Reframing electrolyte coordination for low-temperature lithium-metal batteries
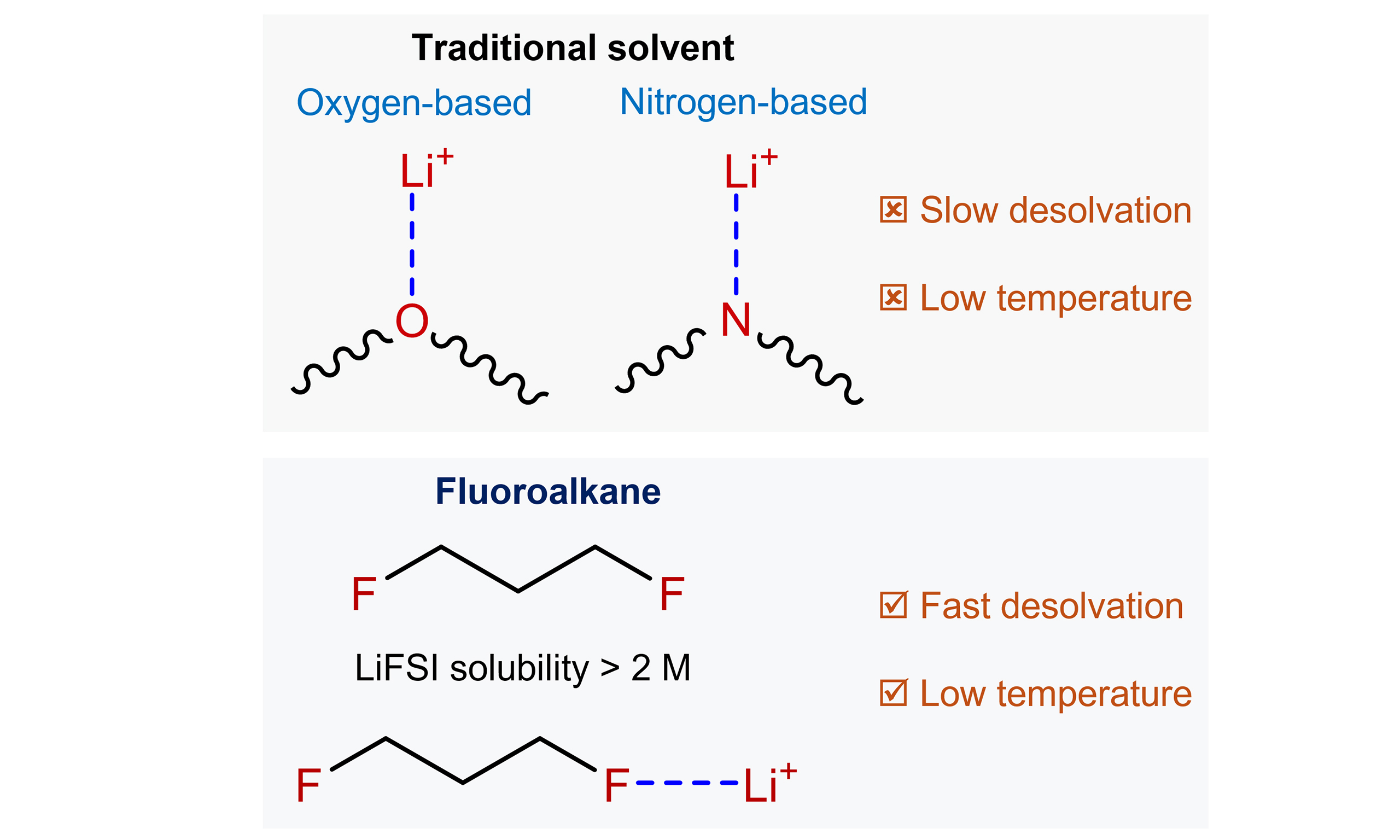
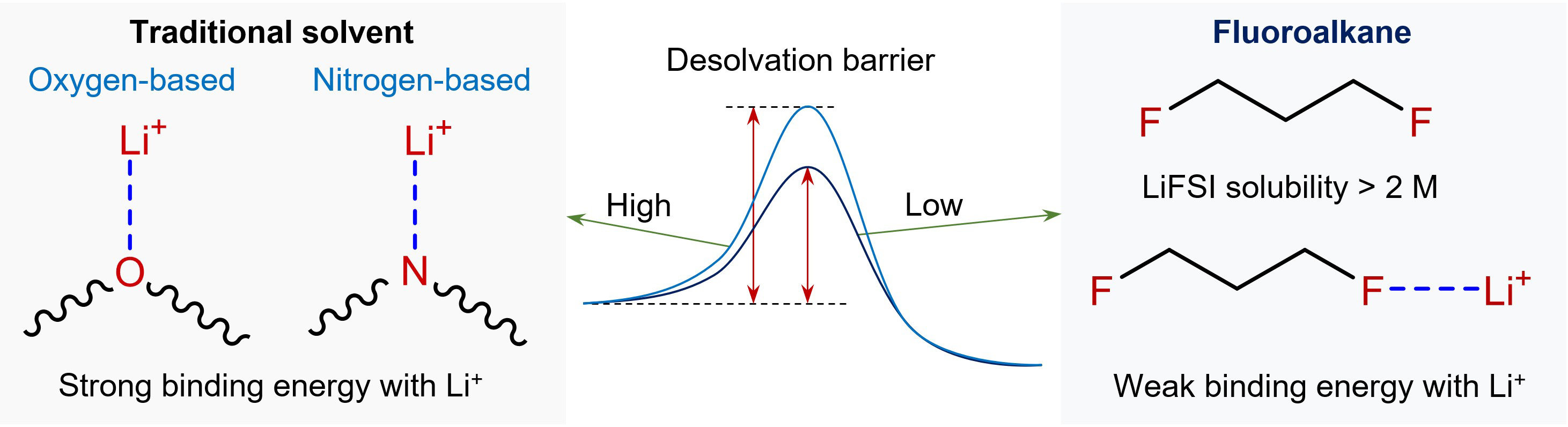
For several decades, electrolyte chemistry in batteries has followed a fundamental principle: to enable rapid ion transport in liquid electrolytes, solvents should exhibit sufficiently strong dipole-ion interactions to dissolve large amounts of lithium salt. As a consequence, molecules containing oxygen and nitrogen donor atoms have dominated electrolyte solvent design, ranging from carbonates in traditional lithium-ion batteries to ethers in many emerging systems[1,2]. Unfortunately, this prevailing approach involves an inherent trade-off. Strong coordination between these donor atoms (N, O) and Li+ facilitates salt dissociation in the solvent. However, it also hinders the release of Li+ from the inner solvation shell at the electrode surface. Even worse, this desolvation process is particularly challenging at low temperatures [Figure 1].
Figure 1. Comparison of desolvation barriers between conventional oxygen-/nitrogen-based solvents and fluoroalkanes.
Recently, Wu et al.[3] from Nankai University proposed a new class of alternative electrolyte solvents: hydrofluorocarbons. Fluorine has historically been regarded as a weak ligand for dissolving lithium salts. Here, the authors innovatively employ fluorine as the sole coordinating atom. Their research demonstrates that carefully designed monofluorinated alkanes can dissolve lithium salts at practical concentrations exceeding 2 M while maintaining weak Li+ F interactions, thereby facilitating interfacial charge transfer. This study challenges the traditional view in electrolyte coordination chemistry that high ionic conductivity requires strong solvation.
The main conceptual breakthrough is the achievement of molecular equilibrium. Fluorinated solvents have traditionally received limited attention due to their weak donor capacity, which restricts salt solubility.
This novel electrolyte design is worthy of attention because traditional designs often involve compromises. Lower viscosity is conductive to mass transport but may decrease electrochemical stability or ion dissociation. Similarly, weak coordination can facilitate desolvation but might also downgrade electrolyte conductivity. Encouragingly, DFP seems to strike a favorable balance. Solvation structure analysis via Raman spectroscopy and molecular dynamics simulations shows that the DFP-based electrolyte is mainly composed of ion-pair aggregates rather than solvent-separated ion pairs, unlike conventional oxygen-based solvents.
This insight is important for lithium-metal batteries, which offer significantly higher energy density than today's graphite-based cells but are limited by unstable interfaces, low Coulombic efficiency, and severe performance degradation at low temperatures. In the new study by Wu et al.[3], lithium plating and stripping in DFP-based electrolytes achieve Coulombic efficiencies of up to 99.7%, and the exchange current density at
The research further highlights that the primary solvation shell is not merely a structural feature but a critical determinant of overall battery performance. Recent research has increasingly focused on localized high-concentration electrolytes, anion-rich solvation structures, and interphase engineering.
Nevertheless, the study should not be seen as a universal endorsement of all fluorinated solvents. Molecular engineering remains complicated. Extending carbon chains or altering fluorine substitution patterns can increase boiling points and broaden the operating temperature window, but not all hydrofluorocarbons are stable against lithium metal. The authors address this issue by developing 1,3,5-trifluoropentane, demonstrating that compatibility can be recovered. Notably, its boiling point increases to 120~130 °C, compared with 43 °C for DFP. This highlights an important message: fluorine coordination offers opportunities, but only carefully designed molecular structures can effectively utilize them.
Wu et al.'s contribution[3] is of great importance because it broadens the conceptual framework of electrolyte chemistry in battery research. Traditionally, electrolyte development has relied on modifying existing solvents through additives, diluents, or concentration adjustments. In contrast, Wu et al.[3] reexamine the donor atom itself. Their hydrofluorocarbon electrolytes suggest that improved battery performance might not stem from strengthening lithium binding but from facilitating its release. This subtle shift in perspective offers a promising route toward energy-dense batteries with reliable performance under low-temperature conditions.
DECLARATIONS
Authors’ contributions
wrote and revised the manuscript: Xiong, Q.; Zhi, C.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
None.
Conflicts of interest
Zhi, C. is an International Advisory Editorial Board Member of the journal Energy Z. However, he was not involved in any stage of the editorial process, notably including reviewer selection, manuscript handling, or decision making. The other author declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Xu, K. Nonaqueous liquid electrolytes for lithium-based rechargeable batteries. Chem. Rev. 2004, 104, 4303-418.
2. Li, M.; Wang, C.; Chen, Z.; Xu, K.; Lu, J. New concepts in electrolytes. Chem. Rev. 2020, 120, 6783-819.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].