Reconstructing strategies for precision diagnosis and treatment of liver cancer based on multi-modal data
Abstract
Liver cancer, particularly hepatocellular carcinoma (HCC), poses a severe global public health threat owing to its high incidence, frequent late-stage diagnosis, and poor 5-year survival rate. Conventional approaches to liver cancer diagnosis and treatment are limited by their reliance on subjective physician experience, uniform and undifferentiated treatment strategies, and imprecise prognostic assessment. This review synthesizes studies published between 2019 and 2025 on the application of multi-modal data in liver cancer care, including computed tomography (CT), magnetic resonance imaging (MRI), pathology, and multi-omics data. We explore the utility of single-modal data analysis including the role of CT or MRI in enhancing diagnostic accuracy and the application of pathological data. Subsequently, the review focuses on multi-modal data fusion strategies, including feature-level, decision-level, and modal-level fusion, which collectively support precision diagnosis, personalized treatment recommendation, and accurate prognosis prediction in clinical practice. Additionally, it addresses critical challenges such as data heterogeneity and low physician acceptance of integrated data-driven tools, while outlining future directions including the development of standardized multi-modal data ecosystems. This review highlights multi-modal data as a core driver of precision liver cancer care, with the objective of accelerating its translation into routine clinical practice.
Keywords
INTRODUCTION
Liver cancer, as one of the most prevalent and lethal malignancies worldwide, poses an enormous threat to global public health. According to the latest global cancer statistics, it ranks as the sixth most diagnosed cancer and the third leading cause of cancer-related deaths, with an estimated 865,269 new cases and 757,948 deaths annually[1]. Hepatocellular carcinoma (HCC), accounting for approximately 75%-85% of all liver cancer cases, is particularly prominent in regions such as East Asia and sub-Saharan Africa, where chronic hepatitis B virus (HBV) infection and alcohol exposure are highly prevalent[2,3]. This review primarily focuses on HCC, which is the most clinically prevalent and well-studied subtype in the context of multi-modal data integration. For intrahepatic cholangiocarcinoma (ICC), combined Hepatocellular-Cholangiocarcinoma (HCC-CCA), and liver metastases, we briefly discuss their relevant multi-modal data applications where research evidence is available, while noting the current lack of large-scale multi-modal studies for these subtypes due to their relatively low incidence and heterogeneous pathological characteristics. Notably, in China alone, the incidence of liver cancer accounts for over 50% of the global total, and lots of patients are diagnosed at an advanced stage due to the lack of effective early screening strategies[4,5]. This grim clinical reality underscores an urgent need to revolutionize the current paradigm of liver cancer diagnosis and treatment.
Traditional approaches to liver cancer management are plagued by inherent limitations that hinder the achievement of precision medicine. In the diagnostic phase, the identification of liver nodules and differentiation between benign and malignant lesions heavily rely on the subjective experience of radiologists and pathologists. For instance, the interpretation of imaging modalities such as ultrasound (US), computed tomography (CT), and magnetic resonance imaging (MRI) exhibits significant inter-observer variability, especially for small lesions which are often missed or misclassified[6]. Pathological analysis, the gold standard for HCC diagnosis, is time-consuming, with microscopic evaluation of whole-slide imaging (WSI) taking up to 40 min per sample[7]. In terms of treatment, a uniform and undifferentiated strategy, which includes transcatheter arterial chemoembolization (TACE), surgical resection, and targeted therapy, fails to account for tumor heterogeneity at the morphological, cellular, and molecular levels[8,9]. This leads to suboptimal treatment responses, with nearly 50% of patients experiencing recurrence or metastasis with curative-intent treatment[10,11]. Prognostic assessment, relying on single clinical indicators such as alpha-fetoprotein (AFP) and Tumor-Node-Metastasis (TNM) staging, also lacks sufficient accuracy to stratify patients into risk groups and guide personalized follow-up plans[12,13].
In recent years, the rapid advancement of multi-modal data acquisition and artificial intelligence (AI) technology has opened new avenues for addressing these unmet needs in liver cancer care. Multi-modal data encompassing macroscopic tumor morphology, vascular status, pathological cellular features and multi-omics profiles integrates information from multiple sources that reflect tumor characteristics and provides a comprehensive perspective covering the macroscopic, microscopic and molecular dimensions of liver cancer, thus facilitating a more holistic understanding of tumor biology[14,15]. AI, particularly deep learning and machine learning algorithms, excels at automated feature extraction, pattern recognition, and predictive modeling[16-19]. For example, AI-driven imaging analysis can achieve a sensitivity of over 90% for liver nodule detection and identify patients as low risk with a high Negative Predictive Value (NPV) of 99.0%[20-22]. AI-based pathological analysis is capable of classifying image patches as either HCC or intrahepatic cholangiocarcinoma with an accuracy of 0.88, and AI-integrated analysis can reach an area under the curve (AUC) of 0.84 for early recurrence prediction[23,24]. More importantly, the integration of multi-modal data via AI has emerged as a transformative approach for precision liver cancer diagnosis and treatment, enabling personalized clinical decision-making through accurate tumor characterization. AI-powered multi-modal fusion leverages complementary strengths across data types and overcomes limitations of individual modalities, thereby improving performance over single-modal approaches in key clinical tasks, including early diagnosis, treatment decision-making, and prognosis prediction[25-27].
For this narrative review, we systematically searched peer-reviewed studies published between January 2019 and June 2025 in the PubMed, Web of Science, Embase, and China National Knowledge Infrastructure (CNKI) databases. The key retrieval terms included combinations of “liver cancer/hepatocellular carcinoma”, “multimodal/multi-modal data”, “artificial intelligence/machine learning/deep learning”, “precision diagnosis/treatment”, “imaging/pathology/multi-omics”, and “data fusion”. We included original research articles, clinical trials, and systematic reviews that focused on the application of multi-modal data and AI in liver cancer diagnosis, prognosis, and treatment decision-making. We excluded non-English articles, conference abstracts, preprints without peer review. After screening titles, abstracts, and full texts, a total of 137 high-quality studies were finally included in this review for synthesis and analysis. This review aims to systematically synthesize the latest advances in multi-modal data analysis for liver cancer. It will first overview the applications of single-modal data as the foundation of precision care, then delve into the theoretical basis, technical strategies, and clinical applications of multi-modal fusion. Additionally, the current challenges in data quality, model interpretability, and clinical translation will be discussed, along with future directions to advance the integration of multi-modal into routine clinical practice. Ultimately, this review seeks to highlight the pivotal role of multi-modal in revolutionizing liver cancer care and improving patient outcomes.
SINGLE-MODAL DATA ANALYSIS IN LIVER CANCER: FOUNDATIONS FOR PRECISION
Single-modality data serves as the fundamental building block for understanding HCC biology and guiding clinical decisions. Unlike multi-modality data, which integrates information from multiple sources but may face challenges in data fusion and standardization, single-modality data offers the advantages of relatively straightforward acquisition, standardized protocols, and deep phenotypic characterization within a specific dimension. Imaging data primarily reflects the macroscopic structural and functional changes of the liver and tumors, providing critical information for tumor detection, localization, and staging[28]. Pathological data reveals the microscopic cellular and histological features that determine tumor aggressiveness and treatment sensitivity[29]. The application of AI to these data types enables the extraction of hidden, high-dimensional features that are imperceptible to the human eye, thereby enhancing the accuracy and efficiency of clinical assessments[30] [Table 1].
Single-modality AI applications in hepatocellular carcinoma
| Modality | Main clinical tasks | Typical performance metrics | Key limitations | References |
| US/CEUS | Liver nodule detection; benign-malignant differentiation; CEUS phase analysis | Nodule detection: Sens = 90.2%, Spec = 78.0%; benign-malignant differentiation: AUC = 0.934; CEUS diagnosis: Acc = 85.0% | High operator dependence; low resolution for small lesions (< 1 cm); poor consistency in elastography parameter measurement | [41-43] |
| Contrast-enhanced CT | HCC segmentation; portal vein invasion detection; radiomic subtype classification | Tumor segmentation: DSC = 0.97; portal vein invasion: Sens = 87.0%, Spec = 66.0% | Poor soft tissue contrast for small HCC; vascular enhancement artifacts affect segmentation | [47-50] |
| DCE-MRI | Small HCC detection (≤ 3 cm); DCE-MRI TIC analysis; post-treatment recurrence prediction | Small HCC detection: Sens = 97.0%, Spec = 97.0%; TIC quantitative analysis: ICC = 0.92 | High scan cost; long acquisition time; motion/breathing artifacts; limited availability in low-resource regions | [53,54] |
| Digital pathology/WSI | HCC/intrahepatic cholangiocarcinoma classification; MVI detection; tumor grading/staging; cancer cell identification | HCC/intrahepatic cholangiocarcinoma classification: Acc = 0.90; MVI detection: Acc = 94.25% | Time-consuming WSI scanning; subjective annotation standards; lack of 3D spatial tumor context | [64,68] |
| Genomics (ctDNA/tissue) | Driver gene mutation detection (TP53/CTNNB1); MRD monitoring; genetic risk prediction | Mutation detection: F1 = 0.91; recurrence risk: agreement = 80.0% | Low ctDNA allele frequency in early HCC; sequencing errors; inability to reflect spatial molecular heterogeneity; high test cost | [77,78,81,83] |
| Proteomics/Metabolomics | Serum biomarker panel development; Metabolic subtype classification; early HCC diagnosis; prognostic marker screening | 4-protein panel diagnosis: AUC = 0.890, Sens = 0.909 | Low specificity in chronic liver disease patients; serum protein degradation; large sample size requirement for panel validation | [87,88] |
Analysis of tumor macroscopic features from images
Imaging technology has long been an indispensable tool in the clinical workflow of HCC, from screening high-risk populations to monitoring treatment response[31-33]. The integration of AI with various imaging modalities has revolutionized the analysis of tumor macroscopic features, enabling more precise clinical decision-making, including US, CT and MRI[34].
US is the most widely used initial screening tool for HCC due to its non-invasiveness, portability, lack of ionizing radiation, and cost-effectiveness[35,36]. However, the interpretation of US images is highly dependent on the operator's experience, leading to variability in diagnostic accuracy[37,38]. AI has demonstrated significant potential in addressing these limitations, with key application directions including liver nodule detection, benign-malignant differentiation, and contrast-enhanced US (CEUS) analysis[39,40].
In liver nodule detection, AI models, particularly deep learning-based object detection algorithms, have achieved remarkable performance. A study by Lu et al. developed an AI system for automatic detection of HCC nodules in US images, achieving a sensitivity of over 90% and a specificity of 78%, outperforming junior and intermediate radiologists. This high sensitivity is crucial for screening high-risk populations, as it minimizes the risk of missed diagnoses of early-stage tumors[41]. For the differentiation of benign and malignant liver nodules, AI models can integrate multiple US features, including grayscale texture, blood flow characteristics, and elastography parameters, to establish a comprehensive classification model. Hu et al. proposed an AI model that combined conventional US and shear wave elastography data, achieving an AUC of 0.934 for distinguishing HCC from benign nodules, which was significantly higher than the accuracy of experienced radiologists[42]. CEUS, which evaluates tumor vascularization by injecting contrast agents, provides additional functional information for HCC diagnosis. AI models can automatically segment the enhancement phases of CEUS and extract dynamic enhancement features to improve diagnostic accuracy. Ding et al. developed a temporal convolutional network for CEUS analysis, which accurately identified the characteristic “wash-in and wash-out” enhancement pattern of HCC, with an overall diagnostic accuracy of 85%[43].
CT, especially contrast-enhanced CT, is a cornerstone imaging modality for HCC diagnosis and staging, offering high spatial resolution and clear visualization of tumor-vascular relationships[44]. AI applications in CT imaging mainly focus on tumor segmentation, vascular invasion assessment, and subtype classification based on radiomics[45,46].
Tumor segmentation is a prerequisite for quantitative analysis of tumor features, such as volume and growth rate. Traditional manual segmentation is time-consuming and subjective, while AI-based automatic segmentation models have shown superior efficiency and consistency. Özcan et al. developed a U-shaped Convolutional Network (U-Net)-based model for automatic segmentation of HCC in contrast-enhanced CT images, achieving a dice similarity coefficient (DSC) of 0.97[47]. Vascular invasion is also a critical prognostic factor for HCC and directly influences treatment strategy selection[48]. AI models can automatically identify and assess vascular invasion by analyzing the continuity and enhancement of blood vessels in CT images[49]. A study by Xiao et al. applied a Convolutional Neural Network (CNN) to contrast-enhanced CT data, achieving a sensitivity of 87% and a specificity of 66% for detecting portal vein invasion, which was comparable to the performance of senior radiologists[50].
MRI offers superior soft tissue contrast and multi-parametric imaging capabilities, making it the most sensitive modality for detecting small HCC and evaluating tumor function[51]. With the advancement of deep learning, especially the application of Transformer architecture, AI has further expanded the clinical utility of MRI in HCC management. Early detection of small HCC is crucial for improving patient survival, as these tumors are often curable with surgical resection or ablation[52]. However, small HCC nodules may have indistinct features on conventional MRI, leading to missed diagnoses[6]. Zhong et al. proposed a Liver Imaging Reporting and Data System (LI-RADS) model for small HCC detection in T1 and T2-weighted MRI images, achieving a sensitivity of 97% and a specificity of 97%[53]. Dynamic contrast-enhanced MRI (DCE-MRI), which evaluates the dynamic enhancement of tumors over time, provides important information about tumor angiogenesis and perfusion, making it a valuable tool for monitoring treatment efficacy. AI models can automatically analyze DCE-MRI time-intensity curves (TICs) to extract quantitative parameters, such as peak enhancement time and washout rate, which reflect changes in tumor vascularity after treatment[54,55].
Analysis of tumor microscopic features in pathological examination
Pathological examination is the gold standard for HCC diagnosis, as it directly reveals the microscopic biological characteristics of tumors, which are critical for determining prognosis and guiding treatment[56]. The advent of digital pathology, particularly WSI, has enabled the digitization of pathological slides, facilitating the application of AI for high-throughput and quantitative analysis of tumor microscopic features[57]. WSI technology scans conventional glass slides at high resolution (up to 0.25 μm/pixel), generating digital images that can be viewed, analyzed, and shared electronically[58]. WSI preserves the full histological context of the tissue sample, providing a rich dataset for AI analysis[59-61]. The main AI tasks in digital pathology for HCC include cancer cell identification, tumor grading and staging, and microvascular invasion (MVI) detection, which has significant clinical translational value, particularly in improving diagnostic efficiency and guiding adjuvant treatment[62].
AI models can automatically pre-screen slides, identify suspicious regions, and provide preliminary diagnostic suggestions, significantly reducing the workload of pathologists[63]. Then, they can distinguish liver tumors and perform multi-class cancer classification with higher sensitivity and specificity[64]. Guiding adjuvant treatment is another key clinical application. MVI status is a critical determinant of adjuvant treatment in HCC patients after surgical resection[65]. Patients with MVI are at high risk of recurrence and may benefit from adjuvant therapies such as TACE, targeted therapy, or immunotherapy[66,67]. AI-based MVI detection can provide accurate and timely information to guide treatment decisions. A prospective clinical trial conducted by Zhang et al. enrolled 753 HCC patients who had undergone surgical resection, based on which an AI-based diagnostic model for MVI was developed. This model demonstrated a high diagnostic accuracy of 94.25% in the independent external validation set. Notably, MVI-Artificial Intelligence Decision-Making Model (MVI-AIDM) can provide spatial information regarding MVI, a key piece of data that is critical for the accurate prediction of postoperative HCC recurrence[68].
Analysis of tumor molecular features from multi-omics data
HCC exhibits profound molecular heterogeneity, which underlies its diverse clinical manifestations, treatment responses, and prognostic outcomes[69,70]. Traditional single-marker approaches fail to capture the complex molecular landscape of HCC, limiting the precision of diagnosis and treatment. Multi-omics data, which integrates information from genomics, transcriptomics, proteomics, and metabolomics, provides a comprehensive view of tumor molecular characteristics[71-73]. The integration of AI with multi-omics data enables the mining of hidden molecular patterns, offering unprecedented insights into HCC pathogenesis and personalized medicine[74,75].
Genomic analysis, particularly of circulating tumor DNA (ctDNA) and tissue DNA, has emerged as a powerful tool for investigating the genetic basis of HCC[76]. AI has significantly enhanced the utility of genomic data by enabling sensitive detection of mutations, accurate prediction of genetic risk, and reliable monitoring of minimal residual disease (MRD). One of the key applications of AI in HCC genomics is the detection of driver gene mutation, such as Tumor Protein 53 (TP53) and Catenin Beta 1 (CTNNB1), which are frequently mutated in HCC and play critical roles in tumor initiation and progression[77,78]. Traditional sequencing analysis methods often face challenges in distinguishing true mutations from sequencing errors, especially in low-allele-frequency ctDNA samples. AI models, such as deep learning-based variant callers, can integrate multiple sequencing features to improve mutation detection accuracy[79]. Genetic risk prediction is another important application of AI in HCC genomics. By integrating genomic data with clinical and environmental factors, AI models can identify individuals at high risk of HCC development or recurrence, facilitating early screening and intervention[80]. Shen et al. constructed a machine learning model that used seven genetic variants to predict HCC recurrence risk. The agreement rate between the results from the decision tree model and the clinical observation reached 80%. This risk prediction model can help prioritize high-risk individuals for intensive screening, reducing the burden of population-wide screening[81].
MRD monitoring, which detects residual tumor cells after curative treatment, is crucial for predicting recurrence and guiding adjuvant therapy[82]. AI models can analyze ctDNA dynamics to identify MRD and predict recurrence risk. Huang et al. found that serial blood samples collected for profiling ctDNA mutations can predict tumor recurrence after liver transplantation[83]. Baseline ctDNA mutational profiles were compared with those of matched tumor tissues. This early detection of MRD enables timely intervention, potentially improving patient survival. In addition to mutation detection and risk prediction, AI can also assist in molecular subtype classification and prognostic gene marker screening[84]. Chen et al. applied a computational framework to integrate multi-omics data from HCC patients using the latest ten different clustering algorithms. They distinguished two subtypes of liver cancer and found that patients with subtype 2 exhibited superior overall survival (OS). AI-based subtype classification can provide valuable information for tailoring treatment strategies, such as using targeted therapy for the proliferative subtype and metabolic modulators for the metabolic subtype[85].
Proteomics and metabolomics provide complementary information to genomics, reflecting the functional state of tumors at the protein and metabolite levels[86]. AI models can integrate proteomic and metabolomic data to identify biomarkers for early diagnosis and survival prediction[87]. Xing et al. conducted a large-scale proteomic study of serum samples from 1,000 HCC patients. They identified a 4-protein panel for HCC and healthy person diagnosis. This panel achieved an AUC of 0.890 and a sensitivity of 0.909. The 4-protein panel was particularly effective in distinguishing patients with liver cancer and healthy persons[88].
MULTI-MODAL DATA FUSION FOR PRECISION IN LIVER CANCER DIAGNOSIS AND TREATMENT
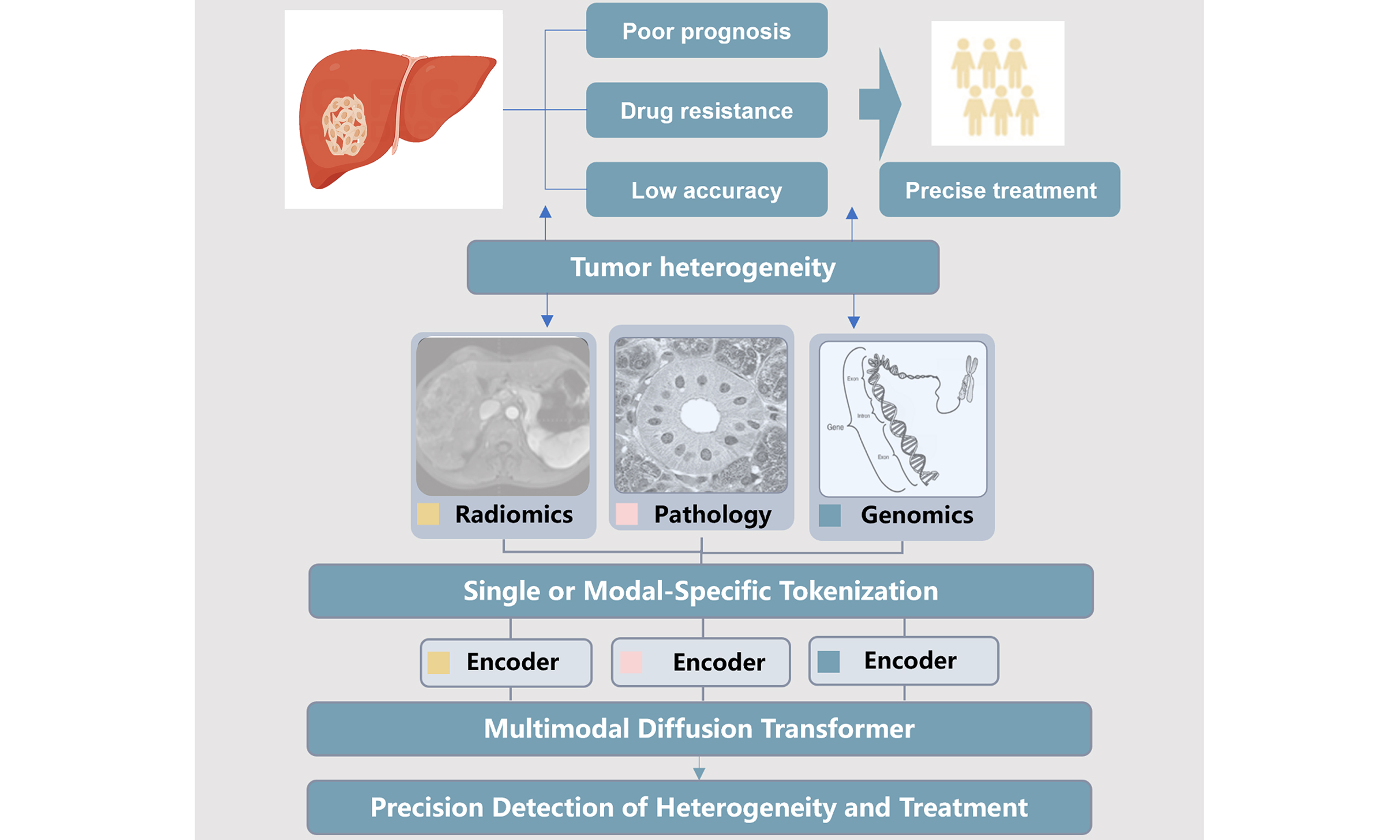
While single-modality data has laid a solid foundation for HCC precision medicine, each modality inherently has limitations. Images capture macroscopic structure but lack molecular insights; pathology reveals microscopic features yet lacks spatial context of the entire tumor, and multi-omics deciphers molecular heterogeneity but cannot reflect morphological characteristics[89-91]. AI-powered multi-modal fusion, which integrates complementary information from multiple modalities, has emerged as a transformative approach to overcome these limitations and achieve a more comprehensive understanding of HCC. This section elaborates on the theoretical basis, technical strategies, and clinical applications of multi-modal fusion [Table 2].
Multi-modal data fusion for hepatocellular carcinoma
| Modality combinations | Fusion strategy | Clinical endpoint | Validation type | Interpretability method used | References |
| Digital histopathology (WSI) + Genomics (MSI molecular detection) | Feature-level fusion | MSI status prediction in cancer via histology; non-invasive surrogate for molecular MSI detection | Multi-center external validation | Histopathological feature heatmap visualization; deep learning attention region mapping; MSI-associated morphological feature mining | [99] |
| Contrast-enhanced CT/MRI Radiomics + Clinical covariates (cirrhosis background) | Feature-level fusion | HCC diagnosis; HCC recurrence prediction after surgical resection; radiomics machine-learning signature construction and validation | External multi-center | Radiomic feature selection and ranking; machine-learning signature weight visualization; diagnostic performance subgroup analysis (cirrhosis subtype) | [101-103] |
| Multi-parametric imaging (CT/MRI) + Transcriptomics (transcriptome subclasses/signatures) | Modal-level fusion | HCC transcriptome subclass non-invasive prediction; imaging surrogate marker identification for transcriptome signatures; tumor biological behavior correlation analysis | Internal + external validation | Imaging feature-transcriptome signature correlation analysis; subtype-specific imaging phenotype visualization; radiomic signature enrichment analysis | [105] |
| Digital pathology/WSI + clinical data + ctDNA | Multi-level fusion (feature+decision) | Postoperative recurrence prediction; adjuvant therapy guidance (TACE/lenvatinib) | Prospective multi-center | Grad-CAM (WSI); SHAP values (clinical/omics) | [107,108] |
| CT radiomics + Genomics (ADH1A gene/retinol metabolism pathway) | Feature-level fusion | HCC OS prediction; retinol metabolism pathway-related molecular feature correlation analysis | External multi-center | Radiomic feature ranking; pathway enrichment analysis; ADH1A expression-radiomic feature correlation plot | [109] |
The theoretical foundation of multi-modal fusion lies in the complementarity of cross-dimensional information and the integrity of tumor phenotypic characterization[92-94]. Tumor development is a complex process involving the interaction of macroscopic structure, microscopic morphology, and molecular regulation[78,95]. No single modality can fully capture the multi-level characteristics of tumors, and the limitations of individual modalities often restrict the accuracy of clinical assessments[96,97]. Multi-modal fusion addresses these limitations by integrating information from different modalities to construct a comprehensive model that encompasses the macro, micro, and molecular scale dimensions. This integrated model achieves three core objectives to advance medical research and clinical practice. First, it mitigates single-modality information limitations by fusing imaging spatial data with omics molecular data, enabling accurate identification of region-specific molecular alterations[98]. Second, it reduces uncertainty in clinical assessments via cross-validating multi-modal conclusions, significantly enhancing result reliability and reproducibility[92]. Third, it facilitates discovery of novel cross-modal associations providing new insights into tumor biology[99]. Collectively, this model serves as a robust tool to support personalized diagnosis, evidence-based treatment decision-making and precision prognosis prediction, bridging the gap between multi-omics research and clinical utility in precision oncology.
In HCC research and clinical practice, the radio-genomics approach has made substantial progress in establishing associations between specific imaging traits and underlying molecular characteristics[100,101]. It correlates contrast-enhanced CT/MRI features including arterial hyperenhancement, washout, and irregular tumor margins with angiogenic pathway activation, cellular proliferation, and aggressive tumor behavior. For example, a non-invasive radiological signature linked to the “proliferation class” of HCC enables identification of patients at high risk of early recurrence[102]. Radiomic models also show utility in predicting mutations in key genes such as CTNNB1, which correlate with less aggressive tumor phenotypes and hypo-vascular imaging appearances[103]. Notably, this radio-genomic approach overcomes the spatial limitations of traditional biopsy by enabling non-invasive mapping of molecular alterations across the entire tumor volume, a capability critical for guiding targeted therapy selection and predicting disease progression[104,105].
The ultimate value of multi-modal data fusion lies in the development of clinical decision support systems that generate actionable clinical recommendations for diagnosis, treatment selection, and prognosis prediction[99]. For patients with newly diagnosed HCC, such systems can synthesize multi-dimensional data including clinical variables, radiological phenotypes, AI-derived pathological scores, and circulating biomarkers to construct a comprehensive composite risk profile[106].
Clinical variables include age and liver function, radiological phenotypes come from CT/MRI, AI-derived pathological scores are from biopsy whole-slide images, and circulating biomarkers include AFP and ctDNA. This profile not only predicts OS but also forecasts response to specific interventions such as lenvatinib, immune checkpoint inhibitors, or transarterial chemoembolization[107-109]. Additionally, multi-modal clinical decision support systems reduce diagnostic and prognostic uncertainty through cross-validation of findings across data types[110,111]. This process boosts the reliability and reproducibility of clinical assessments.
Collectively, multi-modal data fusion is reshaping the landscape of HCC management by translating the abstract concept of precision oncology into tangible clinical tools. By integrating macroscopic, microscopic, and molecular information, this approach transcends the limitations of one-dimensional assessments, enabling a comprehensive understanding of each patient’s unique tumor biology and driving more personalized, effective HCC care. Macroscopic information refers to radiological data, microscopic information refers to pathological data, and molecular information includes genomic data and biomarker data.
CURRENT CHALLENGES IN MULTI-MODAL LIVER CANCER CARE
Despite the significant progress made in integrating AI with single-modality data for HCC precision diagnosis and treatment, several challenges remain to be addressed for broader clinical implementation[112].
One of the major challenges is the lack of standardized and high-quality datasets. AI models require large-scale, well-annotated datasets for training and validation; however, current datasets often suffer from heterogeneity in data acquisition protocols, annotation standards, and patient populations[113,114]. For example, imaging data may vary across different scanners and institutions, while pathological data may have different staining protocols and annotation criteria. This heterogeneity can reduce the generalizability of AI models. To address this issue, international consortia and research institutions should collaborate to establish standardized data acquisition and annotation protocols, and share large-scale multi-center datasets such as Global Alliance for Genomics and Health (GA4GH)[115]. The development of federated learning (FL), which enables model training across multiple institutions without sharing raw data, can also help overcome data privacy and accessibility issues[116].
Another challenge is the interpretability of AI models. Most state-of-the-art AI models, such as deep learning models, are “black boxes” that make predictions without providing clear explanations of the underlying reasoning[117,118]. This lack of interpretability limits clinical trust and acceptance, as clinicians need to understand how AI models arrive at their conclusions to make informed decisions. To improve model interpretability, researchers are developing explainable AI (XAI) techniques, such as attention mechanisms and gradient-weighted class activation mapping (Grad-CAM), which can visualize the regions of the image or features that the model uses for prediction[119,120]. For example, Grad-CAM can highlight the tumor regions in imaging data that the AI model uses for diagnosis, providing a visual explanation for clinicians[121]. Integrating XAI into clinical AI systems will be crucial for their widespread adoption.
The clinical validation and regulatory approval of AI models are also significant challenges. Most AI models are currently developed and validated in retrospective studies, and their performance in prospective clinical trials remains to be demonstrated[122,123]. Beyond data heterogeneity and poor model interpretability, multi-modal AI studies targeting liver cancer are confronted with several typical failure modes that severely impede their clinical translation and practical application. First and foremost, dataset shift is prevalent due to variations in imaging scanners, pathological staining protocols, and multi-omics sequencing platforms among different institutions, which significantly compromises the generalizability of models trained on single-center datasets. Secondly, data leakage, characterized by improper partitioning of training and validation sets and the inclusion of post-diagnosis variables in prognostic models, often results in overestimated model performance and misleading evaluations. Thirdly, the absence of standardized gold standards for clinical endpoints undermines the reliability of model training and validation processes, thereby affecting the credibility of research findings. Finally, most current multi-modal models lack robust strategies for addressing missing data, which may lead to model breakdown or biased predictions when applied in real-world clinical scenarios.
FUTURE DIRECTIONS: ADVANCING PRECISION AND CLINICAL IMPACT
Advancing the accuracy and clinical utility of HCC management requires continuous innovation and breakthroughs across multiple technological domains. Recent progress in AI is profoundly transforming this field through several key technological advancements.
XAI facilitates precision diagnosis and treatment of cancer by incorporating attention mechanisms and visualization tools, aiding in the identification of biomarkers and the development of robust predictive models for early detection[124]. The integration of XAI with machine learning improves diagnostic accuracy and aligns with the overarching objectives of precision medicine[125]. FL offers a privacy-enhancing approach by integrating 2D and 3D deep learning models within an FL framework to achieve accurate segmentation of liver and tumor regions in medical images[126]. The FL framework demonstrates considerable potential for improving tumor detection accuracy, indicating strong suitability for integration into clinical and hospital environments.
Medical digital twins represent an emerging paradigm in personalized medicine, structured around five core elements: patients, data connectivity, in-silico patient models, interactive interfaces, and twin synchronization[127,128]. By integrating multi-modal data and leveraging AI, they enable personalized clinical decision-making, disease progression prediction, and treatment simulation, thereby enhancing both the precision and efficiency of healthcare delivery. Optimization of the data ecosystem, particularly through database standardization, is crucial for supporting AI-driven healthcare innovations[129,130]. Multi-modal data, when combined with deep learning techniques, significantly enhances the diagnosis and prediction of HCC. The integration of diverse data sources, such as blood tests, CT, MRI, and biopsy samples, improves diagnostic accuracy over single-modality approaches and contributes to the construction of more reliable risk stratification models[131]. Furthermore, multi-modal AI technology enhances the extraction of clinically relevant features and supports more accurate predictive analytics[132,133]. The application of AI in medicine has been extensively explored. Recent advances in deep learning have substantially improved the analysis of medical images, particularly by extracting diagnostic, prognostic, and predictive information from radiological imaging data[134,135]. To address these evidence quality issues and standardize multi-modal AI research in liver cancer, adherence to specialized reporting and validation frameworks, including TRIPOD-AI, CONSORT-AI, and SPIRIT-AI, is critical. These frameworks guide researchers to standardize study design, data processing, model validation, and result reporting, thereby improving the transparency and reproducibility of research. For future clinical translation, external multi-center validation and prospective clinical impact studies are the core next steps. External validation verifies the generalizability of models in real-world heterogeneous datasets, while prospective studies directly evaluate whether the application of multi-modal AI tools improves clinical outcomes rather than just technical performance metrics.
However, significant challenges remain in translating algorithmic advancements into clinical practice. Many AI models exhibit high accuracy in controlled research settings but are seldom implemented in real-world clinical scenarios[136,137]. Future research should prioritize the development of clinically actionable models that integrate seamlessly into existing healthcare workflows, which involves adapting models to meet clinical requirements and ensuring they are accessible and user-friendly for healthcare professionals.
CONCLUSION
Liver cancer remains a major global health challenge, and traditional clinical care has long fallen short of meeting the demands of precision medicine. This shortfall stems from reliance on subjective diagnostic assessments, uniform treatment strategies that fail to account for individual variability, and imprecise prognostic evaluations. Synthesizing recent evidence, this review confirms that multi-modal data integration, empowered by AI, stands as a transformative solution to address these critical gaps. Single-modal data serves as a solid foundation for this advancement, and AI-enhanced imaging improves the detection of small lesions that may otherwise be overlooked. AI-accelerated pathological analysis streamlines diagnostic workflows, and multi-omics data uncovers the molecular heterogeneity of tumors. More importantly, multi-modal data fusion generates synergies that outperform single-modal tools alone. It reduces variability in diagnostic interpretations, enables the design of personalized treatment plans tailored to individual patient needs, and refines prognostic assessments, which firmly establishes multi-modal data as a core driver of precision liver cancer care.
However, translating these advances into routine clinical practice requires overcoming several key barriers. These include data heterogeneity across collection and analysis platforms, the opacity of some AI systems (often referred to as “black-box” models), limited evidence from large-scale prospective clinical trials, data privacy concerns, and resource shortages in low-resource regions that restrict equitable access. Future efforts must prioritize the development of standardized multi-modal data ecosystems, the advancement of XAI to enhance clinical trust, and the execution of prospective multi-center trials to validate tool performance. Equally critical is fostering interdisciplinary collaboration across clinical specialties and data science, alongside initiatives to expand equitable access.
In conclusion, multi-modal data holds great promise for revolutionizing liver cancer care. Resolving current challenges will accelerate its translation into clinical practice, ultimately reducing the global burden of this devastating disease and improving outcomes for patients worldwide.
DECLARATIONS
Acknowledgments
The authors gratefully acknowledge the support of the Shanghai Institute for Mathematics and Interdisciplinary Sciences (SIMIS) in the preparation of this review.
Authors’ contributions
Conceptualization: Zhou J, Yang XR
Investigation and writing original draft: Miao RZ, Zhu HR, Li TY
Investigation: Miao RZ, Zhu HR, Li TY
Writing - review and editing: Miao RZ, Zhu HR
Funding acquisition: Zhou J, Yang XR
Supervision: Zhou J, Yang XR
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool ChatGPT (OpenAI, San Francisco, USA) was used solely for language editing. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
This work was supported by the Noncommunicable Chronic Diseases-National Science and Technology Major Project (No. 2024ZD0525400), the National Natural Science Foundation of China (No. 82488101) and the 2025 Key Technology R&D Program of the Shanghai Municipal Science and Technology Commission for New Generation Information Technology (No. 25511102600).
Conflicts of interest
Yang XR is an Associate Chief Editor of the journal Hepatoma Research and also served as a Guest Editor for the Special Issue Advancing Multimodal Approaches in Liver Cancer: From Tumor Heterogeneity to Precision Therapies. Yang XR was not involved in any part of the editorial process, including reviewer selection, manuscript handling, or decision-making. The other authors declare no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Bray F, Laversanne M, Sung H, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2024;74:229-63.
2. Llovet JM, Kelley RK, Villanueva A, et al. Hepatocellular carcinoma. Nat Rev Dis Primers 2021;7:6.
3. Singal AG, Kanwal F, Llovet JM. Global trends in hepatocellular carcinoma epidemiology: implications for screening, prevention and therapy. Nat Rev Clin Oncol 2023;20:864-84.
4. Han B, Zheng R, Zeng H, et al. Cancer incidence and mortality in China, 2022. J Natl Cancer Cent 2024;4:47-53.
5. Zhan Z, Chen B, Huang R, et al. Long-term trends and future projections of liver cancer burden in China from 1990 to 2030. Sci Rep 2025;15:13120.
6. Saborido B, Darnell A, Forner A, et al. Diagnostic performance of contrast-enhanced US in small liver nodules not conclusively characterized after MRI in cirrhotic patients. Eur Radiol 2025;35:5771-80.
7. Huang L, Sun H, Sun L, et al. Rapid, label-free histopathological diagnosis of liver cancer based on Raman spectroscopy and deep learning. Nat Commun 2023;14:48.
8. Sangro B, Sarobe P, Hervás-Stubbs S, Melero I. Advances in immunotherapy for hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol 2021;18:525-43.
9. Yang X, Yang C, Zhang S, et al. Precision treatment in advanced hepatocellular carcinoma. Cancer Cell 2024;42:180-97.
10. Shi X, Wang X, Yao W, et al. Mechanism insights and therapeutic intervention of tumor metastasis: latest developments and perspectives. Signal Transduct Target Ther 2024;9:192.
11. Abdelhamed W, El-Kassas M. Hepatocellular carcinoma recurrence: predictors and management. Liver Res 2023;7:321-32.
12. Hu R, Tran B, Li S, et al. Noninvasive prognostication of hepatocellular carcinoma based on cell-free DNA methylation. PLoS ONE 2025;20:e0321736.
13. She S, Xiang Y, Yang M, et al. C-reactive protein is a biomarker of AFP-negative HBV-related hepatocellular carcinoma. Int J Oncol 2015;47:543-54.
14. Steyaert S, Pizurica M, Nagaraj D, et al. Multimodal data fusion for cancer biomarker discovery with deep learning. Nat Mach Intell 2023;5:351-62.
15. Saltz J, Almeida J, Gao Y, et al. Towards generation, management, and exploration of combined radiomics and pathomics datasets for cancer research. AMIA Jt Summits Transl Sci Proc 2017;2017:85-94.
16. Chen RJ, Lu MY, Williamson DFK, et al. Pan-cancer integrative histology-genomic analysis via multimodal deep learning. Cancer Cell 2022;40:865-78.e6.
17. Khan RA, Fu M, Burbridge B, Luo Y, Wu FX. A multi-modal deep neural network for multi-class liver cancer diagnosis. Neural Netw 2023;165:553-61.
18. Shi Y, Wang M, Liu H, Zhao F, Li A, Chen X. MIF: multi-shot interactive fusion model for cancer survival prediction using pathological image and genomic data. IEEE J Biomed Health Inform 2025;29:3247-58.
19. Mohsen F, Ali H, El Hajj N, Shah Z. Artificial intelligence-based methods for fusion of electronic health records and imaging data. Sci Rep 2022;12:17981.
20. Ying H, Liu X, Zhang M, et al. A multicenter clinical AI system study for detection and diagnosis of focal liver lesions. Nat Commun 2024;15:1131.
21. Shan R, Pei C, Fan Q, et al. Artificial intelligence-assisted platform performs high detection ability of hepatocellular carcinoma in CT images: an external clinical validation study. BMC Cancer 2025;25:154.
22. Yin C, Zhang H, Du J, Zhu Y, Zhu H, Yue H. Artificial intelligence in imaging for liver disease diagnosis. Front Med 2025;12:1591523.
23. Kiani A, Uyumazturk B, Rajpurkar P, et al. Impact of a deep learning assistant on the histopathologic classification of liver cancer. NPJ Digit Med 2020;3:23.
24. Gao W, Wang W, Song D, et al. A predictive model integrating deep and radiomics features based on gadobenate dimeglumine-enhanced MRI for postoperative early recurrence of hepatocellular carcinoma. Radiol Med 2022;127:259-71.
25. Soenksen LR, Ma Y, Zeng C, et al. Integrated multimodal artificial intelligence framework for healthcare applications. NPJ Digit Med 2022;5:149.
26. Erion G, Janizek JD, Hudelson C, et al. A cost-aware framework for the development of AI models for healthcare applications. Nat Biomed Eng 2022;6:1384-98.
27. Zhou LQ, Wang JY, Yu SY, et al. Artificial intelligence in medical imaging of the liver. World J Gastroenterol 2019;25:672-82.
28. García-Figueiras R, Baleato-González S, Padhani AR, et al. How clinical imaging can assess cancer biology. Insights Imaging 2019;10:28.
29. Wang Y, Ju X, Hua R, et al. Deep learning analysis of histopathological images predicts immunotherapy prognosis and reveals tumour microenvironment features in non-small cell lung cancer. Br J Cancer 2024;131:1833-45.
30. Woo EG, Burkhart MC, Alsentzer E, Beaulieu-Jones BK. Synthetic data distillation enables the extraction of clinical information at scale. NPJ Digit Med 2025;8:267.
31. Vithayathil M, Koku D, Campani C, et al. Machine learning based radiomic models outperform clinical biomarkers in predicting outcomes after immunotherapy for hepatocellular carcinoma. J Hepatol 2025;83:959-70.
32. Schön F, Kieslich A, Nebelung H, et al. Comparative analysis of radiomics and deep-learning algorithms for survival prediction in hepatocellular carcinoma. Sci Rep 2024;14:590.
33. Wang Q, Li X, Qian B, Hu K, Liu B. Fluorescence imaging in the surgical management of liver cancers: current status and future perspectives. Asian J Surg 2022;45:1375-82.
34. Hennedige T, Venkatesh SK. Imaging of hepatocellular carcinoma: diagnosis, staging and treatment monitoring. Cancer Imaging 2013;12:530-47.
35. Tanaka H. Current role of ultrasound in the diagnosis of hepatocellular carcinoma. J Med Ultrason 2020;47:239-55.
36. Li L, Dioguardi Burgio M, Fetzer DT, et al. Contrast-enhanced ultrasound for hepatocellular carcinoma diagnosis-AJR expert panel narrative review. AJR Am J Roentgenol 2026;226:e2532813.
37. Lee PYC, Mohamed Afif A, Anthony A, Goodyear M, Lombardo P. Ambient light intensity affecting ultrasound operator detection of liver lesions in cine-clips. Radiography 2024;30:1151-7.
38. Davis DP, Campbell CJ, Poste JC, Ma G. The association between operator confidence and accuracy of ultrasonography performed by novice emergency physicians. J Emerg Med 2005;29:259-64.
39. Chaiteerakij R, Ariyaskul D, Kulkraisri K, et al. Artificial intelligence for ultrasonographic detection and diagnosis of hepatocellular carcinoma and cholangiocarcinoma. Sci Rep 2024;14:20617.
40. Wang L, Fatemi M, Alizad A. Artificial intelligence techniques in liver cancer. Front Oncol 2024;14:1415859.
41. Lu RF, She CY, He DN, et al. AI enhanced diagnostic accuracy and workload reduction in hepatocellular carcinoma screening. NPJ Digit Med 2025;8:500.
42. Hu HT, Wang W, Chen LD, et al. Artificial intelligence assists identifying malignant versus benign liver lesions using contrast-enhanced ultrasound. J Gastroenterol Hepatol 2021;36:2875-83.
43. Ding W, Meng Y, Ma J, et al. Contrast-enhanced ultrasound-based AI model for multi-classification of focal liver lesions. J Hepatol 2025;83:426-39.
44. Chartampilas E, Rafailidis V, Georgopoulou V, Kalarakis G, Hatzidakis A, Prassopoulos P. Current imaging diagnosis of hepatocellular carcinoma. Cancers 2022;14:3997.
45. Balaguer-Montero M, Marcos Morales A, Ligero M, et al. A CT-based deep learning-driven tool for automatic liver tumor detection and delineation in patients with cancer. Cell Rep Med 2025;6:102032.
46. Shin H, Hur MH, Song BG, et al. AI model using CT-based imaging biomarkers to predict hepatocellular carcinoma in patients with chronic hepatitis B. J Hepatol 2025;82:1080-8.
47. Özcan F, Uçan ON, Karaçam S, Tunçman D. Fully automatic liver and tumor segmentation from CT image using an AIM-Unet. Bioengineering 2023;10:215.
48. Krishnan MS, Rajan Kd A, Park J, et al. Genomic analysis of vascular invasion in HCC reveals molecular drivers and predictive biomarkers. Hepatology 2021;73:2342-60.
49. Zhou Y, Sun SW, Liu QP, Xu X, Zhang Y, Zhang YD. TED: two-stage expert-guided interpretable diagnosis framework for microvascular invasion in hepatocellular carcinoma. Med Image Anal 2022;82:102575.
50. Xiao H, Guo Y, Zhou Q, et al. Prediction of microvascular invasion in hepatocellular carcinoma with expert-inspiration and skeleton sharing deep learning. Liver Int 2022;42:1423-31.
51. Hoffmann E, Masthoff M, Kunz WG, et al. Multiparametric MRI for characterization of the tumour microenvironment. Nat Rev Clin Oncol 2024;21:428-48.
52. Melendez-Torres J, Singal AG. Early detection of hepatocellular carcinoma: roadmap for improvement. Expert Rev Anticancer Ther 2022;22:621-32.
53. Zhong X, Guan T, Tang D, et al. Differentiation of small (≤ 3 cm) hepatocellular carcinomas from benign nodules in cirrhotic liver: the added additive value of MRI-based radiomics analysis to LI-RADS version 2018 algorithm. BMC Gastroenterol 2021;21:155.
54. Liu Z, Yao B, Wen J, et al. Voxel-wise mapping of DCE-MRI time-intensity-curve profiles enables visualizing and quantifying hemodynamic heterogeneity in breast lesions. Eur Radiol 2024;34:182-92.
55. Paudyal R, Shah AD, Akin O, et al. Artificial Intelligence in CT and MR imaging for oncological applications. Cancers 2023;15:2573.
56. Zhou J, Sun H, Wang Z, et al. Guidelines for the diagnosis and treatment of primary liver cancer (2022 Edition). Liver Cancer 2023;12:405-44.
57. Kiran N, Sapna F, Kiran F, et al. Digital pathology: transforming diagnosis in the digital age. Cureus 2023;15:e44620.
58. Kumar N, Gupta R, Gupta S. Whole slide imaging (WSI) in pathology: current perspectives and future directions. J Digit Imaging 2020;33:1034-40.
59. Yu J, Chen H, Hu L, et al. Exploring multi-instance learning in whole slide imaging: Current and future perspectives. Pathol Res Pract 2025;271:156006.
60. Ding GY, Shi JY, Wang XD, Yan B, Liu XY, Gao Q. Artificial intelligence-based pathological analysis of liver cancer: current advancements and interpretative strategies. ILIVER 2024;3:100082.
61. Khened M, Kori A, Rajkumar H, Krishnamurthi G, Srinivasan B. A generalized deep learning framework for whole-slide image segmentation and analysis. Sci Rep 2021;11:11579.
62. Yates J, Van Allen EM. New horizons at the interface of artificial intelligence and translational cancer research. Cancer Cell 2025;43:708-27.
64. Aatresh AA, Alabhya K, Lal S, Kini J, Saxena PUP. LiverNet: efficient and robust deep learning model for automatic diagnosis of sub-types of liver hepatocellular carcinoma cancer from H&E stained liver histopathology images. Int J Comput Assist Radiol Surg 2021;16:1549-63.
65. Zhang ZH, Jiang C, Qiang ZY, et al. Role of microvascular invasion in early recurrence of hepatocellular carcinoma after liver resection: a literature review. Asian J Surg 2024;47:2138-43.
66. Liang L, Xu ZD, Lu WF, et al. Survival benefit from adjuvant TACE combined with lenvatinib for patients with hepatocellular carcinoma and microvascular invasion after curative hepatectomy. Asian J Surg 2024;47:5106-12.
67. Du JS, Hsu SH, Wang SN. The current and prospective adjuvant therapies for hepatocellular carcinoma. Cancers 2024;16:1422.
68. Zhang X, Yu X, Liang W, et al. Deep learning-based accurate diagnosis and quantitative evaluation of microvascular invasion in hepatocellular carcinoma on whole-slide histopathology images. Cancer Med 2024;13:e7104.
69. Safri F, Nguyen R, Zerehpooshnesfchi S, George J, Qiao L. Heterogeneity of hepatocellular carcinoma: from mechanisms to clinical implications. Cancer Gene Ther 2024;31:1105-12.
70. Guo DZ, Zhang X, Zhang SQ, et al. Single-cell tumor heterogeneity landscape of hepatocellular carcinoma: unraveling the pro-metastatic subtype and its interaction loop with fibroblasts. Mol Cancer 2024;23:157.
71. Lim J, Park C, Kim M, Kim H, Kim J, Lee DS. Advances in single-cell omics and multiomics for high-resolution molecular profiling. Exp Mol Med 2024;56:515-26.
72. Moris D, Martinino A, Schiltz S, et al. Advances in the treatment of hepatocellular carcinoma: an overview of the current and evolving therapeutic landscape for clinicians. CA Cancer J Clin 2025;75:498-527.
73. Yang C, Zhang H, Zhang L, et al. Evolving therapeutic landscape of advanced hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol 2023;20:203-22.
74. Ramesh D, Manickavel P, Ghosh S, Bhat M. The integration of multi-omics with artificial intelligence in hepatology: a comprehensive review of personalized medicine, biomarker identification, and drug discovery. J Clin Exp Hepatol 2025;15:102611.
75. Nam Y, Kim J, Jung SH, et al. Harnessing artificial intelligence in multimodal omics data integration: paving the path for the next frontier in precision medicine. Annu Rev Biomed Data Sci 2024;7:225-50.
76. Bartolomucci A, Nobrega M, Ferrier T, et al. Circulating tumor DNA to monitor treatment response in solid tumors and advance precision oncology. NPJ Precis Oncol 2025;9:84.
77. Perera GS, Huang X, Bagherjeri FA, et al. Rapid and selective detection of TP53 mutations in cancer using a novel conductometric biosensor. Biosens Bioelectron 2025;276:117252.
78. Zhang S, Xiao X, Yi Y, et al. Tumor initiation and early tumorigenesis: molecular mechanisms and interventional targets. Signal Transduct Target Ther 2024;9:149.
79. Abdelwahab O, Torkamaneh D. Artificial intelligence in variant calling: a review. Front Bioinform 2025;5:1574359.
80. Tohme S, Yazdani HO, Rahman A, et al. The use of machine learning to create a risk score to predict survival in patients with hepatocellular carcinoma: a TCGA cohort analysis. Can J Gastroenterol Hepatol 2021;2021:5212953.
81. Shen J, Qi L, Zou Z, et al. Identification of a novel gene signature for the prediction of recurrence in HCC patients by machine learning of genome-wide databases. Sci Rep 2020;10:4435.
82. Pantel K, Alix-Panabières C. Minimal residual disease as a target for liquid biopsy in patients with solid tumours. Nat Rev Clin Oncol 2025;22:65-77.
83. Huang A, Guo DZ, Zhang X, et al. Serial circulating tumor DNA profiling predicts tumor recurrence after liver transplantation for liver cancer. Hepatol Int 2024;18:254-64.
84. Li C, Hu J, Li M, Mao Y, Mao Y. Integrated multi-omics analysis and machine learning refine molecular subtypes and clinical outcome for hepatocellular carcinoma. Hereditas 2025;162:61.
85. Chen S, Li Y, Hu J, et al. Integrating bulk RNA-seq, scRNA-seq, and spatial transcriptomics data to identify novel post-translational modification-related molecular subtypes and therapeutic responses in hepatocellular carcinoma. Cancer Cell Int 2025;25:330.
86. Santorelli L, Caterino M, Costanzo M. Proteomics and metabolomics in biomedicine. Int J Mol Sci 2023;24:16913.
87. Garg M, Karpinski M, Matelska D, et al. Disease prediction with multi-omics and biomarkers empowers case-control genetic discoveries in the UK Biobank. Nat Genet 2024;56:1821-31.
88. Xing X, Cai L, Ouyang J, et al. Proteomics-driven noninvasive screening of circulating serum protein panels for the early diagnosis of hepatocellular carcinoma. Nat Commun 2023;14:8392.
89. Bussi Y, Keren L. Multiplexed image analysis: what have we achieved and where are we headed? Nat Methods 2024;21:2212-5.
90. Heindl A, Nawaz S, Yuan Y. Mapping spatial heterogeneity in the tumor microenvironment: a new era for digital pathology. Lab Invest 2015;95:377-84.
91. Zhu Q, Zhao X, Zhang Y, et al. Single cell multi-omics reveal intra-cell-line heterogeneity across human cancer cell lines. Nat Commun 2023;14:8170.
92. Bera K, Braman N, Gupta A, Velcheti V, Madabhushi A. Predicting cancer outcomes with radiomics and artificial intelligence in radiology. Nat Rev Clin Oncol 2022;19:132-46.
93. Vandereyken K, Sifrim A, Thienpont B, Voet T. Methods and applications for single-cell and spatial multi-omics. Nat Rev Genet 2023;24:494-515.
94. Zhang S, Zhang J, Tian B, Lukasiewicz T, Xu Z. Multi-modal contrastive mutual learning and pseudo-label re-learning for semi-supervised medical image segmentation. Med Image Anal 2023;83:102656.
95. De Visser KE, Joyce JA. The evolving tumor microenvironment: from cancer initiation to metastatic outgrowth. Cancer Cell 2023;41:374-403.
96. Du T, Li W, Wang Z, et al. Overcoming the challenges of multi-modal medical image sharing: a novel data distillation strategy via contrastive learning. Neurocomputing 2025;617:129043.
97. Azam MA, Khan KB, Salahuddin S, et al. A review on multimodal medical image fusion: compendious analysis of medical modalities, multimodal databases, fusion techniques and quality metrics. Comput Biol Med 2022;144:105253.
98. Lewis SM, Asselin-labat M, Nguyen Q, et al. Spatial omics and multiplexed imaging to explore cancer biology. Nat Methods 2021;18:997-1012.
99. Kather JN, Pearson AT, Halama N, et al. Deep learning can predict microsatellite instability directly from histology in gastrointestinal cancer. Nat Med 2019;25:1054-6.
100. Segal E, Sirlin CB, Ooi C, et al. Decoding global gene expression programs in liver cancer by noninvasive imaging. Nat Biotechnol 2007;25:675-80.
101. Harding‐theobald E, Louissaint J, Maraj B, et al. Systematic review: radiomics for the diagnosis and prognosis of hepatocellular carcinoma. Aliment Pharmacol Ther 2021;54:890-901.
102. Ji G, Zhu F, Xu Q, et al. Machine-learning analysis of contrast-enhanced CT radiomics predicts recurrence of hepatocellular carcinoma after resection: a multi-institutional study. EBioMedicine 2019;50:156-65.
103. Mokrane F, Lu L, Vavasseur A, et al. Radiomics machine-learning signature for diagnosis of hepatocellular carcinoma in cirrhotic patients with indeterminate liver nodules. Eur Radiol 2019;30:558-70.
104. Peng J, Kang S, Ning Z, et al. Residual convolutional neural network for predicting response of transarterial chemoembolization in hepatocellular carcinoma from CT imaging. Eur Radiol 2019;30:413-24.
105. Taouli B, Hoshida Y, Kakite S, et al. Imaging-based surrogate markers of transcriptome subclasses and signatures in hepatocellular carcinoma: preliminary results. Eur Radiol 2017;27:4472-81.
106. Huang S, Yang J, Fong S, Zhao Q. Artificial intelligence in cancer diagnosis and prognosis: opportunities and challenges. Cancer Lett 2020;471:61-71.
107. Hectors SJ, Lewis S, Besa C, et al. MRI radiomics features predict immuno-oncological characteristics of hepatocellular carcinoma. Eur Radiol 2020;30:3759-69.
108. Lv X, Zhang P, Zhang E, Yang S. Predictive factors and prognostic models for Hepatic arterial infusion chemotherapy in Hepatocellular carcinoma: a comprehensive review. World J Surg Oncol 2025;23:166.
109. Yu T, Zhan Z, Lin Q. Computed tomography radiomics prediction of survival in hepatocellular carcinoma and is associated with ADH1A expression of the retinol metabolism pathway. Medicine 2025;104:e42792.
110. Tschandl P, Rinner C, Apalla Z, et al. Human-computer collaboration for skin cancer recognition. Nat Med 2020;26:1229-34.
111. Mckinney SM, Sieniek M, Godbole V, et al. International evaluation of an AI system for breast cancer screening. Nature 2020;577:89-94.
112. Kelly CJ, Karthikesalingam A, Suleyman M, Corrado G, King D. Key challenges for delivering clinical impact with artificial intelligence. BMC Med 2019;17:195.
113. Tang Z, Tang Z, Wu J. Anomaly detection in medical via multimodal foundation models. Front Bioeng Biotechnol 2025;13:1644697.
114. Liang W, Tadesse GA, Ho D, et al. Author correction: advances, challenges and opportunities in creating data for trustworthy AI. Nat Mach Intell 2022;4:904.
115. Rehm HL, Page AJ, Smith L, et al. GA4GH: international policies and standards for data sharing across genomic research and healthcare. Cell Genomics 2021;1:100029.
116. Yurdem B, Kuzlu M, Gullu MK, Catak FO, Tabassum M. Federated learning: overview, strategies, applications, tools and future directions. Heliyon 2024;10:e38137.
117. Karako K, Tang W. Applications of and issues with machine learning in medicine: Bridging the gap with explainable AI. BST 2024;18:497-504.
118. Rudin C. Stop explaining black box machine learning models for high stakes decisions and use interpretable models instead. Nat Mach Intell 2019;1:206-15.
119. Liu G, Zhang J, Chan AB, Hsiao JH. Human attention guided explainable artificial intelligence for computer vision models. Neural Netw 2024;177:106392.
120. Satapathy SM, Paul MG, Garg A, Bhatnagar S. GAN-enhanced hybrid deep learning with explainable AI for automated cataract diagnosis. J Med Syst 2025;49:123.
121. Oncu E, Ciftci F. Multimodal AI framework for lung cancer diagnosis: integrating CNN and ANN models for imaging and clinical data analysis. Comput Biol Med 2025;193:110488.
122. Lee S, Kim DW, Oh N, et al. External validation of an artificial intelligence model using clinical variables, including ICD-10 codes, for predicting in-hospital mortality among trauma patients: a multicenter retrospective cohort study. Sci Rep 2025;15:1100.
123. Cheung CY, Ran AR, Wang S, et al. A deep learning model for detection of Alzheimer’s disease based on retinal photographs: a retrospective, multicentre case-control study. The Lancet Digital Health 2022;4:e806-15.
124. Colak C, Yagin FH, Algarni A, Algarni A, Al-hashem F, Ardigò LP. Proposed comprehensive methodology integrated with explainable artificial intelligence for prediction of possible biomarkers in metabolomics panel of plasma samples for breast cancer detection. Medicina 2025;61:581.
125. Bakrania A, Joshi N, Zhao X, Zheng G, Bhat M. Artificial intelligence in liver cancers: decoding the impact of machine learning models in clinical diagnosis of primary liver cancers and liver cancer metastases. Pharmacol Res 2023;189:106706.
126. Lee C, Lien JJ, Chain K, Huang L, Hsu Z. Federated learning-based CT liver tumor detection using a teacher-student SANet with semisupervised learning. BMC Med Imaging 2025;25:250.
127. Sadée C, Testa S, Barba T, et al. Medical digital twins: enabling precision medicine and medical artificial intelligence. The Lancet Digital Health 2025;7:100864.
128. Laubenbacher R, Mehrad B, Shmulevich I, Trayanova N. Digital twins in medicine. Nat Comput Sci 2024;4:184-91.
129. Feldman K, Johnson RA, Chawla NV. The state of data in healthcare: path towards standardization. J Healthc Inform Res 2018;2:248-71.
130. Brancato V, Esposito G, Coppola L, et al. Standardizing digital biobanks: integrating imaging, genomic, and clinical data for precision medicine. J Transl Med 2024;22:136.
131. Al-antari MA. Artificial intelligence for medical diagnostics - existing and future AI technology! Diagnostics 2023;13:688.
132. Parvin N, Joo SW, Jung JH, Mandal TK. Multimodal AI in biomedicine: pioneering the future of biomaterials, diagnostics, and personalized healthcare. Nanomaterials 2025;15:895.
133. Zubair M, Hussain M, Albashrawi MA, Bendechache M, Owais M. A comprehensive review of techniques, algorithms, advancements, challenges, and clinical applications of multi-modal medical image fusion for improved diagnosis. Comput Methods Programs Biomed 2025;272:109014.
134. Chatzipanagiotou OP, Loukas C, Vailas M, et al. Artificial intelligence in hepatocellular carcinoma diagnosis: a comprehensive review of current literature. J Gastroenterol Hepatol 2024;39:1994-2005.
135. Chen X, Wang X, Zhang K, et al. Recent advances and clinical applications of deep learning in medical image analysis. Med Image Anal 2022;79:102444.
136. Van De Sande D, Chung EFF, Oosterhoff J, Van Bommel J, Gommers D, Van Genderen ME. To warrant clinical adoption AI models require a multi-faceted implementation evaluation. npj Digit Med 2024;7:58.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].