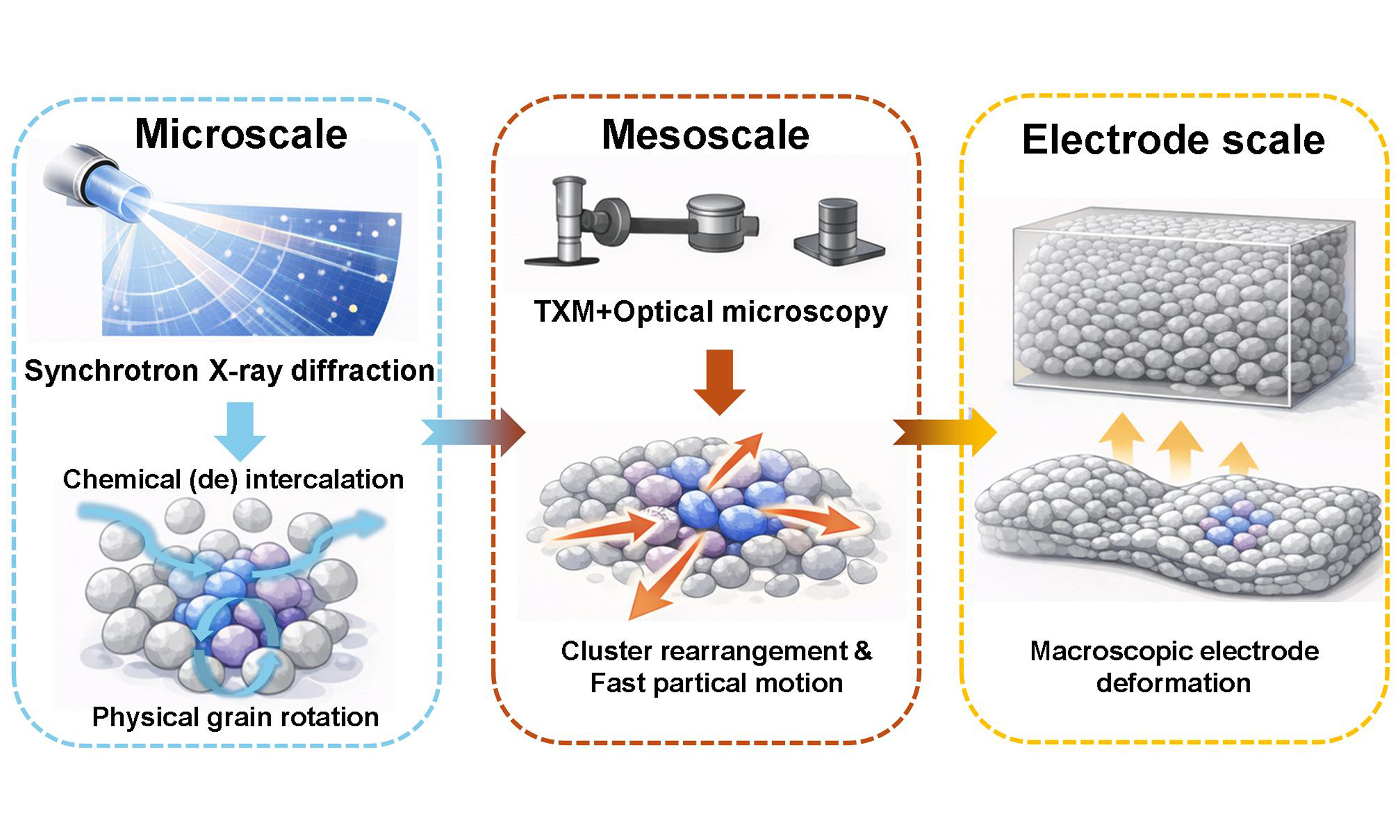
How electrode microstructure evolves in Li-ion batteries
Keywords
Intercalation-based electrodes form the foundation of modern lithium-ion battery technologies, yet their long-term reliability is fundamentally constrained by chemomechanical degradation[1]. Electrochemical cycling induces heterogeneous structural changes within electrodes, leading to cracking, swelling, and irreversible deformation across multiple length scales[2]. Although heterogeneous structural responses under static conditions have been extensively characterized, revealing significant inter- and intra-particle heterogeneity[3,4], how these particle-scale electrochemical differences collectively evolve into electrode-scale strain and macroscopic deformation remains a critical unresolved issue[5]. Writing in Science, Sun et al. provide compelling real-time evidence that electrode deformation emerges from a cascading chemomechanical process driven by asynchronous electrochemical evolution across particles and grains, thereby redefining the origin of mechanical fatigue in intercalation cathodes[6].
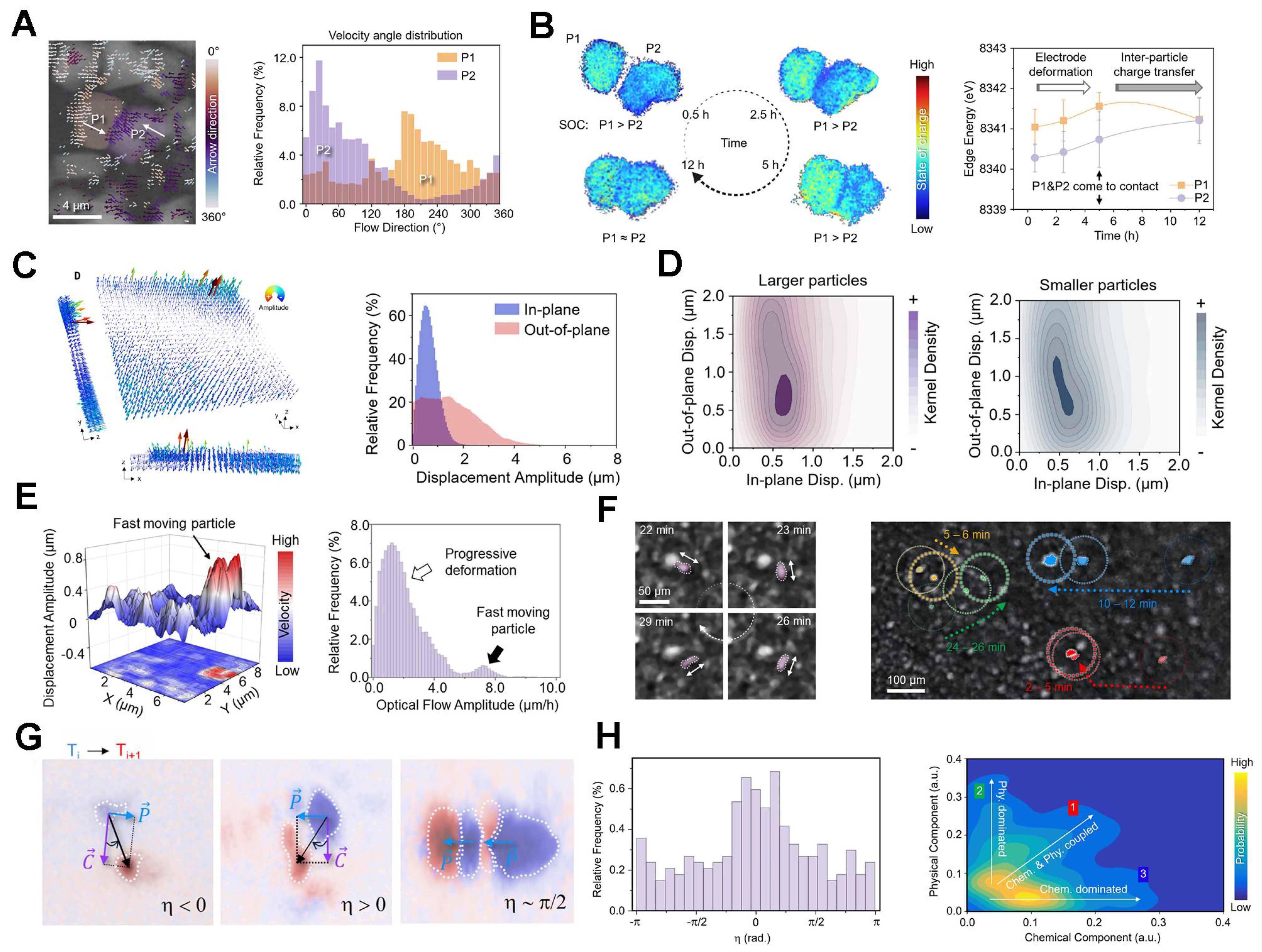
Using multimodal operando microscopy methods, the authors directly visualize how heterogeneous state-of-charge (SOC) evolution generates internal stress that progressively reorganizes electrode microstructures. In a free-standing layered LiNi0.6Mn0.2Co0.2O2 (NMC) cathode, operando spectro-transmission X-ray microscopy (TXM) reveals that neighboring particles undergo coordinated translational and rotational motion during slow charging, gradually moving into physical contact [Figure 1A]. Crucially, despite concurrent delithiation, adjacent particles retain distinct SOCs for extended periods, demonstrating persistent electrochemical asynchronicity at the particle level. These SOC disparities diminish only after physical contact is established, consistent with microscale charge redistribution and redox coupling [Figure 1B]. These observations establish that electrode strain is not merely a passive consequence of uniform lattice expansion, but an emergent response to asynchronous electrochemical evolution among interacting particles.
Figure 1. (A) Optical flow analysis of particle motion and probability distribution of local velocity angles in particles P1 and P2; (B) TXM-based SOC mapping using Ni K-edge energy, along with the evolution of the mean value and standard deviation of Ni K-edge energy in particles P1 and P2; (C) Visualization of electrode strain and probability distributions of the in-plane and out-of-plane components of the deformation field; (D) Kernel density distributions for larger and smaller particles; (E) Amplitude of particle lateral displacement and probability distributions of displacement amplitudes; (F) Localized particle rotation observed via in situ optical microscopy, along with optical imaging of randomly occurring irregular particle movements; (G) Selected Bragg peaks exhibiting different chemomechanical behaviors during cell cycling; (H) Probability distribution of η for all captured Bragg peaks and scatter plot of Bragg peak displacements. Adapted with permission Ref.[6]. Copyright 2025, American Association for the Advancement of Science. TXM: Transmission X-ray microscopy; SOC: state-of-charge.
To assess whether such behavior persists under practical cell conditions, the authors extend their analysis to a compressed single-layer pouch cell using in situ X-ray laminography[7]. Three-dimensional tracking of tens of thousands of particles reveals that substantial electrode strain develops early in the first charging cycle, well before the activation of oxygen redox reactions in Li- and Mn-rich cathodes. Optical-flow analysis uncovers pronounced spatial heterogeneity in the deformation field, with dominant out-of-plane displacement near electrode edges and comparatively weaker in-plane motion toward the interior [Figure 1C]. Particle size-resolved analysis further shows that smaller particles exhibit systematically larger out-of-plane displacements than their larger counterparts, indicating a size-dependent mechanical response, likely arising from faster electrochemical kinetics and enhanced local strain generation [Figure 1D]. Together, these observations highlight the critical role of mechanical mismatch among different cell components and particle populations, demonstrating that electrode deformation is governed not only by active material chemistry but also by microstructural heterogeneity and the mechanical architecture of the full cell stack.
Beyond these gradual deformation modes, Sun et al. identify a previously underappreciated class of sparse, rapid particle-motion events[6]. Statistical analysis reveals that while most particles undergo slow, continuous displacement during charging, a small subset exhibits abrupt, high-velocity rotations and translations [Figure 1E]. Operando optical microscopy confirms that these events correspond to loosely constrained particles that experience sudden mechanical perturbations upon electrochemical activation [Figure 1F]. Although rare, such events represent localized strain-release processes that contribute disproportionately to the heterogeneous evolution of electrode morphology, underscoring the inherently intermittent nature of stress relaxation in composite electrodes.
To elucidate the microscopic origin of these strain dynamics, the authors employ grain-resolved synchrotron X-ray diffraction to decouple chemical and mechanical responses at the crystal-grain level[8]. By tracking Bragg peak trajectories, they distinguish lattice-spacing changes associated with Li-ion (de)intercalation from tangential peak motion arising from physical grain rotation [Figure 1G]. Statistical analysis of hundreds of grains reveals a broad spectrum of behaviors, including chemically dominated, mechanically dominated, and strongly coupled chemomechanical responses. Notably, a substantial fraction of grains undergoes rotation-dominated motion despite being electrochemically inactive, indicating that stress propagates from chemically active regions into neighboring inactive domains [Figure 1H]. This observation establishes a hierarchical strain-transmission network, in which chemically active grains act as primary stress sources while inactive grains participate by accommodating and relaying the transmitted strain.
In summary, this study elucidates the multiscale evolution of “strain cascading” through a multimodal characterization framework. At the microscopic scale, synchrotron X-ray diffraction enables grain-resolved imaging under operating conditions, revealing a fundamental coupling between chemical intercalation/deintercalation and physical grain rotation. This demonstrates that strain originates from asynchrony in lattice breathing. These grains serve as primary sources of strain, where anisotropic lattice expansion generates local stress fields that propagate to neighboring grains, thereby forming mesoscale propagation. Transmission X-ray microscopy, combined with optical microscopy and optical flow analysis under operating conditions, reveals collective cluster rearrangement and rapid random particle motion driven by interparticle charge state heterogeneity. Ultimately, three-dimensional in situ X-ray tomography confirms that the amplification of these microscopic events governs macroscopic electrode deformation, manifesting as significant out-of-plane displacement and overall wrinkling. Collectively, these findings reveal a hierarchical trajectory of strain propagation - from individual chemically triggered points to large-scale mechanical degradation. This strain cascade phenomenon resembles dislocation avalanches in crystalline plasticity, shear band propagation in metallic glasses, and domino-like stress transfer in granular media, involving localized activation nodes, threshold-dependent propagation, heterogeneous spatial distribution, and collective asynchronous responses. However, unlike purely mechanical avalanche phenomena, the strain cascade here is jointly driven by electrochemical heterogeneity and mechanical responses, introducing asynchronous chemical activation as a triggering mechanism.
Importantly, this framework redefines mechanical fatigue as a collective phenomenon that does not require uniform participation of all active materials. Even electrochemically inactive domains contribute to degradation by serving as conduits for stress transmission. These insights carry important implications for battery design, indicating that strategies focused solely on material composition or suppression of lattice expansion may be insufficient to ensure durable operation. Instead, electrodes must be engineered as adaptive, stress-resilient systems, where microstructural design promotes controlled particle connectivity, tunable elasticity, and effective dissipation of transmitted stress. By revealing how local electrochemical asynchronicity drives hierarchical mechanical responses, Sun et al. provide a mechanistic foundation for designing next-generation intercalation cathodes with improved structural stability and extended cycle life[6].
DECLARATIONS
Authors’ contributions
Drafted the manuscript: Yan, W.
Revised and rewrote sections of the manuscript: Li, Y.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the Wallonia Government under the “Plan de Relance” program (2310153-BatFactory) and the China Scholarship Council (No. 202406240020).
Conflicts of interest
Both authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Park, G. T.; Park, N. Y.; Ryu, J. H.; et al. Zero-strain Mn-rich layered cathode for sustainable and high-energy next-generation batteries. Nat Energy 2025, 10, 1215-25.
2. Lu, X.; Bertei, A.; Finegan, D. P.; et al. 3D microstructure design of lithium-ion battery electrodes assisted by X-ray nano-computed tomography and modelling. Nat Commun 2020, 11, 2079.
3. Xiong, J. F.; Wang, M. Y.; Huang, R. B.; Tian, J. H.; Li, J. F.; Tian, Z. Q. Recent progress of in-situ/operando characterization approaches of zinc-air batteries. Chem Synth 2024, 4, 17.
4. Zhang, Y.; Hao, S.; Pei, F.; et al. Operando chemo-mechanical evolution in LiNi0.8Co0.1Mn0.1O2 cathodes. Natl Sci Rev 2024, 11, nwae254.
5. Liu, H.; Strobridge, F. C.; Borkiewicz, O. J.; et al. Capturing metastable structures during high-rate cycling of LiFePO4 nanoparticle electrodes. Science 2014, 344, 1252817.
6. Sun, T.; Qian, G.; Fang, R.; et al. Electrode strain dynamics in layered intercalation battery cathodes. Science 2025, 390, 1272-7.
7. Zan, G.; Qian, G.; Gul, S.; et al. In situ visualization of multicomponents coevolution in a battery pouch cell. Proc Natl Aca Sci U S A 2022, 119, e2203199119.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].