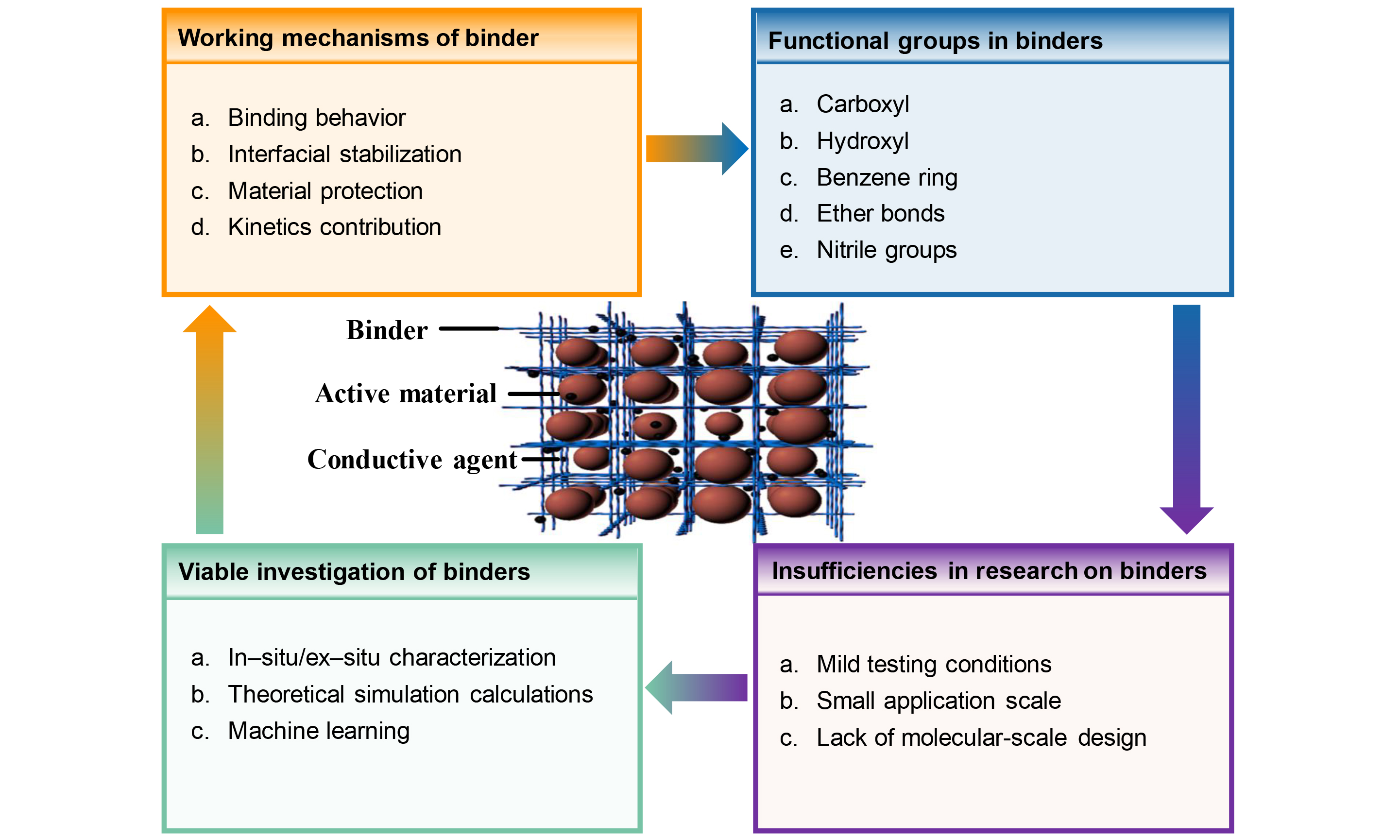
Overview of the functional binders in high-capacity cathodes of lithium-ion battery
Abstract
High-capacity cathode materials for lithium-ion batteries (LIBs), typically represented by nickel (Ni)-rich and lithium (Li)-rich layered oxides, have been regarded as promising candidates for high-performance LIBs owing to their potential to achieve energy densities of up to 400 Wh kg-1. However, challenges such as interfacial instability, bulk structural degradation, dissolution of transition-metal ions, and undesirable side reactions occur during cycling, necessitating strategies to enhance their electrochemical performance. A LIB generally consists of a cathode, anode, electrolyte, separator, and various inactive components, including binder, current collector, and conductive agents. Although the binder constitutes a small portion of the electrode system, it plays a pivotal role in determining the electrode’s electrochemical performance. The utilization of functional binders has proven to be an effective strategy for mitigating the aforementioned challenges associated with Ni- and Li-rich layered oxides. This review provides a comprehensive analysis of emerging binders for Ni- and Li-rich layered oxides, highlighting the effects of functional groups and discussing noteworthy research directions in cathode binder development.
Keywords
INTRODUCTION
The demand for lithium-ion batteries (LIBs) with high energy densities has significantly increased, primarily driven by the growing need for extended driving ranges in electric vehicles[1-3]. Developing high-capacity cathode materials (> 200 mAh g-1) is an effective strategy to break through the energy-density bottleneck of current LIBs[4-7]. Consequently, considerable attention has been directed toward Ni- and Li-rich layered oxides, known for their high capacity and operational voltage[8-10]. However, their intrinsic defects, including interfacial instability, structural degradation, transition metal (TM) ion dissolution, and harmful side reactions, have led to inadequate cycling life[11]. Various strategies, such as controlling nanoparticle granularity, metal doping, and surface coating, have been proposed to modify the cathode materials[12-15]. Nevertheless, these methods are generally characterized by complex procedures, high cost, and low yield, which limits their commercialization.
As an important component, the binder constitutes only a fraction of the overall electrode, yet it plays a pivotal role in determining the cathode’s electrochemical performance[16,17]. Recent studies highlight the multifaceted contributions of well-designed binders for cathodes, encompassing: (i) providing strong binding between active materials and conductive agents, as well as firm adhesion to the current collector; (ii) forming a protective coating layer and serving as an artificial cathode electrolyte interphase (CEI) film to enhance the interfacial stability of active materials; (iii) promoting the transport of Li+ and maintaining an evenly distributed conductive network; (iv) regulating the viscosity, dispersibility, and pH of slurries, as well as the thermal stability and electrochemical stability of cathodes[18-20].
Poly(vinylidene fluoride) (PVDF) is the most commonly employed binder for cathodes. It is a semi-crystalline linear homopolymer that can adhere to material particles through van der Waals forces[21]. The merits of the PVDF binder can be succinctly outlined as follows: (1) slurries containing PVDF binder exhibit good dispersibility; (2) it remains stable under high-voltage conditions on the cathode side; (3) PVDF exhibits low reactivity with the electrolyte[22]. However, due to its inherent limitations such as low ionic conductivity and suboptimal mechanical properties, PVDF falls short of meeting all the requirements for advanced cathodes[23-25].
Recognizing these drawbacks, a multitude of studies have focused on the development of functional binders to replace PVDF[26-28]. The following factors should be considered for the design of the functional binders: (1) suitable viscosity and dispersity; (2) favorable binding properties between the binder, active materials, and current collector; (3) satisfactory thermal and electrochemical stability at high voltage; (4) efficient ion/electron conduction; (5) the capability to inhibit side reactions, alleviate TM ion dissolution, and reduce gas production[29-33]. To date, the design and fabrication of functional binders for Ni- and Li-rich layered oxides have garnered significant attention, and discernible progress has been achieved [Figure 1]. However, the scattered studies on functional binders for the Ni- and Li-rich cathode have not been systematically presented. Given the lack of systematic organization in prior work, we have conducted a comprehensive review of the relevant research. This article is organized as follows: we expound on the influence of functional groups on binders and offer a comprehensive overview of the recent advances in employing functional binders in Ni- and Li-rich layered oxide cathodes. Additionally, a perspective on the future research directions of functional binders for high-capacity cathodes is presented.
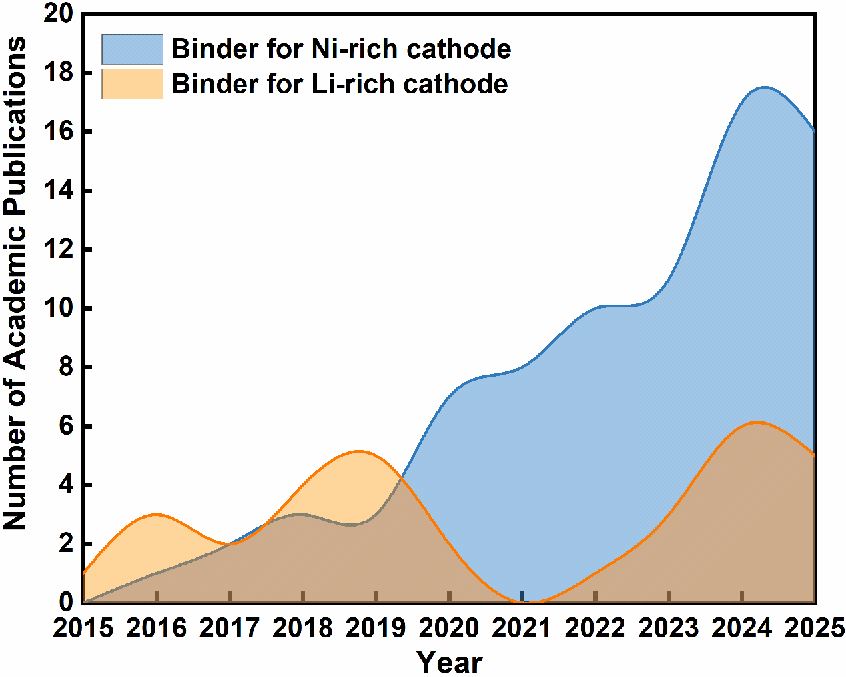
Figure 1. The number of academic publications on binders for Ni- and Li-rich cathodes up to December 31, 2025. Database: Web of Science (https://webofscience.clarivate.cn/wos/woscc/summary/cc2a776a-b3ca-4915-b1b5-89fa140a2377-01a2bc1406/09f9ce99-8b20-4461-bbb5-6a8edcc86175-01a2bbfb11/relevance/1, https://webofscience.clarivate.cn/wos/woscc/summary/39727d87-6cf7-488a-aad4-543460c5a587-01a2bb8a70/9501256c-c8a6-4d1e-8911-359e7ec45a2a-019f0ce736/relevance/1).
TYPES OF BINDER FOR HIGH-CAPACITY CATHODES
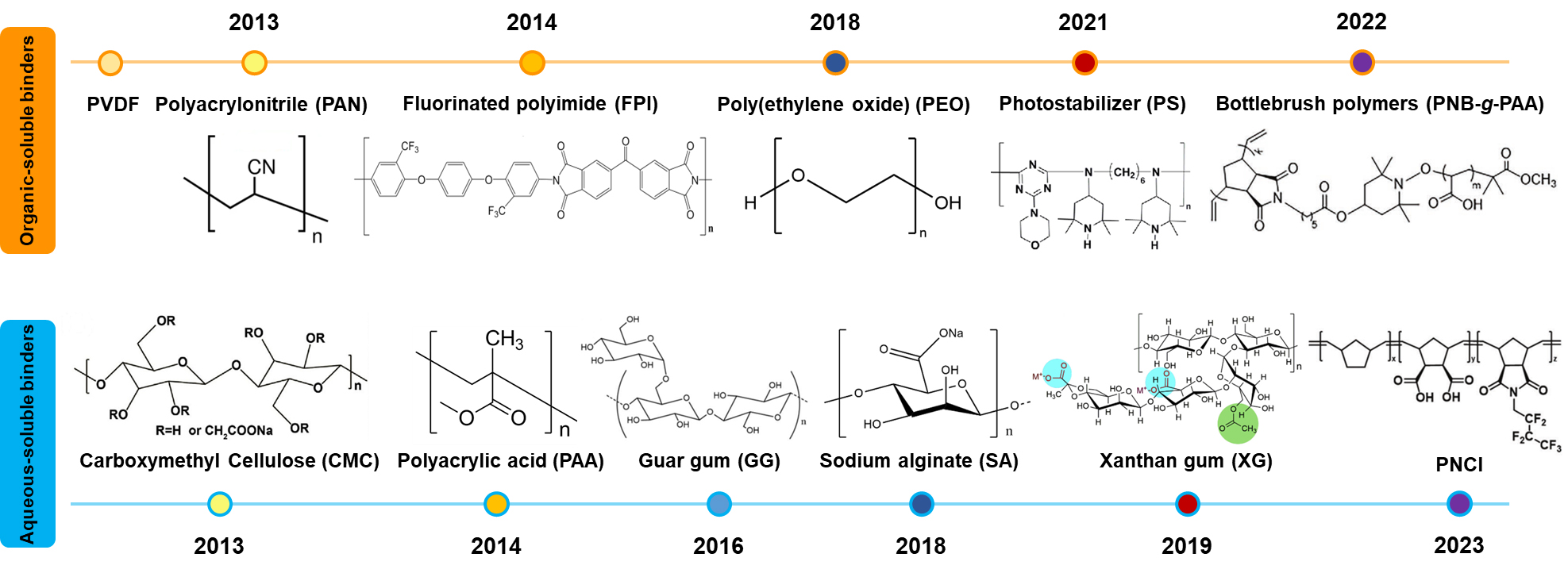
From the commencement of commercial utilization of LIBs to the present, the predominant cathode binder has been organic-soluble, with PVDF standing out as the most extensively employed. Due to its advantages, including superior electrochemical and thermal stability, good dispersion, and affinity to both active material and conductive agent[34,35], PVDF can fulfill most of the requirements for the cathode. However, upon closer consideration of practical factors, certain drawbacks of PVDF prevent it from being the ultimate binder to meet the evolving demands of high-capacity cathode materials in the future[36]. The defects of the PVDF binder can be summarized as follows: (1) poor rate performance caused by low ion and electron conduction[37]; (2) inferior mechanical performance resulting from brittleness; (3) the possibility of loosened or weakened under electrolyte immersion, leading to the loss of binding capability[4]; (4) limited functionalization, making it challenging to adapt to diverse cathode materials or different electrode mass loadings[38]. Consequently, various modification methods for PVDF have been explored, including grafting and compounding with other binders[39,40]. These methods are effective in overcoming the inherent defects of the PVDF binder, thus improving cathode performance. Moreover, alternative organic-soluble binders [polyacrylonitrile (PAN); poly(ethylene oxide) (PEO); and polyimide (PI)] have also been proposed for high-capacity cathodes[4,41]. However, the use of toxic and expensive N-methyl-2-pyrrolidone (NMP) or other organic solvents makes the preparation process based on organic-soluble binders environmentally unfriendly and uneconomical. In this context, it becomes imperative to explore alternative directions for cathode binders.
In conventional cathode production, stringent control over humidity is necessary due to the corrosive impact of moisture on cathode materials. Thanks to rapid advancements in cathode structural optimization and the thoughtful design of aqueous binders, it has become feasible to employ aqueous processes for cathode materials[18]. Well-crafted aqueous binders have the potential to preserve the advantages and functions of typical organic-soluble binders while addressing issues related to toxicity and cost. Some commonly used aqueous binders, such as carboxymethyl cellulose (CMC) and polyacrylic acid (PAA), have already been successfully applied to cathodes and achieved excellent mechanical and electrochemical performances[42-45] [Figure 2]. Additionally, a continuous influx of innovative aqueous cathode binders is being introduced[46-48]. Nevertheless, the large-scale adoption of aqueous cathode binder still encounters several obstacles, primarily encompassing the destruction of interface-bulk structure, dissolution of residual alkali, and exacerbated dissolution of TM ions.
FUNCTIONAL GROUPS IN CATHODE BINDERS
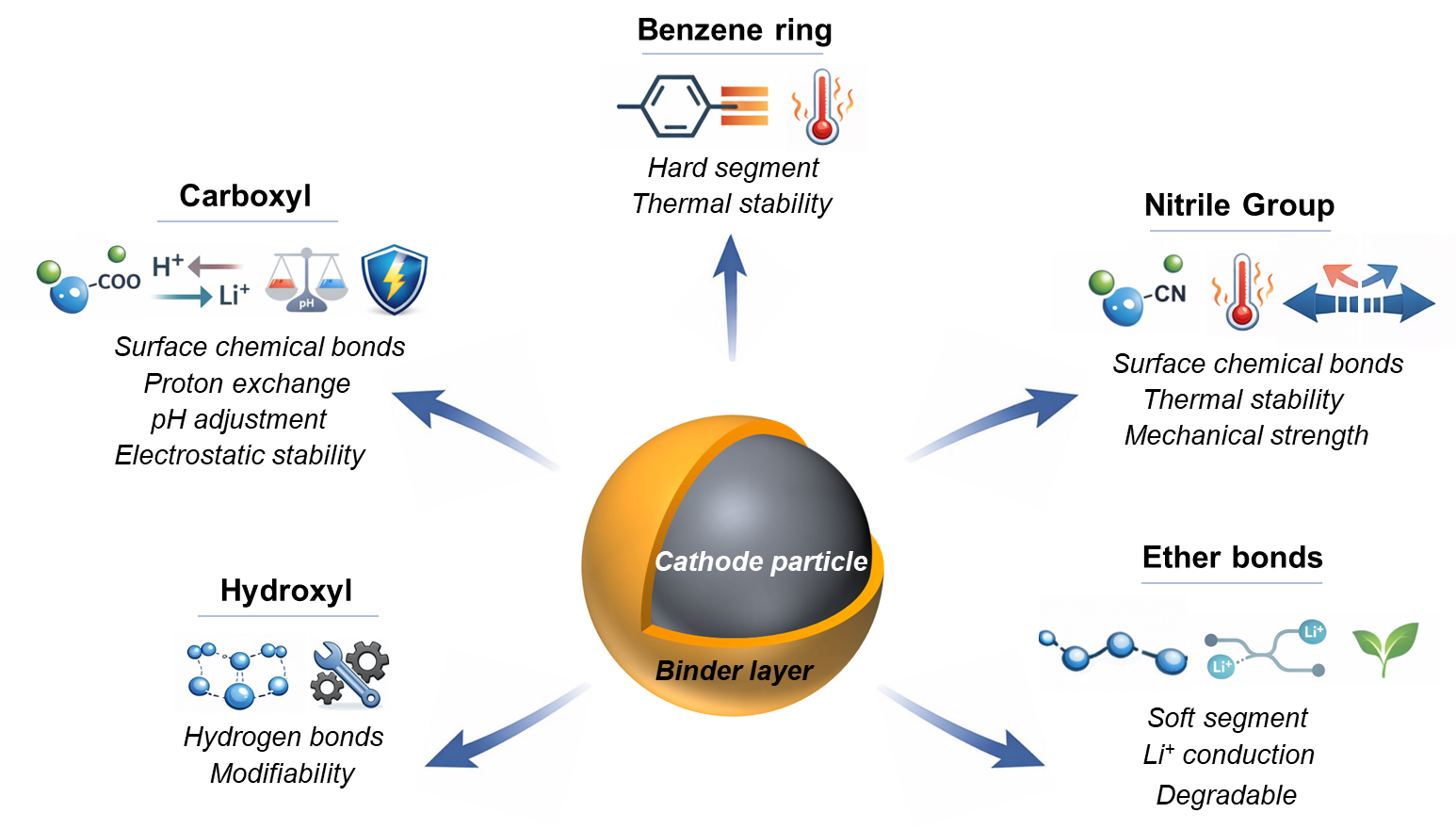
The influence of binders on cathode materials can be encapsulated in terms of interfacial binding and structural stability. Their broader role within the electrode encompasses maintaining electrode integrity and facilitating ion transport. The efficacy of these functions is intricately linked to the functional groups in binders. By deliberately designing binders, it becomes possible to introduce specific functional groups, thus tailoring the enhancement of cathode performance, including both mechanical and electrochemical properties. Figure 3 illustrates the roles of several representative functional groups in binders.
Carboxyl
Carboxyl groups (-COOH), recognized as common active functional groups, are extensively employed to augment the affinity between binders and cathode materials. Their utilization offers a multitude of advantages: (1) ease of implementation due to the abundance of substances containing carboxyl groups; (2) capability to form chemical bonds with the material surface; (3) facilitation of proton exchange, thereby enhancing material stability; (4) pH adjustment of the slurry; (5) provision of electrostatic stability to prevent undesirable particle aggregation. During the cathode preparation process, carboxyl groups can bind to the surface of metal oxide, forming a monodentate –COO-–metal coordination[49,50]. Consequently, the adhesion of the binder to the material surface becomes more robust and uniform. Previous investigations widely document an exchange reaction between H+ and Li+ when subjecting cathode material to an acidic environment[15,45]. In the completely delithiated state of the cathode material, the transference of TM ions occurs from octahedral sites in the TM layer to adjacent tetrahedral sites in the lithium layer. However, the inserted H+ is positioned in Li-deficient octahedral sites, forming a robust covalent bond with the oxygen atom while obstructing the TM ion transport path, hence impeding the migration of TM ions[45].
Maintaining the pH of the cathode slurry in a non-strongly alkaline state is crucial to prevent the corrosion of the Al current collector. As a result, binders containing carboxyl groups are commonly used as a pH-reducing agent to mitigate the potential increase of pH in the cathode slurry[43]. Simultaneously, the addition of carboxylic acid alleviates the slurry agglomeration and mitigates the issue of battery gas generation caused by residual alkali. On another note, the beneficial role of carboxylic groups in improving the slurry manufacturing process is also manifested in optimized electrostatic balance[8]. Specifically, the carboxylic groups bind to the particle surface, ensuring a consistent surface charge. Under the influence of electrostatic repulsion, material particles tend to disperse uniformly rather than agglomerate.
Hydroxyl
Hydroxyls (-OH) are widespread functional groups present in a variety of both natural and synthetic binders. A hydroxyl group consists of one hydrogen atom and one oxygen atom, with the oxygen atom exhibiting greater electronegativity than the hydrogen atom. Due to the polarization effect, there is an uneven charge distribution between the O-H bonds, creating a state where the hydrogen atom behaves akin to a hydrogen ion. When the hydrogen atom encounters another atom with higher electronegativity, electrostatic attraction occurs. This force can be considered as a bridge formed by a hydrogen atom, known as hydrogen bonding. For a binder, hydrogen bonding forces play a pivotal role in bolstering its internal strength, consequently enhancing the overall mechanical property of electrodes.
The strategy of hydrogen bonding has been extensively explored in prior works, and numerous works have reported examples of enhancing the mechanical properties of binders through hydrogen bonding[47,51-53]. Notably, in cases where diverse hydrogen bonds coexist within the binder, a scenario unfolds where gradient hydrogen bonds characterized by distinct binding energies come into existence[54]. Upon the onset of internal stress within the electrodes, the gradual disruption of the hydrogen bonding network ensues. This progressive breakdown acts as a buffer against the stress, effectively preserving the structural integrity of the electrodes.
Beyond their role in mechanical reinforcement, hydroxyl groups offer an additional advantage by conferring significant modifiability to binders. The high reactivity of hydroxyl functional groups facilitates reactions leading to crosslinking between the binder's main chain and various components. This crosslinking imparts diverse and advantageous properties to the binder, including structural stability[54], ion conductivity[55], high voltage resistance[15], amphiphilicity[47], flame retardancy[56], and more.
Benzene ring
The π electrons inherent in the benzene ring showcase a conjugated structure, enabling unrestricted movement throughout the entire ring, in contrast to being confined between two adjacent carbon atoms. This characteristic imparts notable stability to the benzene ring. When integrated into the chain segment of a binder, the benzene ring markedly enhances the mechanical strength of the binder[57,58]. The concept of “soft-hard elastoplasticity” stands as a widely utilized strategy to enhance the mechanical properties of binders[59-62]. Through the alternate incorporation of soft and hard segments into the binder, it achieves a balance between elasticity and structural strength. In this configuration, soft segments serve to mitigate internal stress fluctuations within the electrode, while the hard segments ensure the integrity of the chain structure. Building upon this strategy, the benzene ring is utilized as a “hard segment”. By adjusting the benzene ring content, binders with a harmonious blend of soft-hard characteristics can be realized[4].
The inclusion of benzene rings not only fortifies mechanical strength but also contributes to excellent thermal stability in binders. Numerous studies have documented the remarkable thermal stability of benzene rings, offering a robust strategy for optimizing battery performance, particularly at elevated temperatures[63,64]. What is noteworthy is that compared to the benzene ring, the phenolic group possesses the additional capability to eliminate singlet oxygen[65]. Given the intimate correlation between O2 generation in the cathode and singlet oxygen (1O2), this property becomes crucial. Besides, the electrolyte decomposition under singlet oxygen attack results in the production of CO2 and a series of undesired by-products, significantly influencing the electrochemical performance and safety of batteries. From the binder's perspective, endowing it with the ability to absorb singlet oxygen is highly effective in suppressing gas generation in the cathode and enhancing battery safety. As a result, several studies have incorporated phenolic groups into the cathode binder, notably inhibiting the gas generation and alleviating electrolyte decomposition[65,66].
Ether bonds
In contrast to the aromatic rings discussed earlier, ether bonds (C-O-C) within binders emerge as the epitome of the “soft segment”. These bonds materialize as an oxygen atom bridges two carbon atoms. The relatively diminutive diameter and heightened electronegativity of the oxygen atom induce a denser electron cloud around it, instigating the polarity of the C-O-C bond. With merely two chemical bonds connected to the oxygen atom, the spatial hindrance during rotation is kept to a minimum. This characteristic significantly enhances the flexibility of ether bonds, substantiating their classification as the soft segment in binders[67-69].
The inherent flexibility of ether bonds also enables a more uninhibited swinging of the ether chains. The contribution of binders to Li+ transport is accomplished through the segmented swinging of the binder chains. Specifically, the segmented motion facilitates the migration of lithium ions, enabling their transition from one coordination point to another along the chain or, influenced by an electric field, from one chain to another[70,71]. In essence, the ether bonds promote chain movement, expediting the transport of Li+ within the electrode. Numerous natural binders inherently contain ether bonds, such as guar gum (GG)[51], sodium alginate[72], xanthan gum (XG)[47], etc., and are employed to augment Li+ transport in cathodes.
Ether bonds additionally offer the benefit of facile degradability. Being relatively active, ether bonds are prone to oxidation or even biodegradation[73-75]. For binders, this easy degradability implies a simplified process for recycling batteries, presenting a notable advantage over traditional PVDF binders that struggle with effective removal during recycling. However, the ease of degradation poses a dual challenge, suggesting a potential limitation on the binder’s lifespan. Achieving a delicate equilibrium between the binder’s lifespan and degradability becomes a critical consideration for subsequent researchers.
Nitrile groups
Cyanocompounds, distinguished by the presence of nitrile groups (-C≡N), have garnered significant attention as cathode binders owing to their broad electrochemical window, low viscosity, and high thermostability. Similar to hydroxyl groups, nitrile groups exhibit the capacity to forge robust interactions with the surrounding environment, utilizing hydrogen bonding and dipole-dipole interactions to augment mechanical properties[41,76]. The formidable bond energy inherent in the triple bond of nitrile groups necessitates higher energy for decomposition or reaction. Furthermore, the shorter bond length of the triple bond imparts a more compact molecular structure, endowing binders with superior thermal stability.
Beyond their intrinsic mechanical strength and thermal stability, nitrile groups exert a positive influence on the structure of cathode materials. Upon interfacing with the material surface, one pair of lone electrons in the nitrile group acts as Lewis base and coordinates with TM to form –C≡N → Ni3+ coordination interaction[77-79]. This coordination serves to stabilize the cathode/electrolyte interface via isolating the cathode material from the electrolyte. The isolation plays a pivotal role in extending the service life of active material, underscoring the multifaceted advantages offered by cyanocompounds as cathode binders[80].
BINDERS FOR NI-RICH CATHODES
The high Ni content of Ni-rich layered oxides is a key factor contributing to their high available capacity (≈ 200 mAh g-1); thus, LiNi0.8Co0.1Mn0.1O2 (or compositions with even higher Ni ratios) has garnered considerable attention[81,82]. To unlock the full potential of this promising material, overcoming hurdles such as interfacial/bulk structure instability, TM ions dissolution, susceptibility to corrosion/penetration by the electrolyte, and overall electrode degradation arising from irreversible phase transition must be effectively addressed[83-85]. The development of functional binders is acknowledged as an effective and economical way to overcome the above-mentioned drawbacks associated with Ni-rich layered oxides[29,86].
Ensuring the stability of the material interface is crucial for sustained long-term cycling performance. To achieve this, a protective layer is constructed using a functional binder. In the study by Li et al., the combination of PAA with PVDF demonstrated enhanced performance[87]. It was observed that the residual lithium on the particle surface was partially consumed by PAA. Specifically, PAA decomposes into propionic acid by homolysis reaction under the action of electricity. They proposed that in situ reaction with lithium resulted in the formation of lithium propionic acid (LiPAA). Subsequently, LiPAA was coated on the particle surface and acted as an artificial CEI film to sustain interfacial stability. In comparison to PVDF, the PVDF-LiPAA coating layer exhibits greater uniformity. Moreover, unlike traditional coating methods, the PVDF-LiPAA protective layer is easily formed and does not require additional energy. The PVDF-LiPAA binder prevents the destruction of NCM811 particles and inhibits crack propagation on the particle surfaces, thus enhancing the capacity retention of the LiNi0.8Co0.1Mn0.1O2 (NCM811) cathode from 30.2% to 66.6% after 300 cycles at 1 C[87].
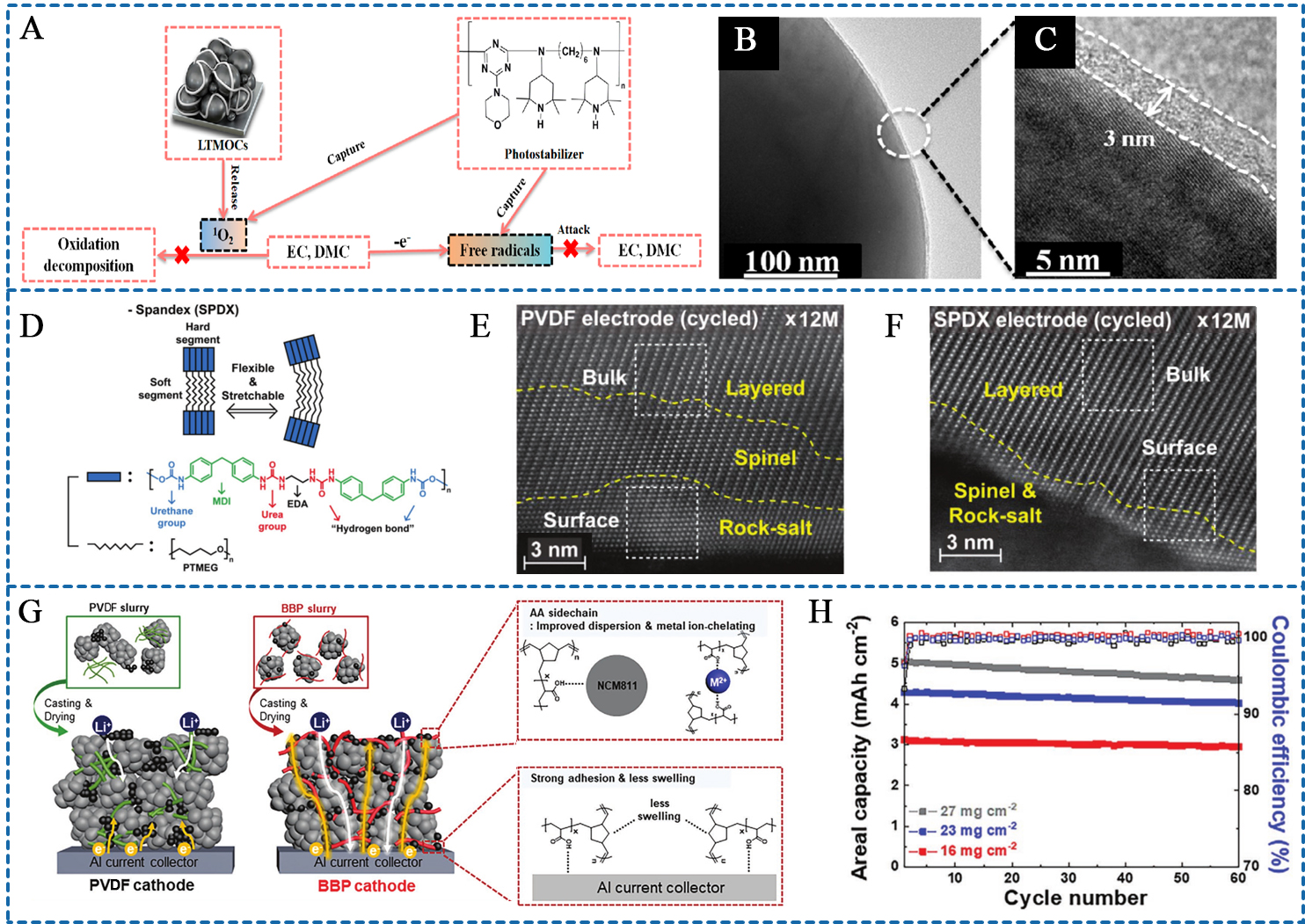
For Ni-rich layered oxides, interface instability also results in material corrosion, triggering the release of O2, CO2, CH4, and other gases. The generation of these various gases not only poses harm to the cathode but also compromises battery safety. It has been demonstrated that the O2 produced in batteries is directly linked to singlet oxygen, while CO2/CH4 generation is derived from the electrolyte attacked by free radicals[88-91]. Therefore, ensuring interfacial stability and suppressing singlet oxygen/free radical generation at the interface holds paramount importance. Drawing inspiration from the natural coping mechanisms against chemical degradation, Cui et al. proposed an antiaging binder [photostabilizer (PS)] with singlet oxygen- and free radicals-scavenging ability[65]. They demonstrated that the hindered amines and electron-deficient heteroaromatic amine motifs confer the PS binder with the capability to scavenge singlet oxygen and free radicals, thereby alleviating electrolyte decomposition at the material interface [Figure 4A]. By using transmission electron microscopy (TEM) images, they found that the SP binder promotes the formation of ultrathin, uniform, and effective CEI films [Figure 4B and C], significantly enhancing the interfacial stability of particles and leading to superior battery performances.
Figure 4. (A) Schematic illustration of the singlet oxygen and free radicals scavenging ability of PS binder; (B and C) TEM images of PS binders-based cathode material surface after 400 cycles; (A-C) are reprinted with permission from Ref.[65], Copyright © 2021, American Chemical Society; (D) Binding scheme of SPDX binder with NCM particle via hydrogen bonding interaction; (E and F) HAADF-STEM images of the PVDF and SPDX-based cathodes after 100 cycles; (D-F) are reprinted with permission from Ref.[57], Copyright © 2020 Wiley; (G) Schematic representation of the BBP and PVDF cathodes; (H) Cycling performance of BBP cathodes as a function of areal mass loading. (G and H) are reprinted with permission from Ref.[92], Copyright © 2021 Wiley. LTMOC: Layered oxides-based cathodes; EC: ethylene carbonate; EMC: ethyl methyl carbonate; SPDX: spandex; PTMEG: poly(tetramethylene ether glycol); MDI: methylene diphenyl diisocyanate; EDA: ethylenediamine; PVDF: poly(vinylidene fluoride); BBP: bottlebrush polymer; NCM: LiNixCoyMn1-x-yO2, (x ≥ 0.8); PS: photostabilizer; TEM: transmission electron microscopy; HAADF-STEM: high-angle annular dark-field scanning transmission electron microscopy.
The high elasticity and strong affinity of the binder to cathode particles facilitate a tight bond with the particle surface, exerting a counterforce during volume changes and effectively inhibiting the formation of internal cracks. In an investigation by Chang et al., the impact of binders on the structural stability of NCM811 materials during cycling was explored, with a particular focus on the mechanical property of elasticity[57]. They reported that this property plays a crucial role in adjusting the shear forces generated during slurry mixing and ensuring uniform coverage of material particles by the binder. As illustrated in Figure 4D, spandex (abbreviated as SPDX) was first reported as a binder for the NCM811 cathode, demonstrating excellent elasticity and adhesion that enable uniform coverage of NCM811 particles. The SPDX binder greatly reduces microcrack formation by directing force toward the center of material particles, thus ameliorating the bulk structural instability caused by increased Ni content. Furthermore, the NCM811 cathode with SPDX binder exhibits minimal irreversible phase transitions, with only thin spinel and rock-salt phases observed in the surface layer [Figure 4E and F]. Due to the improved stability of the bulk/interface structure, the TM ion dissolution of the SPDX-based NCM811 cathode can be inhibited, so as to achieve superior cycling performance.
In addition to charge-discharge capacity, the performance of a cathode at high current densities or elevated mass loadings is a crucial parameter. The rate performance and performance under ultimate mass loading are contingent on the kinetics of Li+ transport. To facilitate the kinetics of Li+, Wang et al. synthesized a poly (imide-siloxane) (PIS) binder with an unobstructed Li+ transport pathway by integrating polydimethylsiloxanes (DMS) with Si-O-Si bonds in aromatic PI[58]. The strategy of combining rigid and flexible chains together provides adequate elasticity while ensuring strong attractive interactions. This combination enables the creation of an interfacial ion/electron conductive environment and widely distributed Li+ transport channels. As a result, the PIS-based NCM811 cathodes exhibit excellent capacity retention of 83% after 500 cycles at 1 C and rate performance of 137.6 mAh g-1 at 5 C under the cutoff voltage of 4.3 V. Furthermore, galvanostatic intermittent titration (GITT) and variable scanning rate cyclic voltammetry (CV) test show the promoted Li+ diffusion coefficient of NCM811 cathodes by PIS binder[58].
The integrity of the conducting network is another critical factor influencing Li+ transport in batteries. A well-developed conductive network can be achieved through the double affinity of binders for both the conductive agent and cathode material. Specifically, the active material-binder-conductive agent package structure can be obtained by capturing and fixing the electrode component with binders. Kim et al. designed an amphiphilic bottlebrush polymers (BBPs) binder through ring-opening metathesis polymerization[92], employing hydrophobic polynorbornene (PNB) as backbones and hydrophilic PAA as side chains, imparting amphipathic properties to the BBPs binder. They suggested that hydrophobic PNB backbones enable the use of non-aqueous solvents in cathode preparation. Concurrently, PAA side chains ensure strong adhesion forces through hydrogen bonding and chelate TM ions. Leveraging the amphiphilicity of the BBPs binder, cathode slurries can be uniformly dispersed, and the fabricated high-mass-loading BBPs-NCM811 cathodes present well-developed electron/ion conducting network [Figure 4G]. A high-mass-loading BBPs-NCM811 cathode (27 mg cm-2, corresponding to 5.2 mAh cm-2) with superior cycling stability is achieved even with the binder content reduced to 1% [Figure 4H].
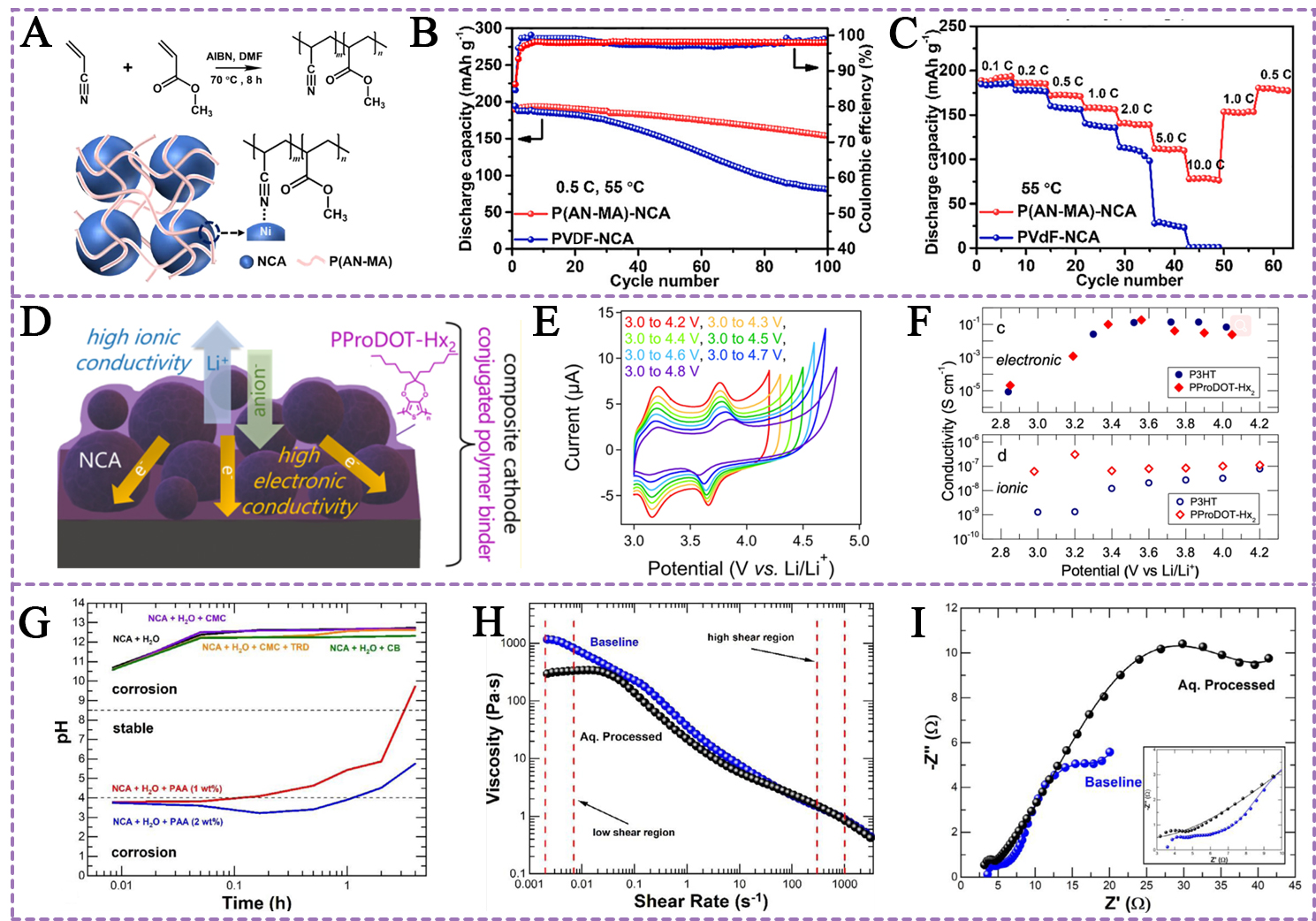
NCA (LiNixCoyAl1-x-yO2, x ≥ 0.8) is another Ni-rich cathode material that also possesses a high theoretical capacity[93,94]. However, the NCA cathode encounters formidable challenges, including the stringent requirements of the fabrication process and poor thermal stability[95]. To address the above factors, a series of functional binders have been proposed. Jin et al. designed a poly (acrylonitrile-co-methyl acrylate) [P(AN-MA] copolymer binder with electron-rich -C≡N groups and high-temperature resistance for LiNi0.8Co0.1Al0.1O2 cathodes[80] [Figure 5A]. They proposed that -C≡N groups enable P(AN-MA) binder to interact with the Ni3+ site on the surface of NCA material particles. The P(AN-MA) binder improves the thermostability of LiNi0.8Co0.1Al0.1O2 cathodes since the bonding between -C≡N groups and Ni3+ site maintains consistent strong adhesive ability even at a high temperature of 55 °C. In addition, the introduction of methyl acrylate (MA) facilitates chain mobility, thereby improving the flexibility of NCA cathodes at high temperatures. Attributed to the above merits, the LiNi0.8Co0.1Al0.1O2 cathode with P(AN-MA) binder delivered a discharge capacity of 155 mAh g-1 with a capacity retention of 81.5% after 100 cycles [Figure 5B]. It also presents excellent rate performance (99 mAh g-1 at 10 C, 1 C = 180 mAh g-1) under the harsh condition of 55 oC [Figure 5C].
Figure 5. (A) Schematic illustration of P(AN-MA) copolymer binder and the interaction between P(AN-MA) binder and NCA material; (B) Long-term cycling performance and (C) rate capability of NCA cathode based on P(AN-MA) and PVDF binders; (A-C) are reprinted with permission from Ref.[80], Copyright © 2021, Elsevier; (D) The sketch map of NCA cathode with PProDOT-Hx2 binder; (E) CV curves of PProDOT-Hx2 film at various potential intervals; (F) Electronic and ionic conductivities of P3HT and PProDOT-Hx2 binders; (D-F) are reprinted with permission from Ref.[98], Copyright © 2020, American Chemical Society; (G) The pH of NCA-binder samples in water; (H) Viscosity profile of baseline and aqueous-processed slurries; (I) Nyquist plot of NCA cathodes with different binders after 100 cycles; (G-I) are reprinted with permission from Ref.[43], Copyright © 2021, Elsevier. AIBN: Azodiisobutyronitrile; DMF: N, N-dimethylformamide; NCA: LiNixCoyAl1-x-yO2, (x ≥ 0.8); P(AN-MA): poly (acrylonitrile-co-methyl acrylate); PVDF: Poly(vinylidene fluoride); PProDOT-Hx2: dihexyl-substituted poly(3,4-propylenedioxythiophene); P3HT: poly(3-hexylthiophene).
Another critical challenge for NCA cathodes is their limited stability and short storage lifetime, which arise from residual alkali introduced during the material synthesis process[96]. Residual alkali species, typically present as LiOH and Li2CO3, lead to increased battery impedance and side reactions with the electrolyte, which in turn cause gas evolution and accelerated interfacial degradation. These processes not only deteriorate the electrochemical performance of batteries but also compromise their safety. Sawangphruk et al. synthesized pre-lithiated Nafion ionomer binder as an innovative solution for LiNi0.8Co0.15Al0.05O2 cathodes using an ion exchange method[97]. To mitigate the impact of residual alkali, ethanol was employed as the solvent for pre-lithiated binder. The modification of the material surface by a pre-lithiated binder-ethanol system is considered the reason for the improvement of the electrochemical performances of the NCA cathode. This is attributed to the different solubility properties of residual alkali in various solvents. Specifically, when the pre-lithiated binder is dissolved in ethanol, the resulting solution tends to solubilize the hydroxide on the surface of the NCA material, thereby altering the interfacial chemical composition and alleviating deterioration caused by residual alkali. The NCA cathode with pre-lithiated binder-ethanol combination delivers an initial discharge capacity of 190 mAh g-1 at 0.5 C and then with 82% capacity retention after 200 cycles. In sharp contrast, the discharge capacity of the NMP-mediated NCA cathode is 171 mAh g-1 at the initial step and reduces to 135 mAh g-1 (79% capacity retention) after 200 cycles.
To enhance the electrochemical kinetics of NCA cathodes, Thompson et al. engineered and fabricated a dihexyl-substituted poly(3,4-propylenedioxythiophene) (PProDOT-Hx2) polymer as an electrochemical binder for LiNi0.8Co0.15Al0.05O2 cathodes[98] [Figure 5D and E]. They found that the open, partly disordered morphology of the PProDOT-Hx2 binder allows efficient ion insertion/deinsertion processes, which facilitate ionic conductivity. The flexible electrochemical doping environment of conjugated polymers provides ample scope for improving electronic conductivity. Simultaneously, the propylenedioxythiophene backbone ensures electrochemical stability, while the hexyl side chains enhance the solubility in nonpolar processing solvents. The advantages of the PProDOT-Hx2 binder, including rapid ionic/electronic conductivity and superior electrochemical stability, enable NCA-PProDOT-Hx2 cathodes to deliver exceptional cycling and rate performance. The PProDOT-Hx2 binder achieves a high electronic conductivity of 0.1 S cm-1 over the potential range of 3.4-4.0 V and an ultrahigh ionic conductivity that is one order of magnitude higher than that of the poly(3-hexylthiophene) (P3HT) binder over the same operating potential range [Figure 5F]. Meanwhile, NCA-PProDOT-Hx2 cathodes exhibit five times higher discharge capacity (111 mAh g-1) compared to NCA-PVDF cathodes at 6 C.
Recently, great interest has been drawn to aqueous binders for NCA cathodes[31,99]. Hawley et al. introduced PAA as an aqueous binder for the LiNi0.8Co0.15Al0.05O2 cathode[43]. In view of the high pH of the NCA material-water system, the acidic PAA binder serves as a corrosion-mitigating and surface-stabilizing agent in the aqueous slurry. Figure 5G illustrates the pH value of NCA-binder samples in water. Additionally, in this study, PAA was found to perform several crucial roles: (1) alleviating the corrosion of aluminum current collector; (2) forming a coating layer on the surface of NCA material to inhibit lithium dissolution; (3) providing an electrostatic equilibrium environment for NCA particles to prevent agglomeration; and (4) exhibiting similar rheological characteristics when combined with CMC and TRD 202A binders [Figure 5H]. The authors found that despite a higher capacity retention rate was achieved for water-involved NCA cathodes, their discharge capacity is lower than that of counterparts in the initial cycling. This can be attributed to the inevitable leaching of lithium during slurry preparation. Furthermore, cracks form on the surface of aqueous-processed NCA cathode, leading to increased charge-transfer resistance (Rct) and limiting reversible capacity at high rates [Figure 5I].
For Ni-rich NCM and NCA materials, ensuring structural stability is a paramount concern. This challenge results in irreversible Ni loss, leading to a persistent decline in capacity. Such problems impede the sustained exceptional performance of Ni-rich cathode materials in electric vehicles or large-scale energy storage devices. Ongoing research on Ni-rich materials underscores the critical importance of enhancing their structural stability. The enhancement of structural stability by binders for Ni-rich materials involves two key aspects: firstly, the formation of a protective layer on the material surface to prevent corrosion from external environments and alleviate internal stresses; secondly, involvement in proton exchange reactions with the materials to augment stability at the crystal structure level. Subsequent research on binders should meticulously consider these two aspects.
BINDERS FOR LI-RICH CATHODES
Li-rich manganese-based layered oxides [xLi2MO3·(1-x)LiMO2 (M = Ni, Co, Mn)] have garnered significant attention as potential candidates for next-generation LIBs due to their high theoretical specific capacity (> 250 mAh g-1)[100-102]. Nevertheless, Li-rich cathode materials suffer from severe capacity loss and voltage decay during cycling, which hinders their commercial applications[72,103]. According to previous researches, the capacity/voltage degradation is mainly caused by the structure instability of Li-rich layered materials, which further leads to irreversible TM migration and undesirable side reactions[104-106]. Addressing these issues, a series of advanced binders have been investigated[51,107]. The key focus in the exploration of Li-rich cathode binder is to play its protective role on material’s interfacial and bulk structure.
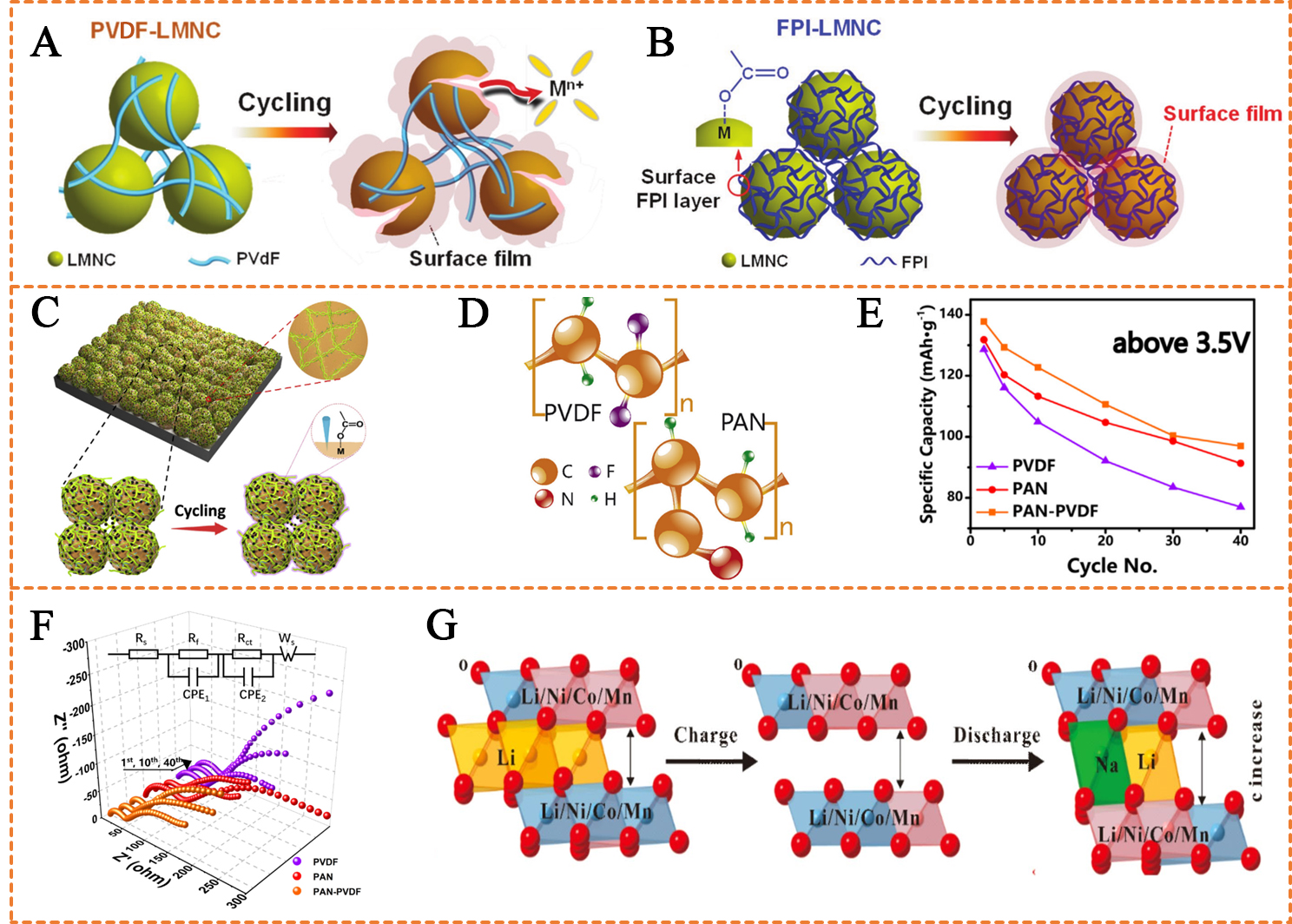
Enhancing interfacial stability by binders can be categorized into two approaches: (1) binder coated on the material surface directly; (2) binder interacts with active material to generate a secondary coating layer. In the first category, the binder layer serves as a protective barrier for active materials. This requires that the binder has superior affinity to the active material to form a uniform coating. For instance, Pham et al. reported modified PVDF with fluorinated PI (FPI) as a novel binder for high-voltage Li-rich layered oxide cathodes[4]. They suggested that the combination of fluorinated PI and PVDF improves the affinity for materials, forming a robust surface coating layer on the Li-rich materials. This coating acts as a barrier, protecting the cathode material and inhibiting metal dissolution and structural degradation [Figure 6A and B]. Similarly, Yin et al. utilized a GG binder for Li-rich cathodes and studied the CEI films, morphology, microstructure, and surface elements of the prepared GG cathodes[51]. They observed that the GG binder forms a stable coating layer on the Li-rich material, alleviating the decomposition of active materials. In addition, the GG binder exhibited stronger adhesion, preventing structural collapse of the material. Attributed to the improved interfacial stability, the resistance of the cathode electrolyte interphase (RCEI) of the Li-rich cathode with the GG binder (88.6 Ω) is significantly lower than that of the cathode using the PVDF binder (243.3 Ω). More importantly, the dissolution of Mn2+ and the surface phase transformation from layered to spinel are also inhibited.
Figure 6. (A) Schematic illustration of PVDF-LMNC and (B) FPI-LMNC cathode before and after 100 cycles; (A and B) are reprinted with permission from Ref.[4], Copyright © 2017, Wiley; (C) Sketch map of XG cathodes; (C) is reprinted with permission from Ref.[47], Copyright © 2019, Elsevier; (D) Sketch map of PAN and PVDF; (D-F) are reprinted with permission from Ref.[41], Copyright © 2017, Elsevier; (E) Divided capacity of PAN-PVDF, PAN and PVDF cathodes above 3.5 V; (F) Nyquist plots of different electrodes in 1st, 10th, 40th at the state of DOD ≈ 50%; (G) Schematic diagram of ion-exchange process provided by CMC during cycling. (G) is reprinted with permission from Ref.[72], Copyright © 2016, Wiley. PVDF: Poly(vinylidene fluoride); LMNC: Li1.13Mn0.463Ni0.203Co0.203O2; FPI: fluorinated polyimide; PAN: polyacrylonitrile; XG: xanthan gum; DOD: depth of discharge: CMC: carboxymethyl cellulose.
In the second category of interfacial protection, the secondary coating layer typically exhibits higher stability and stronger bonding with the material particles. Zhang et al. proposed XG as an aqueous binder for Li-rich cathodes[47] [Figure 6C]. The inter/intramolecular hydrogen bonding and hydrophobic backbone of XG jointly promote the formation of a unique double helix superstructure, resulting in a significant mechanical interlocking effect[108]. Meanwhile, the abundant charged carboxyl groups in XG effectively stabilize the cathode surface through –COO-–metal coordination chemical bonding with material particles. Consequently, the integrity of the Li-rich cathode was ensured, and cathode degradations (particle pulverization, metal ion dissolution, microstructural degradation) were alleviated. The resulting Li-rich cathode with the XG binder delivers an impressive initial discharge capacity of 275.6 mAh g-1, along with 98.4% capacity retention after 200 cycles at 0.1C.
Beyond interface factors, TM ion dissolution and voltage decay in Li-rich materials are also linked to their bulk structure. Binders contribute to material structure stability through two mechanisms: (1) exerting mechanical force to inhibit volume change; (2) inserting into material particles by ion exchange to stabilize the lattice structure. Wu et al. designed a PAN and PVDF composite binder (PAN-PVDF) for layered Li-rich oxides (LLROs) cathodes[41] [Figure 6D]. PAN exhibited excellent electrochemical stability and anti-swelling properties in LLROs cathodes, enhancing material structural-change resistance at voltages higher than 3.5 V [Figure 6E]. On the other hand, the wettability of PVDF in electrolyte ensures good contact between LLROs and electrolyte, thereby resulting in lower interfacial impedance [Figure 6F]. As a consequence, the retention of 40th cycled PAN-PVDF-LLROs cathode is increased to 89.6%, significantly higher than that of the separate PAN (85.1%) and PVDF (77.5%) cathodes. The discharge capacity of the PAN-PVDF-LLROs cathode at 1 C has also been improved to 178 mAh g-1. Zhang et al. initially selected CMC as a binder for the Li-rich cathode[72]. They claimed that the cycling stability of the Li-rich cathode was enhanced due to two key effects: (1) CMC provides a strong adhesive property to ensure consistent electrical contact; (2) CMC stabilizes the crystal structure by intercalating Na+ into the lattice via ion exchange, thus suppressing the voltage fading and TM migration [Figure 6G]. The electrochemical tests demonstrate that the Li-rich cathode with CMC binder exhibited an initial discharge capacity of 226 mAh g-1 with 89% capacity retention after 500 cycles at 200 mA g-1.
Some advanced binders can enhance the stability of both interfacial and bulk structure. Yang et al. proposed a facile surface-modifying strategy for Li-rich cathode by PAA binder[45]. The carboxyl groups in PAA chains can react with oxygen species on the surface of the cathode material to form a tightly coated film. This film isolates Li-rich material particles from the electrolyte, therefore minimizing side effects. Besides that, a spontaneous H+/Li+ exchange reaction occurred between Li-rich material and PAA binder, forming a proton-doped bulk structure. Density functional theory (DFT) calculations demonstrated that the inserted H+ stabilize lattice structure, consequently inhibiting the TM dissolution and voltage decay[45]. The prepared PAA-based cathode exhibits excellent overall electrochemical performance: reveals slightly voltage decay and retains 88% of capacity after 500 cycles at 200 mA g-1.
The structural instability issue, previously discussed in Ni-rich materials, persists in Li-rich materials and is accompanied by voltage decay phenomena. Voltage decay not only leads to a reduction in battery energy density but also adversely affects the normal operation of electronic devices. Furthermore, Li-rich materials exhibit pronounced gas generation during the charging and discharging processes, posing a significant threat to battery safety. Therefore, the binder design for Li-rich cathode materials needs to address both the mitigation of voltage decay and the suppression of gas generation. Specifically, mitigating voltage decay relies on the protective layer formed by the binder, while suppressed gas production depends on the binder's ability to absorb singlet oxygen and radicals.
CONCLUSION AND OUTLOOK
Despite their promising potential for wide applications, the commercialization of high-capacity Ni- and Li-rich layered oxide cathode materials is limited by inherent defects, including interfacial instability, bulk structural degradation, TM ion dissolution, and gas evolution. Among various feasible strategies, increasing attention has been directed toward research on functional binders.
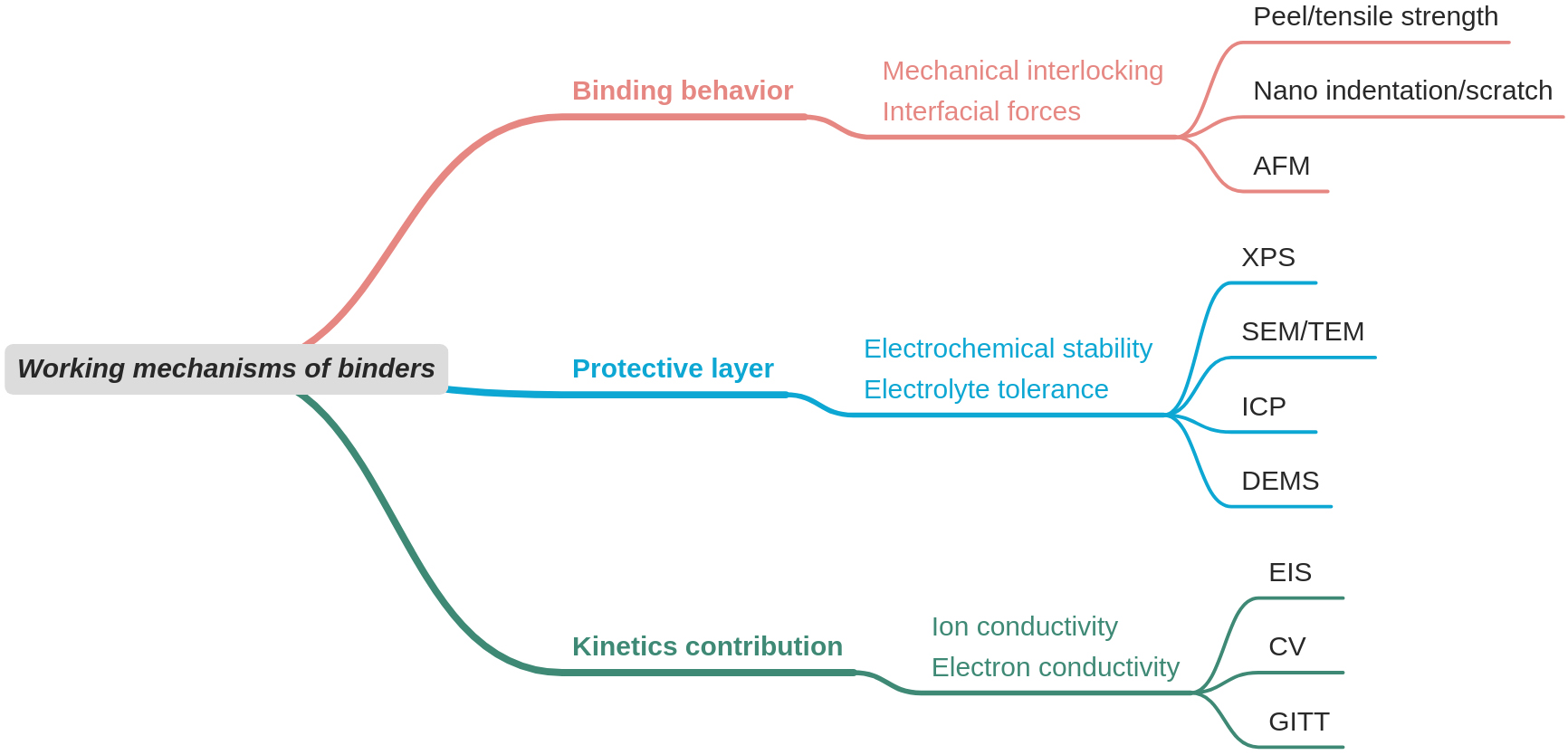
This review highlights recent advances in functional binders for high-capacity Ni- and Li-rich layered oxides. The proposed mechanisms of action of binders are elucidated. The contributions of binders to cathode materials, based on existing studies, can be summarized as follows: (1) the binder provides strong binding between active materials and conductive agents, as well as firm adhesion to the current collector, preventing electrode detachment and cracking[109]; (2) a polymer binder with high strength enhances the internal cohesion of electrodes and maintains electrode integrity; (3) the binder forms a uniform coating layer on the material surface, exerting a counterforce to protect the material structure and inhibit TM ion dissolution. Meanwhile, the binder layer acts as an artificial CEI film to stabilize the cathode interface, preventing corrosion by the electrolyte and reducing gas production; (4) the binder improves the kinetics of Li+ transport by providing “hopping channels”[110]. Li+ can be attracted to 4-5 O/N atoms in binders due to their electronegativity. As binder chains undergo segmental motion, they dissociate from their original coordination sites and subsequently re-coordinate with neighboring sites. In the presence of an electric field, Li+ can gradually move sideways; (5) various functional groups can be introduced to design binders, regulating their dispersibility, pH value, thermostability, electrochemical stability, and other characteristics through compounding, polymerization, grafting, and other methods. Figure 7 provides a schematic illustration that links binder working mechanisms to key performance parameters and the corresponding characterization and validation methods.
Figure 7. Schematic illustration that links binder working mechanisms with key performance parameters and the corresponding characterization and validation methods. AFM: Atomic force microscopy; XPS: X-ray photoelectron spectroscopy; SEM: scanning electron microscopy; TEM: transmission electron microscopy; ICP: inductively coupled plasma; DEMS: differential electrochemical mass spectrometry; EIS: electrochemical impedance spectroscopy; CV: cyclic voltammetry; GITT: galvanostatic intermittent titration technique.
Looking ahead, there are still ample opportunities for further investigation of binders. Electrochemical tests of Ni- and Li-rich layered oxides with different binders reported in publications were mostly performed in the laboratory under mild conditions using coin-cell testing systems; however, actual battery production involves high-temperature operating environments and mass loadings of active material that are three to four orders of magnitude greater than in coin cells, which exacerbates capacity degradation and safety issues. Therefore, it is necessary to verify the practical application of binders by assembling large-scale soft-pack batteries. The effective design of cathode binders requires in-depth analysis of their working mechanisms, which relies on advanced in situ/ex situ characterization methods and theoretical simulations, including spherical electron microscopy[111-113], synchrotron radiation technology[114-118], and DFT calculations of lowest unoccupied molecular orbital (LUMO)/highest occupied molecular orbital (HOMO) energy[119-121]). Exploring the synergistic interactions between binders and cathode materials, as well as the chemical state of the cathode during lithiation and delithiation, provides critical guidance for understanding binder function, and machine learning can be leveraged to design binders more intricately, capitalizing on insights gained from characterization results. For advanced battery systems, cathode binders are required to achieve a high level of comprehensive performance, including enhanced oxidative stability under high operating voltages, robust chemical tolerance against electrolytes and their decomposition products, reliable adhesion at elevated temperatures, and sustained functionality in thick electrodes with high areal loadings. Notably, performance requirements for binders are strongly dependent on the specific cathode: for Ni-rich cathodes, binder design must particularly emphasize stabilizing interfacial and bulk structures, whereas binders for Li-rich cathodes are required to exert additional regulatory effects on severe voltage decay and gas evolution. The improvements implemented to address binder failure mechanisms are effective: binder failure in batteries can be categorized into two primary modes, degradation failure and interface failure. Degradation failure originates from structural damage to the polymer binder due to electrolyte attack, high operating voltages, and accumulated stress, resulting in loss of mechanical integrity, whereas interface failure arises from deterioration of interfacial interactions between the binder and active materials, commonly caused by electrolyte swelling, partial dissolution, and insufficient interfacial adhesion. Both failure modes ultimately lead to active material delamination, inducing degraded cycling stability.
In conclusion, a commercially viable high-energy-density battery requires a reliable high-capacity cathode. As an indispensable electrode component, the binder plays a crucial role in unlocking cathode capacity, not only protecting the cathode during cycling but also being precisely tailored for different cathode materials. Despite significant achievements in high-capacity cathode binder research, further mechanistic studies and practical experiments are needed to translate laboratory results into industrial applications. It is anticipated that, through theoretical design and innovative experimentation, high-energy-density battery technology will achieve a bright and promising future.
DECLARATIONS
Authors’ contributions
Writing-original draft: Qiu, J.; Zhong, L.
Writing-review & editing: Xiao, M.; Wang, S.; Huang, S.
Collecting & analyzing: Wang, S.
Supervision: Han, D.; Meng, Y.
Funding acquisition: Han, D.; Meng, Y.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Key Research and Development Program (2019YFA0705701); the National Natural Science Foundation of China (22179149, 22075329, 22008267, 51573215, and 21978332); the Guangdong Basic and Applied Basic Research Foundation (Grant No. 2021A0505030022); and the Research and Development Project of Henan Academy of Sciences, China (No. 232018002). We also acknowledge financial support from Shandong Lecron Industrial Development Group Co., Ltd., China.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Yang, H.; Wu, H. H.; Ge, M.; et al. Simultaneously dual modification of Ni-rich layered oxide cathode for high-energy lithium-ion batteries. Adv. Funct. Mater. 2019, 29, 1808825.
2. Zhou, Y.; Ding, Y.; Chen, Y.; et al. Thermal degradation of lithium-ion battery cathodes: a machine learning prediction of stability and safety. Energy. Mater. 2025, 5, 500077.
3. Kim, D.; Jayasubramaniyan, S.; Kim, S.; et al. Multi-layered yolk-shell design containing carbon bridge connection for alloying anodes in lithium-ion batteries. Energy. Mater. 2025, 5, 500072.
4. Pham, H. Q.; Kim, G.; Jung, H. M.; Song, S. W. Fluorinated polyimide as a novel high-voltage binder for high-capacity cathode of lithium-ion batteries. Adv. Funct. Mater. 2017, 28, 1704690.
5. Lu, X.; Zhang, X.; Tan, C.; et al. Multi-length scale microstructural design of lithium-ion battery electrodes for improved discharge rate performance. Energy. Environ. Sci. 2021, 14, 5929-46.
6. Feng, X.; Gao, Y.; Ben, L.; Yang, Z.; Wang, Z.; Chen, L. Enhanced electrochemical performance of Ti-doped Li1.2Mn0.54Co0.13Ni0.13O2 for lithium-ion batteries. J. Power. Sources. 2016, 317, 74-80.
7. Xie, X.; Cui, J.; Liu, Z.; et al. Constructing a surface spinel layer to stabilize the oxygen frame of Li-rich layered oxides. Inorg. Chem. Front. 2022, 9, 5828-39.
8. Kuo, J.; Li, C. Water-based process to the preparation of nickel-rich Li(Ni0.8Co0.1Mn0.1)O2 Cathode. J. Electrochem. Soc. 2020, 167, 100504.
9. Elizalde-segovia, R.; Das, P.; Zayat, B.; Irshad, A.; Thompson, B. C.; Narayanan, S. R. Understanding the role of π-conjugated polymers as binders in enabling designs for high-energy/high-rate lithium metal batteries. J. Electrochem. Soc. 2021, 168, 110541.
10. Nitou, M. V. M.; Fang, X.; Wang, J.; et al. The integration of LiNi0.8Co0.1Mn0.1O2 coatings on separators for elevated battery performance. Energy. Mater. 2025, 5, 500018.
11. Kimura, K.; Sakamoto, T.; Mukai, T.; et al. Improvement of the cyclability and coulombic efficiency of Li-ion batteries using Li[Ni0.8Co0.15Al0.05]O2 cathode containing an aqueous binder with pressurized CO2 gas treatment. J. Electrochem. Soc. 2018, 165, A16-20.
12. Loghavi, M. M.; Bahadorikhalili, S.; Lari, N.; Moghim, M. H.; Babaiee, M.; Eqra, R. The effect of crystalline microstructure of PVDF binder on mechanical and electrochemical performance of lithium-ion batteries cathode. Zeitschrift. für. Physikalische. Chemie. 2020, 234, 381-97.
13. Tsai, P.; Wen, B.; Wolfman, M.; et al. Single-particle measurements of electrochemical kinetics in NMC and NCA cathodes for Li-ion batteries. Energy. Environ. Sci. 2018, 11, 860-71.
14. Dong, T.; Mu, P.; Zhang, S.; Zhang, H.; Liu, W.; Cui, G. How do polymer binders assist transition metal oxide cathodes to address the challenge of high-voltage lithium battery applications? Electrochem. Energ. Rev. 2021, 4, 545-65.
15. Zhao, T.; Meng, Y.; Ji, R.; Wu, F.; Li, L.; Chen, R. Maintaining structure and voltage stability of Li-rich cathode materials by green water-soluble binders containing Na+ ions. J. Alloys. Compd. 2019, 811, 152060.
16. Cui, X.; Wang, X.; Pan, Q. Achieving fast and stable Li+ transport in lithium-sulfur battery via a high ionic conduction and high adhesion solid polymer electrolyte. Energy. Mater. 2023, 3, 300034.
17. Zhou, X.; Yang, J.; Wang, R.; Zhang, W.; Yun, S.; Wang, B. Advances in lithium-ion battery materials for ceramic fuel cells. Energy. Mater. 2022, 2, 200041.
18. Brandt, T. G.; Temeche, E.; Tuokkola, A. R.; et al. Nanocomposite Li- and Mn-rich spinel cathodes characterized with a green, aqueous binder system. Chem. Eng. J. 2024, 479, 147419.
19. Gan, Q.; Qin, N.; Guo, H.; et al. Consolidating the vulnerable interphase of Ni-rich layered cathode by multifunctional water-based binder. ACS. Energy. Lett. 2024, 9, 1562-71.
20. Jeong, Y. H.; Won, G.; Kim, S.; et al. A tough, adhesive, and protective binder shield for stabilizing high‐nickel cathodes in lithium‐ion batteries. Adv. Energy. Mater. 2025, 15, e02765.
21. Mazouzi, D.; Karkar, Z.; Reale Hernandez, C.; et al. Critical roles of binders and formulation at multiscales of silicon-based composite electrodes. J. Power. Sources. 2015, 280, 533-49.
22. Zhong, X.; Han, J.; Chen, L.; et al. Binding mechanisms of PVDF in lithium ion batteries. Appl. Surf. Sci. 2021, 553, 149564.
23. Pasquier, A. D.; Disma, F.; Bowmer, T.; Gozdz, A. S.; Amatucci, G.; Tarascon, J. M. Differential scanning calorimetry study of the reactivity of carbon anodes in plastic Li-ion batteries. J. Electrochem. Soc. 2019, 145, 472-7.
24. Zhang, Z.; Ma, J.; Xiao, M.; et al. Poly (propylene carbonate) with extremely alternating structure used as binders for high-loading cathodes by solvent-free method in high-performance NCM811 batteries. Materials. (Basel). 2024, 17, 5466.
25. Kim, H.; Kang, D. H.; Lee, J.; et al. Solvophobic binder crystallinity-tailored advances in solvent-free thick cathodes for high-energy lithium metal batteries. Adv. Funct. Mater. 2025, 2420104.
26. Park, J.; Lee, S.; Choi, W. Surface-intensive doping of Na via pH-dependent polymer for Co-free Ni-rich cathodes in Li-ion batteries. Chem. Eng. J. 2024, 488, 150807.
27. Albers, S.; Timmermann, J.; Brake, T.; et al. Enabling aqueous processing of Ni-rich layered oxide cathodes via systematic modification of biopolymer (polysaccharide)-based binders. Adv. Energy. and. Sustain. Res. 2024, 5, 2400117.
28. Wang, Y.; Fang, Y.; Huang, L.; et al. Stabilizing Nickel-rich cathodes in aqueous process through nanocellulose as water barrier. Adv. Funct. Mater. 2024, 35, 2413865.
29. Xu, Y.; Wang, Y.; Dong, N.; et al. Novel polyimide binder for achieving high-rate capability and long-term cycling stability of LiNi0.8Co0.1Mn0.1O2 cathode via constructing polar and micro-branched crosslinking network structure. J. Energy. Chem. 2023, 76, 19-31.
30. Yu, M.; Wang, Y.; Wang, Z.; et al. Weakened capacity fading of Li-rich cathode via aqueous binder for advanced lithium ion batteries. J. Electrochem. Soc. 2019, 166, A4122-7.
31. Watanabe, T.; Hirai, K.; Ando, F.; et al. Surface double coating of a LiNiaCobAl1-a-bO2 (a > 0.85) cathode with TiOx and Li2CO3 to apply a water-based hybrid polymer binder to Li-ion batteries. RSC. Adv. 2020, 10, 13642-54.
32. Jeong, D.; Kwon, D. S.; Kim, H. J.; Shim, J. Striking a balance: exploring optimal functionalities and composition of highly adhesive and dispersing binders for high-nickel cathodes in lithium-ion batteries. Adv. Energy. Mater. 2023, 13, 2302845.
33. Chen, B.; Zhang, Z.; Wu, C.; et al. Aliphatic polycarbonate-based binders for high-loading cathodes by solvent-free method used in high performance LiFePO4|Li batteries. Materials. (Basel). 2024, 17, 3153.
34. Ko, H.; Kim, M.; Hong, S. Y.; et al. Plasma-assisted mechanochemistry to covalently bond ion-conducting polymers to Ni-rich cathode materials for improved cyclic stability and rate capability. ACS. Appl. Energy. Mater. 2022, 5, 4808-16.
35. Wu, Q.; Ha, S.; Prakash, J.; Dees, D. W.; Lu, W. Investigations on high energy lithium-ion batteries with aqueous binder. Electrochim. Acta. 2013, 114, 1-6.
36. Jang, J.; Ahn, J.; Ahn, J.; et al. A fluorine-free binder with organic-inorganic crosslinked networks enabling structural stability of Ni‐rich layered cathodes in lithium-ion batteries. Adv. Funct. Mater. 2024, 34, 2410866.
37. Watanabe, T.; Yokokawa, T.; Yamada, M.; et al. Surface coating of a LiNixCoyAl1-x-yO2 (x > 0.85) cathode with Li3PO4 for applying a water-based hybrid polymer binder during Li-ion battery preparation. RSC. Adv. 2021, 11, 37150-61.
38. Das, P.; Elizalde-Segovia, R.; Zayat, B.; et al. Enhancing the ionic conductivity of poly(3,4-propylenedioxythiophenes) with oligoether side chains for use as conductive cathode binders in lithium-ion batteries. Chem. Mater. 2022, 34, 2672-86.
39. Lv, M.; Zhao, R.; Hu, Z.; et al. Binder design strategies for cathode materials in advanced secondary batteries. Energy. Environ. Sci. 2024, 17, 4871-906.
40. Nam, M. G.; Moon, J.; Kim, M.; et al. p-phenylenediamine-bridged binder-electrolyte-unified supramolecules for versatile lithium secondary batteries. Adv. Mater. 2024, 36, e2304803.
41. Wu, F.; Li, W.; Chen, L.; et al. Polyacrylonitrile-polyvinylidene fluoride as high-performance composite binder for layered Li-rich oxides. J. Power. Sources. 2017, 359, 226-33.
42. Hawley, W. B.; Parejiya, A.; Bai, Y.; Meyer, H. M.; Wood, D. L.; Li, J. Lithium and transition metal dissolution due to aqueous processing in lithium-ion battery cathode active materials. J. Power. Sources. 2020, 466, 228315.
43. Hawley, W. B.; Meyer, H. M.; Li, J. Enabling aqueous processing for LiNi0.80Co0.15Al0.05O2 (NCA)-based lithium-ion battery cathodes using polyacrylic acid. Electrochim. Acta. 2021, 380, 138203.
44. Laszczynski, N.; Von zamory, J.; Kalhoff, J.; Loeffler, N.; Chakravadhanula, V. S. K.; Passerini, S. Improved performance of VOx‐coated Li‐rich NMC electrodes. ChemElectroChem 2015, 2, 1768-73.
45. Yang, J.; Li, P.; Zhong, F.; et al. Suppressing voltage fading of Li‐rich oxide cathode via building a well-protected and partially-protonated surface by polyacrylic acid binder for cycle-stable Li‐ion batteries. Adv. Energy. Mater. 2020, 10, 1904264.
46. Notake, K.; Gunji, T.; Kokubun, H.; et al. The application of a water-based hybrid polymer binder to a high-voltage and high-capacity Li-rich solid-solution cathode and its performance in Li-ion batteries. J. Appl. Electrochem. 2016, 46, 267-78.
47. Zhang, G.; Qiu, B.; Xia, Y.; et al. Double-helix-superstructure aqueous binder to boost excellent electrochemical performance in Li-rich layered oxide cathode. J. Power. Sources. 2019, 420, 29-37.
48. Zhou, C. C.; Su, Z.; Gao, X. L.; Cao, R.; Yang, S. C.; Liu, X. H. Ultra‐high‐energy lithium‐ion batteries enabled by aligned structured thick electrode design. Rare. Metals. 2021, 41, 14-20.
49. Pham, H. Q.; Lee, J.; Jung, H. M.; Song, S. Non-flammable LiNi0.8Co0.1Mn0.1O2 cathode via functional binder; stabilizing high-voltage interface and performance for safer and high-energy lithium rechargeable batteries. Electrochim. Acta. 2019, 317, 711-21.
50. Huang, X.; You, H.; Yan, X.; et al. Environmentally sustainable lithium-ion battery cathode binders based on cellulose nanocrystals. J. Mater. Chem. A. 2024, 12, 33015-22.
51. Yin, Z.; Zhang, T.; Zhang, S.; et al. Understanding the role of water-soluble guar gum binder in reducing capacity fading and voltage decay of Li-rich cathode for Li-ion batteries. Electrochim. Acta. 2020, 351, 136401.
52. Hwa, Y.; Frischmann, P. D.; Helms, B. A.; Cairns, E. J. Aqueous-processable redox-active supramolecular polymer binders for advanced lithium/sulfur cells. Chem. Mater. 2018, 30, 685-91.
53. Qiu, J.; Wu, S.; Yang, Y.; et al. Aqueous supramolecular binder for a lithium-sulfur battery with flame-retardant property. ACS. Appl. Mater. Interfaces. 2021, 13, 55092-101.
54. Hu, L.; Zhang, X.; Zhao, P.; et al. Gradient H-bonding binder enables stable high-areal-capacity Si-based anodes in pouch cells. Adv. Mater. 2021, 33, e2104416.
55. Li, Z.; Wu, G.; Yang, Y.; et al. An ion‐conductive grafted polymeric binder with practical loading for silicon anode with high interfacial stability in lithium-ion batteries. Adv. Energy. Mater. 2022, 12, 2201197.
56. Liu, H.; Chen, T.; Xu, Z.; Liu, Z.; Yang, J.; Chen, J. High-safety and long-life silicon-based lithium-ion batteries via a multifunctional binder. ACS. Appl. Mater. Interfaces. 2020, 12, 54842-50.
57. Chang, B.; Kim, J.; Cho, Y.; et al. Highly elastic binder for improved cyclability of nickel-rich layered cathode materials in lithium-ion batteries. Adv. Energy. Mater. 2020, 10, 2001069.
58. Wang, Y.; Dong, N.; Liu, B.; et al. Enhanced electrochemical performance of the LiNi0.8Co0.1Mn0.1O2 cathode via in-situ nanoscale surface modification with poly(imide-siloxane) binder. Chem. Eng. J. 2022, 450, 137959.
59. Liu, T.; Chu, Q.; Yan, C.; Zhang, S.; Lin, Z.; Lu, J. Interweaving 3D network binder for high-areal-capacity Si anode through combined hard and soft polymers. Adv. Energy. Mater. 2018, 9, 1802645.
60. Qi, K.; Wang, Y.; Dong, N.; et al. Novel polyimide binders integrated with soft and hard functional segments ensuring long-term high-voltage operating stability of high-energy NCM811 lithium-ion batteries up to 4.5 V. Appl. Energy. 2022, 320, 119282.
61. Yang, J.; Shi, M.; Wu, W.; et al. Cross-linked siloxane-based copolymer binder with combined hardness and softness for stable silicon anodes in Li-ion batteries. J. Mater. Sci. 2021, 56, 20126-37.
62. Jiao, X.; Yin, J.; Xu, X.; et al. Highly energy-dissipative, fast self-healing binder for stable Si anode in lithium-ion batteries. Adv. Funct. Mater. 2020, 31, 2005699.
63. Li, J.; Zhang, B.; Yuan, X.; et al. Benzene ring structural design strategy toward well-balanced thermal and electrical properties in epoxy dielectric polymers. Compos. Part. B:. Eng. 2025, 297, 112308.
64. Li, T.; Chai, S.; Liu, B.; Zhao, C.; Li, H. All‐carbon backbone aromatic polymers for proton exchange membranes. J. Polym. Sci. 2023, 61, 2796-814.
65. Mu, P.; Zhang, H.; Jiang, H.; et al. Bioinspired antiaging binder additive addressing the challenge of chemical degradation of electrolyte at cathode/electrolyte interphase. J. Am. Chem. Soc. 2021, 143, 18041-51.
66. Liu, Z.; Dong, T.; Mu, P.; Zhang, H.; Liu, W.; Cui, G. Interfacial chemistry of vinylphenol-grafted PVDF binder ensuring compatible cathode interphase for lithium batteries. Chem. Eng. J. 2022, 446, 136798.
67. Naga, N.; Inose, D.; Ishida, T.; Kubota, K.; Nageh, H.; Nakano, T. Synthesis of polymer networks by means of addition reactions of tri-amine and poly(ethylene glycol) diacrylate or diglycidyl ether compounds. Polym. Bull. 2020, 78, 2745-63.
68. Talakesh, M. M.; Sadeghi, M.; Chenar, M. P.; Khosravi, A. Gas separation properties of poly(ethylene glycol)/poly(tetramethylene glycol) based polyurethane membranes. J. Membr. Sci. 2012, 415-416, 469-77.
69. Chang, Z.; Zhang, M.; Hudson, A. G.; et al. Synthesis and properties of segmented polyurethanes with triptycene units in the hard segment. Polymer 2013, 54, 6910-7.
70. Bocharova, V.; Sokolov, A. P. Perspectives for polymer electrolytes: a view from fundamentals of ionic conductivity. Macromolecules 2020, 53, 4141-57.
71. Costa, C. M.; Merazzo, K. J.; Gonçalves, R.; Amos, C.; Lanceros-Méndez, S. Magnetically active lithium-ion batteries towards battery performance improvement. iScience 2021, 24, 102691.
72. Zhang, S.; Gu, H.; Pan, H.; et al. A novel strategy to suppress capacity and voltage fading of Li‐ and Mn‐rich layered oxide cathode material for lithium-ion batteries. Adv. Energy. Mater. 2016, 7, 1601066.
73. Kim, Y. M.; Murugesan, K.; Chang, Y. Y.; Kim, E. J.; Chang, Y. S. Degradation of polybrominated diphenyl ethers by a sequential treatment with nanoscale zero valent iron and aerobic biodegradation. J. Chem. Technol. Biotechnol. 2011, 87, 216-24.
74. Arias, K. S.; Climent, M. J.; Corma, A.; Iborra, S. Biomass-derived chemicals: synthesis of biodegradable surfactant ether molecules from hydroxymethylfurfural. ChemSusChem 2014, 7, 210-20.
75. Tang, S.; Baker, G. A.; Zhao, H. Ether- and alcohol-functionalized task-specific ionic liquids: attractive properties and applications. Chem. Soc. Rev. 2012, 41, 4030-66.
76. Pedaballi, S.; Li, C. Aqueous processed Ni-rich Li(Ni0.8Co0.1Mn0.1)O2 cathodes along with water-based binders and a carbon fabric as 3-D conductive host. J. Electrochem. Soc. 2021, 168, 120538.
77. Wang, L.; Ma, Y.; Li, Q.; et al. 1,3,6-Hexanetricarbonitrile as electrolyte additive for enhancing electrochemical performance of high voltage Li-rich layered oxide cathode. J. Power. Sources. 2017, 361, 227-36.
78. Lee, S. H.; Hwang, J. Y.; Park, S. J.; Park, G. T.; Sun, Y. K. Adiponitrile (C6H8N2): a new Bi-functional additive for high-performance Li‐metal batteries. Adv. Funct. Mater. 2019, 29, 1902496.
79. Li, S.; Zhao, D.; Wang, P.; Cui, X.; Tang, F. Electrochemical effect and mechanism of adiponitrile additive for high-voltage electrolyte. Electrochim. Acta. 2016, 222, 668-77.
80. Jin, M.; Li, B.; Hu, L.; Zhao, P.; Zhang, Q.; Song, J. Functional copolymer binder for nickel-rich cathode with exceptional cycling stability at high temperature through coordination interaction. J. Energy. Chem. 2021, 60, 156-61.
81. Xi, Y.; Liu, Y.; Zhang, D.; Jin, S.; Zhang, R.; Jin, M. Comparative study of the electrochemical performance of LiNi0.5Co0.2Mn0.3O2 and LiNi0.8Co0.1Mn0.1O2 cathode materials for lithium ion batteries. Solid. State. Ionics. 2018, 327, 27-31.
82. Li, W.; Wu, K.; Feng, H.; et al. Atomic layer deposition of ultrafine Pd nanoparticles for enhancing the rate capability of LiNi0.8Co0.1Mn0.1O2 cathode. Tungsten 2022, 4, 346-55.
83. Gan, Z.; Hu, G.; Peng, Z.; Cao, Y.; Tong, H.; Du, K. Surface modification of LiNi0.8Co0.1Mn0.1O2 by WO3 as a cathode material for LIB. Appl. Surf. Sci. 2019, 481, 1228-38.
84. Chen, S.; He, T.; Su, Y.; et al. Ni-rich LiNi0.8CO0.1Mn0.1O2 oxide coated by dual-conductive layers as high performance cathode material for lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2017, 9, 29732-43.
85. Wang, Y.; Dong, N.; Liu, B.; Tian, G.; Qi, S.; Wu, D. Self-adaptive gel poly(imide-siloxane) binder ensuring stable cathode-electrolyte interface for achieving high-performance NCM811 cathode in lithium-ion batteries. Energy. Storage. Mater. 2023, 56, 621-30.
86. Xu, Y.; Chafi, F. Z.; Chen, P.; et al. More than just a binder: versatile block copolymer enhances the electrochemical performance of a nickel-rich cathode. ACS. Appl. Polym. Mater. 2023, 5, 4654-63.
87. Li, R. Bai, C.-J.; Liu, H.; et al. New insights into the mechanism of enhanced performance of Li[Ni0.8Co0.1Mn0.1]O2 with a polyacrylic acid-modified binder. ACS. Appl. Mater. Interfaces. 2021, 13, 10064-70.
88. Wandt, J.; Freiberg, A. T.; Ogrodnik, A.; Gasteiger, H. A. Singlet oxygen evolution from layered transition metal oxide cathode materials and its implications for lithium-ion batteries. Mater. Today. 2018, 21, 825-33.
89. Freiberg, A. T. S.; Roos, M. K.; Wandt, J.; de Vivie-Riedle, R.; Gasteiger, H. A. Singlet oxygen reactivity with carbonate solvents used for Li-ion battery electrolytes. J. Phys. Chem. A. 2018, 122, 8828-39.
90. Hatsukade, T.; Schiele, A.; Hartmann, P.; Brezesinski, T.; Janek, J. Origin of carbon dioxide evolved during cycling of nickel-rich layered NCM cathodes. ACS. Appl. Mater. Interfaces. 2018, 10, 38892-9.
91. Rinkel, B. L. D.; Hall, D. S.; Temprano, I.; Grey, C. P. Electrolyte oxidation pathways in lithium-ion batteries. J. Am. Chem. Soc. 2020, 142, 15058-74.
92. Kim, N. Y.; Moon, J.; Ryou, M. H.; et al. Amphiphilic bottlebrush polymeric binders for high-mass-loading cathodes in lithium-ion batteries. Adv. Energy. Mater. 2021, 12, 2102109.
93. Xiao, P.; Shi, T.; Huang, W.; Ceder, G. Understanding surface densified phases in Ni-rich layered compounds. ACS. Energy. Lett. 2019, 4, 811-8.
94. Kleiner, K.; Dixon, D.; Jakes, P.; et al. Fatigue of LiNi0.8Co0.15Al0.05O2 in commercial Li ion batteries. J. Power. Sources. 2015, 273, 70-82.
95. Zhuang, G. V.; Chen, G.; Shim, J.; Song, X.; Ross, P. N.; Richardson, T. J. Li2CO3 in LiNi0.8Co0.15Al0.05O2 cathodes and its effects on capacity and power. J. Power. Sources. 2004, 134, 293-7.
96. Kim, J.; Lee, H.; Cha, H.; Yoon, M.; Park, M.; Cho, J. Prospect and reality of Ni‐rich cathode for commercialization. Adv. Energy. Mater. 2017, 8, 1702028.
97. Vadivel, S.; Sawangphruk, M. Prelithiated perfluoro-ionomer as an alternative binder for the state-of-the-art Ni-rich LiNi0.8Co0.15Al0.05O2 cathode of next-generation lithium-ion batteries. J. Mater. Chem. A. 2020, 8, 20714-24.
98. Das, P.; Zayat, B.; Wei, Q.; et al. Dihexyl-Substituted Poly(3,4-Propylenedioxythiophene) as a dual ionic and electronic conductive cathode binder for lithium-ion batteries. Chem. Mater. 2020, 32, 9176-89.
99. Tanabe, T.; Liu, Y.; Miyamoto, K.; et al. Synthesis of water-resistant thin TiOx layer-coated high-voltage and high-capacity LiNiaCobAl1-a-bO2 (a > 0.85) cathode and its cathode performance to apply a water-based hybrid polymer binder to Li-Ion batteries. Electrochim. Acta. 2017, 258, 1348-55.
100. Nayak, P. K.; Erickson, E. M.; Schipper, F.; et al. Review on challenges and recent advances in the electrochemical performance of high capacity Li‐ and Mn‐rich cathode materials for Li‐Ion batteries. Adv. Energy. Mater. 2017, 8, 1702397.
101. Yang, J.; Xia, Y. Suppressing the phase transition of the layered Ni-rich oxide cathode during high-voltage cycling by introducing low-content Li2MnO3. ACS. Appl. Mater. Interfaces. 2016, 8, 1297-308.
102. Pedaballi, S.; Li, C. Effects of surface modification and organic binder type on cell performance of water-processed Ni-rich Li(Ni0.8Co0.1Mn0.1)O2 cathodes. J. Power. Sources. 2020, 472, 228552.
103. Yan, W.; Xie, Y.; Jiang, J.; et al. Enhanced rate performance of Al-doped Li-rich layered cathode material via nucleation and post-solvothermal method. ACS. Sustainable. Chem. Eng. 2018, 6, 4625-32.
104. Pan, H.; Zhang, S.; Chen, J.; et al. Li- and Mn-rich layered oxide cathode materials for lithium-ion batteries: a review from fundamentals to research progress and applications. Mol. Syst. Des. Eng. 2018, 3, 748-803.
105. Zhang, S. J.; Deng, Y. P.; Wu, Q. H.; et al. Sodium-alginate-based binders for lithium-rich cathode materials in lithium-ion batteries to suppress voltage and capacity fading. ChemElectroChem 2018, 5, 1321-9.
106. Pham, H. Q.; Hwang, E.; Kwon, Y.; Song, S. Understanding the interfacial phenomena of a 4.7 V and 55 °C Li-ion battery with Li-rich layered oxide cathode and graphite anode and its correlation to high-energy cycling performance. J. Power. Sources. 2016, 323, 220-30.
107. Iturrondobeitia, A.; Kvasha, A.; Lopez Del Amo, J.; et al. A comparative study of aqueous and organic processed Li1.2Ni0.2Mn0.6O2 Li-rich cathode materials for advanced lithium-ion batteries. Electrochim. Acta. 2017, 247, 420-5.
108. Chen, H.; Ling, M.; Hencz, L.; et al. Exploring chemical, mechanical, and electrical functionalities of binders for advanced energy-storage devices. Chem. Rev. 2018, 118, 8936-82.
109. Liu, J.; Galpaya, D. G. D.; Yan, L.; et al. Exploiting a robust biopolymer network binder for an ultrahigh-areal-capacity Li-S battery. Energy. Environ. Sci. 2017, 10, 750-5.
110. Liu, J.; Zhang, Q.; Zhang, T.; Li, J. T.; Huang, L.; Sun, S. G. A robust ion-conductive biopolymer as a binder for Si anodes of lithium-ion batteries. Adv. Funct. Mater. 2015, 25, 3599-605.
111. Kong, D.; Hu, J.; Chen, Z.; et al. Ti-gradient doping to stabilize layered surface structure for high performance high‐Ni oxide cathode of Li‐ion battery. Adv. Energy. Mater. 2019, 9, 1901756.
112. Zhang, Q.; Liu, K.; Li, C.; et al. The surface triple-coupling on single crystalline cathode for lithium ion batteries. Nano. Energy. 2021, 86, 106096.
113. Qiu, L.; Xiang, W.; Tian, W.; et al. Polyanion and cation co-doping stabilized Ni-rich Ni-Co-Al material as cathode with enhanced electrochemical performance for Li-ion battery. Nano. Energy. 2019, 63, 103818.
114. Liu, X.; Wang, D.; Liu, G.; et al. Distinct charge dynamics in battery electrodes revealed by in situ and operando soft X-ray spectroscopy. Nat. Commun. 2013, 4, 2568.
115. Ulvestad, A.; Singer, A.; Cho, H. M.; et al. Single particle nanomechanics in operando batteries via lensless strain mapping. Nano. Lett. 2014, 14, 5123-7.
116. Xu, J.; Sun, M.; Qiao, R.; et al. Elucidating anionic oxygen activity in lithium-rich layered oxides. Nat. Commun. 2018, 9, 947.
117. Song, J.; Li, B.; Chen, Y.; et al. A high-performance Li-Mn-O Li-rich cathode material with rhombohedral symmetry via intralayer Li/Mn disordering. Adv. Mater. 2020, 32, e2000190.
118. Liu, S.; Liu, Z.; Shen, X.; et al. Surface doping to enhance structural integrity and performance of Li‐rich layered oxide. Adv. Energy. Mater. 2018, 8, 1802105.
119. Cui, C.; Ji, X.; Wang, P.; et al. Integrating multiredox centers into one framework for high-performance organic Li-ion battery cathodes. ACS. Energy. Lett. 2019, 5, 224-31.
120. Yang, J.; Xiong, P.; Shi, Y.; et al. Rational molecular design of benzoquinone-derived cathode materials for high-performance lithium-ion batteries. Adv. Funct. Mater. 2020, 30, 1909597.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].