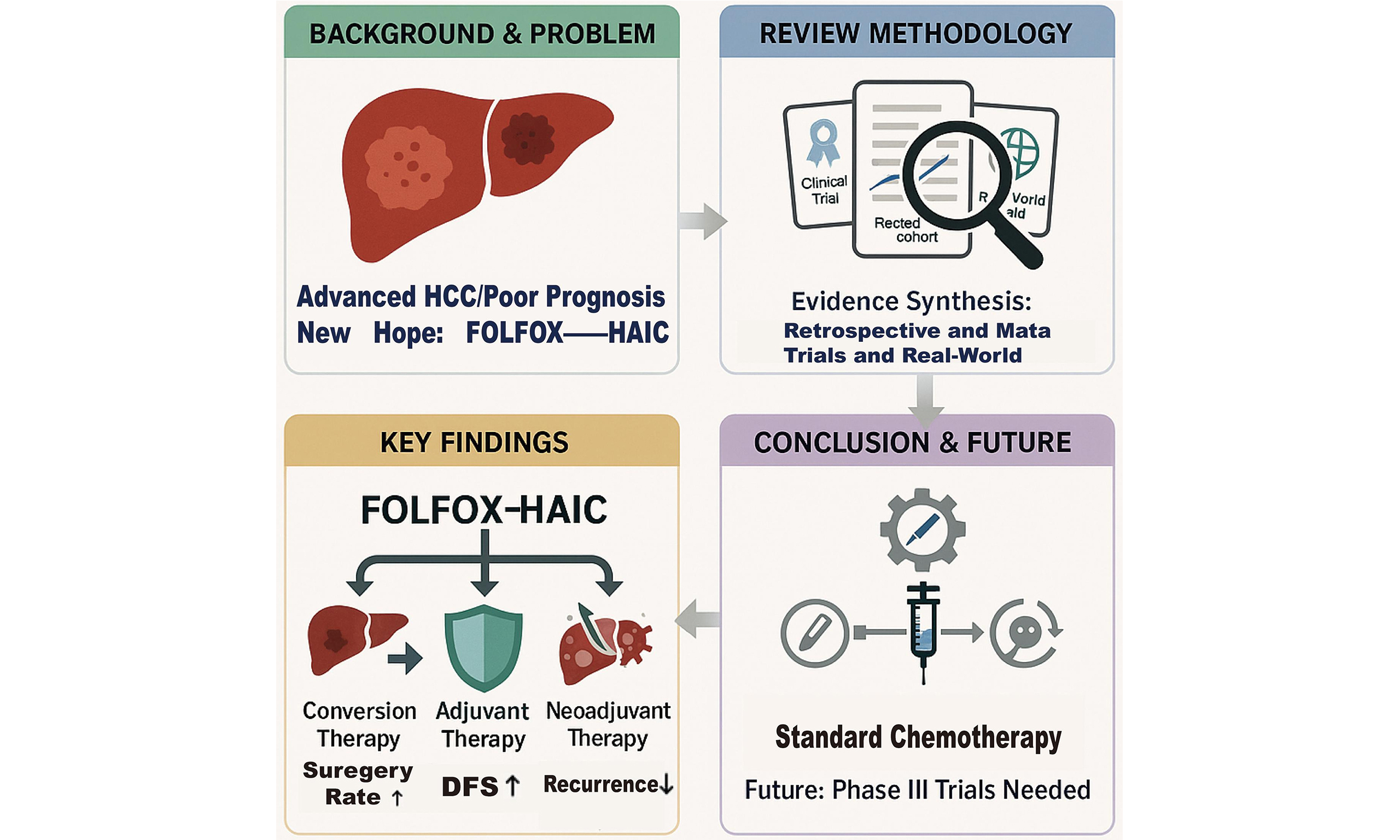
Review of advances in FOLFOX-HAIC for conversion, neoadjuvant, and adjuvant therapy in hepatocellular carcinoma
Abstract
FOLFOX (oxaliplatin, leucovorin, 5-fluorouracil)-based hepatic arterial infusion chemotherapy (FOLFOX-HAIC) has emerged as a promising locoregional strategy in the multimodal management of hepatocellular carcinoma (HCC), particularly for patients with intermediate or advanced-stage disease. This narrative review summarizes recent clinical evidence on the application of FOLFOX-HAIC across the therapeutic continuum: in conversion, neoadjuvant, and adjuvant settings. We conducted an analysis based on a search of PubMed, Embase, China National Knowledge Infrastructure (CNKI) and conference abstracts, which incorporated phase II/III trials, retrospective cohort studies, multicenter real-world studies, and meta-analyses. Evidence indicates that FOLFOX-HAIC, especially when combined with targeted therapy and immunotherapy, yields superior objective response rates and higher rates of conversion to curative resection compared to conventional transarterial chemoembolization (TACE). Furthermore, as postoperative adjuvant therapy, it significantly reduces recurrence risk and prolongs survival in high-risk patients, such as those with microvascular invasion. In the neoadjuvant setting, these combinations can induce profound pathological responses, potentially improving outcomes for resectable patients at high risk of recurrence. Consequently, FOLFOX-HAIC represents an evolving cornerstone of HCC therapy. However, current evidence is largely derived from retrospective and small prospective studies, underscoring the urgent need for large-scale phase III randomized controlled trials to standardize regimens, optimize patient selection, and confirm long-term survival benefits.
Keywords
INTRODUCTION
Hepatocellular carcinoma (HCC) is a common malignancy with a poor prognosis, often diagnosed at intermediate or advanced stages, which precludes curative resection[1-3]. This has driven the need for effective locoregional and systemic combination therapies. Compared with conventional transarterial chemoembolization (TACE), hepatic arterial infusion chemotherapy (HAIC) delivers chemotherapeutic agents directly into the artery supplying the tumor, thereby achieving higher intra-tumoral drug concentrations and enhanced local antitumor effects, while potentially mitigating systemic toxicity[4]. Recent studies have demonstrated the efficacy of FOLFOX (oxaliplatin, leucovorin, 5-fluorouracil)-based HAIC (hereafter FOLFOX-HAIC) in controlling intrahepatic disease, facilitating conversion to resection, and providing perioperative benefits - effects that appear significantly enhanced when combined with targeted therapy or immunotherapy[5]. This review aims to summarize current evidence for FOLFOX-HAIC in conversion, adjuvant, and neoadjuvant settings, and to discuss its limitations and future directions.
METHODOLOGY
This narrative review was conducted to summarize the recent clinical evidence on FOLFOX-HAIC in conversion, neoadjuvant, and adjuvant therapy for HCC.
We performed literature searches in PubMed, Embase, China National Knowledge Infrastructure (CNKI) and major international conference abstract databases. The search strategy combined relevant keywords with controlled vocabulary terms [e.g., Medical Subject Headings (MeSH) in PubMed, Emtree in Embase] such as “hepatocellular carcinoma”, “HCC”, “FOLFOX”, “hepatic arterial infusion chemotherapy”, “HAIC”, “conversion therapy”, “downstaging”, “neoadjuvant therapy”, “adjuvant therapy”, and “perioperative” using Boolean operators (“AND”, “OR”). No start date restriction was applied to ensure inclusion of the most recent evidence.
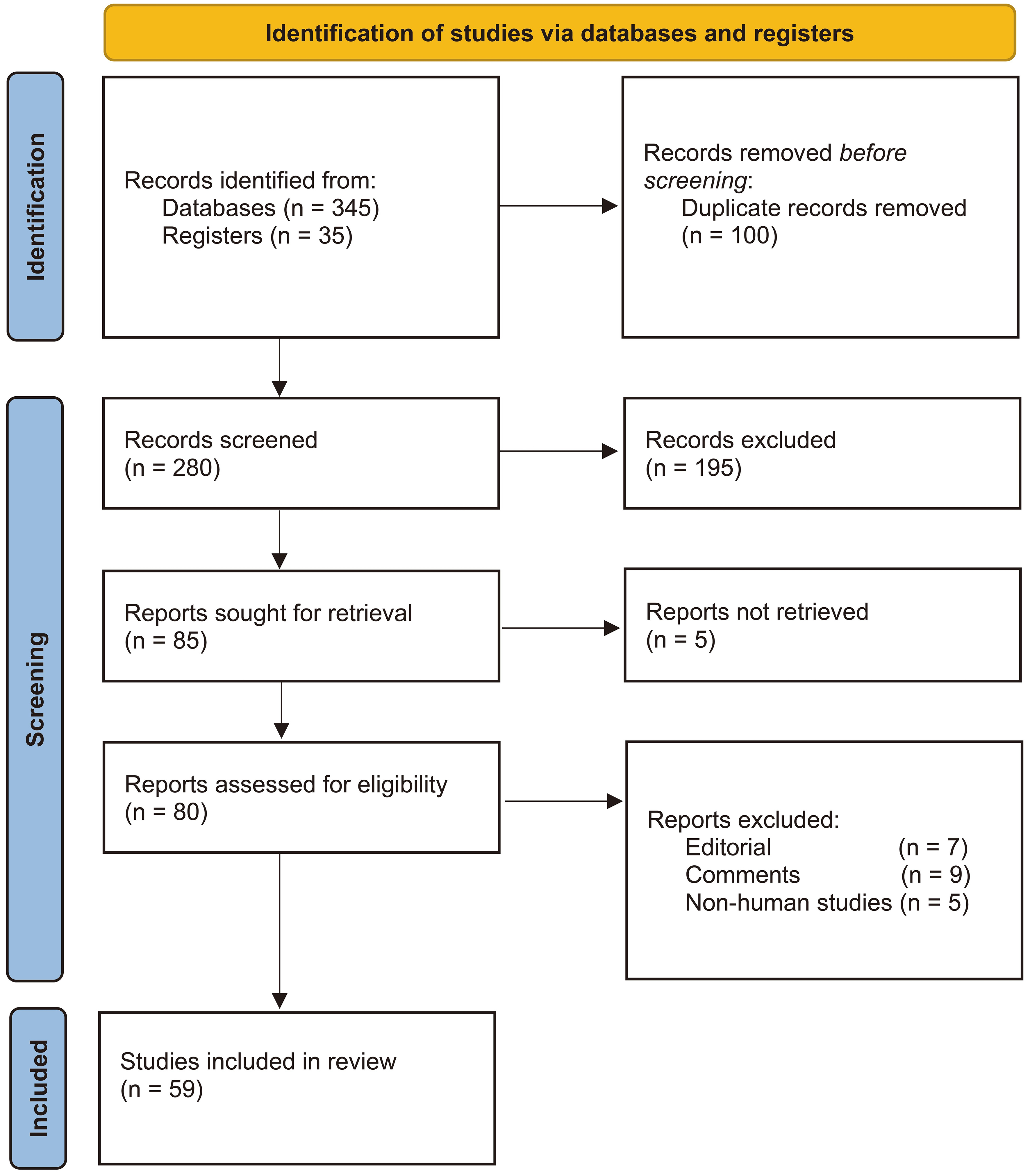
The literature screening followed a standard process. Initially, approximately 380 records were retrieved. After removing duplicates, roughly 280 unique records underwent title and abstract screening. Subsequently, about 80 articles were selected for full-text review. Finally, 59 studies met the inclusion criteria and were included in this review. The inclusion criteria were as follows: studies focusing on FOLFOX-HAIC in conversion, neoadjuvant, or adjuvant settings for HCC; published in English. The main exclusion criteria included: editorials, comments, non-human studies, and studies for which the full text was unavailable. The figures provided for the screening process are illustrative of the workflow; the precise numbers may vary with future literature updates [Figure 1].
To ensure transparent evaluation of the evidence, we assessed the quality of key outcomes [e.g., objective response rate (ORR), conversion-to-surgery rate, overall survival (OS), disease-free survival (DFS)] for major findings. This assessment was guided by the principles of the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) framework[6]. We considered study design (randomized trial vs. observational study), risk of bias, consistency of results, directness of evidence, precision of effect estimates, and potential for publication bias. Based on this evaluation, the overall certainty of evidence supporting a conclusion is qualitatively described in the narrative as high, moderate, low, or very low. (High, meaning we are very confident that the true effect lies close to that of the estimate; Moderate, meaning we are moderately confident in the effect estimate, and the true effect is likely to be close to the estimate but there is a possibility that it is substantially different; Low, meaning our confidence in the effect estimate is limited, and the true effect may be substantially different from the estimate; and Very low, meaning we have very little confidence in the effect estimate, and the true effect is likely to be substantially different). This approach prioritizes findings from high-quality trials.
Within the text, we have indicated the study design and level of evidence where relevant [e.g., randomized controlled trial (RCT), phase II trial, retrospective analysis]. Citation numbering follows that of the source text. This manuscript is a narrative review and, therefore, did not adhere to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. [Table 1 and Supplementary Table 1].
GRADE evaluation
| Quality of evidence | Description |
| High | We are very confident that the true effect lies close to that of the estimate of the effect. Future research is very unlikely to change our confidence in the estimate |
| Moderate | We are moderately confident in the effect estimate. Future research is likely to have an important impact on our confidence in the estimate and may change the estimate |
| Low | Our confidence in the effect estimate is limited. The true effect may be substantially different from the estimate |
| Very Low | We have very little confidence in the effect estimate. The true effect is likely to be substantially different from the estimate |
ADVANCES IN CONVERSION THERAPY
Conversion therapy aims to render initially unresectable or borderline-resectable tumors operable, enabling curative resection and improving long-term survival[7]. For intermediate-to-advanced HCC, TACE has been a standard locoregional option[8]. However, growing evidence suggests that FOLFOX-HAIC yields superior conversion outcomes compared to TACE[4,9]. Moreover, in advanced HCC with portal vein tumor thrombosis (PVTT), adding FOLFOX-HAIC to systemic regimens can further improve prognosis[10]. The following sections summarize evidence for FOLFOX-HAIC, both as monotherapy and in combination with targeted agents, immunotherapy, or multimodal approaches, within the conversion therapy setting.
FOLFOX-HAIC monotherapy as conversion therapy
Multiple studies have demonstrated that FOLFOX-HAIC monotherapy is superior to TACE for conversion. A study reported at the 2020 European Society for Medical Oncology (ESMO) meeting showed a surgical conversion rate of 23.8% for the FOLFOX-HAIC group vs. 11.5% for contemporaneous TACE group[11] (Quality of Evidence: Moderate). Li et al. subsequently confirmed this advantage in a randomized phase III trial: the conversion rate was 24% in the FOLFOX-HAIC arm vs. 12% in the TACE arm; ORR was 46% vs. 18%; median OS was 12.9 months vs. 8.2 months [HR (hazard ratio) = 0.56, P < 0.05]. These results indicate that compared with TACE, FOLFOX-HAIC can substantially improve tumor response and prolong survival. Notably, the study suggested that if patients receive conversion therapy but ultimately do not undergo resection, OS differences between HAIC and TACE are not significant - implying that HAIC’s maximal benefit is realized through enabling surgery; if surgery is not achieved, the long-term survival advantage may diminish[9] (Quality of Evidence: High). Retrospective cohort data further support long-term benefits after successful HAIC conversion: Wang et al. reported that the 5year survival rate is approaching that of initially resectable patients after HAIC followed by surgery (approximately 48.7% vs. 52.1%). However, repeated HAIC cycles may show diminishing marginal benefit and raise safety concerns: efficacy appears to plateau beyond four cycles while cumulative hepatotoxicity increases, requiring careful risk-benefit assessment[12] (Quality of Evidence: Low). Importantly, the cumulative hepatotoxicity from repeated FOLFOX-HAIC cycles may extend beyond transient transaminitis. The oxaliplatin component is associated with the development of sinusoidal obstruction syndrome, which can exacerbate underlying hepatic fibrosis and contribute to increased portal hypertension over time[13]. This underscores the critical need for vigilant monitoring of liver stiffness (e.g., via elastography) and portal pressure dynamics in patients undergoing multiple treatment cycles, particularly those with pre-existing advanced cirrhosis. Overall, current evidence establishes FOLFOX-HAIC as a promising conversion modality, but many supporting studies are from single centers, with limited sample sizes and heterogeneous patient populations.
FOLFOX-HAIC combined with targeted therapy as conversion therapy
Combining HAIC with molecular targeted agents may produce local-systemic synergy and improve conversion efficacy. A phase III trial by He et al. showed that FOLFOX-HAIC combined with sorafenib significantly outperformed sorafenib alone: median OS was 13.37 months vs. 7.13 months (HR = 0.52, P < 0.001), ORR rose from 5.7% to 52.4%, and conversion rate improved from 2.1% to 12.8%. This demonstrates that even when patients receive systemic targeted therapy, adding HAIC yields substantial incremental survival benefit[10] (Quality of Evidence: High). Safety data from a phase II study by Zheng et al. indicated similar rates of grade 3-4 adverse events between HAIC combined with sorafenib (91%) and sorafenib alone (88%), suggesting comparable tolerability[14] (Quality of Evidence: Moderate). A retrospective analysis by Yang et al. reported a pathological complete response (pCR) rate of 68.2% in patients treated with HAIC combined with targeted therapy - markedly higher than with HAIC alone[15] (Quality of Evidence: Low). Collectively, these studies suggest that HAIC combined with targeted agents can increase downstaging and resection rates without markedly increasing toxicity. With the advent of next-generation tyrosine kinase inhibitors (TKIs) such as lenvatinib and donafenib, HAIC combined with targeted regimens may achieve even greater conversion efficacy.
FOLFOX-HAIC combined with immunotherapy as conversion therapy
Immune checkpoint inhibitor (ICI) monotherapy typically yields ORRs below 20% in advanced HCC[16], but HAIC combined with ICI may overcome this limitation. Although large RCTs are lacking, several prospective and retrospective studies have reported encouraging results. Li et al. presented a prospective single-arm phase II study at ESMO 2025 evaluating HAIC combined with ivonescimab [a programmed cell death protein 1 (PD-1)/vascular endothelial growth factor (VEGF) bispecific antibody] in first-line treatment of unresectable HCC: among 12 evaluable patients, ORR by modified Response Evaluation Criteria in Solid Tumors (mRECIST) was 75.0%, disease control rate (DCR) 91.7%, and conversion rate 58.3%; grade 3 treatment-related adverse events occurred in 22.2%, with no grade 4 events, suggesting favorable efficacy and manageable safety[17] (Quality of Evidence: Low). Zhao et al. conducted a phase II trial in advanced HCC with Vp3/4 PVTT; FOLFOX-HAIC combined with the programmed cell death-ligand 1 (PD-L1) antibody durvalumab achieved an ORR of 47.4%, substantially higher than historical rates for ICI monotherapy[18] (Quality of Evidence: Low). Liu et al. reported HAIC combined with PD-1 inhibitor sintilimab and a bevacizumab biosimilar in unresectable HCC, showing ORR of 58.6% and pCR of 46.7%, indicating strong antitumor activity[19] (Quality of Evidence: Low). A multicenter retrospective analysis by Xin et al. described atezolizumab (PD-L1) plus bevacizumab together with FOLFOX-HAIC for advanced HCC, yielding ORR of 67.3% and enabling curative resection in 13.4% of patients[20] (Quality of Evidence: Low). In summary, HAIC combined with immunotherapy has shown promising preliminary conversion efficacy, but studies are generally small.
FOLFOX-HAIC combined with targeted-immunotherapy as conversion therapy
Emerging triple or quadruple regimens that combine HAIC combined with targeted agents and ICIs aim to maximize local and systemic synergy. These multimodal strategies have produced breakthrough results in conversion therapy, markedly improving response and conversion rates. Pan et al. reported a phase II study in high-risk PVTT patients using HAIC combined with PD-1 (toripalimab) and TKI (lenvatinib): median progression-free survival (PFS) was 15 months and ORR reached 68.97% (mRECIST), with 24.1% of patients converted to surgery[21] (Quality of Evidence: Low). Zhao et al. found that triple therapy (HAIC combined with TKI and PD-1 inhibitor) vs. dual targeted-immunotherapy significantly extended median OS from 16.6 to 21.0 months and median PFS from 8.9 to 11.6 months, while ORR increased approximately threefold (53.2% vs. 17.0%)[22] (Quality of Evidence: Low). For patients with special anatomy, such as arteriovenous shunts [arterioportal shunt (APS)], Liu et al. reported that triple therapy achieved APS closure in 74.1% of cases, with a median OS of 10 months[23] (Quality of Evidence: Low). Some groups have explored adding transarterial embolization (TAE) to form a quadruple regimen; Chen et al. compared HAIC combined with TKI, PD-1 and TAE vs. HAIC combined with TKI and PD-1 and found that adding TAE extended median OS from 11.3 to 14.1 months and increased ORR from 52% to 72%, although efficacy for single giant tumors (> 15 cm) was limited[24] (Quality of Evidence: Low). These data inform optimization of combination regimens. Investigators have also explored biomarkers to predict benefit from combined regimens. Lai et al. identified baseline serum CCL28 [Chemokine (C-C motif) ligand 28] and betacellulin (BTC) levels associated with response to HAIC combined with TKI and PD-1 inhibitor; 6-month PFS rates differed by marker strata and the overall 6-month PFS reached 80.6% with median OS of 17.9 months[25] (Quality of Evidence: Very Low). Liver function (Child-Pugh class) also substantially affects prognosis with HAIC combined with combination therapy, indicating hepatic reserve is an important predictor of benefit[26]. Safety profiles of HAIC combined with targeted and immune agents have generally been acceptable: reported grade 3-4 adverse events are typically below 30%, mainly hematologic toxicities and hypertension, with no clear signal of increased irreversible toxicity[27] (Quality of Evidence: Low). Importantly, studies have not reported treatmentrelated deaths[21-23,25,28,29]. Overall, in carefully selected patients, HAIC combined with multimodal therapy has an acceptable risk-benefit ratio. A 2023 Chinese expert consensus noted that while these combinations show improved response and survival trends, they remain exploratory[30]. A recent large realworld study of 318 advanced HCC patients further supported multimodal HAIC combined with TKI and PD-1 therapy: ORR 47.1%, DCR 85.5%, median PFS ≈11.4 months, median OS 21.7 months; 34.6% of patients underwent successful surgery after treatment. Subgroup analysis showed that for intermediatestage [Barcelona Clinic Liver Cancer (BCLC)-B] patients successfully converted to surgery, prognosis was markedly better than for nonconverted patients (median OS 29.3 months vs. 19.7 months)[31] (Quality of Evidence: Moderate). This study provides strong clinical support for HAICbased targeted-immune conversion therapy. In summary, HAIC combined with targeted and immunotherapy provides an effective conversion strategy for advanced HCC, significantly improving survival and resection rates. Optimal patient selection based on biomarkers and liver function is key, with a manageable safety profile.
ADVANCES IN POSTOPERATIVE ADJUVANT THERAPY
Although curative resection is a key treatment for early HCC, high postoperative recurrence remains a major challenge[32]. Patients with highrisk pathological features (e.g., tumor diameter ≥ 5 cm, microvascular invasion (MVI), macrovascular invasion/PVTT) have poor longterm outcomes after surgery alone[33]. To reduce recurrence and improve survival, HAICbased adjuvant strategies have been investigated. The following summarizes FOLFOX-HAIC monotherapy and combinations with targeted agents and/or immunotherapy in the adjuvant setting.
FOLFOX-HAIC monotherapy as adjuvant treatment
Evidence increasingly shows that postoperative FOLFOX-HAIC infusion can improve outcomes in highrisk HCC. A network metaanalysis comparing multiple postoperative adjuvant approaches ranked HAIC highest for OS benefit (HR ≈ 0.44), with benefits most pronounced in subgroups with PVTT and vascular invasion[34] (Quality of Evidence: Moderate). Another metaanalysis by Hu et al. confirmed that adjuvant HAIC significantly reduced death and recurrence vs. surgery alone (OS HR ≈ 0.69; DFS HR ≈ 0.64), with the greatest benefit in patients with PVI or MVI (OS HR ≈ 0.43)[35] (Quality of Evidence: Moderate). These highlevel syntheses support HAIC as an effective adjuvant option. Prospective and retrospective studies also directly validate HAIC’s adjuvant value. A prospective randomized trial by Li et al. reported that in high-risk HCC patients, postoperative FOLFOX-HAIC extended median recurrence-free survival (RFS)/DFS) from 10.0 to 20.3 months and reduced the 2-year cumulative recurrence rate from 55.7% to 40.1% (absolute reduction ≈ 15.6%)[36] (Quality of Evidence: High). A recent multicenter study by Wen et al. pooled 549 MVI-positive patients (from two RCT populations) and used propensity score matching to compare postoperative strategies: median DFS in the HAIC group reached 63.2 months vs. 21.7 months in the TACE group and 11.2 months in surgery alone (P < 0.05). Five-year OS in the HAIC group was 72.8%, compared with 29.8% for TACE and 53.4% for surgery alone, with HAIC showing a highly significant advantage. Notably, TACE did not significantly improve OS vs. no adjuvant therapy (P = 0.279), whereas HAIC markedly delayed recurrence and improved survival[37] (Quality of Evidence: Low). These findings strongly support FOLFOX-HAIC as an effective adjuvant therapy in high-risk patients, particularly MVI-positive cases. Given HAIC’s pharmacokinetic advantage of high intrahepatic drug concentration with relatively controlled systemic toxicity, it is becoming an effective strategy to prevent postoperative recurrence. Nevertheless, much of the evidence remains retrospective and some prospective trials are small.
FOLFOX-HAIC combined with targeted adjuvant therapy
HAIC combined with targeted therapy has been explored in the adjuvant setting to achieve both local eradication of residual disease and inhibition of systemic micrometastases. Long et al. conducted a prospective phase II trial of postoperative FOLFOX-HAIC followed by sequential donafenib (a domestic TKI) in 18 highrisk patients, with median followup of 15 months: 1year RFS was 64.9% and median RFS was 14.3 months. In the subgroup with tumor diameter ≥ 5 cm and MVI, the combination yielded improved RFS vs. historical controls. The proposed mechanism is HAIC eradication of microscopic residual disease and TKI suppression of angiogenesis. Reported adverse events included hand-foot skin reaction (72.2%) and diarrhea (38.9%); grade ≥ 3 events occurred in 33.3% without treatmentrelated deaths, and the toxicity profile was comparable to TKI monotherapy[38] (Quality of Evidence: Low). Although feasible and tolerable, this single-arm, small-sample study lacks a control comparison and thus has limited persuasive power.
FOLFOX-HAIC combined with immunotherapy as adjuvant therapy
Immunotherapy in the adjuvant setting for highrisk HCC is of growing interest. Studies have explored postoperative HAIC combined with PD-1/PDL1 inhibitors to achieve local plus systemic synergy. Liang et al. reported that in MVIpositive patients, adjuvant HAIC combined with PD-1 inhibitor significantly prolonged DFS vs. HAIC alone (HR ≈ 0.41, P = 0.031), corresponding to an approximately 60% reduction in recurrence risk. Importantly, the combination did not significantly increase treatmentrelated adverse events (P > 0.05), indicating acceptable safety. Although OS differences were not yet statistically significant (P = 0.124), longer followup and larger samples may reveal survival benefits[39] (Quality of Evidence: Very Low). These data suggest that combining HAIC’s local control with immunotherapy’s systemic modulation may reduce recurrence; however, current evidence is preliminary and further trials are required.
FOLFOX-HAIC combined with targeted-immunotherapy as adjuvant therapy
Studies combining HAIC, targeted agents and immunotherapy in the adjuvant setting are at an early stage and limited in number. Nevertheless, preliminary data suggest potential incremental benefits. Although direct trials of HAIC combined with TKI and ICI adjuvant therapy are not yet available, results from adjuvant targeted-immune combinations are informative. A retrospective analysis by Yang et al. showed that postoperative immunotherapy plus targeted therapy significantly extended median DFS vs. surgery alone (22 months vs. 11 months), reducing recurrence risk by about 54% (HR = 0.46, 95%CI: 0.24-0.90), particularly in patients with MVI[40] (Quality of Evidence: Low). A multicenter realworld study by Ouyang et al. found that postoperative camrelizumab (PD-1) plus apatinib (antiangiogenic) in MVIpositive resected patients reduced recurrence risk by 53% (HR ≈ 0.47, P < 0.001) with grade 3-4 treatmentrelated events occurring in 20.7%, mainly controllable hypertension and proteinuria[41] (Quality of Evidence: Low). These findings indicate that targeted-immune adjuvant regimens effectively clear microscopic residual disease and delay recurrence. Theoretically, adding HAIC to this combination could further enhance local control. Thus, adjuvant HAIC combined with targeted and immune triple therapy merits prospective evaluation in ultrahighrisk populations (e.g., large tumors with MVI) to determine whether it confers additional benefit over dual therapy and whether the safety profile is acceptable. If proven, such a regimen could set a new standard for postoperative adjuvant therapy.
Advances in neoadjuvant therapy
Neoadjuvant therapy refers to preoperative systemic or regional treatment for resectable tumors to reduce tumor volume, eradicate micrometastases, and improve surgical and postoperative outcomes[42]. For some intermediate or locally advanced but technically resectable HCC, preoperative tumor downstaging is increasingly attempted. FOLFOX-HAIC has shown encouraging results in this domain; the following sections summarize HAIC monotherapy and combination neoadjuvant strategies.
FOLFOX-HAIC monotherapy as neoadjuvant treatment
HAIC as neoadjuvant therapy for resectable HCC is increasingly recognized. Several studies indicate that preoperative FOLFOX-HAIC improves longterm outcomes. In a retrospective comparison, Hu et al. evaluated preoperative HAIC followed by hepatectomy vs. upfront resection: 1 and 3year OS in the neoadjuvant HAIC group were 95.2% and 75.9%, significantly higher than 85.4% and 67.2% in the upfront surgery group; 1 and 3year RFS were 66.0% and 51.4% vs. 54.1% and 42.1% respectively, suggesting reduced postoperative recurrence with neoadjuvant HAIC[43] (Quality of Evidence: Low). Although neoadjuvant therapy may increase surgical difficulty (e.g., adhesions, fibrosis), overall benefit deserves attention. Pan et al. compared neoadjuvant HAIC with postoperative portal vein chemotherapy pump infusion: preoperative HAIC was associated with longer median OS (26.71 months vs. 19.47 months) and markedly longer median PFS (37.57 months vs. 6.73 months), smaller resection liver volume and better perioperative safety[44] (Quality of Evidence: Low). Overall, while most neoadjuvant HAIC studies are retrospective or small, results suggest neoadjuvant HAIC can improve diseasefree and OS in selected patients.
FOLFOX-HAIC combined with targeted neoadjuvant therapy
For technically resectable but biologically aggressive midtolate-stage HCC, preoperative combination systemic therapy shows promise. HAIC combined with TKIs has been explored as neoadjuvant therapy. Shi et al. reported a single-center retrospective study comparing HAIC combined with lenvatinib as neoadjuvant treatment followed by resection vs. upfront resection (82 patient vs. 72 patients): median OS favored the neoadjuvant group (40.1 months vs. 35.9 months, P = 0.001), and median PFS was 32.8 months vs. 23.8 months (P = 0.0023). Patients achieving radiologic complete response (CR) showed the most pronounced survival benefit - their median duration of response was not reached - whereas partial responders had a median duration of 28.9 months; CR patients had significantly longer OS than partial response (PR) patients (40.0 months vs. 29.1 months, P = 0.037). These results suggest deep preoperative responses positively impact prognosis. Preoperative adverse events were similar between groups (grade ≥ 3 AE (Adverse Event) 15.9% vs. 6.6%, P = 0.064) with no grade 4-5 events. Subgroup analysis indicated that patients with more advanced stage (BCLC intermediate/advanced) derived larger benefit (HR ≈ 0.47), while early-stage (BCLC A) patients did not clearly benefit[45] (Quality of Evidence: Low). Given the singlecenter retrospective design, selection bias is a limitation.
FOLFOX-HAIC combined with immunotherapy as neoadjuvant therapy
Immunotherapy has also advanced rapidly as a neoadjuvant modality in HCC. Combining HAIC with ICIs before surgery may achieve high rates of tumor necrosis and shrinkage in a short preoperative interval.
FOLFOX-HAIC combined with targeted-immunotherapy as neoadjuvant therapy
Triple therapy (HAIC combined with TKI and ICI) as neoadjuvant treatment is an evolving frontier.
DISCUSSION
This review synthesizes the evidence on the application of FOLFOX-HAIC in the conversion, neoadjuvant, and adjuvant therapy of HCC. The core finding indicates that FOLFOX-HAIC is evolving beyond a traditional palliative locoregional modality into a central strategy capable of profoundly altering the disease course. Its value is particularly pronounced when combined with targeted therapy and immunotherapy, enhancing objective response and conversion-to-surgery rates, thereby creating curative opportunities for patients who are initially unresectable or at high risk of recurrence, and significantly delaying postoperative relapse. Compelling evidence converges to show that for patients with macrovascular invasion, massive tumor burden, or poor response to conventional TACE, FOLFOX-HAIC-based strategies offer significantly superior tumor control and survival benefits compared to TACE alone.
Despite its promise, the integration of FOLFOX-HAIC into mainstream HCC management faces multiple challenges and uncertainties. First, there is a lack of standardization in treatment protocols. The optimal combination regimen, number of cycles, and sequencing remain undefined. Clinically, the decision to combine TACE with HAIC is typically based on anticipating scenarios where TACE monotherapy is likely ineffective or high-risk. These scenarios primarily include massive tumor burden (e.g., single tumor > 10 cm or multiple large tumors) with complex vasculature[9], making complete embolization difficult; the presence of major portal vein tumor thrombus (Vp3-4)[48], where embolization may exacerbate liver dysfunction or be ineffective; and imaging features predictive of TACE resistance, such as significant APS, which lead to washout of embolic agents, reducing TACE efficacy and increasing the risk of non-target embolization[49]. In these contexts, the combined strategy aims to synergize the ischemic effect of TACE with the sustained, high-concentration cytotoxic drug penetration from HAIC, overcoming tumor heterogeneity and hypoxic microenvironments to achieve deeper tumor kill. However, this combination significantly alters the adverse effect profile[50].Compared to TACE alone, which primarily causes post-embolization syndrome (fever, pain, transient transaminitis), the addition of FOLFOX-HAIC superimposes the classic toxicities of oxaliplatin and fluorouracil. This includes a higher frequency and severity of myelosuppression, peripheral neurotoxicity, and a greater risk of more severe and prolonged liver injury, such as drug-induced liver injury and sinusoidal obstruction syndrome[51]. Consequently, the decision to employ combination therapy necessitates a comprehensive evaluation of tumor biology, hepatic functional reserve, and patient performance status, carefully weighing the potential for deep response against the increased toxic burden.
Second, accurately distinguishing the etiology of liver injury is paramount when patients receiving combined FOLFOX-HAIC, targeted therapy, and immunotherapy develop abnormal liver function, as this dictates vastly different management strategies. Liver injury related to FOLFOX-HAIC often bears the hallmarks of regional chemotherapy, is temporally related to infusion cycles, and presents as transient elevations in transaminases and bilirubin[36]. Injury caused by targeted agents is often insidious, showing a progressive cholestatic or hepatocellular pattern related to continuous drug exposure[52]. In contrast, immune-related hepatitis induced by ICIs has a distinct timeline and biochemical signature[53]. Clinical differentiation requires integrating the drug administration history, injury pattern, autoantibody screening, and imaging findings to determine whether to adjust HAIC cycles, reduce or withhold targeted therapy, or initiate immunosuppressive treatment.
Furthermore, current evidence is limited by its hierarchy and methodological constraints. Most supporting data originate from retrospective studies and single-arm phase II trials, lacking large-scale phase III RCTs. This raises questions about the generalizability of the findings and leaves room for selection bias. A key cognitive limitation is the absence of precise predictive tools. The high heterogeneity of HCC means that not all patients benefit equally from intensive HAIC-based regimens. The current reliance on clinical staging for patient selection is crude, potentially exposing some patients to unnecessary treatment toxicity while withholding the most effective therapy from others who might benefit. Encouragingly, the identification of reliable predictive biomarkers has become a focal point of research to achieve personalized HAIC application, with preliminary progress being made. Explorations are underway across multiple dimensions: at the serological level, beyond alpha-fetoprotein (AFP), novel circulating factors such as baseline levels of the chemokine CCL28 and BTC have been associated with response to HAIC combined with targeted and immunotherapy[25,54,55]; liver function reserve [e.g., Child-Pugh class, Albumin-Bilirubin (ALBI) grade] remains the cornerstone for assessing treatment tolerance and survival benefit. At the imaging level, quantitative radiomic features extracted from contrast-enhanced computed tomography (CT) or magnetic resonance imaging (MRI), reflecting tumor perfusion and heterogeneity, offer a novel non-invasive approach to predict HAIC response and the likelihood of conversion resection[56]. Furthermore, pathological response assessment based on post-treatment specimens (e.g., degree of tumor necrosis) is itself a powerful prognostic indicator, and in-depth investigation into its underlying molecular features (e.g., markers related to angiogenesis or immune cell infiltration) is elucidating the mechanisms of treatment response. Dynamic early on-treatment assessment also holds significant value; for instance, the dynamic change in AFP levels (AFP response) after 1-2 cycles of HAIC can serve as an early indicator for predicting ultimate efficacy and guiding subsequent decisions[57]. Although these emerging biomarkers still require validation in large-scale prospective cohorts, they collectively mark a substantive move in the field beyond sole reliance on clinical staging towards achieving precision medicine.
Based on the above analysis, we propose a hierarchical and integrated treatment framework based on therapeutic goals and tumor biology. In this framework, FOLFOX-HAIC is not a standalone technique but a dynamic “platform” for combination. For advanced patients with high tumor burden where “conversion” is the goal, FOLFOX-HAIC should act as a “potentiator” for systemic therapy, forming the core of a powerful induction regimen. For resectable patients with high-risk features, FOLFOX-HAIC can serve as a “cleaner” in the neoadjuvant or adjuvant setting to eradicate micrometastases and reduce recurrence risk.
Furthermore, it is imperative to contextualize FOLFOX-HAIC within the broader spectrum of locoregional therapies for HCC, particularly in comparison to transarterial radioembolization (TARE) with yttrium-90 (Y90). While direct head-to-head studies are currently scarce, emerging data offer valuable insights into their respective niches. A recent network meta-analysis positioned oxaliplatin-based HAIC favorably for OS benefit in advanced HCC. In intermediate-stage disease, especially with large or multinodular tumors, FOLFOX-HAIC has shown superior objective response and conversion rates over conventional TACE, underscoring its potency for downstaging[58]. Conversely, Y90-TARE, particularly in the form of radiation segmentectomy for early-stage solitary HCC, demonstrates exceptional efficacy in achieving profound local tumor control and durable responses. The cited 2018 study reports that a tumoricidal dose (> 190 Gy) can achieve complete pathological necrosis, with 5-year local tumor control rates of 72% and survival outcomes comparable to curative-intent therapies for selected patients.[59]. This evolving landscape suggests that therapeutic choice may be guided by specific clinical scenarios: FOLFOX-HAIC, particularly in combination with systemic therapy, appears optimal for aggressive downstaging in advanced, high-tumor-burden disease, whereas Y90-TARE may be a superior ablative strategy for localized disease control or bridging to transplantation. Ultimately, the integration of both locoregional modalities with systemic targeted and immunotherapy represents a key frontier, as exemplified by ongoing trials such as the ROWAN study (TheraSphere With Durvalumab and Tremelimumab for Hepatocellular Carcinoma; ClinicalTrials.gov NCT05063565).
Beyond clinical efficacy and safety, the widespread adoption of FOLFOX-HAIC faces significant practical and systemic barriers critical for healthcare planning. Its implementation is highly resource-intensive, demanding specialized interventional radiology expertise for precise catheter management, dedicated pharmacy support, and nursing teams proficient in prolonged infusion protocols - requirements that may restrict its availability to major tertiary centers. Additionally, its cost-effectiveness relative to established modalities such as TACE remains an unresolved question. Although FOLFOX-HAIC may improve conversion rates and survival, the substantial upfront costs of the procedure, implanted devices, chemotherapeutic agents, and extended hospitalization necessitate comprehensive health economic analyses across diverse healthcare settings. Equitable access is also a pressing concern, as disparities in infrastructure and reimbursement policies could exacerbate existing inequalities in HCC care. Addressing these barriers through specialized training, protocol simplification, and rigorous cost-utility studies is essential to translate the demonstrated benefits of FOLFOX-HAIC into broader real-world practice.
To overcome current limitations and establish its standard role, future research must prioritize several concrete directions. The foremost task is to conduct rigorously designed multicenter phase III trials directly comparing FOLFOX-HAIC-based combinations against standard therapies across HCC stages, which is fundamental for securing definitive guideline recommendations. Concurrently, research must focus on integrating multi-omics data to construct composite predictive models, emphasizing early on-treatment dynamic changes as indicators for real-time therapeutic adjustment. There is an urgent need to accelerate the discovery and validation of predictive biomarkers to optimize patient selection, maximize efficacy, and minimize toxicity. Optimizing combination regimens and technical standards through systematic research is equally crucial to define efficacy-safety boundaries and standardize procedures for catheter management, drug infusion, and toxicity monitoring. Finally, dedicated health economics and implementation science research are needed to evaluate cost-effectiveness and develop strategies to promote equitable access.
CONCLUSION
In conclusion, FOLFOX-HAIC is evolving from a complementary locoregional modality into a cornerstone of multimodal HCC therapy. With the accumulation of evidence and ongoing clinical innovation, we can anticipate a future where standardized and individualized HAIC strategies will expand surgical opportunities, reduce recurrence, and improve long-term outcomes for patients with HCC. Ongoing clinical research and multidisciplinary collaboration will be pivotal to realizing the full potential of HAIC-based combination therapies and bringing new hope to patients affected by this formidable disease.
DECLARATIONS
Authors’ contributions
Conceptualization and study design: Zhu C, Peng T
Literature search and data curation: He Y, Mo S, Wang Y, Shao J
Formal analysis: He Y, Mo S
Writing - original draft: Wang Y, Shao J, Zhu C
Writing - review and editing: Zhu C
Critical revision for important intellectual content: Han C
Supervision and project administration: Peng T
Review, editing, and final approval of the manuscript: Zhu C
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool Deepseek (DeepSeek-V4, released 2026) was used solely for language editing. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
This work was supported by the Medical Excellence Award Funded by the Creative Research Development Grant from the First Affiliated Hospital of Guangxi Medical University (Grant No. 2021006), the First-class Discipline Innovation-driven Talent Program of Guangxi Medical University, the Guangxi Medical and Health Appropriate Technology Development and Application Project (Nos. S2021100 and S2022065), the National Natural Science Foundation of China (Nos. 81802874 and 82260548), the Natural Science Foundation of the Guangxi Province of China (Grant No. 2024GXNSFAA010347), the Guangxi Key R&D Program (GKEAB18221019), and the Guangxi Key Laboratory of Enhanced Recovery After Surgery for Gastrointestinal Cancer.
Conflicts of interest
Peng T is an Editorial Board Member of the journal Hepatoma Research. Peng T was not involved in any steps of the editorial processing, notably including reviewers’ selection, manuscript handling, or decision-making. The other authors declare that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007;132:2557-76.
2. Saito A, Toyoda H, Kobayashi M, et al. Prediction of early recurrence of hepatocellular carcinoma after resection using digital pathology images assessed by machine learning. Mod Pathol. 2021;34:417-25.
3. Singh SP, Madke T, Chand P. Global epidemiology of hepatocellular carcinoma. J Clin Exp Hepatol. 2025;15:102446.
4. Chen S, Yuan B, Yu W, Wang X, He C, Chen C. Comparison of arterial infusion chemotherapy and chemoembolization for locally advanced hepatocellular carcinoma: a multicenter retrospective study. J Gastrointest Surg. 2022;26:2292-300.
5. Pan Y, Yuan Z, Wang J, et al. Survival benefit and impact of adjuvant therapies following FOLFOX-HAIC-based conversion therapy with unresectable hepatocellular carcinoma: a retrospective cohort study. J Cancer Res Clin Oncol. 2023;149:14761-74.
6. Guyatt GH, Oxman AD, Vist GE, et al.; GRADE Working Group. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336:924-6.
7. Zhang W, Zhang K, Liu C, et al. Hepatic arterial infusion chemotherapy combined with anti-PD-1/PD-L1 immunotherapy and molecularly targeted agents for advanced hepatocellular carcinoma: a real world study. Front Immunol. 2023;14:1127349.
8. Tanaka T. Transarterial chemoembolization for hepatocellular carcinoma: current role and techniques. Interv Radiol. 2025;10:e20240016.
9. Li QJ, He MK, Chen HW, et al. Hepatic arterial infusion of oxaliplatin, fluorouracil, and leucovorin versus transarterial chemoembolization for large hepatocellular carcinoma: a randomized phase III trial. J Clin Oncol. 2022;40:150-60.
10. He M, Li Q, Zou R, et al. Sorafenib plus hepatic arterial infusion of oxaliplatin, fluorouracil, and leucovorin vs sorafenib alone for hepatocellular carcinoma with portal vein invasion: a randomized clinical trial. JAMA Oncol. 2019;5:953-60.
11. Zhou H, Song T. Conversion therapy and maintenance therapy for primary hepatocellular carcinoma. Biosci Trends. 2021;15:155-60.
12. Wang J, Zheng Z, Wu T, et al. Hepatic arterial infusion chemotherapy as a timing strategy for conversion surgery to treat hepatocellular carcinoma: a single-center real-world study. J Hepatocell Carcinoma. 2022;9:999-1010.
13. Zhu C, Ren X, Liu D, Zhang C. Oxaliplatin-induced hepatic sinusoidal obstruction syndrome. Toxicology. 2021;460:152882.
14. Zheng K, Zhu X, Fu S, et al. Sorafenib plus hepatic arterial infusion chemotherapy versus sorafenib for hepatocellular carcinoma with major portal vein tumor thrombosis: a randomized trial. Radiology. 2022;303:455-64.
15. Yang M, Jiang X, Liu H, et al. Efficacy and safety of HAIC combined with tyrosine kinase inhibitors versus HAIC monotherapy for advanced hepatocellular carcinoma: a multicenter propensity score matching analysis. Front Pharmacol. 2024;15:1410767.
16. Abou-Alfa GK, Meyer T, Cheng AL, et al. Cabozantinib in patients with advanced and progressing hepatocellular carcinoma. N Engl J Med. 2018;379:54-63.
17. Li H, Li R, Zhang X, et al. Ivonescimab (PD-1/VEGF bispecific antibody) combined with HAIC for first-line treatment of unresectable hepatocellular carcinoma (uHCC): a prospective, single-arm phase II study. JCO. 2025;43:e16212.
18. Zhao M, Lyu N, Zhong S, et al. 983P Safety and efficacy of durvalumab plus hepatic artery infusion chemotherapy in HCC with severe portal vein tumor thrombosis (Vp3/4) - the DurHope study. Ann Oncol. 2023;34:S608.
19. Liu D, Mu H, Liu C, et al. Sintilimab, bevacizumab biosimilar, and HAIC for unresectable hepatocellular carcinoma conversion therapy: a prospective, single-arm phase II trial. Neoplasma. 2023;70:811-8.
20. Xin Y, Cao F, Yang H, et al. Efficacy and safety of atezolizumab plus bevacizumab combined with hepatic arterial infusion chemotherapy for advanced hepatocellular carcinoma. Front Immunol. 2022;13:929141.
21. Pan L, Sun R, He Q, et al. Hepatic artery infusion chemotherapy (HAIC) combined with tislelizumab and lenvatinib for initial unresectable hepatocellular carcinoma (HCC) with portal vein tumor thrombus: a prospective, single-arm phase II trial. J Clin Oncol. 2024;42:4103.
22. Zhao Z, Jiang X, Wen S, Hao Y. Efficiency and safety of HAIC combined with lenvatinib and tislelizumab for advanced hepatocellular carcinoma with high tumor burden: a multicenter propensity score matching analysis. Front Pharmacol. 2024;15:1499269.
23. Liu G, Zhu D, He Q, et al. Hepatic arterial infusion chemotherapy combined with lenvatinib and PD-1 inhibitors for managing arterioportal shunt in hepatocellular carcinoma with portal vein tumor thrombus: a retrospective cohort study. J Hepatocell Carcinoma. 2024;11:1415-28.
24. Chen S, Shi F, Wu Z, et al. Hepatic arterial infusion chemotherapy plus lenvatinib and tislelizumab with or without transhepatic arterial embolization for unresectable hepatocellular carcinoma with portal vein tumor thrombus and high tumor burden: a multicenter retrospective study. J Hepatocell Carcinoma. 2023;10:1209-22.
25. Lai Z, He M, Bu X, et al. Lenvatinib, toripalimab plus hepatic arterial infusion chemotherapy in patients with high-risk advanced hepatocellular carcinoma: a biomolecular exploratory, phase II trial. Eur J Cancer. 2022;174:68-77.
26. Luo L, Xiao Y, Zhu G, et al. Hepatic arterial infusion chemotherapy combined with PD-1 inhibitors and tyrosine kinase inhibitors for unresectable hepatocellular carcinoma: a tertiary medical center experience. Front Oncol. 2022;12:1004652.
27. Zhang N, Zhang Y, Yu B, et al. Adjuvant HAIC combined with anlotinib and TQB2450 for resected high-risk hepatocellular carcinoma. Innovation. 2025;6:100910.
28. Lin ZP, Hu XL, Chen D, et al. Efficacy and safety of targeted therapy plus immunotherapy combined with hepatic artery infusion chemotherapy (FOLFOX) for unresectable hepatocarcinoma. World J Gastroenterol. 2024;30:2321-31.
29. Li R, Wang X, Li H, et al. Hepatic arterial infusion chemotherapy combined lenvatinib and PD-1 inhibitor showed improved survival for infiltrative hepatocellular carcinoma: a multicenter cohort study. J Hepatocell Carcinoma. 2024;11:1727-40.
30. Yang Y, Sun J, Cai J, et al.; Chinese Association of Liver Cancer and Chinese Medical Doctor Association. Chinese expert consensus on the whole-course management of hepatocellular carcinoma (2023 Edition). Liver Cancer. 2025;14:311-33.
31. Zhang W, Zhao X, Gao W, et al. Conversion study of hepatocellular carcinoma using HAIC combined with lenvatinib and PD-1/L1 immunotherapy under the guidance of BCLC staging. Front Immunol. 2025;16:1596864.
32. Nevola R, Ruocco R, Criscuolo L, et al. Predictors of early and late hepatocellular carcinoma recurrence. World J Gastroenterol. 2023;29:1243-60.
33. Chi QY, Liu QY, Wang SJ, et al. Lenvatinib combined with anti-PD-1 antibodies plus locoregional treatment for initial unresectable hepatocellular carcinoma with portal vein tumor thrombosis: a multicenter real-world study. BMC Cancer. 2025;25:1162.
34. Liu Y, Wang Y, Guo X, et al. Comparative effectiveness of adjuvant treatment for resected hepatocellular carcinoma: a systematic review and network meta-analysis. Front Oncol. 2021;11:709278.
35. Hu L, Zheng Y, Lin J, Shi X, Wang A. Does adjuvant hepatic artery infusion chemotherapy improve patient outcomes for hepatocellular carcinoma following liver resection? A meta-analysis. World J Surg Oncol. 2023;21:121.
36. Li SH, Mei J, Cheng Y, et al. Postoperative adjuvant hepatic arterial infusion chemotherapy with FOLFOX in hepatocellular carcinoma with microvascular invasion: a multicenter, phase III, randomized study. J Clin Oncol. 2023;41:1898-908.
37. Wen Y, Lu L, Mei J, et al. Hepatic arterial infusion chemotherapy vs transcatheter arterial chemoembolization as adjuvant therapy following surgery for MVI-positive hepatocellular carcinoma: a multicenter propensity score matching analysis. J Hepatocell Carcinoma. 2024;11:665-78.
38. Long Y, Huang Z, He X, et al. Postoperative adjuvant hepatic arterial infusion chemotherapy combined with donafenib in hepatocellular carcinoma with solitary large tumor and microvascular invasion: a multicenter, prospective, single-arm, phase II trial. J Clin Oncol. 2024;42:e16227.
39. Liang Y, Zhong D, Shang J, et al. Efficacy and safety of postoperative adjuvant HAIC with FOLFOX combining PD-1 inhibitors in HCC patients with microvascular invasion: a propensity score matching analysis. BMC Cancer. 2025;25:418.
40. Yang J, Jiang S, Chen Y, Zhang J, Deng Y. Adjuvant ICIs plus targeted therapies reduce HCC recurrence after hepatectomy in patients with high risk of recurrence. Curr Oncol. 2023;30:1708-19.
41. Ouyang J, Yang Y, Zhou Y, et al. Adjuvant camrelizumab plus apatinib in resected hepatocellular carcinoma with microvascular invasion: a multi-center real world study. Hepatobiliary Surg Nutr. 2024;13:616-31.
42. Singal AG, Yarchoan M, Yopp A, Sapisochin G, Pinato DJ, Pillai A. Neoadjuvant and adjuvant systemic therapy in HCC: current status and the future. Hepatol Commun. 2024;8:e0430.
43. Hu Z, Yang Z, Pan Y, et al. Survival benefit of preoperative hepatic arterial infusion of oxaliplatin, fluorouracil, and leucovorin followed by hepatectomy for hepatocellular carcinoma. Front Pharmacol. 2023;14:1210835.
44. Pan Y, Mei J, Chen J, et al. Comparison between portal vein perfusion chemotherapy and neoadjuvant hepatic arterial infusion chemotherapy for resectable intermediate to advanced stage hepatocellular carcinoma. Ann Surg Oncol. 2022;29:2016-29.
45. Shi Y, Chen K, Li X, et al. Prognosis of neoadjuvant HAIC and lenvatinib followed by surgery versus direct resection for resectable or borderline resectable hepatocellular carcinoma: a real-world study. J Hepatocell Carcinoma. 2024;11:2063-76.
46. Tao P, Wei Y, Zhu G, et al. 187P Neoadjuvant cadonilimab (PD-1/CTLA-4 bispecific antibody) plus transhepatic arterial infusion chemotherapy (HAIC) for resectable multinodular CNLC Ib/IIa hepatocellular carcinoma (Car-Hero). Ann Oncol 2023;34:S1547-8. Available from: https://www.annalsofoncology.org/article/S0923. [Last accessed on 24 Apr 2026].
47. Wu X, Wang Y, Wang S, et al. Neoadjuvant targeted immunotherapy followed by surgical resection versus upfront surgery for hepatocellular carcinoma with macrovascular invasion: a multicenter study. J Cancer. 2024;15:3024-33.
48. Yuan Y, He W, Yang Z, et al. TACE-HAIC combined with targeted therapy and immunotherapy versus TACE alone for hepatocellular carcinoma with portal vein tumour thrombus: a propensity score matching study. Int J Surg. 2023;109:1222-30.
49. Zhao W, Liu C, Wu Y, et al. Transarterial chemoembolization (TACE)-hepatic arterial infusion chemotherapy (HAIC) combined with PD-1 inhibitors plus lenvatinib as a preoperative conversion therapy for nonmetastatic advanced hepatocellular carcinoma: a single center experience. Transl Cancer Res. 2024;13:2315-31.
50. Wu C, Dong Y, Li X, et al. TACE-HAIC versus HAIC combined with TKIs and ICIs for hepatocellular carcinoma with a high tumor burden - a propensity-score matching comparative study. Front Immunol. 2025;16:1664756.
51. Yang J, Shang X, Li J, Wei N. Comparative study on the efficacy and safety of transarterial chemoembolization combined with hepatic arterial infusion chemotherapy for large unresectable hepatocellular carcinoma. J Gastrointest Oncol. 2024;15:346-55.
52. Abza GB, De Vos K, Annaert P. Inhibition of ABC transporters by sorafenib and lenvatinib: implications for drug-induced cholestasis. Xenobiotica. 2025;55:99-109.
53. Hountondji L, Ferreira De Matos C, Lebossé F, et al. Clinical pattern of checkpoint inhibitor-induced liver injury in a multicentre cohort. JHEP Rep. 2023;5:100719.
54. Tang HH, Zhang MQ, Zhang ZC, et al. Prognostic nutritional index predicts survival in intermediate and advanced hepatocellular carcinoma treated with hepatic arterial infusion chemotherapy combined with PD-(L)1 inhibitors and molecular targeted therapies. BMC Cancer. 2025;25:603.
55. Lv X, Zhang PB, Zhang EL, Yang S. Predictive factors and prognostic models for Hepatic arterial infusion chemotherapy in hepatocellular carcinoma: a comprehensive review. World J Surg Oncol. 2025;23:166.
56. Zhao Y, Huang F, Liu S, et al. Prediction of therapeutic response of unresectable hepatocellular carcinoma to hepatic arterial infusion chemotherapy based on pretherapeutic MRI radiomics and Albumin-Bilirubin score. J Cancer Res Clin Oncol. 2023;149:5181-92.
57. Zhu AX, Dayyani F, Yen CJ, et al. Alpha-fetoprotein as a potential surrogate biomarker for atezolizumab + bevacizumab treatment of hepatocellular carcinoma. Clin Cancer Res. 2022;28:3537-45.
58. Li J, Yang B, Teng Z, Liu Y, Li D, Qu X. Efficacy and safety of first-line treatments for advanced hepatocellular carcinoma patients: a systematic review and network meta-analysis. Front Immunol. 2024;15:1430196.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].