Conversion and neoadjuvant systemic therapy for intrahepatic cholangiocarcinoma: from technical resectability to biology-driven surgical selection - a systematic review and meta-analysis
Abstract
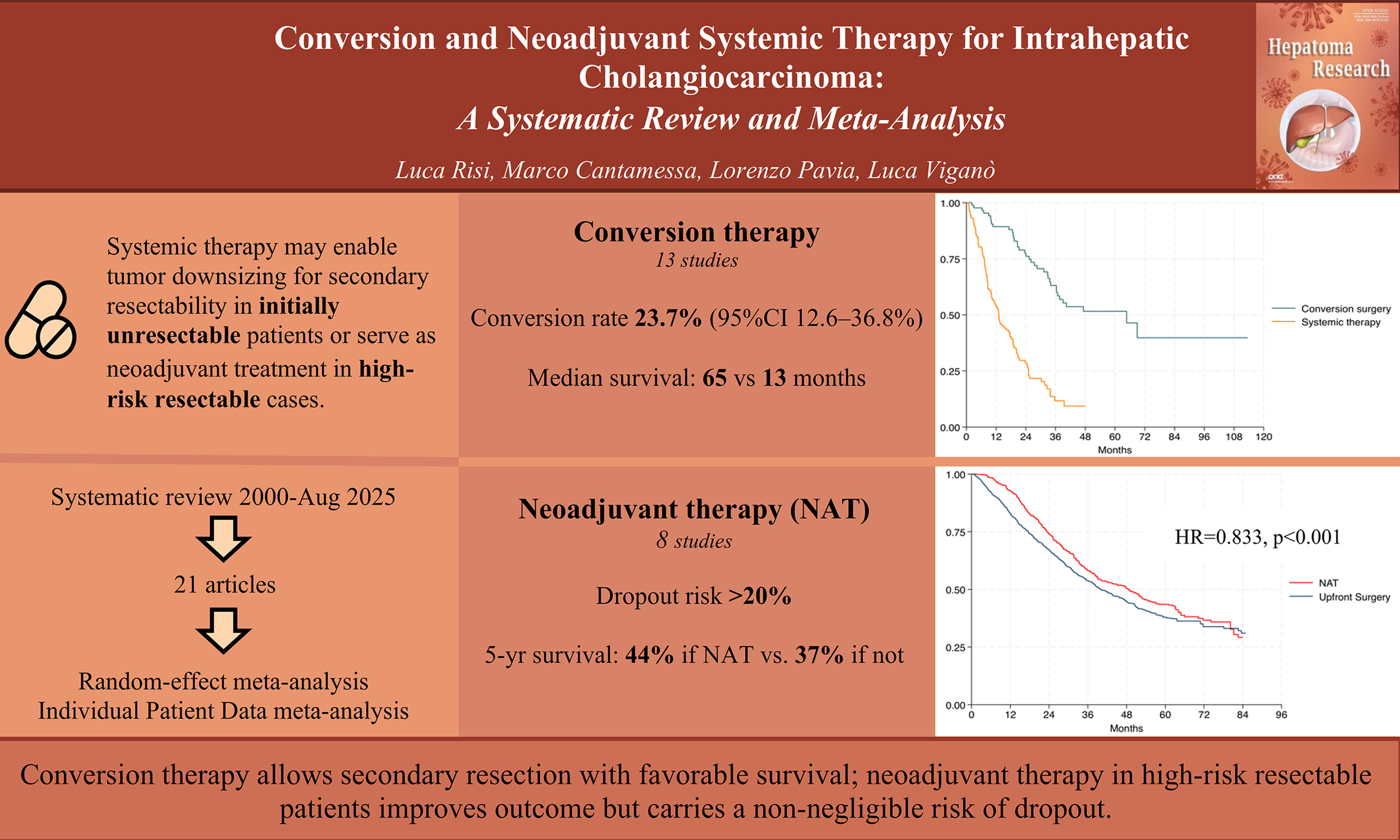
Aim: Intrahepatic cholangiocarcinoma (iCCA) is a rare liver malignancy with poor prognosis. Surgery remains the cornerstone of treatment; however, systemic therapy may enable tumor downsizing for secondary resectability in initially unresectable patients or serve as neoadjuvant treatment in high-risk resectable cases. This study aimed to systematically summarize the literature to clarify the outcomes of conversion and neoadjuvant systemic therapy in iCCA.
Methods: We conducted a systematic review of English-language studies published before August 2025. Studies were eligible if they included at least ten iCCA patients receiving conversion or neoadjuvant systemic therapy and reported treatment outcomes. Aggregated data meta-analyses were performed for categorical outcomes and individual patient-data meta-analyses were performed to evaluate overall survival (OS).
Results: Twenty-one studies (13 conversion, 8 neoadjuvant) including a median of 45 patients per study [interquartile range (IQR) 23-77] were analyzed. In the conversion setting, the pooled secondary resectability rate was 23.7%. Median OS was 64.7 months after conversion surgery vs. 13.1 months after systemic therapy alone. In the neoadjuvant setting, the dropout risk exceeded 20%; however, when the entire treatment sequence was completed, preoperative therapy was associated with improved OS from diagnosis compared with upfront surgery [5-year OS: 44% vs. 37%; hazard ratio (HR) = 0.833, P < 0.001]. Pooled postoperative mortality and severe morbidity rates were 1.2% and 19.1%, with similar outcomes in conversion and neoadjuvant groups.
Conclusion: Systemic therapy is a key adjunct in iCCA management without compromising perioperative safety. Conversion therapy enables secondary resection with favorable long-term outcomes; neoadjuvant therapy in high-risk resectable patients improves postoperative survival but carries a non-negligible risk of treatment dropout.
Keywords
INTRODUCTION
Intrahepatic cholangiocarcinoma (iCCA) is a rare malignancy with a globally increasing incidence, accounting for approximately 15% of primary liver tumors and 10%-20% of biliary tract cancers[1]. Most patients with iCCA have a poor prognosis due to the aggressive nature of the disease, its frequent presentation at advanced stages, and the limited efficacy of standard chemotherapy regimens[1,2]. In early-stage disease, surgery combined with adjuvant chemotherapy remains the cornerstone of treatment[3,4]. For advanced stages, systemic therapy represents the standard of care, with survival outcomes improving following the introduction of targeted therapies and immunotherapy[5-7].
The majority of patients are diagnosed with unresectable disease[1]. As observed in other liver tumors, in selected patients, systemic therapy may induce sufficient tumor shrinkage to allow conversion to secondary resectability. In hepatocellular carcinoma and colorectal liver metastases, conversion therapy followed by surgery has been associated with a significant survival advantage compared with palliative treatment alone[8-11]. Preliminary findings suggest similar results in iCCA[12], although available studies are heterogeneous in terms of therapeutic approach (systemic therapy, radioembolization, intra-arterial chemotherapy or radiotherapy), inclusion criteria, and conversion rates.
Conversely, liver resection with lymphadenectomy is recommended for all patients with technically resectable disease[13-15]. Advances in liver surgery have enabled resection even in patients with extensive tumor burden or complex disease presentations[16,17]. However, about 15%-20% of patients experience very early recurrence (≤ 6 months) and early cancer-related death, without deriving a meaningful survival benefit from surgery[18-21]. Oncologic resectability, defined as resection associated with a survival benefit, does not necessarily coincide with technical resectability, and the former should guide multidisciplinary decision-making[22-24]. Patients who are technically resectable but of uncertain oncologic resectability may undergo preoperative “selection” through neoadjuvant treatment[25]. Theoretically, this approach may help identify patients with more favorable tumor biology while also reducing tumor size, facilitating safer resections, and improving R0 rates[24,25]. In colorectal liver metastases, neoadjuvant therapy has become standard practice in most cases[26]. Data regarding its use in iCCA remain preliminary, heterogeneous, and challenging to interpret when considered individually.
The present study aims to clarify the role of systemic therapies in conversion strategies for initially unresectable iCCA and in neoadjuvant settings for resectable disease through a systematic review and meta-analysis of the available literature.
METHODS
The present review aimed to address the following key clinical questions regarding the use of systemic therapy as conversion or neoadjuvant treatment in patients with iCCA.
The first PICOTS concerns conversion therapy: Do patients who undergo resection after conversion therapy achieve a survival benefit compared with those treated with systemic therapy alone?
In details:
P (Patients): Patients with unresectable iCCA
I (Intervention): Conversion strategy
C (Comparison): Systemic therapy
O (Outcomes): overall survival (OS)
T (Timing): Outcomes at 12, 36, and 60 months
S (Study design): Systematic review of the available original articles and meta-analysis of results.
The second PICOTS concerns neoadjuvant therapy: Does neoadjuvant therapy followed by surgery provide any survival benefit compared with upfront surgery?
In details:
P (Patients): Patients with resectable iCCA
I (Intervention): Neoadjuvant therapy
C (Comparison): Upfront surgery
O (Outcomes): OS
T (Timing): Outcomes at 12, 36, and 60 months
S (Study design): Systematic review of the available original articles and meta-analysis of results.
In patients undergoing conversion therapy, the primary endpoints were to assess the secondary resectability rate of initially unresectable cases and their oncological outcomes after surgery compared with patients receiving palliative systemic therapy. In patients receiving neoadjuvant therapy, the primary endpoints were the dropout rate during preoperative systemic therapy and the oncological outcomes of resected patients after systemic therapy compared with those undergoing upfront surgery. In both groups, the secondary endpoints included the radiological and pathological response to systemic therapy, and the 90-day postoperative outcomes of liver resection in pretreated patients.
Literature search and inclusion criteria
A systematic literature review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (checklist available as Supplementary Table 1). The research protocol was registered in the International Prospective Register of Systematic Reviews (PROSPERO; http://www.crd.york.ac.uk/PROSPERO) under the registration number CRD420251153620.
For this review, we included studies meeting the following criteria: (1) published in English; (2) involving patients with a primary diagnosis of iCCA confirmed by histology; (3) reporting the administration of conversion or neoadjuvant systemic therapy; and (4) providing data on secondary resection rate or post-resection outcomes. Studies were excluded if they: reported only palliative therapies outside of a conversion strategy; evaluated neoadjuvant or conversion approaches other than systemic therapy; did not report outcome data regarding conversion rates or long-term results; included patients with recurrent iCCA; or analyzed biliary tract cancers without presenting a specific subgroup analysis for iCCA. Studies including mixed populations (i.e., patients with recurrent disease, extrahepatic cholangiocarcinoma, or mixed preoperative treatments such as systemic therapy combined with other approaches) were considered eligible if data of the population of interest was separately reported or if other subgroups represented less than 30% of the total cohort.
The literature search was carried out in the PubMed database, restricted to studies conducted in humans and released between 01/01/2000 and 31/07/2025. The following search query was used: “(((conversion or downstaging or downsizing or neoadjuvant treatment or neoadjuvant therapy or initially unresectable) AND (cholangiocarcinoma or biliary cancer or biliary tumor or biliary tract cancer or biliary tract tumor)) AND (surgery or resection or hepatectomy) AND ((“2000/01/01”[Date - Publication]: “2025/07/31”[Date - Publication]))) AND (“english”[Language])”.
Study selection
Screening of the studies identified through the electronic search was performed using the Rayyan platform (www.rayyan.ai)[27]. The software automatically detected duplicates, which were subsequently verified and removed manually by one author (Viganò L). No additional automated tools were used. After duplicate removal, the titles and abstracts of the remaining articles were independently screened by four authors (Risi L, Cantamessa M, Pavia L, and Viganò L), with each article assessed by two reviewers. In case of disagreement, a third independent reviewer was consulted to reach a final decision. Full texts of the selected articles were then retrieved and independently evaluated for eligibility by two authors, with a third reviewer again involved in case of conflict. The reference lists of included studies were also screened for additional eligible articles.
All original studies were included, regardless of design (randomized controlled trials, cohort studies, case-control studies, or case series), provided that at least 10 patients were analyzed. Case reports, book chapters, editorials, previously published reviews or meta-analyses, conference proceedings, and consensus statements were excluded.
When multiple papers originated from the same institution or registry, the enrollment periods and inclusion criteria were carefully evaluated. If a patient overlap greater than 50% was identified, only the most recent or highest-quality study was included.
Data extraction
Two authors independently extracted and cross-checked relevant data from the selected articles. The following information was collected: (1) Article details: first author, corresponding author’s institution, year of publication, and journal; (2) Study design; (3) Cohort characteristics: enrolling institution(s), number of patients, enrollment period, inclusion and exclusion criteria, definitions of unresectability, borderline resectability, and high-risk status, age, and sex; (4) Tumor characteristics: number and size of lesions, nodal and metastatic status, staging, organ or vascular invasion, and carbohydrate antigen (CA) 19-9 levels; (5) Conversion or neoadjuvant therapy: type, regimen, duration, and associated treatments; (6) Therapy effectiveness: radiological and pathological response rates, conversion rate among initially unresectable patients, and dropout rate among initially resectable patients; (7) Liver resection: procedure performed (extent and associated procedures), postoperative mortality, overall morbidity, and severe morbidity; (8) Long-term outcomes: OS and recurrence-free survival (RFS).
Quality assessment
Two authors (Pavia L and Viganò L) independently assessed the quality of the comparative studies using the Newcastle-Ottawa Scale (NOS)[28]. In the event of any discrepancies, a consensus was reached through discussion with a third author (Risi L).
Statistical analysis
Categorical variables were reported as numbers and percentages, while continuous variables were reported as medians with interquartile ranges (IQR) or means with standard deviation. A meta-analysis of aggregated data was performed for dichotomous variables, and an individual patient-data meta-analysis was performed for survival outcomes. Heterogeneity across studies was evaluated using the Cochrane Q test, and the I2 statistic was used to quantify the proportion of total variability attributable to between-study differences. I2 values of 25%, 50%, and 75% were considered to indicate low, moderate, and high heterogeneity, respectively[29]. For binary outcomes (i.e., conversion rate, R0 resection rate, radiological and pathological response, severe morbidity, and mortality), a descriptive meta-analysis was conducted to present pooled estimates of variables of interest. No comparative meta-analysis was performed due to the limited availability of comparative data. A random-effects model was applied using the Freeman-Tukey double arcsine transformation for single proportions[30,31]. For the individual patient-data meta-analysis, the primary objective was to obtain pooled survival estimates for each treatment group (systemic therapy, conversion strategy, neoadjuvant treatment, and upfront surgery), and the secondary objective was to compare survival outcomes among them. Survival data were extracted from published Kaplan-Meier curves using DigitizeIt software, or from patient-level time-to-event data when available. The algorithm by Guyot et al. was applied to reconstruct patient-level survival data, estimating time-to-event parameters while maintaining the monotonicity constraint[32-34]. For each study, the reconstructed survival data were reviewed by comparing them with the original published data. A one-stage meta-analysis was conducted using Cox proportional hazards models with a gamma shared frailty term to account for baseline heterogeneity among studies or groups due to unmeasured covariates[35,36]. The proportional hazards assumption was assessed visually by comparing predicted vs. observed survival functions, and analytically by examining scaled Schoenfeld residuals and performing the Grambsch-Therneau test. The difference and ratio in restricted mean survival time (RMST) between groups were then estimated as a nonparametric measure of survival comparison[37,38]. Finally, hazard ratios (HR) and confidence intervals (CI) were obtained for each study using reconstructed individual patient datasets, and a pooled HR was calculated through a conventional two-step meta-analysis[35].
A sensitivity analysis was performed on a selected group of studies, separately evaluating the conversion therapy and neoadjuvant therapy groups. Two conditions were explored: (1) studies with a NOS score ≥ 6; and (2) studies adopting a homogeneous definition of unresectable or high-risk iCCA. For the latter criterion, the following definitions were applied: in the conversion therapy group, studies defining unresectability based on hepatic involvement, vascular and/or biliary involvement, and nodal status; in the neoadjuvant therapy group, studies providing detailed information on the selection criteria for high-risk patients.
P-values less than 0.05 were considered statistically significant for all tests. Publication bias was assessed visually by funnel plots and by using the Egger test to assess for a small-study effect. Statistical analysis was conducted using STATA version SE 19 (StataCorp, College Station, Texas, USA).
RESULTS
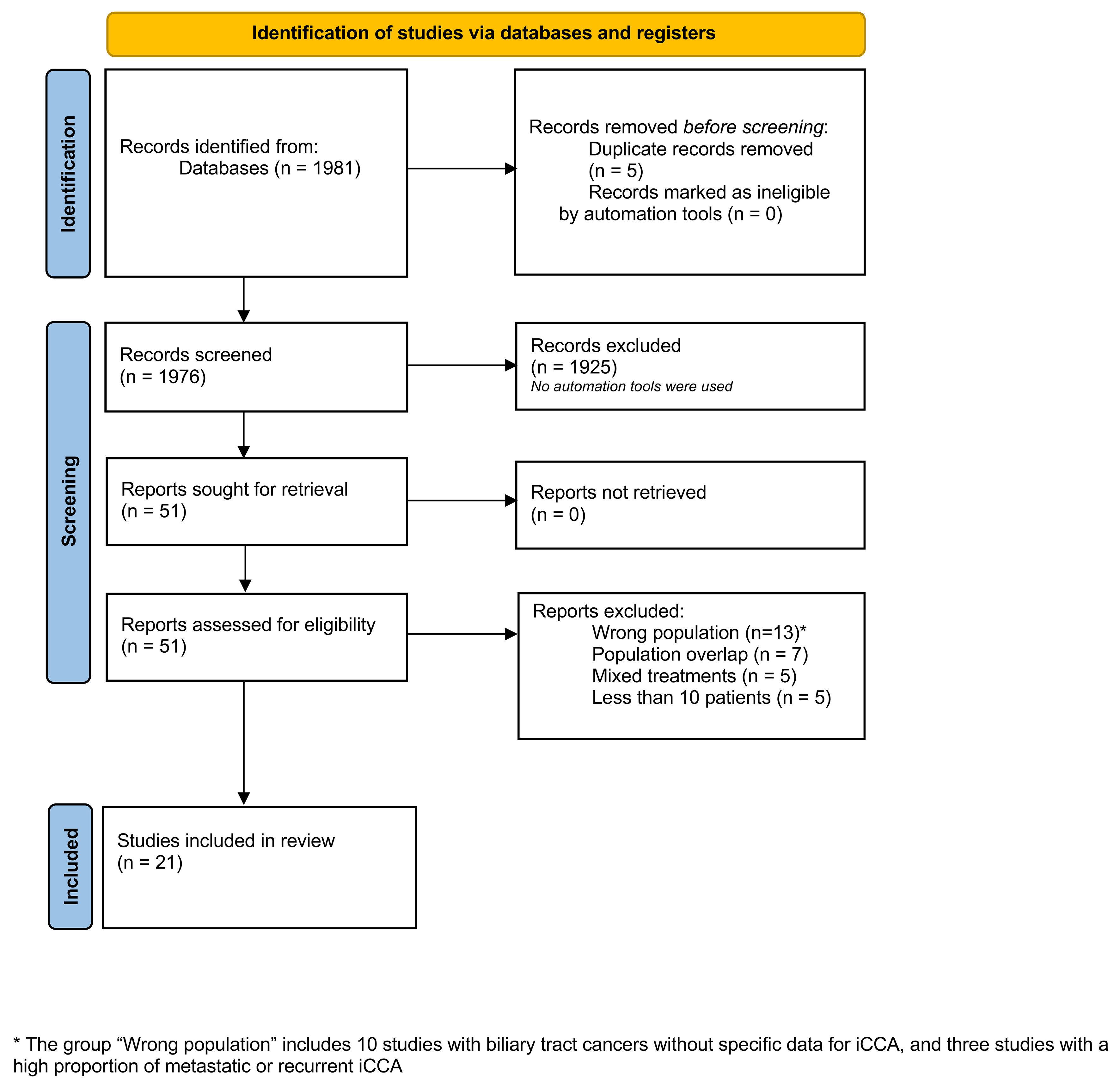
Online search conducted in PubMed database identified 1,981 papers. Initial screening led to the exclusion of five duplicates and 1,925 articles based on title and abstract evaluation. The full texts of the remaining 51 papers were retrieved and analyzed, of which 21 were retained for the present analysis[39-59]. The selection process is summarized in Figure 1.
Figure 1. PRISMA 2020 flowchart summarizing the selection process. iCCA: Intrahepatic cholangiocarcinoma; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
The characteristics of the included papers are reported in Table 1. All studies except three were published since 2020, and six (29%) were multicenter. The papers included one randomized controlled trial (gemcitabine, cisplatin, and S-1 chemotherapy vs. gemcitabine and cisplatin in a conversion setting), two prospective series, and 18 retrospective analyses. Three studies, all comparing neoadjuvant therapy followed by surgery with upfront surgery, used propensity score matching analysis[44,51,58].
Characteristics of the included studies
| Author | Year | Study design | Setting | Enrolment period | Number of patients | ITT |
| Conversion therapy | ||||||
| Dong et al.[39] | 2025 | Retrospective | Monocentric | 2019-2024 | 57 | yes |
| Garita et al.[45] | 2025 | Retrospective | Monocentric | 2015-2022 | 23 | yes |
| Wu et al.[59] | 2025 | Retrospective | Monocentric | 2019-2023 | 151 | no |
| Murakami et al.[52] | 2025 | Retrospective | Monocentric | 2007-2018 | 35 | yes |
| Kosaka et al.[48] | 2025 | Retrospective | Monocentric | 2018-2022 | 23 | yes |
| Nakamura et al.[40] | 2023 | RCT | Multicentric | 2014-2016 | 77 | yes |
| Choi et al.[43] | 2023 | Retrospective | Monocentric | 2019-2021 | 34 | yes |
| Nooijen et al.[53] | 2021 | Retrospective | Monocentric | 2016- 2019 | 13 | yes |
| Wang et al.[57] | 2021 | Retrospective | Monocentric | 2013-2020 | 28 | yes |
| Riby et al.[54] | 2020 | Retrospective | Monocentric | 1997-2017 | 13 | no |
| Le Roy et al.[49] | 2017 | Retrospective | Monocentric | 2000-2013 | 74 | yes |
| Konstantinidis et al.[47] | 2016 | Retrospective | Monocentric | 2000-2012 | 74 | yes |
| Kato et al.[46] | 2015 | Prospective | Monocentric | 2011-2014 | 25 | yes |
| Neoadjuvant therapy | ||||||
| Wehrle et al.[58] | 2024 | Retrospective | Multicentric | 2004-2019 | 364 | no |
| Van Treeck et al.[56] | 2024 | Retrospective | Monocentric | 2014-2020 | 47 | no |
| Maithel et al.[50] | 2023 | Prospective | Multicentric | 2018-2021 | 30 | yes |
| Alaimo et al.[41] | 2023 | Retrospective | Multicentric | 2010-2018 | 480 | no |
| Ayabe et al.[42] | 2023 | Retrospective | Monocentric | 2003-2019 | 45 | no |
| Choi et al.[44] | 2022 | Retrospective | Multicentric | 2014-2018 | 196 | no |
| Sutton et al.[55] | 2021 | Retrospective | Monocentric | 2004-2017 | 10 | no |
| Mason et al.[51] | 2021 | Retrospective | Multicentric | 2006-2016 | 607 | no |
Thirteen analyses addressed the outcomes of conversion therapy, while eight addressed the outcomes of neoadjuvant therapy. The distinction between conversion and neoadjuvant settings was based on the definition of iCCA as unresectable or resectable, the inclusion criteria that allowed classification of patients as resectable or unresectable, or the authors’ statements in the Methods section. Notably, one study combined the two groups (conversion and neoadjuvant) without reporting separate results. Because this study focused exclusively on resected patients and included a predominance of neoadjuvant over conversion cases, it was classified within the neoadjuvant group[42].
The median number of included patients per study was 45 (IQR 23-77), and their characteristics are summarized in Table 2.
Characteristics of the included patients
| Author | Number of patients | Age, years | Sex (F) | Number | Size (mm) | Ca 19-9 (U/mL) | T stage | N+ | M+ | Staging | ||||
| T0-2 | T3 | T4 | I | II | III | |||||||||
| Conversion therapy | ||||||||||||||
| Dong et al.[39] | 57 | 55†/59††* | 32 (45%)a | - | 67†/74††* | 40†/229††* | - | - | - | - | 15 (21%)a | 14 (10%)a | 19 (27%)a | 23 (32%)a |
| Garita et al.[45] | 23 | 64 (57-72)* | 11 (48%) | Multiple 16 (70%) | 70 (27-173)§ | 37 (2-26,960)§ | - | - | - | - | 0% | 0 (0%) | 5 (22%) | 18 (78%) |
| Wu et al.[59] | 151 | ≥ 65 years 51 (34%) | 50 (33%) | Multiple 48 (32%) | 50* | - | - | - | - | 51 (34%) | 8 (5%) | 28 (19%) | 123 (81%) | 0% |
| Murakami et al.[52] | 35 | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Kosaka et al.[48] | 23 | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Nakamura et al.[40] | 77 | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Choi et al.[43] | 34 | 68 (50-78)§ | 20 (59%) | - | - | 10 (3-5,546)§ | - | - | - | - | 0% | - | - | - |
| Nooijen et al.[53] | 13 | 62 ± 8‡ | 5 (38%) | - | 103 (64-120)* | 766 ± 1,251.6‡ | - | - | - | - | - | - | - | - |
| Wang et al.[57] | 28 | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Riby et al.[54] | 13 | 60 (47-71)§ | 5 (38%) | 1 (1-10)§ | 70 (30-120)§ | - | - | - | - | - | 0% | - | - | - |
| Le Roy et al.[49] | 74 | 60 (52-68)* | 34 (46%) | 3.7 ± 4‡ | 75 (57-100)* | 466 (50-1,156)* | - | - | - | 31/66b | 28 (38%) | - | - | - |
| Konstantinidis et al.[47] | 74 | - | - | - | - | - | - | - | - | - | 0% | - | - | - |
| Kato et al.[46] | 25 | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Neoadjuvant therapy | ||||||||||||||
| Wehrle et al.[58] | 364 | 60.1 ± 11.9‡ | 225 (62%) | - | 83 ± 79‡ | 49.1 ± 29.9‡ | 316 (87%) | 48 (13%) | 0% | 116 (32%) | 0% | - | - | - |
| Van Treeck et al.[56] | 47 | 62 (55-69)* | 26 (55%) | - | 69 (50-85)* | - | 44 (94%) | 0% | 3 (6%) | 19 (40%) | 0% | 7 (15%) | 19 (40%) | 21 (45%) |
| Maithel et al.[50] | 30 | 60.5 (39-78)# | 12 (40%) | 3.5 (2-7)* | 55 (30-74)*,c | 14.8 (3.8-258.8)* | - | - | - | 8/22 (36%) | 0% | - | - | - |
| Alaimo et al.[41] | 480 | ≥ 70 years 98 (20%) | 245 (51%) | - | - | - | 388 (81%) | 92 (19%) | 125 (26%) | 0% | - | - | - | |
| Ayabe et al.[42] | 45 | 61 (53-67)* | 26 (58%) | - | - | ≥ 35 U/mL 27 (60%) | - | - | - | - | 6 (13%) | - | 7 (16%) | 32 (71%) |
| Choi et al.[44] | 196 | 60.8 ± 11,3‡ | 114 (58%) | - | - | - | 137 (70%)d | 38 (19%)d | 34 (17%) | 5 (3%) | - | - | - | |
| Sutton et al.[55] | 10 | 58 (44-66)* | 7 (70%) | - | - | 62 (46-1,445)* | - | - | - | - | 0% | 3 (30%) | 3 (30%) | 4 (40%) |
| Mason et al.[51] | 607 | 59.2 ± 11,3‡ | 317 (52%) | - | > 50 mm 273 (45%)e | - | 444 (73%)e | 89 (15%)e | 22 (4%)e | 118 (19%) | 0% | - | - | - |
According to the NOS, the overall quality of comparative studies was low, with only two studies scoring 7 or higher [Supplementary Table 2].
Indications to treatment and regimens
Unresectability in conversion therapy group
Ten of the 13 studies provided definitions of unresectability, which are summarized in Table 3. Resectability was based on the following criteria: extent of hepatic involvement (n = 9 studies, including tumor size and location, and the presence of intrahepatic metastases); vascular involvement (n = 10 studies, mainly defined as bilateral and/or unreconstructable infiltration or encasement of the intrahepatic portal vein or hepatic artery, or infiltration of the three hepatic veins); biliary infiltration (n = 4 studies, defined as bilateral and unreconstructable); nodal status (n = 7 studies, including distant lymph-node metastases or extensive lymph node involvement); and liver function and volume (n = 8 studies). Additional criteria mentioned in single studies included infiltration of contiguous organs, presence of portal vein thrombosis, and CA 19-9 levels. Distant metastases were excluded in four studies, included in three, and not specified in six.
Inclusion criteria of studies with unresectable patients undergoing conversion systemic therapy
| Liver involvement | Vascular involvement | Biliary involvement | N status | LFT/FLR | Other criteria | ||||
| Details | Details | Details | |||||||
| Dong et al.[39] | √ † | √ | - | √ | Distant N+ | ||||
| Wu et al.[59] | √ # | √ | - | √ | - | √ | |||
| Murakami et al.[52] | √ * | √ | Unreconstructable HA/PV All three HVs involv | √ | Bilateral involv | √ | |||
| Kosaka et al.[48] | √ | √ | Unreconstructable | √ | Unreconstructable | √ | Extranodal ext.; Distant N+ | √ | Infiltration of contiguous organs |
| Choi et al.[43] | √ | Bilateral involv. of HA Unreconstructable PV | √ | Bilateral involv** | √ | Beyond N2*** | |||
| Nooijen et al.[53] | √ | √ | - | √ | Portal thrombosis | ||||
| Riby et al.[54] | √ # | √ | - | √ | - | √ | |||
| Le Roy et al.[49] | √ # | √ | - | √ | - | √ | Ca 19-9 | ||
| Kato et al.[46] | √ | √ | Unreconstructable HA/PV/HVs | √ | Unreconstructable | √ | |||
| Konstantinidis et al.[47] | √ † | √ | Unreconstructable | √ | Extended N+ | √ | |||
Indications to neoadjuvant therapy
The indication for neoadjuvant therapy was specified in three of the eight studies, one of which generically reported “high-risk patients”[56]. The remaining two studies provided a detailed description of the inclusion criteria [Table 4]. In the study by Maithel et al., patients were enrolled if they were M0 and had one of the following conditions: T-stage Ib or higher tumors; solitary lesion > 5 cm; multifocal tumors or satellite lesions confined to the same liver lobe as the dominant lesion; major vascular invasion; or suspicious or involved regional lymph nodes[50]. In the study by Ayabe et al., inclusion criteria were regional lymphadenopathy, multifocal disease or satellite lesions requiring extensive resection, or vascular involvement with either tumor thrombus or arterial encasement[42].
Selection criteria of studies including resectable patients undergoing neoadjuvant systemic therapy
| Solitary > 5 cm | Solitary + satellite nodules | Multiple | Invasion of | c/pN+ | Ca 19-9 (U/mL) | Expected R1 | ||||
| Vascular structures (T2) | Visceral peritoneum (T3) | Adjacent organs (T4) | Diaphragm | |||||||
| Studies included in the review | ||||||||||
| Maithel et al.[50] | √ | √ | √ | √ | √ | √ | √ | |||
| Ayabe et al.[42] | √ | √ | √* | √ | ||||||
| Ongoing trials | ||||||||||
| NCT06903273 | √ | √ | √ | √ | √ | √ | √ | ≥ 200 | ||
| NCT04989218 | √ | √ | √ | √ | √ | √ | √ | |||
| NCT06050252 | √ | √ | √ | √ | √ # | > 200 | ||||
| NCT05557578 | √ | √ | √ | √ | > 200 | |||||
| NCT04669496 | √ | √ | √ | √ | > 37‡ | |||||
| NCT04506281 | √ | √ | √ | √ | > 200 | |||||
| NCT06037980 | √ | √ | √ | √ | √ | √ ± | > 100 | √ | ||
| NCT06721286 | √ | √ | √ | √ | ||||||
Treatment details
Treatment regimens, number of cycles, and associated locoregional treatments are detailed in Table 5. In both the conversion and neoadjuvant settings, most patients received a gemcitabine - cisplatin-based regimen. Immunotherapy or targeted therapies were used in only a few studies in the conversion
Details of administered systemic therapy
| Author | Pts | Systemic therapy regimen | Number of cycles | Other treatment | |
| Conversion therapy | |||||
| Dong et al.[39] | 57 | Gemcitabine - Cisplatin - Nab-paclitaxel (n = 57) | 4 (4-5)*** | Y | SIRT (6%)* |
| Garita et al.[45] | 23 | Gemcitabine - Cisplatin (n = 20) Gemcitabine - Cisplatin - other (n = 3) | - | N | - |
| Wu et al.[59] | 151 | Gemcitabine - Oxaliplatin - Lenvatinib - antiPD1 (n = 151) | 6 to 8 | N | - |
| Murakami et al.[52] | 35 | Gemcitabine-based (n = 35) | - | - | - |
| Kosaka et al.[48] | 23 | Gemcitabine - Cisplatin + S1 (n = 23) | - | N | - |
| Nakamura et al.[40] | 77 | Gemcitabine - Cisplatin +/- S1 (number not detailed) | - | N | - |
| Choi et al.[43] | 34 | Gemcitabine - Cisplatin - Nab-paclitaxel (n = 34) | 6*** | N | - |
| Nooijen et al.[53] | 13 | Gemcitabine - Cisplatin (n = 12) Chemoradiotherapy (n = 1) | 6 (3-7.5)*** | Y | ChemoRT (8%) SIRT (8%) |
| Wang et al.[57] | 28 | Gemcitabine-based (n = 19) Immuno and/or targeted therapy (n = 5) 5-Fluorouracil (n = 4) | - | N | - |
| Riby et al.[54] | 13 | Gemcitabine - Oxaliplatin/Folfirinox/Cisplatin-based (number not detailed) | - | N | - |
| Le Roy et al.[49] | 74 | Gemcitabine - Oxaliplatin (n = 44) Gemcitabine - other (n = 4) Gemcitabine (n = 3) Other (n = 19, 5-Fluorouracil/Oxaliplatin/Irinotecan) | 6 (4-8)*** | Y | SIRT or HAI (5%) |
| Konstantinidis et al.[47] | 74 | Gemcitabine-based (n = 58) Other agents (n = 16) | - | N | - |
| Kato et al.[46] | 25 | Gemcitabine - Cisplatin (n = 25) | - | N | - |
| Neoadjuvant therapy | |||||
| Wehrle et al.[58] | 364 | Multi-agent chemotherapy (n = 364) | - | Y | RT (12%) |
| Van Treeck et al.[56] | 47 | Gemcitabine - Cisplatin (n = 30)** Gemcitabine - Cisplatin - Nab-paclitaxel (n = 5) Gemcitabine - Oxaliplatin/Carboplatin (n = 5) FOLFOX / CAPOX (n = 4) Other (n = 4) | 4 (3-6)*** | N | - |
| Maithel et al.[50] | 30 | Gemcitabine - Cisplatin - Nab-paclitaxel (n = 30) | - | N | - |
| Alaimo et al.[41] | 480 | Multi-agent chemotherapy (n = 368) | - | N | - |
| Ayabe et al.[42] | 45 | Gemcitabine - Cisplatin (n = 25) Gemcitabine - Cisplatin - Nab-paclitaxel (n = 10) FOLFOX (n = 3) Gemcitabine - Oxaliplatin/Carboplatin (n = 3) Gemcitabine - Capecitabine (n = 2) Gemcitabine - Irinotecan (n = 1) Gemcitabine - Oxaliplatin - Adriamycin (n = 1) | 6 (4-8)*** | N | - |
| Choi et al.[44] | 196 | - | - | Y | HAI (4%) HAI + local ablation (0.5%) |
| Sutton et al.[55] | 10 | Gemcitabine - Platinum-based (n = 9) FOLFOX (n = 1) | 14 weeks (4-20)**** | N | |
| Mason et al.[51] | 607 | Multi-agent chemotherapy (n = 327) Single-agent chemotherapy (n = 72) RT/ChemoRT (n = 172) | - | Y | RT/ChemoRT (28%) |
Effectiveness of conversion therapy
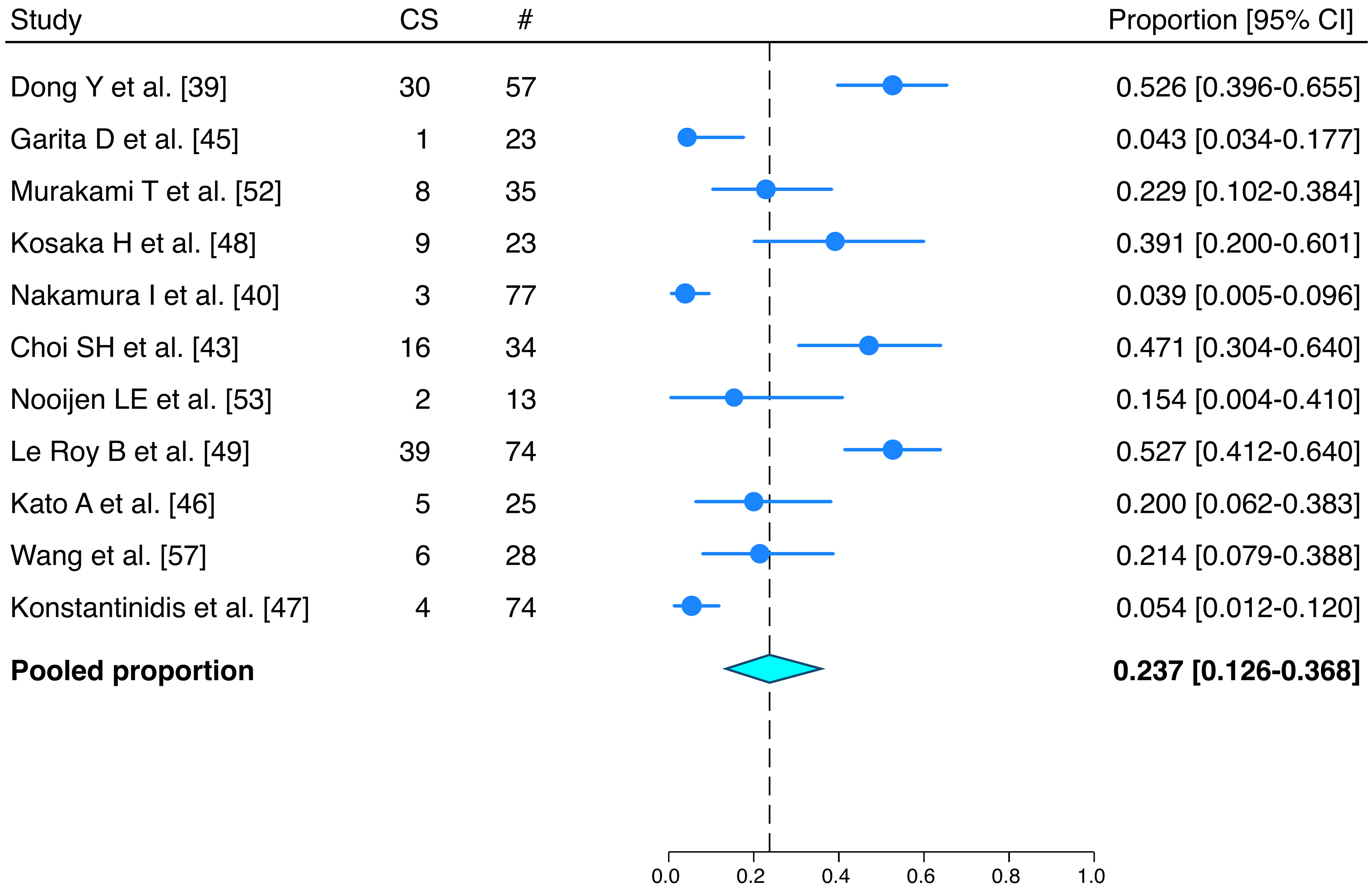
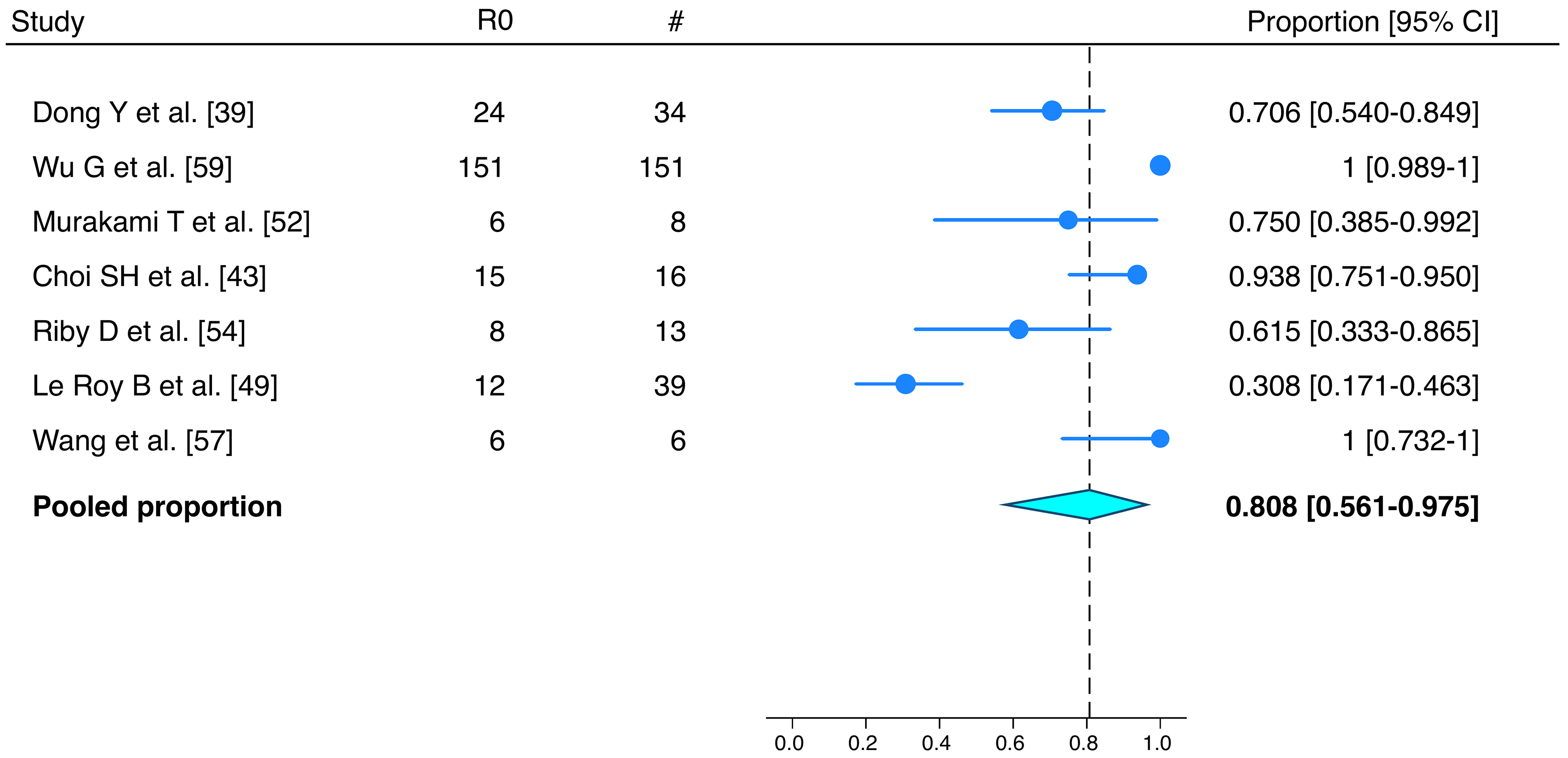
Of the 13 studies analyzing conversion therapy, 11 adopted an intention-to-treat design, including the entire population of treated patients, while two[54,59] focused only on those who underwent resection [Table 1]. In the meta-analysis, the pooled conversion rate was 23.7% (95%CI: 12.6%-36.8%) [Figure 2]. Among resected patients, the completeness of surgery was reported in seven studies[39,43,49,52,54,57,50], with a pooled R0 resection rate of 80.8% (95%CI: 56.1%-97.5%) [Figure 3]. The complete pathological response rate was reported in three studies[39,43,59], with a pooled rate of 1.9% (95%CI: < 0.1%-8.2%).
Figure 2. Forest plot of the meta-analysis of conversion rates. CS: Conversion surgery; CI: confidence intervals.
Survival of secondarily resected patients
Six studies reported OS from diagnosis in patients who underwent conversion surgery[39,40,49,52,57,59]. One study[59] was excluded from the individual patient-data meta-analysis because it did not provide either the number at risk or the number of events. For OS rates in the palliative chemotherapy group (non-conversion), three studies[39,49,56] were included in the meta-analysis. Among these, two provided a direct comparison with converted patients[39,49], both suggesting higher survival in the conversion surgery group. However, no comparative meta-analysis between converted and non-converted patients was performed due to the limited number of comparative studies and patients.
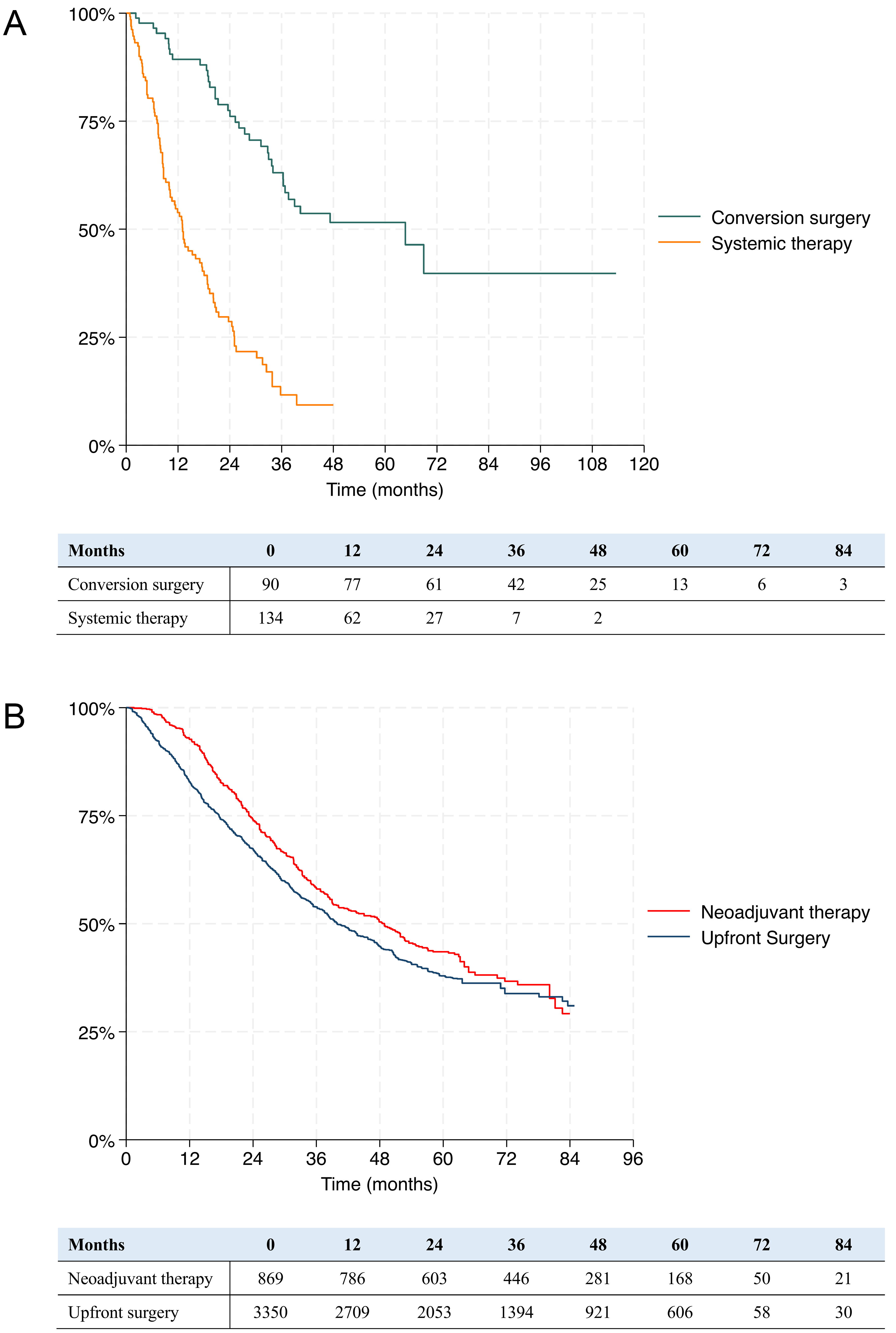
In the descriptive meta-analysis, the conversion surgery group (n = 90 patients) had 1-, 3-, and 5-year OS rates from diagnosis of 89.7% (95%CI: 81.1%-94.5%), 64.2% (95%CI: 52.3%-73.9%), and 52.5% (95%CI: 40%-63.5%), respectively. The 134 patients receiving palliative systemic therapy had 1- and 3-year OS rates of 53.9% (95%CI: 44.5%-62.3%) and 11.7% (95%CI: 5.6%-20.2%), respectively. The median survival time was 64.7 months after conversion surgery and 13.1 months after palliative systemic therapy. Survival curves are shown in Figure 4A.
Figure 4. Kaplan-Meier curve for cumulative overall survival in (A) resected and unresected patients after conversion therapy, and in (B) neoadjuvant and upfront surgery group (P < 0.001, Wilcoxon test).
Five studies reported RFS after conversion surgery[39,40,49,54,57]. In the descriptive meta-analysis (n = 94 patients), the 1-, 3-, and 5-year RFS rates were 61.5% (95%CI: 50.3%-70.9%), 33.5% (95%CI: 22.9%-44.3%), and 29.5% (95%CI: 19.2%-40.6%), respectively. The median RFS time was 17.8 months.
Additional survival data have been reported. Le Roy et al. compared patients undergoing conversion surgery with those receiving upfront surgery and found similar median OS (24.1 months vs. 25.7 months)[49]. Wu et al. demonstrated that both OS and RFS after conversion surgery are associated with pathological tumor response[59].
Effectiveness of neoadjuvant therapy
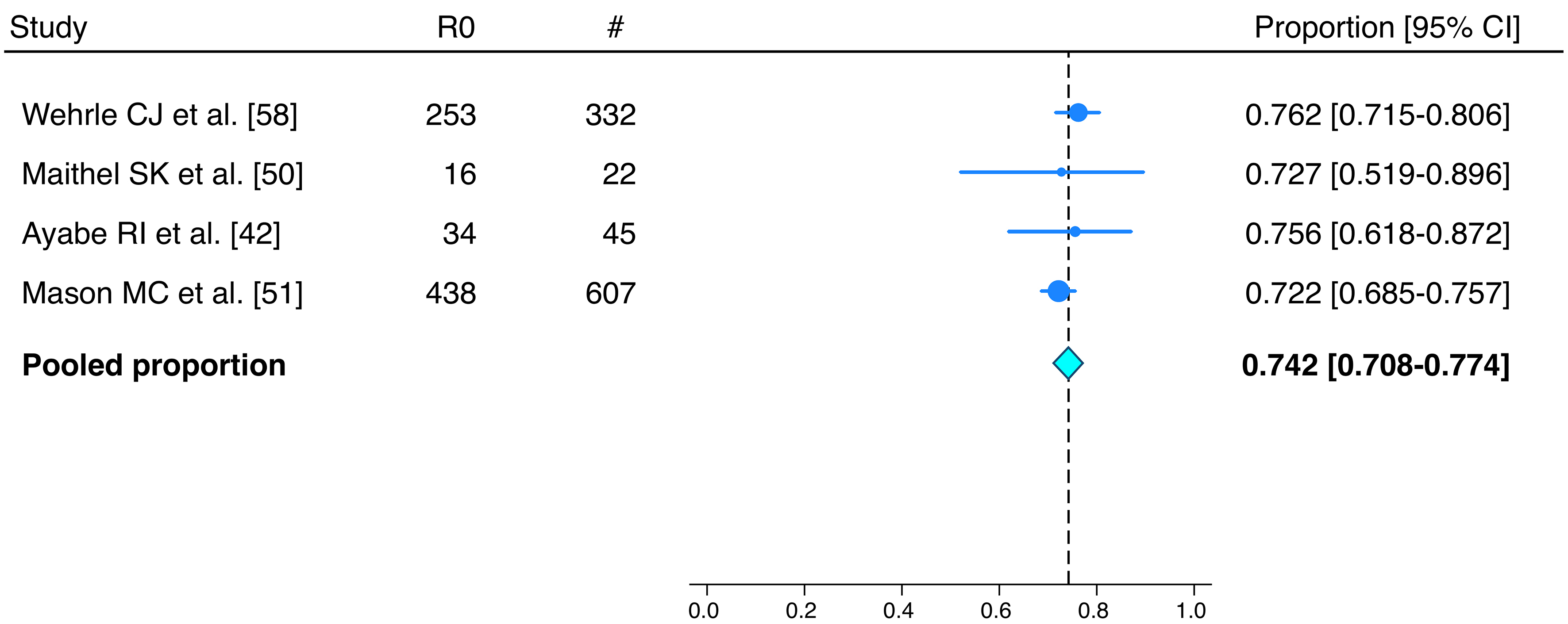
Only the prospective study by Maithel et al. had an intention-to-treat design and reported that approximately one-fourth of patients did not undergo surgery (7 of 29): five experienced preoperative disease progression on imaging, and two were found to have unresectable disease at exploration[50]. Of the eight studies analyzed, only two reported the radiological response rate according to the Response Evaluation Criteria in Solid Tumors (RECIST) criteria, with the following results: partial response in 24.5% of cases (95%CI: 11.7%-39.8%), stable disease in 65.3% (95%CI: 49.2%-79.9%), and disease progression in 10.0% (95%CI: 1.4%-21.5%)[50,55]. Downstaging was reported in two studies, with rates of 17.7% in the series by Alaimo et al. and 46.8% in the series by Van Treeck et al.[41,56]. After surgery, completeness of resection was reported in four studies[42,50,51,58], with a pooled R0 resection rate of 74.2% (95%CI: 70.8%-77.4%) [Figure 5]. The complete pathological response rate was reported in four studies[42,50,55,56], with a pooled rate of 0.7% (95%CI: < 0.1%-4.4%).
Does neoadjuvant treatment provide a survival benefit over upfront surgery?
All studies except one[44] analyzed OS rates, from diagnosis in most cases. Of these, five[41,50,55,56,58] compared the outcomes of patients receiving neoadjuvant chemotherapy followed by surgery with those of upfront surgery, but only four were included in the individual patient-data meta-analysis, as one study[50] did not provide either the number at risk or the number of events. The 869 patients who underwent surgery after neoadjuvant treatment had 1-, 3-, and 5-year OS rates from diagnosis of 92.7% (95%CI: 90.7%-94.2%), 58.2% (95%CI: 54.7%-61.5%), and 43.5% (95%CI: 39.7%-47.2%), respectively. Patients who underwent upfront surgery in the survival meta-analysis were 3,350 and they showed corresponding survival rates of 82.8% (95%CI: 81.5%-84.1%), 53.9% (95%CI: 52.1%-55.6%), and 37.1% (95%CI: 35.1%-39.1%), respectively, which were statistically significantly lower (P < 0.001). Survival curves are shown in Figure 4B.
In the one-stage meta-analysis using Cox proportional hazards models, neoadjuvant systemic therapy followed by surgery was associated with significantly improved OS from diagnosis compared with upfront surgery (HR = 0.833, 95%CI: 0.751-0.923; P < 0.001). Neoadjuvant systemic therapy was also associated with a significant increase of 3.6 months of RMST at five years (95%CI: 2.1-5.2; P < 0.001) which translates into a relative increase of life expectancy of 9.6% (95%CI: P < 0.001). In the two-stage meta-analysis the pooled HR was 0.845 (95%CI: 0.729-0.981; P = 0.027).
Only one study reported the OS rates after surgery, showing no significant difference between the two groups (median OS: 48.3 months with neoadjuvant treatment vs. 38.8 months without; P = 0.160)[58]. RFS data were reported in four studies[42,50,55,56]; however, no meta-analysis was performed due to heterogeneity and incomplete data. Median RFS from surgery was 7.1 months[50], 11 months[42], and 13 months[55], respectively, whereas Van Treeck et al. reported a median RFS of 23 months from diagnosis[56]. Only two analyses compared neoadjuvant and upfront surgery groups, and both found no significant differences in RFS: HR = 1.20 (95%CI: 0.60-2.41; P = 0.609, RFS from diagnosis) in Van Treeck et al., and HR = 0.54 (95%CI: 0.18-1.62; P = 0.27, RFS from surgery at three years) in Sutton et al.[55,56].
Additional benefits of neoadjuvant therapy
Other outcomes beyond survival have also been explored. Comparing patients with and without neoadjuvant systemic therapy, Wehrle et al. reported that the former group had a higher number of retrieved lymph nodes (OR = 1.26, 95%CI: 1.09-1.88) and a lower number of positive nodes (OR = 0.67, 95%CI: 0.49-0.93)[58]. Van Treeck et al. hypothesized that an objective response to therapy could facilitate surgery[56]; however, the R0 resection rate did not differ between patients with and without preoperative chemotherapy in the two studies addressing this issue (75.3% vs. 76.5%, OR = 0.91, Wehrle et al.; OR for R1 resection 1.49, 95%CI: 0.36-6.18; P = 0.58, Sutton et al.)[55,58].
Two studies, both excluded from review because of population overlap, compared the impact of neoadjuvant vs. adjuvant treatment and reported conflicting results[60,61]. Parente et al., in a series of 399 patients, found no survival difference between the two approaches, whereas Yadav et al. observed a significant survival benefit for neoadjuvant over adjuvant therapy (median OS: 40.3 months vs. 32.8 months, HR = 0.78, 95%CI: 0.64-0.94, P = 0.01)[60,61].
Short-term results after preoperative systemic therapy
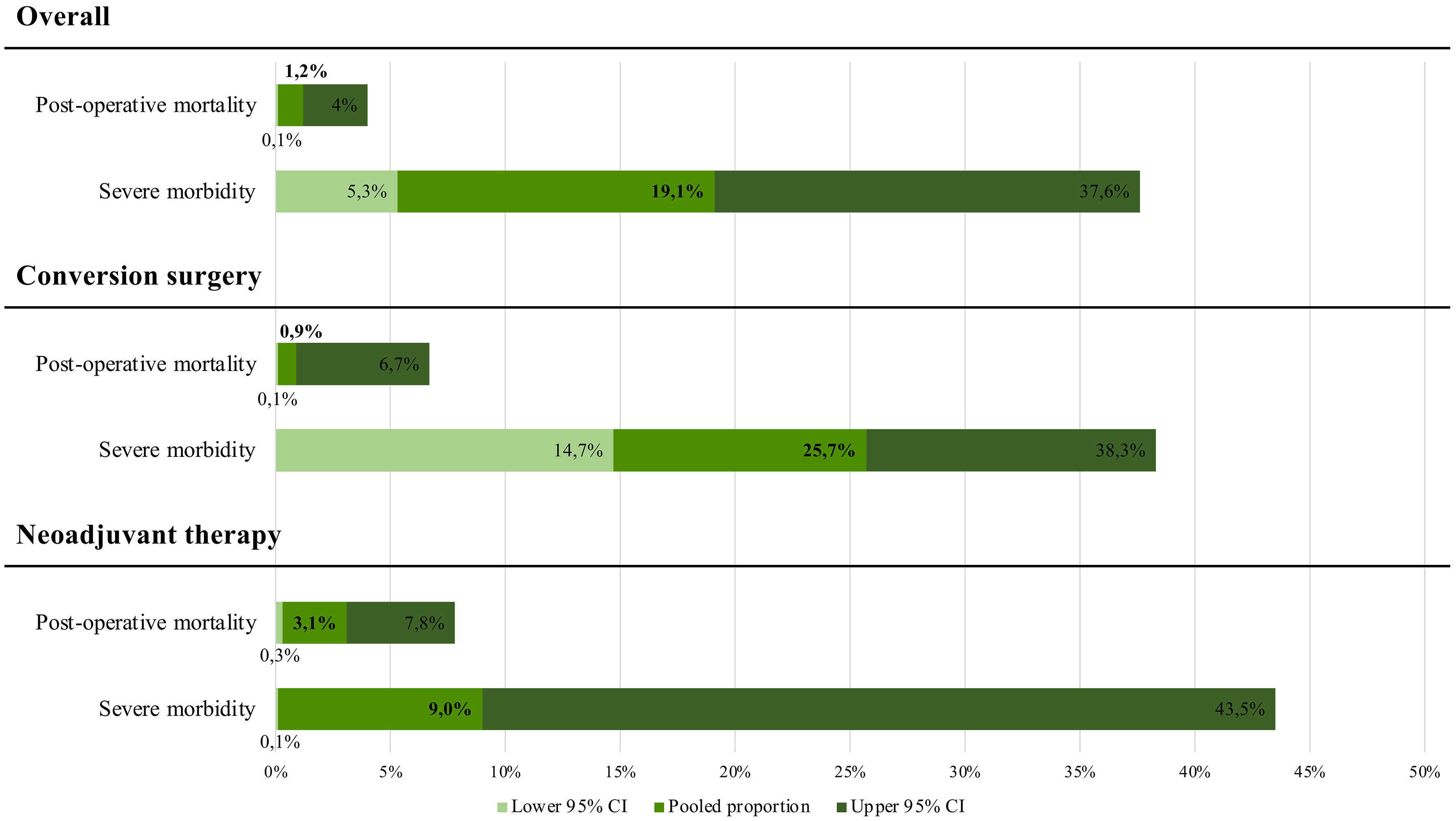
Postoperative outcomes were analyzed in nine studies (seven in the conversion group and two in the neoadjuvant group). Mortality was defined at 90 days after surgery in eight studies[39,40,48-50,52,54,57], and at 30 days in one study[44]. The pooled postoperative mortality rate was 1.2% (95%CI: < 0.1%-4%), being 0.9% (95%CI: < 0.1%-6.7%) in the conversion group and 3.1% (95%CI: 0.3%-7.8%) in the neoadjuvant group. Severe morbidity rates were reported by five studies (three conversion and two neoadjuvant)[44,49,50,52,54], with a pooled rate of 19.1% (95%CI: 5.3%-37.6%), being 25.7% (95%CI: 14.7%-38.3%) in the conversion group and 9% (95%CI: < 0.1%-43.5%) in the neoadjuvant group. Results of the descriptive meta-analysis for short-term outcomes are shown in Figure 6.
Figure 6. Pooled results of post-operative outcomes (mortality and severe morbidity) overall and stratified by conversion and upfront surgery groups. CI: Confidence intervals.
Two studies compared postoperative outcomes between upfront surgery and surgery following conversion therapy[49,54]. Overall morbidity, severe morbidity, and mortality rates were similar between groups, with the only exception being a higher incidence of pulmonary complications in the chemotherapy group reported by Riby et al. (23% vs. 4%, P = 0.04)[54].
Choi et al. specifically compared operative risks in patients with and without neoadjuvant chemotherapy using a propensity score-matched analysis[44]. They found that neoadjuvant therapy was not associated with an increased risk of postoperative mortality or morbidity; however, it was correlated with greater blood loss requiring transfusion (43.2% vs. 29.4%, P = 0.001), more frequent use of the Pringle maneuver (33.9% vs. 25.0%, P = 0.03), and longer operative time (345.4 min vs. 314.8 min, P = 0.02). Similarly, Sutton et al. and Wehrle et al. found no increase in severe morbidity after neoadjuvant therapy, although Wehrle et al. reported prolonged hospitalization in treated patients (OR for no chemotherapy, 0.64; 95%CI: 0.52-0.79)[55,58].
Sensitivity analysis
Details of the sensitivity analysis are summarized in Supplementary Table 3. Considering articles with a NOS score ≥ 6, one study was included in the conversion therapy group[40] and four in the neoadjuvant therapy group[44,51,55,58]. Meta-analysis was feasible only for R0 resection rate and OS in the neoadjuvant therapy group. In both analyses, results were consistent with the global analysis with pooled R0 resection rate being 73.9% and pooled 5-year OS being 47.7% for neoadjuvant therapy vs. 34.3% for upfront surgery (P = 0.002).
When restricting the analysis to studies with a homogeneous definition of unresectability, six studies were included[39,47-49,54,59]. The pooled conversion rate was 35.0%, higher than in the global analysis (23.7%). Pooled rates of R0 resection (72.3%), pathological complete response (2.8%), postoperative mortality (1.5%), and severe morbidity (22.7%) were comparable to the global analysis. Survival outcomes were also similar, with a pooled 3-year OS of 62.1% for patients undergoing conversion surgery vs. 14.7% for unresected patients, and a pooled 3-year RFS of 34.6% among resected patients.
Considering the analyses adopting a homogeneous definition of high-risk iCCA, two studies were included[42,50]. Meta-analysis was feasible only for the R0 resection and pathological complete response rates, both consistent with the global analysis (74.7% and 2.3%, respectively); survival outcomes could not be pooled.
Publication bias
Funnel plots constructed for each score showed symmetry and the Egger test did not show a possible small-study effect. This suggested that publication bias was not substantial and unlikely to impact any conclusions.
DISCUSSION
The present study, by pooling available evidence through rigorous methodology, demonstrates that systemic therapy is central to a biology-driven approach in the management of patients with iCCA, without adversely affecting postoperative recovery. In selected patients with initially unresectable disease, systemic therapy can achieve conversion to resectability in approximately one-fourth of cases, resulting in excellent long-term survival rates approaching those of patients who are upfront resectable. In high-risk resectable patients, neoadjuvant treatment was associated with superior survival compared with upfront surgery when the treatment strategy was successfully completed, although a dropout rate exceeding 20% and potential biases in the available studies warrant careful consideration.
Understanding tumor biology is fundamental to a true precision-medicine approach. Surgical oncologists must shift their focus from technical to oncologic resectability, identifying which patients, and at what time, truly benefit from surgery[22,25]. In this context, collaboration between surgical and medical oncologists within hepatobiliary multidisciplinary teams has intensified, driven by the increasing aggressiveness of surgical approaches and the growing effectiveness of modern systemic therapies[5-7,16,17,62,63]. Conversion and neoadjuvant therapies are now routinely considered in the management of liver malignancies, although evidence supporting their specific benefits in iCCA remains limited.
Before discussing the outcomes of onco-surgical strategies, a preliminary comment is needed regarding the definitions of unresectable and high-risk iCCA. Despite ongoing efforts to reach a consensus, substantial heterogeneity persists in liver disease resectability criteria[64]. Interestingly, some consistency emerged among the analyzed studies, with common criteria for unresectability including bilateral vascular and/or biliary involvement. Some authors more precisely defined the limit as the technical unreconstructability of hepatic vessels or bile ducts[43,46-48,52]. Choi et al. specifically defined the limit for biliary reconstruction as bilateral tumor extension beyond the borders of the respective portal veins[43]. Murakami et al. proposed a definition of unresectability as tumor extension more distal than 10 mm from both the U point (the inflection between the umbilical portion and transverse sections of the left portal vein) and the P point (the junction between the anterior and posterior branches of the right portal vein)[52]. Another consistent criterion was an insufficient future liver remnant, although liver hypertrophy techniques may be applied to overcome this limitation[65]. Some agreement was also observed regarding distant or extensive lymph node involvement and intrahepatic metastases, although for the latter, the distinction between multifocal and metastatic disease remains blurred. On the contrary, identifying high-risk patients within the technically resectable group, and thus defining appropriate indications for neoadjuvant therapy, remains far less well established. Only two of the eight studies provided explicit definitions[42,50], highlighting substantial uncertainty, with N+ tumors and multifocal disease being the most commonly shared indications. However, when considering ongoing trials, such uncertainty appears to be more theoretical than practical. Indeed, current studies evaluating neoadjuvant systemic therapy for resectable iCCA adopt fairly homogeneous inclusion criteria [Table 4], based on risk factors for early postoperative recurrence and established prognostic factors[18-21,66,67]. De facto, all studies included patients with a solitary tumor > 5 cm, multiple tumors, tumors with vascular invasion, or confirmed or suspected nodal involvement. Consequently, if these studies yield positive results, the indication for neoadjuvant therapy will be established. Debate remains regarding the assessment of satellite nodules, the CA 19-9 cutoff value, and the likelihood of achieving a complete resection. Although this latter criterion has been included in only one trial (NCT06037980), we consider R0 resection mandatory and believe that, when it cannot be reasonably ensured, this should constitute an indication for preoperative treatment.
Most iCCA patients are unresectable at diagnosis, and systemic therapy represents their only potential treatment[1]; surgery can be considered secondarily if sufficient tumor shrinkage or downstaging occurs. The concept of conversion therapy in hepatic tumors, initially introduced by Bismuth et al. in 1996 for colorectal liver metastases[68], has become a cornerstone in liver oncology, shifting secondary resectability from an occasional outcome to a defined treatment goal. Several conversion strategies for iCCA have been explored, often including radioembolization or intra-arterial treatments[69-71]. The present analysis focused exclusively on systemic therapies and confirmed that, in selected patient cohorts, secondary resectability can be achieved in approximately one-fourth of cases (even up to one-third of cases in studies that explicitly detailed resectability criteria), with excellent R0 resection rates, exceeding 80%. Even if conversion rates varied across studies, patients who achieved secondary resection consistently showed excellent survival outcomes, with 5-year survival rates approaching those of upfront resectable patients and markedly exceeding those of unresected cases. With the advent of modern systemic treatments, including immunotherapy and targeted agents[5-7], response rates are expected to improve further increasing the number of converted patients.
The present meta-analysis highlighted several relevant points. The main finding favored neoadjuvant therapy: patients who completed the full treatment sequence (preoperative therapy followed by surgery) achieved higher OS from diagnosis than those undergoing upfront surgery (44% vs. 37% at five years), corresponding to an expected survival gain of approximately 3.6 months. Additionally, Wehrle et al. reported a lower number of positive lymph nodes in patients receiving preoperative treatment[58]. However, these positive outcomes should be interpreted with caution for at least two reasons. First, neoadjuvant therapy did not result in higher OS or RFS from surgery, nor in increased R0 resection rates. Second, all comparative studies analyzing OS from diagnosis included only resected patients, excluding those who dropped out from the neoadjuvant arm, thereby introducing a potential immortal time bias. Radiological disease progression occurred in approximately 10% of patients, consistent with rates generally reported and accepted for colorectal liver metastases[72]. However, the overall dropout rate reported by the only available intention-to-treat study exceeded 20% (7/29)[50]. Such a high dropout rate suggests that the survival benefit observed in the neoadjuvant group compared with upfront surgery could be substantially attenuated in an intention-to-treat perspective and raises concerns about the potential loss of a curative opportunity in patients who were initially resectable. Ongoing trials will answer these questions, but some considerations can already be made. The high dropout risk could be perceived differently when evaluated in the light of the previously mentioned 15%-20% risk of futile surgery after upfront resection due to very-early recurrence, a risk that may be even higher in patients with negative prognostic factors selected for neoadjuvant strategies[18-21]. Would neoadjuvant therapy be considered a failure if it achieves survival similar to upfront surgery, given that it would spare a futile resection in approximately one-fifth of patients? Furthermore, the dropout risk is expected to decrease with the adoption of immune checkpoint inhibitors and targeted therapies, which are associated with higher response rates compared with standard chemotherapy[5-7].
A further point of interest is the potential negative impact of preoperative therapy on postoperative outcomes. Early reports in patients with colorectal liver metastases undergoing hepatectomy after systemic therapy indicated higher blood loss, increased risk of liver failure, and even higher mortality compared with untreated patients[73,74]. However, surgeons have learned to manage chemotherapy-related parenchymal injuries by adopting an adequate chemotherapy-to-surgery interval and by setting a higher cut-off value for the minimal volume of the future liver remnant[75]. The experience accumulated in metastatic patients has been valuable for iCCA. Indeed, the present study confirmed low mortality rates and acceptable rates of severe morbidity after surgery in patients receiving preoperative chemotherapy, with minimal impact. Initially unresectable patients experienced slightly higher severe morbidity rates, but this is more likely attributable to the complexity of surgery rather than systemic treatment. Some concerns remain regarding increased blood loss, which warrant further investigation. In patients with colorectal liver metastases, chemotherapy has been associated with fragile livers, which may lead to increased intraoperative complexity and a higher bleeding risk[75]. In iCCA patients, the scenario may be even more critical, as chemotherapy-related liver injury can be associated with pre-existing underlying fibrosis or cirrhosis, and tumors often present as large, deep-located lesions.
The present analysis is clinically relevant because it addresses several open questions. First, it highlights the importance of defining oncological resectability in iCCA, which is crucial for optimizing outcomes beyond purely technical considerations. Despite the heterogeneity of the literature, a relatively consistent definition of unresectability emerged, along with concordant characteristics of candidates for neoadjuvant therapy. Second, it clarifies the outcomes of standard systemic chemotherapy, highlighting both established evidence and existing knowledge gaps. Although no data are currently available for immunotherapy and targeted therapies, this should not be considered a major limitation, as not all ongoing neoadjuvant trials include immunotherapy and targeted therapies are still considered second-line options. The results reported here provide an essential reference for comparisons with future treatment strategies. Third, the favorable results observed in the conversion setting - both in terms of conversion rate and post-resection survival - support the development of a systematic approach aimed at achieving secondary resectability. Finally, the benefits of neoadjuvant treatment in resectable patients, together with the absence of any negative impact on postoperative outcomes, support this strategy whenever the advantage of upfront surgery is highly uncertain.
Some limitations should be acknowledged. First, most of the included studies were retrospective and had major limitations in design. Among conversion analyses, most were exploratory and did not include a well-designed comparison with other populations; in the neoadjuvant setting, only one had an intention-to-treat design, limiting a comprehensive assessment of the true impact of systemic therapy. No study explored the predictive factors of dropout, which could be valuable when scheduling a neoadjuvant therapy. Second, inclusion criteria were heterogeneous or, in some cases, not clearly defined, making direct comparisons challenging and pooled data difficult to interpret. Even the definitions of resectability and high-risk patients are not universally standardized, further complicating consistent classification of results. Finally, although most studies were recent, modern immunotherapies and targeted agents were rarely included. While a greater effect and benefit from preoperative treatment can be hypothesized, new specific evidence is needed.
In conclusion, secondary resectability should be the primary goal of systemic therapy in initially unresectable patients. Indications for neoadjuvant therapy should still be considered with caution, and its benefits require further validation through additional data; however, a short preoperative treatment may be considered in resectable patients with aggressive disease features or at high risk of incomplete surgery, acknowledging a non-negligible dropout rate (approximately 20%). If ongoing neoadjuvant trials yield positive results, based on their homogeneous inclusion criteria, indications will likely be rapidly extended to all patients with a solitary tumor > 5 cm, multiple lesions, N+ disease, or vascular invasion. The findings of the present review should guide multidisciplinary decision-making while awaiting conclusive evidence.
DECLARATIONS
Acknowledgments
This work was performed under the framework of the European Network for the Study of Cholangiocarcinoma (ENSCCA) and the COST Action Collaborations (COST Action CA22125 Precision-BTC-Net).
Authors’ contributions
Contributed to the conception and design of the work, data acquisition, analysis, interpretation, and manuscript revision: Risi L, Viganò L
Contributed to the design of the work, data acquisition and interpretation, and manuscript draft preparation: Cantamessa M, Pavia L
All authors have read and approved the final version of the manuscript.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
Not applicable.
Conflicts of interest
Not applicable.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Banales JM, Marin JJG, Lamarca A, et al. Cholangiocarcinoma 2020: the next horizon in mechanisms and management. Nat Rev Gastroenterol Hepatol. 2020;17:557-88.
2. Kelley RK, Bridgewater J, Gores GJ, Zhu AX. Systemic therapies for intrahepatic cholangiocarcinoma. J Hepatol. 2020;72:353-63.
3. Mazzaferro V, Gorgen A, Roayaie S, Droz Dit Busset M, Sapisochin G. Liver resection and transplantation for intrahepatic cholangiocarcinoma. J Hepatol. 2020;72:364-77.
4. Berardi G, Risi L, Muttillo EM, et al. Anatomic versus non-anatomic liver resection for intrahepatic cholangiocarcinoma: a systematic review and patient-level meta-analysis. Ann Surg Oncol. 2024;31:9170-82.
5. Kelley RK, Ueno M, Yoo C, et al.; KEYNOTE-966 Investigators. Pembrolizumab in combination with gemcitabine and cisplatin compared with gemcitabine and cisplatin alone for patients with advanced biliary tract cancer (KEYNOTE-966): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2023;401:1853-65.
6. Oh DY, He AR, Bouattour M, et al. Durvalumab or placebo plus gemcitabine and cisplatin in participants with advanced biliary tract cancer (TOPAZ-1): updated overall survival from a randomised phase 3 study. Lancet Gastroenterol Hepatol. 2024;9:694-704.
7. Tesini G, Ibrahim H, Rimassa L, Braconi C. Evolving precision: updates in targeted therapy for cholangiocarcinoma. Hepatology. 2025;Epub ahead of print.
8. Villard C, Habib M, Nordenvall C, Nilsson PJ, Jorns C, Sparrelid E. Conversion therapy in patients with colorectal liver metastases. Eur J Surg Oncol. 2021;47:2038-45.
9. Wang MD, Xu XJ, Wang KC, et al. Conversion therapy for advanced hepatocellular carcinoma in the era of precision medicine: current status, challenges and opportunities. Cancer Sci. 2024;115:2159-69.
10. Xu H, Zhang H, Li B, Chen K, Wei Y. Systemic conversion therapies for initially unresectable hepatocellular carcinoma: a systematic review and meta-analysis. BMC Cancer. 2024;24:1008.
11. Adam R, Wicherts DA, de Haas RJ, et al. Patients with initially unresectable colorectal liver metastases: is there a possibility of cure? J Clin Oncol. 2009;27:1829-35.
12. Perri G, Engstrand J, Wright RD, et al. The evolving concept of conversion surgery for upfront unresectable upper gastrointestinal and hepato-pancreato-biliary cancers: comprehensive review. BJS Open. 2025;9:zraf070.
13. Association for the Study of the Liver. EASL-ILCA Clinical Practice Guidelines on the management of intrahepatic cholangiocarcinoma. J Hepatol. 2023;79:181-208.
14. Vogel A, Bridgewater J, Edeline J, et al.; ESMO Guidelines Committee. Electronic address: [email protected]. Biliary tract cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023;34:127-40.
15. Benson AB, D’Angelica MI, Abrams T, et al. NCCN Guidelines® insights: biliary tract cancers, version 2.2023. J Natl Compr Canc Netw. 2023;21:694-704.
16. Mehrabi A, Golriz M, Ramouz A, et al. Promising outcomes of modified ALPPS for staged hepatectomy in cholangiocarcinoma. Cancers. 2023;15:5613.
17. Conci S, Viganò L, Ercolani G, et al. Outcomes of vascular resection associated with curative intent hepatectomy for intrahepatic cholangiocarcinoma. Eur J Surg Oncol. 2020;46:1727-33.
18. Tsilimigras DI, Sahara K, Wu L, et al. Very early recurrence after liver resection for intrahepatic cholangiocarcinoma: considering alternative treatment approaches. JAMA Surg. 2020;155:823-31.
19. Choi WJ, Williams PJ, Claasen MPAW, et al. Systematic review and meta-analysis of prognostic factors for early recurrence in intrahepatic cholangiocarcinoma after curative-intent resection. Ann Surg Oncol. 2022;Epub ahead of print.
20. Moazzam Z, Alaimo L, Endo Y, et al. Combined tumor burden score and carbohydrate antigen 19-9 grading system to predict outcomes among patients with intrahepatic cholangiocarcinoma. J Am Coll Surg. 2023;236:804-13.
21. Nassar A, Tzedakis S, Sindayigaya R, et al. Factors of early recurrence after resection for intrahepatic cholangiocarcinoma. World J Surg. 2022;46:2459-67.
22. Schadde E, Grunhagen DJ, Verhoef C, Krzywon L, Metrakos P. Limitations in resectability of colorectal liver metastases 2020 - a systematic approach for clinicians and patients. Semin Cancer Biol. 2021;71:10-20.
23. Conci S, Ruzzenente A, Viganò L, et al. Patterns of distribution of hepatic nodules (single, satellites or multifocal) in intrahepatic cholangiocarcinoma: prognostic impact after surgery. Ann Surg Oncol. 2018;25:3719-27.
24. Zhang B, Wang XY, Yang LY, et al. Genetic predictors of postoperative recurrence in node-negative intrahepatic cholangiocarcinoma. Updates Surg. 2025;77:675-84.
25. Viganò L, Risi L, Ammirabile A. New perspectives in biology-driven treatment of cholangiocarcinoma: from oncological resectability to genetic breakthroughs and AI-powered imaging. Hepatobiliary Surg Nutr. 2025;14:703-8.
26. Cervantes A, Adam R, Roselló S, et al.; ESMO Guidelines Committee. Electronic address: [email protected]. Metastatic colorectal cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023;34:10-32.
27. Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A. Rayyan-a web and mobile app for systematic reviews. Syst Rev. 2016;5:210.
28. Wells GA, O’Connell D, Peterson J, Welch V, Losos M, Tugwell P. The newcastle-ottawa scale (NOS) for assessing the quality of non-randomized studies in meta-analysis. Available from: https://www.researchgate.net/publication/261773681_The_Newcastle-Ottawa_Scale_NOS_for_Assessing_the_Quality_of_Non-Randomized_Studies_in_Meta-Analysis. [Last accessed on 13 May 2026].
29. Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21:1539-58.
30. Riley RD, Higgins JP, Deeks JJ. Interpretation of random effects meta-analyses. BMJ. 2011;342:d549.
31. Chen Y, Chen D, Wang Y, Han Y. Using freeman-tukey double arcsine transformation in meta-analysis of single proportions. Aesthetic Plast Surg. 2023;47:83-4.
32. Guyot P, Ades AE, Ouwens MJ, Welton NJ. Enhanced secondary analysis of survival data: reconstructing the data from published Kaplan-Meier survival curves. BMC Med Res Methodol. 2012;12:9.
33. Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials. 2007;8:16.
34. Wei Y, Royston P. Reconstructing time-to-event data from published Kaplan-Meier curves. Stata J. 2017;17:786-802.
35. Burke DL, Ensor J, Riley RD. Meta-analysis using individual participant data: one-stage and two-stage approaches, and why they may differ. Stat Med. 2017;36:855-75.
36. de Jong VMT, Moons KGM, Riley RD, et al. Individual participant data meta-analysis of intervention studies with time-to-event outcomes: a review of the methodology and an applied example. Res Synth Methods. 2020;11:148-68.
37. Royston P, Parmar MK. The use of restricted mean survival time to estimate the treatment effect in randomized clinical trials when the proportional hazards assumption is in doubt. Stat Med. 2011;30:2409-21.
38. Lueza B, Rotolo F, Bonastre J, Pignon JP, Michiels S. Erratum to: bias and precision of methods for estimating the difference in restricted mean survival time from an individual patient data meta-analysis. BMC Med Res Methodol. 2016;16:71.
39. Dong Y, Santol J, Podrascanin V, et al. Conversion to resection after neoadjuvant gemcitabine, cisplatin, and nab-paclitaxel for borderline resectable CCA: a real-world exploratory analysis. Ann Surg Oncol. 2025;32:7215-27.
40. Nakamura I, Hatano E, Baba H, et al. Impact of conversion surgery after chemotherapy in patients with initially unresectable and recurrent biliary tract cancer. Ann Gastroenterol Surg. 2023;7:1009-20.
41. Alaimo L, Moazzam Z, Lima HA, et al. Impact of staging concordance and downstaging after neoadjuvant therapy on survival following resection of intrahepatic cholangiocarcinoma: a bayesian analysis. Ann Surg Oncol. 2023;30:4799-808.
42. Ayabe RI, Paez-Arango N, Estrella JS, et al. Neoadjuvant chemotherapy for high-risk intrahepatic cholangiocarcinoma - does pathologic response mean better outcomes? HPB. 2023;25:472-80.
43. Choi SH, Kang I, Lee SH, et al. Clinical feasibility of curative surgery after nab-paclitaxel plus gemcitabine-cisplatin chemotherapy in patients with locally advanced cholangiocarcinoma. Surgery. 2023;173:280-8.
44. Choi WJ, Ivanics T, Claasen MPAW, Gallinger S, Hansen B, Sapisochin G. Is it safe to administer neoadjuvant chemotherapy to patients undergoing hepatectomy for intrahepatic cholangiocarcinoma? ACS-NSQIP propensity-matched analysis. HPB. 2022;24:1535-42.
45. Garita E, Sarwar A, Tahir M, et al. Yttrium90-TARE with or without chemotherapy may improve survival in patients with locally advanced unresectable intrahepatic cholangiocarcinoma. Discov Oncol. 2025;16:1386.
46. Kato A, Shimizu H, Ohtsuka M, et al. Downsizing chemotherapy for initially unresectable locally advanced biliary tract cancer patients treated with gemcitabine plus cisplatin combination therapy followed by radical surgery. Ann Surg Oncol. 2015;22:S1093-9.
47. Konstantinidis IT, Groot Koerkamp B, Do RK, et al. Unresectable intrahepatic cholangiocarcinoma: Systemic plus hepatic arterial infusion chemotherapy is associated with longer survival in comparison with systemic chemotherapy alone. Cancer. 2016;122:758-65.
48. Kosaka H, Matsui K, Ikeura T, et al. Prognostic impact of combination therapy with gemcitabine and cisplatin plus S-1 and subsequent conversion surgery for initially unresectable upper biliary tract cancers. Surg Today. 2025;55:351-9.
49. Le Roy B, Gelli M, Pittau G, et al. Neoadjuvant chemotherapy for initially unresectable intrahepatic cholangiocarcinoma. Br J Surg. 2018;105:839-47.
50. Maithel SK, Keilson JM, Cao HST, et al. NEO-GAP: a single-arm, phase II feasibility trial of neoadjuvant gemcitabine, cisplatin, and nab-paclitaxel for resectable, high-risk intrahepatic cholangiocarcinoma. Ann Surg Oncol. 2023;30:6558-66.
51. Mason MC, Massarweh NN, Tzeng CD, et al. Time to rethink upfront surgery for resectable intrahepatic cholangiocarcinoma? Implications from the neoadjuvant experience. Ann Surg Oncol. 2021;28:6725-35.
52. Murakami T, Matsuyama R, Yabushita Y, et al. Efficacy of conversion surgery for initially unresectable biliary tract cancer that has responded to down-staging chemotherapy. Cancers. 2025;17:873.
53. Nooijen LE, Franken LC, Belkouz A, et al. Efficacy and safety of gemcitabine plus cisplatin as potential preoperative chemotherapy in locally advanced intrahepatic, perihilar, and mid-cholangiocarcinoma: a retrospective cohort study. Am J Clin Oncol. 2021;44:526-32.
54. Riby D, Mazzotta AD, Bergeat D, et al. Downstaging with radioembolization or chemotherapy for initially unresectable intrahepatic cholangiocarcinoma. Ann Surg Oncol. 2020;27:3729-37.
55. Sutton TL, Billingsley KG, Walker BS, et al. Neoadjuvant chemotherapy is associated with improved survival in patients undergoing hepatic resection for intrahepatic cholangiocarcinoma. Am J Surg. 2021;221:1182-7.
56. Van Treeck BJ, Olave MC, Watkins RD, et al. Neoadjuvant therapy leads to objective response in intrahepatic cholangiocarcinoma. HPB. 2024;26:938-48.
57. Wang Z, Jin Y, Guo Y, et al. Conversion therapy of intrahepatic cholangiocarcinoma is associated with improved prognosis and verified by a case of patient-derived organoid. Cancers. 2021;13:1179.
58. Wehrle CJ, Chang J, Woo K, et al. Neoadjuvant therapy reduces node positivity but does not confer survival benefit versus up-front resection for resectable intrahepatic cholangiocarcinoma: a propensity-matched analysis. J Surg Oncol. 2024;130:453-61.
59. Wu G, Chen X, Luo R, et al. Histopathologic grading of residual tumor predicts survival of intrahepatic cholangiocarcinoma patients treated with neoadjuvant therapy: major pathologic response and its clinical significance. Am J Surg Pathol. 2025;49:578-87.
60. Parente A, Kamarajah SK, Baia M, et al. Neoadjuvant chemotherapy for intrahepatic, perihilar, and distal cholangiocarcinoma: a national population-based comparative cohort study. J Gastrointest Surg. 2023;27:741-9.
61. Yadav S, Xie H, Bin-Riaz I, et al. Neoadjuvant vs. adjuvant chemotherapy for cholangiocarcinoma: a propensity score matched analysis. Eur J Surg Oncol. 2019;45:1432-8.
62. Lai Q, Parisse S, Ginanni Corradini S, et al. Evolution of transplant oncology indications: a single-institution experience over 40 years. Updates Surg. 2024;76:911-21.
63. Kim KD, Lee JE, Kim J, et al. Laparoscopic liver resection as a treatment option for intrahepatic cholangiocarcinoma. Updates Surg. 2024;76:869-78.
64. Huiskens J, Bolhuis K, Engelbrecht MR, et al.; Dutch Colorectal Cancer Group. Outcomes of resectability assessment of the dutch colorectal cancer group liver metastases expert panel. J Am Coll Surg. 2019;229:523-32.e2.
65. Serenari M, Ratti F, Hilal MA, et al. Liver hypertrophy techniques: a position paper from the Italian Group of Regenerative and Occlusive Worldwide-used techniques of hepatic Hypertrophy (I GROWtoH). Updates Surg. 2025;77:2155-71.
66. Farges O, Fuks D, Boleslawski E, et al. Influence of surgical margins on outcome in patients with intrahepatic cholangiocarcinoma: a multicenter study by the AFC-IHCC-2009 study group. Ann Surg. 2011;254:824-29.
67. Sposito C, Maspero M, Cucchetti A, Mazzaferro V. A snapshot on current approaches to lymphadenectomy in liver resection for intrahepatic cholangiocarcinoma: results from an international survey. Updates Surg. 2024;76:1797-805.
68. Bismuth H, Adam R, Lévi F, et al. Resection of nonresectable liver metastases from colorectal cancer after neoadjuvant chemotherapy. Ann Surg. 1996;224:509-20.
69. Schartz DA, Porter M, Schartz E, et al. Transarterial Yttrium-90 radioembolization for unresectable intrahepatic cholangiocarcinoma: a systematic review and meta-analysis. J Vasc Interv Radiol. 2022;33:679-86.
70. Savic LJ, Chapiro J, Geschwind JH. Intra-arterial embolotherapy for intrahepatic cholangiocarcinoma: update and future prospects. Hepatobiliary Surg Nutr. 2017;6:7-21.
71. Liu JJ, Zhou M, Yuan T, Huang ZY, Zhang ZY. Conversion treatment for advanced intrahepatic cholangiocarcinoma: opportunities and challenges. World J Gastroenterol. 2025;31:104901.
72. Nordlinger B, Sorbye H, Glimelius B, et al.; EORTC Gastro-Intestinal Tract Cancer Group, Cancer Research UK; Arbeitsgruppe Lebermetastasen und-tumoren in der Chirurgischen Arbeitsgemeinschaft Onkologie (ALM-CAO); Australasian Gastro-Intestinal Trials Group (AGITG); Fédération Francophone de Cancérologie Digestive (FFCD). Perioperative chemotherapy with FOLFOX4 and surgery versus surgery alone for resectable liver metastases from colorectal cancer (EORTC Intergroup trial 40983): a randomised controlled trial. Lancet. 2008;371:1007-16.
73. Vauthey JN, Pawlik TM, Ribero D, et al. Chemotherapy regimen predicts steatohepatitis and an increase in 90-day mortality after surgery for hepatic colorectal metastases. J Clin Oncol. 2006;24:2065-72.
74. Rubbia-Brandt L, Audard V, Sartoretti P, et al. Severe hepatic sinusoidal obstruction associated with oxaliplatin-based chemotherapy in patients with metastatic colorectal cancer. Ann Oncol. 2004;15:460-6.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].