Tailored separators for diverse batteries: design, modification, and prospects
Abstract
With the diversification of energy storage technologies and the pressing demands for high-performance batteries, separators have transcended their conventional role as inert barriers to emerge as critical enablers of performance, safety, and longevity. To address the unique operational challenges across emerging battery chemistries - including lithium-ion, lithium-metal, and multivalent-ion systems - innovative separator design has become a key research focus. This review begins by outlining the fundamental properties and essential functions of separators to provide guiding principles for functional engineering. It then critically surveys recent advances in separator development tailored for diverse battery systems, systematically analyzing their critical roles, performance improvements, and persistent challenges. Emphasis is placed not only on modification strategies and implementation techniques, but also on the underlying working mechanisms and design rationales behind emerging functional separators. The realized functions are classified and discussed in detail, offering insights into how separators can be engineered to meet specific electrochemical and interfacial requirements. Finally, forward-looking perspectives on future research directions are provided, aiming to bridge fundamental understanding with practical application. By synthesizing current knowledge and highlighting future opportunities, this review seeks to foster the rational design of high-performance separators and accelerate their deployment in next-generation energy storage systems.
Keywords
INTRODUCTION
A battery is an energy conversion and storage device that transforms chemical or physical energy into electrical energy through chemical reactions or physical processes. In a narrow sense, it typically refers to a chemical battery, a device that converts chemical energy into electrical energy[1]. If this device can only convert chemical energy into electrical energy, it is called a primary battery; if it can convert chemical energy into electrical energy and vice versa, it is called a secondary battery[2,3].
A classic battery structure consists of four primary materials - cathode, anode, electrolyte, and separator - along with other auxiliary materials[4]. Within this system, the separator is one of the critical internal components, positioned between the cathode and anode. Its primary function is to prevent direct contact between the electrodes, thereby avoiding battery short circuits, while simultaneously allowing ions to pass through to facilitate controlled electrochemical reactions[5,6]. Considering the two primary functions, the former is more critical and fundamental than the latter. If the separator did not permit ion transport, the battery system would be incapable of energy conversion. However, if the separator failed to isolate different electrodes, it would often signify a complete loss of control over the battery reaction. Therefore, the foremost characteristic of a separator is the ability to isolate the electrodes and prevent short circuits[7,8]. To achieve this, the separator must possess sufficient stability - the capacity to maintain its structural integrity under any conditions, thereby ensuring effective electrode separation[9].
As battery technologies advance toward higher energy density, faster charging, and longer cycle life, separators have evolved beyond passive physical barriers[10]. Their microstructural features - including pore size distribution, porosity, and tortuosity - not only influence ionic transport but also dictate the spatial distribution of charge carriers within the cell[11,12]. Uneven distribution of ions can locally amplify concentration gradients, leading to concentration polarization at electrode surfaces. Over repeated cycling, such microstructural variations can be progressively magnified, manifesting as inhomogeneous reactions, differential solid electrolyte interphase (SEI) formation, and variations in long-term cycling stability[13-15].
Beyond structural constraints, however, a more critical limitation arises from the intrinsic material nature of separators employed in current commercial systems. Widely used polyolefin separators, while offering good mechanical robustness and chemical stability, are inherently inert and exhibit minimal active interaction with electrolyte species or electrode interfaces[5,16,17]. This chemical passivity fundamentally restricts their ability to regulate ion solvation environments, control the spatial distribution of functional additives, or modulate interfacial reaction kinetics, rendering them increasingly inadequate under demanding operating conditions[18-20]. Consequently, modern separator design has progressively shifted from a paradigm of “passive isolation” toward one of “active regulation,” in which separators are recognized not merely as structural components, but as key functional materials that shape the internal electrochemical environment, govern interfacial processes, and ultimately determine overall battery performance and durability.
Building on this perspective, this review provides a comprehensive overview of the role of separators in regulating battery performance across different systems. To enhance narrative coherence and distill transferable separator design principles, a unified framework is adopted that links separator parameters with the physicochemical processes they regulate, the failure modes they target, and the key performance metrics used for evaluation [Table 1]. Within this framework, the critical properties of separators and their influence on ion transport, interfacial stability, and thermal safety are first discussed. It then analyzes, in the context of lithium-ion, lithium-metal, and multivalent-ion batteries, the challenges associated with each system and how separators, through structure-function design, can mitigate these issues. The review highlights the critical considerations for developing advanced separators and outlines future research directions to guide the research of next-generation battery technologies.
Separator design framework table
| Separator design feature | Controlled parameter | Targeted failure mode | Representative metric |
| Thickness/Porosity/Tortuosity[21-26] | Ion transport; Ion flux distribution; Electrolyte uptake | Concentration polarization, Local depletion, Dendrite growth | Ionic conductivity; Transference number; Gurley value; MacMullin number |
| Pore size[27-30] | Ion transport; Size of shuttle species, | Concentration polarization; active-species shuttling; Interfacial instability | Ionic conductivity; Gurley value; CE |
| Surface polarity[31-35] | Wettability; Dynamic evolution of the solvation process; Interfacial ion supply | Li/Na plating; Li/Na/Zn dendrites; Unstable SEI; Unstable Li+ flux | Contact angle; Electrolyte uptake; Rct; CE |
| Thermal dimensional stability[36-39] | Structural integrity under abuse; Thermal regulation of pore structure | Internal short circuit, Side reactions shuttling, Thermal runaway | Thermal shrinkage; T1, T2, T3 |
| Mechanical compliance and structural resilience[40-42] | Transport continuity under deformation or plating/stripping | pore collapse under compression, dendrite penetration | Cycling stability, Tensile/compressive strain |
PROPERTIES AND REQUIREMENTS OF SEPARATORS
The stability imperative: preventing internal short circuits
From the perspective of stability, primary batteries differ from secondary batteries in that they do not undergo repeated material and energy conversion processes internally[43]. The storage duration of primary batteries typically far exceeds their discharge time, so separators used in primary batteries generally prioritize stability during long-term storage. For example, in zinc-manganese batteries, paper-based separators could meet stability requirements[44]. Subsequently, rechargeable secondary batteries gradually became commercialized. In nickel-cadmium batteries, separators are primarily made of materials such as non-woven fabrics and nylon. However, these materials possess limited mechanical strength and chemical stability, making them inadequate for meeting the demands of high-performance batteries[45]. Lithium-ion batteries impose far more stringent requirements on separator stability because they operate with longer cycle life, larger cell capacity, higher energy density, and more severe consequences once electrochemical runaway occurs[24,46].
The preliminary answer is polyolefin separators. In the first generation of commercial lithium-ion batteries, microporous membranes made from polyolefin materials such as polyethylene (PE) and polypropylene (PP) were widely used[47]. First, polyolefin materials exhibit excellent chemical and electrochemical stability, remaining inert to all substances within the battery system - neither undergoing chemical reactions nor electrochemical interactions during storage or operation[48]. Second, polyolefin microporous membranes exhibit excellent mechanical stability, including high tensile strength, puncture resistance, and peel strength[49]. Finally, these membranes possess thermal stability, maintaining structural integrity below their melting point while closing their pores above their melting temperature to block the lithium-ion flow, thereby providing a safety mechanism[50]. Building upon these properties, composite-structured polyolefin microporous membranes have been developed, such as the PP/PE/PP three-layer structure. This design combines the advantages of different materials, delivering relatively superior thermal stability and mechanical strength[51].
Another design significantly enhancing thermal stability and mechanical strength is the ceramic-coated separator based on polyolefin microporous membranes, which represents the mainstream technical solution currently used in commercial lithium-ion batteries[52,53]. Inorganic ceramics (such as alumina or boehmite), binders, dispersants, and other components are dispersed in a liquid (typically water) to form a slurry. This slurry is then coated onto the surface of the polyolefin microporous membrane. After removing the liquid phase, an inorganic ceramic coating is obtained[54]. The inherent mechanical and thermal stability of the inorganic ceramic layer further enhances the overall properties of the separator, achieving tensile strength of 1,000 kg cm-2, puncture resistance of 300 g/20 μm, and thermal stability exceeding the melting temperature of polyolefin[46]. Specifically, when heat-resistant polymer materials such as polyimide, aramid, or polydopamine are incorporated into the inorganic ceramic layer, the dimensional integrity and mechanical stability can be maintained at temperatures exceeding 200 °C[55,56].
The designs of current commercial lithium-ion battery separators predominantly rely on polyolefin microporous membranes; their mature manufacturing processes enable mass production, offering significant advantages for current applications. While optimizing these established polyolefin-based separators remains a key research focus, the development of next-generation separator technologies that move beyond polyolefins is widely recognized as a critical challenge. Exploring alternative materials has yielded promising candidates with superior properties, albeit often at a higher manufacturing cost. For instance, polyimide (PI)[57] nonwoven separators exhibit excellent high-temperature stability and electrolyte wettability; cellulose-based separators, inspired by early separator designs, allow for more intricate pore structure engineering[58]; poly(ether ether ketone) (PEEK)[59], through sulfonation and lithiation - a concept adapted from fuel cell proton exchange membrane modifications - enables the design of advanced functional groups. Notably, polymers of intrinsic microporosity (PIMs) form microporous solids due to their rigid[60,61], contorted macromolecular chains that pack inefficiently; lacking a cross-linked covalent network, PIMs are soluble in organic solvents, facilitating their processing into films, coatings, or fibers. Currently, the practical application of these non-polyolefin-based separators is constrained by manufacturing costs and process maturity. Nevertheless, they hold significant promise for elevating separator performance and expanding applications beyond lithium-ion batteries to other energy storage systems. For example, the application of PIMs in zinc-ion batteries will be discussed in detail in the following section Tuning size effects through pore-structure engineering.
Structural design for ion permeability: porosity vs. thickness
In the previous section, we primarily focused on the stability aspects of battery separators. Beyond this insulating function, however, a high-performance separator must simultaneously facilitate efficient ionic conduction to sustain reversible electrochemical processes and enable battery operation. This requirement is primarily fulfilled through the absorption of liquid electrolyte and the establishment of continuous ion-transport pathways within the separator matrix[62]. This is why the polyolefin separator, the preliminary solution mentioned in the previous section, requires fabrication into microporous membranes. Building on this foundation, considering that polyolefin microporous membranes have become the cornerstone of commercial lithium-ion battery separator manufacturing, we must approach pore-related issues with greater caution and rigour[63].
First, critical issues remain regarding the pore structure parameters, specifically size and distribution. Concerning pore size, to effectively inhibit the diffusion of cathode materials to the anode side, the membrane’s pores must be designed with diameters smaller than the characteristic particle size of the electrode active materials[64,65]. As for porosity, it is directly correlated with the electrolyte absorption capacity of the separator. Higher porosity enhances electrolyte uptake, thereby facilitating ion transport between the electrodes and reducing the internal resistance of the battery[66]. However, high porosity can compromise the mechanical integrity of the separator, thereby posing potential risks to the safety performance of the battery. Hence, the porosity should be maintained in a reasonable range, generally 40% ~ 60%[66-69]. Overall, the pores of traditional polyolefin separators are smaller than 1 μm, and the porosity is about 40%, effectively eliminating potential safety hazards[70].
Second, pore morphology also constitutes a critical factor that requires careful consideration. If the electrolyte uniformly saturates the pores within the separator, unobstructed ion transport channels can be maintained throughout battery operation[13]. Conversely, an inhomogeneous pore distribution within the separator can lead to localized insufficient wetting. This results in a non-uniform current distribution and a correspondingly uneven ion flux, which may readily promote dendrite formation[71]. In addition to pore uniformity, the specific pore morphology should also be taken into consideration. In polyolefin microporous membranes, faster phase separation tends to yield finger-like macropore structures, whereas a slower phase separation process generally results in sponge-like pore morphology[72]. It is necessary to control the process and reasonably regulate these two morphologies to ensure that the pores possess a certain degree of tortuosity without excessively prolonging the ion migration path[73]. Regarding specific characterization methods, direct observation techniques such as electron microscopy can be employed to examine whether the desired pore morphology has been achieved[74]. Provided that the membranes are produced under an identical manufacturing process, parameters such as the Gurley value and MacMullin number can be utilized for characterization - offering a practical assessment of membrane permeability in terms of both gas and liquid transport - based on considerations of testing convenience and cost-effectiveness[75].
Furthermore, it is essential to evaluate the relevant parameters of separator pores in conjunction with its thickness. Since the separator does not participate directly in electrochemical reactions, it does not contribute to the energy density of the battery[22]. Amid the ongoing pursuit of higher energy density in batteries, it may appear advantageous to minimize the mass and thickness of the separator through optimized manufacturing processes[76,77]. Currently, leading separator manufacturers can produce polyolefin separators as thin as 4 μm or even less at their process extremes. However, questions arise regarding whether excessively thin and porous separators possess sufficient inherent strength to meet stability requirements[47]. Simultaneously, concerns persist about whether the relevant pore parameters of separators produced at these process extremes can maintain long-term stability. In fact, a closer examination reveals that the aforementioned separator characteristics - including porosity, pore size, and pore morphology - exert an indirect yet significant influence on overall battery performance[78]. Recent studies over the past five years have shown that the ion conductivity of commercial polyolefin separators typically ranges from 0.3 to 0.9 mS cm-1. The design of different separators significantly impacts the ion conductivity of the battery[79-81]. Consequently, while pursuing thinner separators, manufacturers often reduce porosity and alter pore morphology to maintain structural integrity, which can adversely affect electrolyte wetting properties[82]. In practical separator development, a holistic optimization of thickness and pore-related parameters is essential to achieve an optimal performance balance. Currently, polyolefin separators for power lithium-ion batteries have reached thicknesses as low as 7 μm, while those for energy storage and consumer electronics typically measure around 9 μm[82]. Correspondingly, at these thickness levels, the characterization and control of pores have matured within the industry, with established development approaches aimed at achieving a balanced performance profile across all aspects.
Functional evolution: from passive conduit to active promoter
Based on achieving an optimal balance between properties such as porosity and thickness, separators have been engineered, manufactured, and deployed. Notably, polyolefin separators have emerged as the preferred option across diverse battery systems[83]. However, under real-world operating conditions - which often deviate substantially from idealized laboratory environments - the exclusive reliance on polyolefin separators proves inadequate.
Take power lithium-ion batteries as an example: First, the intrinsic design requirements of power batteries necessitate higher energy and power density, driving the need for separators with reduced thickness and optimized porosity[84]. Second, the dynamic charge/discharge profiles encountered during vehicle acceleration and deceleration differ substantially from the steady-state conditions typical of laboratory testing, elevating the risk of electrical abuse such as overcharge or over-discharge. Third, operational accidents can subject battery packs to mechanical abuse - including impact, compression, or penetration. Finally, heat accumulation during normal operation or resulting from the aforementioned electrical or mechanical abuse can induce thermal runaway (TR)[85]. In summary, when considering the specific design requirements, operational dynamics, and potential abuse scenarios, it is evident that conventional polyolefin separators alone are insufficient to meet the critical performance and safety demands of power lithium-ion batteries[86].
To address these critical performance and safety requirements, researchers are conducting targeted R&D on separators to endow them with specific technical properties - a process widely referred to as functionalization or functional modification[66,87]. Based on the intended performance outcomes, functional modification can be broadly categorized into two main types: enhancement of intrinsic properties and integration of supplementary functionalities. The former typically focuses on improving separator stability, further enhancing its reliability as a battery component separating the cathode and anode while balancing factors such as porosity and thickness[87]. This approach has seen considerable research and application, and the most common method for enhancing the intrinsic functions of separators is the ceramic-coated separator mentioned in Section The stability imperative: preventing internal short circuits. The inorganic ceramic layer significantly improves the overall dimensional stability, mechanical stability, and thermal stability of the separator[88], which will not be elaborated upon here.
Regarding the integration of supplementary functionalities, this concept originated primarily from the practical demands of industrial battery manufacturing[89]. In actual battery manufacturing, separators and electrodes are assembled into formed cells through winding or stacking processes, followed by encapsulation in rigid cases or aluminum-laminated film pouches and subsequent electrolyte filling. During this process, challenges such as gas entrapment, interfacial gaps between the separator and electrodes, and separator wrinkling inevitably arise[90]. Such macroscopic defects at the interface lead to uneven internal resistance distribution. During battery cycling, localized over-charging or over-discharging may occur, thereby affecting battery consistency and cycle stability[91]. The most widely adopted solution involves coating the separator with a thin polymer layer, typically polyvinylidene fluoride (PVDF), and introducing an additional hot-pressing step during cell assembly. This process promotes electrode-separator adhesion, eliminates entrapped air within the interfacial gaps, and in practice, also enhances the structural rigidity of the cell while maintaining uniform thickness consistency. Furthermore, the strong affinity of PVDF for the electrolyte enhances the interfacial electrolyte retention capacity, thereby contributing to improved battery performance[91]. Consequently, researchers have shifted focus toward integrating supplementary functionalities into separators to enhance their adaptability across diverse application scenarios.
Moreover, inspired by advances in solid-state electrolyte technology, a growing number of studies no longer regard separators merely as “passive” physical barriers. Instead, they are focusing on developing them into “active” multifunctional integrated components[92,93]. In solid-state batteries, solid electrolytes serve dual roles as both separators and electrolytes. Considering the technological continuity in transitioning to solid-state batteries, researchers are beginning to treat separators and electrolytes as an integrated system, evaluating their interactions and overall performance holistically[94]. Currently implemented semi-solid and quasi-solid batteries adopt this strategy, actively advancing novel integrated electrolyte-separator materials. These materials function as the primary ion-transport medium while concurrently providing mechanical support and interfacial stabilization[95,96].
Compared to complex electrolyte compositions, separators feature simpler structures, clearer functional mechanisms, and more diverse control methods, making them a preferred focus for related research[97]. Such studies can generally be classified into two categories: one involves modifying the separator’s surface or bulk structure to impart ionic conductivity, while the other enhances the ionic conductivity of electrolytes within the porous network and at interfacial regions through molecular or chemical interactions[98]. The most representative approach in this field involves leveraging the abundant pore structures within the separator to construct functionalized networks that enhance ionic conductivity. This strategy of leveraging tailored pore structures represents a distinctive advantage inherent to separators, a dimension seldom explored in solid-state electrolyte research. A detailed overview of this research trend - transforming separators from passive ion-permeable components into active ion-conducting elements - will be provided in Section The optimization of solvation structure.
The separator value chain: from laboratory to commercialization
Whether employing passive or active separators, their functional design is grounded in empirical rationale. In general, the function of separators originates from the practical requirements of battery systems, while their performance parameters are often critically dependent on the manufacturing process employed. Indeed, such functional modifications are guided by specific research, development, and manufacturing paradigms. A fundamental principle is that any given separator must be paired with a tailored preparation technique[99].
The following are representative preparation methods for common separator types: PP microporous separators are typically fabricated using dry-stretching processes; PE microporous separators employ wet-stretching methods; polymer nonwoven separators are prepared via electrospinning, meltblown spinning, or centrifugal spinning; ceramic-coated separators rely on anilox roll-coating techniques; and polymer-coated separators are generally manufactured through spray-coating of dilute slurries[99-104]. Polyolefin membranes form the established foundation of separator technology. Accordingly, the following discussion will focus on their two primary manufacturing processes: dry and wet processing.
The dry process involves a sequence of four principal steps: melting, extrusion, annealing, and stretching, which collectively induce porosity to form the microporous membrane. The process begins by heating and extruding the polymer into a thin sheet. Subsequently, thermal annealing is performed to develop oriented microcrystalline structures. The final pore structure is then created through controlled uniaxial or biaxial stretching[100]. Uniaxially stretched separators possess anisotropic tensile properties, with significantly higher strength in the longitudinal direction than in the transverse direction. This directional strength profile therefore helps minimize breakage during demanding assembly operations like winding. Comparatively, biaxially stretched separators, while having lower overall tensile strength, offer markedly better thermal shrinkage resistance[100]. This type of method is only applicable to semi-crystalline polymers, such as PP resin.
The wet stretching process is primarily applied to PE and operates on the principle of thermally induced phase separation for pore formation. Initially, a high-boiling-point nucleating agent is blended and melted with the polyolefin to create a homogeneous mixture. Upon rapid cooling, the polyolefin crystallizes into a film, while the pore-forming agent induces phase separation. Subsequently, the film is stretched to finalize the microporous membrane structure. The pore-forming agent is subsequently extracted, thereby creating a permanent porous network at the sites where it was originally dispersed[100,105]. The phase inversion process, involving polymer dissolution, film casting, and solvent precipitation, relies on a ternary system of polymer, solvent, and non-solvent. The mutual diffusion of solvent and non-solvent induces thermodynamic instability, which is the fundamental mechanism for phase separation and subsequent pore generation. Thus, pore morphology can be adjusted by precisely controlling the phase separation process[100,105].
Given that separators are essential battery components, research must be firmly rooted in application requirements. Therefore, after identifying a preparation method, the research approach - especially for functionalization - should systematically convert battery-level challenges into precise technical specifications and target parameters for the separator[106]. Take power lithium-ion batteries as an example. Separator research in this context must address the actual operating conditions, which include high energy density, large single-cell capacity, fluctuating currents, and the critical need for stability under TR scenarios triggered by mechanical abuse[107,108]. Extensive data from traditional battery systems allows researchers to simplify requirements into a few key metrics, making performance improvements more targeted.
In transitioning from laboratory validation to industrial production, the decisive factor often shifts from technical feasibility to comprehensive cost optimization, provided multiple methods meet the core requirements. For example, in fabricating high-temperature-stable separators, both polydopamine and phenol-formaldehyde resin coatings achieve the target of enhanced dimensional stability[109]. However, the significantly lower cost of phenol-formaldehyde resin renders its application and potential further modification a more commercially viable and industrially rational choice[109]. Comprehensive cost evaluation also accounts for process complexity, not just material expense. Consider polymer-coated ceramic separators: although gravure and spray coating both achieve coverage, the gravure process requires iterative slurry formulation, frequent material swaps, and extensive roller maintenance during development and production. This inherent complexity often makes spray coating a more efficient and cost-effective industrial choice[102]. The latter only necessitates preparing polymer dilute solutions at a fixed concentration, with actual development involving spray flow rate control to produce membranes with different polymer loadings[103,104]. Consequently, the current large-scale production of PVDF-coated separators is predominantly based on the spray coating method using aqueous dilute slutions.
Therefore, in functional separator research, battery application constitutes both the origin and the final destination of the development cycle. This closed-loop approach necessitates not only bench-level testing of the separator’s physicochemical properties but, crucially, its system-level validation within complete battery cells[110,111]. Typically, both separator manufacturers and battery cell producers conduct independent yet collaborative evaluations of functional separators within full battery systems, using established benchmark data.
Consequently, a universal framework for functionalized separator research is proposed. It encompasses the translation of specific battery requirements into concrete separator parameters, the application of tailored functionalization methods, and the critical integration of cost and manufacturability assessments, culminating in validation within full battery cells[112]. This framework aims to achieve the desired performance while fulfilling the initial application requirements.
Separator theoretical design: simulation modeling for battery separator failure analysis
The design of battery separators plays a pivotal role in the performance and safety of modern energy storage systems. As battery technology advances, relying solely on traditional experimental methods to predict separator behavior under diverse conditions becomes increasingly difficult. Simulation modeling techniques, such as finite element analysis (FEA) and molecular dynamics (MD), have proven invaluable in addressing these challenges by offering critical insights into separator failure mechanisms. These models predict separator behavior under stress conditions like mechanical deformation, thermal runaway, and electrolyte degradation[113,114]. By simulating environmental factors such as temperature, pressure, and chemical interactions, researchers can identify potential failure points early in the design process and improve separator integrity.
In addition, the pore structure of separators significantly influences lithium-ion transport and interfacial stability. Changes in pore structure can impact lithium-ion solvation dynamics, affecting transport efficiency and interfacial stability[115]. Simulation techniques help model these effects, enabling the development of separators that optimize both ionic conductivity and mechanical strength. This approach ultimately enhances battery safety and performance. Integrating simulation modeling into separator development accelerates innovation, reducing the time and costs typically associated with experimental testing. By predicting separator performance based on design parameters like pore size and surface characteristics, researchers can optimize separator design and ensure the development of high-performance, reliable battery systems.
SEPARATOR DESIGNS FOR LITHIUM- AND SODIUM-ION BATTERIES
The pursuit of higher energy density and ultrafast charging makes the electrochemical and thermal processes in lithium-ion batteries more intense and more strongly coupled, thereby creating a more complex internal reaction environment[54,116,117]. As a consequence, local imbalances and parasitic reactions are more likely to accumulate, increasing the susceptibility of the system to TR[118-120]. Extensive studies have systematically analyzed the TR process in batteries[121]. Among these, Feng et al. proposed a representative framework that divides TR evolution into distinct stages marked by three characteristic temperatures (T1, T2, and T3)[122,123]. The onset of TR originates from the breakdown of the SEI on the anode. Once the SEI protection is lost, the exposed anode active materials react directly with electrolyte solvents, triggering self-heating within the cell. The temperature at which self-heating initiates is defined as T1, representing the initial stage of TR[124]. With continuous heat accumulation, interfacial side reactions are progressively amplified, accelerating the heat generation rate; the temperature at which the self-heating rate reaches 1 °C min-1 is defined as T2, denoting the triggering point of TR[54]. Extensive studies have shown that structural failure of the separator at this stage often serves as a critical trigger for TR[125,126]. Once the separator collapses or melts, direct contact between the electrodes induces intense Joule heating, rapidly initiating a cascade of exothermic reactions accompanied by the generation of large amounts of flammable gases[127]. As a result, the cell temperature rises sharply, potentially leading to combustion or even explosion. The maximum temperature reached during TR is defined as T3, representing the severity of the event. Relying solely on enhancing the thermal limit of separators is insufficient to fundamentally suppress the initiation and propagation of TR[128,129]. Addressing this challenge requires comprehensive safety strategies that account for the full TR pathway and provide protection at critical exothermic stages and structural failure events. The separator not only functions as a physical barrier to effectively prevent direct contact between the cathode and anode, but also serves as a reservoir and transport medium for the electrolyte, establishing extensive and intimate interfacial contact with the electrodes. During the evolution of battery TR, the separator acts as a passive barrier in the subsequent reaction processes, mitigating TR propagation and severity by delaying structural failure and restricting the transport of reactive species and heat[121].
Moreover, sodium-ion batteries, benefiting from the abundant availability, wide distribution, and low cost of sodium resources, are widely regarded as an important complement to lithium-ion batteries. Compared with lithium-ion systems, they generally exhibit superior fast-charging capability and low-temperature adaptability, endowing them with distinct potential for high-power and low-temperature energy-storage applications[130]. However, this also implies that separator design for sodium-ion batteries cannot simply follow the design principles established for lithium-ion batteries. Instead, beyond ensuring reliable safety isolation, it must also accommodate rapid Na+ transport, sustained wettability under low-temperature conditions, and stable interfacial regulation[131]. In particular, the hard-carbon anode often suffers from unstable sodium-storage behavior, facile SEI instability, and irreversible sodium loss during cycling, further underscoring the critical role of separator design in regulating interfacial reactions and improving overall system stability.
The passive barrier: halting thermal runaway at the separator
The primary function of a separator is to prevent direct electrode contact while enabling stable ion transport, making its dimensional stability under thermal and mechanical stress critical for battery safety[22]. However, commercial polyethylene separators have inherently limited thermal dimensional stability. Under high-rate cycling or thermal abuse conditions, they are prone to shrinkage or local melting, compromising electrical isolation and potentially triggering internal short circuits [Figure 1A][117]. Separator shrinkage at this stage is also considered a decisive factor influencing the T2 stage during TR propagation. To mitigate this, organic or inorganic coatings can form high-modulus support networks that partially restrict shrinkage and pore collapse[132]. Yet, once the polymer substrate softens at elevated temperatures, the coating provides only partial restraint, revealing the intrinsic limitation of this approach. Strengthening the interfacial interactions between the coating and substrate - for instance, through hydrogen bonding or covalent linkages - can enhance multiphase interfacial stability and further delay membrane shrinkage at high temperatures [Figure 1B][133]. Furthermore, achieving an integrated separator architecture via outer-layer encapsulation represents an effective strategy for improving overall thermal dimensional stability [Figure 1C][134].
Figure 1. (A) Schematic illustration of the correlation between TR evolution and separator failure in lithium-ion batteries; (A) are reprinted with permission from Ref[117], Copyright © 2018 Elsevier; (B) PDA-enabled interfacial bonding between the ceramic coating and the polymer substrate; (B) are reprinted with permission from Ref[133], Copyright © 2022 Elsevier; (C) Outer PDA-coated ceramic separator with strengthened ceramic-substrate interfacial adhesion; (C) are reprinted with permission from Ref[134], Copyright © 2016 Royal Society of Chemistry; (D) Mesoporous polyimide separator prepared via the phase inversion method; (D) are reprinted with permission from Ref[140], Copyright © 2022 American Chemical Society; (E) Coaxial electrospun PAN-core/PVDF-HFP-shell polymer framework with enhanced thermal robustness; (E) are reprinted with permission from Ref[141], Copyright © 2023 Springer Nature. PD: Polydopamine; PE: polyethylene; PDA: polydopamine; CCS: ceramic coated separator; TR: thermal runaway; PAN: polyacrylonitrile; PVDF-HFP: polyvinylidene fluoride-hexafluoropropylene.
However, as battery operating conditions extend to higher temperatures, increased energy densities, or extreme abuse cases, the inherently low melting point and poor thermal stability of polyolefins present a critical bottleneck[101]. Therefore, structural approaches that merely retard failure are insufficient for safety; instead, a more fundamental solution requires shifting to substrate materials with higher intrinsic thermal stability, such as PI[50,78,135-139].
The fabrication of high-dimensionally-stable polymer membranes primarily relies on techniques such as phase inversion, electrospinning, papermaking, and casting. The different mechanisms of membrane formation and porous structural construction inherent to each method directly govern key performance parameters of the final membrane, including its pore size distribution, porosity, and long-term dimensional stability in service[24,62]. Guo et al.[140] employed solvent-nonsolvent exchange-induced phase separation to construct a continuous porous framework, which was subsequently stabilized via curing or imidization to stabilize the pore architecture. This approach enables precise control over porosity, pore size distribution, and pore connectivity while preserving the intrinsic thermal stability of the PI membrane [Figure 1D][140]. In contrast, Chen et al.[141] exploited the structural design flexibility of coaxial electrospinning to fabricate a polyacrylonitrile (PAN) core/PVDF-HFP shell membrane, integrating a thermally robust PAN framework with vermiculite nanoparticles dispersed in the shell layer. This layered structure effectively prevents structural collapse at high temperatures, maintaining relatively stable dimensional stability even at 160 °C. In comparison, polyolefin separators have completely shrunk [Figure 1E][141]. Although selecting substrate materials with enhanced dimensional stability can significantly increase the separator’s failure temperature, the high cost of such materials contradicts the development principles of current lithium-ion batteries, preventing their widespread adoption.
While advances in materials and structural design can markedly enhance separator thermal dimensional stability and delay high-temperature physical failure, battery TR is an inherently continuous process driven by coupled chemical and transport dynamics, not a mere outcome of structural collapse[142,143]. Hence, merely raising the thermal tolerance of separators cannot fundamentally curb TR onset and propagation. It is noteworthy that, beyond serving as a physical barrier isolating the electrodes, the separator also acts as a reservoir and transport medium for the electrolyte, establishing extensive and intimate interfacial contact with the electrodes. Consequently, during the evolution of battery TR, the separator can function not only as a passive barrier in subsequent reaction processes but also provide protection at critical exothermic stages and structural failure points - through strategies such as optimizing interfacial stability and restricting the transport of reactive species and heat- thereby mitigating the propagation and severity of TR. The following discussion will focus on how separator functionalization can systematically enhance battery performance throughout the entire TR process.
Interfacial stability: separator engineered with a protective layer
During the progression of TR in lithium-ion batteries, interfacial stability is a critical determinant of its initiation, while the accumulation of byproducts from parasitic reactions can compromise the steady-state integrity of the electrode interface[144]. The separator, as a key component preventing direct electrode contact, adheres closely to the electrode surface and can be modified with functional surface coatings to scavenge these reactive byproducts, providing passive protection that mitigates local imbalances and side-reaction accumulation[145].
Among common commercial electrolytes, LiPF6 is widely used due to its excellent ionic conductivity, favorable interfacial film formation, and industrial maturity[146]. However, it suffers from intrinsic thermal and chemical instability: in the presence of trace water or under elevated temperatures, LiPF6 readily decomposes to generate HF, which can severely damage electrode structures and current collectors. By enabling the in-situ capture or neutralization of HF, functionalized separators such as Al2O3, SiO2, polydopamine (PDA), and metal-organic frameworks (MOFs) can reduce its reactivity, thereby shielding both electrode interfaces and current collectors from corrosive attack.[147-150].
Beyond electrolyte-induced crosstalk at the electrodes, layered cathodes with high capacity face systemic structural degradation under high-voltage and high-temperature conditions. This includes oxygen release, the dissolution of transition metals like Mn and cation migration[151,152]. These dissolved metal ions migrate to the anode, not only disrupting ion transport and SEI stability but also, in the case of Mn2+, actively catalyzing electrolyte decomposition reactions[153]. To address these risks, an interfacial engineering strategy involves incorporating functional moieties onto the separator that selectively impede transition-metal migration while preserving unimpeded Li2+ mobility, thereby synergistically stabilizing the electrode-electrolyte interface[154,155]. To inhibit transition-metal shuttling, separators are modified through physical blocking (e.g., reduced pore size, graphene/carbon barriers) or chemical adsorption using polar-group-rich materials like polydopamine, polyethyleneimine, boehmite, and zeolitic imidazolate framework (ZIF)-8. A representative example is the work by Callegari et al., where a coating of chitosan-modified alumina nanoparticles on the separator selectively captured Mn2+ and Ni2+ via chemical adsorption, thereby mitigating crossover and enhancing cycling stability [Figure 2A][156].
Figure 2. (A) Separator coated with chitosan-modified Al2O3 nanoparticles for selective scavenging of Mn2+ and Ni2+; (A) are reprinted with permission from Ref[156], Copyright © 2024 Wiley; (B) High-temperature-induced Diels-Alder reactions of electrolyte solvents form oligomers that block separator pores, reducing Li+ transport and triggering thermal shutdown; (B) are reprinted with permission from Ref[160], Copyright © 2025 Wiley; (C) Al2O3/APP “sandwich” separator on polyethylene, sealed with phenolic resin; APP carbonization upon heating provides a protective barrier, improving battery safety; (C) are reprinted with permission from Ref[170], Copyright © 2020 Wiley; (D) Silica microcapsules containing phase-change materials on the separator enable heat-triggered release of flame retardants, enhancing thermal safety; (D) are reprinted with permission from Ref[54] ,Copyright © 2022 Elsevier. RT: Room temperature; VC-(DMFu)m: Vinyl Carbamate-(2,5-Dimethylfuran)m; APP: ammonium polyphosphate; PE: polyethylene; PPA: polyphenylenamine; PFR: polyfluorene resin; PCM: polymer composite membrane; TEP: triethyl phosphate.
Thermal safety: separator with smart pore-closing functionality
As internal cross-talk reactions intensify, the released heat accumulates, causing the cell temperature to rise. Once the temperature reaches the separator’s melting point, direct contact between the anode and cathode may occur, triggering TR. Commercial polyolefin separators, with their abundant porous structure, facilitate such cross-talk reactions, while their relatively low melting point renders them prone to shrinkage, thereby increasing the risk of internal short circuits[157].
Although the mechanisms of parasitic cross-talk reactions are complex and multifaceted, they fundamentally rely on the electrolyte as a propagation medium within the cell[158]. As a critical component, the separator can be designed with materials of differentiated thermal shrinkage behavior to achieve thermally responsive pore regulation, thereby blocking the propagation of parasitic reactions and mitigating TR[29,158,159]. In addition, designing electrolytes that undergo in situ solidification at elevated temperatures to block separator pores has also been widely employed to suppress cross-talk reactions and enhance battery thermal safety. Liu et al. reported a thermoresponsive electrolyte system that undergoes a Diels-Alder reaction at elevated temperatures to form oligomeric species[120]. This transformation simultaneously reduces ionic conductivity and blocks separator pores, enabling a two-step intelligent regulation of TR, ranging from an early-warning response at approximately 100 °C to complete shutdown at 120 °C. Notably, the separator maintains dimensional stability after shutdown [Figure 2B][160].
Beyond TR caused by thermal abuse, battery failure also stems from electrochemical abuse (e.g., overcharging, high-voltage transients) under extreme conditions. Such abuse similarly triggers core failure mechanisms - including transition-metal dissolution, uncontrolled lithium plating, and current-collector corrosion - which severely degrade interfacial stability, accelerate parasitic reactions, and ultimately lead to TR[161,162]. To address this issue, a design strategy inspired by thermal-shutdown separators can be adopted by constructing a potential-responsive functional coating on the separator surface. This coating can induce adaptive pore contraction or closure under abnormal polarization or overpotential conditions, thereby effectively suppressing detrimental ion transport, blocking the amplification of side reactions, and ultimately enhancing the intrinsic safety of batteries under electrochemical abuse conditions. For example, Wang et al. employed electroactive carbazole-based polymers as functional separator coatings, in which voltage-triggered electrochemical state transitions under overcharge conditions induce reversible closure of the separator pore structure, thereby effectively regulating ion transport during abnormal voltage excursions[163].
Fire resistance: separator enhanced with flame-retardant additives
In the late stages of TR, continued exothermic reactions and structural degradation heat the electrolyte, inducing its decomposition and generating flammable gases. Under conditions such as internal short-circuit heating or oxygen release from the cathode, these gases can ignite, further exacerbating the severity of the
Flame-retardant coatings on separators are a well-established strategy to suppress electrolyte combustion during TR. Based on their dominant mechanism, they are classified as inorganic or organic. Inorganic coatings [e.g., boehmite, Al(OH)3] undergo endothermic decomposition at high temperatures, releasing water vapor that quenches flames via localized cooling and dilution of flammable gases[167,168]. Organic flame-retardant coatings, in contrast, often incorporate P-, F-, or Br-containing additives. These act either by quenching radical species to terminate gas-phase combustion chains, or by forming an insulating char barrier through high-temperature carbonization, thereby impeding heat and fuel diffusion[169]. Leveraging these distinct but synergistic mechanisms, flame-retardant coatings on separators deliver an effective strategy for reducing fire intensity and alleviating the severity of TR incidents. Peng et al.[170] developed a thermally stable and flame-retardant smart separator by constructing an Al2O3/ ammonium polyphosphate (APP) sandwich structure on a polyethylene membrane and encapsulating it with phenolic resin; upon heating, APP carbonizes to form a protective barrier that suppresses combustion and enhances battery safety. ARC test results show that, compared to the unmodified membrane, the peak temperature during TR (T3) is reduced by approximately 280 °C [Figure 2C][170]. In addition, core-shell separator designs that encapsulate flame retardants within organic or inorganic shells enable temperature-triggered release and represent an effective strategy for mitigating TR. As an example, Liu et al. designed silica microcapsules that encapsulate both phase-change materials and flame-retardant agents to demonstrate a strategy for thermally triggered retardant release[120]. This approach effectively suppresses electrolyte combustion during the late phase of TR [Figure 2D][54].
Constructing separators from materials that combine high thermal stability with intrinsic flame retardancy is an effective materials-level strategy to enhance the safety of lithium-ion batteries. Engineering polymers such as PI, poly (p-phenylene terephthalamide) (PAA), and PEEK not only exhibit high dimensional stability, but also demonstrate excellent inherent non-flammability due to their high bond-energy backbones. During TR, these separators can maintain structural integrity over a broad temperature range while avoiding becoming combustible themselves. Their stable porous frameworks also retard rapid electrolyte release and atomization, thereby reducing the instantaneous generation of flammable vapors. In addition, some high-thermal-stability polymers form continuous carbonaceous layers upon decomposition, which act as thermal and mass-transfer barriers to suppress thermal feedback and limit fuel supply. Furthermore, synergistic integration with flame-retardant coatings can further enhance the separator’s ability to mitigate TR[171,172].
Overall, separator-based flame-retardant strategies integrate two complementary mechanisms: active functional coatings that mediate combustion via endothermic decomposition, radical scavenging, or triggered release during late-stage TR, and passive, intrinsically nonflammable materials that bolster safety by preserving structural integrity, retarding electrolyte release/vaporization, and forming high-temperature carbon barriers. Together, these event-responsive and material-intrinsic approaches provide synergistic safety enhancement for high-energy-density batteries.
The active regulator: separator-enabled electrolyte engineering for battery longevity
Owing to its intrinsic chemical inertness, the separator has long been regarded as a passive safety component in battery systems. However, growing understanding of transport phenomena and interfacial reactions has revealed that the separator can also actively influence electrolyte behavior across multiple length scales. Because the separator maintains continuous contact with both electrodes and its three-dimensional porous structure is fully infiltrated by electrolyte, it can regulate ion transport at the interfacial scale and influence electrolyte distribution and retention at the bulk scale, placing it at a critical nexus of multiscale electrochemical processes. Accordingly, rational structural and chemical design of separators provides a means to modulate electrolyte-related physicochemical processes, which in turn can influence reaction kinetics, interfacial stability, and long-term cycling behavior. In this section, separator-enabled electrolyte engineering is discussed from three interconnected dimensions: microscopic regulation - control of the local solvation structure, interfacial regulation - control of film formation reactions, and spatial regulation - design of electrolyte distribution and transport continuity
Molecular-level design: regulation of ion transport behavior
Interfacial stability is primarily dictated by the uniformity of interfacial ion flux and the rational regulation of the dynamic Li+ desolvation process[173]. For separators, pore architecture and thickness are the principal parameters governing ion transport. In general, high porosity, large pore size, and reduced thickness facilitate lower transport resistance, thereby improving ion supply at the interface and promoting interfacial stability. Nevertheless, optimal separator design should not pursue transport performance alone, but instead balance ion-transport efficiency with mechanical robustness.
From a mechanistic perspective, regulating interfacial Li+ desolvation kinetics essentially involves tuning the binding energy between Li+ and solvent molecules[174-176], as well as the corresponding decoordination process. Because the separator remains in continuous contact with both the electrodes and the electrolyte, its surface chemistry provides a valuable handle for modulating interfacial ion-transport behavior. By grafting polar functional groups onto the separator surface or introducing polar coatings, the interactions with solvent molecules and anions can, to some extent, reconstruct the local solvation environment and lower the energy barrier associated with Li+ desolvation, thereby improving interfacial reaction kinetics[177]. Meanwhile, the increased surface polarity of the separator also enhances electrolyte wettability and promotes electrolyte enrichment at the interface, which in turn alleviates concentration polarization arising from excessive local ion-flux gradients. Collectively, these effects help suppress interfacial lithium plating and favor the formation and evolution of a more stable SEI[178,179]. For example, Cheng et al. used a copper-based metal-organic framework functionalized hydroxyapatite/bacterial cellulose (Cu-MOF@HB) separator with abundant Cu2+ sites to accelerate Li+ desolvation and, combined with a high transference number of 0.74 and high ion conductivity of 0.95 mS cm-1, achieve uniform interfacial ion release, suppressing Li accumulation on the anode[111]. These results indicate that the synergistic design of separator structure and chemistry can reduce the binding energy between lithium ions and solvent molecules, while optimizing the desolvation and transport processes, thereby directly enhancing the kinetic performance and interfacial stability under fast-charging conditions [Figure 3A][111].
Figure 3. (A) Cu-MOF@HB separator featuring abundant Cu2+ sites to promote Li+ desolvation; (A) are reprinted with permission from Ref[111], Copyright © 2025 Wiley; (B) Simulated ion flux within the separator featuring different pore architectures; (B) are reprinted with permission from Ref[83], Copyright © 2024 Wiley; (C) Radiation-induced grafting of C=O and C-O functional groups onto polypropylene separators; (C) are reprinted with permission from Ref[182], Copyright © 2021 Springer Nature; (D) Volume expansion of the silicon anode leads to collapse of the separator structure, resulting in battery failure; (D) are reprinted with permission from Ref[189], Copyright © 2024 Springer Nature. Cu-MOF@HB: a copper-based metal-organic framework functionalized hydroxyapatite/bacterial cellulose; NF: nylon fber; PE: polyethylene.
Interfacial regulation: controlled release and anchoring of electrolyte additives
Beyond preventing direct electrode contact and enabling ion transport, the separator can also serve as an important functional reservoir within the battery. Its abundant porous structure provides effective sites for the storage and localized enrichment of sparingly soluble electrolyte additives. Accordingly, separators with larger pore size and higher porosity are generally more favorable for additive loading and electrolyte retention. The accompanying decline in mechanical strength can, however, be mitigated through surface-engineering strategies such as ceramic coatings.
Additives such as LiNO3[180] and sulfonates[181], can be gradually released through slow dissolution from the separator, thereby promoting an increased inorganic fraction in the SEI. This modification boosts ionic transport and thermal stability, which collectively sustain the structural and electrochemical reversibility of the anode interface during high-temperature, high-load operation. Furthermore, although phosphorus-based flame retardants help improve battery fire resistance, their direct addition to the electrolyte can trigger interfacial side reactions with the graphite anode, degrading Coulombic efficiency. In contrast, anchoring them onto polyolefin separator surfaces via radiation-induced grafting maintains flame-retardant functionality while eliminating interference with the anode interface[182]. Along the same line, chemically grafting flame-retardant additives into separator coatings offers an additional route to balance safety enhancement with electrochemical stability. By immobilizing the active species within the coating matrix, their flame-retardant function can be retained while avoiding direct participation in interfacial electrochemical reactions.
Spatial regulation: designing electrolyte distribution
In lithium-ion batteries, solvent molecules serve as the primary transport medium for Li+ ions. Adequate electrolyte wetting within porous electrodes, together with its stable retention during cycling, is essential for sustaining continuous ion transport, suppressing interfacial side reactions, and ensuring long-term operational stability of the cell [Figure 3B][83]. Commercial polyolefin separators, owing to their intrinsically low surface energy and nonpolar chemical nature, exhibit limited wettability and retention toward polar electrolytes[157]. This deficiency not only retards electrolyte infiltration during cell fabrication but also promotes nonuniform electrolyte distribution at the electrode/separator interface during prolonged cycling, leading to locally hindered ion transport and even electrolyte-depleted regions. Although separators with larger pore size and higher porosity can, to some extent, facilitate electrolyte penetration, the improvement remains limited. Conventional modification strategies primarily enhance the wettability of the external separator surface, while the internal pore channels still retain their low-surface-energy and nonpolar characteristics, thereby hindering complete wetting throughout the entire pore network[183].
Under conditions of thick electrodes or restricted electrolyte volume, this limitation further exacerbates uneven electrolyte distribution and interfacial instability[184,185]. Accordingly, fundamentally enhancing the wetting and retention of electrolyte within the separator requires systematic modulation of both the polymer matrix and the surface chemistry of the pore network. Among various strategies to enhance electrolyte affinity of separators, γ-ray irradiation is recognized as a representative bulk activation method. Sheng et al. systematically varied the γ-ray dose (20-100 kGy) applied to commercial PE separators, demonstrating that high-energy irradiation effectively cleaves the polymer chains and induces surface oxidation, thereby introducing polar functional groups such as C=O and C-O within the separator matrix [Figure 3C][182]. Beyond physical irradiation approaches, chemical modification of the polyolefin backbone has also demonstrated notable advantages in tuning polarity. Na and colleagues reported a strategy introducing catechol functional groups into the polyolefin main chain, which significantly enhanced the surface polarity and wettability of the material, enabling more stable interactions with polar media[186].
For high-volume-expansion anodes, sustained mechanical compression can cause traditional microporous separators to collapse or locally block their pores, markedly hindering ion transport, amplifying electrode polarization, and accelerating performance degradation[187,188]. Under extreme conditions, such mechanically induced transport limitations may even act as a potential trigger for cell failure [Figure 3D][189]. Enhancing the flexibility and deformation adaptability of separators during cycling is a key strategy to mitigate mechanical mismatch at the interface between silicon-based anodes and the separator. Compared with conventional polyolefin separators, high-performance polymers such as PI, PAN, and bacterial cellulose exhibit lower elastic moduli and superior toughness, enabling them to absorb mechanical stress from the anode through reversible deformation[190]. These materials can be processed into porous fibrous networks via electrospinning as functional coatings or directly fabricated into self-supporting membranes, preserving continuous ion transport pathways while reducing the risk of microstructural damage caused by electrode volume expansion.
From lithium-ion battery to sodium-ion battery
Sodium-ion batteries, due to the abundant natural reserves and low cost of sodium, are widely regarded as an ideal complement to lithium-ion batteries[191-193]. The relatively smaller Stokes radius of Na+ compared to lithium ions facilitates faster ion migration within the electrodes and lowers the desolvation energy barrier, enabling sodium-ion batteries to maintain excellent power performance even under high-rate and low-temperature conditions[194,195]. Therefore, they are particularly well-suited for high-power devices and applications in high-altitude or cold environments. From a separator design perspective, minimizing ion transport resistance by reducing separator thickness and optimizing pore structure has become a key focus in the development of sodium-ion battery separators[196-198]. At the same time, under low-temperature conditions, the increased electrolyte viscosity together with the decline in separator wettability can further weaken electrolyte infiltration and retention within the pore network, thereby hindering ion transport and aggravating interfacial polarization. Therefore, separators for sodium-ion batteries must not only exhibit low transport resistance, but also maintain excellent low-temperature wettability and electrolyte retention to ensure continuous and stable ion transport under sub-ambient conditions. In addition, effective control of separator cost remains a critical factor in enabling the broader adoption of sodium-ion batteries.
Although sodium-ion batteries generally exhibit lower energy density than lithium-ion batteries, this does not necessarily translate into improved safety. The hard carbon anodes commonly employed in sodium-ion systems tend to form quasi-metallic sodium clusters and SEI layers rich in soluble components, leading to intrinsically poor interfacial thermal stability and an increased propensity for severe exothermic reactions under abusive conditions[199,200]. In addition, the interplay between Na+ desolvation energy and deposition potential renders sodium plating more favorable at the anode surface, thereby exacerbating interfacial instability and leading to irreversible sodium loss[201-204]. Correspondingly, sodium-ion batteries have been reported to undergo noticeable self-heating at lower temperatures compared with lithium-ion systems, suggesting relatively poorer interfacial thermal stability[199]. Accordingly, stabilizing the hard carbon anode interface represents a key challenge for advancing sodium-ion batteries. As in lithium-ion batteries, functional surface engineering of separators offers an effective strategy to regulate the dynamic evolution of ion desolvation at the molecular level while enhancing Na+ transport, thereby improving interfacial stability and retarding early-stage heat generation[205-208]. Meanwhile, the intrinsic thermal stability of the separator remains a fundamental prerequisite for sodium-ion battery design[198,205,209].
SEPARATOR DESIGNS FOR LITHIUM METAL BATTERIES
As conventional lithium-ion batteries approach their intrinsic energy-density limits, further gains from optimizing electrode composition or microstructure become increasingly limited. This limitation arises from the restricted capacity of intercalation-type anodes and the unavoidable fraction of inactive components. In this context, redesigning the anode at the system level emerges as a necessary strategy to overcome energy-density bottlenecks[210]. Lithium metal, with its ultrahigh theoretical specific capacity (3,860 mAh g-1) and lowest electrochemical reduction potential (-3.04 V vs. SHE), not only enables significant improvements in both gravimetric and volumetric energy density but also simplifies anode architecture and reduces inactive mass, providing a fundamental materials basis for high-energy systems. Consequently, replacing conventional graphite with lithium metal is widely recognized as a critical pathway to surpass the current energy-density ceiling of lithium-ion batteries and support long-range electric vehicles and advanced stationary storage applications. However, this transition introduces new challenges in interfacial stability, dendrite formation, and overall safety, driving the concurrent evolution of electrolyte, separator, and interface engineering strategies.
Research progress of separators for lithium metal anodes
The electrochemical reaction in the graphite anode, a typical porous electrode, proceeds via the bulk insertion/extraction of Li+ ions. This mechanism is distinct from surface-limited reactions, as it occurs throughout the entire electrode volume. [Figure 4A][211-213]. Conversely, the lithium metal anode is a conventional planar electrode. Its electrochemical reaction - the plating and stripping of lithium - is primarily an interfacial event, predominantly occurring at the electrode surface[Figure 4B][214,215]. Unlike a porous electrode, the flat surface of lithium metal cannot homogenize the high local current density inherent to its plating/stripping reactions. The resulting concentrated ionic flux promotes inhomogeneous lithium deposition, which manifests as dendrite growth, severe interfacial fluctuations, and cumulative degradation of the SEI. The separator is not merely a physical barrier but a critical determinant of lithium metal anode behavior. In direct contact with the lithium surface, its structural and chemical properties profoundly affect local current distribution, dendrite growth patterns, and the stability of the interfacial films. Consequently, optimizing the separator design to regulate lithium deposition behavior has emerged as a key and more targeted strategy for solving interfacial problems in lithium metal batteries, as it is far more effective and direct than managing the interface through a porous electrode matrix.
Figure 4. (A) Separator-graphite electrode interface and (B) Separator-lithium metal electrode interface. SEI: Solid electrolyte interphase.
Ion transport regulation via pore structure design
The function of separators in lithium metal batteries has transcended their conventional role as passive physical barriers between the cathode and anode, evolving into a critical functional component that governs ion transport kinetics and interfacial stability. From a structural perspective, typical polyolefin separators consist of a polymer matrix and a three-dimensionally interconnected pore network. The polymer matrix provides essential mechanical strength, chemical stability, and structural integrity, yet its intrinsic ion-insulating nature precludes direct involvement in ion conduction. In contrast, the electrolyte-saturated pore network serves as the primary pathway for lithium-ion transport, “matrix-pore” dual-phase composite structure renders ion transport within the separator highly dependent on its microstructural characteristics. Structural defects such as non-uniform pore size distribution, inadequate pore connectivity, or excessively high pore tortuosity impose significant geometric constraints on lithium ions during transmembrane migration, leading to increased overall ionic transport resistance and heterogeneous local flux distribution.
Critically, the inherent ion-blocking property of the polymer matrix results in spatially discontinuous ion flux distribution within the separator, forcing ionic transport to concentrate within a limited number of effective channels. For lithium metal anode systems, such heterogeneous ion supply directly exacerbates interfacial polarization, causing local current densities to deviate substantially from the macroscopic average. This disruption in the kinetic equilibrium of lithium deposition/dissolution promotes preferential heterogeneous nucleation and oriented growth of lithium in regions of high ion flux, ultimately leading to dendritic morphologies[217]. Conversely, in areas with insufficient ion supply, discontinuous lithium deposition and repeated fracture-reconstruction cycles of the SEI occur, thereby accelerating interfacial degradation processes.
This insight has redirected separator design for lithium metal batteries (LMBs) from maximizing porosity to precisely modulating ionic flux distribution[83]. Ideally, Li+ migration across the separator should follow a vertical trajectory. To achieve this, vertically aligned or low-tortuosity pores are engineered to specifically reduce diffusion resistance, ensuring a continuous and uniform ionic flux along the thickness direction. This effectively mitigates concentration gradients and local polarization effects at the electrode-separator interface that arise from transport limitations. As demonstrated by Lee et al.[218], separators with deliberately designed tortuosity gradients - exhibiting finger-like (LT, τ = 1.13), mixed finger-sponge (MT, τ = 1.52), and sponge-like (HT, τ = 2.88) morphologies [Figure 5A] - were simulated to reveal that vertically aligned channels in the LT separator enable homogeneous Li+ distribution by suppressing through-thickness concentration gradients [Figure 5B]. This uniform ion supply reduces interfacial reaction heterogeneity on the lithium metal anode, promoting smooth deposition and ultimately yielding a stable LT-Li4‖NCM811 full cell with 80% capacity retention after 205 cycles.
Figure 5. (A) SEM images of separators with different tortuosity and corresponding effects on Li+ deposition behavior; (B) Concentration distribution of Li+ in LT, MT, and HT host structures; (A and B) are reprinted with permission from Ref[218], Copyright © 2025 Wiley. LT: Finger-like; MT: mixed finger–sponge; HT: sponge-like; SEM: scanning electron microscope.
Building upon this foundation, hierarchical pore architectures extend the design space for pore-structure regulation. In such architectures, macropores establish low-impedance, fast ion-transport pathways, while meso- and micropores function as local “flux regulators” near the electrode interface. In this context, Yi et al.[114] developed the S7540 separator, which integrates ultra-high porosity with a broad, multimodal pore-size distribution, thereby leveraging a synergistic ion-transport mechanism across macro (> 1 μm) and micro-/mesoporous (< 100 nm) regimes. This hierarchical architecture enables length scale coupled enhancement of ionic conduction (2.45 mS cm-1). Beyond hierarchical porosity, gradient pore architectures represent another effective design strategy, in which the pore size varies continuously across the separator thickness[83]. Separators with asymmetric or graded pore distributions offer a distinct advantage: a higher pore density or smaller pore size on the lithium metal side forms an interfacial ionic-flux buffering layer, while the bulk of the separator retains high overall transport efficiency[113,219]. This spatially asymmetric design enables the pore architecture to actively modulate lithium deposition kinetics, transforming it from a passive ion-conduction medium into a dynamic interfacial regulator.
Beyond the physical architecture, the chemical nature of separator pore surfaces also plays a critical role in regulating lithium metal interfacial behavior. Pores are not merely channels for ion transport; they form confined microenvironments where the chemical functional groups on pore walls significantly influence the solvation structure of Li+, the extent of anion participation, and the orientation of solvent molecules. Compared with the bulk electrolyte, this confined environment enhances surface-ion interactions, thereby governing ion distribution and transport kinetics within the pores. Accordingly, various surface grafting and modification methods - such as ultraviolet, electron-beam, plasma, and γ-ray irradiation - have been developed in recent years. These techniques enable the introduction of diverse functional groups while preserving pore connectivity. The chemical and electronic properties of these groups directly dictate local solvation configurations and ion-transport behavior. Specifically, introducing functional groups with varying polarities, Lewis acid-base characteristics, or charge properties onto the surface and pore walls of separators enables selective interactions with solvent molecules and/or Li+, thereby reconstructing the local solvation environment within the confined pores[220]. For example, functional groups such as amino (-NH2), carboxyl (-COOH), and fluorinated (-F), which exhibit distinct electronic effects[221], are able to form weak coordination interactions with Li+. These interactions establish interfacial hopping sites along pore surfaces, promoting partial de-solvation and enabling Li+ migration through surface-assisted pathways. Consequently, ion transport transitions from solely relying on bulk electrolyte conduction to a hybrid mechanism that combines bulk diffusion with interfacial surface conduction. Conversely, charged or strongly polar functional groups can selectively adsorb or restrict anions, thereby raising the Li+ transference number. Representative examples such as sulfonate groups (-SO3-) have demonstrated effective regulation of anion distribution and improved uniformity of Li+ transport[53]. The incorporation of such ion-selective sites alleviates concentration gradients and polarization caused by pore-scale heterogeneity or local electrolyte depletion - a factor especially critical for lithium metal anodes governed by interfacial reactions.
Based on the above mechanism, the function of separator pores has gradually evolved from the traditional role of serving as passive electrolyte reservoirs to acting as chemically active microenvironments capable of regulating ion behavior. Within the confined pore space, the introduction of functional groups with Li+ affinity or coordination capability can modulate the local solvation structure, facilitate the partial de-solvation of Li+, and create continuous ion migration sites along the pore walls. As a result, Li+ transport can be guided along preferential pathways to a certain extent. In this context, separator designs that endow pore surfaces with ion-regulation capability can be regarded as a form of “active separator”.
It should be emphasized that although this “active” characteristic shares certain similarities with the ion-transport behavior observed in solid polymer electrolytes, ion conduction within separators still predominantly relies on the liquid electrolyte confined in the porous structure. The functional groups introduced on the pore walls mainly regulate the ion solvation structure, local ion concentration, and migration pathways through interfacial interactions. Their role is therefore essentially associated with interfacial regulation and transport assistance, rather than forming a continuous primary ion-conducting phase. Consequently, the function of such modified separators should be understood as modulating the local ion-transport environment and interfacial reaction kinetics, rather than replacing the electrolyte as the dominant medium for ion conduction.
From this perspective, the concept of “active separators” reflects an important evolution in separator design philosophy, shifting from purely structural optimization toward a comprehensive strategy that integrates structure, surface chemistry, and interfacial behavior regulation. By modulating ion flux distribution, solvation structures, and interfacial reaction processes at the pore scale, such functionalized separators can more effectively address key challenges associated with lithium metal anodes, including dendrite growth, interfacial instability, and enhanced polarization. Consequently, these advanced separator designs provide new insights and promising pathways for the practical implementation of lithium metal batteries.
Interfacial stabilization via separator-mediated SEI control
Building upon the discussion of how the separator’s pore structure - both physically and chemically - regulates Li+ solvation in the electrolyte, this influence extends beyond the liquid phase to include bulk-phase solvation effects on interfacial processes. This chapter will now examine functional coating strategies for separators, with a focus on how tailored coating designs can enhance the formation and stability of the SEI layer.
Polymers, due to their wide variety of chemical systems, have been widely favored for designing coatings with different mechanical, transport, and electrochemical properties. Huang et al.[222] recently discussed the role of polymer coatings in addressing the various stages of lithium heterogeneous deposition. They found that inert polymers like PDMS or PEO could suppress interfacial side reactions, thereby improving the composition of the SEI layer. Moreover, Wang et al.[223] developed a high-performance separator for lithium metal batteries by coating a PP separator with a functional polymer layer composed of polyaniline (PANI) and polyvinylidene fluoride-hexafluoropropylene (PVDF-HFP) [Figure 6A]. From the perspective of ion transport properties, the PSPH@PP separator exhibits a higher ionic conductivity (0.947 mS cm-1) than the pristine PP separator (0.768 mS cm-1). Consistently, the Li+ transference number of the PSPH@PP separator (0.765) is also significantly higher than that of the PP separator (0.451). The conjugated structure of PANI in the PSPH coating promotes electron delocalization and interface homogenization, significantly reducing the tip effect caused by uneven current density and resulting in a more uniform SEI structure. Moreover, the synergistic interaction between the π-conjugated structure and -NH+ groups in the coating further facilitates anion decomposition, thereby promoting Li+ desolvation and optimizing SEI composition [Figure 6B and C]. The high content of inorganic components notably enhances the structural stability of the SEI [Figure 6D]. Yang et al.[92] synthesized a lithium-affinity donor-acceptor polymer (ArMT), based on a triazine ring, and coated it onto a PP separator, as shown in Figure 6E. The study indicates that ArMT exhibits a stronger binding affinity toward ethylene carbonate/ diethyl carbonate (EC/DEC) solvent molecules than toward Li+. Compared with the pristine PP separator, the Li+ transference number of the ArMT@PP separator increases from 0.55 to 0.67. This behavior suggests that the preferential interaction with solvent molecules can stabilize the local solvation environment, which in turn facilitates the formation of a more uniform and stable SEI [Figure 6F and G]. Time-of-flight secondary ion mass spectrometry (TOF-SIMS) depth profiling reveals a shallow distribution of LiF2- and PO2- on the SEI surface [Figure 6H and I], indicating that passivation is confined mainly to the surface and results in a thinner, more stable SEI layer. This SEI structure improves Li+ conductivity, mitigates uneven lithium deposition during cycling, and enhances battery cycling stability.
Figure 6. (A) Current density distribution on the lithium metal surface and the corresponding magnified image; (B) Percentage of LiF and Li3N components in the SEI under different separators; (C) Schematic of SEI formation under PP and PSPH@PP separators; (A-C) are reprinted with permission from Ref[223], Copyright © 2025 Wiley; (D) Chemical structure of ArMT; (E and F) C1s and Li1s XPS spectra of lithium metal under PP and ArMT@PP separators after cycling tests; (G and H) TOF-SIMS depth profiling maps showing radial distributions of Li-, C-, LiF2-, and PO2- fragments from lithium metal under corresponding PP and ArMT@PP separators; (I) Schematic illustration of SEI formation mechanisms for conventional separators versus FSL-coated separators; (D-I) are reprinted with permission from Ref[92], Copyright © 2025 Wiley; (J) Schematic image of SEI formation mechanism; (J) are reprinted with permission from Ref[224], Copyright © 2024 Wiley. PSPH: PP: polypropylene; ArMT: a lithium-affinity donor-acceptor polymer; SEI: solid electrolyte interphase; F-SAM: fluorinated self-assembled monolayer; FSL: 3,3,3-trifluoropropylsilane sacrificial layer; XPS: X-ray photoelectron spectroscopy; TOF-SIMS: time-of-flight secondary ion mass spectrometry; ArMT@PP: a lithium-affinity donor-acceptor polymer on PP separator.
In addition, sacrificial polymer coatings have also attracted considerable attention. These coatings can be functionalized with reactive groups to promote targeted reactions with lithium metal, thereby facilitating the formation of a favorable SEI composition. Jin et al.[224] utilized a fluorinated self-assembled monolayer (F-SAM, 3,3,3-trifluoropropylsilane) as a sacrificial layer, as shown in Figure 6J. The C-F bonds in this structure react with lithium metal during cell fabrication and early cycling to form a stable SEI layer. Beyond such fluorinated sacrificial layers, other polymers - such as those containing ether or amide groups - can also serve as functional coatings.
Encapsulating functional additives within the pore structure of porous materials enables their gradual release during charge-discharge cycles, thereby allowing continuous modulation of the SEI. Liao et al.[220] used high-surface-area hollow mesoporous silica (HMS) as a LiNO3 storage carrier to obtain the commercial polyolefin separator (CSN) [Figure 7A]. The results indicate that the coating interface enriches NO3-, promoting its involvement in regulating the interfacial solvation structure of Li+. X-ray photoelectron spectroscopy (XPS) analysis further reveals that the continuously released LiNO3 is reduced on the lithium anode surface, forming an SEI layer rich in highly ion-conductive lithium nitride (Li3N) [Figure 7B], thereby enabling long-term stable and uniform migration and deposition of Li+.
Figure 7. (A) Schematic diagram of the protection mechanism of HMS@LiNO3 particles; (B) XPS analysis of SEI layers on Li metal surfaces by adopted CSN separator; (A and B) are reprinted with permission from Ref[220] , Copyright © 2025 Elsevier; (C) Schematic diagram of transport and desolvation processes of Li+ within COF channels; (D) Schematic diagram of transport and desolvation processes of Li+; (C and D) are reprinted with permission from Ref[226], Copyright © 2025 American Chemical Society; (E) Structural diagram of the COF-Cl; (F) Ionic conductivity and Li+ transference number plot; (G and H) F 1s and Li 1s of Li anode after 20 cycles in Li//Li symmetric cells with different separators; (I) Area ratio of various components in the Li 1s spectra of different separators; (E-I) are reprinted with permission from Ref[20], Copyright © 2025 Wiley. HMS: Hollow mesoporous silica; EC: ethylene carbonate; FEC: fluoroethylene Carbonate; DMC: dimethyl carbonate; SEI: solid electrolyte interphase; COF: covalent organic framework; CSN: commercial polyolefin separator.
Based on these developments, organic framework materials featuring highly tunable structures and abundant functional sites - notably MOFs and covalent organic frameworks (COFs) - have emerged as a prominent research focus. Chang et al.[225] reported the successful in-situ growth of ZIF-8 on a PP separator, enabling the realization of a 350 Wh/kg lithium metal pouch cell, demonstrating the potential of MOF materials in improving battery energy density. In certain framework structures, the presence of functional groups such as -SO3H, -NH2, and -F can further enhance the stability of the SEI. Chen et al.[226] proposed a PP separator modified with an anionic covalent organic framework (Bd-COF) [Figure 7C]. The abundant -SO3H groups in the Bd-COF channels strongly interact with Li(EC)4+, promoting partial desolvation of Li+. This effectively reduces solvent-related side reactions on the electrode surface and significantly lowers interfacial impedance [Figure 7D]. Yao et al.[20] employed a cationic COF - composed of positively charged organic units and weakly bonded F- ions - to modify a commercial PP separator (COF-F@PP, Figure 7E). The positively charged framework confines anions and interacts strongly with solvents, enhancing Li+ desolvation and raising the Li+ transference number to 0.66 [Figure 7F], while simultaneously suppressing interfacial decomposition and promoting the formation of an LiF-rich SEI [Figure 7G-I].
Similarly, encapsulating functional substances or additives within the pore structures of such organic frameworks enables precise regulation of the SEI. For instance, embedding polyoxometalates (e.g., phosphotungstic acid, PW12)[227] or LiNO3[228] into UIO-66 for coating modification of commercial PP separators represents an effective approach. In the former case, PW12 promotes the desolvation of Li+ within the UIO-66 pores, thereby accelerating ion transport and facilitating the formation of an SEI enriched with LiF [Figure 8A-C]. In the latter, the gradual release of LiNO3 contributes to a homogeneous, inorganic-rich SEI (primarily composed of LiF, Li3N, LiNxOy), which effectively stabilizes the lithium metal anode during long-term cycling [Figure 8D and E].
Figure 8. (A) Schematic representation of POM@MOF-coated separators enabled selective ion-transport behavior; (B) Binding site of Li+ with EC, DEC, UIO-66, and PW12; (C) 1s, Li 1s and F 1s XPS spectra of cycled Li anode with PW12@UIO66‐PP; (A-C) are reprinted with permission from Ref[227], Copyright © 2025 Wiley; (D) Rational design of LNO@UIO-66F/PI separator in LMBs; (E) Diagram illustrating the SEI composition and its key components formed during cycling with the designed separator; (D and E) are reprinted with permission from Ref[228], Copyright © 2025 Wiley. POM: Polyoxometalate; MOF: metal-organic framework; PP: Polypropylene; EC:ethylene carbonate DEC: diethyl carbonate; PW12: phosphotungstic acid; PW12@UIO66‐PP: phosphotungstic acid doped on polypropylene-modified UIO-66; PI: polyimide; SEI: solid electrolyte interphase; LMB: lithium metal battery; XPS: X-ray photoelectron spectroscopy.
Dendrite inhibition via functional separator design
The uneven distribution of Li+ at the interface leads to localized current density concentration, resulting in heterogeneous nucleation and preferential growth of lithium metal along with additional deposition overpotential. Research indicates that the overpotential generated by accelerated electrodeposition accumulates at dendritic tips, forming localized “hot spots” that further lower the nucleation barrier and promote directional dendrite growth. This process is exacerbated under coupled electrochemical-thermal effects, creating an unstable dendrite-prone environment. Therefore, enhancing interfacial heat dissipation and reducing local thermal accumulation have been recognized as effective strategies to suppress dendrite growth. Building on this, Han et al.[229] designed a rapid-heat-dissipation separator by coating a high-thermal-conductivity graphene layer onto a PP separator. During battery operation, this separator functions as a “heat sink”, significantly improving the effective thermal conductivity at the lithium metal interface, reducing local temperature gradients, thereby inhibiting dendrite formation and enhancing the overall stability of the lithium metal anode interface.
However, passive strategies such as thermal management or mechanical confinement alone are insufficient to fundamentally eliminate the risk of dendrite growth and separator penetration. Inhomogeneous lithium deposition at the interface is inevitable, and localized current concentration can still promote preferential dendrite growth. Sole reliance on mechanical suppression offers limited physical restraint, leaving short-circuit risks unresolved. Consequently, introducing functional reactive coatings on the separator anode side has emerged as a more proactive regulatory approach. Materials such as silicon[230] and black phosphorus[231] leverage their reversible reactions or alloying characteristics with lithium. When dendrites contact the coating, these materials preferentially consume active lithium, reconstitute the interfacial composition, or induce uniform interfacial reactions. This mechanism passivates and self-suppresses dendritic tips, effectively reducing the likelihood of continued dendrite growth and separator puncture, thereby significantly enhancing battery safety and cycling stability.
Towards advanced separators for lithium metal batteries: design and engineering exemplified by Li-S systems
Lithium metal batteries can directly supply lithium from the anode and therefore enable the use of high-capacity lithium-free cathodes such as sulfur and oxygen[232]. However, these conversion-type cathodes rely on conversion reactions rather than intercalation mechanisms. Although they increase energy density, they also tend to aggravate interfacial degradation of the lithium metal anode, thereby posing significant challenges to commercialization. As a critical battery component, the separator faces more demanding performance requirements in such systems. The following section will take lithium-sulfur batteries as an example to systematically analyze the challenges they impose on separator design and the corresponding mitigation strategies[233,234].
In lithium-sulfur batteries (LSBs), the solid-liquid-solid reaction mechanism rather than a simple two-phase transformation is central to their electrochemical processes. During discharge, S8 is reduced to a series of soluble long-chain lithium polysulfides (Li2S8 → Li2S6 → Li2S4). As further lithium is added, these polysulfides are converted into shorter-chain solid compounds (Li2S4 → Li2S2 → Li2S)[235,236]. However, long-chain polysulfides exhibit significant solubility in the electrolyte, allowing them to diffuse from the cathode to the anode, where they react with lithium metal, forming insulating compounds like Li2S2 and Li2S. This leads to the well-known polysulfide shuttle effect, which results in severe loss of active sulfur material, low Coulombic efficiency, and accelerated degradation of the lithium metal anode. This solid-liquid-solid mechanism is inherently more complex than the simple ion insertion and extraction seen in traditional intercalation electrodes[237-239]. It not only requires the dissolution and migration of materials but also involves their subsequent redeposition, which necessitates careful design and optimization of separators, electrolytes, and electrode materials to ensure efficient cycling and minimize issues such as polysulfide shuttling, dendrite formation, and capacity fade.
As a result, the separator must not only allow efficient ion transport but also manage the interactions between the dissolved species and the electrode materials. Therefore, the separator design must ensure: (1) Effective Containment: Preventing the migration of soluble reaction intermediates across the separator, which can lead to capacity loss and other unwanted side reactions; (2) Improved Ionic Conductivity: Facilitating the efficient transport of lithium ions across the separator without introducing significant resistance, particularly during the dissolution and redeposition processes of active materials; (3) Interface Stability: Providing a stable interface between the electrolyte and electrode to support the complex dissolution-redeposition cycles, preventing degradation of the separator and maintaining overall battery performance.
Design strategies for physical isolation and confinement
In Li-S batteries, the polysulfide shuttle effect leads to distinct formation and evolution mechanisms of the SEI on the lithium metal surface compared to graphite anodes. Graphite anodes generally develop a stable SEI layer that suppresses side reactions, whereas the SEI on lithium metal anodes is inherently unstable and subjected to far greater impact than transition metal crossover in lithium-ion batteries. This is because polysulfide dissolution and migration are intrinsic to the reaction pathway of Li-S batteries. Dissolved polysulfides migrate across the separator along the concentration gradient during charge-discharge cycles and reach the lithium metal anode surface. Due to the absence of a stable, graphite-like SEI barrier, polysulfides react directly with lithium metal, forming non-conductive species such as Li2S2. This process not only continuously depletes active sulfur species but also promotes lithium dendrite growth and SEI fracture, thereby accelerating battery performance degradation[240,241].
Therefore, in Li-S batteries, the selective barrier function of the separator is particularly critical. The separator must not only provide Li+ conducting channels but also serve as an effective barrier to prevent polysulfide migration. Physical blocking represents one of the most direct and widely adopted strategies to suppress polysulfide shuttling. This strategy mainly regulates the transport pathways and diffusion behavior of polysulfides, delaying or even blocking their transmembrane migration at the structural level, thereby effectively confining active sulfur species within the cathode region.
A widely adopted strategy involves incorporating an independent interlayer on the cathode side of commercial polyolefin separators. This design aims to suppress polysulfide migration by constructing more tortuous diffusion pathways. Carbon-based materials, including carbon black, graphene, and carbon nanotubes, are commonly employed as interlayer materials owing to their excellent conductivity, mechanical stability, and processability. These materials not only mitigate polysulfide shuttling but also enhance overall separator performance by improving electrical conductivity and providing a stable scaffold for efficient sulfur utilization. As early as 2012, Su et al. introduced a microporous carbon paper (MCP) interlayer between the cathode and separator to facilitate polysulfide adsorption in the electrolyte[242]. Experimental results indicated that after 100 cycles, the specific surface area of the MCP decreased substantially from 659 m2 g-1 to 61 m2 g-1, and the total pore volume declined from 1.94 cm3 g-1 to 0.28 cm3 g-1[242]. These changes directly demonstrate the effective accommodation and retention of soluble polysulfides within its pore structure during cycling. Furthermore, this conductive carbon material acts as a secondary current collector, enhancing active-material utilization. With this conductive porous interlayer, a pure sulfur cathode delivered an initial discharge capacity of 1,367 mA h g-1 and retained about 85% capacity after 1,000 cycles at 1 C (1 C = 1,675 mA g-1), with an average Coulombic efficiency of 97.6%[242].
Building upon the independent interlayer approach, researchers have further extended their efforts to the separator body itself, directly constructing functionalized coatings on the separator surface. This strategy enables more precise regulation of the polysulfide migration pathways. Wang et al. coated a polypropylene (PP) separator with a two-dimensional porous carbon sheet (LPC) that combines nanoscale thickness with naturally stacked layered structures, effectively suppressing polysulfide migration[243]. Beyond carbon-based materials, coating compositions have been extended to include inorganic ceramics and polymers. In addition, Wang et al.[244] proposed a novel functionalization strategy for PP separators through the in-situ polymerization of thioctic acid (PTA). Compared with direct coating methods, this heat-triggered ring-opening polymerization process not only simplifies operation but also preserves the intrinsic nanoporous structure of the separator[244]. The abundant carboxyl groups in the PTA side chains electrostatically stabilize polysulfides on the cathode side of the separator, thereby promoting polysulfide conversion and significantly enhancing overall battery performance.
Multilayer structure design is also one of the key strategies to enhance the physical isolation effect of separators. A typical asymmetric separator usually features a dense barrier layer on the cathode side, combined with a porous support layer, introducing a gradient of pore size and transport resistance along the thickness direction of the separator. For instance, Zhou et al. developed a bilayer-coated separator (SPLAOMS) by combining LiAlO2 nanorods (LAO) and conductive carbon (SP)[245]. In this structure, the Super P layer primarily acts through physical isolation to inhibit the diffusion of polysulfides, thereby improving sulfur utilization, while the LAO layer enhances ion flux by optimizing Li+ conductivity. This structural design effectively balances the issues of ion conduction and polysulfide shuttle suppression, becoming an important direction in the development of physical isolation-type separators. Furthermore, functional polymer coatings can utilize steric hindrance or electrostatic interactions to suppress the shuttle effect of soluble polysulfides and facilitate Li+ deposition kinetics. Ko et al. explored clay-based layered materials with low lithium-ion diffusion barriers, such as lithium-montmorillonite (0.15 eV), zinc sulfide (ZnS, 0.494 eV), magnesium oxide (MgO, 0.45 eV), aluminum oxide (Al2O3, 1.22 eV), and cerium oxide (CeO2, 0.66 eV)[246]. These materials enable facile lithium-ion diffusion within the sulfur cathode, thereby improving the electrochemical performance of Li-S batteries[246].
Overall, physical isolation-dominated separators provide an important structural foundation for suppressing polysulfide shuttling in lithium-sulfur batteries, and their design concepts have, to some extent, improved the cycling stability of the battery. However, under the practical operating conditions of high-energy-density lithium-sulfur batteries, relying solely on physical isolation is often insufficient to achieve long-term stable polysulfide confinement. This limitation has prompted further research into integrating chemical adsorption and catalytic functionalities, which can synergistically work with the physical isolation effect to enhance the overall performance and stability of the separator.
Design strategies for chemical adsorption and immobilization
Due to the weak adsorption energy of van der Waals forces, physical adsorption or electrostatic repulsion alone is insufficient to effectively prevent polysulfide diffusion. In contrast, chemisorption - with its stronger adsorption energy - restricts polysulfide migration through electron transfer, exchange, or sharing with polysulfides. Taking polar molecules as an example, their separated positive and negative charge centers result in uneven charge distribution or electric dipoles, forming polar covalent bonds. Atoms with lower electronegativity in these molecules exhibit relatively positive characteristics within the covalent bonds. Consequently, polar-polar interactions can occur between these less electronegative atoms and polysulfide ions carrying excess negative charge, thereby enhancing polysulfide adsorption. In recent years, designing separators modified with polar materials has become one of the most effective strategies for suppressing polysulfide shuttling.
Introducing heteroatoms to non-polar carbon materials can create quasi-polar sites, and thus help to enhance the interaction between carbon materials and polysulfides. By using graphene as a model, the graphene nanoribbons with different doped heteroatoms (e.g., B, N, O, F, P, S, and Cl)[247] have been used to evaluate polysulfide-entrapping capabilities of heteroatoms Figure 9A and B). Heteroatoms with high electronegativity and appropriate sizes, such as N and O, facilitate the formation of doping atom-Li bonds and enable the adsorption of polysulfides through electrostatic dipole-dipole interactions. Li and co-workers demonstrated that the pyridine N-doped and pyrrolic N-doped graphene exhibited high binding energy of 1.85 eV and 1.40 eV toward Li2S4, respectively, both much higher than that of graphene (0.01 eV)[248]. The Bader charge transfer analysis demonstrated that the polysulfide trapping mechanism of N-doped graphene was due to the neutral π network of the pristine graphene being influenced by N heteroatoms, which effectively facilitated the transfer of charge from polysulfides to the N-doped graphene. Fang and co-workers reported a functional PP separator coated with N-doped porous carbon nanosheets (G@PC/PP) for LSBs [Figure 9C-E][249]. Profiting from high N content and high specific surface area, the G@PC/PP separator with a low carbon loading of 0.075 mg cm-2 exhibited a more favorable polysulfide-entrapping capability than the G@PC/PP separators with lower N contents and the nonpolar carbon material modified PP separators. The LSB with a G@PC/PP separator and a sulfur loading of 3.5 mg cm-2 delivered a high initial discharge capacity of 851 mAh g-1 and maintained a discharge capacity of 754 mAh g-1 after 500 cycles at 1 C. In addition to single element doping, Huang and co-workers designed a lightweight sulfur-nitrogen dual-doped graphene (SNGE) interlayer to inhibit the polysulfide shuttling [Figure 9F][250]. Apart from abundant pore structures coupled with a high heteroatom doping concentration, the SNGE interlayer also has a good electrical conductivity, which can effectively modulate the transformation of Li2S2/Li2S. Likewise, introducing heteroatoms with lower electronegativity (e.g., B and P) can construct relative electrically positive sites to get strong interaction with electronegative polysulfides. Chen’s group fabricated a N, P co-doped honeycomb-like carbon structure (HNPC) to modify separators[251]. The strong adsorption of HNPC toward polysulfides was attributed to the formation of strong N-Li and P-S bonds between N, P heteroatoms and polysulfides. An HNPC-modified separator coupled with a high sulfur loading cathode (4 mg cm-2) enabled a high initial specific capacity of 1,387 mAh g-1 and the cell maintained 930 mA h g-1 after 200 cycles at 0.2 C. In addition to the heteroatoms-doping strategy, the introduction of polar functional groups can also regulate the polarity of carbon materials. Hydroxyl[252,253], carboxyl[254], ether[255,256], ester[257], and amino groups have been proven to be able to inhibit the shuttle effect through their interactions with polysulfides [Figure 9G][253,258].
Figure 9. (A) The schematic diagram of X-doped nanocarbon materials (X = N, O, F, B, P, S, Cl) and PBE level optimized structure of Li2S, Li2S4, Li2S8, S8 molecules; (B) The binding energies of Li2S, Li2S4, Li2S8, and S8 interacting with X-doped GNRs and pristine (undoped) GN. Reproduced with permission; (A and B) are reprinted with permission from Ref[247], Copyright © 2016 Wiley; (C) Schematic illustration of shuttling effect; (D) The separator with a barrier layer for suppressing the polysulfide migration; (E) The synthetic procedures for the G@PC nanosheets. Reproduced with permission; (C-E) are reprinted with permission from Ref[249], Copyright © 2018 Elsevier; (F) Schematic of the electrode configuration for the LSB with the SNGE interlayer. Reproduced with permission; (F) are reprinted with permission from Ref[250], Copyright © 2025 Royal Society of Chemistry; (G) The summary of binding energies between Li2S4 and GA/graphene; (G) are reprinted with permission from Ref[250], Copyright © 2018 Wiley. GO: Graphene oxide; PC: porous carbon nanosheet; PB: Prussian blue; PP: polypropylene; SNGE: sulfur-nitrogen dual-doped graphene; GNR: graphene nanoribbon; GN: graphene nanoplatelet; PBE: Perdew-Burke-Ernzerhof; GA: graphene aerogel; LSB: lithium-sulfur battery.
Traditional carbon-based coatings, while capable of adsorbing polysulfides, face limitations in their adsorption capacity due to the inherent surface chemistry of the materials, and they often perform poorly at high discharge rates. To effectively address the issues of polysulfide dissolution and migration, several novel materials, including metal oxides, sulfides, nitrides, and organic framework materials (MOFs, COFs), have been proposed for separator coatings[259-262]. These materials have shown great potential in the adsorption and stabilization of polysulfides. Metal oxides, such as TiO2 and Al2O3, are particularly effective in adsorbing polysulfides due to their surface chemical reactivity. This helps inhibit the dissolution and migration of polysulfides in the electrolyte, thus improving the cycling stability of the battery. However, their low conductivity limits their performance at high discharge rates. By combining them with conductive materials, this drawback can be mitigated, enhancing their overall performance. Meanwhile, metal sulfides like MoS2 and FeS2 not only exhibit strong chemical adsorption but can also catalyze the conversion of polysulfides, further slowing down the shuttle effect and improving the overall stability of the battery. Their high catalytic activity allows the battery to maintain excellent performance over extended cycles, and their relatively high conductivity ensures good rate performance.
In addition, organic framework materials (MOFs and COFs) have become important in lithium-sulfur battery separator research due to their highly tunable pore structures and multifunctional surface modification capabilities. The pore size, morphology, and surface chemical functionalities of these materials can be precisely designed to limit the diffusion and migration of dissolved polysulfides, particularly preventing their entry into the electrolyte. By forming strong chemical interactions with polysulfides, these framework materials not only enhance the adsorption of polysulfides but also stabilize them by tuning the interfacial solvation structure. The metal ions or organic ligands in MOFs and COFs provide abundant coordination sites that interact with the sulfur atoms of polysulfides through van der Waals forces, hydrogen bonds, and coordination bonds, thereby significantly increasing their adsorption strength and inhibiting the dissolution and migration of polysulfides in the electrolyte. Especially in MOFs, the tunability of metal centers allows different types of metal ions to form stable coordination structures with various sulfur atoms in polysulfides, optimizing the adsorption and stabilization of polysulfides. Additionally, the large surface area and pore volume of MOFs and COFs enable the accommodation of more polysulfide molecules and, through the molecular sieve effect within the pores, facilitate directional adsorption of polysulfides. More importantly, these materials can precisely control the diffusion pathways of polysulfides through pore size regulation, effectively preventing their free migration in the electrolyte. This significantly improves the energy efficiency and cycling life of lithium-sulfur batteries.
Design strategies for catalytic conversion and redox mediation
In addition to the severe polysulfide shuttle effect, the sluggish kinetic conversion of polysulfides also affects the electrochemical performance of LSBs. More specifically, at the beginning of the discharge process, the highly soluble long-chain polysulfides would inevitably diffuse into the electrolyte under the concentration gradient and gradually move toward the counter electrode, especially in the case of high sulfur loading conditions. Therefore, accelerating the conversion of highly soluble Li2Sn (4 < n < 8) to insoluble Li2S2/Li2S is of vital importance to polysulfide shuttle suppression. Similarly, if the kinetically sluggish Li2S2/Li2S cannot be oxidized to long-chain polysulfides swiftly during the charging process, there will be irreversible loss of active materials and increased polarization. Therefore, in Li-S batteries, the separator should not only confine polysulfides to the cathode side but also accelerate the conversion kinetics of these intermediates, thereby evolving into a truly functional “active separator” capable of modulating interfacial reactions.
Metal-free catalysts have garnered significant attention in Li-S batteries due to their light weight, low cost, and abundance. Carbon nitrides, for instance, exhibit unique electron distributions that enable effective polysulfide adsorption and catalysis. Garapati et al. employed g-C3N4 nanosheets (GCN) coated on a carbon-fabric gas-diffusion layer (GCN-GDL) to adsorb and catalyze polysulfides[263]. Beyond strong adsorption, the GCN-GDL interlayer enhanced electron transfer and Li+ diffusion, leading to sharper CV peaks and shifted redox potentials - indicating accelerated polysulfide conversion kinetics. Zheng et al. developed a multifunctional ion-sieve composite of g-C3N4, BN, and graphene sheets. In this design, the 3 Å nanopores of g-C3N4 selectively sieve polysulfides while permitting Li+ migration; graphene provides conductive pathways, and BN acts as a catalytic bridge between graphene and g-C3N4 to promote polysulfide redox[264]. Given its ability to form P-S and P-Li bonds with polysulfides, red phosphorus (RP) has also been explored as a weak Lewis-base coating on PP separators. DFT and XPS analyses confirm that RP donates electrons to establish these bonds. The resulting polythionate complexes indicate strong immobilization and catalytic conversion of polysulfides. Surface O=P-O groups on RP further transform into Li3PO4 during cycling, promoting Li+ transport and accelerating polysulfide reaction kinetics.
SEPARATOR DESIGNS FOR MULTIVALENT BATTERIES
Multivalent-ion batteries differ fundamentally from monovalent-ion batteries in solvent selection due to the distinct physicochemical characteristics of multivalent ions.
This necessitates the use of strong polar solvents (such as H2O) as the electrolyte for multivalent-ion batteries. This also presents a different challenge for the optimization of the separators of multivalent-ion batteries [Figure 10A][265]. On the one hand, the strong polarity and high dielectric constant of water enable effective dissolution of most multivalent-ion salts. On the other hand, aqueous solvents offer practical advantages, including wide availability, low cost, and environmental benignity. Due to the universality of aqueous electrolyte, this chapter focuses more on the optimization strategies of the separator under aqueous conditions.
Figure 10. (A) Schematic diagram of aqueous zinc-ion batteries; (A) are reprinted with permission from Ref[265], Copyright © 2023 Wiley; (B) Schematic diagram depicting the limitations of GF separators; (B) are reprinted with permission from Ref[278], Copyright © 2025 Wiley; (C) Schematic diagram depicting the preparation and function of MXene-GF separator; (C) are reprinted with permission from Ref[278], Copyright © 2022 Wiley; (D) Schematic diagram depicting the preparation and function of UiO-66 functionalized separator; (D) are reprinted with permission from Ref[285], Copyright © 2022 Springer Nature; (E) Schematic diagram depicting the preparation processes of CF and CF-SO3 separators; (E) are reprinted with permission from Ref[288], Copyright © 2024 Wiley. GF: Glass fiber; CF: carbon fiber.
Whether it’s a multivalent or monovalent ion battery separator, most strategies involve using fiber-based separators [e.g., glass fiber (GF) separators] to accommodate aqueous electrolytes. However, the optimization of the monovalent ionic battery separator system often cannot be directly applied to the multivalent-ionic battery separator system. As mentioned before, modern separators for monovalent-ion batteries have gradually shifted from a paradigm of “passive isolation” to one of “active regulation”. The “active regulation” and “passive isolation” in separator design becomes significantly more intricate for multivalent-ion batteries. Regarding active regulation strategies, multivalent systems impose far more stringent requirements for homogenizing ion flux. This requires a multifaceted strategy that addresses several factors, including the large ionic radii of multivalent cations, the precise engineering of sieving pore sizes, high desolvation energy barriers, complex anion solvation dynamics, local pH modulation, suppression of the hydrogen evolution reaction (HER), and mitigation of salt hydrolysis (e.g., the hydrolysis of ZnSO4)[266-268]. Conversely, concerning passive isolation strategies, while the non-flammability of aqueous electrolytes alleviates thermal runaway concerns, the design focus shifts critically toward optimizing the wettability and affinity between the separator and the aqueous medium. Furthermore, overcoming the inherent mechanical limitations of conventional fiber-based separators (such as GF) to ensure structural integrity against dendrite penetration remains a pivotal challenge in these aqueous multivalent systems.
Besides, the use of aqueous electrolytes also imposes more stringent requirements on the physical and chemical properties of separators. Firstly, aqueous systems require separator materials or interfaces with sufficient hydrophilicity to ensure adequate electrolyte wetting and continuous ion transport. Secondly, the inherently high desolvation energy of multivalent-ions necessitates separators that can facilitate or accelerate the desolvation process during ion migration. Finally, the relatively narrow electrochemical stability window of aqueous electrolytes demands that separators be capable of regulating water activity at the electrode interface, thereby suppressing parasitic reactions such as HER and electrode corrosion.
To address these challenges, GF separators (e.g., Whatman) are commonly used in multivalent-ion batteries. However, these separators were originally designed for filtration applications, and their highly tortuous and disordered pore structures are not optimized for electrochemical energy storage systems. To fundamentally overcome these limitations and develop separators suitable for multivalent-ion batteries, it is essential to refocus on the intrinsic properties of the charge carriers. Accordingly, the modification of separator materials through “active regulation” and “passive isolation” should be guided by the high charge density of multivalent-ions and their specific solvation characteristics.
The physicochemical properties of separators directly determine the cycling stability and safety of batteries. Because aqueous electrolytes are nonflammable, passive-isolation strategies for separators in multivalent-ion batteries should balance electrolyte affinity with suppression of free water. These strategies should alsoconsider the relationship between separator mechanical properties and resistance to zinc dendrite penetration. In this context, the affinity between the electrolyte and the separator is a critical issue in multivalent batteries. Conventional polyolefin separators, such as PE and PP, are widely used in monovalent-ion batteries but are inherently hydrophobic and poorly compatible with the aqueous electrolytes commonly used in multivalent-ion systems. As a result, fiber-based separators are often adopted instead. However, their high porosity can make suppression of free water more difficult, thereby exacerbating HER at the negative electrode interface and promoting electrode corrosion. To achieve superior mechanical properties, the thickness of the fiber-based separator is much greater than that of the commercial polyolefin separators. However, the excessively high thickness often further restricts the application of multivalent-ion batteries and to some extent limits the "active control" strategy. For active separator-control strategies in multivalent-ion batteries, greater attention should be paid to the difficulty of regulating solvation structures caused by high ionic charge density, including low transference numbers, high desolvation energy barriers, anion solvation, and large solvation-shell sizes. Generally, these differences originate from the high charge density of multivalent-ions[269-272]: (1) The high charge density of multivalent-ions leads to distinctive hydration characteristics. As summarized in Table 2[273-277], which compares the difference between ionic radii and hydrated ionic radii of K+, Na+, Li+, Ca2+, Mg2+ and Zn2+. Li+ and Mg2+ have comparable ionic radii, yet their hydrated radii are also similar; In contrast, Li⁺ and Na+ differ significantly in ionic radius but exhibit comparable hydrated radii. The unique hydrated radii of multivalent ions necessitate more rigorous design considerations for separators in multivalent batteries. Key structural features - such as porosity, pore geometry, and pore-size distribution - must be carefully tailored accordingly; (2) The high charge density of multivalent-ions is accompanied by high desolvation energy barriers. As a result, strongly polar electrolytes are usually required to effectively solvate multivalent-ions, in order to provide sufficient concentrations of charge carrier for multivalent-ion batteries. Thus, electrolyte-separator compatibility is a critical requirement for separators in multivalent-ion batteries, particularly when highly polar electrolytes are employed. Given the strong coupling between ionic charge density and solvation behavior, the deployment of multivalent-ion batteries elevates the demands on the physicochemical characteristics of separators.
The ionic radii and hydrated ionic radii of K+, Na+, Li+, Ca2+, Mg2+ and Zn2+
Commonly used separators in multivalent-ion batteries (e.g. Magnesium-ion and zinc-ion batteries) include the following categories[54,271,278,279]: (1) Polyolefin separators (PE, PP). Due to their low cost, ultrathin thickness and high mechanical strength, polyolefin separators are widely used in batteries. However, the low porosity and poor compatibility with polar electrolytes (such as aqueous electrolytes) further limited their application in multivalent-ion batteries; (2) Cellulose-based separators. As the most abundant biomass material, cellulose has been widely applied in various fields owing to its low cost, environmental friendliness and renewability. As an eco-friendly separator material, cellulose-based separators have garnered considerable attention owing to their excellent wettability, high porosity, flexibility, and good thermal stability. However, cellulose is prone to degradation in electrolytes, which can break down its mechanical strength. This raises safety concerns such as internal short circuits and dendrite penetration during long-term cycling; (3) GF separators. Benefiting from mature industrial manufacturing processes and excellent wettability toward aqueous electrolytes, GF separators have become a popular choice in academic research. Composed primarily of SiO2, GF separators feature a three-dimensional fibrous network characterized by high porosity and outstanding electrolyte wettability. This architecture provides efficient transport pathways for multivalent-ions, anions, and their solvation shells, thereby supporting rapid reaction kinetics in batteries. While GF separators serve as an important reference in aqueous multivalent-battery research, commercial products like “Whatman” glass-microfiber filters carry a design legacy rooted in filtration, not electrochemical systems. Their properties and surface functionalities are not tailored for electrochemical use, often exhibiting inherent deficits such as highly tortuous pores, inadequate mechanical strength, excessive hydrophilicity (promoting side reactions), and excessive thickness. These characteristics hinder their ability to meet the requirements of modern battery technologies for high energy density and long cycle life [Figure 10B][278].
In order to enable separators for multivalent-ion batteries to be compatible with modern battery technologies and manufacturing processes, they are generally expected to possess the following functionalities through “active regulation”: (1) High ionic transport kinetics: The separators should enhance the transference numbers of multivalent-ion and their solvation structure (interacted with anions) with distinct radii; (2) Superior mechanical performance and chemical stability: The separators should resistance to electrolyte corrosion or degradation under long cycle life, ensuring the safety and operation of multivalent-ion batteries. (3) Good compatibility with electrolytes: the separators should exhibit a well-balanced hydrophilic-hydrophobic character. On the one hand, it must retain sufficient aqueous electrolyte to enable rapid ionic transport and stable battery operation; on the other hand, excessive water uptake should be suppressed to avoid interfacial side reactions, such as hydrogen evolution and dendrite growth. Although separators do not participate directly in electrochemical reactions, they serve as critical regulators of ionic transport in the electrolyte, as well as stabilizers of interfacial processes. Beyond their primary role of physically isolating the cathode and anode, the pore structure and interfacial properties of separators can tune ion solvation structures and transport behaviors, thereby enhancing the performance of multivalent-ion batteries. The design of separators for multivalent-ion batteries is often optimized through “active regulation” strategies surface modification, interfacial coatings, and in situ formation, including (1) regulation of internal electric field, (2) tuning size effects through pore-structure engineering and (3) optimization of desolvation energy [Table 3], leading to the improvement of mechanical property, electrolyte compatibility and electrochemical performance. The design strategies of separators will be discussed below (take zinc-ion batteries as representative examples):
Optimization strategies for the separators of multi-valent ion batteries
| Battery | Separator | Modified material | Ref. | |
| The regulation of internal electric field | ||||
| Zinc-ion | GF | MXene | [282] | |
| Zinc-ion | Nanofiber | B | [280] | |
| Zinc-ion | GF | PANI | [283] | |
| Zinc-ion | PTFE | PDA/BTO | [293] | |
| Magnesium-ion | GF | Graphene@PVP | [284] | |
| Tuning size effects through pore-structure engineering | ||||
| Zinc-ion | GF | UIO-66 | [285] | |
| Zinc-ion | GF | PEDOT:PSS | [286] | |
| Zinc-ion | PIM | Polymer | [60] | |
| Magnesium-ion | GF | ZIF-8 | [287] | |
| The optimization of solvation structure | ||||
| Zinc-ion | GF | -CN | [289] | |
| Zinc-ion | Cellulose | -CFSO3 | [288] | |
| Zinc-ion | Gel | Zinc alginate | [292] | |
| Magnesium-ion | GF | Ether/-NH2 | [294] | |
| Magnesium-ion | SPEEK | -SO3H | [291] | |
The regulation of internal electric field
In zinc-ion batteries, uneven ion flux accumulation and inherent corrosion in aqueous electrolytes deteriorate interfacial heterogeneity, resulting in dendrite formation and battery failure[270,280,281]. The electric field is the driving force for the migration of multivalent-ions between the electrodes, hence, regulating the internal electric field in separators could benefit the uniform multivalent-ion flux and the delayed growth of dendrites. The Butler-Volmer equation is usually used to described the interaction between electric field distribution and local current density, which highlights that uniform electric field is essential for smooth deposition. To enhance the performance of multivalent metal anode, the dielectric materials are incorporated with separators to form the uniform electric field for deposition, suppressing uneven metal deposition and dendrite growth.
Mxene and PANI are common conductive materials used in the modification of separators [Figure 10C][282,283]: Mxene displays adjustable dielectric constants and offers directional electrical field to expedite Zn2+ flux and repel anions, while PANI-modified layer weakens the zincophilicity of the separator surface to prevent the growth of zinc dendrites toward the separator, homogenizes the interfacial electric field distribution, and induces uniform Zn2+ plating/stripping, thereby significantly enhancing the reversibility of the zinc metal anode. Additionally, graphene@PVP (polyvinylpyrrolidone) layer on separator could establish an internal electric field, thereby promoting charge transfer in magnesium-ion batteries[284].
Tuning size effects through pore-structure engineering
Based on Sand’s time equation, the initial growth time of dendrites could be delayed by reducing the current density, increasing the transference number of cations or limiting the anion transport. In hence, tuning size effects through pore-structure engineering such as MOFs and functional groups with unique structure. Song et al.[285] reported that the unique pore-structure of UIO-66 could effectively enhances the transport ability of charge carriers and demonstrates preferential orientation of (002) crystal plane, which is favorable for corrosion resistance and dendrite-free zinc deposition [Figure 10D]. The poly(3,4-ethylenedioxythiophene):polystyrene sulfonate (PEDOT:PSS) upper layer on a GF[286], possesses intrinsic ionic/electronic conductivity and abundant sulfonic acid groups, enabling a more uniform electric-field distribution and unique “cage” structurer while effectively suppressing anion migration. As for Mg2+-transport, ZIF-8 with large-area and defect-free MOF membrane eliminating the nonselective inter-crystalline diffusion of electrolytes and enabling selective transport of Mg2+ ions, while simultaneously precisely excluding solvent molecules from the MOF channels[287]. Furthermore, Tan et al. constructed a PIM layer with a rigid twisted structure and sub-nanometer-sized micropores (0.5 ~ 0.9 nm) on the membrane surface, the unique pore-channel confinement effect is utilized to uniformly distribute the Zn2+ flux and hinder the mobility of anions, eliminating local electric field concentration[60]. At the same time, the process of Zn2+ desolvation is accelerated to reduce the erosion of active water molecules on the interface. Thus, in the absence of external additives, the physical/electrochemical inhibition of zinc dendrites and the efficient blocking of hydrogen evolution/corrosion side reactions are achieved simultaneously. This significantly improves the cycling stability and Coulomb efficiency of aqueous zinc-ion batteries[60].
The optimization of solvation structure
Due to the high charge density of multivalent-ions, Zn2+ is prone to form stable solvation structures with solvents and anions, which is not conducive to the uniform deposition of working ions. Zhou et al.[288] developed a sulfonated cellulose separator with low thickness, exceptional mechanical strength and large ionic conductivity [Figure 10E]. Benefiting from the electrostatic repulsion between the -SO3- groups and SO42- anions, as well as the strong interactions between -SO3- groups and Zn2+ ions, the migration of SO42- anions can be effectively restricted, facilitating the desolvation of hydrated Zn2+ ions. Furthermore, Li et al.[289] designed a PAN-zinc trifluoromethanesulfonate (ZnTFSI) nanofibre synergistic glass fibre (PZ-GF) to modulate the interfacial deposition kinetics. Effective synergistic interactions between abundant C≡N functional groups and Zn2+ ions lead to a homogeneous electric-field distribution on the electrode surface, thereby promoting uniform Zn2+ deposition on the zinc anode and significantly suppressing rapid dendrite growth. Additionally, a strongly nucleophilic amidogen (such as -NH2 and -SO3H) donor for competing solvation coordination has been demonstrated to be effective in regulating the solvation energy of Zn2+ ions[288-292].
Although numerous separator-modification strategies have been developed for multivalent-ion batteries and have shown encouraging performance gains, their practical deployment still faces three major challenges: (1) balancing thickness with mechanical strength to resist dendrite penetration; (2) optimizing separator mass fraction relative to battery energy density; and (3) developing passive-isolation strategies that simultaneously enhance affinity for strongly polar solvents (e.g., water) while suppressing interfacial side reactions. In conclusion, starting from the application requirements of multivalent-ion batteries, targeted modification of separators is essential for performance enhancement and large-scale application of multivalent-ion batteries.
CONCLUSION AND OUTLOOK
In electrochemical energy storage systems, such as lithium-ion batteries, the separator plays an irreplaceable role by physically isolating the anode and cathode to prevent internal short circuits while maintaining the ionic transport channels required for electrochemical reactions. Key physical properties of the separator, including thickness, pore structure, wettability, and thermal dimensional stability, directly influence the battery’s electrochemical performance, safety, and cycle life. The design and improvement of separators must be fundamentally rooted in the functional requirements imposed by the battery. The design principles underlying existing commercial separators, in fact, provide valuable references for the development of separators in battery systems that have not yet been fully commercialized. While the development directions and characteristics of different battery systems vary, modification strategies for separators must address both common requirements for fulfilling basic functions and distinctions dictated by the specific application demands of each system. Furthermore, it is noteworthy that modifying one property of a separator often leads to corresponding changes in its other inherent characteristics. Therefore, a deep understanding of these intrinsic interrelationships is essential for balancing performance requirements while meeting the demands for the commercial application of separators.
With the rapid development of high-energy-density batteries, lithium metal, sodium-ion, solid-state batteries, and other emerging systems, traditional polyolefin separators are increasingly unable to meet the growing demands for safety, rate performance, and cycle stability due to insufficient heat resistance, electrolyte affinity, interface compatibility, and mechanical strength. This challenge has driven separator research from being a passive isolation material to an active functional component, with significant progress achieved through surface modification, composite designs, novel polymers, and inorganic-organic hybrid strategies. As battery applications continue to expand into electric vehicles, grid-level energy storage, wearable electronics, and extreme environments, separator design must evolve further toward multifunctionality, system adaptability, and intelligent architectures.
Future separator designs should, while ensuring basic safety functions, focus on enhancing dimensional stability and interface integrity under complex electrochemical conditions and abuse scenarios, particularly ensuring the reliability of ultrathin separators under the coupled stresses of chemistry, electrochemistry, thermodynamics, and mechanics. Additionally, separator design must address the differentiated needs of various battery systems, optimizing the balance among key factors such as mechanical strength, ionic transport, thermal stability, electrolyte compatibility, energy density, cost, and scalability.
DECLARATIONS
Authors’ contributions
Conceptualization: Chen, Q.; Yang, X.; Wang, C.
Data curation: Chen, Q.; Yang, X.; Wang, C.
Writing - original draft: Chen, Q.; Yang, X.; Wang, C.
Review & editing: Chen, Q.; Yang, X.; Wang, C.; Xu, Y.; Gao, Q.; Gao. Y.
Supervision: Huang, B.; Li, R.; Fang, X.; Zhang, P.; Zhao, J.
Writing: Review and Editing: Huang, B.; Li, R.; Fang, X.; Zhang, P.; Zhao, J.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work is supported by the National Key Research and Development Program of China (Grant No. 2021YFB2400300), the High-level Talent Team Project of Fujian Agriculture and Forestry University (118360020).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Gao, Y.; Pan, Z.; Sun, J.; Liu, Z.; Wang, J. High-energy batteries: beyond lithium-ion and their long road to commercialisation. Nanomicro. Lett. 2022, 14, 94.
2. Thakur, J.; Phogat, P. S. H. R. E. Y. A.; Jha, R.; Singh, S. Non-rechargeable batteries: a review of primary battery technology and future trends. Phys. Chem. Chem. Phys. 2025, 27, 4045-77.
3. Miao, J.; Lang, Z.; Xue, T.; et al. Revival of zeolite-templated nanocarbon materials: recent advances in energy storage and conversion. Adv. Sci. (Weinh). 2020, 7, 2001335.
4. Ye, Z.; Jiang, Y.; Li, L.; Wu, F.; Chen, R. Rational design of MOF-based materials for next-generation rechargeable batteries. Nanomicro. Lett. 2021, 13, 203.
5. Yang, Y.; Wang, W.; Meng, G.; Zhang, J. Function-directed design of battery separators based on microporous polyolefin membranes. J. Mater. Chem. A. 2022, 10, 14137-70.
6. Huang, X.; He, R.; Li, M.; Chee, M. O. L.; Dong, P.; Lu, J. Functionalized separator for next-generation batteries. Mater. Today. 2020, 41, 143-55.
7. Xing, J.; Li, J.; Fan, W.; et al. A review on nanofibrous separators towards enhanced mechanical properties for lithium-ion batteries. Compos. Part. B-Eng. 2022, 243, 110105.
8. Wu, Y.; Wu, Q.; Sun, M.; Zeng, Z.; Cheng, S.; Xie, J. Enhancing safety in lithium batteries: a review on functional separators controlling substance and heat during thermal runaway. Adv. Funct. Mater. 2025, 35, 2425698.
9. Hu, W.; Fu, W.; Jhulki, S.; et al. Heat-resistant Al2O3 nanowire-polyetherimide separator for safer and faster lithium-ion batteries. J. Mater. Sci. Technol. 2023, 142, 112-20.
10. Tang, H.; Yue, L.; Mu, X.; Xia, H.; Zhou, H. Phosphorus-based anodes for fast-charging lithium-ion batteries: advances, challenges and prospects. Energy. Mater. 2025, 5, 500151.
11. Permatasari, A.; So, M.; Nguyen, V. L.; Yano, T.; Inoue, G. Porosity and tortuosity dynamics and their impact on lithium-ion battery performance with different separator structures. J. Power. Sources. 2025, 647, 237278.
12. Wang, W.; Hao, F.; Mukherjee, P. P. Mechanistics of lithium-metal battery performance by separator architecture design. ACS. Appl. Mater. Interfaces. 2020, 12, 556-66.
13. Ji, Y.; Yang, C.; Han, J.; He, W. Functional separators for modulating Li-ion flux toward uniform Li deposition: a review. Adv. Energy. Mater. 2024, 14, 2402329.
14. Wang, Y.; Chang, X.; Li, Z.; et al. Preventing sudden death of high-energy lithium-ion batteries at elevated temperature through interfacial ion-flux rectification. Adv. Funct. Mater. 2022, 33, 2208329.
15. Zhao, S.; Ma, M.; Gao, L.; et al. Engineering the Li-ion flux and interfacial chemistry toward a stable Li metal anode via a simple separator coating strategy. New. J. Chem. 2023, 47, 7986-94.
16. Hamenu, L.; Mohammed, L.; Attah, J.; et al. LiNi1/3Mn1/3Co1/3O2/graphite cells adopting polyolefin and non-polyolefin separators for potential application in industrial manufacturing of energy storage devices. Electrochem. Commun. 2024, 158, 107644.
17. Zheng, S.; Mo, L.; Chen, K.; et al. Precise control of Li+ directed transport via electronegative polymer brushes on polyolefin separators for dendrite-free lithium deposition. Adv. Funct. Mater. 2022, 32, 2201430.
18. Lei, S.; Zeng, Z.; Liu, M.; Zhang, H.; Cheng, S.; Xie, J. Balanced solvation/de-solvation of electrolyte facilitates Li-ion intercalation for fast charging and low-temperature Li-ion batteries. Nano. Energy. 2022, 98, 107265.
19. Liao, L.; Wang, F.; He, J.; Ma, L.; Liu, Q.; Deng, Y. Ultrathin TA‐MXene modification layer with sub-nanometer interlayer channels suppresses polysulfide shuttling and accelerates Li+ desolvation. Adv. Funct. Mater. 2025, 36, e12612.
20. Yao, S.; Yang, Y.; Liang, Z.; et al. A dual-functional cationic covalent organic frameworks modified separator for high energy lithium metal batteries. Adv. Funct. Mater. 2023, 33, 2212466.
21. Huang, X.; Ellison, N. Fabricating a high performance composite separator with a small thickness for lithium ion batteries. Compos. Sci. Technol. 2018, 168, 346-52.
22. Lingappan, N.; Lee, W.; Passerini, S.; Pecht, M. A comprehensive review of separator membranes in lithium-ion batteries. Renew. Sust. Energ. Rev. 2023, 187, 113726.
23. Pan, R.; Wang, Z.; Sun, R.; et al. Thickness difference induced pore structure variations in cellulosic separators for lithium-ion batteries. Cellulose 2017, 24, 2903-11.
24. Yu, Y.; Liu, M.; Chen, Z.; et al. Advances in nonwoven-based separators for lithium-ion batteries. Adv. Fiber. Mater. 2023, 5, 1827-51.
25. Zahn, R.; Lagadec, M. F.; Hess, M.; Wood, V. Improving ionic conductivity and lithium-ion transference number in lithium-ion battery separators. ACS. Appl. Mater. Interfaces. 2016, 8, 32637-42.
26. Zhu, G.; He, Y.; Deng, Y.; et al. Dependence of separator thickness on Li-ion battery energy density. J. Electrochem. Soc. 2021, 168, 110545.
27. Liu, S.; Cheng, S.; Huang, C.; et al. Nanoporous aramid nanofiber separators with high modulus and thermal stability for safe lithium-ion batteries. Small 2024, 20, e2404639.
28. Liu, Y.; Li, C.; Li, C.; et al. Porous, robust, thermally stable, and flame retardant nanocellulose/polyimide separators for safe lithium-ion batteries. J. Mater. Chem. A. 2023, 11, 23360-9.
29. Song, Y.; Liu, X.; Ren, D.; et al. Simultaneously blocking chemical crosstalk and internal short circuit via gel-stretching derived nanoporous non-shrinkage separator for safe lithium-ion batteries. Adv. Mater. 2022, 34, e2106335.
30. Wang, Y.; Guo, M.; Fu, H.; et al. Thermotolerant separator of cross-linked polyimide fibers with narrowed pore size for lithium-ion batteries. J. Membr. Sci. 2022, 662, 121004.
31. Sheng, L.; Song, L.; Gong, H.; et al. Polyethylene separator grafting with polar monomer for enhancing the lithium-ion transport property. J. Power. Sources. 2020, 479, 228812.
32. Sheng, L.; Wang, Q.; Liu, X.; et al. Suppressing electrolyte-lithium metal reactivity via Li+-desolvation in uniform nano-porous separator. Nat. Commun. 2022, 13, 172.
33. Xiao, S.; Fu, Q.; Wang, X.; et al. Polar-rich-group triazine-based covalent organic frameworks modified separators with de-solvation effect enables uniform Li deposition for stable lithium-metal anode. J. Energy. Chem. 2025, 105, 823-34.
34. Ma, H.; Yu, J.; Chen, M.; et al. Amino-enabled desolvation sieving effect realizes dendrite-inhibiting thin separator for durable aqueous zinc-ion batteries. Adv. Funct. Mater. 2023, 33, 2307384.
35. Subramanyan, K.; Jyothilakshmi, S.; Ulaganathan, M.; Lee, Y.; Aravindan, V. An efficient upcycling of graphite anode and separator for Na-ion Batteries via solvent-co-intercalation process. Carbon 2024, 216, 118525.
36. Choi, S.; Hassan, S. Z.; Roh, Y.; et al. Comprehensive crosslinking strategy using fluorinated azide to enhance the thermal stability of ceramic-coated separators for Li-ion batteries. Chem. Eng. J. 2024, 498, 155238.
37. Luo, M.; Zhang, X.; Wang, S.; et al. A thermal-ball-valve structure separator for highly safe lithium-ion batteries. Small 2024, 20, e2309523.
38. Shen, X.; Zhang, L.; Zhu, J.; Duan, X. Super electrolyte-philic all-inorganic ceramic separators for highly robust lithium batteries. Energy. Storage. Mater. 2025, 80, 104389.
39. Wei, Z.; Zhang, N.; Feng, T.; Wu, F.; Zhao, T.; Chen, R. A copolymer microspheres-coated separator to enhance thermal stability of lithium-sulfur batteries. Chem. Eng. J. 2022, 430, 132678.
40. Sun, S.; Rui, B.; Tan, X.; Bahuguna, S.; Zhou, J.; Xu, J. Effects of electrolyte, state of charge, and strain rate on the mechanical properties of lithium-ion battery electrodes and separators. J. Mater. Chem. A. 2025, 13, 20673-87.
41. Wang, S.; Ma, Z.; Zhao, W.; et al. Temperature-shift-induced mechanical property evolution of lithium-ion battery separator using cyclic nanoindentation. ACS. Appl. Mater. Interfaces. 2022, 14, 47578-86.
42. Zhao, Q.; Wu, X.; Li, S.; et al. Boosting thermal and mechanical properties: achieving high-safety separator chemically bonded with nano TiN particles for high performance lithium-ion batteries. Small 2023, 19, e2300378.
43. Pandey, A.; Rawat, K.; Phogat, P. Shreya; Jha, R.; Singh, S. Next-generation energy storage: a deep dive into experimental and emerging battery technologies. J. Alloys. Compd. 2025, 1014, 178781.
44. Rodrigues-marinho, T.; Miranda, D.; Barbosa, J. C.; Gonçalves, R.; Lanceros-Méndez, S.; Costa, C. M. Separator membranes for aqueous zinc-manganese oxide batteries: a comprehensive review on experimental results and theoretical simulations. Sustain. Energ. Fuels. 2025, 9, 1432-46.
45. Chek, Y. W.; Lee, P. K.; Yu, J. N.; Wang, B.; Ang, D. T. Review - separators for rechargeable alkaline zinc batteries: challenges and progress. J. Electrochem. Soc. 2025, 172, 020509.
46. Francis, C. F. J.; Kyratzis, I. L.; Best, A. S. Lithium-ion battery separators for ionic-liquid electrolytes: a review. Adv. Mater. 2020, 32, e1904205.
47. Zhong, S.; Yuan, B.; Guang, Z.; et al. Recent progress in thin separators for upgraded lithium ion batteries. Energy. Storage. Mater. 2021, 41, 805-41.
48. Liu, Q.; Yu, Z.; Hu, Y.; Tang, M.; Zhang, Q.; Ye, M. Designing advanced separators toward lithium-ion batteries. Adv. Energy. Mater. 2025, 15, e03217.
49. Heidari, A. A.; Mahdavi, H. Recent development of polyolefin-based microporous separators for Li-ion batteries: a review. Chem. Rec. 2020, 20, 570-95.
50. Dai, X.; Zhang, X.; Wen, J.; et al. Research progress on high-temperature resistant polymer separators for lithium-ion batteries. Energy. Storage. Mater. 2022, 51, 638-59.
51. Li, H.; Wang, H.; Xu, Z.; et al. Thermal-responsive and fire-resistant materials for high-safety lithium-ion batteries. Small 2021, 17, e2103679.
52. Xing, J.; Bliznakov, S.; Bonville, L.; Oljaca, M.; Maric, R. A review of nonaqueous electrolytes, binders, and separators for lithium-ion batteries. Electrochem. Energy. Rev. 2022, 5, 14.
53. Zhang, Y.; Lu, J.; Jin, Z.; et al. Covalently anchoring an ultrathin conformal SiOx coating on polyolefin separator for enhanced lithium metal battery performance. Adv. Funct. Mater. 2024, 35, 2417160.
54. Liu, Z.; Peng, Y.; Meng, T.; Yu, L.; Wang, S.; Hu, X. Thermal-triggered fire-extinguishing separators by phase change materials for high-safety lithium-ion batteries. Energy. Storage. Mater. 2022, 47, 445-52.
55. Wu, D.; Dong, N.; Wang, R.; Qi, S.; Liu, B.; Wu, D. In situ construction of high-safety and non-flammable polyimide “ceramic” lithium-ion battery separator via SiO2 nano-encapsulation. Chem. Eng. J. 2021, 420, 129992.
56. Zhu, J.; Huang, L.; Xu, L.; et al. Bio-based furan aramid/ceramic-coated lithium-ion battery separators with high ionic conductivity, wettability and safety via in situ lithium compensation strategy. J. Energy. Chem. 2026, 114, 630-8.
57. Lin, D.; Zhuo, D.; Liu, Y.; Cui, Y. All-integrated bifunctional separator for Li dendrite detection via novel solution synthesis of a thermostable polyimide separator. J. Am. Chem. Soc. 2016, 138, 11044-50.
58. Liao, T.; Huang, S.; Wang, L.; Liu, W.; Wang, X.; Zhang, W. Biomimetic ultrathin cellulose composite separators for high-performance lithium-sulfur batteries. Adv. Funct. Mater. 2025, 36, e19474.
59. Weng, Y. T.; Liu, H. W.; Pei, A.; et al. An ultrathin ionomer interphase for high efficiency lithium anode in carbonate based electrolyte. Nat. Commun. 2019, 10, 5824.
60. Tan, R.; He, H.; Wang, A.; et al. Interfacial engineering of polymer membranes with intrinsic microporosity for dendrite-free zinc metal batteries. Angew. Chem. Int. Ed. Engl. 2024, 63, e202409322.
61. Mckeown, N. B. The synthesis of polymers of intrinsic microporosity (PIMs). Sci. China. Chem. 2017, 60, 1023-32.
62. Lee, H.; Yanilmaz, M.; Toprakci, O.; Fu, K.; Zhang, X. A review of recent developments in membrane separators for rechargeable lithium-ion batteries. Energy. Environ. Sci. 2014, 7, 3857-86.
63. Costa, C. M.; Lee, Y.; Kim, J.; Lee, S.; Lanceros-méndez, S. Recent advances on separator membranes for lithium-ion battery applications: from porous membranes to solid electrolytes. Energy. Storage. Mater. 2019, 22, 346-75.
64. Manly, A. J.; Tenhaeff, W. E. One-step fabrication of robust lithium ion battery separators by polymerization-induced phase separation. J. Mater. Chem. A. 2022, 10, 10557-68.
65. Su, M.; Huang, G.; Wang, S.; Wang, Y.; Wang, H. High safety separators for rechargeable lithium batteries. Sci. China. Chem. 2021, 64, 1131-56.
66. Fan, Z.; Chen, X.; Shi, J.; et al. Functionalized separators boosting electrochemical performances for lithium batteries. Nanomicro. Lett. 2025, 17, 128.
67. Ko, Y.; Jung, H. Y.; Jung, H. W.; Ko, S. M.; Lee, J. T.; You, J. A new strategy for accelerating redox kinetics in lithium-ion batteries: highly porous poly(ethylene glycol)/nanocellulose separators. Carbohydr. Polym. 2025, 356, 123372.
68. Liu, F.; Chuan, X. Recent developments in natural mineral-based separators for lithium-ion batteries. RSC. Adv. 2021, 11, 16633-44.
69. Babiker, D. M.; Usha, Z. R.; Wan, C.; Hassaan, M. M. E.; Chen, X.; Li, L. Recent progress of composite polyethylene separators for lithium/sodium batteries. J. Power. Sources. 2023, 564, 232853.
70. Li, Z.; Jiao, S.; Yu, D.; et al. Cationic-polymer-functionalized separator as a high-efficiency polysulfide shuttle barrier for long-life Li-S battery. ACS. Appl. Energy. Mater. 2021, 4, 2914-21.
71. Yang, M.; Jue, N.; Chen, Y.; Wang, Y. Improving cyclability of lithium metal anode via constructing atomic interlamellar ion channel for lithium sulfur battery. Nanoscale. Res. Lett. 2021, 16, 52.
72. Waqas, M.; Ali, S.; Feng, C.; Chen, D.; Han, J.; He, W. Recent development in separators for high-temperature lithium-ion batteries. Small 2019, 15, e1901689.
73. l’Abee, R.; Darosa, F.; Armstrong, M. J.; Hantel, M. M.; Mourzagh, D. High temperature stable Li-ion battery separators based on polyetherimides with improved electrolyte compatibility. J. Power. Sources. 2017, 345, 202-11.
74. Yi, W. J.; Niu, B.; Hu, H.; et al. In-situ grafting strategy enables functional separator for advanced lithium-sulfur batteries. Small 2025, 21, e2407982.
75. Takeda, S.; Saito, Y.; Mukai, T.; Senoh, H.; Kaneko, I.; Yoshitake, H. Effect of the stretching process of polyethylene separators on rate capability of lithium-ion batteries. J. Phys. Chem. C. 2021, 125, 12496-503.
76. Hu, M.; Ma, Q.; Yuan, Y.; et al. Grafting polyethyleneimine on electrospun nanofiber separator to stabilize lithium metal anode for lithium sulfur batteries. Chem. Eng. J. 2020, 388, 124258.
77. Gu, M.; Lee, J.; Kim, Y.; et al. Inhibiting the shuttle effect in lithium-sulfur batteries using a layer-by-layer assembled ion-permselective separator. RSC. Adv. 2014, 4, 46940-6.
78. Trinh, H. N.; Eesaee, M.; Shahgaldi, S.; Singh, J.; Hoang, T. L. G.; Nguyen-Tri, P. Advancements and challenges in polymer-based separators for lithium-ion batteries. Energy. Storage. Mater. 2025, 77, 104153.
79. Li, B.; Liu, Y.; Han, X.; et al. Electrospun nanofiber surface-modified polyethylene separator for enhanced cycling stability and low-temperature performance of sodium-ion batteries. Chem. Eng. J. 2024, 501, 157803.
80. Mun, J.; Yim, T.; Gap Kwon, Y.; Jae Kim, K. Self-assembled nano-silica-embedded polyethylene separator with outstanding physicochemical and thermal properties for advanced sodium ion batteries. Chem. Eng. J. 2021, 405, 125844.
81. Luo, W.; Cheng, S.; Wu, M.; Zhang, X.; Yang, D.; Rui, X. A review of advanced separators for rechargeable batteries. J. Power. Sources. 2021, 509, 230372.
82. Wen, Z.; Tian, X.; Zhang, Y.; et al. The influence of separator thickness on the physical properties of separator and the electrochemical properties of lithium-ion battery. J. Electrochem. Soc. 2025, 172, 090506.
83. Jia, H.; Zeng, C.; Lim, H. S.; et al. Important role of ion flux regulated by separators in lithium metal batteries. Adv. Mater. 2024, 36, e2311312.
84. Ping, W.; Xue, B.; Zhang, F.; Huang, X.; Chen, L.; Xiang, H. Ultrathin cellulose composite separator for high-energy density lithium-ion batteries. Adv. Funct. Mater. 2025, 36, e09803.
85. Ouyang, D.; Chung, Y.; Liu, J.; et al. Characteristics and mechanisms of as well as evaluation methods and countermeasures for thermal runaway propagation in lithium-ion batteries. Progr. Energy. Combust. Sci. 2025, 108, 101209.
86. Hwang, Y.; Kim, M. Effect of a polypropylene separator with a thin electrospun ceramic/polymer coating on the thermal and electrochemical properties of lithium-ion batteries. Polymers. (Basel). 2024, 16.
87. Seo, J.; Im, J.; Kim, M.; Song, D.; Yoon, S.; Cho, K. Y. Recent progress of advanced functional separators in lithium metal batteries. Small 2024, 20, e2312132.
88. Gogia, A.; Wang, Y.; Rai, A. K.; Bhattacharya, R.; Subramanyam, G.; Kumar, J. Binder-free, thin-film ceramic-coated separators for improved safety of lithium-ion batteries. ACS. Omega. 2021, 6, 4204-11.
89. Manoharan, A.; Chong, J. J.; Choong, Z. J.; et al. Optimizing lithium-ion battery manufacturing with digitalization and AI-driven frameworks. Int. J. Adv. Manuf. Technol. 2025, 142, 1-37.
90. Örüm Aydin, A.; Zajonz, F.; Günther, T.; Dermenci, K.; Berecibar, M.; Urrutia, L. Lithium-ion battery manufacturing: industrial view on processing challenges, possible solutions and recent advances. Batteries 2023, 9, 555.
91. YAN, X.; HU, Z.; LIU, F.; et al. The origin and elimination of separator wrinkles in lithium-ion batteries. Energy. Storage. Sci. Technol. 2021, 10, 156.
92. Yang, T.; Xu, X.; Chen, S.; et al. A lithiophilic donor-acceptor polymer modified separator for high-performance lithium metal batteries. Angew. Chem. Int. Ed. Engl. 2025, 64, e202420973.
93. Cao, M.; Bu, F.; Liu, X.; Ng, C. H.; Guan, C. Beyond separation: multifunctional separators in rechargeable batteries. Adv. Energy. Mater. 2025, 15, 2502540.
94. Zhang, N.; Zhao, X.; Liu, G.; et al. Solid electrolyte membranes for all-solid-state rechargeable batteries. eTransportation 2024, 20, 100319.
95. Zhang, Y.; Bahi, A.; Ko, F.; Liu, J. Polyacrylonitrile-reinforced composite gel polymer electrolytes for stable potassium metal anodes. Small 2022, 18, e2107186.
96. Ma, Y.; Wang, C.; Yang, K.; et al. Ultrathin and robust composite electrolyte for stable solid-state lithium metal batteries. ACS. Appl. Mater. Interfaces. 2023, 15, 17978-85.
97. Likitaporn, C.; Prathumrat, P.; Senthilkumar, N.; Tanalue, N.; Wongsalam, T.; Okhawilai, M. Engineering the separators for high electrolyte uptakes in Li-ion batteries. Journal. of. Energy. Storage. 2024, 101, 113861.
98. Han, D.; Lee, J.; Bang, Y.; et al. Molecularly engineered membrane-driven interphase stabilization of electrodes for Li||NCM811 cells under practical operating conditions. Energy. Environ. Sci. 2025, 18, 10112-24.
99. Zhang, J.; Liu, Z.; Dang, C.; et al. From lab to industry: high-safety separators for lithium-ion/-metal batteries. Matter 2025, 8, 102101.
100. Mun, S. C.; Won, J. H. Manufacturing processes of microporous polyolefin separators for lithium-ion batteries and correlations between mechanical and physical properties. Crystals 2021, 11, 1013.
101. Sun, X.; Zhou, Y.; Li, D.; et al. A review of electrospun separators for lithium‐based batteries: progress and application prospects. Carbon. Energy. 2024, 6, e539.
102. Lee, S. H.; Kim, J.; Kim, B. H.; Yoon, S.; Cho, K. Y. Delamination-free multifunctional separator for long-term stability of lithium-ion batteries. Small 2019, 15, e1804980.
103. Liang, X.; Zhou, Y.; Liu, D.; Wang, H.; Sun, J.; Cai, X. Optimization polypropylene separator by ultrathin Ti0.6Fe0.4O2 nanosheet coating for Li-metal battery. J. Power. Sources. 2025, 640, 236765.
104. Shen, L.; Liu, X.; Dong, J.; et al. Functional lithiophilic polymer modified separator for dendrite-free and pulverization-free lithium metal batteries. J. Energy. Chem. 2021, 52, 262-8.
105. Ding, L.; Li, D.; Du, F.; Zhang, D.; Zhang, S.; Wu, T. Novel preparation of lithium‐ion battery wet‐processed separator based on the synergistic effect of porous skeleton nano-Al2O3 in situ blending and synchro‐draw. Polym. Int. 2022, 72, 61-70.
106. Lei, Y.; Xu, L.; Chan, Q. N.; et al. Recent advances in separator design for lithium metal batteries without dendrite formation: implications for electric vehicles. eTransportation 2024, 20, 100330.
107. Zhang, J.; Huang, H.; Zhang, G.; Dai, Z.; Wen, Y.; Jiang, L. Cycle life studies of lithium-ion power batteries for electric vehicles: a review. J. Energy. Storage. 2024, 93, 112231.
108. Guo, X.; Yang, Y.; Shi, C.; Xu, M.; Liu, Y.; Zou, D. Monitoring and control of internal temperature in power batteries: a comprehensive review. Energy. Storage. Mater. 2025, 75, 104051.
109. Gu, Q.; Xue, H.; Li, Z.; Song, J.; Sun, Z. High-performance polyethylene separators for lithium-ion batteries modified by phenolic resin. J. Power. Sources. 2021, 483, 229155.
110. Lin, C.; Ou, S.; Liu, B.; et al. Enhanced electrochemical performance of zinc-ion batteries using functionalized nano-chitin separators. Adv. Compos. Hybrid. Mater. 2025, 8, 218.
111. Cheng, L.; Zhang, Y.; Li, D.; et al. Cu‐MOF functionalized hydroxyapatite/bacterial cellulose composite separator enabling dendrite-free fast charging and enhanced thermal safety for high-loading graphite anode. Adv. Funct. Mater. 2025, 36, e15523.
112. Ju, Y.; Kim, B.; Youn, B.; Song, Y.; Jang, H.; Lee, D. Towards dendrite-free lithium-ion batteries: a comprehensive review of functional separator technologies. J. Energy. Storage. 2025, 127, 116957.
113. Li, Y.; Wang, Y.; Chen, B.; et al. Proactive lithium dendrite regulation enabled by manipulating separator microstructure using high-fidelity phase-field simulation. Adv. Energy. Mater. 2025, 15, 2500503.
114. Yi, X.; Yang, Y.; Song, J.; et al. Strategically tailored polyethylene separator parameters enable cost-effective, facile, and scalable development of ultra-stable liquid and all-solid-state lithium batteries. Energy. Storage. Mater. 2025, 77, 104191.
115. Zhao, D. Y.; Zhou, S.; Huang, Y. F.; et al. Simulation of the cation solvation structure of the electrolyte in separator nanopores of a lithium ion battery. J. Phys. Chem. B. 2025, 129, 6721-30.
116. Wang, Y.; Feng, X.; Huang, W.; He, X.; Wang, L.; Ouyang, M. Challenges and opportunities to mitigate the catastrophic thermal runaway of high-energy batteries. Adv. Energy. Mater. 2023, 13, 2203841.
117. Feng, X.; Ouyang, M.; Liu, X.; Lu, L.; Xia, Y.; He, X. Thermal runaway mechanism of lithium ion battery for electric vehicles: a review. Energy. Storage. Mater. 2018, 10, 246-67.
118. Wu, Y.; Xie, L.; Ming, H.; et al. an empirical model for the design of batteries with high energy density. ACS. Energy. Lett. 2020, 5, 807-16.
119. Liu, X.; Ren, D.; Hsu, H.; et al. Thermal runaway of lithium-ion batteries without internal short circuit. Joule 2018, 2, 2047-64.
120. Liu, Y.; Tian, Y.; Fu, Z.; et al. Thermite reaction-induced thermal runaway of lithium-ion batteries. Adv. Mater. 2025, 37, e10486.
121. Chen, G.; Tan, R.; Zeng, C.; et al. Developing safe and high-performance lithium-ion batteries: strategies and approaches. Prog. Mater. Sci. 2025, 154, 101516.
122. Feng, X.; Ren, D.; He, X.; Ouyang, M. Mitigating thermal runaway of lithium-ion batteries. Joule 2020, 4, 743-70.
123. Feng, X.; Zhang, F.; Huang, W.; Peng, Y.; Xu, C.; Ouyang, M. Mechanism of internal thermal runaway propagation in blade batteries. J. Energy. Chem. 2024, 89, 184-94.
124. Ding, M.; Feng, X.; Peng, Y.; et al. Electrolyte induced synergistic construction of cathode electrolyte interphase and capture of reactive free radicals for safer high energy density lithium-ion battery. J. Energy. Chem. 2023, 87, 207-14.
125. Ren, D.; Feng, X.; Liu, L.; et al. Investigating the relationship between internal short circuit and thermal runaway of lithium-ion batteries under thermal abuse condition. Energy. Storage. Mater. 2021, 34, 563-73.
126. Liu, L.; Feng, X.; Rahe, C.; et al. Internal short circuit evaluation and corresponding failure mode analysis for lithium-ion batteries. J. Energy. Chem. 2021, 61, 269-80.
127. Lai, X.; Jin, C.; Yi, W.; et al. Mechanism, modeling, detection, and prevention of the internal short circuit in lithium-ion batteries: recent advances and perspectives. Energy. Storage. Mater. 2021, 35, 470-99.
128. Zhao, L.; Hou, J.; Feng, X.; et al. The trade-off characteristic between battery thermal runaway and combustion. Energy. Storage. Mater. 2024, 69, 103380.
129. Chen, S.; Wei, X.; Wu, H.; et al. Multi-functional thermal barrier suppresses battery thermal runaway propagation and degradation. Renew. Sust. Energ. Rev. 2025, 223, 116056.
130. Nayak, P. K.; Yang, L.; Brehm, W.; Adelhelm, P. From lithium-ion to sodium-ion batteries: advantages, challenges, and surprises. Angew. Chem. Int. Ed. Engl. 2018, 57, 102-20.
131. Dong, Y.; Zeng, Q.; Ding, L.; et al. Modulating Na plating morphology via interfacial design to achieve energy-dense and fast-charging sodium-ion batteries. Nano. Energy. 2025, 136, 110777.
132. Li, Y.; Yu, L.; Hu, W.; Hu, X. Thermotolerant separators for safe lithium-ion batteries under extreme conditions. J. Mater. Chem. A. 2020, 8, 20294-317.
133. Roh, Y.; Jin, D.; Kim, E.; et al. Highly improved thermal stability of the ceramic coating layer on the polyethylene separator via chemical crosslinking between ceramic particles and polymeric binders. Chem. Eng. J. 2022, 433, 134501.
134. Dai, J.; Shi, C.; Li, C.; et al. A rational design of separator with substantially enhanced thermal features for lithium-ion batteries by the polydopamine-ceramic composite modification of polyolefin membranes. Energy. Environ. Sci. 2016, 9, 3252-61.
135. Zou, Z.; Yin, M.; Yin, P.; Hu, Z.; Wang, D.; Pu, H. Facile preparation of surface-modified polypropylene nanofiber separators with enhanced ionic transport and welding performance for lithium-ion batteries. Nano. Energy. 2024, 127, 109774.
136. Zhang, M.; Wang, L.; Xu, H.; Song, Y.; He, X. Polyimides as promising materials for lithium-ion batteries: a review. Nanomicro. Lett. 2023, 15, 135.
137. Wang, X.; Lin, Z.; Fu, Y.; et al. High-temperature resistant semi-crystalline poly(ether ether ketone) separator. Energy. Storage. Materials. 2025, 82, 104642.
138. Li, M.; Yuan, J.; Wang, K.; et al. Advances in thermal stable separators and solid electrolytes for high-temperature lithium-ion batteries. Energy. Storage. Mater. 2025, 77, 104163.
139. Chang, H.; Kang, G.; Zhang, Z.; Liu, W.; Jin, Y. Evaluation of asymmetric poly(vinylidene fluoride)-coated polyimide separator with three-dimensionally homogeneous microporous structure for high-safety lithium-ion battery. Energy. Mater. 2024, 4, 400052.
140. Guo, D.; Mu, L.; Lin, F.; Liu, G. Mesoporous polyimide thin films as dendrite-suppressing separators for lithium-metal batteries. ACS. Nano. 2024, 18, 155-63.
141. Chen, Z.; Guan, M.; Cheng, Y.; et al. Boehmite-enhanced poly(vinylidene fluoride-co-hexafluoropropylene)/polyacrylonitrile (PVDF-HFP/PAN) coaxial electrospun nanofiber hybrid membrane: a superior separator for lithium-ion batteries. Adv. Compos. Hybrid. Mater. 2023, 6, 219.
142. Lai, X.; Meng, Z.; Zhang, F.; et al. Mitigating thermal runaway hazard of high-energy lithium-ion batteries by poison agent. J. Energy. Chem. 2023, 83, 3-15.
143. Finegan, D. P.; Darcy, E.; Keyser, M.; et al. Identifying the cause of rupture of li-ion batteries during thermal runaway. Adv. Sci. (Weinh). 2018, 5, 1700369.
144. Zeng, W.; Mei, W.; He, X.; et al. Insights into the coupled thermal behavior of silicon/graphite composite anodes in high energy density lithium-ion battery. Energy. Storage. Mater. 2026, 84, 104841.
145. He, T.; Gadkari, S.; Zhang, T.; et al. Investigation of the internal physical and chemical changes of a cylindrical lithium-ion battery during thermal runaway. J. Clean. Prod. 2024, 434, 140548.
146. Spotte-Smith, E. W. C.; Petrocelli, T. B.; Patel, H. D.; Blau, S. M.; Persson, K. A. Elementary decomposition mechanisms of lithium hexafluorophosphate in battery electrolytes and interphases. ACS. Energy. Lett. 2022, 8, 347-55.
147. Nam, S.; Lee, J.; Kim, Y.; et al. From HF scavenging to Li‐ion transport enhancement: multifunctional separator enabling stable Li metal batteries in carbonate-based electrolytes. Adv. Energy. Mater. 2025, 16, e05601.
148. Banerjee, A.; Ziv, B.; Shilina, Y.; Luski, S.; Aurbach, D.; Halalay, I. C. Acid-scavenging separators: a novel route for improving Li-ion batteries’ durability. ACS. Energy. Lett. 2017, 2, 2388-93.
149. Qiu, Z.; Yuan, S.; Wang, Z.; et al. Construction of silica-oxygen-borate hybrid networks on Al2O3-coated polyethylene separators realizing multifunction for high-performance lithium ion batteries. J. Power. Sources. 2020, 472, 228445.
150. Yang, Y.; Yang, T.; Liu, L.; et al. Green in situ synthesis of ZIF-8 membranes on the inner-surface of PESf hollow fibers and application in hydrogen separation. Green. Chem. 2025, 27, 5282-94.
151. Li, W.; Song, B.; Manthiram, A. High-voltage positive electrode materials for lithium-ion batteries. Chem. Soc. Rev. 2017, 46, 3006-59.
152. Cho, J.; Park, J.; Lee, M.; Song, H.; Lee, S. A polymer electrolyte-skinned active material strategy toward high-voltage lithium ion batteries: a polyimide-coated LiNi0.5Mn1.5O4 spinel cathode material case. Energy. Environ. Sci. 2012, 5, 7124.
153. Zhang, Z.; Said, S.; Lovett, A. J.; et al. The influence of cathode degradation products on the anode interface in lithium-ion batteries. ACS. Nano. 2024, 18, 9389-402.
154. Zhong, S.; Xie, H.; Han, Y.; et al. Engineering interfacial immunity in high-nickel batteries with a mediating biodegradable separator. Adv. Energy. Mater. 2025, 16, e05311.
155. An, Q.; Liu, Q.; Mao, P.; et al. Developing the tandem structure to regulate interfacial chemistry and promote ion transport kinetics toward high-voltage lithium metal batteries. Angew. Chem. Int. Ed. Engl. 2025, 64, e202422539.
156. Callegari, D.; Canini, M.; Davino, S.; Coduri, M.; Mustarelli, P.; Quartarone, E. Chitosan-decorated alumina hybrid nanoparticles as smart scavengers of HF and dissolved transition metals in lithium-ion batteries. Adv. Funct. Mater. 2024, 34, 2406315.
157. Lagadec, M. F.; Zahn, R.; Wood, V. Characterization and performance evaluation of lithium-ion battery separators. Nat. Energy. 2018, 4, 16-25.
158. Jo, S.; Seo, S.; Kang, S. K.; et al. Thermal runaway mechanism in Ni-rich cathode full cells of lithium-ion batteries: the role of multidirectional crosstalk. Adv. Mater. 2024, 36, e2402024.
159. Song, Y.; Wang, L.; Sheng, L.; et al. The significance of mitigating crosstalk in lithium-ion batteries: a review. Energy. Environ. Sci. 2023, 16, 1943-63.
160. Ghosh, A.; Tian, S.; Zhang, M.; et al. Deciphering a new electrolyte formulation for intelligent modulation of thermal runaway to improve the safety of lithium-ion batteries. Adv. Funct. Mater. 2025, 35, 2502761.
161. Liu, J.; Wang, Z.; Bai, J. Influences of multi factors on thermal runaway induced by overcharging of lithium-ion battery. J. Energy. Chem. 2022, 70, 531-41.
162. Wang, Z.; Yuan, J.; Zhu, X.; et al. Overcharge-to-thermal-runaway behavior and safety assessment of commercial lithium-ion cells with different cathode materials: a comparison study. J. Energy. Chem. 2021, 55, 484-98.
163. Wang, B.; Gao, L.; Yang, Z.; et al. Interphase engineering enabled by using a separator with electrochemically active carbazole polymers for lithium-ion batteries. Polymers. (Basel). 2025, 17.
164. Hou, J.; Feng, X.; Wang, L.; et al. Unlocking the self-supported thermal runaway of high-energy lithium-ion batteries. Energy. Storage. Mater. 2021, 39, 395-402.
165. Sharifi‐Asl, S.; Lu, J.; Amine, K.; Shahbazian‐Yassar, R. Oxygen release degradation in Li-ion battery cathode materials: mechanisms and mitigating approaches. Adv. Energy. Mater. 2019, 9, 1900551.
166. Jiang, Z.; Liu, C.; Huang, L.; et al. Interfacial reactions take the lead: elucidating the dominant role of cathode-electrolyte interactions in triggering thermal runaway of high-nickel lithium-ion batteries. Energy. Environ. Sci. 2025, 18, 8232-43.
167. Zhou, J.; Zhang, C.; Xie, C.; et al. A low-cost Al(OH)3‐modified fire-retardant and shuttle-limiting separator for safe and stable lithium-sulfur batteries. Adv. Energy. Mater. 2023, 14, 2303063.
168. Chen, X.; Yu, Z.; Cui, W.; et al. Surfactant-Enabled BM particle-embedded coaxial PVDF/PEI electrospun membranes enhancing lithium-ion battery safety. Chem. Eng. J. 2025, 503, 158225.
169. Zhang, X.; Sun, Q.; Zhen, C.; et al. Recent progress in flame-retardant separators for safe lithium-ion batteries. Energy. Storage. Mater. 2021, 37, 628-47.
170. Peng, L.; Kong, X.; Li, H.; et al. A rational design for a high-safety lithium-ion battery assembled with a heatproof-fireproof bifunctional separator. Adv. Funct. Mater. 2020, 31, 2008537.
171. Zhou, Z.; Chen, B.; Fang, T.; et al. A multifunctional separator enables safe and durable lithium/magnesium-sulfur batteries under elevated temperature. Adv. Energy. Mater. 2019, 10, 1902023.
172. Tong, B.; Li, X. Towards separator safety of lithium-ion batteries: a review. Mater. Chem. Front. 2024, 8, 309-40.
173. Sheng, Y.; Yue, X.; Hao, W.; Ding, L.; Zhu, J.; Liang, Z. Heterojunction-coated graphite anode enables fast charging via built-in electric field-regulated interfacial li-ion transport. ACS. Energy. Lett. 2025, 10, 5075-83.
174. Su, F.; Lin, Y.; Dou, X.; et al. Compact ion-pair aggregates dominated electrolytes enable high-performance low-temperature lithium-ion batteries. Angew. Chem. Int. Ed. Engl. 2025, 64, e202510647.
175. Yuan, S.; Cao, S.; Chen, X.; et al. Deshielding anions enable solvation chemistry control of LiPF6-based electrolyte toward low-temperature lithium-ion batteries. Adv. Mater. 2024, 36, e2311327.
176. Zhu, Z.; Ji, J.; Qi, X.; et al. Rational anion selection of the electrolyte additive for highly reversible lithium plating/stripping. Adv. Energy. Mater. 2023, 13, 2300936.
177. Wang, Z.; Wang, H.; Qi, S.; et al. Structural regulation chemistry of lithium ion solvation for lithium batteries. EcoMat 2022, 4, e12200.
178. Lu, D.; Zhang, S.; Li, J.; et al. Transformed solvation structure of noncoordinating flame-retardant assisted propylene carbonate enabling high voltage Li-ion batteries with high safety and long cyclability. Adv. Energy. Mater. 2023, 13, 2300684.
179. Zhang, Y.; Cheng, L.; Zhu, Y.; et al. Reversible Li plating regulation on graphite anode through a barium sulfate nanofibers-based dielectric separator for fast charging and high-safety lithium-ion battery. J. Energy. Chem. 2025, 101, 511-23.
180. Stuckenberg, S.; Bela, M. M.; Lechtenfeld, C. T.; et al. Influence of LiNO3 on the lithium metal deposition behavior in carbonate-based liquid electrolytes and on the electrochemical performance in zero-excess lithium metal batteries. Small 2024, 20, e2305203.
181. Jia, H.; Liu, J.; Liu, B.; et al. Lignin-based separators for lithium-ion batteries via a dry fibrillation method. Adv. Mater. 2025, 37, e2419694.
182. Sheng, L.; Zhang, Y.; Xie, X.; et al. Polyethylene separator activated by γ-ray irradiation for improving lithium-based battery performance. J. Mater. Sci. 2021, 56, 20026-36.
183. Fu, K.; Li, X.; Sun, K.; et al. Thick electrode design for lithium-ion batteries from an ion-electron coupled transport perspective: from independent regulation to cooperative design. Energy. Z. 2026, 2, 200002.
184. Niu, X.; Lu, Y.; Chen, P.; et al. Gradient-matched microstructural engineering for fast-charging, damage-tolerant thick electrodes of lithium-ion batteries. Adv. Energy. Mater. 2025, 15, 2502245.
185. Zhao, C. X.; Wang, Z.; Jacobson, D.; et al. Electrolytes that reduce electro-osmotic drag improve fast charging of lithium-ion batteries. Science 2025, 390, 745-50.
186. Na, Y.; Chen, C. Catechol-functionalized polyolefins. Angew. Chem. Int. Ed. Engl. 2020, 59, 7953-9.
187. Liu, J.; Wan, Y.; Wang, K.; et al. Constructing an enhanced charge-mass transfer passage for silicon anodes to achieve increased capacity under high-rate conditions. Energy. Mater. 2025, 5, 500094.
188. Nelson, A.; Mateti, S.; Chen, Y.; Sharma, N.; Han, Q.; Rahman, M. M. Creating value added nano silicon anodes from end-of-life photovoltaic modules: recovery, nano structuring, and the impact of ball milling and binder on its electrochemical performance. Energy. Mater. 2024, 4, 400046.
189. Seo, J. Y.; Kim, S.; Kim, J. H.; et al. Mechanical shutdown of battery separators: silicon anode failure. Nat. Commun. 2024, 15, 10134.
190. Liu, X.; Zhao, J.; Wang, J.; et al. Electrolyte-philic and thermal-resistant polyimide separator enhances the performance of flexible silicon/carbon nanofibers for lithium-ion batteries. J. Energy. Storage. 2022, 54, 105324.
191. Hwang, J. Y.; Myung, S. T.; Sun, Y. K. Sodium-ion batteries: present and future. Chem. Soc. Rev. 2017, 46, 3529-614.
192. Xue, C.; Zhang, Y.; Zhang, Y.; et al. Fe single-atom-regulated carbon nanofibers for high-performance lithium/sodium ion battery anode. Energy. Mater. 2025, 5, 500113.
193. Recent commentaries on the expected performance, advantages and applications of sodium-ion batteries. Energy. Mater. 2023.
194. Li, M.; Zhuo, H.; Jing, Q.; et al. Low‐temperature performance of Na‐ion batteries. Carbon. Energy. 2024, 6, e546.
195. Zhao, Y.; Wang, L. P.; Sougrati, M. T.; et al. A review on design strategies for carbon based metal oxides and sulfides nanocomposites for high performance Li and Na ion battery anodes. Adv. Energy. Mater. 2016, 7, 1601424.
196. Chen, J.; Adit, G.; Li, L.; Zhang, Y.; Chua, D. H. C.; Lee, P. S. Optimization strategies toward functional sodium‐ion batteries. Energ. Environ. Mater. 2023, 6, e12633.
197. Hu, J.; Xu, L.; Li, X.; et al. Pre-sodiation strategies for constructing high-performance sodium-ion batteries. J. Mater. Chem. A. 2025, 13, 3206-35.
198. Zhang, L.; Li, X.; Yang, M.; Chen, W. High-safety separators for lithium-ion batteries and sodium-ion batteries: advances and perspective. Energy. Storage. Mater. 2021, 41, 522-45.
199. Li, C.; Du, L.; Ju, J.; et al. Assessment of safety concerns in sodium-ion batteries. Adv. Energy. Mater. 2025, 15, e03209.
200. Niu, J.; Dong, J.; Zhang, X.; et al. Sodium cluster-driven safety concerns of sodium-ion batteries. Energy. Environ. Sci. 2025, 18, 2474-84.
201. Aniskevich, Y.; Yu, J. H.; Kim, J. Y.; Komaba, S.; Myung, S. T. Tracking sodium cluster dynamics in hard carbon with a low specific surface area for sodium-ion batteries. Adv. Energy. Mater. 2024, 14, 2304300.
202. Wang, Z.; Feng, X.; Bai, Y.; et al. Probing the energy storage mechanism of quasi-metallic Na in hard carbon for sodium-ion batteries. Adv. Energy. Mater. 2021, 11, 2003854.
203. Zeng, Y.; Yang, J.; Yang, H.; Yang, Y.; Zhao, J. Bridging microstructure and sodium-ion storage mechanism in hard carbon for sodium ion batteries. ACS. Energy. Lett. 2024, 9, 1184-91.
204. Zhou, H.; Song, Y.; Zhang, B.; et al. Overview of electrochemical competing process of sodium storage and metal plating in hard carbon anode of sodium ion battery. Energy. Storage. Mater. 2024, 71, 103645.
205. Du, L.; Xu, G.; Zhang, Z.; et al. Multifunctional polyimide separator enabling the long-life operation of sodium ion batteries at elevated temperatures. ACS. Energy. Lett. 2025, 10, 3941-51.
206. Li, X.; Zhang, T.; Zhao, Y.; et al. Enhancing robustness and charge transfer kinetics of sodium-ion batteries through introduction of anionic anchoring separators. J. Am. Chem. Soc. 2025, 147, 8488-99.
207. Wang, J.; Wang, Z.; Ni, J.; Li, L. Electrospinning for flexible sodium-ion batteries. Energy. Storage. Mater. 2022, 45, 704-19.
208. Yang, J. L.; Zhao, X. X.; Zhang, W.; et al. “Pore-hopping” ion transport in cellulose-based separator towards high-performance sodium-ion batteries. Angew. Chem. Int. Ed. Engl. 2023, 62, e202300258.
209. Yang, J. L.; Zhao, X. X.; Liu, H. H.; et al. Nature-inspired separator with thermal sealing reinforcement toward sustainable sodium-ion batteries. ACS. Nano. 2025, 19, 15983-93.
210. Tang, W.; Ma, J.; Zhang, X.; et al. Interfacial strategies towards highly stable Li-metal anode of liquid-based Li-metal batteries. Energy. Storage. Mater. 2024, 64, 103084.
211. Hu, Y.; Chen, Z.; Wang, Y.; et al. Review of thin lithium metal battery anode fabrication - microstructure - electrochemistry relations. Adv. Mater. 2026, 38, e11817.
212. Dong, Y.; Liu, C.; Rui, M.; et al. Review on graphite anodes for fast-charging lithium-ion batteries: mechanism, modification and characterizations. Adv. Funct. Mater. 2025, 35, 2506190.
213. Song, B.; Luo, Z.; Liu, X.; et al. Bridging donor ligands enable an ultrastable graphite anode for sodium-ion batteries. Angew. Chem. Int. Ed. Engl. 2025, 64, e202503027.
214. Ko, S.; Obukata, T.; Shimada, T.; et al. Electrode potential influences the reversibility of lithium-metal anodes. Nat. Energy. 2022, 7, 1217-24.
215. He, X.; Zheng, S.; Sun, K.; et al. Operando electrode-scale stress characterization revealing the Li+ insertion mechanism of graphite anode. Energy. Storage. Mater. 2025, 82, 104629.
216. Wang, C.; Ouyang, T.; Wang, X.; et al. Accelerating lithium ion transport via increasing the entropy of the electrolyte for stable lithium metal batteries. J. Energy. Chem. 2024, 99, 384-92.
217. Zhao, Z.; Nian, B.; Lei, Y.; et al. Passivation layers in Mg-metal batteries: robust interphases for Li-metal batteries. Adv. Mater. 2024, 36, e2402626.
218. Lee, G.; Han, D. Y.; Park, S.; et al. Regulating polymer demixing dynamics to construct a low-tortuosity host for stable high-energy-density lithium metal batteries. Adv. Mater. 2026, 38, e10919.
219. You, J.; Lu, J.; Liu, C.; et al. A rationally thin composite membrane with differentiated pore structure for industrial-scale alkaline water electrolysis. Nat. Commun. 2025, 16, 5981.
220. Liao, C.; Zheng, T.; Zhu, J.; et al. Engineering durable nitrate-enriched solvation sheaths in carbonate electrolytes through functional separator design for high-voltage lithium metal batteries. Nano. Energy. 2025, 142, 111283.
221. Cao, S.; He, X.; Chen, M.; et al. A CF4 plasma functionalized polypropylene separator for dendrite-free lithium metal anodes. J. Mater. Chem. A. 2023, 11, 7545-55.
222. Huang, Z.; Lyu, H.; Greenburg, L. C.; Cui, Y.; Bao, Z. Stabilizing lithium-metal electrodes with polymer coatings. Nat. Energy. 2025, 10, 811-23.
223. Wang, K.; Zhao, T.; Lv, R.; et al. Lithium metal batteries enabled by ion flux-regulating coating on separator. Adv. Funct. Mater. 2025, 35, e08164.
224. Jin, H.; Pyo, S.; Seo, H.; et al. LiF-rich solid electrolyte interphase formation by establishing sacrificial layer on the separator. Small 2024, 20, e2401928.
225. Chang, Z.; Yang, H.; Pan, A.; He, P.; Zhou, H. An improved 9 micron thick separator for a 350 Wh/kg lithium metal rechargeable pouch cell. Nat. Commun. 2022, 13, 6788.
226. Chen, J.; Tang, Z.; Zhu, D.; et al. Stabilizing Li-metal electrode via anion-induced desolvation in a covalent organic framework separator. ACS. Nano. 2025, 19, 9232-40.
227. Liu, Y.; Hou, T.; Zhang, W.; et al. Anion‐repulsive polyoxometalate@MOF‐modified separators for dendrite‐free and high‐rate lithium batteries. Interd. Mater. 2024, 4, 190-200.
228. Du, M.; He, Z.; Zhang, Y.; Cai, Y. P.; Zheng, Q. Sustainable release of LiNO3 from a fluorine-decorated metal-organic framework separator to enable high-performance Li‐metal batteries in carbonate electrolytes. Adv. Energy. Mater. 2025, 15, 2403674.
229. Han, D.; Wang, X.; Zhou, Y. N.; et al. A graphene-coated thermal conductive separator to eliminate the dendrite-induced local hotspots for stable lithium cycling. Adv. Energy. Mater. 2022, 12, 2201190.
230. Park, B. K.; Nam, D. W.; Ma, D.; et al. Strategic design of a functional separator with dual-acting Si/SiOx nanocomposite for dendrite-free Li-metal batteries. ACS. Nano. 2025, 19, 26659-72.
231. Wang, Y.; Zhang, Y.; Zhang, Y.; et al. Dual protection of Li-metal anode by black phosphorus-modified separator via modulating solid electrolyte interphase and eliminating Li dendrites. Small. Methods. 2025, 9, e2500201.
232. Feng, S.; Fu, Z. H.; Chen, X.; Zhang, Q. A review on theoretical models for lithium-sulfur battery cathodes. InfoMat 2022, 4, e12304.
233. Li, S.; Fan, Z. Encapsulation methods of sulfur particles for lithium-sulfur batteries: a review. Energy. Storage. Mater. 2021, 34, 107-27.
234. Wang, J.; Han, W. Q. A review of heteroatom doped materials for advanced lithium-sulfur batteries. Adv. Funct. Mater. 2021, 32, 2107166.
235. Li, M.; Wang, J.; Cao, G.; et al. Optimization of single-atom catalysts for sulfur cathode in lithium-sulfur batteries: a review. Nano. Energy. 2025, 143, 111330.
236. Zhu, X.; Iqbal, S.; Kumar, N.; et al. The catalytic chemistry for high-performance lithium-sulfur batteries: a review and prospects. Adv. Funct. Mater. 2025, 35, e11659.
237. Chen, M.; Shao, M.; Jin, J.; Cui, L.; Tu, H.; Fu, X. Configurational and structural design of separators toward shuttling-free and dendrite-free lithium-sulfur batteries: a review. Energy. Storage. Mater. 2022, 47, 629-48.
238. Rosenman, A.; Markevich, E.; Salitra, G.; Aurbach, D.; Garsuch, A.; Chesneau, F. F. Review on Li-sulfur battery systems: an integral perspective. Adv. Energy. Mater. 2015, 5, 1500212.
239. Yan, Y.; Zhou, X.; He, J.; et al. Self-supporting metal-organic frameworks (MOFs) and their derivatives for lithium-sulfur batteries: a comprehensive review. Coord. Chem. Rev. 2026, 548, 217218.
240. Fu, X.; Wang, Y.; Scudiero, L.; Zhong, W. A polymeric nanocomposite interlayer as ion-transport-regulator for trapping polysulfides and stabilizing lithium metal. Energy. Storage. Mater. 2018, 15, 447-57.
241. Hou, L.; Zhang, X.; Li, B.; Zhang, Q. Challenges and promises of lithium metal anode by soluble polysulfides in practical lithium-sulfur batteries. Mater. Today. 2021, 45, 62-76.
242. Su, Y. S.; Manthiram, A. Lithium-sulphur batteries with a microporous carbon paper as a bifunctional interlayer. Nat. Commun. 2012, 3, 1166.
243. Wang, X.; Zou, N.; Qiao, Y.; et al. Spatially structured lamellar porous Carbon-CoS2 separator for enhanced adsorption and conversion of lithium polysulfides in high-performance Li-S batteries. Chem. Eng. J. 2025, 515, 163529.
244. Wang, L.; Yue, K.; Qiao, Q.; et al. In situ self-polymerization of thioctic acid enabled interphase engineering towards high-performance lithium-sulfur battery. Adv. Energy. Mater. 2024, 15, 2402617.
245. Zhou, X.; Li, X.; Li, Z.; et al. Ten micrometer thick polyethylene separator modified by α-LiAlO2@γ-Al2O3 nanosheets for simultaneous suppression of Li dendrite growth and polysulfide shuttling in Li-S batteries. Mater. Today. Energy. 2022, 26, 100990.
246. Ko, I.; Ha, S.; Shin, W. H.; et al. Clay-mineral-coated separators for lithium-ion batteries: exploring the relationships between clay mineral morphology and separator performance. Adv. Sci. (Weinh). 2025, 12, e10779.
247. Hou, T. Z.; Chen, X.; Peng, H. J.; et al. Design principles for heteroatom-doped nanocarbon to achieve strong anchoring of polysulfides for lithium-sulfur batteries. Small 2016, 12, 3283-91.
248. Li, Q.; Song, Y.; Xu, R.; et al. Biotemplating growth of nepenthes-like N-doped graphene as a bifunctional polysulfide scavenger for Li-S batteries. ACS. Nano. 2018, 12, 10240-50.
249. Pei, F.; Lin, L.; Fu, A.; et al. A two-dimensional porous carbon-modified separator for high-energy-density Li-S batteries. Joule 2018, 2, 323-36.
250. Wang, L.; Yang, Z.; Nie, H.; et al. A lightweight multifunctional interlayer of sulfur-nitrogen dual-doped graphene for ultrafast, long-life lithium-sulfur batteries. J. Mater. Chem. A. 2016, 4, 15343-52.
251. Zeng, P.; Huang, L.; Zhang, X.; Zhang, R.; Wu, L.; Chen, Y. Long-life and high-areal-capacity lithium-sulfur batteries realized by a honeycomb-like N, P dual-doped carbon modified separator. Chem. Eng. J. 2018, 349, 327-37.
252. Ponraj, R.; Kannan, A. G.; Ahn, J. H.; et al. Effective trapping of lithium polysulfides using a functionalized carbon nanotube-coated separator for lithium-sulfur cells with enhanced cycling stability. ACS. Appl. Mater. Interfaces. 2017, 9, 38445-54.
253. Tu, S.; Chen, X.; Zhao, X.; et al. A polysulfide-immobilizing polymer retards the shuttling of polysulfide intermediates in lithium-sulfur batteries. Adv. Mater. 2018, 30, e1804581.
254. Qiao, X.; Wang, C.; Zang, J.; et al. Conductive inks composed of multicomponent carbon nanomaterials and hydrophilic polymer binders for high-energy-density lithium-sulfur batteries. Energy. Storage. Materials. 2022, 49, 236-45.
255. Luo, L.; Chung, S.; Manthiram, A. A trifunctional multi-walled carbon nanotubes/polyethylene glycol (MWCNT/PEG)-coated separator through a layer-by-layer coating strategy for high-energy Li-S batteries. J. Mater. Chem. A. 2016, 4, 16805-11.
256. Chung, S. H.; Manthiram, A. A polyethylene glycol-supported microporous carbon coating as a polysulfide trap for utilizing pure sulfur cathodes in lithium-sulfur batteries. Adv. Mater. 2014, 26, 7352-7.
257. Shaibani, M.; Akbari, A.; Sheath, P.; et al. Suppressed polysulfide crossover in Li-S batteries through a high-flux graphene oxide membrane supported on a sulfur cathode. ACS. Nano. 2016, 10, 7768-79.
258. Kong, W.; Yan, L.; Luo, Y.; et al. Ultrathin MnO2/graphene oxide/carbon nanotube interlayer as efficient polysulfide-trapping shield for high-performance Li-S batteries. Adv. Funct. Mater. 2017, 27, 1606663.
259. Fan, B.; Chen, W.; Li, K.; et al. Synergistic adsorption and catalytic effects of Ti3C2Tx/CoO/MoO3 composite on lithium polysulfides for high‐performance lithium–sulfur batteries. Interd. Mater. 2024, 3, 726-37.
260. Gu, H.; Yue, W.; Hu, J.; et al. Asymmetrically coordinated Cu-N1C2 single-atom catalyst immobilized on Ti3C2Tx MXene as separator coating for lithium-sulfur batteries. Adv. Energy. Mater. 2023, 13, 2204014.
261. Kang, X.; He, T.; Dang, H.; et al. Designing amino functionalized titanium-organic framework on separators toward sieving and redistribution of polysulfides in lithium-sulfur batteries. Nanomicro. Lett. 2025, 17, 277.
262. Wang, T.; He, J.; Zhu, Z.; et al. Heterostructures regulating lithium polysulfides for advanced lithium-sulfur batteries. Adv. Mater. 2023, 35, e2303520.
263. Garapati, M. S. V.s., A. P.; Sundara, R. Synergy between partially exfoliated carbon nanotubes-sulfur cathode and nitrogen rich dual function interlayer for high performance lithium sulfur battery. Carbon 2019, 147, 364-76.
264. Deng, D. R.; Bai, C. D.; Xue, F.; et al. Multifunctional ion-sieve constructed by 2D materials as an interlayer for Li-S batteries. ACS. Appl. Mater. Interfaces. 2019, 11, 11474-80.
265. Zong, Y.; He, H.; Wang, Y.; et al. Functionalized separator strategies toward advanced aqueous zinc-ion batteries. Adv. Energy. Mater. 2023, 13, 2300403.
266. Chen, S.; Wang, Y.; Sun, Y.; et al. Research status and prospect of separators for magnesium-sulfur batteries. J. Energy. Chem. 2023, 87, 225-46.
267. Fang, Y.; Xie, X.; Zhang, B.; et al. Regulating zinc deposition behaviors by the conditioner of PAN separator for zinc-ion batteries. Adv. Funct. Mater. 2021, 32, 2109671.
268. Wang, T.; Xiao, Y.; Xiang, W.; Tang, S.; Yu, J. S. Stable zinc electrode/separator interface enabled by phthalocyanine-modified separator for advanced zinc metal batteries. Small 2025, 21, e2503907.
269. Yang, Z. The size and structure of selected hydrated ions and implications for ion channel selectivity. RSC. Adv. 2015, 5, 1213-9.
270. Wan, H.; Xu, J.; Wang, C. Designing electrolytes and interphases for high-energy lithium batteries. Nat. Rev. Chem. 2024, 8, 30-44.
271. Tian, Y.; Zeng, G.; Rutt, A.; et al. Promises and challenges of next-generation “beyond Li-ion” batteries for electric vehicles and grid decarbonization. Chem. Rev. 2021, 121, 1623-69.
272. Lee, Y.; Thirumalai, D.; Hyeon, C. Ultrasensitivity of water exchange kinetics to the size of metal ion. J. Am. Chem. Soc. 2017, 139, 12334-7.
273. Kazemzadeh, H.; Karimi-sabet, J.; Towfighi Darian, J.; Adhami, A. Evaluation of polymer inclusion membrane efficiency in selective separation of lithium ion from aqueous solution. Sep. Purif. Technol. 2020, 251, 117298.
274. Sun, D. F.; Wang, Z. J.; Tian, T.; et al. Constructing oxygen deficiency‐rich V2O3@PEDOT cathode for high‐performance aqueous zinc‐ion batteries. Rare. Metals. 2023, 43, 635-46.
275. Wang, Y. Y.; Song, X.; Liu, S.; Li, G. R.; Ye, S. H.; Gao, X. P. Elucidating the effect of the dopant ionic radius on the structure and electrochemical performance of Ni-rich layered oxides for lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2021, 13, 56233-41.
276. Yu, T.; Kim, J.; Hwang, J.; et al. High-energy O3-Na1-2xCax[Ni0.5Mn0.5]O2 cathodes for long-life sodium-ion batteries. J. Mater. Chem. A. 2020, 8, 13776-86.
277. Lee, Y.; Kim, H.; Son, H.; Kim, M.; Lee, G. Engineering of protective surfaces for retarding selenium leaching by self-growth of rGO@Ni Se on carbon nanofibers for potassium ion batteries. J. Alloys. Compd. 2024, 1000, 175071.
278. Gu, W.; Wu, K.; Huang, J.; et al. The role and mechanism of separators in aqueous zinc metal batteries: a critical review. Adv. Energy. Mater. 2025, 15, 2502652.
279. Li, B.; Zeng, Y.; Zhang, W.; et al. Separator designs for aqueous zinc-ion batteries. Sci. Bull. (Beijing). 2024, 69, 688-703.
280. Cao, J.; Rao, X.; Qian, S.; et al. Dynamic Zn2+‐coordinating oxygen sites and electric field modulation in boron-integrated cellulose nanofiber separators for stable zinc‐ion batteries. Adv. Energy. Mater. 2025, 15, e03368.
281. Sui, B. B.; Sha, L.; Wang, P. F.; et al. Salt solution etching to construct micro-gullies on the surface of Zn anodes enhances anodes performance in aqueous zinc-ion batteries. J. Colloid. Interface. Sci. 2024, 653, 159-69.
282. Su, Y.; Liu, B.; Zhang, Q.; et al. Printing‐Scalable Ti3C2Tx MXene‐decorated Janus separator with expedited Zn2+ flux toward stabilized Zn anodes. Adv. Funct. Mater. 2022, 32, 2204306.
283. Wu, F.; Du, F.; Ruan, P.; et al. Regulating zinc deposition behaviors by using a functional PANI modification layer on a separator for high performance aqueous zinc-ion batteries. J. Mater. Chem. A. 2023, 11, 11254-63.
284. Xue, X.; Huang, T.; Zhang, Y.; et al. Synergy of electrolyte manipulation and separator functionalization enables ultralong-life nonaqueous magnesium-organic batteries. J. Mater. Chem. A. 2024, 12, 24955-65.
285. Song, Y.; Ruan, P.; Mao, C.; et al. Metal-organic frameworks functionalized separators for robust aqueous zinc-ion batteries. Nanomicro. Lett. 2022, 14, 218.
286. Zhang, Y.; Zeng, Z.; Yang, S.; Zhang, Y.; Ma, Y.; Wang, Z. A nanocellulose-mediated, multiscale ion-sieving separator with selective Zn2+ channels for durable aqueous zinc-based batteries. Energy. Storage. Mater. 2023, 57, 557-67.
287. Zhang, Y.; Li, J.; Zhao, W.; et al. Defect-free metal-organic framework membrane for precise ion/solvent separation toward highly stable magnesium metal anode. Adv. Mater. 2022, 34, e2108114.
288. Zhou, W.; Yang, M.; Chen, M.; et al. Ion-sieving effect enabled by sulfonation of cellulose separator realizing dendrite-free Zn deposition. Adv. Funct. Mater. 2024, 34, 2315444.
289. Li, R.; Sui, B.; Wang, P.; et al. Modulation of zinc deposition behaviour by electrostatically spun PAN-ZnTFSI fibre layer on separator for aqueous zinc ion batteries. J. Alloys. Compd. 2024, 997, 174899.
290. Wang, F.; Hua, H.; Wu, D.; et al. Solvent molecule design enables excellent charge transfer kinetics for a magnesium metal anode. ACS. Energy. Lett. 2022, 8, 780-9.
291. Ren, W.; Ng, M. S.; Zhang, Y.; et al. Fluorine-free ion-selective membrane with enhanced Mg2+ Transport for Mg-organic batteries. ACS. Nano. 2025, 19, 5781-8.
292. Yan, L.; Zhang, Q.; Zhang, Z.; et al. Hydrogel electrolyte membrane with regulated pore effect to stabilize zinc anode in aqueous zinc-ion batteries. J. Membr. Sci. 2024, 690, 122243.
293. Mao, Y.; Liu, S.; Liu, J.; et al. Ultrathin polydopamine/BaTiO3 modified PTFE separator with synergistic hydrophilic and electric field regulation toward stable zinc deposition. ACS. Appl. Polym. Mater. 2025, 7, 14281-91.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data



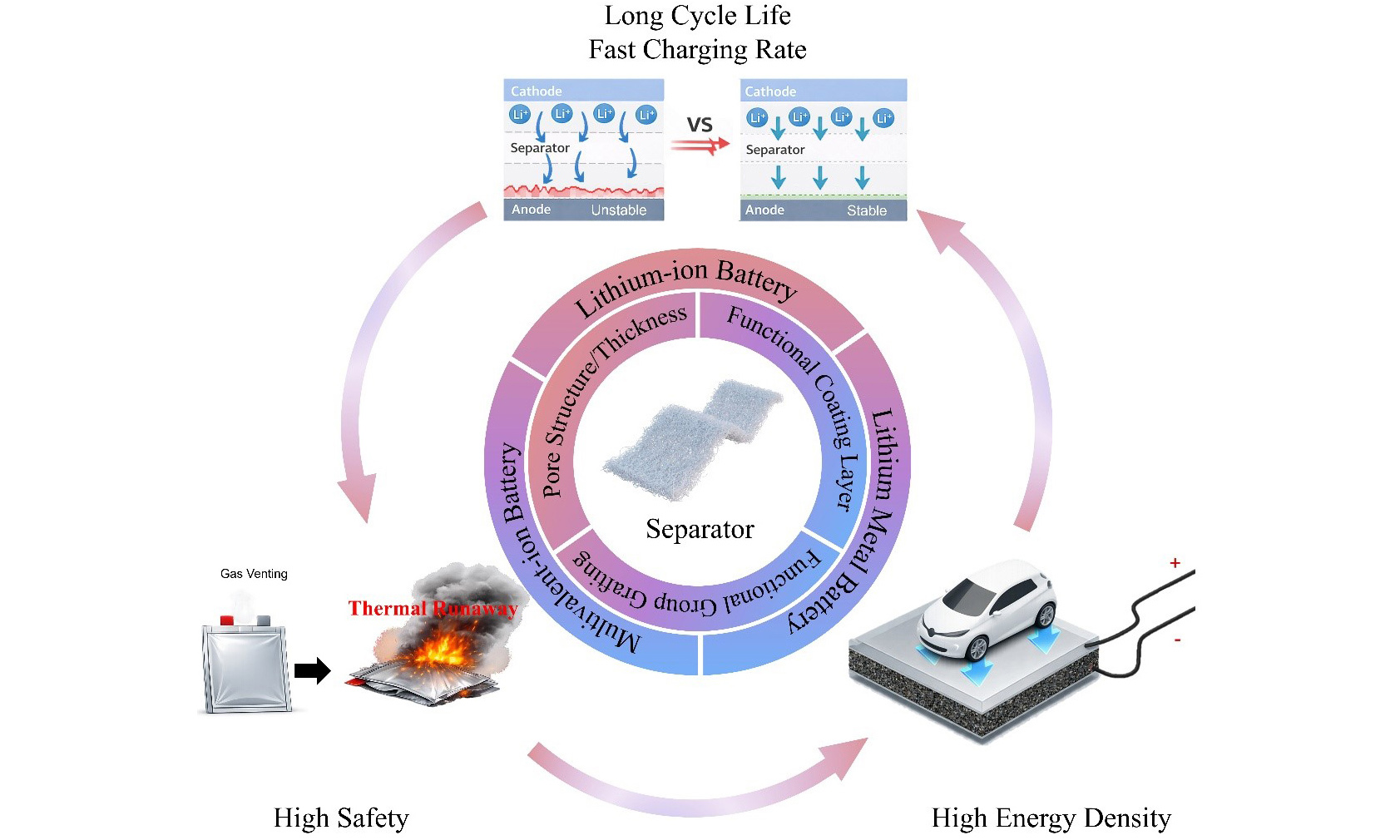
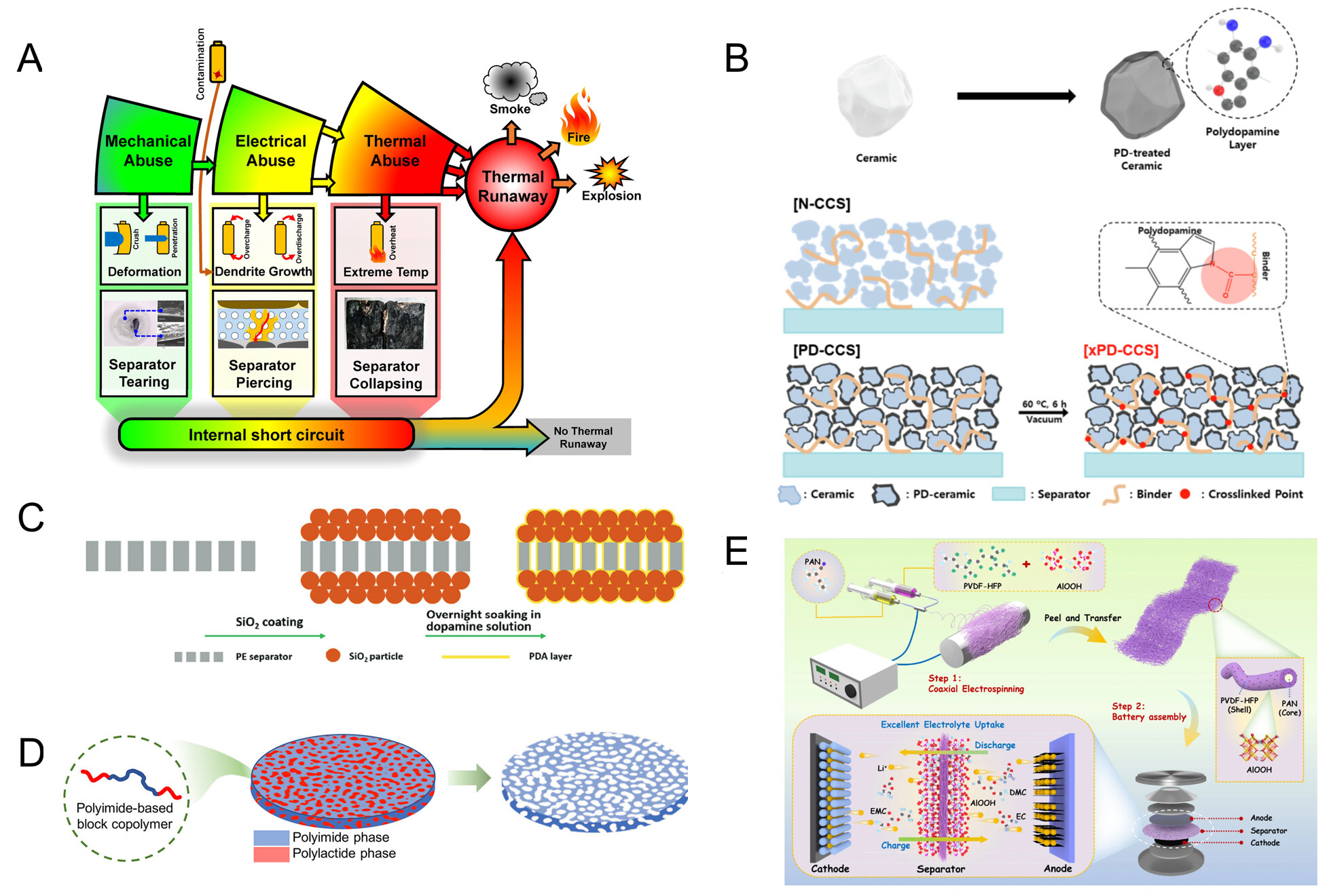
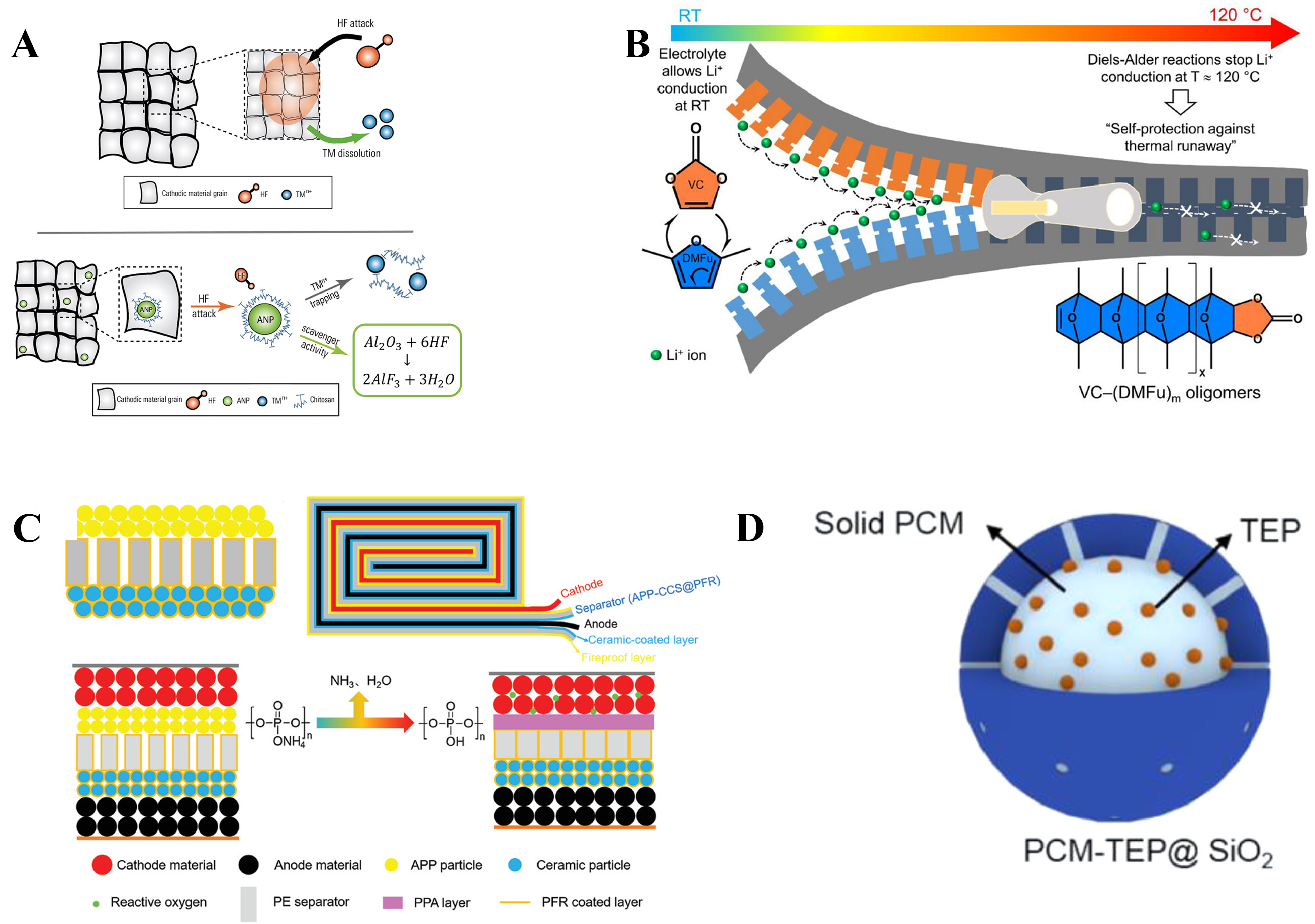
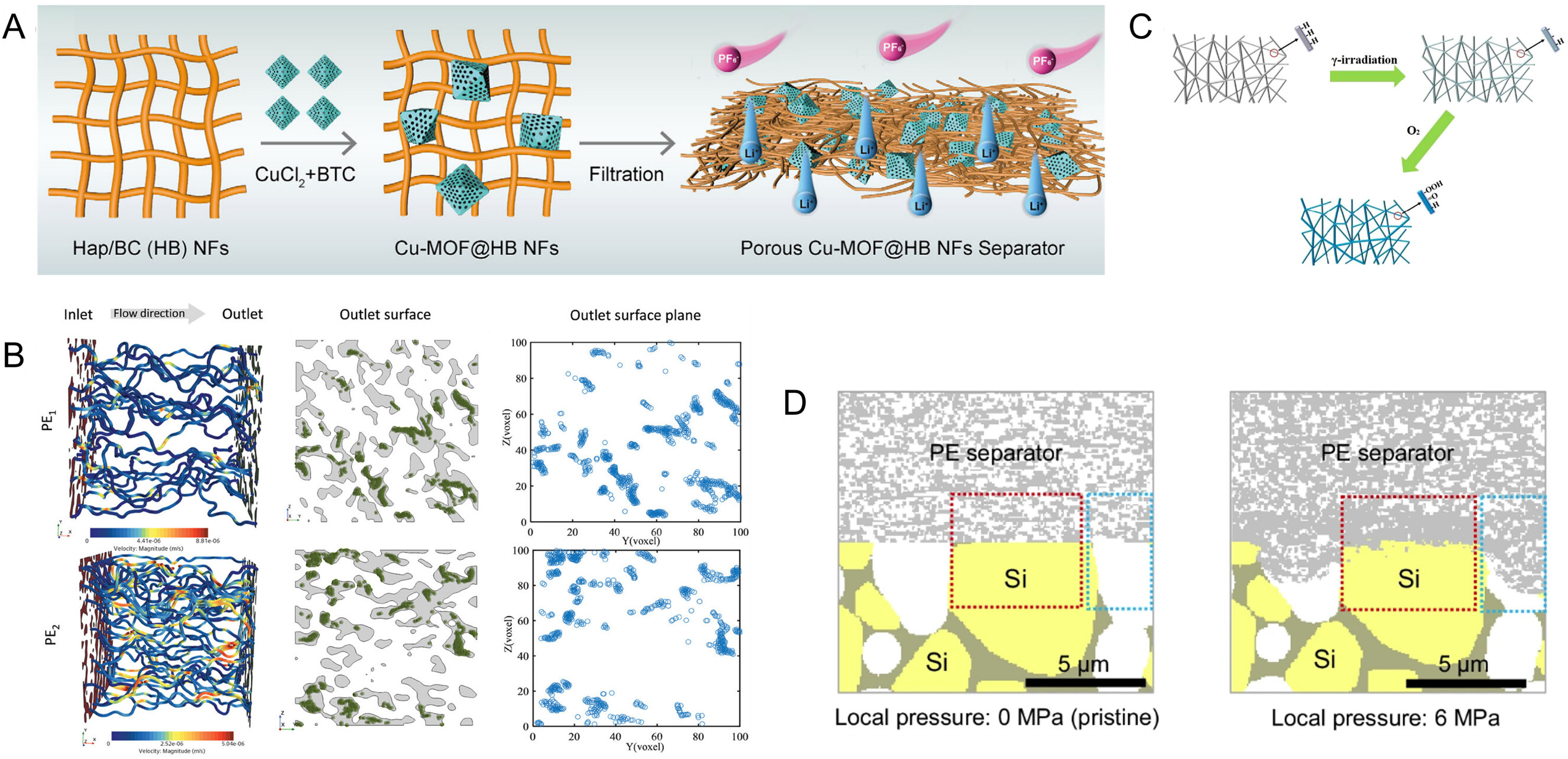
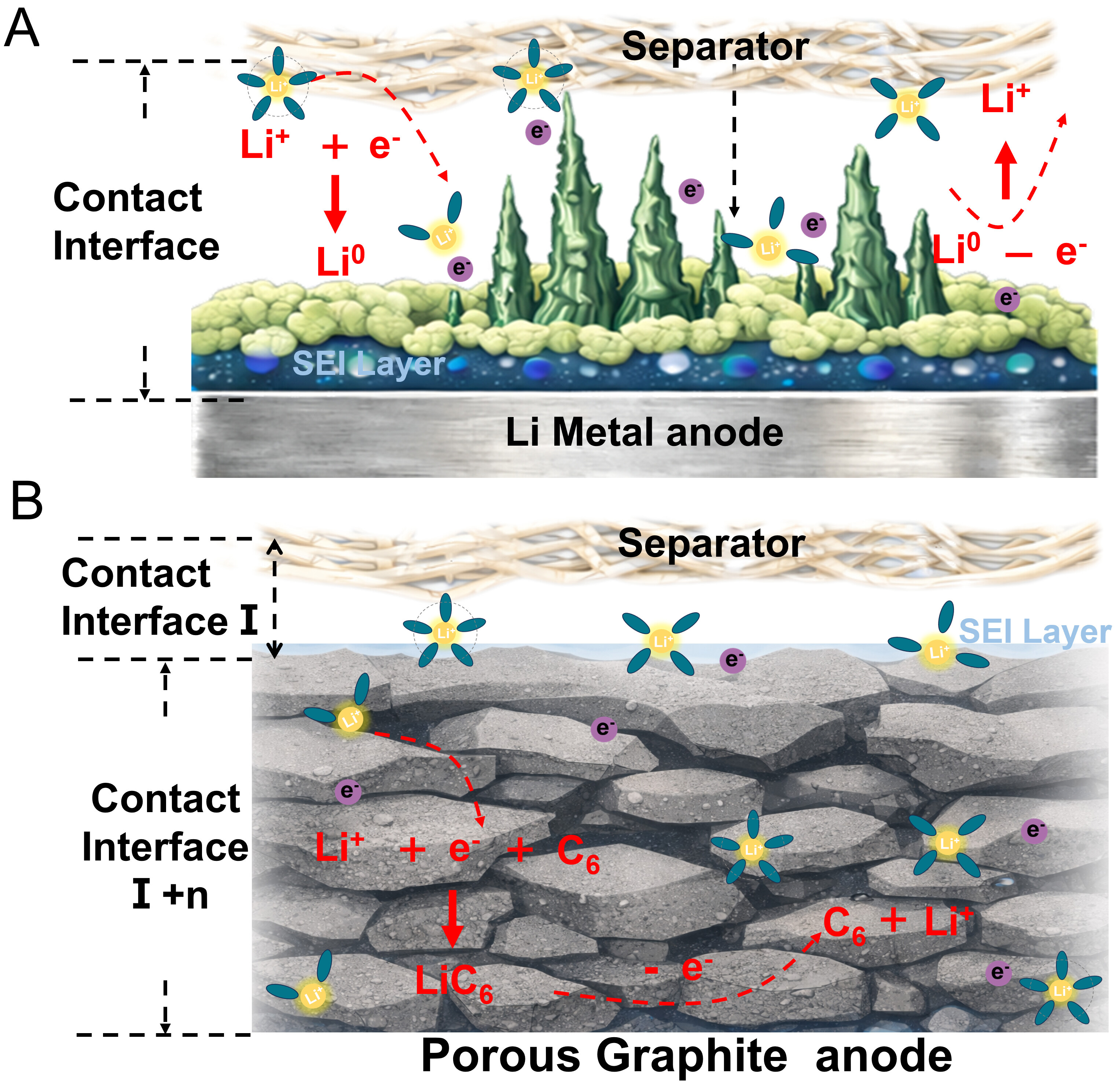
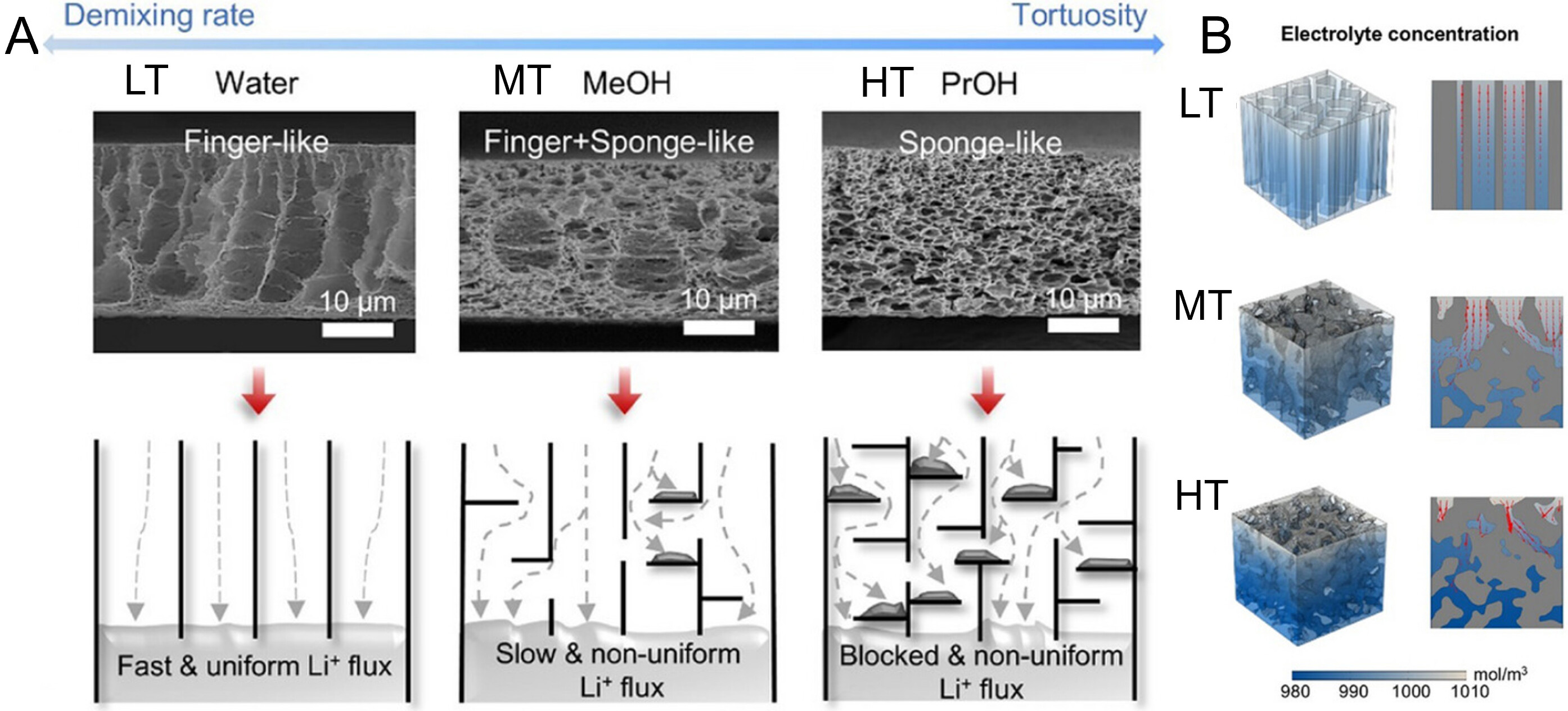
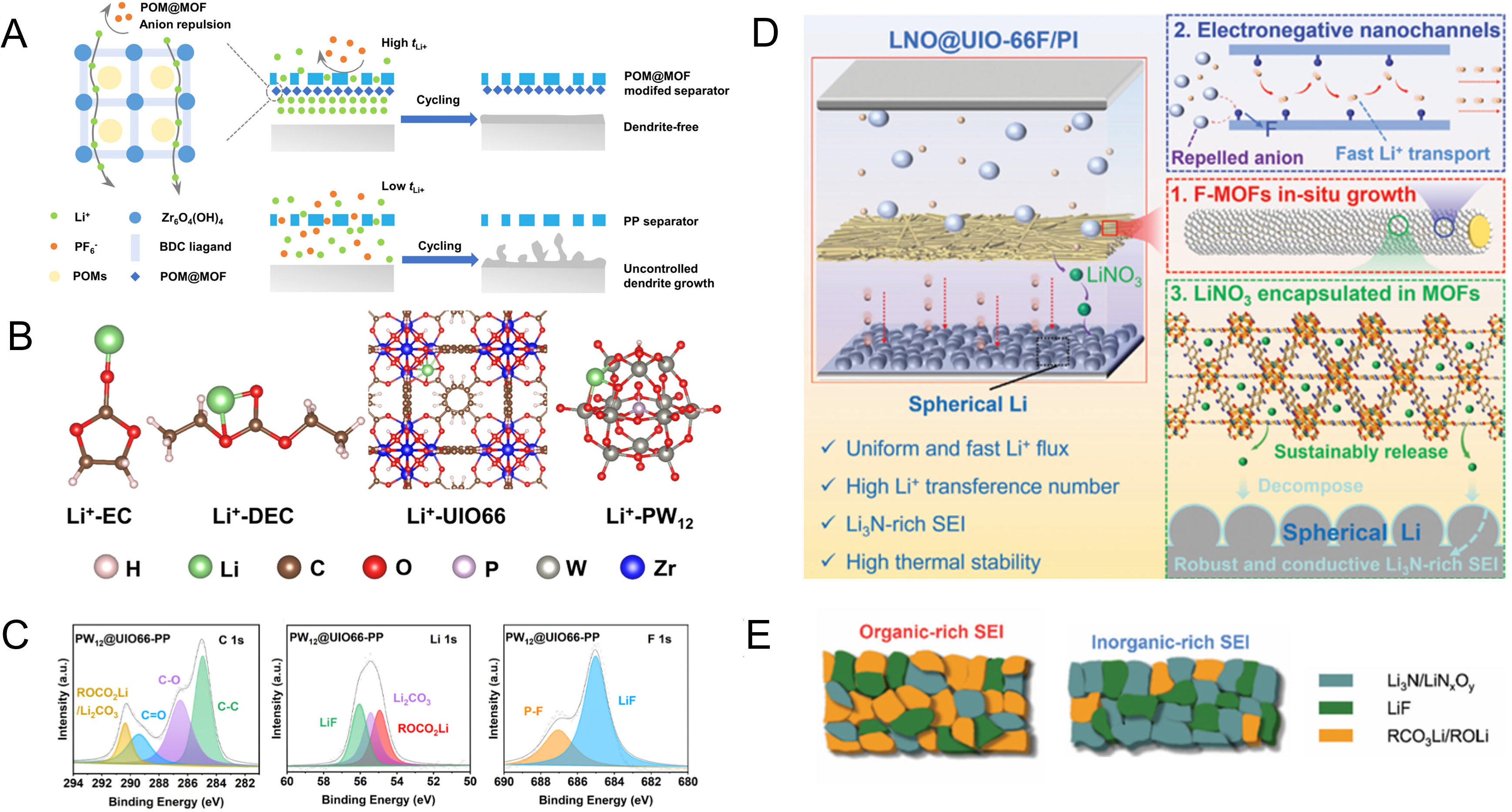








Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].