The evolving role of contrast-enhanced ultrasound in hepatocellular carcinoma: from diagnosis to therapeutic monitoring
Abstract
Hepatocellular carcinoma (HCC) remains a significant global health issue, linked to chronic liver diseases such as viral hepatitis, cirrhosis, and metabolic dysfunction-related steatohepatitis. Early, accurate diagnosis is vital for treatment, but many cases are diagnosed at advanced stage. Contrast-enhanced ultrasound (CEUS) is a valuable, radiation-free tool for real-time liver lesion assessment with high accuracy. This review examines the growing role of ultrasound and CEUS in diagnosing, monitoring, and post-treatment care of HCC. CEUS has sensitivity and specificity similar to computed tomography and magnetic resonance imaging, especially for nodules ≥ 1 cm, and helps clarify uncertain Liver Imaging Reporting and Data System findings. Dynamic CEUS improves diagnosis by allowing microvascular perfusion measurement. Artificial intelligence (AI) and machine learning integration promises automated lesion classification and better consistency. CEUS is especially useful in outpatient and resource-limited settings, enabling quick decision-making and reducing delays. Meta-analyses and studies support CEUS for initial detection and post-treatment follow-up. As advanced ultrasound becomes more accessible, CEUS can be more widely used in hepatology. Future steps include standard protocols, clinician training, and AI integration. Overall, CEUS complements other imaging methods and aids precision medicine in liver cancer management.
Keywords
INTRODUCTION
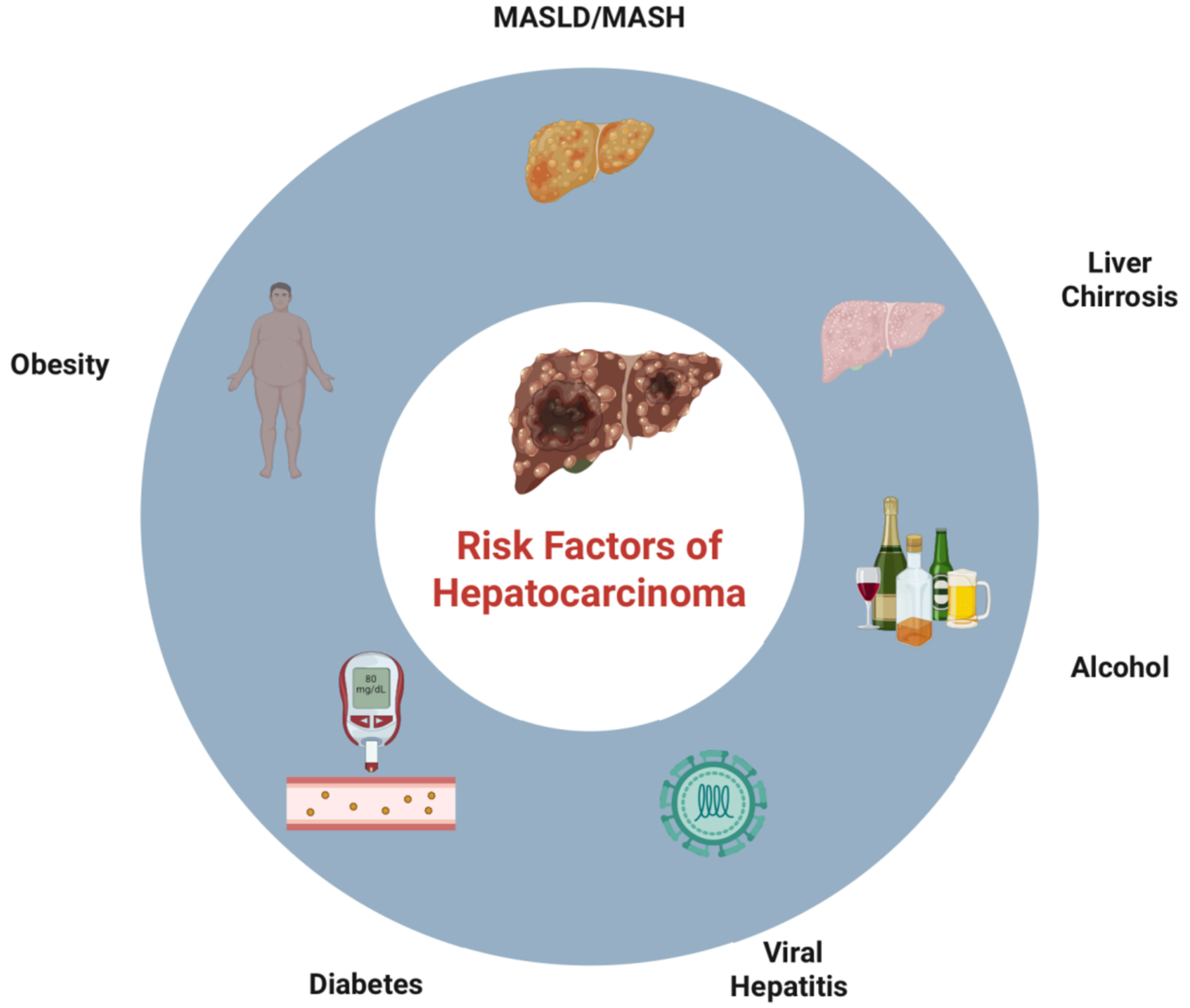
Hepatocellular carcinoma (HCC) is the most common primary liver malignancy and one of the leading causes of cancer-related mortality worldwide. According to the Global Cancer Statistics (GLOBOCAN) 2020 report, HCC ranks sixth in incidence among all malignant tumors and third in cancer-related deaths, with over 900,000 new cases and more than 800,000 deaths estimated each year[1]. The geographic distribution of the disease is heterogeneous, with significantly higher prevalence in East Asian countries and sub-Saharan Africa, where chronic hepatitis B virus infection is highly endemic. In Western countries, the leading cause is chronic hepatitis C virus infection, followed by metabolic dysfunction-associated steatotic liver disease (MASLD) and steatohepatitis (MASH), excessive alcohol consumption, and other causes of chronic liver disease leading to cirrhosis. Liver cirrhosis is considered the most critical risk factor for HCC development, present in approximately 80%-90% of patients at diagnosis. However, recent studies have shown a progressive increase in the incidence of HCC even in non-cirrhotic livers, particularly in patients with advanced MASH or significant fibrosis, especially those with metabolic syndrome, type 2 diabetes mellitus, and obesity, as shown in Figure 1[2]. Early diagnosis of HCC is crucial for improving prognosis and expanding curative treatment options. When detected at an early stage, HCC can be successfully treated with surgical resection, liver transplantation, or locoregional ablation (radiofrequency or microwave), achieving 5-year survival rates above 70%. However, most patients are diagnosed at an advanced stage, when therapeutic options are limited and survival is significantly reduced[3,4]. For this reason, the international guidelines recommend regular surveillance programs for patients at risk, particularly those with cirrhosis or advanced fibrosis. In this setting, noninvasive liver imaging, particularly conventional ultrasound and contrast-enhanced ultrasound (CEUS), plays a central role in screening, diagnosing, and monitoring suspicious liver nodules[3-5]. CEUS, through the use of intravascular, non-nephrotoxic microbubbles, enables real-time evaluation of hepatic vascular perfusion, allowing for high diagnostic accuracy in lesion characterization and in identifying the typical vascular pattern of HCC. Recent studies have demonstrated that CEUS is comparable to computed tomography (CT) and magnetic resonance imaging (MRI) in terms of sensitivity and specificity, especially for nodules < 2 cm[6-8]. This narrative review aims to comprehensively examine the role of conventional ultrasound and CEUS in the diagnosis and surveillance of HCC, highlighting their strengths, limitations, and integration into clinical practice.
Figure 1. Visual description of HCC-associated risk factors. Created in BioRender. https://BioRender.com/onozqd8. MASLD: Metabolic dysfunction-associated steatotic liver disease; MASH: metabolic dysfunction-associated steatohepatitis; HCC: hepatocellular carcinoma.
ULTRASOUND: FROM PHYSICAL PRINCIPLES TO CLINICAL APPLICATION
Brightness mode (B-mode) ultrasonography is the fundamental modality for morphological evaluation of the liver. It operates through high-frequency ultrasound waves (> 2 MHz), which are reflected to varying degrees depending on the acoustic impedance of the tissues. The resulting echoes are processed in real time to generate two-dimensional grayscale images that delineate hepatic parenchymal structures[9-11]. The examination is typically performed using a convex probe (3.5-5 MHz) placed on the patient’s upper abdomen in a supine or lateral decubitus position. Proper patient preparation, including fasting for at least
CLINICAL APPLICATIONS OF CEUS
CEUS utilizes intravascular microbubbles composed of gas, such as sulfur hexafluoride in Sonovue or perfluorobutane in Sonazoid, encapsulated within lipid or protein shells. These microbubbles, with a diameter between 1 and 4 micrometers, are too large to extravasate into the interstitial space and therefore remain confined within the bloodstream. When insonated, they oscillate and emit nonlinear harmonic signals, which can be detected in real time, enabling high sensitivity in the assessment of microvascular perfusion[20]. Sonovue acts solely as a blood-pool agent, allowing for the visualization of the conventional arterial, portal, and late vascular phases. In contrast, Sonazoid also enables imaging during the Kupffer phase, which occurs between 10 and 30 min after injection. This phase reflects the activity of liver-resident macrophages (Kupffer cells) and proves especially useful for identifying lesions that lack such cells, including HCC and metastases[21]. Comparative studies have shown that both contrast agents offer similar sensitivity, approximately 80%, and extremely high specificity, reaching 100%, in the diagnosis of HCC. While the Kupffer phase provides additional diagnostic information, it does not appear to increase sensitivity significantly[22]. The CEUS examination consists of multiple phases. The arterial phase, which occurs within the first 10-30 s after contrast administration, is characterized by rapid tumor enhancement, known as arterial phase hyperenhancement (APHE). This typically appears as a strong, early hyperintensity of the lesion relative to the surrounding liver parenchyma, a hallmark feature of HCC[23,24]. During the portal venous phase (30-120 s), the hepatic parenchyma enhances more intensely, while malignant lesions often become iso- or hypoenhancing, displaying earlier washout compared to healthy liver tissue[22]. This is followed by the late phase (beyond 120 s), during which washout becomes more pronounced. A delayed and mild washout is characteristic of HCC and is observed in over 95% of cases that initially exhibit APHE[25]. With Sonazoid, it is also possible to perform imaging during the Kupffer phase, typically 10-15 min after contrast injection. During this phase, normal liver tissue retains the microbubbles due to the presence of Kupffer cells and appears uniformly echogenic. In contrast, lesions lacking Kupffer cells remain hypoenhancing, creating a so-called “Kupffer defect”[24]. Together, these phases reflect both the tumor’s vascular architecture and the functional integrity of the hepatic macrophage system. APHE combined with delayed washout remains a key diagnostic criterion for HCC[25]. To facilitate standardized interpretation, the CEUS Liver Imaging Reporting and Data System (LI-RADS), developed by the American College of Radiology, provides a classification framework for hepatic lesions in at-risk patients, ranging from LR-1 (definitely benign) to LR-5 (definitely HCC). This system is based on enhancement and washout patterns observed during CEUS[26]. For LR-5 lesions, diagnostic specificity can reach 95%-100%, with interobserver agreement exceeding 90% in experienced centers. Currently, the integration of the Kupffer phase into LI-RADS is under investigation, as the official criteria apply exclusively to blood-pool agents, such as Sonovue[22,27-32]. Table 1 shows the typical imaging findings of common hepatic nodules. CEUS plays a valuable role in the management of HCC during both radiofrequency ablation (RFA) and surgical resection. In the periprocedural setting, CEUS improves lesion conspicuity and targeting, particularly for small or iso-echoic HCC nodules that are poorly visible on B-mode ultrasound, thereby facilitating accurate needle placement during RFA. Immediately after ablation, CEUS allows real-time assessment of treatment efficacy by demonstrating the absence of intralesional enhancement and enabling prompt detection of residual viable tumor, which may be retreated during the same session. In surgical resection, intraoperative CEUS enhances detection of additional small lesions and refines margin assessment, contributing to more accurate surgical planning. However, CEUS does not replace CT or MRI for preoperative staging or postoperative surveillance, which remain essential for comprehensive evaluation of disease extent and recurrence[33].
Typical imaging findings of common hepatic nodules across B-mode ultrasound, CEUS, and CT/MRI
| Lesion | B-mode ultrasound (typical) | CEUS (typical enhancement pattern) | CT/MRI (typical dynamic pattern) | Key clue(s) for differentiation |
| HCC | Variable echogenicity; may be hypo/iso/hyperechoic; may show mosaic/heterogeneity | Non-rim APHE followed by late, mild washout; with Sonazoid: Kupffer-phase defect | Arterial hyperenhancement + washout (portal/delayed) ± enhancing capsule; ancillary features may support diagnosis | Washout late and mild is typical for HCC in at-risk patients (CEUS LI-RADS concept) |
| Hemangioma | Often well-defined, hyperechoic (may be heterogeneous if large) | Peripheral, discontinuous nodular enhancement in arterial phase with progressive centripetal fill-in; usually no washout (persistent enhancement) | Peripheral nodular discontinuous enhancement with progressive fill-in on delayed phases | “Peripheral nodular + fill-in” is the classic hallmark |
| FNH | Often iso/hypoechoic; may show central scar (not always visible) | Rapid, intense arterial hyperenhancement often with centrifugal spoke-wheel pattern; iso/hyperenhancement in portal and late phases (no washout) | Strong arterial enhancement; typically iso in portal/delayed; central scar may enhance late (MRI) | No washout + spoke-wheel arterial pattern strongly suggests FNH |
| HCA | Variable; may be heterogeneous; may show intralesional fat/hemorrhage signs | Usually arterial hyperenhancement, often heterogeneous; portal/late phase isoenhancement is common; washout may occur in a minority (pitfall) | Arterial hyperenhancement; variable portal/delayed behavior; MRI may show fat/hemorrhage depending on subtype | Clinical context (young women, hormones) + variable imaging; can overlap with HCC → consider MRI/subtyping when needed |
| iCCA | Often hypoechoic/heterogeneous; may be ill-defined | Frequently rim APHE with early and/or marked washout (LR-M pattern) | Peripheral/rim arterial enhancement with progressive delayed enhancement; capsular retraction may be present | Rim APHE + early/marked washout favors non-HCC malignancy |
| Metastases | Often multiple; target/bull’s-eye appearance may be seen | Commonly rim APHE and early, marked washout | Variable; often peripheral enhancement; diffusion restriction on MRI | Variable; often peripheral enhancement; diffusion |
COMPARATIVE EVALUATION OF CEUS VS. CT AND MRI
Advantages of CEUS include an excellent safety profile (absence of ionizing radiation, nephrotoxicity, or systemic reactions to the contrast agent), the capability for real-time imaging, cost-effectiveness, and portability, features particularly beneficial in patients with fragile health or those hospitalized. Furthermore, CEUS is repeatable and can be utilized to guide or monitor locoregional treatments[27,34-36]. Among its limitations are a narrow field of view, operator dependence, suboptimal performance in obese patients or those with deep or gas-masked lesions, and challenges in assessing multifocal disease. Efforts are ongoing to standardize the technique, particularly with the introduction of novel agents, such as Sonazoid, which enables imaging during the macrophage phase[36-39]. For nodules larger than 1 cm, CEUS has demonstrated diagnostic accuracy comparable to CT and MRI, with sensitivity ranging from 85%-95% and specificity between 90%-98%. In some instances, CEUS can clarify indeterminate MRI findings (LI-RADS 3/4), particularly when Kupffer phase imaging is employed[28,40-45]. Because CEUS contrast agents remain strictly intravascular, this modality provides superior visualization of the vascular architecture of focal liver lesions compared to CT and MRI, proving especially useful for characterizing indeterminate nodules[46-48]. When performed by experienced operators, CEUS can reduce the need for second-level examinations, thereby lowering costs and providing immediate diagnostic feedback[49,50]. It is particularly advantageous for patients with anxiety or claustrophobia towards tomographic imaging techniques[51-52]. Recent literature supports the role of CEUS in hepatology practice. In this regard, a recent meta-analysis of nine studies and 1,434 patients reported sensitivity of 92% and specificity of 93%, with an area under the curve(AUC) of 0.95 and a diagnostic odds ratio (DOR) > 150, underscoring the impact of technological advancements and operator experience on diagnostic performance[53]. Direct comparative studies have shown that, for hepatic lesions ≥ 1 cm, CEUS sensitivity for detecting APHE is comparable to or superior to that of MRI. Specificity is also high, ranging from 90% to 98%, consistent with values reported for CT and MRI[54,55]. Limitations of CEUS include a lower diagnostic yield for deep, multifocal, or anatomically challenging-to-visualize lesions, particularly in contexts where tomographic imaging provides a broader overview and is less operator-dependent. Nevertheless, CEUS remains a valid, noninvasive, and real-time imaging option for characterizing hepatic lesions[56]. Recently, the use of CEUS in follow-up after locoregional treatments [e.g., transarterial chemoembolization (TACE) or percutaneous ablation] was analyzed, demonstrating 85% sensitivity and 94% specificity, confirming its efficacy in early detection of tumor recurrence or persistence[57]. A summary of recent meta-analyses on CEUS is shown in Table 2.
Summary of recent meta-analyses on CEUS
| Reference | Sample size | Number of studies | Aim | Sensitivity | Specificity |
| Zhang et al. (2023)[52] | 1,434 | 9 | Evaluation of CEUS for early diagnosis of HCC | 0.92 | 0.93 |
| Wang et al. (2023)[54] | 831 | 5 | Diagnostic performance of CEUS LI-RADS algorithms for HCC | 0.79 for LI-RADS 5 0.86 for LI-RADS 4/5 | 0.81 for LI-RADS 5 0.70 for LI-RADS 4/5 |
| Zhang et al. (2017)[56] | More than 4,827 patients (the numbers of patients were not mentioned in two studies | 53 | Evaluation of CEUS for diagnosis of HCC | 0.85 | 0.91 |
Overall, these data confirm CEUS as a reliable, safe, and highly performant technique, effective for both initial diagnosis and post-treatment surveillance, with performance comparable, and in some aspects superior, to CT and MRI[54]. CEUS is particularly advantageous for patients contraindicated for contrast-enhanced CT or MRI, such as those with chronic or advanced renal disease or known allergies to iodine or gadolinium. It is important to emphasize that CEUS contrast agents are not nephrotoxic and do not carry a risk of nephrogenic systemic fibrosis. According to the 2020 guidelines of the World Federation for Ultrasound in Medicine and Biology, CEUS is recommended as a first-line modality in patients with renal insufficiency and focal liver lesions, regardless of cirrhosis status. In cirrhotic patients, CEUS can be used for initial diagnosis, whereas CT/MRI are indicated for comprehensive staging when feasible[58]. Indeed, CEUS contrast agents contain neither iodine nor gadolinium; they consist of inert gas microbubbles stabilized by phospholipid or protein shells, exhibiting an excellent safety profile with severe adverse reactions occurring in less than 0.01% of cases. In this way, recent studies conducted in high-risk populations have confirmed that CEUS can be performed safely without the need for premedication or precautionary measures[59]. In patients with renal insufficiency, Sonazoid (perflubutane-based) has demonstrated an excellent safety profile, with no requirement for renal function screening. Furthermore, no increase in adverse events even in renally impaired patients, without the need for hydration or preventive
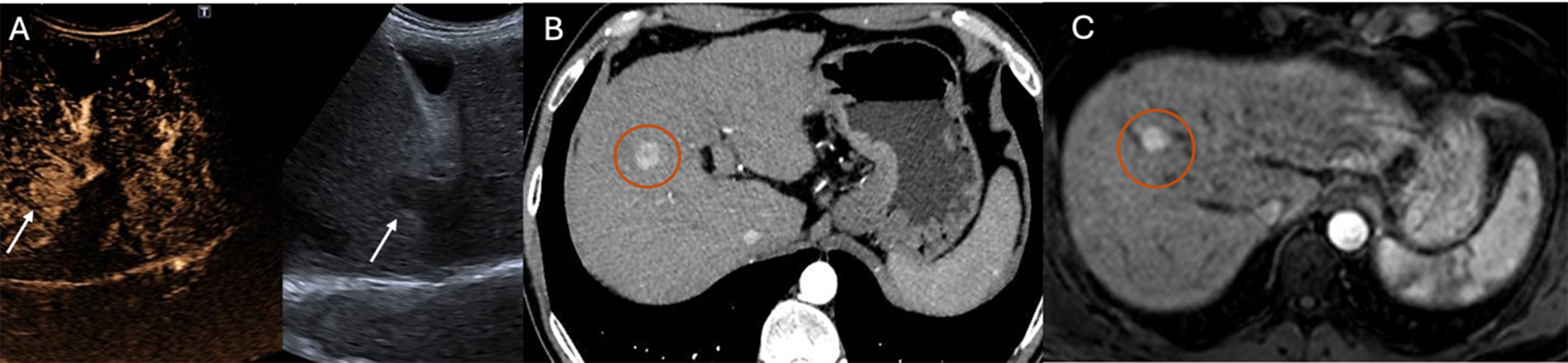
Figure 2. Detection of HCC using CEUS after contrast agent administration (SonoVue®) (A), arterial-phase CT (B), and arterial-phase MRI (C). Arrows in (A) indicate the HCC lesion, while circles in (B and C) highlight the tumor area identified on cross-sectional imaging. HCC: Hepatocellular carcinoma; CEUS: contrast-enhanced ultrasound; CT: computed tomography; MRI: magnetic resonance imaging.
Comparative summary of advantages, limitations, and preferred use of CEUS, CT, and MRI in the diagnostic pathway
| Modality | Key advantages | Key limitations | Preferred clinical use |
| CEUS | No ionizing radiation; excellent safety profile; non-nephrotoxic contrast agent; real-time assessment; portable and cost-effective; repeatable; useful to guide/monitor locoregional treatments | Limited field of view; operator dependence; reduced performance in obesity or deep/gas-masked lesions; limited assessment of multifocal disease and extrahepatic spread (whole-liver staging not feasible) | First-line problem-solving for targeted characterization of a known lesion (especially in patients unsuitable for iodinated/gadolinium contrast); outpatient/point-of-care characterization; early post-treatment assessment of residual/recurrent disease |
| CT (multiphase) | Whole-liver coverage; fast acquisition; widely available; less operator-dependent; suitable for global assessment and staging | Ionizing radiation; iodinated contrast-related risks (allergy/ nephrotoxicity); less suitable for repeated follow-up in some patients | Comprehensive staging (whole liver, vascular invasion, extrahepatic disease) and second-line when CEUS is inconclusive or technically limited |
| MRI (dynamic contrast-enhanced) | No ionizing radiation; high soft-tissue contrast; multiparametric evaluation; strong overall performance for characterization and staging | Longer acquisition time; limited availability/cost; contraindications in some patients; patient intolerance/claustrophobia; gadolinium-related concerns in severe renal impairment | Second-line characterization of indeterminate lesions and staging when feasible; preferred when a broader, multiparametric assessment is required |
CLINICAL CONSIDERATIONS AND FUTURE PERSPECTIVES
The use of CEUS significantly impacts clinical practice, particularly when prompt therapeutic decisions are required. It enables the real-time characterization of liver lesions, providing immediate access to treatment options, streamlining the diagnostic process, and reducing reliance on more invasive or expensive tests. In multidisciplinary teams, CEUS is a helpful tool for guiding management, assisting in deciding between surgery, ablation, or active surveillance[36]. The growing availability of advanced ultrasound machines in hepatology clinics enables the direct incorporation of CEUS into outpatient visits. This approach enables quick, point-of-care diagnoses, enhances patient adherence to follow-up, and reduces referrals to radiology or hospitals for further tests[61-63]. Additionally, CEUS in remote or resource-limited centers broadens diagnostic access across larger areas. Successful implementation in outpatient settings requires proper training for medical staff and logistical planning for contrast agent supplies. Despite these challenges, the clinical and organizational advantages, such as increased efficiency and patient-centered care, make CEUS a crucial part of liver disease diagnosis[64-67]. Recent advances include the integration of artificial intelligence (AI) into CEUS, offering new diagnostic and treatment insights. With the increasing use of immune checkpoint inhibitors in the treatment of HCC, there is growing interest in imaging biomarkers capable of capturing early and dynamic treatment responses. CEUS, through real-time evaluation of tumor microvascular perfusion, may provide complementary information to conventional size-based criteria by detecting early changes in intratumoral enhancement that reflect immune-mediated vascular remodeling or necrosis. This may be particularly relevant in the context of immunotherapy, where atypical response patterns such as pseudoprogression or delayed responses can limit the accuracy of CT- or MRI-based assessments alone. Although current evidence remains limited and CEUS is not yet incorporated into standardized response criteria for immunotherapy, preliminary data suggest that CEUS-derived perfusion parameters could contribute to early response monitoring and treatment adaptation. Further prospective studies are needed to validate the role of CEUS as an adjunct imaging tool for immunotherapy response assessment in HCC[68]. AI holds promise for automating the analysis of enhancement patterns, aiding in the classification of liver nodules, and predicting treatment outcomes. These technologies help reduce variability between observers and standardize interpretations across operators. Machine learning models trained on large CEUS datasets can identify features of malignant lesions, boosting sensitivity and accuracy beyond traditional visual assessment[69,70]. Furthermore, dynamic CEUS (D-CEUS), combined with advanced software, enables the objective measurement of microvascular blood flow, providing clinicians with more precise data for personalized cancer monitoring[68]. Based on current research, an integrated diagnostic approach can be suggested for managing liver nodules in cirrhotic patients. When a suspicious lesion ≥ 1 cm is found, initial assessment should include B-mode ultrasound combined with CEUS. If CEUS shows a typical pattern, APHE followed by washout, HCC can be diagnosed, and the lesion classified as LI-RADS 5[36]. In cases with atypical vascular features, further evaluation with MRI or CT is recommended. For indeterminate nodules (LI-RADS 3 or 4), D-CEUS offers detailed perfusion analysis[33]. If uncertainty persists, a biopsy may be necessary to obtain a definitive diagnosis. Post-treatment, early CEUS and follow-up D-CEUS at one month are advised to evaluate treatment response and detect residual or recurrent disease. These protocols, supported by multicenter studies and expert hepatology groups, have proven to improve diagnostic accuracy and resource management[50]. Future perspectives and validated clinical applications of CEUS are summarized in Table 4.
Validated clinical applications and future perspectives in the clinical application of CEUS
| Category | Item | Descriptions | Expected impact | References |
| Already validated/currently applied | Integration of CEUS in outpatient hepatology clinics | Routine use of CEUS during outpatient diagnostic practice for real-time characterization of liver lesions | Rapid diagnosis, improved follow-up adherence, reduced need for radiological referrals | [36,61,63] |
| Already validated/currently applied | Integrated diagnostic algorithms | Structured approach using B-mode ultrasound, CEUS, D-CEUS, MRI/CT, and biopsy | Improved accuracy in HCC diagnosis and treatment stratification | [33,36,50] |
| Already validated/currently applied | Post-treatment CEUS surveillance | Early CEUS and 1-month D-CEUS to assess therapy response | Early detection of residual or recurrent disease | [50] |
| Future perspective | Use of CEUS in remote and resource-limited settings | Deployment of CEUS where access to CT/MRI is limited | Expanded diagnostic access and equity in care delivery | [61-63] |
| Future perspective | Staff training and logistical support | Training clinicians and ensuring contrast agent availability in outpatient settings | Essential for safe, effective CEUS implementation in decentralized settings | [64-67] |
| Future perspective | AI integration | Application of AI algorithms to better evaluate CEUS patterns | Standardization, reduced interobserver variability, improved diagnostic accuracy | [68,69] |
| Future perspective | Machine learning for predictive modeling | Development of machine learning models based on large CEUS datasets | Enhanced lesion classification and outcome prediction | [68,69] |
| Future perspective | D-CEUS with perfusion quantification | Use of software-assisted D-CEUS to measure microvascular flow | Personalized monitoring of tumor response and recurrence | [68] |
CONCLUSIONS
HCC is a major challenge in hepatology, with rising incidence and associations with chronic liver conditions. Ultrasound imaging, particularly B-mode ultrasound, is crucial for diagnosis, surveillance, and monitoring, although it is limited in detecting small or deep lesions[70]. CEUS is increasingly used as a second-line tool due to real-time vascular assessment, no ionizing radiation, and safety in renal impairment. Studies show that the sensitivity and specificity of CEUS are comparable or superior to CT and MRI, making it reliable for diagnosing and characterizing HCC and other liver lesions. Integrating CEUS speeds up patient stratification and decision-making, improving management. It is also valuable for early post-treatment monitoring of therapies such as ablation and TACE, detecting residual tumor or progression. D-CEUS provides quantitative microvascular perfusion data, adding functional insights[71]. AI technologies promise further advancements by enabling automated interpretation and reducing variability. These tools position CEUS as a vital part of liver oncology and precision medicine. This review emphasizes CEUS as both a complement to cross-sectional imaging and a standalone tool, especially for indeterminate nodules, improving LI-RADS classification and guiding therapy, potentially reducing invasive procedures. Broader CEUS use in hepatology, with training and standardized protocols, can enhance early HCC diagnosis and healthcare outcomes, especially in resource-limited centers.
DECLARATIONS
Acknowledgements
The authors thank Corrado Pelaia for assistance with Figure 1 & Graphic Abstract creation using BioRender.com (Graphic Abstract was created in BioRender. https://BioRender.com/ufsof1r).
Authors’ contributions
Conceptualized and designed the review: Abenavoli L, Laganà D
Wrote, reviewed, and edited the manuscript: Gambardella ML, Passante E, La Torre G
Provided figures and tables: Passante E, Battaglia C
Reviewed and approved the final manuscript as submitted: Manti F, Console D
Read and approved the final manuscript: Abenavoli L, Luzza F, Laganà D
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
None.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209-49.
2. Scarlata GGM, Cicino C, Spagnuolo R, et al. Impact of diet and gut microbiota changes in the development of hepatocellular carcinoma. Hepatoma Res. 2024;10:19.
3. Kim TH, Kim SY, Tang A, Lee JM. Comparison of international guidelines for noninvasive diagnosis of hepatocellular carcinoma: 2018 update. Clin Mol Hepatol. 2019;25:245-63.
4. Kim TK, Jang HJ. Contrast-enhanced ultrasound in the diagnosis of nodules in liver cirrhosis. World J Gastroenterol. 2014;20:3590-6.
5. Ciocalteu A, Iordache S, Cazacu SM, et al. Role of contrast-enhanced ultrasonography in hepatocellular carcinoma by using LI-RADS and ancillary features: a single tertiary centre experience. Diagnostics. 2021;11:2232.
6. Duparc F, Grignon B, Bonnel F, Macchi V, Douard R. Liver anatomy: from Couinaud to the transplantation. Surg Radiol Anat. 2020;42:1405.
7. Fasel JHD. Human liver territories: think beyond the 8-segments scheme. Clin Anat. 2017;30:974-7.
9. Tyagi G, Jha RK. Portal vein variations, clinical correlation, and embryological explanation: a review article. Cureus. 2023;15:e36400.
10. Mao W, Jiang X, Cao Y, et al. A practical study of the hepatic vascular system anatomy of the caudate lobe. Quant Imaging Med Surg. 2021;11:1313-21.
11. D’Souza JC, Sultan LR, Hunt SJ, et al. B-mode ultrasound for the assessment of hepatic fibrosis: a quantitative multiparametric analysis for a radiomics approach. Sci Rep. 2019;9:8708.
12. Hirooka M, Koizumi Y, Sunago K, et al. Efficacy of B-mode ultrasound-based attenuation for the diagnosis of hepatic steatosis: a systematic review/meta-analysis. J Med Ultraso. 2022;49:199-210.
13. Uggowitzer M, Kugler C, Machan L, et al. Power Doppler imaging and evaluation of the resistive index in focal nodular hyperplasia of the liver. Abdom Imaging. 1997;22:268-73.
14. Cannella R, Giambelluca D, Pellegrinelli A, Cabassa P. Color Doppler ultrasound in portal hypertension: a closer look at left gastric vein hemodynamics. J Ultrasound Med. 2021;40:7-14.
15. Iranpour P, Lall C, Houshyar R, et al. Altered Doppler flow patterns in cirrhosis patients: an overview. Ultrasonography. 2016;35:3-12.
16. Chawla Y, Kumar S, Dhiman RK, Suri S, Dilawari JB. Duplex Doppler sonography in patients with Budd-Chiari syndrome. J Gastroenterol Hepatol. 1999;14:904-7.
17. Lee DH, Lee JY, Han JK. Superb microvascular imaging technology for ultrasound examinations: Initial experiences for hepatic tumors. Eur J Radiol. 2016;85:2090-5.
18. Shankar N, Kuo L, Krugliak Cleveland N, et al. Point-of-care ultrasound in gastroenterology and hepatology. Clin Gastroenterol Hepatol. 2025;23:1277-90.
19. Ying X, Dong S, Zhao Y, Chen Z, Jiang J, Shi H. Research progress on contrast-enhanced ultrasound (CEUS) assisted diagnosis and treatment in liver-related diseases. Int J Med Sci. 2025;22:1092-108.
20. Friedrich-Rust M, Klopffleisch T, Nierhoff J, et al. Contrast-enhanced ultrasound for the differentiation of benign and malignant focal liver lesions: a meta-analysis. Liver Int. 2013;33:739-55.
21. Goto E, Masuzaki R, Tateishi R, et al. Value of post-vascular phase (Kupffer imaging) by contrast-enhanced ultrasonography using Sonazoid in the detection of hepatocellular carcinoma. J Gastroenterol. 2012;47:477-85.
22. Huang J, Gao L, Li J, et al. Head-to-head comparison of Sonazoid and SonoVue in the diagnosis of hepatocellular carcinoma for patients at high risk. Front Oncol. 2023;13:1140277.
23. Westwood M, Joore M, Grutters J, et al. Contrast-enhanced ultrasound using SonoVue® (sulphur hexafluoride microbubbles) compared with contrast-enhanced computed tomography and contrast-enhanced magnetic resonance imaging for the characterisation of focal liver lesions and detection of liver metastases: a systematic review and cost-effectiveness analysis. Health Technol Assess. 2013;17:1-243.
24. Ren J, Lu Q, Fei X, et al. Assessment of arterial-phase hyperenhancement and late-phase washout of hepatocellular carcinoma-a meta-analysis of contrast-enhanced ultrasound (CEUS) with SonoVue® and Sonazoid®. Eur Radiol. 2024;34:3795-812.
25. Kang HJ, Kim JH, Yoo J, Han JK. Diagnostic criteria of perfluorobutane-enhanced ultrasonography for diagnosing hepatocellular carcinoma in high-risk individuals: how is late washout determined? Ultrasonography. 2022;41:530-42.
26. D’Onofrio M, Crosara S, De Robertis R, Canestrini S, Mucelli RP. Contrast-enhanced ultrasound of focal liver lesions. AJR Am J Roentgenol. 2015;205:W56-66.
27. Sawatzki M, Meyenberger C, Brand S, Semela D. Contrast-enhanced ultrasound (CEUS) has excellent diagnostic accuracy in differentiating focal liver lesions: results from a Swiss tertiary gastroenterological centre. Swiss Med Wkly. 2019;149:w20087.
28. Schwarze V, Marschner C, Völckers W, et al. Diagnostic value of contrast-enhanced ultrasound versus computed tomography for hepatocellular carcinoma: a retrospective, single-center evaluation of 234 patients. J Int Med Res. 2020;48:300060520930151.
29. Ghazanfar H, Javed N, Qasim A, et al. Metabolic dysfunction-associated steatohepatitis and progression to hepatocellular carcinoma: a literature review. Cancers. 2024;16:1214.
30. Chen HJ, Huang TX, Jiang YX, Chen X, Wang AF. Multifunctional roles of inflammation and its causative factors in primary liver cancer: a literature review. World J Hepatol. 2023;15:1258-71.
31. Hansmann J, Ray CE Jr
32. Beudeker BJB, Fu S, Balderramo D, et al. Validation and optimization of AFP-based biomarker panels for early HCC detection in Latin America and Europe. Hepatol Commun. 2023;7:e0264.
33. Cao J, Dong Y, Xu X, et al. LI-RADS CEUS nonradiation TRA version 2024: application on HCC patients treated with ablation treatment. Ultrasound Med Biol. 2025;51:1308-15.
34. Han K, Kim JH. Transarterial chemoembolization in hepatocellular carcinoma treatment: Barcelona clinic liver cancer staging system. World J Gastroenterol. 2015;21:10327-35.
35. Zhang Y, Li Q, Li L, et al. Diagnostic performance of modified contrast-enhanced ultrasound liver imaging reporting and data system in patients without risk factors for hepatocellular carcinoma: comparison with world federation for ultrasound in medicine and biology guideline. Ultrasound Med Biol. 2024;50:243-50.
36. Piscaglia F, Wilson SR, Lyshchik A, et al. American college of radiology contrast enhanced ultrasound liver imaging reporting and data system (CEUS LI-RADS) for the diagnosis of hepatocellular carcinoma: a pictorial essay. Ultraschall Med. 2017;38:320-4.
37. Terzi E, Iavarone M, Pompili M, et al.; CEUS LI-RADS Italy study group collaborators. Contrast ultrasound LI-RADS LR-5 identifies hepatocellular carcinoma in cirrhosis in a multicenter restropective study of 1,006 nodules. J Hepatol. 2018;68:485-92.
38. Quaia E, Alaimo V, Baratella E, et al. Effect of observer experience in the differentiation between benign and malignant liver tumors after ultrasound contrast agent injection. J Ultrasound Med. 2010;29:25-36.
39. Park HJ, Kim SY. Imaging modalities for hepatocellular carcinoma surveillance: expanding horizons beyond ultrasound. J Liver Cancer. 2020;20:99-105.
40. Melendez-Torres J, Singal AG. Early detection of hepatocellular carcinoma: roadmap for improvement. Expert Rev Anticancer Ther. 2022;22:621-32.
41. Tzartzeva K, Obi J, Rich NE, et al. Surveillance imaging and alpha fetoprotein for early detection of hepatocellular carcinoma in patients with cirrhosis: a meta-analysis. Gastroenterology. 2018;154:1706-18.e1.
42. Association for the Study of the Liver. EASL clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol. 2018;69:182-236.
43. Tarao K, Nozaki A, Komatsu H, et al. Comparison of unenhanced magnetic resonance imaging and ultrasound in detecting very small hepatocellular carcinoma. World J Hepatol. 2021;13:699-708.
44. Mei Q, Yu M, Chen Q. Clinical value of contrast-enhanced ultrasound in early diagnosis of small hepatocellular carcinoma (≤ 2 cm). World J Clin Cases. 2022;10:8525-34.
45. Claudon M, Dietrich CF, Choi BI, et al. Guidelines and good clinical practice recommendations for contrast enhanced ultrasound (CEUS) in the liver-update 2012: a WFUMB-EFSUMB initiative in cooperation with representatives of AFSUMB, AIUM, ASUM, FLAUS and ICUS. Ultraschall Med. 2013;34:11-29.
46. Marrero JA, Kulik LM, Sirlin CB, et al. Diagnosis, staging, and management of hepatocellular carcinoma: 2018 practice guidance by the american association for the study of liver diseases. Hepatology. 2018;68:723-50.
47. Sidhu PS, Cantisani V, Dietrich CF, et al. The EFSUMB guidelines and recommendations for the clinical practice of contrast-enhanced ultrasound (CEUS) in non-hepatic applications: update 2017 (long version). Ultraschall Med. 2018;39:e2-44.
48. Wang DC, Jang HJ, Kim TK. Characterization of indeterminate liver lesions on CT and MRI with contrast-enhanced ultrasound: what is the evidence? AJR Am J Roentgenol. 2020;214:1295-304.
49. Seitz K, Strobel D, Bernatik T, et al. Contrast-enhanced ultrasound (CEUS) for the characterization of focal liver lesions - prospective comparison in clinical practice: CEUS vs. CT (DEGUM multicenter trial). Parts of this manuscript were presented at the ultrasound Dreiländertreffen 2008, Davos. Ultraschall Med. 2009;30:383-9.
50. Lanka B, Jang HJ, Kim TK, Burns PN, Wilson SR. Impact of contrast-enhanced ultrasonography in a tertiary clinical practice. J Ultrasound Med. 2007;26:1703-14.
51. Enders J, Zimmermann E, Rief M, et al. Reduction of claustrophobia during magnetic resonance imaging: methods and design of the “CLAUSTRO” randomized controlled trial. BMC Med Imaging. 2011;11:4.
52. Zhang Z, Ma C, Luo Y. Diagnostic value of liver contrast-enhanced ultrasound in early hepatocellular carcinoma: a systematic review and meta-analysis. J Gastrointest Oncol. 2023;14:626-35.
53. Candita G, Rossi S, Cwiklinska K, et al. Imaging diagnosis of hepatocellular carcinoma: a state-of-the-art review. Diagnostics. 2023;13:625.
54. Wang H, Cao J, Fan H, Huang J, Zhang H, Ling W. Compared with CT/MRI LI-RADS, whether CEUS LI-RADS is worth popularizing in diagnosis of hepatocellular carcinoma? Quant Imaging Med Surg. 2023;13:4919-32.
55. Wilson SR, Burns PN. Microbubble-enhanced US in body imaging: what role? Radiology. 2010;257:24-39.
56. Zhang J, Yu Y, Li Y, Wei L. Diagnostic value of contrast-enhanced ultrasound in hepatocellular carcinoma: a meta-analysis with evidence from 1998 to 2016. Oncotarget. 2017;8:75418-26.
57. Hai Y, Savsani E, Chong W, Eisenbrey J, Lyshchik A. Meta-analysis and systematic review of contrast-enhanced ultrasound in evaluating the treatment response after locoregional therapy of hepatocellular carcinoma. Abdom Radiol. 2021;46:5162-79.
58. Dietrich CF, Nolsøe CP, Barr RG, et al. Guidelines and good clinical practice recommendations for contrast-enhanced ultrasound (CEUS) in the liver-update 2020 WFUMB in cooperation with EFSUMB, AFSUMB, AIUM, and FLAUS. Ultrasound Med Biol. 2020;46:2579-604.
59. Bartolotta TV, Taibbi A, Midiri M, Lagalla R. Contrast-enhanced ultrasound of hepatocellular carcinoma: where do we stand? Ultrasonography. 2019;38:200-14.
60. Nakai M, Sato M, Sahara S, et al. Radiofrequency ablation assisted by real-time virtual sonography and CT for hepatocellular carcinoma undetectable by conventional sonography. Cardiovasc Intervent Radiol. 2009;32:62-9.
61. Huang J, Chen W, Yao S. Assessing diagnostic value of contrast-enhanced ultrasound and contrast-enhanced computed tomography in detecting small hepatocellular carcinoma: a meta-analysis. Medicine. 2017;96:e7555.
63. Wicks JS, Dale BS, Ruffolo L, et al. Comparable and complimentary modalities for treatment of small-sized HCC: surgical resection, radiofrequency ablation, and microwave ablation. J Clin Med. 2023;12:5006.
64. Dong Y, Qiu Y, Yang D, et al. Potential application of dynamic contrast enhanced ultrasound in predicting microvascular invasion of hepatocellular carcinoma. Clin Hemorheol Microcirc. 2021;77:461-9.
65. Chernyak V, Fowler KJ, Do RKG, et al. LI-RADS: looking back, looking forward. Radiology. 2023;307:e222801.
66. Cai WJ, Ying M, Zheng RQ, et al. Contrast-enhanced ultrasound liver imaging reporting and data system in hepatocellular carcinoma ≤ 5 cm: biological characteristics and patient outcomes. Liver Cancer. 2023;12:356-71.
67. Lyshchik A, Kono Y, Dietrich CF, et al. Contrast-enhanced ultrasound of the liver: technical and lexicon recommendations from the ACR CEUS LI-RADS working group. Abdom Radiol. 2018;43:861-79.
68. Tong J, Tan Y, Ouyang W, Chang H. Targeting immune checkpoints in hepatocellular carcinoma therapy: toward combination strategies with curative potential. Exp Hematol Oncol. 2025;14:65.
69. Brooks JA, Kallenbach M, Radu IP, et al. Artificial intelligence for contrast-enhanced ultrasound of the liver: a systematic review. Digestion. 2025;106:227-44.
70. Abenavoli L, Scarlata GGM, Gambardella ML, et al. Combined model for the diagnosis of hepatocellular carcinoma: a pilot study comparing the liver to spleen volume ratio and liver vein to cava attenuation. J Clin Med. 2025;14:4306.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].