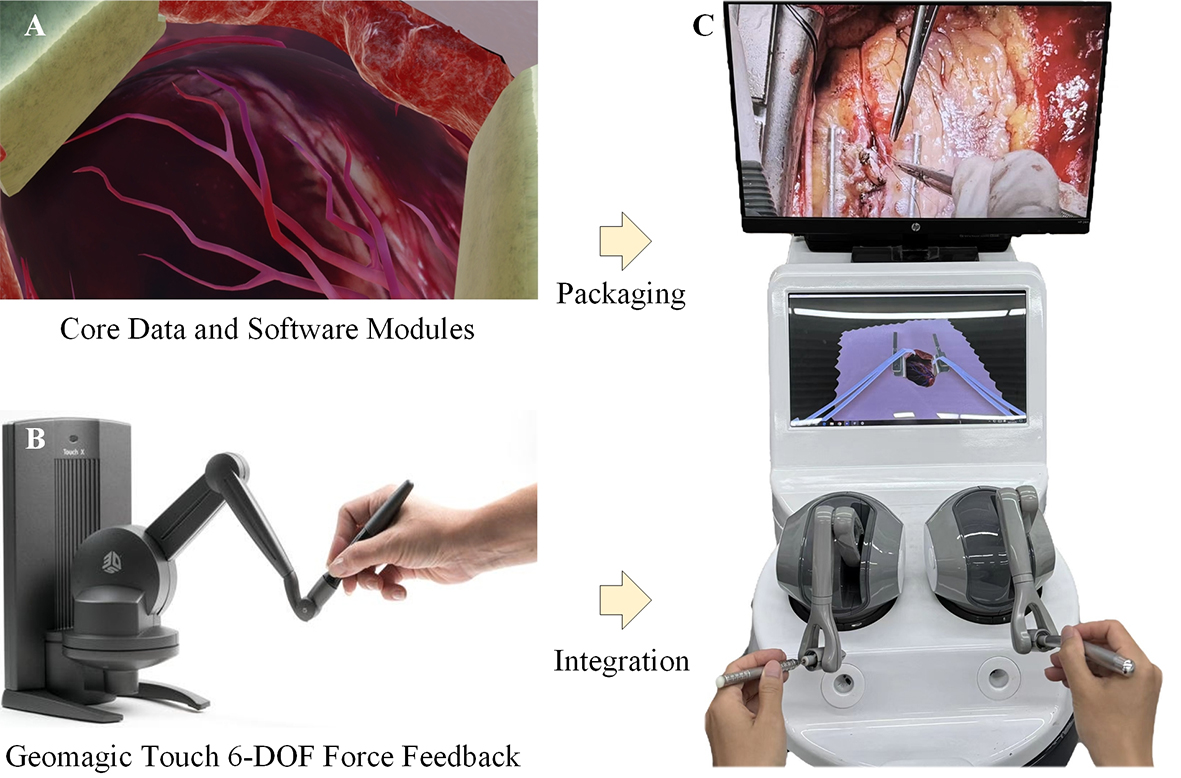
In this study, a prototype of the MICS simulator was developed by integrating hardware and software
Figure 3. The development of the MICS simulator prototype. (A) Illustration of core data and software modules; (B) Force feedback hardware (Geomagic Touch) with 6 degrees of freedom (6-DOF); (C) Simulator prototype.
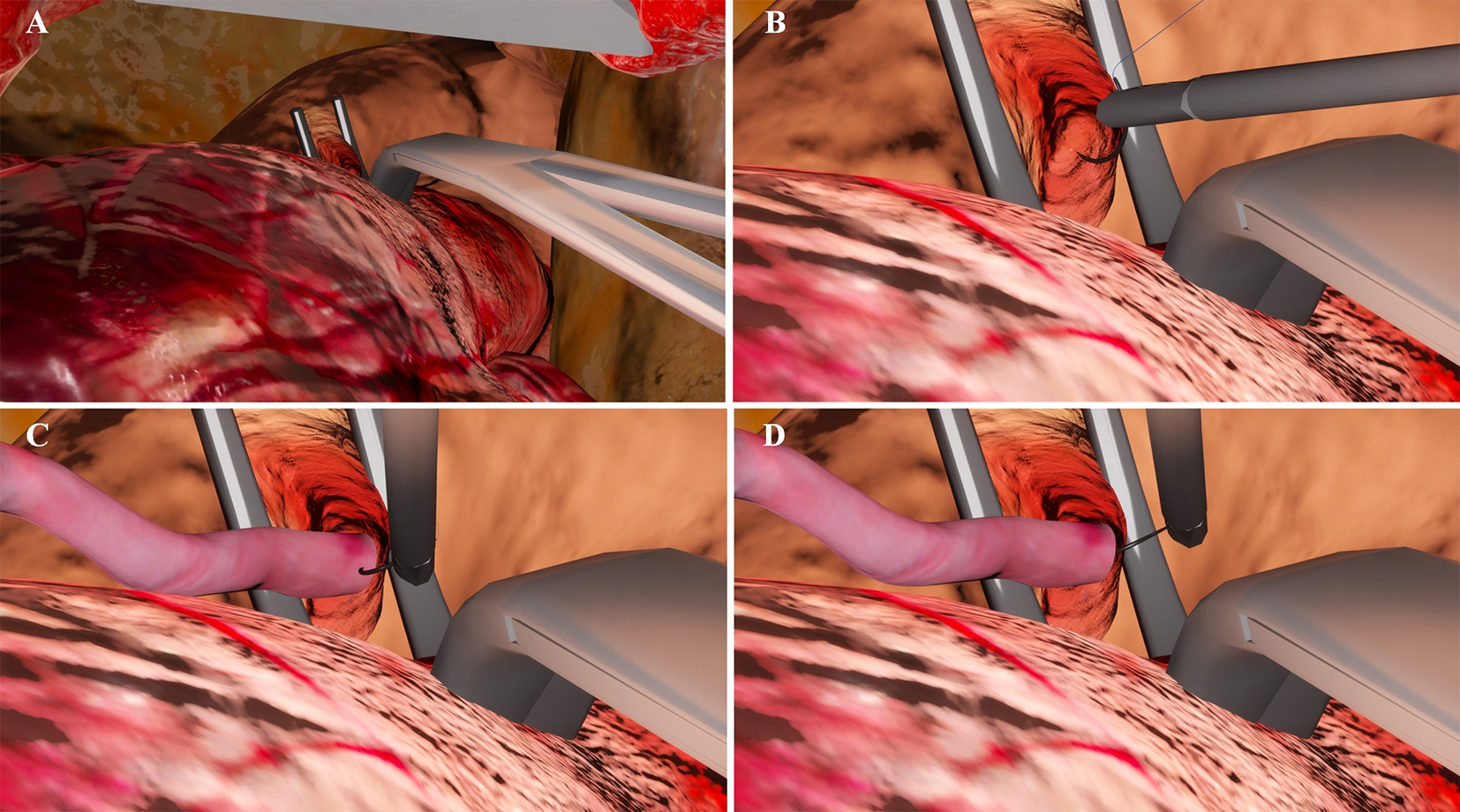
Considering the characteristics of proximal anastomosis surgery—specifically, its high precision requirements and the challenges in exposing the surgical field—this study creates a high-fidelity training environment by spatially positioning the virtual camera. The camera coordinates are meticulously aligned with the clinical operation origin, which is the chest wall incision between the second and third ICSs of the patient. This setup simulates the actual eye position of the surgeon during the procedure through a limited incision. The camera is angled at 15° to 30° downward towards the heart, accurately reproducing the surgeon's overhead perspective. This alignment ensures that the spatial relationships, size proportions, and anatomical occlusion effects of the ascending aorta exposure area, aortic clamping position, graft vessel path, and anastomosis operation area in the simulation are consistent with those encountered in real surgery. The four subgraphs in Figure 4 present the simulation results at different key moments of the proximal anastomosis operation.
Figure 4. Simulation results at different key moments of the proximal anastomosis. The simulation was implemented on Windows 11. Images were captured using the built-in screenshot utility of Windows and formatted in Microsoft Visual Studio. (A) Partial occlusion of the aorta; (B) The circular needle penetrates the aorta; (C) The circular needle penetrates the graft vessel; (D) Suturing the graft vessel to aorta.
The high-precision 3D model accurately renders the color and texture of human tissues, providing an immersive multi-sensory experience. The system simulates the key steps of proximal anastomosis, including the partial occlusion of the aorta by a side-biting clamp to create a bloodless anastomosis area, and the coordinated manipulation of the circular needle by both hands to penetrate the aortic intima and graft vessel. It displays vascular deformation under suture traction in real-time, revealing the interaction between the needle, suture, and tissue. The virtual instruments support dynamic adaptation: the needle holder can switch between long-handled and fine models as needed, and the size and type of the circular needle can be parameterized to match different surgical methods. Medical students can develop precise operation rhythm and blood vessel protection strength through repeated practice, enhancing spatial perception and suture tension control, significantly shortening the transition from theoretical learning to clinical operations.
VR validation
This study developed and implemented a preliminary multi-dimensional expert evaluation system. Beyond functional demonstration, the system integrates validity verification, reliability testing, and performance benchmarking. Its objective is to assess the system's core value from the perspective of clinical experts, confirming the virtual simulation module's high technical usability and realism, its role in shortening the learning curve, and its potential as a surgical skill assessment and training tool.
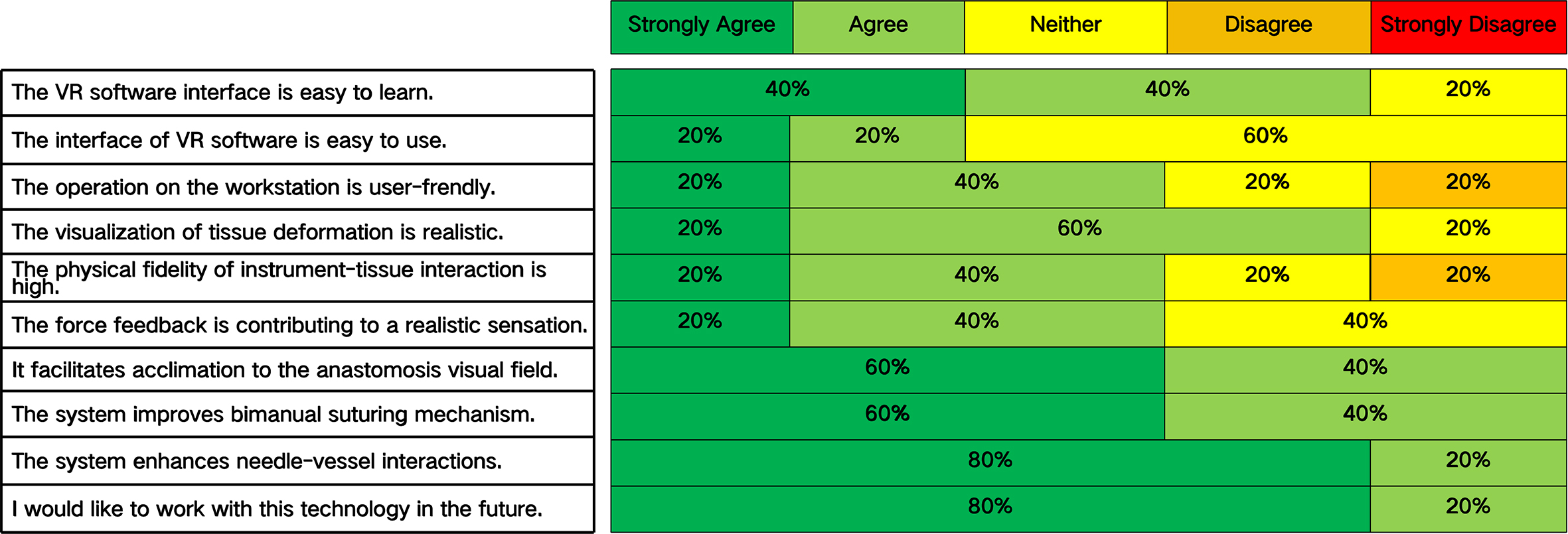
After integrating all data and functional modules, ten clinical surgeons (mean clinical experience: 12.5 ± 4.3 years) evaluated the system using structured questionnaires. As summarized in Figure 5, expert ratings revealed positive feedback across all dimensions, with detailed analyses as follows: (1) Usability: The average score was 0.6, indicating that despite incorporating complex engines and interactive logic, the system's user interface and workflow are user-friendly with a low learning curve. Experts noted its ease of use without lengthy pre-training—critical for clinical implementation. Minor suggestions on interface icon clarity provided direction for subsequent iterations; (2) Realism: Covering visual fidelity and operational feedback, this dimension achieved an average score of 0.67. Experts acknowledged the system's fidelity in organ geometry, tissue texture, and surgical scene rendering, effectively creating an immersive environment. Its simulation of standard surgical procedures aligns closely with clinical practice, demonstrating effectiveness beyond visual simulation. The system accurately reproduces tactile sensations and mechanical responses during operations, such as differentiated resistance feedback for various tissue layers during dissection. This distinguishes it from graphics-focused counterparts and facilitates skill transfer to real surgical scenarios; (3) Learning curve shortening and future potential: This dimension achieved the highest average score of 1. VR simulation replicates stage-specific clinical challenges in the learning curve (e.g., bimanual suturing mechanics, deep surgical obstacle coordination, needle-thread-vessel interaction angles). Such training reduces bleeding from uneven suturing and shortens surgical time. Experts expressed strong expectations for its application in standardized resident assessment, high-difficulty surgical rehearsal, rare case library development, and remote collaborative training—supporting clinical translation and commercialization.
Figure 5. Questionnaire results on the designed dimensions measuring virtual reality simulator for MICS CABG.
Based on expert ratings, the system's overall validity index was calculated as 0.78-exceeding the conventional 0.7 threshold. This confirms good construct validity, demonstrating that the designed dimensions effectively measure the core characteristics of a preliminary surgical simulation system.
DISCUSSION
The evolution of MICS CABG marks a significant advancement in cardiac surgery, effectively combining the principles of complete revascularization with minimally invasive approaches.
MICS CABG has gained widespread acceptance due to its advantages including reduced trauma, faster recovery, and unrestricted graft placement options[1-6], making it currently the most applicable minimally invasive approach. Compared to conventional sternotomy, MICS CABG uses an intercostal incision approach that requires surgeons to adapt to specialized instruments and develop personalized exposure techniques, particularly for proximal anastomosis[7-12]. These demands significantly elevate the surgeon's skill requirements, necessitating substantial case accumulation to master the techniques and achieve stable operative times and outcomes.
MICS CABG exhibits a distinct learning curve[7,20]. The CUSUM analysis used in this study, originally developed to quantify learning processes and identify proficiency milestones, has become an objective method in medical education for assessing minimum case requirements for clinical skill acquisition[21]. The learning curve from MIDCAB (Minimally Invasive Direct Coronary Artery Bypass) to MICS CABG, based on our team’s previous research, conforms to a cubic fitting formula, with inflection points at 27 cases for single-vessel and 59 cases for multi-vessel procedures[20]. This study targets proximal anastomosis, a critical technical link. The surgical learning curve is a multi-phasic process, characterized by initial rapid skill improvement, an intermediate refinement phase, and a final stabilization phase. The cubic regression model is identified as the most optimal after regression-curve fitting analysis[20]. Linear regression models can only describe unidirectional linear changes, while quadratic regression models cannot fully capture the three phases of proficiency development observed in our study. Operative time, the core indicator in learning curve research, can be influenced by confounding factors such as patient comorbidities and graft number. More grafts require additional anastomotic procedures, contributing to prolonged operative time in the early learning stage. Additionally, marginal differences in percutaneous coronary intervention (PCI) history may lead to altered vessel anatomy, increasing anastomotic difficulty. A subgroup analysis of patients with and without PCI history confirmed that the learning curve trajectory (inflection point at case 52) was consistent across both subgroups. Although this proximal anastomosis learning curve study may be influenced by comorbidities and operator-dependent factors, it provides objectively reproducible experience that offers practical clinical protocols and technical references to help other teams shorten their learning curves and reduce training costs.
The critical aspect of the MICS CABG learning curve lies in proximal anastomosis techniques, with the core challenge being optimal visual exposure[10]. Our experience shows that pericardial suspension with external traction, right-sided aortic compression using small gauze, and the strategic placement of specialized angled clamps can create sufficient working space while preserving the pulmonary artery "no-touch" technique. This approach eliminates the need for cardiac stabilizers that could compress the right ventricular outflow tract. Furthermore, conducting a trial clamp prior to aortotomy, without significantly reducing blood pressure, helps minimize the hemodynamic impact on the great vessels.
The advantages of minimally invasive surgery must never compromise safety. Advancements in VR, artificial intelligence (AI), and human-machine interfaces make surgical simulation crucial for shortening learning curves[22]. Our study created a highly realistic cardiovascular model and coronary artery bypass grafting (CABG) simulation scenario using medical image processing and geometric-physical modeling. This multimodal data-driven approach reconstructs the spatial relationships between anatomical structures and simulates tissue interactions during surgical maneuvers. As a result, it offers practical insights into instrument-tissue mechanics, which are essential for developing high-fidelity surgical simulation systems[23].
Leveraging Unity3D's strengths in realistic rendering and efficient simulation, we developed a proximal anastomosis training system that includes vessel manipulation and suturing techniques, significantly enhancing simulation fidelity. The integration of visual-haptic feedback, instrument control logic, and vascular/suture behavior modeling enables trainees to master essential surgical skills in an immersive environment.
Enhanced visual guidance enables repetitive precision practice to develop damage-avoidance capabilities[24]. The clinical value of the VR simulation is evident in two key areas: (1) In surgical training, its accurate instrument feedback and tailored pathological scenarios transform traditional animal labs and observational learning into quantifiable, reproducible skill-building loops. This approach accelerates the development of operative and spatial decision-making abilities; (2) In surgical planning, the system facilitates patient-specific model reconstruction from computed tomography (CT) data. This allows for preoperative virtual testing of various approaches for target vessel exposure, evaluation of clamp forces to prevent intimal injury, and optimization of suture paths to reduce anastomotic stress concentration. As a result, risk mitigation is shifted to the planning stage, establishing a new paradigm of precise rehearsal followed by safe implementation.
Proximal anastomosis poses the primary technical challenge in MICS CABG, often leading to increased operative duration and blood loss. To achieve consistent perioperative outcomes, strict quality control measures are essential. Our VR modeling incorporates key parameters extracted from preoperative imaging and intraoperative data collection. The geometrically and physically accurate modeling of organs and instruments provides clinically relevant anatomical representations and dynamic interactions, enhancing the understanding of anastomotic pathway planning and instrument-tissue mechanics. By seamlessly integrating visual-haptic feedback with instrument control logic and vascular/suture behavior simulation, the system enables immersive training in core surgical skills.
Through reinforced visual guidance, trainees can repeatedly practice suturing precision and develop critical damage-avoidance abilities[25], establishing an effective approach to shorten the surgical learning curve with significant clinical application value.
This study validated the core value of the VR simulation system for MICS CABG by assessing its usability, realism, and ability to shorten the learning curve. Expert evaluations revealed a significant connection between the outcomes of VR simulation and the clinical learning curve.
The essence of the clinical learning curve lies in surgeons gradually improving operational proficiency, decision-making accuracy, and reducing complications through repeated practice[7]. A key challenge for novice surgeons is the dilemma of "low fault tolerance" combined with "high pressure" in real surgical scenarios, which complicates their ability to safely navigate skill bottlenecks. The VR simulation system developed in this study accurately replicates critical clinical challenges in MICS CABG, including bimanual suturing mechanics, coordination around deep surgical obstacles, and control of the needle-thread-vessel interaction angle. This system offers a solution for the "controllable crossing" of the learning curve and establishes a coherent narrative chain of "simulation skill acquisition—clinical ability transfer—learning curve shortening."
The core value of VR simulation in direct clinical practice guidance lies in the transferable skill training brought by its "situational reproduction" and "feedback accuracy"[26]. Expert evaluation in this study revealed that the system's high realism in differentiating resistance feedback from tissue layers—such as the varying resistance encountered when dissecting fascia versus muscle tissue—and its accurate mechanical responses to surgical operations (including cutting, suturing, and retraction). These features enable surgeons to effectively transfer the skills acquired in the simulated environment to real surgical scenarios. This transfer effect is particularly significant in reducing the risk of complications during the "unskilled skill stage" of the learning curve, such as minimizing bleeding caused by uneven suturing, and in enhancing the efficiency of skill acquisition, thereby shortening the transition period from "novice" to "proficient operator." Additionally, the VR simulation system facilitates "targeted training" that addresses specific challenges at different stages of the learning curve, allowing for repeated simulation drills focused on individual surgeons' weak links, such as deep aorta exposure. This personalized training model enhances the practical value of simulation outcomes in clinical settings, making the process of shortening the learning curve more focused and effective.
LIMITATIONS
Firstly, this study was a retrospective observational study and is subject to the inherent limitations of such designs. Secondly, the reliance on a single-surgeon series in the learning curve study may limit generalizability, as the outcomes may be influenced by specific techniques and institutional protocols unique to that surgeon. Furthermore, the validation system has not yet established comprehensive validity and reliability; it lacks direct assessment of construct and predictive validity, as well as systematic reliability testing. To demonstrate its efficacy in accelerating the acquisition of surgical competency among trainees, prospective, multi-user validation studies are necessary.
CONCLUSION
This study quantitatively identifies the learning curve inflection point for proximal anastomosis in MICS CABG within a single-surgeon series. Surpassing this threshold is associated with significantly reduced operative duration while maintaining equivalent perioperative safety profiles. A key contribution of our work is the use of a substantial single-institution procedural cohort (n = 132 consecutive cases) for clinical learning curve analysis, combined with the systematic integration of a high-fidelity VR simulation platform. Additionally, we developed and validated a VR surgical simulation that provides a biomechanically realistic and haptically integrated environment for practicing critical procedural steps. The preliminary VR prototype system developed in this study will undergo iterative hardware and software upgrades through continuous data accumulation, ultimately establishing an integrated surgical training ecosystem that combines skill development, preoperative planning, and risk rehearsal. This approach aims to systematically shorten the learning curve and advance surgical training toward a new paradigm of precision and efficiency.
DECLARATIONS
Authors’ contributions
Made substantial contributions to the conception and design of the study and performed data analysis and interpretation: Liu J, Cui J, Li S, Liang L
Performed data acquisition and provided technical and material support: Chi L, Shen J
Availability of data and materials
The data supporting their findings are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the Capital Health Research and Development of Special (No. 2022-2Z-20611).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
The research was performed in accordance with the Declaration of Helsinki. Approval for the study was granted by the institutional ethics committee of Beijing Anzhen Hospital (Approval number: 2026012X). The ethics committee waived the need for informed consent to participate in the study.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. McGinn JT Jr, Usman S, Lapierre H, Pothula VR, Mesana TG, Ruel M. Minimally invasive coronary artery bypass grafting: dual-center experience in 450 consecutive patients. Circulation. 2009;120:S78-84.
2. Guo MH, Wells GA, Glineur D, et al. Minimally Invasive coronary surgery compared to STernotomy coronary artery bypass grafting: the MIST trial. Contemp Clin Trials. 2019;78:140-5.
3. Snegirev MA, Paivin AA, Denisyuk DO, et al. Minimally invasive multivessel coronary bypass surgery: angiographic patency data. J Card Surg. 2020;35:620-5.
4. Nambala S, Mishra YK, Ruel M. Less invasive multivessel coronary artery bypass grafting: now is the time. Curr Opin Cardiol. 2021;36:735-9.
5. Ruel M, Halkos ME. Minimally invasive coronary artery bypass grafting is the future: pro. Semin Thorac Cardiovasc Surg. 2025;37:34-42.
6. Sef D, Thet MS, Hashim SA, Kikuchi K. Minimally invasive coronary artery bypass grafting for multivessel coronary artery disease: a systematic review. Innovations. 2024;19:351-9.
7. Une D, Lapierre H, Sohmer B, Rai V, Ruel M. Can minimally invasive coronary artery bypass grafting be initiated and practiced safely?: a learning curve analysis. Innovations. 2013;8:403-9.
8. Rodriguez ML, Lapierre HR, Sohmer B, Ruel JP, Ruel MA. Predictors and outcomes of sternotomy conversion and cardiopulmonary bypass assistance in minimally invasive coronary artery bypass grafting. Innovations. 2016;11:315-20.
9. Rodriguez ML, Lapierre HR, Sohmer B, Glineur D, Ruel M. Mid-term follow-up of minimally invasive multivessel coronary artery bypass grafting: is the early learning phase detrimental? Innovations. 2017;12:116-20.
10. Kikuchi K, Endo Y. Assistive techniques for proximal anastomosis in minimally invasive coronary artery bypass grafting. Innovations. 2017;12:224-6.
11. Une D, Sakaguchi T. Initiation and modification of minimally invasive coronary artery bypass grafting. Gen Thorac Cardiovasc Surg. 2019;67:349-54.
12. Qureshi SH, Ruel M. The 7 pillars of multivessel minimally invasive coronary surgery. Innovations. 2021;16:216-7.
13. Ocagli H, Cella N, Stivanello L, Degan M, Canova C. The Barthel index as an indicator of hospital outcomes: a retrospective cross-sectional study with healthcare data from older people. J Adv Nurs. 2021;77:1751-61.
14. Yushkevich PA, Yang Gao, Gerig G. ITK-SNAP: an interactive tool for semi-automatic segmentation of multi-modality biomedical images. Annu Int Conf IEEE Eng Med Biol Soc. 2016;2016:3342-5.
15. Li S, Cui J, Hao A, Zhang S, Zhao Q. Design and evaluation of personalized percutaneous coronary intervention surgery simulation system. IEEE Trans Vis Comput Graph. 2021;27:4150-60.
16. De Boer A, Van Der Schoot A, Bijl H. Mesh deformation based on radial basis function interpolation. Comput Struct. 2007;85:784-95.
18. Li C, Yu P, Liu T, et al. MGPBD: a multigrid accelerated global XPBD solver. In: Alford G, Zhang H, Schulz A, editors. Proceedings of the Special Interest Group on Computer Graphics and Interactive Techniques Conference Conference Papers, 2025 Aug 10-14; Vancouver, Canada. New York: Association for Computing Machinery; 2025. pp. 1-11.
19. Sadeghi AH, Bakhuis W, Van Schaagen F, et al. Immersive 3D virtual reality imaging in planning minimally invasive and complex adult cardiac surgery. Eur Heart J Digit Health. 2020;1:62-70.
20. Liu J, Kong Q, Tang Z, et al. Analysis of the learning curve for minimally invasive coronary artery bypass grafting. Chin J Clin Thorac Cardiovasc Surg. 2021;28:639-44. (in Chinese). Available from: https://www.cnki.com.cn/Article/CJFDTotal-ZXYX202106006.htm [Last accessed on 16 Apr 2026].
21. Wohl H. The cusum plot: its utility in the analysis of clinical data. N Engl J Med. 1977;296:1044-5.
22. Chen R, Cui J, Li S, Hao A. A coupling physics model for real-time 4D simulation of cardiac electromechanics. Comput Aided Des. 2024;175:103747.
23. Shahrezaei A, Sohani M, Taherkhani S, Zarghami SY. The impact of surgical simulation and training technologies on general surgery education. BMC Med Educ. 2024;24:1297.
24. Nia P, Daemen JHT, Maessen JG. Development of a high-fidelity minimally invasive mitral valve surgery simulator. J Thorac Cardiovasc Surg. 2019;157:1567-74.
25. Pan J, Zhang L, Yu P, et al. Real-time VR simulation of laparoscopic cholecystectomy based on parallel position-based dynamics in GPU. In: Argelaguet F, Bruder G, Kopper R, et al., editors. 2020 IEEE Conference on Virtual Reality and 3D User Interfaces (VR), 2020 Mar 22-26; Atlanta, USA. New York: IEEE; 2020. pp. 548-56.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].