Current research status and progress of laser therapy in wound healing
Abstract
A wound is a disruption in the integrity of the skin or other body tissues. The application of laser therapy can facilitate microscopic debridement by utilizing photothermal action to remove necrotic tissue and employing photobiological regulation to expedite epithelial growth while reducing the duration of acute inflammation. Laser therapy can regulate Heat Shock Proteins, Matrix Metalloproteinases, and inflammatory factors in wound tissues. This promotes wound re-epithelialization and organizes collagen fibers, helping to reduce scar formation. Additionally, laser therapy utilizes photochemical action for sterilization and photomechanical action to disrupt bacterial biofilms, thereby facilitating drug delivery and absorption. Numerous studies have substantiated the efficacy of laser therapy in promoting chronic wound re-epithelialization, diminishing scarring in acute wounds, and enhancing both the speed and quality of overall wound healing. However, further large-scale studies are required to explore the optimal parameter range for laser wound therapy. Consequently, laser therapy represents a promising option for managing various types of wounds, offering prospects for further reduction in scar formation.
Keywords
INTRODUCTION
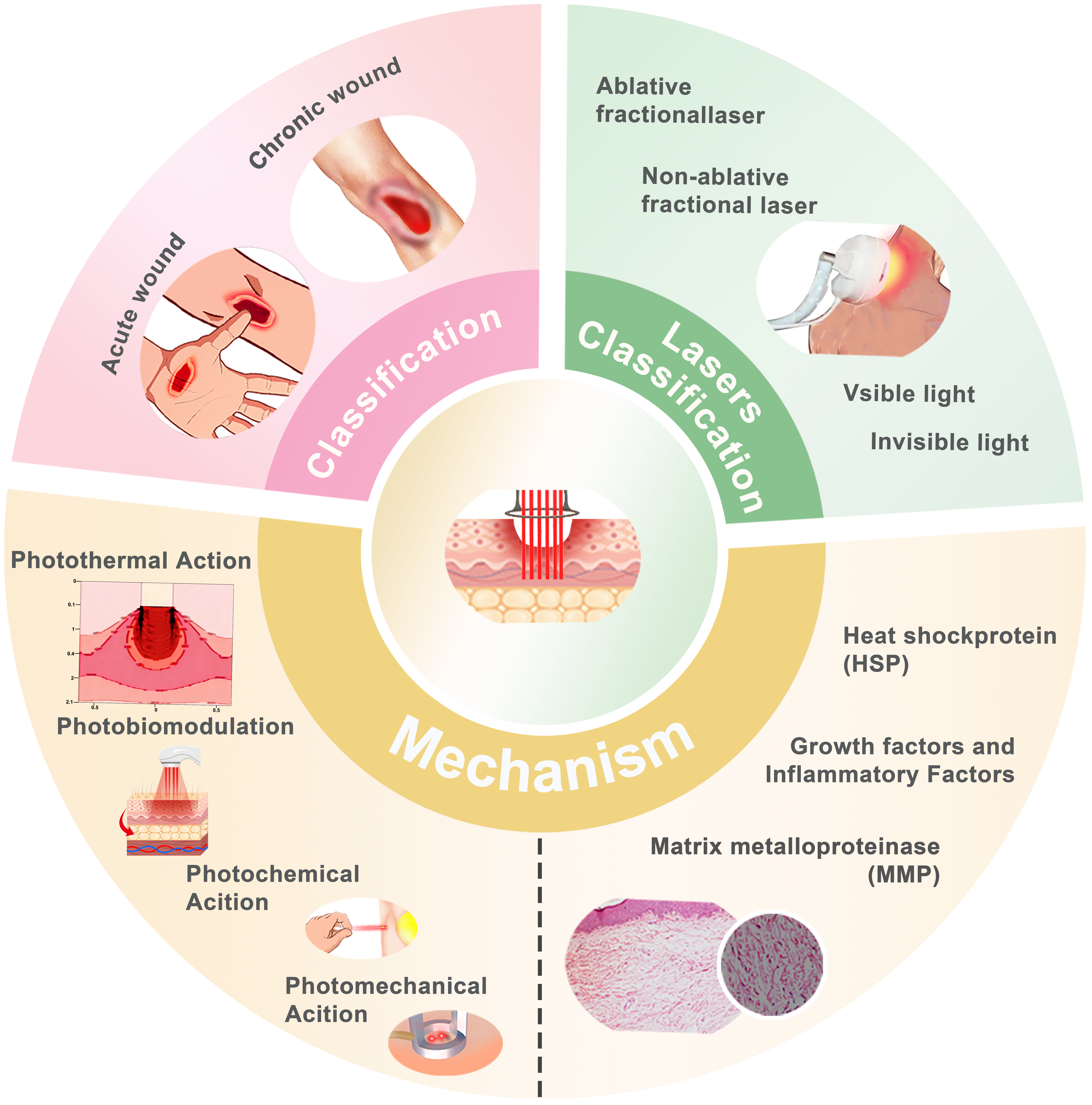
A wound refers to damage to normal tissue caused by external and internal injury factors, often resulting in compromised skin integrity and the loss of healthy tissue. Acute and chronic wounds lack strict temporal demarcation; typically, a wound that is prolonged and difficult to heal is classified as a chronic wound[1]. The etiology of chronic wounds is diverse, encompassing pressure ulcers, diabetic skin ulcers, venous ulcers, burns or post-traumatic residual wounds, radiation ulcers, cancerous ulcers, and more[2]. Acute wounds are usually caused by trauma or surgery.
Various therapy options are available for wounds, such as debridement, negative pressure suction technology, and growth factor application; however, these methods can be associated with prolonged therapy times and high costs[3,4]. The long-term treatment of many chronic refractory wounds places significant economic and psychological burdens on patients[5-7]. In developed countries, it is estimated that 2%-5% of total healthcare spending is allocated to chronic wound care[8]. For acute wound treatment, primary methods include debridement and suturing, but these methods may present challenges such as significant pain, extensive tissue defects, high costs, and poor efficacy.
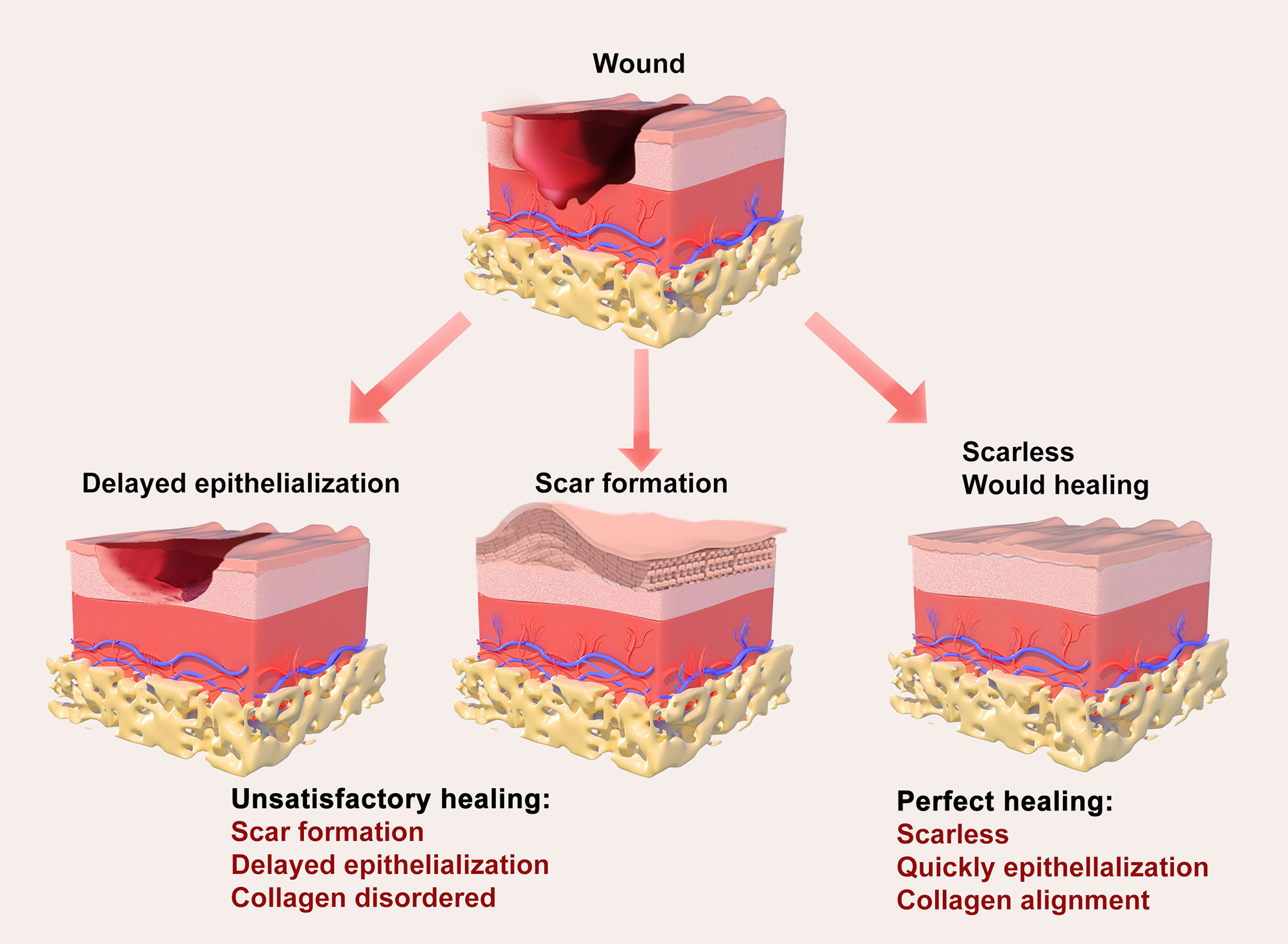
The main challenges in wound healing are delayed epithelialization and scar formation. Delayed epithelialization not only escalates therapy costs but also imposes significant psychological and physical burdens on patients. Excessive scarring can greatly impact patients, causing issues such as pain, itching, contractures, and movement limitations[8,9]. Thus, promoting wound re-epithelization and reducing scar formation represent major clinical challenges.
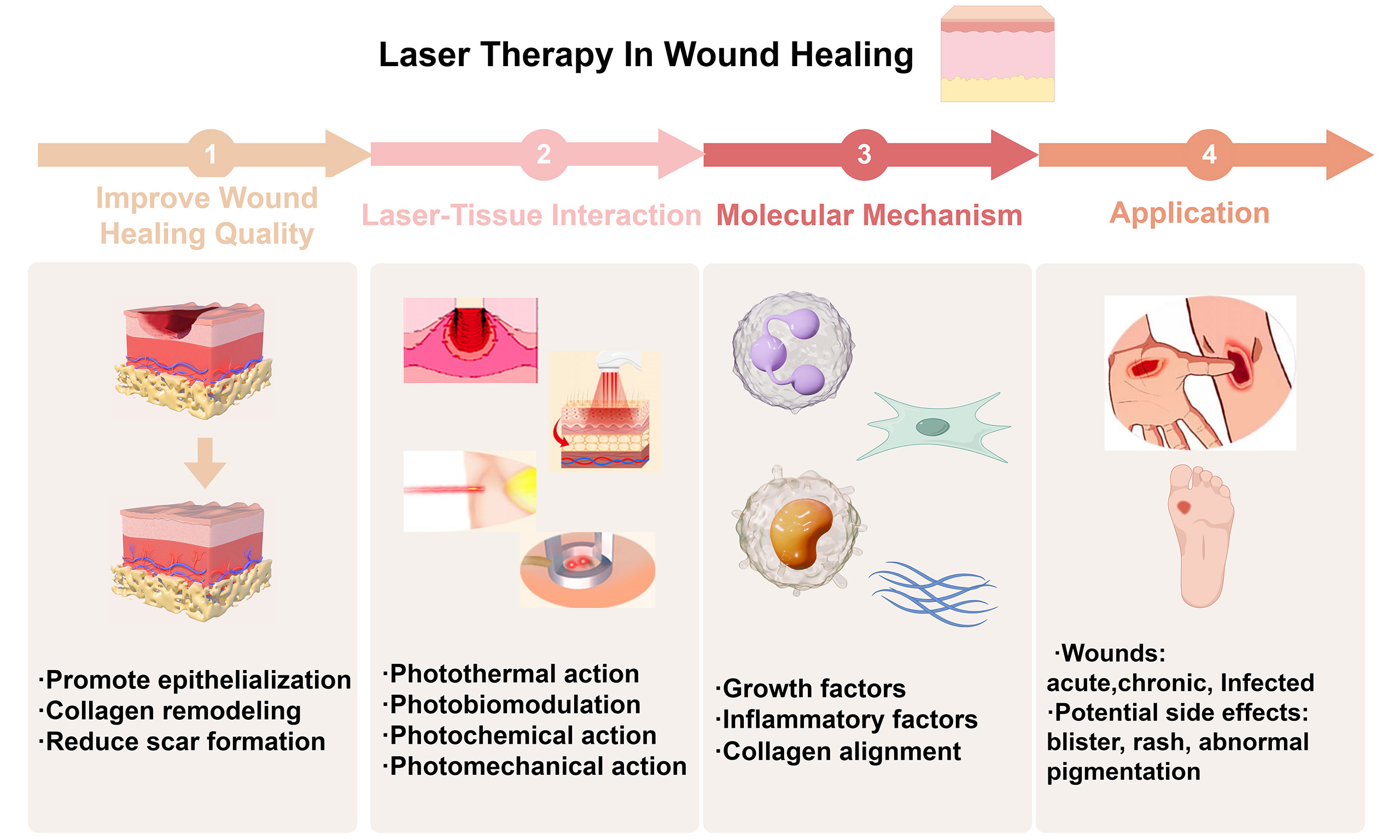
Recent studies suggest that laser therapy can upregulate tissue growth factors via photobiological regulation, thereby promoting wound re-epithelialization. In addition to conventional non-surgical and surgical therapies for scars, recent research has validated the effectiveness of lasers in preventing and treating scars[10]. Several medical institutions have employed laser therapy for acute and chronic wounds, demonstrating its effectiveness and safety, though there is still a lack of large-scale systematic studies. This review summarizes the relevant mechanisms and previous studies on how laser therapy can promote wound re-epithelization and reduce scar formation, offering new possibilities for wound treatment [Figure 1].
WOUND HEALING
Wound healing involves the regeneration of the dermis and epidermis following skin injury. It is a complex, dynamic process that consists of three overlapping stages: inflammation, proliferation, and remodeling. With the progression of wound healing and the completion of the remodeling phase, scar formation follows.
The primary aim of wound therapy is tissue regeneration to promote healing. Effective debridement is essential for good wound bed preparation, aiming to remove barriers such as necrotic tissue, senescent and non-migrating cells, bacteria, and foreign bodies[11]. These barriers interfere with healing and impede the migration of keratinocytes across the wound bed, preventing epithelialization[11]. Debridement plays a crucial role in promoting wound healing, with various options available, including surgical, autolytic, enzymatic, biological, ultrasonic, and “water knife” debridement[12,13].
Surgical debridement is associated with significant pain, requires anesthesia, and can result in extensive tissue damage, imposing high demands on the patient’s overall condition[12]. Autolytic debridement creates a moist wound environment to facilitate the dissolution of necrotic tissue using endogenous enzymes; however, it is time-consuming and does not effectively control infection. Enzymatic debridement can be costly and may inhibit immune cell function, while biological debridement may lead to poor patient acceptance and dependence[14]. In contrast, ultrasonic debridement allows for the selective removal of necrotic tissue with minimal pain and trauma but is unsuitable for wounds on the head, neck, or perineum[15,16]. “Water knife” debridement is a relatively new method, but it is limited by its accuracy in removing necrotic tissue[17,18].
In addition to traditional debridement methods, laser debridement offers an innovative approach. This technique enables precise debridement, effectively alleviating pain, minimizing bleeding, and reducing healing time[19-21]. The application of laser therapy on wounds results in minimal tissue damage at a low cost, making it a valuable alternative worth considering.
Regarding the choice of debridement degree, chronic wounds (e.g., diabetic foot ulcers, venous ulcers) require more frequent and thorough debridement tailored to individual wound characteristics. This involves removing necrotic tissue and biofilms to promote granulation tissue formation[22,23]. In acute wounds (e.g., surgical wounds and burns), caution is necessary during debridement, as excessive removal can enlarge the wound and delay healing[23]. Enzymatic debridement is effective for scab removal in burn wounds[24,25]. For infected wounds (e.g., necrotic soft tissue infections), early and extensive debridement followed by negative pressure wound therapy (NPWT) is essential to enhance healing rates[26,27].
Reducing scar formation is another aim of wound therapy. A range of therapeutic strategies is employed to minimize and prevent scars. Non-surgical therapies include corticosteroids, fluorouracil, and stress therapy, while more aggressive approaches involve various surgical procedures and skin grinding[28,29]. However, surgical interventions often result in linear scars, and non-surgical therapies carry the risk of scar hyperplasia and contracture.
Laser therapy has emerged as a preferred treatment for many types of scars[30-32]. The core mechanism of laser therapy for scarring is the synergistic effect of physical disruption, molecular signal modulation and combination therapy to achieve collagen remodeling, vascular normalization and inflammation abatement, ultimately improving the appearance and function of the scar[33,34]. It meets patients’ needs for non-invasive and cosmetic solutions in scar management. The National Expert Consensus on Early Management of Scars[35] and the 2018 Expert Consensus on the Therapy of Skin Traumatic Scars with Photoelectric Technology[36] both advocate for comprehensive anti-scar therapy as soon as possible, with ablative fractional laser (AFL) being a key therapeutic modality [Figure 2].
Lasers used for wound therapy generally fall into two categories: fractional lasers (FL) and light therapy. Common AFLs include CO2 lasers and erbium-doped yttrium aluminium garnet (Er:YAG) laser therapy. Light therapy is further divided into visible light therapy, which includes red and blue light therapies, and invisible light therapy, which includes infrared and ultraviolet therapies. FL promotes wound healing through a molecular cascade involving heat shock proteins (HSPs), matrix metalloproteinases (MMPs), and inflammatory responses. Collagen remodeling, potentially achieved by reducing type I collagen and increasing type III collagen, improves the final texture and thickness of scars while accelerating the healing of unstable wounds.
Existing studies have confirmed that FL can promote the healing of chronic wounds that are difficult to treat. Additionally, FL has been shown to improve the quality of healing in early wounds, resulting in lighter final scars[37-39]. However, the specific mechanism remains unclear and may be related to the effects of lasers on skin and micro-regulation [Table 1].
Classification of lasers for wound therapy
| Lasers | Wavelength (nm) | Target | Indication |
| Ablative fractional laser[40] | |||
| CO2 | 10,600 | Water | Vaporization and stripping, thermal effect |
| Er:YAG | 2,940 | Water | Vaporization and stripping, thermal effect |
| Non-ablative fractional laser[40] | |||
| Affirm | 1,470, 1,340, 694 | Water | Thermal effect |
| Fraxel | 1,550 | Water | Thermal effect |
| Visible light[41] | |||
| Red light | 620-700 | Hemoglobin | Photochemical effect |
| Blue light | 400-500 | - | Photochemical effect |
| Invisible light[41] | |||
| Near-infrared light | 700-1,440 | - | Photobiomodulation |
Mechanisms of laser-tissue interaction
Photothermal action
High-intensity lasers have a significant thermal effect on tissue[42,43]. The combined action of temperature and time can result in heat coagulation, leading to cell necrosis, hemostasis, and changes in the extracellular matrix (ECM). During laser therapy, the temperature of vaporized tissue is often above 100 °C, resulting in rapid drying and charring of the wound[44]. When using a CO2 laser with continuous low-energy output, the skin tissue temperature fluctuates between 120-200 °C without causing carbonization. The CO2 laser acts at a depth of about 20 µm, but thermal coagulation damage can occur at depths of approximately 4 mm due to heat conduction effects, resulting in tissue solidification, drying, and even scorching. This ablative effect can be utilized in scar therapy and micro-debridement. There was a threshold effect of laser light on wounds, with excessive energy leading to tissue damage. For example, a dose range of 4-8 J/cm2 shows optimal healing in animal models[45].
Under thermal stimulation, tissue initiates regeneration and rearrangement within the collagen fibers. As a result, laser debridement offers more precise micro-debridement while minimizing damage to surrounding tissue. Selective photothermal action involves selecting the appropriate wavelength, energy, and pulse width based on the biological characteristics of different tissues, ensuring effective treatment of pathological tissue while minimizing damage to healthy surrounding tissue. At specific wavelengths, lasers can target vessels or pigmented cells with minimal impact on adjacent tissues[46].
Scars and vascular diseases can be treated using the selective photothermal action of lasers. In particular, the 585 nm pulsed dye laser is effective in closing micro-vessels and treating scars by inhibiting excessive wound vascularization, reducing blood vessel density, and nutrient delivery to achieve the desired effect of reducing scar formation caused by abnormal hyperplasia of local micro-vessels[46]. Previous studies concluded that moderate vascularization of tissue promotes wound re-epithelization, while excessive vascularization is associated with scar formation[47]. Therefore, laser therapy may effectively close microvasculature and reduce scar formation. A laser can also remove tattoos and pigments. If the skin absorbs high-energy photons in a short period, it may lead to the disintegration of physical tissue. The ultra-short pulse width and peak power energy are absorbed by pigment particles, producing a photoacoustic effect that results in pressure accumulation and subsequent rupture of these particles. The disintegrated pigment particles are then more easily ingested and metabolized by antigen-presenting cells, achieving depigmentation effects[48,49].
In summary, the effects of laser on wounds through photothermal action include: (1) coagulating necrotic tissue for debridement; (2) promoting wound re-epithelization by facilitating collagen fiber rearrangement; and (3) selectively acting on microvessels or pigmented nevi to inhibit excessive vascularization or pigmentation of tissues.
Photobiomodulation
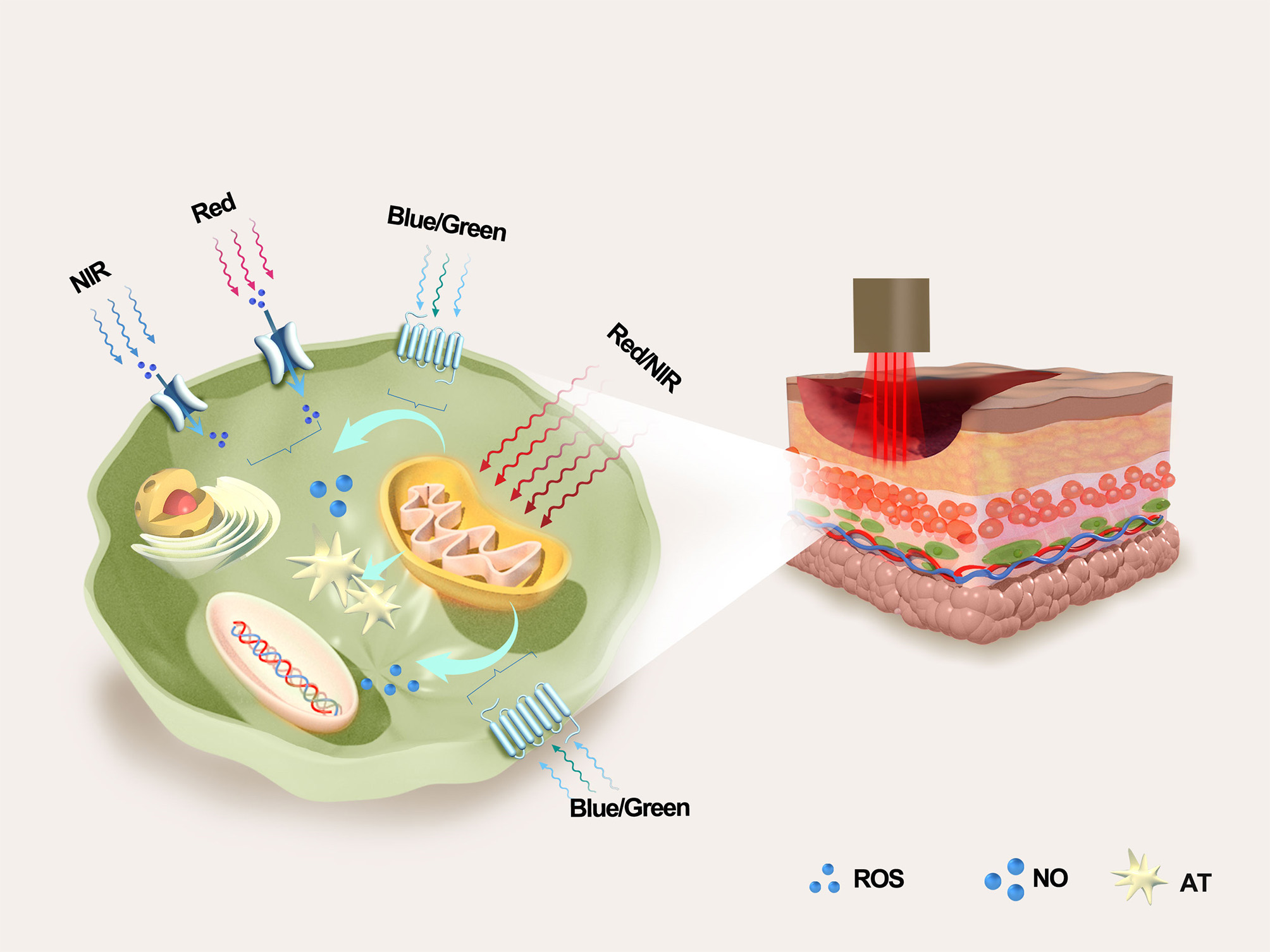
The mechanisms of laser-tissue interaction encompass not only photothermal effects but also photobiomodulation (PBM). PBM, also known as low-level laser therapy (LLLT), utilizes lasers or light emitting diode (LED) light to produce low-intensity light. PBM is non-invasive and has been shown to decrease inflammation and provide pain relief[50]. Endre Mester observed that exposing the shaved backs of mice to low-energy lasers accelerated hair regeneration and promoted wound healing[51]. This phenomenon is now referred to as the “photobiomodulation” effect[52,53]. Since then, significant progress has been made in the basic research and clinical application of low-energy lasers in wound healing and other fields. The effectiveness of PBM follows the Arndt-Schulz law: low doses (1-10 J/cm2) are effective, whereas higher doses can inhibit the therapeutic effect. For example, the 660 nm laser optimally promotes vascular proliferation at 1.0 J/cm2[54,55].
The pathways of PBM action primarily include the mitochondrial cytochrome c oxidase (CCO)-adenosine triphosphate (ATP) pathway, the dissociated nitric oxide (NO) pathway, reactive oxygen release, oxidation–reduction (REDOX) signaling pathways, membrane receptors, and photosensitive ion channels[50,56,57]. Light-sensitive target chromophores generated from the mitochondria include ATP, reactive oxygen species (ROS), and NO, which enter an excited state after absorbing specific wavelengths of light. This leads to accelerated electron transfer within the mitochondrial respiratory chain, increased ATP synthesis, and changes in the REDOX state of the mitochondria[58]. Although the CCO-ATP signaling pathway is the core mechanism of PBM, its efficacy demonstrates significant dependence on wavelength and dose. For instance, studies have revealed that an 810 nm laser effectively promotes healing in a porcine skin wound model, whereas a 1,064 nm laser exhibits no significant effect under identical parameters. This indicates that longer wavelengths may necessitate distinct parameter windows due to diminished CCO absorption efficiency[59].
Simultaneously, there is an increase in the release of downstream growth factors. The binding of these growth factors to receptors on the cell surface induces signaling pathways that transmit signals to the
In conclusion, the effects of PBM on wounds include restoring cell function, reducing inflammation and pain, and enhancing tissue regeneration and wound healing[63] [Figure 3].
Figure 3. Chromophores involved in PBM include mitochondrial CCO, membrane receptors such as opsins and light-sensitive ion channels. The three main effectors (ATP, ROS, and NO) are generated by the mitochondria, as shown here. PBM: photobiomodulation; CCO: cytochrome c oxidase; ATP: Adenosine triphosphate; ROS: reactive oxygen species; NO: nitric oxide; CNG: cyclic nucleotide-gated ion channel; NIR: near-infrared; AT: active thiol species.
Photochemical action
The photochemical effect refers to the chemical reactions triggered by the absorption of energy from external photons by the molecules of a substance[64]. Currently, the most common application of photochemical effects is photodynamic therapy (PDT), which utilizes the chemical action of lasers to sterilize and effectively treat contaminated wounds[65]. This meta-analysis incorporated data from 6 randomized controlled trials (RCTs), encompassing 458 patients with 467 diabetic foot ulcers (DFUs), to evaluate the efficacy and safety of PDT versus SOC (standard of care) in managing DFU. The study suggested that PDT might offer an enhanced healing rate for DFUs compared to SOC alone, potentially leading to improved patient outcomes[66]. The energy of photochemistry is dependent on the photosensitizer concentration and the duration of exposure. For example, blue light (400-500 nm) requires precise energy control to avoid cytotoxicity[67,68]. In conclusion, the photochemical effect of lasers on wounds can sterilize the wound.
Photomechanical action
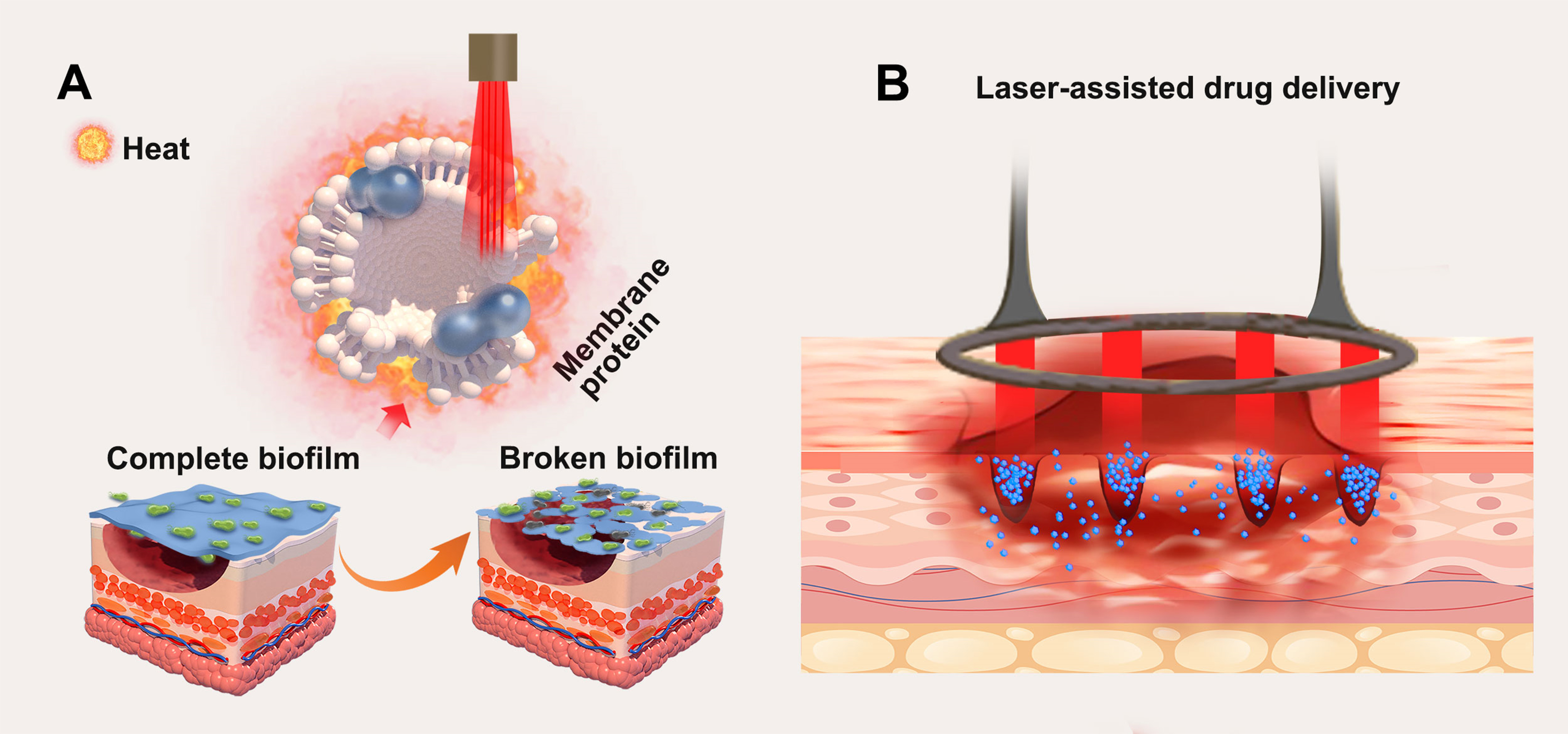
The application of photomechanical action involves using pressure waves generated by laser energy to mechanically disrupt bacterial biofilms [Figure 4A]. The presence of pathogenic bacteria on wounds has been found to delay healing. Various lasers, especially AFL, operate in a repair mode focused on the microthermal zones (MTZ). This laser stimulates wound regeneration by creating microcellular thermal holes within the epidermis and dermis. These pores not only remove aged and necrotic tissue but also promote the regeneration of both the dermis and epidermis. Through these microscopic thermal holes, oxygen and circulation within the wound are increased, effectively blocking the action of biofilms while preserving up to 95% of the dermis intact. As a result, laser-induced damage can be repaired more rapidly.
Figure 4. Laser therapy in wounds can (A) destroy the biofilm on the wound surface; (B) increase drug delivery through MTZ. MTZ: Microcellular thermal damage zone.
In 2017, Nathan confirmed that lasers can generate shock waves to destroy biofilms and significantly reduce bacterial load. Furthermore, medications such as antibiotics, silicone, or hormone drugs can be administered via MTZ, increasing the area of contact between the drug and the tissue, thereby enhancing drug absorption rates[69,70] [Figure 4A].
In summary, lasers can significantly promote wound epithelialization[20,21,71]. The effects of PBM on wounds include reducing inflammation and pain, as well as enhancing tissue regeneration and healing [Figure 4B]. The photochemical effect of lasers can sterilize wounds. Through photomechanical action, lasers effectively reduce bacterial load, destroy biofilms, and improve drug delivery. However, the interaction between lasers and tissue constitutes a continuous, dynamically evolving process. At the instant of laser application to tissue, photothermal effects invariably occur. The selection of different laser types, wavelengths, energy densities, pulse widths, and treatment modalities determines which effect predominates during the therapeutic process. The ultimate outcome represents a dynamic balance of all parameters.
Molecular mechanism of laser therapy in wound healing
Growth factors and inflammatory factors
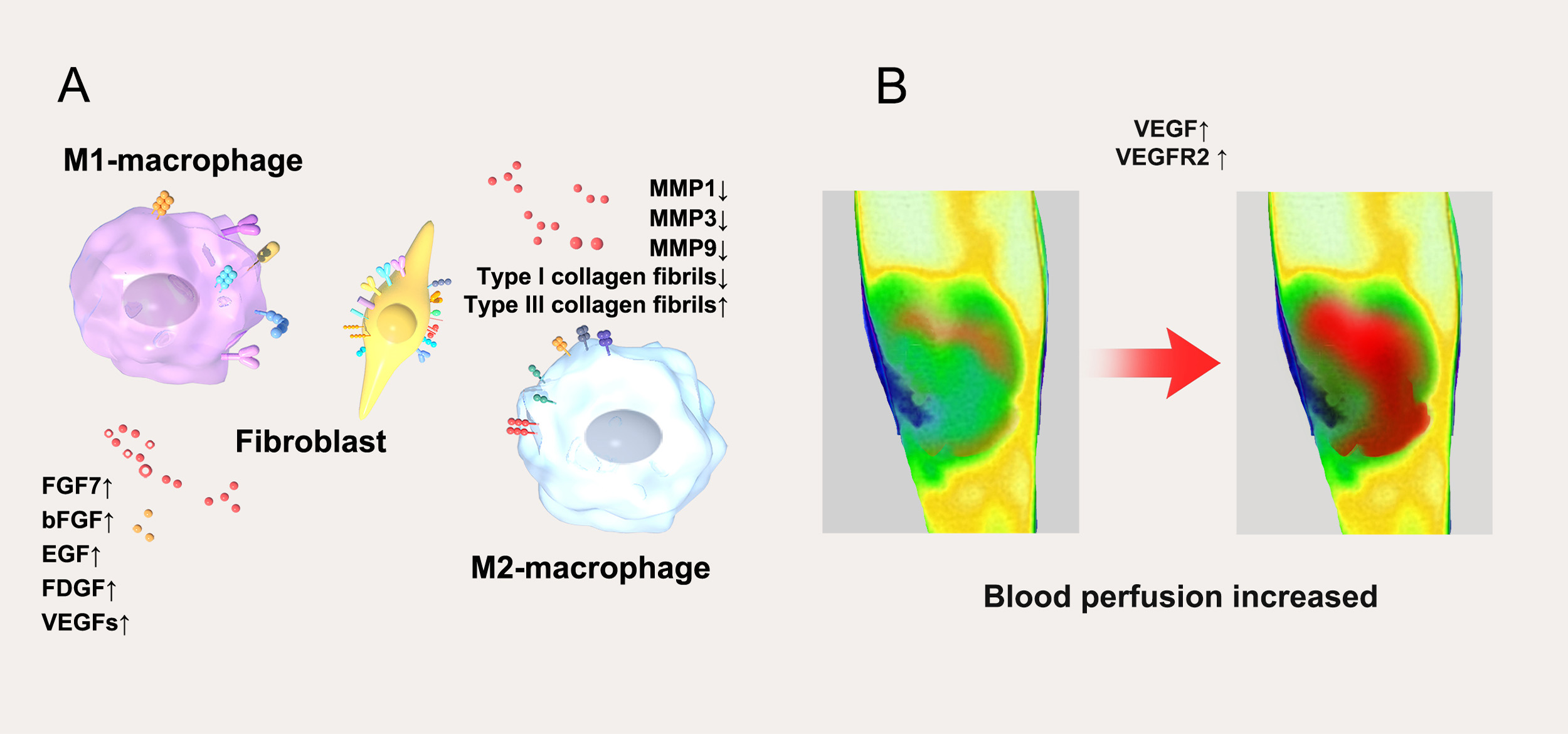
Various lasers, such as AFL and Er:YAG lasers, initiate the involvement of neutrophils and macrophages in the initial stages of wound healing. The highly organized process of wound healing requires coordinated regulation of cell proliferation, migration, and tissue remodeling, primarily mediated by polypeptide growth factors, including members of the fibroblast growth factor family[72]. Previous studies have demonstrated the significance of keratinocyte growth factor fibroblast growth factor 7 (FGF7) in promoting wound healing by regulating the production and degradation of the ECM; FGF7 expression increases with CO2 laser therapy[73]. Additionally, vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF) play crucial roles in angiogenesis. Laser therapy can stimulate the production of various growth factors, including bFGF, epidermal growth factor (EGF), platelet-derived growth factor (PDGF), and VEGF[71,74]. Compared to surgical debridement, laser therapy results in increased local blood flow signals, confirming that it promotes angiogenesis and tissue regeneration[75,76] [Figure 5A].
Figure 5. Laser therapy in wounds resulted in (A) upregulation of growth factors and downregulation of inflammatory factors; (B) increased blood perfusion of the wound. M1-macrophage: Classically activated macrophage; FGF7: fibroblast growth factor 7; bFGF: basic fibroblast growth factor; EGF: epidermal growth factor; FDGF: fibroblast-derived growth factor; VEGFs: vascular endothelial growth factors; MMP: matrix metalloproteinase; M2-macrophage: alternatively activated macrophage; VEGF: vascular endothelial growth factor; VEGFR2: vascular endothelial growth factor receptor 2.
Laser therapy can enhance blood perfusion in wounds. This study involved 54 patients with chronic wounds, who were randomly divided into two groups. One group received traditional sharp instrument/surgical debridement [routine treatment (RT) group], while the other group was treated with CO2 laser debridement [laser treatment (LT) group]. Results indicated that the LT group exhibited a significantly higher percentage of wound perfusion compared to normal periwound skin perfusion (calculated as wound perfusion/normal periwound skin perfusion × 100%) on the 1st, 7th, and 14th days after therapy. Blood perfusion in the wounds increased significantly on the second day post-treatment and continued to rise until day 14. These findings suggest that laser therapy promotes an increase in wound blood perfusion[77].
The underlying mechanism may be related to the biological effects of the laser on the wound. Laser stimulation increases the levels of VEGF and its receptor (VEGFR2) in the skin, thereby enhancing blood perfusion in the wound. This has also been confirmed in previous studies[78,79]. VEGF is a crucial pro-angiogenic factor that binds to its receptor on the cell surface, triggering signaling cascades that significantly influence the process of neovascularization [Figure 5B]. Therefore, laser-induced hyperaemia is not an isolated physiological phenomenon but a crucial mechanism driven by the VEGF/VEGFR2 signaling axis. This mechanism directly addresses core clinical objectives: accelerating wound bed preparation and reducing tissue hypoxia, offering significant therapeutic value - particularly for ischaemic chronic wounds such as diabetic foot ulcers. Recent animal studies further confirm that laser pretreatment administered one week prior to flap surgery significantly enhances flap survival rates[80] [Figure 5B].
Tissue injury is associated with acute inflammation and the invasion of neutrophils. Chemokines are known to regulate the recruitment of neutrophils to the wound site during acute inflammatory responses. Several studies have indicated that chemokines also play roles in regulating epithelialization, tissue remodeling, and angiogenesis, highlighting their importance in wound healing[71]. Research by Schmitt et al. has shown that chemokine levels in human skin increase after laser therapy. Specifically, increased expression of C-X-C motif chemokine ligand 12 (CXCL12) was observed during burn wound healing, indicating its significant role in post-burn recovery. CXCL12 is also a chemokine for hematopoietic stem cells; elevating its levels in the wound may enhance the recruitment of stem cells to promote healing[81-83].
Laser therapy induces a shift from a chronic inflammatory state to an acute inflammatory state. Once inflammation is resolved and the wound is cleared of contamination, healing progresses to the proliferative stage. The transition from the inflammatory to the proliferative phase is a key step in healing. Analysis of skin samples obtained at indicated times after laser therapy using real-time polymerase chain reaction for gene expression shows a significant increase in levels of interleukin (IL)-1β and IL-8 in the wound following treatment. This study demonstrates that laser therapy can influence the expression of inflammatory mediators in human skin in vivo, inducing a shift from a chronic inflammatory state to an acute one[71,84]. Once in the proliferative phase, keratinocytes begin to proliferate and migrate across the wound bed to re-epithelialize the wound[85,86].
The coordinated changes in the aforementioned growth factors and inflammatory mediators collectively underpin the clinical benefits observed with laser therapy. The induction of VEGF and bFGF is directly linked to enhanced blood perfusion in chronic wounds and accelerated granulation tissue formation. Concurrently, the laser-induced shift from chronic to acute inflammation - characterized by a transient elevation followed by a rapid decline of early inflammatory mediators such as IL-1β and IL-8 - represents a critical step in overcoming chronic wound healing stagnation and driving progression into the proliferative phase. This inflammatory 'reset,' combined with subsequent upregulation of growth factors such as FGF7, synergistically promotes keratinocyte migration and re-epithelization.
HSPs and MMP
HSPs significantly increase following the elevation of local skin temperature due to laser therapy. They not only protect tissues from injury but also play crucial roles in tissue remodeling and wound healing. The heightened expression of Heat Shock Protein (HSP)27 helps reduce cell damage during emergency states[87]. Additionally, HSP72, HSP47, and HSP70 support normal skin remodeling and minimize scar formation[71]. Researchers have observed an increase in HSP content after treating normal skin with lasers, noting that HSP70 and HSP72 levels began to rise one hour post-treatment, peaked between 3 and 14 days, and gradually decreased thereafter[88,89]. Furthermore, HSP47 is linked to collagen folding and secretion. A study by Köhler et al. revealed that skin levels of HSP47 increased seven days after laser therapy and persisted for over three months[89]. The enhanced expression of HSP70 promotes fibroblast proliferation and migration, while HSP90 supports fibroblast migration and granulation tissue formation, both contributing synergistically to protein remodeling[90-92]. CO2 laser therapy has been shown to immediately induce the expression of HSP47 and HSP70 in scars while maintaining them within a reasonable range, promoting normal transforming growth factor beta (TGF-β) expression at the site treated for MTZ repair. However, the comprehensive effects of long-term HSP expression on scars and wounds remain unclear, necessitating further studies.
MMPs play a complex and crucial role in wound healing and scar formation[93]. During physiological healing, specific MMPs - such as MMP-1, MMP-2, MMP-3, and MMP-9 - are essential for keratinocyte migration, granulation tissue formation, and ECM remodeling[93]. For instance, MMP-1, also known as interstitial collagenase, hydrolyzes intact fibrillar collagen. Studies have shown that long-pulse neodymium-doped yttrium aluminum garnet (Nd:YAG) laser therapy can affect MMP-1 expression, with varying outcomes depending on energy levels one month post-treatment. At low energy, MMP-1 expression was lower, while slightly higher energy resulted in increased MMP-1 expression. MMP-3 is believed to facilitate fibroblast contraction and initiate wound contraction[94]. Research by Jeffery indicates that the laser-induced inflammatory response from CO2 lasers stimulates the production of new collagen, which may benefit acute and chronic refractory wounds, atrophic scars, and other non-inflammatory conditions[71]. MMP-9 is upregulated early in re-epithelization to drive keratinocyte migration[93], while MMP-1 initiates the degradation of type I and III collagens, creating conditions for subsequent new collagen deposition and remodeling. However, in chronic wounds and hypertrophic scars, persistent inflammatory signals lead to dysregulated expression of MMPs (and their tissue inhibitors, TIMPs), resulting in excessive ECM degradation or abnormal deposition. This imbalance hinders healing and promotes pathological scar formation[95].
In summary, lasers promote wound healing and scar repair through a mechanism partly rooted in their precise regulation of MMPs. Laser-induced moderate inflammation triggers the essential expression of MMPs to drive tissue regeneration, while subsequent modulation of the MMP/TIMP guides ECM remodeling toward a more normal, ordered structure. This process not only ensures effective healing but also enhances scar texture, color, and elasticity, and minimizes scar hyperplasia.
THE APPLICATION OF LASER THERAPY IN WOUND HEALING
Laser therapy has emerged as a versatile tool in both cosmetic and wound care, demonstrating unique advantages in wound management. Research increasingly supports the efficacy of lasers not only for scar management but also for wound debridement and therapy.
The first report on the use of CO2 laser therapy for chronic wounds appeared in 2012, showcasing its success in treating three chronic refractory wounds. Notably, chronic ulcers associated with old scars healed within two weeks of treatment[96]. In 2017, a study utilizing 2,940 nm Er:YAG laser therapy for a non-healing burn wound reported successful healing after five months, with significant improvements in scar texture and flexibility during follow-up. Researchers suggested that laser-induced MTZ promotes healing “from the outside in”, facilitating granulation tissue development and new collagen formation[97].
Despite promising findings, scientific animal studies validating these results are limited. One study involving diabetic mice showed that CO2 laser therapy on wound edges significantly promoted healing across varying pulse energies. Notably, low-fluence AFL (2 mJ/spot) was as effective as higher fluences (6 and
Subsequent systematic case studies primarily focused on challenging chronic wounds have demonstrated varying degrees of efficacy for AFL in promoting healing. A retrospective cohort study involving 164 patients indicated significantly improved wound outcomes in the CO2 laser group compared to conventional treatments, with reductions in wound area and improved Bates-Jensen Wound Assessment Tool (BWAT) scores.
AFL not only enhances healing but also exhibits anti-infective properties. A randomized controlled study involving 54 patients confirmed that laser debridement effectively reduces bacterial load and patient discomfort compared to traditional surgical methods. Microbial infection, often leading to biofilm formation, significantly hinders healing[99]. Laser therapy’s photothermal action allows for biofilm removal while increasing oxygen levels in the wound and reducing anaerobic bacterial colonization. Additionally, laser therapy has been shown to upregulate regulatory inflammatory factors, decreasing TGF-β levels while increasing VEGF expression to promote vascular hyperplasia[98].
Early intervention is critical for minimizing scar formation, and AFL has been shown to effectively promote healing while also serving as a preventive measure against scarring. For instance, the study by Rohrer and Ugent (2002) on YAG laser therapy for surgical incisions reported no side effects, and while results were not statistically significant, scar appearance improved post-therapy[100]. In 2017, Du et al. applied AFL to 10 acute wounds in Asian patients before scar repair surgery, resulting in significant aesthetic improvements after eight months[33].
Karmisholt et al. conducted a study with 32 postoperative patients that compared treated and untreated surgical scars, finding that early non-ablative fractional laser (NAFL) therapy improved scar outcomes. In another study, Hiren assessed the impact of 2,940 nm Er:YAG laser therapy on surgical scars, demonstrating that early intervention post-surgery improved scar appearance[21].
Despite several clinical studies confirming AFL’s potential in enhancing scar outcomes for acute wounds, the specific mechanisms underlying these effects remain unclear. Moreover, there is a notable lack of large-scale, long-term randomized controlled studies with uniform intervention timing to further validate the effectiveness and mechanisms of scar prevention [Table 2].
Previous research on laser therapy in wounds
| Study | Laser type & device | Wound | Dosage parameters (SI units) | Sample size | Classification | FDA/NMPA approval status (for indication) | Key conclusion |
| Lee HN[98] | CO2 (AFL, Lutronic) | Diabetic mice | Energy density: 2, 6, 12 mJ/cm2; Spot density: 300 spots/cm2; Spot size: 120 μm; Frequency: 1-2/week | N = 8 | AFL | CO2 AFL is FDA-approved for skin resurfacing and scar treatment | AFL therapy effectively promoted wound healing in diabetic mouse models |
| Jiang B[99] | CO2 (ML-2030CI) | Chronic human wounds | Energy density: 60-140 mJ/cm2 (defocused); Dwell time: 4-6 s; Passes: continuous mode, single session | N = 54 | AFL | Device approved. Laser debridement is a well-established clinical practice; specific approval varies by region | The CO2 laser exhibits higher accuracy, more effectively controls wound infection, and promotes an increase in wound blood perfusion |
| Tania JP[101] | CO2 (AFL, Lutronic Deep FX) | Chronic human wounds | Energy density: 30 or 50 mJ/cm2; Density: 5%; Frequency: 150 Hz; Passes: 1, single session | N = 3 (cases) | AFL | FDA-approved for skin resurfacing. Use for chronic wounds represents evidence-based practice extension | Lasers promote wound healing and remodel collage |
| Babak H[19] | Er:YAG | Chronic human wounds | Energy density: 50 J/cm2; Spot size: 4 mm; Dwell time: 0.5 s; Overlap: 50%; Passes: 1-2 | N = 22 | AFL | FDA/NMPA-approved for skin ablation. Debridement is a common and accepted clinical application | Laser debridement can effectively reduce the bacterial load within the wound and alleviate patient discomfort |
| Du F[33] | CO2 (AFL, AcuPulse®) | Acute human wounds | Energy (Deep): 20-22.5 mJ, Density: 5%; Energy (Superficial): 80-100 mJ, Density: 40%; Passes: 2 (combined); Sessions: 2 | N = 10 | AFL | FDA-approved for scar revision. Parameters fall within the approved treatment range for scar management | All patients exhibited significant improvement in aesthetic scars |
| Karmisholt KE[21] | Erbium-glass (NAFL, Icon) | Acute human wounds | Energy/microbeam: 40-50 mJ; Density: 115 microbeams/cm2; Stacks/passes: 3 stacks, 2 passes; Session: 1 | N = 32 | NAFL | FDA-approved for treatment of moderate to severe facial wrinkles and acne scars | NAFL-treated scars showed subtle improvement compared with untreated control scars |
| Zhang Y[102] | CO2 (AFL, Lumenis) | Acute human wounds | Energy density: 10-15 mJ; Density: 5%; Frequency: 300 Hz; Sessions: 1-3 (3-month intervals) | N = 18 | AFL | FDA-approved for scar treatment. Multi-session approach is standard clinical protocol | All the patients in the therapy group had significant improvements in their surgical scars after the laser therapy |
Laser therapy also has side effects in wound healing. If the laser energy is high, it may cause delayed tissue repair or excessive inflammation due to thermal damage[103,104]; if the laser is not followed by standardized care, it may cause hyperpigmentation or hypopigmentation[105,106]; if the laser energy is too high or the post-laser cooling treatment is not in place, it may cause local oedema, blisters, pain, and other discomforts[107,108]. A study by John compiled side effects following 163 treatments with different laser types, revealed that postoperative pain was the most common side effect (37%), followed by blistering (27%), hypopigmentation (12%), fever (10%) and rash (7%)[108,109]. It is necessary for the operator to standardize the parameters according to the type and depth of wounds, and to combine with standardized care, to minimize the risks. Prior to laser treatment, special attention must be given to mitigating complication risks in patients who have recently used photosensitizing medications or who have a history of abnormal scarring. During the procedure, both the clinician and the patient are required to wear appropriate protective eyewear. Following laser treatment, immediate and thorough cutaneous cooling is essential; strict photoprotection measures - including broad-spectrum sunscreen use, physical sun avoidance, and protective clothing - must be instituted; and mechanical irritation (e.g., scratching or rubbing) must be actively avoided. Early recognition and prompt, proactive management of potential complications are critical. Collectively, these precautions significantly reduce the incidence of post-laser side effects.
CONCLUSION
In summary, laser therapy enhances wound healing through its photothermal effects and PBM. It effectively regulates local HSP, MMP, and cytokines at a microscopic level, promoting healing, reducing inflammation, and facilitating collagen remodeling. Compared to traditional surgical debridement, AFL offers advantages such as decreased patient pain, reduced bleeding, and precise tissue removal. For chronic wounds, laser therapy improves blood perfusion, inhibits infection, and accelerates healing. Additionally, AFL therapy applied to acute wounds can help minimize scar formation. However, further large-scale studies are required to explore the optimal parameter range for laser wound therapy and to clarify the mechanisms by which lasers act on the wound surface. Prior to implementing laser therapy on wounds, all healthcare personnel must undergo safety training to minimize the occurrence of associated side effects. As a progressive and effective treatment modality, laser wound therapy shows great promise as a novel therapeutic approach in wound management, offering prospects for further reduction in scar formation.
DECLARATIONS
Acknowledgments
The Graphical Abstract was created with Figdraw (www.figdraw.com, ID: WTOOS5c700).
Authors’ contributions
Conception and design of the study: Ni X
Acquisition of data: Liu X, Zhao X, Zhuang Z
Drafting of the manuscript: Liu X
Critical revision of the manuscript: Ni X, Chen K
All authors have read and approved the final manuscript.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool ChatGPT (version GPT-4, released 2023-03-14) was used solely for language editing. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
None.
Conflict of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Kyaw BM, Järbrink K, Martinengo L, Car J, Harding K, Schmidtchen A. Need for improved definition of “chronic wounds” in clinical studies. Acta Derm Venereol. 2018;98:157-8.
3. Rezaie F, Momeni-Moghaddam M, Naderi-Meshkin H. Regeneration and repair of skin wounds: various strategies for treatment. Int J Low Extrem Wounds. 2019;18:247-61.
4. Martinengo L, Olsson M, Bajpai R, et al. Prevalence of chronic wounds in the general population: systematic review and meta-analysis of observational studies. Ann Epidemiol. 2019;29:8-15.
5. Olsson M, Järbrink K, Divakar U, et al. The humanistic and economic burden of chronic wounds: a systematic review. Wound Repair Regen. 2019;27:114-25.
6. Graves N, Phillips CJ, Harding K. A narrative review of the epidemiology and economics of chronic wounds. Br J Dermatol. 2022;187:141-8.
7. Maxant G, Pastrav M, Gogeneata I, Bajcz C, Bertaux AC. Clinical and medico-economic benefits of remote monitoring of chronic wounds. Int Wound J. 2025;22:e70140.
8. Nussbaum SR, Carter MJ, Fife CE, et al. An economic evaluation of the impact, cost, and medicare policy implications of chronic nonhealing wounds. Value Health. 2018;21:27-32.
9. Hsieh JC, Maisel-Campbell AL, Joshi CJ, Zielinski E, Galiano RD. Daily quality-of-life impact of scars: an interview-based foundational study of patient-reported themes. Plast Reconstr Surg Glob Open. 2021;9:e3522.
10. Pirakitikulr N, Martin JJ, Wester ST. Laser resurfacing for the management of periorbital scarring. Plast Aesthet Res. 2020;7:67.
12. Wynn M. The benefits and harms of cleansing for acute traumatic wounds: a narrative review. Adv Skin Wound Care. 2021;34:488-92.
13. Warriner RA 3rd, Wilcox JR, Carter MJ, Stewart DG. More frequent visits to wound care clinics result in faster times to close diabetic foot and venous leg ulcers. Adv Skin Wound Care. 2012;25:494-501.
14. De Decker I, De Graeve L, Hoeksema H, et al. Enzymatic debridement: past, present, and future. Acta Chir Belg. 2022;122:279-95.
15. Liu E, Hu X, Zhang W, et al. Efficacy and safety of ultrasound-assisted wound debridement in the treatment of diabetic foot ulcers: a systematic review and meta-analysis of 11 randomized controlled trials. Front Endocrinol. 2024;15:1393251.
16. Kataoka Y, Kunimitsu M, Nakagami G, Koudounas S, Weller CD, Sanada H. Effectiveness of ultrasonic debridement on reduction of bacteria and biofilm in patients with chronic wounds: a scoping review. Int Wound J. 2021;18:176-86.
17. Bowers S, Franco E. Chronic wounds: evaluation and management. Am Fam Physician. 2020;101:159-66.
18. Legemate CM, Kwa KAA, Goei H, et al.; HyCon Study Group. Hydrosurgical and conventional debridement of burns: randomized clinical trial. Br J Surg. 2022;109:332-9.
19. Hajhosseini B, Chiou GJ, Dori G, et al. Er:YAG laser vs. sharp debridement in management of chronic wounds: effects on pain and bacterial load. Wound Repair Regen. 2020;28:118-25.
20. Sherrill JD, Finlay D, Binder RL, et al. Transcriptomic analysis of human skin wound healing and rejuvenation following ablative fractional laser treatment. PLoS One. 2021;16:e0260095.
21. Karmisholt KE, Banzhaf CA, Glud M, et al. Laser treatments in early wound healing improve scar appearance: a randomized split-wound trial with nonablative fractional laser exposures vs. untreated controls. Br J Dermatol. 2018;179:1307-14.
22. Parikh UM, Mentz J, Collier I, et al. Strategies to minimize surgical scarring: translation of lessons learned from bedside to bench and back. Adv Wound Care. 2022;11:311-29.
23. Nube VL, Alison JA, Twigg SM. Diabetic foot ulcers: weekly versus second-weekly conservative sharp wound debridement. J Wound Care. 2023;32:383-90.
24. Ditmars FS, Kay KE, Broderick TC, Fagg WS. Use of amniotic membrane in hard-to-heal wounds: a multicentre retrospective study. J Wound Care. 2024;33:S44-50.
25. Mathioudaki E, Vitsos A, Rallis MC. Proteolytic enzymes and wound debridement: a literature review. Wounds. 2024;36:357-65.
26. Afzal H, Dawson E, Fonseca R, et al. Negative pressure wound therapy with and without instillation in necrotizing soft tissue infections. Surg Infect. 2024;25:199-205.
27. Matthews MR, Fernández LG, Hermans MH, Chakravarthy D. The efficient application of instilling negative pressure wound therapy with a hypochlorous acid-preserved wound cleanser: a case series and practical advice. Wounds. 2024;36:148-53.
28. Gold MH, McGuire M, Mustoe TA, et al.; International Advisory Panel on Scar Management. Updated international clinical recommendations on scar management: part 2 - algorithms for scar prevention and treatment. Dermatol Surg. 2014;40:825-31.
29. Yang X, Lohsiriwat V, Chang FCS, et al. Real-world management of abnormal scarring using topical silicone gel: expert consensus and case series from the Asian SCARS Expert Group. Drugs Context. 2023;12.
30. Seago M, Shumaker PR, Spring LK, et al. Laser treatment of traumatic scars and contractures: 2020 International Consensus Recommendations. Lasers Surg Med. 2020;52:96-116.
31. Ma Y, Barnes SP, Chen YY, Moiemen NS, Lord JM, Sardeli AV. Influence of scar age, laser type and laser treatment intervals on adult burn scars: a systematic review and meta-analysis. PLoS One. 2023;18:e0292097.
32. Anderson RR, Donelan MB, Hivnor C, et al. Laser treatment of traumatic scars with an emphasis on ablative fractional laser resurfacing: consensus report. JAMA Dermatol. 2014;150:187-93.
33. Du F, Yu Y, Zhou Z, Wang L, Zheng S. Early treatment using fractional CO2 laser before skin suture during scar revision surgery in Asians. J Cosmet Laser Ther. 2018;20:102-5.
34. Nunez JH, Strong AL, Comish P, et al. A review of laser therapies for the treatment of scarring and vascular anomalies. Adv Wound Care. 2023;12:68-84.
35. Zhang Y; Chinese Association of Plastics and Aesthetics Scar Medicine Branch. National expert consensus on early management of scars (2020 version). Zhonghua Shao Shang Za Zhi. 2021;37:113-25. (in Chinese).
36. Lv K, Xia Z; Chinese Consensus Panel on the Prevention and Treatment of Scars. Chinese expert consensus on clinical prevention and treatment of scar. Burns Trauma. 2018;6:27.
37. Shen S, Cai Y, Song X, Xiang W. The efficacy of fractional carbon dioxide laser in surgical scars treatment: a system review and meta-analysis. Aesthetic Plast Surg. 2023;47:340-50.
38. Friedman O, Gofstein D, Arad E, Gur E, Sprecher E, Artzi O. Laser pretreatment for the attenuation of planned surgical scars: a randomized self-controlled hemi-scar pilot study. J Plast Reconstr Aesthet Surg. 2020;73:893-8.
39. Aoki A, Mizutani K, Taniguchi Y, et al. Current status of Er:YAG laser in periodontal surgery. Jpn Dent Sci Rev. 2024;60:1-14.
40. Haykal D, Cartier H, Goldberg D, Gold M. Advancements in laser technologies for skin rejuvenation: a comprehensive review of efficacy and safety. J Cosmet Dermatol. 2024;23:3078-89.
41. Maghfour J, Ozog DM, Mineroff J, Jagdeo J, Kohli I, Lim HW. Photobiomodulation CME part I: overview and mechanism of action. J Am Acad Dermatol. 2024;91:793-802.
43. Thomsen S. Pathologic analysis of photothermal and photomechanical effects of laser-tissue interactions. Photochem Photobiol. 1991;53:825-35.
44. Walsh JT Jr, Flotte TJ, Deutsch TF. Er:YAG laser ablation of tissue: effect of pulse duration and tissue type on thermal damage. Lasers Surg Med. 1989;9:314-26.
45. Karkada G, Maiya GA, Arany P, Kg MR, Adiga S, Kamath SU. Dose-response relationship of photobiomodulation therapy and oxidative stress markers in healing dynamics of diabetic neuropathic ulcers in Wistar rats. J Diabetes Metab Disord. 2023;22:393-400.
46. Anderson RR, Parrish JA. Selective photothermolysis: precise microsurgery by selective absorption of pulsed radiation. Science. 1983;220:524-7.
47. DiPietro LA. Angiogenesis and wound repair: when enough is enough. J Leukoc Biol. 2016;100:979-84.
48. Yu W, Zhu J, Yu W, Lyu D, Lin X, Zhang Z. A split-face, single-blinded, randomized controlled comparison of alexandrite 755-nm picosecond laser versus alexandrite 755-nm nanosecond laser in the treatment of acquired bilateral nevus of Ota-like macules. J Am Acad Dermatol. 2018;79:479-86.
49. Ma SY, Gong YQ, Zhang WJ, et al. Split-face comparison of the efficacy of picosecond 532 nm Nd:YAG laser and Q-switched 755 nm Alexandrite laser for treatment of freckles. J Cosmet Laser Ther. 2022;24:22-7.
50. Leyane TS, Jere SW, Houreld NN. Cellular signalling and photobiomodulation in chronic wound repair. Int J Mol Sci. 2021;22.
51. Mester E, Spiry T, Szende B. Effect of laser rays on wound healing. Bull Soc Int Chir. 1973;32:169-73.
52. Mester E, Ludány G, Sellyei M, Szende B, Gyenes G, Tota GJ. Studies on the inhibiting and activating effects of laser beams. Langenbecks Arch Chir. 1968;322:1022-7. (in German).
53. Ryu HS, Lim NK, Padalhin AR, et al. Improved healing and macrophage polarization in oral ulcers treated with photobiomodulation (PBM). Lasers Surg Med. 2022;54:600-10.
54. Zhang G, Yi L, Wang C, et al. Photobiomodulation promotes angiogenesis in wound healing through stimulating the nuclear translocation of VEGFR2 and STAT3. J Photochem Photobiol B. 2022;237:112573.
55. Mathioudaki E, Rallis M, Politopoulos K, Alexandratou E. Photobiomodulation and wound healing: low-level laser therapy at 661 nm in a scratch assay keratinocyte model. Ann Biomed Eng. 2024;52:376-85.
56. Barolet AC, Villarreal AM, Jfri A, Litvinov IV, Barolet D. Low-intensity visible and near-infrared light-induced cell signaling pathways in the skin: a comprehensive review. Photobiomodul Photomed Laser Surg. 2023;41:147-66.
57. Kasowanjete P, Houreld NN, Abrahamse H. The effect of photomodulation on fibroblast growth factor and the Ras/MAPK signalling pathway: a review. J Wound Care. 2022;31:832-45.
58. Karu T. Mitochondrial mechanisms of photobiomodulation in context of new data about multiple roles of ATP. Photomed Laser Surg. 2010;28:159-60.
59. Chung H, Dai T, Sharma SK, Huang YY, Carroll JD, Hamblin MR. The nuts and bolts of low-level laser (light) therapy. Ann Biomed Eng. 2012;40:516-33.
60. Dompe C, Moncrieff L, Matys J, et al. Photobiomodulation-underlying mechanism and clinical applications. J Clin Med. 2020;9.
61. Glass GE. Photobiomodulation: a review of the molecular evidence for low level light therapy. J Plast Reconstr Aesthet Surg. 2021;74:1050-60.
62. da Silva TG, Ribeiro RS, Mencalha AL, de Souza Fonseca A. Photobiomodulation at molecular, cellular, and systemic levels. Lasers Med Sci. 2023;38:136.
63. Mineroff J, Maghfour J, Ozog DM, Lim HW, Kohli I, Jagdeo J. Photobiomodulation CME part II: clinical applications in dermatology. J Am Acad Dermatol. 2024;91:805-15.
64. Agostinis P, Berg K, Cengel KA, et al. Photodynamic therapy of cancer: an update. CA Cancer J Clin. 2011;61:250-81.
65. Warrier A, Mazumder N, Prabhu S, Satyamoorthy K, Murali TS. Photodynamic therapy to control microbial biofilms. Photodiagnosis Photodyn Ther. 2021;33:102090.
66. Hou C, Zhang L, Wang L, et al. A meta-analysis and systematic review of photodynamic therapy for diabetic foot ulcers. Photodiagnosis Photodyn Ther. 2024;48:104228.
67. Zhang D, Leong ASW, McMullin G. Blue light therapy in the management of chronic wounds: a narrative review of its physiological basis and clinical evidence. Wounds. 2023;35:91-8.
68. Tripodi N, Sidiroglou F, Apostolopoulos V, Feehan J. Transcriptome analysis of the effects of polarized photobiomodulation on human dermal fibroblasts. J Photochem Photobiol B. 2023;242:112696.
69. Francis NC, Yao W, Grundfest WS, Taylor ZD. Laser-generated shockwaves as a treatment to reduce bacterial load and disrupt biofilm. IEEE Trans Biomed Eng. 2017;64:882-9.
70. Yao W, Kuan EC, Francis NC, St John MA, Grundfest WS, Taylor ZD. Laser-generated shockwaves enhance antibacterial activity against biofilms in vitro. Lasers Surg Med. 2017;49:539-47.
71. Orringer JS, Sachs DL, Shao Y, et al. Direct quantitative comparison of molecular responses in photodamaged human skin to fractionated and fully ablative carbon dioxide laser resurfacing. Dermatol Surg. 2012;38:1668-77.
72. Maddaluno L, Urwyler C, Werner S. Fibroblast growth factors: key players in regeneration and tissue repair. Development. 2017;144:4047-60.
73. Werner S, Peters KG, Longaker MT, Fuller-Pace F, Banda MJ, Williams LT. Large induction of keratinocyte growth factor expression in the dermis during wound healing. Proc Natl Acad Sci U S A. 1992;89:6896-900.
74. Prignano F, Campolmi P, Bonan P, et al. Fractional CO2 laser: a novel therapeutic device upon photobiomodulation of tissue remodeling and cytokine pathway of tissue repair. Dermatol Ther. 2009;22 Suppl 1:S8-15.
75. Bai J, Li L, Kou N, et al. Low level laser therapy promotes bone regeneration by coupling angiogenesis and osteogenesis. Stem Cell Res Ther. 2021;12:432.
76. Mirsky N, Krispel Y, Shoshany Y, Maltz L, Oron U. Promotion of angiogenesis by low energy laser irradiation. Antioxid Redox Signal. 2002;4:785-90.
77. Jiang B, Tang R, Zheng D, et al. Evaluation of the efficacy of ultrapulsed CO2 laser in chronic wounds. Lasers Surg Med. 2021;53:443-9.
78. El Nawam H, El Backly R, Zaky A, Abdallah A. Low-level laser therapy affects dentinogenesis and angiogenesis of in vitro 3D cultures of dentin-pulp complex. Lasers Med Sci. 2019;34:1689-98.
79. Keshri GK, Yadav A, Verma S, Kumar B, Gupta A. Effects of pulsed 810 nm Al-Ga-As diode laser on wound healing under immunosuppression: a molecular insight. Lasers Surg Med. 2020;52:424-36.
80. Kang D. Enhancing Skin flap survival with preoperative carbon dioxide fractional laser treatment: a novel approach in reconstructive surgery. J Craniofac Surg. 2025;36:334-8.
81. Cambier S, Gouwy M, Proost P. The chemokines CXCL8 and CXCL12: molecular and functional properties, role in disease and efforts towards pharmacological intervention. Cell Mol Immunol. 2023;20:217-51.
82. Restivo TE, Mace KA, Harken AH, Young DM. Application of the chemokine CXCL12 expression plasmid restores wound healing to near normal in a diabetic mouse model. J Trauma. 2010;69:392-8.
83. Schmitt L, Huth S, Amann PM, et al. Direct biological effects of fractional ultrapulsed CO2 laser irradiation on keratinocytes and fibroblasts in human organotypic full-thickness 3D skin models. Lasers Med Sci. 2018;33:765-72.
84. Helbig D, Bodendorf MO, Grunewald S, Kendler M, Simon JC, Paasch U. Immunohistochemical investigation of wound healing in response to fractional photothermolysis. J Biomed Opt. 2009;14:064044.
85. Peña OA, Martin P. Cellular and molecular mechanisms of skin wound healing. Nat Rev Mol Cell Biol. 2024;25:599-616.
86. Rousselle P, Braye F, Dayan G. Re-epithelialization of adult skin wounds: cellular mechanisms and therapeutic strategies. Adv Drug Deliv Rev. 2019;146:344-65.
87. Dillmann WH. Heat shock proteins and protection against ischemic injury. Infect Dis Obstet Gynecol. 1999;7:55-7.
88. Williams RS. Heat shock protein 47: a chaperone for the fibrous cap? Circulation 2000;101:1227-8.
89. Köhler A, Mörgelin M, Gebauer JM, et al. New specific HSP47 functions in collagen subfamily chaperoning. FASEB J. 2020;34:12040-52.
90. Genest O, Wickner S, Doyle SM. Hsp90 and Hsp70 chaperones: collaborators in protein remodeling. J Biol Chem. 2019;294:2109-20.
91. Makboul M, Makboul R, Abdelhafez AH, Hassan SS, Youssif SM. Evaluation of the effect of fractional CO2 laser on histopathological picture and TGF-β1 expression in hypertrophic scar. J Cosmet Dermatol. 2014;13:169-79.
92. Paasch U, Sonja G, Haedersdal M. Synergistic skin heat shock protein expression in response to combined laser treatment with a diode laser and ablative fractional lasers. Int J Hyperthermia. 2014;30:245-9.
93. Salo T, Mäkelä M, Kylmäniemi M, Autio-Harmainen H, Larjava H. Expression of matrix metalloproteinase-2 and -9 during early human wound healing. Lab Investig J Tech Methods Pathol 1994;70:176-82.
94. Kümper M, Zamek J, Steinkamp J, Pach E, Mauch C, Zigrino P. Role of MMP3 and fibroblast-MMP14 in skin homeostasis and repair. Eur J Cell Biol. 2022;101:151276.
95. Kostov K, Blazhev A. Changes in serum levels of matrix metalloproteinase-1 and tissue inhibitor of metalloproteinases-1 in Patients with essential hypertension. Bioengineering. 2022;9:119.
96. Shumaker PR, Kwan JM, Badiavas EV, Waibel J, Davis S, Uebelhoer NS. Rapid healing of scar-associated chronic wounds after ablative fractional resurfacing. Arch Dermatol. 2012;148:1289-93.
97. Madni TD, Lu KB, Imran JB, et al. Chronic burn wound treatment by Erbium: YAG fractional ablation: first described report and literature review. Burns. 2019;45:256-8.
98. Lee HN, Bae JM, Goo BCL, Park YM. Promotion of wound healing through low-fluence ablative fractional laser treatment in diabetic mice. Lasers Med Sci. 2019;34:421-5.
99. Guan H, Zhang D, Ma X, et al. Efficacy and safety of CO2 laser in the treatment of chronic wounds: a retrospective matched cohort trial. Lasers Surg Med. 2022;54:490-501.
100. Rohrer TE, Ugent SJ. Evaluating the efficacy of using a short-pulsed erbium:YAG laser for intraoperative resurfacing of surgical wounds. Lasers Surg Med. 2002;30:101-5.
101. Phillips TJ, Morton LM, Uebelhoer NS, Dover JS. Ablative fractional carbon dioxide laser in the treatment of chronic, posttraumatic, lower-extremity ulcers in elderly patients. JAMA Dermatol. 2015;151:868-71.
102. Zhang Y, Liu Y, Cai B, et al. Improvement of surgical scars by early intervention with carbon dioxide fractional laser. Lasers Surg Med. 2020;52:137-48.
103. Weinstein Velez M, Prezzano J, Bell M, Widgerow A. A single center, prospective, randomized, blinded study to evaluate the efficacy and safety of a topical tripeptide/hexapeptide anhydrous gel when used pre- and post- hybrid fractional laser for the treatment of acne scars. Clin Cosmet Investig Dermatol. 2022;15:2763-74.
104. Kuppa SS, Kang JY, Kim JY, et al. Red-light LED therapy promotes wound regeneration by upregulating COL1A1, COL2A1, VEGF and reducing IL-1β for anti-inflammation. Lasers Med Sci. 2025;40:171.
105. Riedemann HI, Marquardt Y, Jansen M, Baron JM, Huth S. Biological effect of laser-assisted scar healing (LASH) on standardized human three-dimensional wound healing skin models using fractional non-ablative 1540 nm Er:Glass or 1550 nm diode lasers. Lasers Surg Med. 2024;56:100-6.
106. Pulumati A, Jaalouk D, Algarin YA, Nouri K. The role of 755-nm alexandrite picosecond laser in melasma management. Arch Dermatol Res. 2023;316:60.
107. Bao MZ, Zhou LB, Zhao L, et al. Efficacy of lidocaine wet compress combined with red-light irradiation for chronic wounds. World J Clin Cases. 2023;11:7277-83.
108. Prudente D, Hauser F, Mettraux G, Di Bella E, Krejci I. Efficacy of one-time application of low-level laser therapy in the management of complications after third molar surgery: a retrospective practice-based study. Am J Dent. 2023;36:21-4.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].