Latest review articles on ageing published on Ageing Research Reviews
Our staff editors continue to share exciting, interesting, and thought-provoking reading material in the recommended articles series. This week, we would like to share several latest articles on ageing.
Title: Neuronal excitation/inhibition imbalance: core element of a translational perspective on Alzheimer pathophysiology
Authors: Fernando Maestú, Willem de Haan, Marc Aurel Busche, Javier DeFelipe
Type: Review
Highlights:
• The E/I balance is a fundamental mechanism for maintaining effective neuronal communication and support cognitive performance.
• AD research has been approached by different levels of analysis with no integration between them. Results need harmonization in a common framework.
• E/I imbalance is the endpoint of all AD-related micro-level synaptic pathology, and the starting point of the network disruption and cognitive impairment.
• Computational neurophysiological models can integrate data from different levels, creating a framework to test hypotheses and propose new strategies of intervention.
Abstract:
Our incomplete understanding of the link between Alzheimer’s Disease pathology and symptomatology is a crucial obstacle for therapeutic success. Recently, translational studies have begun to connect the dots between protein alterations and deposition, brain network dysfunction and cognitive deficits. Disturbance of neuronal activity, and in particular an imbalance in underlying excitation/inhibition (E/I), appears early in AD, and can be regarded as forming a central link between structural brain pathology and cognitive dysfunction. While there are emerging (non-)pharmacological options to influence this imbalance, the complexity of human brain dynamics has hindered identification of an optimal approach. We suggest that focusing on the integration of neurophysiological aspects of AD at the micro-, meso- and macroscale, with the support of computational network modeling, can unite fundamental and clinical knowledge, provide a general framework, and suggest rational therapeutic targets.
Access this article: https://doi.org/10.1016/j.arr.2021.101372
Title: The role of senescence in the pathogenesis of atrial fibrillation: A target process for health improvement and drug development
Authors: Guangran Guo, Steven Watterson, Shu-Dong Zhang, Anthony Bjourson, Victoria McGilligan, Aaron Peace, Taranjit Singh Rai
Type: Review
Highlights:
• Atrial fibrillation (AF) is an ageing-related chronic disease.
• Atrial electrical and structural remodeling act as substrate for AF maintenance.
• Atrial fibrosis is regarded as basis for atrial fibrillation development and maintenance.
• Senescence contributes to AF by causing atrial fibrosis through extracellular matrix, Angiotensin II, TGF-ß/Smad and thrombin signalling.
• Eliminating senescent cardiac cells by means of senolytic compounds, immune cells, CAR T cells and sEVs may attenuate atrial fibrillation.
Abstract:
Cellular senescence is a state of growth arrest that occurs after cells encounter various stresses. Senescence contributes to tumour suppression, embryonic development, and wound healing. It impacts on the pathology of various diseases by secreting inflammatory chemokines, immune modulators and other bioactive factors. These secretory biosignatures ultimately cause inflammation, tissue fibrosis, immunosenescence and many ageing-related diseases such as atrial fibrillation (AF). Because the molecular mechanisms underpinning AF development remain unclear, current treatments are suboptimal and have serious side effects. In this review, we summarize recent results describing the role of senescence in AF. We propose that senescence factors induce AF and have a causative role. Hence, targeting senescence and its secretory phenotype may attenuate AF.
Access this article: https://doi.org/10.1016/j.arr.2021.101363
Title: Altered endocytosis in cellular senescence
Authors: Eun-Young Shin, Nak-Kyun Soung, Martin Alexander Schwartz, Eung-Gook Kim
Type: Review
Highlights:
• Dysfunctional endocytic components contribute to cellular senescence.
• Downregulation of amphiphysin-1 and βPIX induces senescence through altered CME.
• Overexpression of ING1a induces cellular senescence through altered CME.
• Elevated caveolin-1 regulates CavME and induces senescence.
Abstract:
Cellular senescence occurs in response to diverse stresses (e.g., telomere shortening, DNA damage, oxidative stress, oncogene activation). A growing body of evidence indicates that alterations in multiple components of endocytic pathways contribute to cellular senescence. Clathrin-mediated endocytosis (CME) and caveolae-mediated endocytosis (CavME) represent major types of endocytosis that are implicated in senescence. More recent research has also identified a chromatin modifier and tumor suppressor that contributes to the induction of senescence via altered endocytosis. Here, molecular regulators of aberrant endocytosis-induced senescence are reviewed and discussed in the context of their capacity to serve as senescence-inducing stressors or modifiers.
Access this article: https://doi.org/10.1016/j.arr.2021.101332
Title: Emerging pathogenic role of peripheral blood factors following BBB disruption in neurodegenerative disease
Authors: Min-Tae Jeon, Kyu-Sung Kim, Eun Seon Kim, Suji Lee, Jieun Kim, Hyang-Sook Hoe, Do-Geun Kim
Type: Review
Highlights:
• Regulating inflammation in the brain and peripheral organs is an important strategy to develop the therapeutics for neurodegenerative diseases.
• The blood-brain barrier (BBB) is disrupted in patients with NDs and mouse models of NDs.
• Strengthening BBB integrity and preventing BBB disruption may attenuate ND progression and associated functional loss.
Abstract:
The responses of central nervous system (CNS) cells such as neurons and glia in neurodegenerative diseases (NDs) suggest that regulation of neuronal and glial functions could be a strategy for ND prevention and/or treatment. However, attempts to develop such therapeutics for NDs have been hindered by the challenge of blood-brain barrier (BBB) permeability and continued constitutive neuronal loss. These limitations indicate the need for additional perspectives for the prevention/treatment of NDs. In particular, the disruption of the blood-brain barrier (BBB) that accompanies NDs allows brain infiltration by peripheral factors, which may stimulate innate immune responses involved in the progression of neurodegeneration. The accumulation of blood factors like thrombin, fibrinogen, c-reactive protein (CRP) and complement components in the brain has been observed in NDs and may activate the innate immune system in the CNS. Thus, strengthening the integrity of the BBB may enhance its protective role to attenuate ND progression and functional loss. In this review, we describe the innate immune system in the CNS and the contribution of blood factors to the role of the CNS immune system in neurodegeneration and neuroprotection.
Access this article: https://doi.org/10.1016/j.arr.2021.101333
Title: Senescence in tissue samples of humans with age-related diseases: A systematic review
Authors: Camilla S.L. Tuttle, Suzanne W.M. Luesken, Mariette E.C. Waaijer, Andrea B. Maier
Type: Review
Highlights:
• Higher numbers of senescent cells have been implicated in age-related diseases.
• In humans, senescence is most studied in the heart and the respiratory system.
• The cell cycle regulator p16ink4a is most often investigated.
• Higher numbers of senescent cells are observed in human diseased tissues.
• The higher expression is independent of the marker used to detect senescence.
Abstract:
Background
Higher numbers of senescent cells have been implicated in age-related disease pathologies. However, whether different diseases have different senescent phenotypes is unknown. Here we provide a systematic overview of the current available evidence of senescent cells in age-related diseases pathologies in humans and the markers currently used to detect senescence levels in humans.
Methods
PubMed, Web of Science and EMBASE were systematically searched from inception to the 29th of September 2019, using keywords related to ‘senescence’, ‘age-related diseases’ and ‘biopsies’.
Results
In total 12,590 articles were retrieved of which 103 articles were included in this review. The role of senescence in age-related disease has been assessed in 9 different human organ system and 27 different age-related diseases of which heart (27/103) and the respiratory systems (18/103) are the most investigated. Overall, 27 different markers of senescence have been used to determine cellular senescence and the cell cycle regulator p16ink4a is most often used (23/27 age-related pathologies).
Conclusion
This review demonstrates that a higher expression of senescence markers are observed within disease pathologies. However, not all markers to detect senescence have been assessed in all tissue types.
Access this article: https://doi.org/10.1016/j.arr.2021.101334
Title: Reactive astrocytes: The nexus of pathological and clinical hallmarks of Alzheimer’s disease
Authors: Brittani R. Price, Lance A. Johnson, Christopher M. Norris
Type: Review
Highlights:
• Many Alzheimer’s disease pathophysiological processes are linked through astrocyte reactivity and/or dysfunction.
• Astrocyte signaling mechanisms are extensively intertwined and likely impact one another in deleterious ways.
• Modifying or resolving reactive astrocyte phenotypes could lead to effective anti-AD treatments.
Abstract:
Astrocyte reactivity is a hallmark of neuroinflammation that arises with Alzheimer’s disease (AD) and nearly every other neurodegenerative condition. While astrocytes certainly contribute to classic inflammatory processes (e.g. cytokine release, waste clearance, and tissue repair), newly emerging technologies for measuring and targeting cell specific activities in the brain have uncovered essential roles for astrocytes in synapse function, brain metabolism, neurovascular coupling, and sleep/wake patterns. In this review, we use a holistic approach to incorporate, and expand upon, classic neuroinflammatory concepts to consider how astrocyte dysfunction/reactivity modulates multiple pathological and clinical hallmarks of AD. Our ever-evolving understanding of astrocyte signaling in neurodegeneration is not only revealing new drug targets and treatments for dementia but is suggesting we reimagine AD pathophysiological mechanisms.
Graphical abstract:
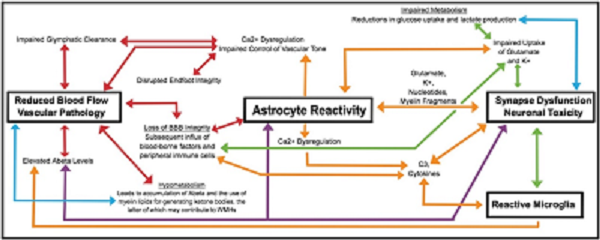
Access this article: https://doi.org/10.1016/j.arr.2021.101335
Title: Hypoxia and brain aging: Neurodegeneration or neuroprotection?
Authors: Johannes Burtscher, Robert T. Mallet, Martin Burtscher, Grégoire P. Millet
Type: Review
Highlights:
• Hypoxia is a central factor in brain aging and the development of age-related neurological diseases.
• Severe hypoxia is detrimental to the brain and is implicated in the pathogeneses of neurodegenerative diseases.
• The outcome of brain hypoxia depends on the type, severity, exposure duration and frequency of hypoxia.
• Adaptations of the brain to moderate hypoxia can be harnessed for neuroprotection.
• Hypoxia conditioning is a potential treatment strategy for neurodegenerative diseases.
Abstract:
The absolute reliance of the mammalian brain on oxygen to generate ATP renders it acutely vulnerable to hypoxia, whether at high altitude or in clinical settings of anemia or pulmonary disease. Hypoxia is pivotal to the pathogeneses of myriad neurological disorders, including Alzheimer’s, Parkinson’s and other age-related neurodegenerative diseases. Conversely, reduced environmental oxygen, e.g. sojourns or residing at high altitudes, may impart favorable effects on aging and mortality. Moreover, controlled hypoxia exposure may represent a treatment strategy for age-related neurological disorders. This review discusses evidence of hypoxia’s beneficial vs. detrimental impacts on the aging brain and the molecular mechanisms that mediate these divergent effects. It draws upon an extensive literature search on the effects of hypoxia/altitude on brain aging, and detailed analysis of all identified studies directly comparing brain responses to hypoxia in young vs. aged humans or rodents. Special attention is directed toward the risks vs. benefits of hypoxia exposure to the elderly, and potential therapeutic applications of hypoxia for neurodegenerative diseases. Finally, important questions for future research are discussed.
Access this article: https://doi.org/10.1016/j.arr.2021.101343







