Overcoming challenges in sustaining newborn screening in low-middle-income countries: the Philippine newborn screening system
Abstract
Newborn screening (NBS) was introduced in the Philippines in 1996, and the Newborn Screening Act of 2004 mandated its provision to all Filipino newborns. The program initially covered five conditions and has expanded to the current panel of 29 conditions. This report focuses on the steps taken for successful NBS implementation and the challenges that must be overcome to make NBS sustainable. While often considered a public health program, NBS is really a system of interacting parts that must be carefully considered and planned prior to embarking on their implementation. The basic challenges are the same in both high-income and low-middle-income countries (LMICs), but they are more difficult to overcome in LMICs. In addition to the technical aspects of screening, including supplies and maintenance, considerations must include human resources, professional and public education, and government support. Challenges may occur at any point in implementation and continuation, and it is important to learn from the experiences of others in order to make the process more efficient. Here, we report on the experiences in one LMIC, the Philippines, in creating and sustaining a NBS system so that others may gain from these experiences.
Keywords
INTRODUCTION
NBS is recognized globally as a successful public health prevention program that decreases morbidity and mortality through early screening, diagnosis, and treatment[1]. Program availability, newborn coverage, and number of screened disorders vary worldwide, i.e., North America (35 disorders, 100% coverage), Latin America (2-20+ disorders 10%-80% coverage), Africa (1 disorder, < 2% coverage), East Mediterranean and East Asia (5-20+ disorders, 60%-100% coverage)[2-4].
Almost half of the world’s 134 million babies reside in the Asia Pacific (APAC) region[5]. Countries in the region vary widely in size, economic development, language, culture, religion, and geography. APAC NBS progress is variable and can be broadly divided into three groups of countries: developed programs (Australia, China, Hong Kong, Japan, Korea, Malaysia, Sri Lanka, New Zealand, Philippines, Singapore, Taiwan, and Thailand) (1-30+ disorders; 80% to 100%); developing programs (Bangladesh, India, Indonesia, Cambodia, Laos, Mongolia, Nepal, Pakistan, Vietnam) (1-4+ disorders; < 1% to 5%); and undeveloped programs (Bhutan, Brunei Darussalam, Myanmar, North Korea, and Papua New Guinea). The undeveloped and underdeveloped programs are in low-income countries (LICs) and LMICs, challenged by poor economies, low literacy, inadequate health education, and poor government support[2,4,6]. In countries where screening is just developing or in the planning stages, there may be multiple projects ongoing within the country, often unknown to each other. In some countries, NBS may not appear to exist, but its availability from international screening laboratories may mean that a limited number are receiving the service anyway. Reviews of NBS activities in India, Malaysia, and Vietnam provide examples of some of the APAC NBS issues[7-9].
Examples of international NBS collaborations that were useful in establishing NBS in developing settings exist, and these should be reviewed and considered as part of any planning process. Collaborations may include both commercial and/or non-commercial sources. For example, a portion of the Polynesian islands access NBS through the New Zealand screening program[4]. Specialists from Singapore, Thailand, regions of China, Malaysia, and Vietnam have participated in training programs originating in Australia[4]. Collaboration with a German NBS program is providing laboratory services in Laos and a pilot project in Nepal is sending specimens to a commercial resource in India[10]. Instrument manufacturers have provided instrument training both onsite and abroad. International collaborative support for developing programs also exists within the region. In the latter case, six regional workshops since 2008 have resulted in written declarations by attendees aimed at providing development goals and encouraging related government support (at least philosophically)[11].
The Philippine NBS program (PNBSP) began as a pilot in 1996, utilizing the laboratory services of the NBS program in Sydney, Australia. Since then, the PNBSP has continued to grow and mature until it now provides screening services across the entire country, is part of the national insurance program, and is viewed by some as a worthwhile model of sustainability. Here, we review the development of NBS in the Philippines, including challenges and solutions, in the hope that these experiences can be helpful in developing NBS programs. It is not our intent to prescribe how NBS should be developed, but rather to review the experiences with one NBS program that has been initiated in LMICs and has become a successful, sustainable, and thriving system.
THE PHILIPPINE NBS SYSTEM
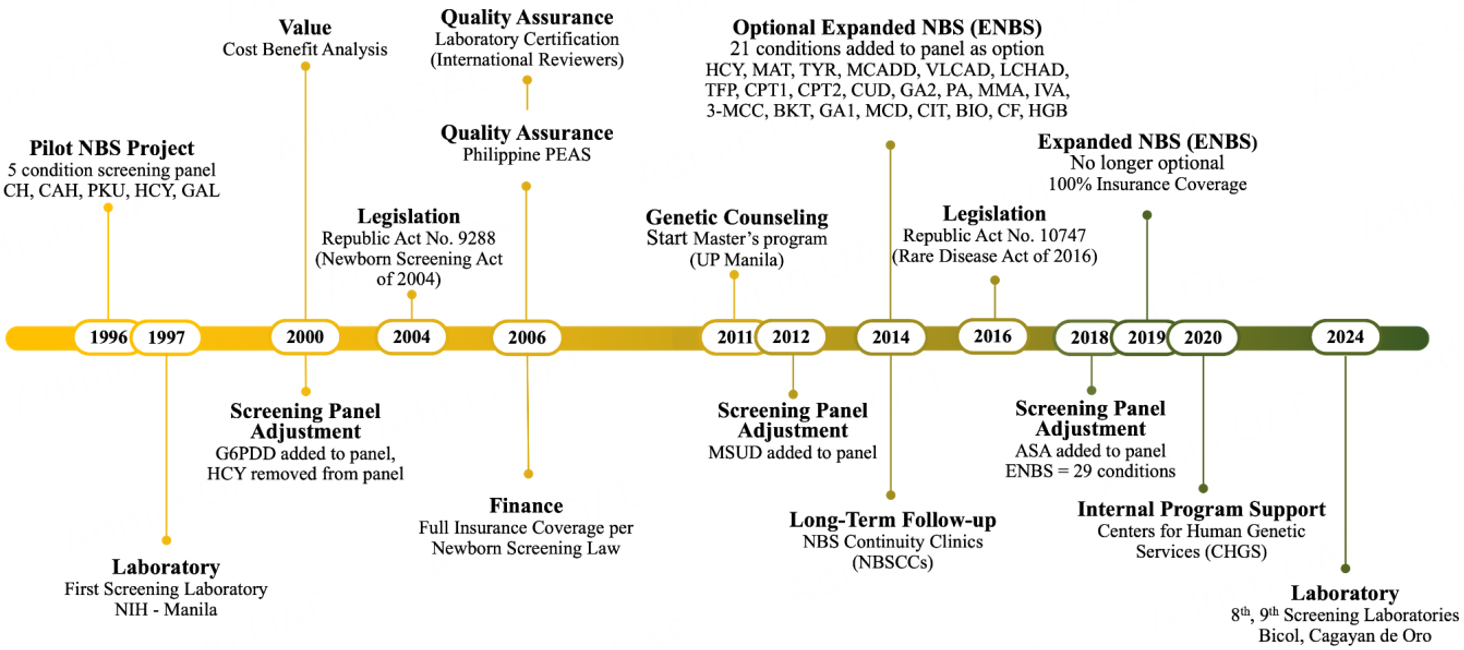
The Philippines is an archipelago with 7,600+ islands grouped into three major island chains: Luzon (north), Visayas (central), and Mindanao (south). It is classified as a LMIC in the Asia-Pacific and is home to a population of over 111 million. Administratively, the country is divided into 17 regions[12]. The NBS system had its origin in Metro Manila when collaborators from 24 hospitals in Metro Manila developed incidence data for six disorders that were thought to be of sufficient prevalence to justify NBS: congenital hypothyroidism (CH); congenital adrenal hyperplasia (CAH); galactosemia (GAL); phenylketonuria (PKU); homocystinuria (HCY); and glucose-6-phosphate dehydrogenase deficiency (G6PDD). Leadership for this PNBSP was provided by two pediatricians from the University of the Philippines Manila who were passionately interested in NBS. Funding came from a small break-even fee (approximately US$ 11) charged to each patient, with billing to individual hospitals based on the number of specimen collection cards requested. Through professional contacts, collaborative laboratory services were obtained from the NBS laboratory in Sydney, Australia. For the first year of the project, dried blood spot (DBS) specimens were transported daily to Sydney while data accumulated and local laboratory capabilities developed[13]. This project became the anchor point of the Philippine NBS timeline, as shown in Figure 1.
The PNBSP data confirmed the direction that NBS should take, and routine screening for five disorders (excluding G6PDD) began in 1996 in the 24 pilot hospitals. Data on G6PDD continued to be collected until it was added to the screening panel in 2000. In 1998, the PNBSP data were presented to the Department of Health (DOH) for consideration for nationwide implementation. Implementation was not approved due to logistical concerns and a lack of available funding. As an alternative, the DOH expressed its support to NBS in March 1999, with its inclusion in “Children’s Health 2025”, which was part of the road map for safeguarding the rights of Filipino children from 2000-2025. In 2000, an official administrative order (AO) was issued, setting the direction of NBS for the next five years[14,15]. This DOH AO provided provisions to initiate the NBS system, directing all DOH-retained and renationalized hospitals to participate in NBS by 2000; all other hospitals, both private and government, were required to participate in the project by 2004. In 2000, G6PDD was officially added to the screening panel and HCY was removed as a cost-saving strategy due to insufficient case findings.
Specimen collection was initially set at 48 hours after birth but was changed in 2000 to 24 hours based on data that showed little effect on screening results by this change[15,16]. A cost-benefit analysis in 2000 revealed a benefit-to-cost ratio of 4:1 (net benefit of US$ 11.42 Million annually) for the 5-condition screening panel (CH, CAH, GAL, PKU, G6PDD)[17]. This cost-benefit analysis became a reference document for the enactment of the National NBS Law of 2004 (Republic Act No. 9288)[18], which was written in response to poor uptake of NBS in prior years and required significant lobbying efforts for more than a year (an unusually short period for a law such as this). The passage of the law was integral to the inclusion of NBS into the public healthcare system. Further, in compliance with the 2004 NBS law, program costs were carefully calculated and included in the newborn care package of the Philippine Health Insurance Corporation (PhilHealth). In 2006, PhilHealth released a circular on the funding of newborn screening under a case payment scheme for reimbursement at a fixed price of PhP500[19]. The NBS fee subsequently increased in 2018 to PhP1,750 following the expansion of the screening panel to include 21 additional congenital disorders[20].
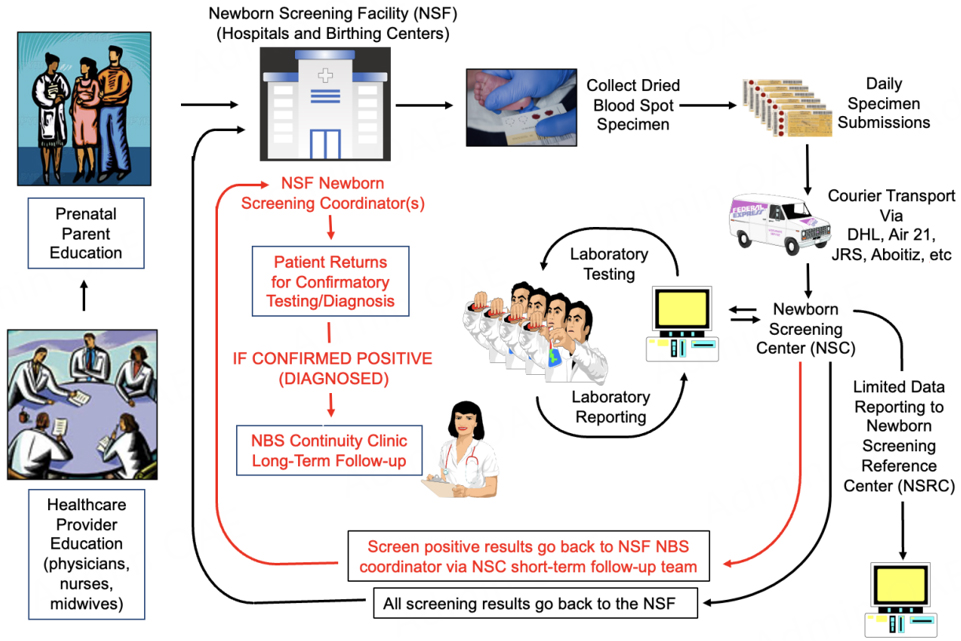
Figure 2 provides a schematic diagram showing the basic flow of the Philippine NBS system. Education about NBS is done either prenatally, as part of parent education, or just before the collection of the screening specimen. In compliance with the law, the parent/guardian is informed about NBS and given the opportunity to dissent. NBS by heelprick is performed after 24 hours following the birth at a Newborn Screening Facility (NSF-hospital or birthing center) by a healthcare professional trained in collecting heelstick blood. Specimens are submitted to a regional Newborn Screening Center (NSC-screening laboratory). A short-term follow-up team (pediatrician and/or nurse) from the NSC informs the NSF of screen-positive patients and NBS coordinators at the NSFs track/recall patients needing confirmatory testing. Diagnosed patients are referred for long-term follow-up to the NBS Continuity Clinics (see below). A database of diagnosed patients is kept at each NSC and the Newborn Screening Reference Center (NSRC-national NBS oversight center). Residual specimens remaining after NBS are retained indefinitely.
As shown in Figure 1, the PNBSP slowly grew and developed from a 5-condition panel in 1996 to a 29-condition panel today and from a single laboratory to nine by the end of 2024. Data have been periodically evaluated to determine whether the screening panel was appropriate, and adjustments have been made when and where possible. Based on clinical data, MSUD was added in 2012. In the Implementing Rules and Regulations of the NBS law[21], a National Technical Working Group on Newborn Screening periodically reviews and recommends to the DOH Advisory Committee on Newborn Screening regarding the addition or removal of conditions to the NBS panel. While considering the design of a pilot study for possible NBS expansion, data from the California (CA) NBS program were analyzed because of their large immigrant Filipino population. A detailed review of the expanded NBS data from CA provided sufficient case findings in a large enough Filipino population to serve as a surrogate pilot study. The reviewed NBS conditions in CA were those consistent with the U.S. Recommended Uniform Screening Panel (RUSP). In 2014, based on the NBS data from CA and with sufficient program savings to purchase MS/MS and other necessary equipment, the PNBSP screening panel was increased to include 21 additional conditions. This expanded NBS panel was made available as an option to parents at a break-even fee. Significant local case findings led to the addition of ASA to the screening panel in 2018, and in 2019, with increased insurance reimbursement, ENBS for 29 total screening conditions was made permanent. The screening panel currently includes the conditions shown in Table 1[22]. From a coverage of less than 1% in 1996, coverage peaked at 93.5% before the COVID-19 pandemic. Coverage dipped to 88.1% during the pandemic and is now (2023) returning to its pre-pandemic coverage.
Conditions included in the Philippine newborn screening program as of 2023
| Amino acid disorders | Endocrine disorders | Fatty acid oxidation disorders | Organic acidurias | Urea cycle defects | Hemoglobin disorders | Others |
| Homocystinuria (HCY) | Congenital adrenal hyperplasia (CAH) | Medium chain-acyl-CoA dehydrogenase deficiency (MCAD) | Propionic acidemia (PA) | Argininosuccinic aciduria (ASA) | Detectable hemoglobinopathies and thalassemias (HGB) | Biotinidase deficiency (BIO) |
| Methionine adenosine Transferase deficiency (MAT) [Hypermethioninemia] | Congenital Hypothyroidism (CH) | Very long Chain-acyl- CoA Dehydrogenase deficiency (VLCAD) | Methylmalonic academia (MMA) | Citrullinemia (CIT) | Cystic fibrosis (CF) | |
| Maple syrup urine disease (MSUD) | Long chain hydroxy-acyl- COA dehydrogenase deficiency (LCHAD) | Isovaleric acidemia (IVA) | Galactosemia (GAL) | |||
| Phenylketonuria (PKU) [Hyperphenylalaninemia, 6-pyruvoyl-tetrahydropterin synthase deficiency] | Trifunctional protein deficiency (TFP) | 3-methylcrotonyl carboxylase deficiency (3MCC) | Glucose-6-phosphate dehydrogenase deficiency (G6PDD) | |||
| Tyrosinemia (TYR) [Tyrosinemia type I, II, III] | Carnitine palmitoyl Transferase deficiency (CPT1) | Beta-Ketothiolase deficiency (BKT) | ||||
| Carnitine palmitoyl Transferase deficiency 2 (CPT2) | Glutaric aciduria type (GA1) | |||||
| Carnitine uptake defect (CUD) | Multiple carboxylase deficiency (MCD) | |||||
| Glutaric aciduria type 2 (GA2) |
The archipelagic nature of the Philippines poses the additional challenge of implementation across the 7200+ NSFs. This challenge has been met by the appointment of a NBS coordinator for NSF. This coordinator is responsible for ensuring that every baby born in the NSF undergoes NBS. Responsibilities also include educating healthcare workers and the family, collecting samples, and arranging for the courier service to pick-up and transport specimens to the designated NSC. The NSC performs the screening tests, releases the screening results, completes short-term follow-up activities, and then refers confirmed cases to a Newborn Screening Continuity Clinic (NBSCC). In the short-term follow-up process, the NBS coordinator at the NSF can be contacted to facilitate additional testing for screen-positive newborns, aiding in the diagnostic process.
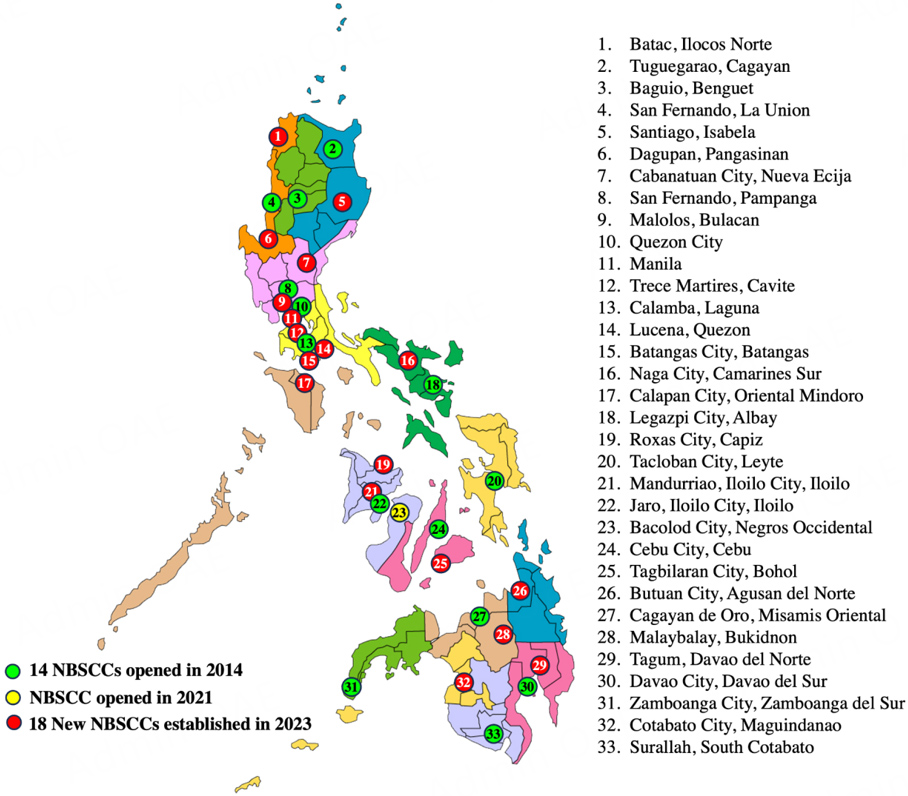
The NBSCCs and Centers for Human Genetic Services (CHGSs) were created to assist in providing patient care for diagnosed cases. The NBSCCs were first established in 2014 to strengthen the referral network and to ensure the appropriate treatment/management of confirmed cases[23]. The establishment of the NBSCCs was in response to the lack of medical facilities with capabilities for long-term patient care of patients diagnosed with a condition in the ENBS panel. NBSCCs are manned by a pediatrician and a nurse, who take charge of long-term care, including referrals to specialists[24]. For patients with metabolic conditions, the NBSCCs organize the delivery of medical foods. Currently, there are 33 NBSCCs strategically located throughout the Philippine archipelago [Figure 3]. Additional satellite clinics were also established to expand the treatment network. The NBSCCs coordinate with other stakeholders (i.e., local government units - LGUs, CHGSs, DOH-CHDs) to ensure delivery of services to patients[24].
The CHGSs were established in 2020 and are composed of facilities that cover island-wide services (Luzon, Visayas, and Mindanao) and facilitate comprehensive clinical evaluation, appropriate case management (diagnostic and therapeutic), and genetic counseling services for families or individuals with genetic conditions. The CHGS core teams include a clinical geneticist, a pediatrician or family physician, a nurse, a dietitian/nutritionist, a pharmacist, a genetic counselor, a psychologist or social worker, and administrative staff. Their responsibilities include facilitating comprehensive clinical management and genetic counseling services for families and individuals; assisting NSCs, NSFs, and NBSCCs in acute and long-term case management; serving as the central repository of medical foods, orphan drugs and products, and other treatment needs not readily available locally and/or commercially; coordinating the distribution of medical food, supplies, and drugs to NSCs and NBSCCs; conducting research that contributes evidence-based data for directing future strategic plans of the national NBS program; collaborating with the Institute of Human Genetics, National Institutes of Health (NIH-IHG)-in developing management guidelines and other related advocacy materials targeting relevant stakeholders; submitting periodic reports to NIH-IHG; and assisting the Newborn Screening Reference Center (NSRC) in its capacity building amongst health personnel of the NBSCCs.
In 2020, the COVID-19 pandemic challenged healthcare systems worldwide. In the Philippines, strict border controls were enacted between provinces, challenging the way in which NBS samples, medical foods, supplies, and medicines could be transported. The ingenuity and dedication of the staff at the NSFs, NSCs, NBSCCs, CHGSs, NSRC, and DOH regional offices led to the successful continuation of NBS despite the COVID-19 pandemic. Disaster contingency plans for overcoming the resulting challenges (operations, transportation, supply chain, communication, safety, etc.) were enacted by all groups. The NSFs, in collaboration with the DOH regional offices, sent samples with personnel who were allowed to cross the borders (policemen, firemen, military, navy, etc.). NSCs are divided internally into two cross-trained laboratory teams to enable continued laboratory services should COVID-positive cases arise in team members. Follow-up of patients shifted to teleconsultations as the main mode of patient contact[24-27]. As a result, the PNBSP is developing updated contingency plans to respond not only to pandemics but also to natural and man-made disasters. Being located in the Pacific Ring of Fire, the NBS program must be able to respond effectively in emergency situations.
DISCUSSION
FACTORS CONTRIBUTING TO SUCCESSFUL NBS IN THE PHILIPPINES
Policy development
With the poor uptake of NBS in the early years, national policy development became the major thrust of NBS champions. When NBS uptake failed to increase significantly, NBS champions moved forward with a request for a national law requiring that NBS be offered to all parents of newborns. The 2004 NBS law also integrated NBS into the public healthcare system. The Newborn Screening Act of 2004 has had the most significant impact on national implementation. The law included five key elements: (1) DOH was made the lead agency for NBS implementation; (2) NBS was covered by government insurance; (3) a Newborn Screening Reference Center (NSRC), a university-based technical implementation partner, was created; (4) health workers were required to inform parents and guardians about NBS; (5) NBS was required to be offered for issuance of a birthing facilities’ license to operate and accreditation[18].
The full engagement of the DOH contributed to the progressive success of the PNBSP by increasing the number of NSFs (from 24 hospitals in 1996 to 324 at the time the law was passed, 5,616 in 2014, and 7,200+ until now), and by increasing NBS coverage (from less than 1% in 1996 to 93.5% in 2019). The NBS law has been used as the basis for target setting at the DOH. In 2018, the “National Policy and Strategic Framework on Expanded Newborn Screening for 2017-2030” was formulated, which details the NBS targets by 2030: 95% newborn screening coverage; 90% of health facilities with maternity and newborn services include expanded NBS (ENBS); 100% PhilHealth coverage of the newborn care package including ENBS; 95% of facilities maintaining a greater than 99% satisfactory specimen rate; 95% of monitoring data generated from DOH regional offices and NSFs through an online monitoring system; and 85% of identified strategically-located provinces with established long-term management system for confirmed positive NBS patients[28].
Funding
The NBS law included payment for NBS in the benefits package of the Philippine Health Insurance Corporation (PhilHealth), which oversees the National Health Insurance Program[19,20]. Program costs to be reimbursed included stakeholder education, laboratory testing, sample transport, short-term follow-up, and overhead expenses, among others. The law provided for administrative support by including that at least 4% of the NBS fee go to the DOH Centers for Health Development (CHD) for follow-up services, education, and other relevant activities. At least 4% was also allocated to the NSCs for human resource development, equipment maintenance, and equipment replacement. At least 4% was also allocated to the Newborn Screening Reference Center (NSRC) for overall program supervision, training, continuing education, national data collection, program monitoring and quality assurance, and other operational expenses[18]. The ENBS fee augments the cost of treatment and management of patients[29].
Communication strategies
Communication strategies have been important from the beginning and their mechanisms for sharing NBS program information have evolved over the years. While maintaining applicable traditional measures (brochures, newsletters, posters, newspapers), the program has added digital media in recent years. Pertinent information on NBS can be accessed through the NSRC website, social media (i.e., Facebook and Twitter), webinars, and online newsletters [Figure 4].
The “Newborn Screening in Focus” series is a pre-recorded weekly video discussion with experts, field implementers, patients, and policy makers on different NBS topics [Figure 5]. It is aired through TVUP, an Internet television (webcast) network operated by the University of the Philippines, which delivers free content for information and educational purposes. The first season was launched in February 2023. YouTube views have ranged from 110+ to 2300+ per episode, not including downloaded materials used for lectures.
Community engagement
The presence of an empowered collaborative stakeholder community has also contributed to the success of the PNBSP. The national NBS system includes engagement with experts, health professionals, staff, advocates, academe and civil society, and the religious sector. Engagement with these program partners includes activities compliant with Presidential Order No. 540, which declares the first week of October of each year as National Newborn Screening Week[30]. With the DOH and the IHG as national focal points, activities are organized to commemorate this event and ensure that information about the availability and benefits of NBS is expeditiously and widely disseminated to the general public. Activity co-sponsors have included local government units, NBS coordinators in NSFs, private companies (sponsoring baby and family expos), and volunteers for interviews in radio programs.
The NBS agenda has also been disseminated and strengthened through the initiatives of various advocacy organizations such as Volunteer Youth Leaders for Health (VYLH)-Philippines, and the Philippine Society for Orphan Disorders (PSOD). VYLH is a network of proactive, service-oriented, empowering, and visionary leaders mobilizing the youth towards a healthy Philippines[31]. PSOD is a non-stock and non-profit organization established in 2006 to provide patient support for rare diseases[32]. Both groups were instrumental in the lobbying efforts for the enactment of the Rare Disease Act of 2016 or Republic Act No. 10747[33], which supported some of the conditions in the ENBS panel.
Panels of medical specialty experts assumed the responsibility for formulating algorithms for screening, confirmation/diagnosis, and treatment. They also provided canned responses to frequently asked questions from both doctors and patients. Separate medical expert panels were created to handle metabolic disorders, G6PD deficiency, hemoglobinopathies and thalassemias, congenital hypothyroidism, congenital adrenal hyperplasia, and cystic fibrosis.
Capacity building/cross training
From the outset, there has been a demand for trained healthcare providers who could assist with short-term follow-up. In 2011, the Master of Science in Genetic Counseling program was approved for implementation at the University of the Philippines Manila[34,35]. The 2-year program trains genetic counseling professionals to be equipped with the competencies necessary to counsel and empower patients and families to make informed genetics-related decisions. Some of these trained genetic counselors (GCs) are currently providing services to the NSCs, including in-person genetic counseling and telegenetics. In 2022, the GCs organized themselves to establish the Philippine Society of Genetic Counselors, Inc., with the commitment to sustain a community of highly trained GCs by sharing expertise and best practices relative to education, research, and professional practice[36].
Quality assurance
Quality services are critical in the delivery of all NBS services. The Philippine Performance Evaluation and Assessment Scheme (PPEAS) is a quality checklist designed to monitor quality and guide improvements in the PNBSP. The PPEAS was developed on a similar PEAS developed for US NBS programs. PPEAS aims to help ensure and improve NBS quality at regional and local NSFs, as required by Republic Act No. 9288. Several evaluation tools were developed for different NBS system components: NSCs, NSFs, and CHDs. More recently, PPEAS tools have been developed for the NBSCCs[37]. Each NSC is a participant in the Newborn Screening Quality Assurance Program (NSQAP) of the Centers for Disease Control and Prevention (CDC), USA and the G6PDD External Quality Assurance Program of Preventive Maintenance Foundation in Taiwan. These programs periodically provide unknown samples in a graded external Quality Assurance exercise. The DOH also requires external review assessment as part of its NSC licensure requirement, and a review team of international experts (Dr. Bradford Therrell from the USA and Dr. Veronica Wiley from Australia), a local pathologist, and representatives from the DOH and NSRC provide periodic expert reviews as part of this process.
Oversight issues
Despite the overall success of the program, there are continuing national challenges yet to be fully addressed by the NSRC [Table 2]. Policies must be developed to address the timeliness of specimen submission and recall of screen-positive patients, which pose a particular challenge for patients from remote communities in the mountains or on remote islands. For screen-positive patients already presenting with symptoms upon recall, coordination with the needed subspecialist must be done to initiate acute care management. When a remote patient is confirmed, arrangements for follow-up and delivery of metabolic food (for metabolic patients) and drugs pose a logistical issue. In some instances, collaboration with the military has been necessary as the only viable alternative to accomplish this task. The presence of these geographically isolated and disadvantaged areas (GIDA) also presents a challenge in delivering continuing education to NBS frontliners. Telecommunications technology is currently being used to keep the program implementers updated. A locally developed ENBS mobile app now provides a one-stop communications hub for NBS information. The app streamlines messages to all healthcare workers about program activities and monitors compliance of newborn screening facilities.
Summary of challenges and possible solutions
| Challenges | Possible solutions |
| Quality of specimen submission during NBS initial test | Re-training screeners at the NSFs |
| Efficiency of recall of screened positive newborns | Collaborations with LGUs and DOH-CHDs for reaching patients living in difficult-to-reach places, i.e., islands and mountains Training healthcare workers in communities to perform heel prick and to help in the recall of patients |
| Prompt treatment of diagnosed patients | Early coordination with the needed subspecialist to initiate management of patients who already present symptoms on recall |
| Presence of geographically isolated and disadvantaged areas | Collaborations with LGUs in education and implementation of the program Use of telecommunications technology to keep the program implementers updated |
| Maintain quality testing in the NBS laboratories | Initiate and ensure compliance with standards among the NSCs through ISO 15189 certification |
| Expansion of the treatment network | Coordination with DOH regional offices and provincial offices in identifying new sites for the continuity and satellite clinics |
CONCLUSION
The challenges to implementing sustainable national newborn screening in developing health systems have been previously outlined[38]. The implementation of NBS in the Philippines has been successful because of attention to overcoming these challenges that we have discussed in this paper. The success of the PNBSP has been attributed to a mix of actions and activities, including national policies, local health ordinances, dedicated program partners, health professionals, policymakers, and civil organizations (professional health societies, NGOs, parent support groups, etc.). The inclusion of a funding mechanism in the law was a tactical strategy to help ensure the sustainability of the NBS program. Good leadership is essential for program development and sustainability and has been present in the PNBSP, from planning the pilot to nationwide expansion. Education using traditional (print) and evolving technologies (digital) has been a continuous and necessary component of the program. It ensures that parents, NBS coordinators, policy makers, and program staff are informed about new program information. Technical, medical, and logistical support have been present from the early days of the program and continue today, placing the program on a firm foundation as it moves forward. Continuous quality improvement has been integrated into the various components of PNBSP through the development and implementation of PPEAS including external laboratory certification with international experts. As a result of these efforts, the PNBSP has been successful and sustainable and is a potential model for other developing programs with a similar environment.
DECLARATIONS
Acknowledgments
We thank the Newborn Screening Reference Center of the National Institutes of Health, University of the Philippines Manila for sharing updated information on the Philippine Newborn Screening Program.
Authors’ contributions
Contributed to planning, securing information, analyzing data, and writing and editing of the manuscript: Padilla CD, Abadingo ME, Munda KV, Therrell BL
Availability of data and materials
Not applicable.
Financial support and sponsorship
None.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Written informed consent for publication was obtained from all individuals appearing in Figure 5, including the interviewer/host and the four interviewees. All individuals have authorized the use of photographs for academic publications, educational materials, and online dissemination.
Copyright
© The Author(s) 2023.
REFERENCES
1. Centers for Disease Control and Prevention (CDC). Ten great public health achievements--United States, 2001-2010. MMWR Morb Mortal Wkly Rep. 2011;60:619-23.
2. Therrell BL, Padilla CD, Loeber JG, et al. Current status of newborn screening worldwide: 2015. Semin Perinatol. 2015;39:171-87.
3. Bonham J, Leung-Pineda V. Report on global newborn screening IFCC/ISNS task. Available from: https://cms.ifcc.org/media/479858/3-tf-nbs-van-leung-pineda.pdf [Last accessed on 4 Dec 2023].
4. Wiley V, Webster D, Loeber G. Screening pathways through China, the Asia Pacific region, the world. Int J Neonatal Screen. 2019;5:26.
5. Our World in Data. Births and deaths per year, world. Available from: https://ourworldindata.org/grapher/births-and-deaths-projected-to-2100 [[Last accessed on 4 Dec 2023].
6. Howson CP, Cedergren B, Giugliani R, et al. Universal newborn screening: a roadmap for action. Mol Genet Metab. 2018;124:177-83.
7. Mookken T. Universal implementation of newborn screening in India. Int J Neonatal Screen. 2020;6:24.
8. Leong YH, Gan CY, Tan MA, Majid MI. Present status and future concerns of expanded newborn screening in malaysia: sustainability, challenges and perspectives. Malays J Med Sci. 2014;21:63-7.
9. Nguyen TT, Le QT, Hoang DT, et al. Massively parallel sequencing uncovered disease-associated variant spectra of glucose-6-phosphate dehydrogenase deficiency, phenylketonuria and galactosemia in Vietnamese pregnant women. Mol Genet Genomic Med. 2022;10:e1959.
10. Vesaphong P, Lindemuth L, Istaz B, Kedsatha P, Saysanasongkham B, Hoehn T. Establishment of a sustainable newborn TSH-screening program in the People's Democratic Republic of Laos. Early Hum Dev. 2021;154:105306.
11. Padilla CD, Therrell BL Jr; Working Group of the Asia Pacific Society for Human Genetics on Consolidating Newborn Screening Efforts in the Asia Pacific Region. Consolidating newborn screening efforts in the Asia Pacific region : networking and shared education. J Community Genet. 2012;3:35-45.
12. Philippine Statistics Authority. 2022 Philippine national demographic and health survey. Available from: https://library.psa.gov.ph/cgi-bin/koha/opac-detail.pl?biblionumber=15634 [Last accessed on 4 Dec 2023].
13. Padilla CD. Newborn screening in the Philippines. Southeast Asian J Trop Med Public Health. 2003;34 Suppl 3:87-8.
14. Department of Health. Administrative order No. 1-A, s. 2000: policies on the nationwide implementation of newborn screening. Available from: https://dmas.doh.gov.ph:8083/Rest/GetFile?id=336241 [Last accessed on 4 Dec 2023].
15. Padilla CD, Fagela-Domingo C. Implementation of newborn screening in the Philippines. Philipp J Pediatr 2002;51:2-10. Available from: https://www.herdin.ph/index.php/component/herdin/?view=research&cid=33913 [Last accessed on 4 Dec 2023].
16. Fagela-Domingo C, Padilla CD. Newborn screening for congenital hypothyroidism in early discharged infants. Southeast Asian J Trop Med Public Health. 2003;34 Suppl 3:165-9.
17. David-Padilla C, Dans LF, Tamondong Jr MR, Bernal RMS, Laceste JJO, Capistrano-Estrada S. Cost-benefit analysis of the newborn screening program of the Philippines. Acta Med Philipp 2009;43:46-52. Available from: https://actamedicaphilippina.upm.edu.ph/index.php/acta/article/view/2500 [Last accessed on 7 Dec 2023].
18. Republic Act 9288. An act promulgating a comprehensive policy and a national system for ensuring newborn screening. Available from: https://lawphil.net/statutes/repacts/ra2004/ra_9288_2004.html [Last accessed on 4 Dec 2023].
19. Philippine Health Insurance Corporation. PhilHealth Circular No. 34, s-2006. Available from: https://www.philhealth.gov.ph/circulars/2006/circ34_2006.pdf [Last accessed on 4 Dec 2023].
20. Philippine Health Insurance Corporation. PhilHealth Circular No. 2018-0021. Available from: https://www.philhealth.gov.ph/circulars/2018/circ2018-0021.pdf [Last accessed on 4 Dec 2023].
21. Republic of the Philippines Department of Health. Rules and regulations implementing republic act No. 9288. https://lawphil.net/statutes/repacts/ra2004/pdf/irr_9288_2004.pdf [Last accessed on 7 Dec 2023].
22. Padilla CD, Therrell BL Jr, Alcausin MMLB, et al. Successful implementation of expanded newborn screening in the philippines using tandem mass spectrometry. Int J Neonatal Screen. 2022;8:8.
23. Department of Health. Administrative order No. 2014-0035: implementing guidelines on the setting-up of newborn screening continuity clinics. Available from: https://dmas.doh.gov.ph:8083/Rest/GetFile?id=336996 [Last accessed on 4 Dec 2023].
24. Maceda EBG, Abadingo ME, Panol KAR, et al. Newborn screening long-term follow-up clinics (continuity clinics) in the Philippines during the COVID-19 pandemic: continuing quality patient care. Int J Neonatal Screen. 2022;9:2.
25. Padilla PJD, Manalo EM. Newborn Screening knowledge, attitudes and practices among obstetrics-gynecology residents, pediatric residents, and newborn screening nurses in a tertiary government hospital in the Philippines during the COVID-19 pandemic. Int J Neonatal Screen. 2023;9:19.
26. Tumulak MJR, Pascua AV, Jover EJM, Guerbo RJ, Canoy GMR, Laurino MY. Genetic counseling in the time of COVID-19: the Philippine experience with telegenetics. J Genet Couns. 2021;30:1285-91.
27. Koracin V, Loeber JG, Mlinaric M, Battelino T, Bonham JR, Groselj U; COVID-NBS ISNS global network. Global impact of COVID-19 on newborn screening programmes. BMJ Glob Health. 2022;7:e007780.
28. Department of Health. Administrative order No. 2018-0025: national policy and strategic framework on expanded newborn screening for 2017-2030. Available from: https://dmas.doh.gov.ph:8083/Rest/GetFile?id=618688 [Last accessed on 4 Dec 2023].
29. Department of Health. Administrative order 2020-0052: revised guidelines on the implementation of the expanded newborn screening program. Available from: https://law.upd.edu.ph/wp-content/uploads/2021/02/DOH-Administrative-Order-No-2020-0052.pdf [Last accessed on 4 Dec 2023].
30.
31. Padilla CD, Sur ALD, Pascual RJP, et al. Volunteer youth leaders for health-Philippines: providing a mechanism for youth empowerment towards advocacy for birth defects prevention and care. Acta Med Philipp. 2017;51:167-74.
32. National Organization for Rare Disorders (NORD). Philippine society for orphan disorders. Available from: https://rarediseases.org/non-member-patient/philippine-society-for-orphan-disorders/ [Last accessed on 4 Dec 2023].
33. Republic Act No. 10747. Rare diseases act of the Philippines. Available from: https://www.officialgazette.gov.ph/2016/03/03/republic-act-no-10747/ [Last accessed on 7 Dec 2023].
34. Laurino MY, David-Padilla C, Alcausin MMB, Silao CLT, Cutiongco-de la Paz EM. A master of science in genetic counseling program in the Philippines. Acta Med Philipp 2011;45:7-9. Available from: https://actamedicaphilippina.upm.edu.ph/index.php/acta/article/view/4738 [Last accessed on 7 Dec 2023].
35. Laurino MY, Padilla CD. Genetic counseling training in the Philippines. J Genet Couns. 2013;22:865-8.
36. Abad PJ, Tumulak M, Guerbo R, et al. Landscape of genetic counseling in the philippines. J Genet Couns. 2023:1-9.
37. Padilla CD, Therrell BL, Panol KAR, et al. Philippine performance evaluation and assessment scheme (PPEAS): experiences in newborn screening system quality improvement. Int J Neonatal Screen. 2020;6:95.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.




















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].