Sulfur-defect-rich Bi2S3/ZnIn2S4 Z-scheme heterojunction for highly efficient H2O2 photosynthesis in pure water
Abstract
Photocatalytic conversion of O2 and H2O provides a green and low-cost route for H2O2 synthesis; however, most reaction systems involve sacrificial agents, and achieving efficient photosynthesis of H2O2 in pure water remains a challenge. In this work, a Z-scheme Bi2S3/ZnIn2S4 heterojunction with rich sulfur defects was prepared by a one-step hydrothermal method. The combination of Bi2S3 with ZnIn2S4 greatly enhanced visible-light absorption. The intimate heterojunction interface bonded through sulfur bridge efficiently promoted the separation and migration of photogenerated carriers. Moreover, the enlarged specific surface area, the existence of sulfur defects and the increase of surface hydrophobicity facilitated the oxygen reduction reaction. As a result, the H2O2 production rate of the Bi2S3/ZnIn2S4 heterojunction in pure water under visible light reached 1,634 μmol·g-1·h-1, which was 5.3 and 43.0 times that of ZnIn2S4 and Bi2S3, respectively. This work provides new ideas for the construction of novel heterojunction photocatalysts for H2O2 production.
Keywords
INTRODUCTION
Hydrogen peroxide (H2O2), a green oxidizing agent, is widely used in fields such as food, medicine and chemical synthesis. In recent years, the demand for H2O2 has continued to increase and is projected to reach 5.7 million tons annually by 2027[1,2]. Currently, about 98% of industrial H2O2 is produced via the anthraquinone process. However, this method suffers from high energy consumption, a complex process, and severe environmental pollution, which are inconsistent with the principles of green chemistry[3]. Photocatalytic technology provides a green and cost-effective alternative for H2O2 production, driven by solar energy and utilizing oxygen and water as feedstocks[4,5].
Photocatalytic H2O2 production can proceed via two routes: oxygen reduction reaction (ORR) and water oxidation reaction (WOR), with the ORR pathway being predominant[6-9]. Depending on the photocatalyst, two types of ORR are possible: (i) a two-step single-electron ORR [O2 + e- → ·O2-, ·O2- + e- + 2H+ → H2O2,
Various photocatalysts such as metal oxides, metal sulfides, polymeric carbon nitrides and metal organic frameworks have been applied in the H2O2 photosynthesis. Among them, ZnIn2S4 (ZIS) shows a great potential because of its visible-light response, suitable energy band positions and unique 2D layered structure[14-17]. However, pure ZIS usually suffers from severe charge recombination, insufficient active sites and photocorrosion problem, resulting in unsatisfactory photocatalytic activity and stability. In order to improve the photocatalytic activity, some strategies including defect engineering and heterojunction construction, etc. were proposed to modify ZIS[18,19]. For example, sulfur defects and zinc defects were introduced into ZIS lattice to improve the carrier transfer efficiency and also serve as active sites for the reaction[20-22]. By constructing heterojunctions with other semiconductors, the spatial separation of electrons and holes was realized and the charge separation efficiency was improved[23]. An intimate heterojunction interface is of great importance to the transport of photogenerated charges, and thus affects the photocatalytic performance. Recent studies demonstrated that constructing a heterojunction by combining two semiconductors containing a same element facilitates the formation of intimate interface[24]. For example, Che et al. prepared a Zn3In2S6@CdS heterojunction with matched interfacial lattices and S covalent bonding bridges, which provide robust charge transfer channels[25]. Benefiting from the rapid interfacial charge separation and transfer, the heterojunction enables efficient H2 production coupled with H2O2 photosynthesis. Yang et al. developed a In2O3/In2S3 heterojunction for photocatalytic CO2 reduction[26]. The tight interface between the two phases ensures high-speed transport of electrons, contributing to the superior CO2 reduction performance.
Bismuth (Bi)-based materials such as Bi-metals, Bi-multicomponent oxides, Bi-oxyhalogens, Bi-sulfides, etc. have triggered much attention as photocatalysts because of their suitable band gap, unique crystal structure, diverse composition, abundant atomic coordination and superior physicochemical properties[27]. Most Bi-based materials can be stimulated by visible and near-infrared light. Bi-based materials also feature a 2D layered structure with a large surface area[28,29]. They are widely used in the field of photocatalysis including CO2 reduction, water splitting, chemical synthesis and pollutant degradation[30,31]. Among all the Bi-based photocatalysts, Bi2S3 (BS) has attracted significant interest owing to its advantages such as low cost, low toxicity, and good photosensitivity[32]. It has a relatively narrow band gap (Eg = 1.3-1.7 eV) with a response to visible and near-infrared light[33]. Unfortunately, the limited redox potential, the high electron-hole recombination rate and the easy photocorrosion limit its photocatalytic applications[34]. To overcome these limitations, constructing heterojunctions with other semiconductors is a feasible strategy to improve the photocatalytic activity and stability of BS. Since both ZIS and BS contain sulfur, there is a strong possibility of constructing a Bi2S3/ZIS (BZIS) heterojunction with a tight interface through sulfur bonding.
In the present work, a BZIS heterojunction with intimate interface and rich sulfur vacancies was synthesized by a one-step hydrothermal method. The heterojunction exhibits excellent photocatalytic performance for H2O2 production in pure water. The effects of heterojunction construction and sulfur defect introduction on the photocatalytic performance were systematically investigated, and the charge transfer route and the photocatalytic mechanism were proposed.
EXPERIMENTAL
Reagents and materials
All reagents and materials are of analytical grade, and no further purification was required before use. Zinc chloride (ZnCl2, 98%, AR), indium chloride tetrahydrate (InCl3, 99.9%, AR), bismuth nitrate pentahydrate [Bi(NO3)3·5H2O, 98%, ACS], 5,5-dimethyl-1-pyrroline-N-oxide (DMPO, 97%, AR) and thioacetamide (TAA, 99%, ACS) were purchased from Aladdin Biochemical Technology Co., Ltd. Citric acid monohydrate (CA, 99.5%, AR) and potassium iodide (KI, 99%, AR) were purchased from Macklin Biochemical Technology Co., Ltd. Silver nitrate (AgNO3, 99.8%, AR) and p-benzoquinone (BQ, 98%, AR) were purchased from Sinopharm Chemical Reagent Co., Ltd. Potassium biphthalate (C8H5O4K, 98%, AR), triethanolamine (TEOA, 99%, AR) and absolute ethanol (99.7%, AR) were purchased from Tianjin Damao Chemical Preparations Factory. Tert-butyl alcohol (TBA, 99.5%, AR) was purchased from Tianjin Kemiou Chemical Reagent Co., Ltd.
Preparation of photocatalysts
BZIS heterojunctions were synthesized by a one-step hydrothermal method according to a previously reported method with some modifications[35]. For a typical synthesis, 2 mmol of ZnCl2, 4 mmol of InCl3, 1 g of citric acid (CA), 0.1 mmol of Bi(NO3)3·5H2O and 21 mmol of thioacetamide (TAA) were dissolved in
Characterizations
X-ray diffraction (XRD) patterns of the samples were collected using a SmartLab 9 kW X-ray diffractometer (Rigaku Corporation, Japan) with Cu Kα radiation (λ = 1.5406 Å). Structural and morphological characteristics were examined using a scanning electron microscope (SEM, SU8220, Hitachi, Japan) operated at 5.0 kV and a transmission electron microscope (TEM, HT7700 EXALENS, Hitachi, Japan) operated at
Photoelectrochemical performance test
Photoelectrochemical performance was tested using an electrochemical workstation (CHI 760E, Chenhua, China). A 0.5 mol·L-1 Na2SO4 solution was used as electrolyte and a 300 W Xe lamp served as light source. A Pt wire and an Ag/AgCl electrode were employed as the counter and reference electrodes, respectively. The working electrode was prepared by dip-coating a sample film onto a piece of fluorine-doped tin oxide (FTO) glass
Photocatalytic H2O2 production
The photocatalytic H2O2 production performance of the photocatalysts was evaluated in pure water under visible light. In detail, 15 mg of photocatalyst was well dispersed in 30 mL of deionized water by ultrasonication for 15 min. After O2 was bubbled in the solution for 30 min, the reaction began under the irradiation of a 300 W Xe lamp ( > 420 nm). During the reaction of 120 min, 1 mL of the solution was extracted every 30 min. The H2O2 concentration was measured by iodometry. The solution was mixed with
RESULTS AND DISCUSSION
Synthesis and structural characterizations
Figure 1A illustrates the synthesis process of BZIS heterojunction. This one-step hydrothermal approach ensures the intimate interfacial contact between BS and ZIS. The morphology and structure of the samples were observed by SEM and TEM. From the SEM images, ZIS shows nano-flower morphology with a diameter of ~5 µm which is assembled by nanosheets [Figure 1B], while BS exhibits nanorod structure [Figure 1C]. For the BZIS heterojunction, both nanorods and nanosheets can be observed, which are closely interwoven with each other [Figure 1D]. The TEM image provides further evidence that BS nanorods are encapsulated by ZIS nanosheets [Figure 1E]. A high-resolution TEM (HRTEM) image [Figure 1F] shows clear lattice spacings of 0.353 and 0.292 nm, which match well with the (130) plane of BS and the (104) plane of ZIS, respectively, indicating the intimate interfacial contact between the two phases. Elemental mapping images [Figure 1G] reveal that Zn, In, Bi and S elements are homogeneously distributed within the catalyst, confirming the successful preparation of BZIS heterojunction.
Figure 1. (A) Schematic illustration of the synthesis of BZIS heterojunctions; SEM images of (B) ZIS, (C) BS and (D) BZIS-1/20, (E) TEM image and (F) HRTEM image of BZIS-1/20; and (G) elemental mapping images of Zn, In, Bi and S. BZIS: Bi2S3/ZnIn2S4; SEM: scanning electron microscope; ZIS: ZnIn2S4; BS: Bi2S3; TEM: transmission electron microscope; HRTEM: high-resolution transmission electron microscope.
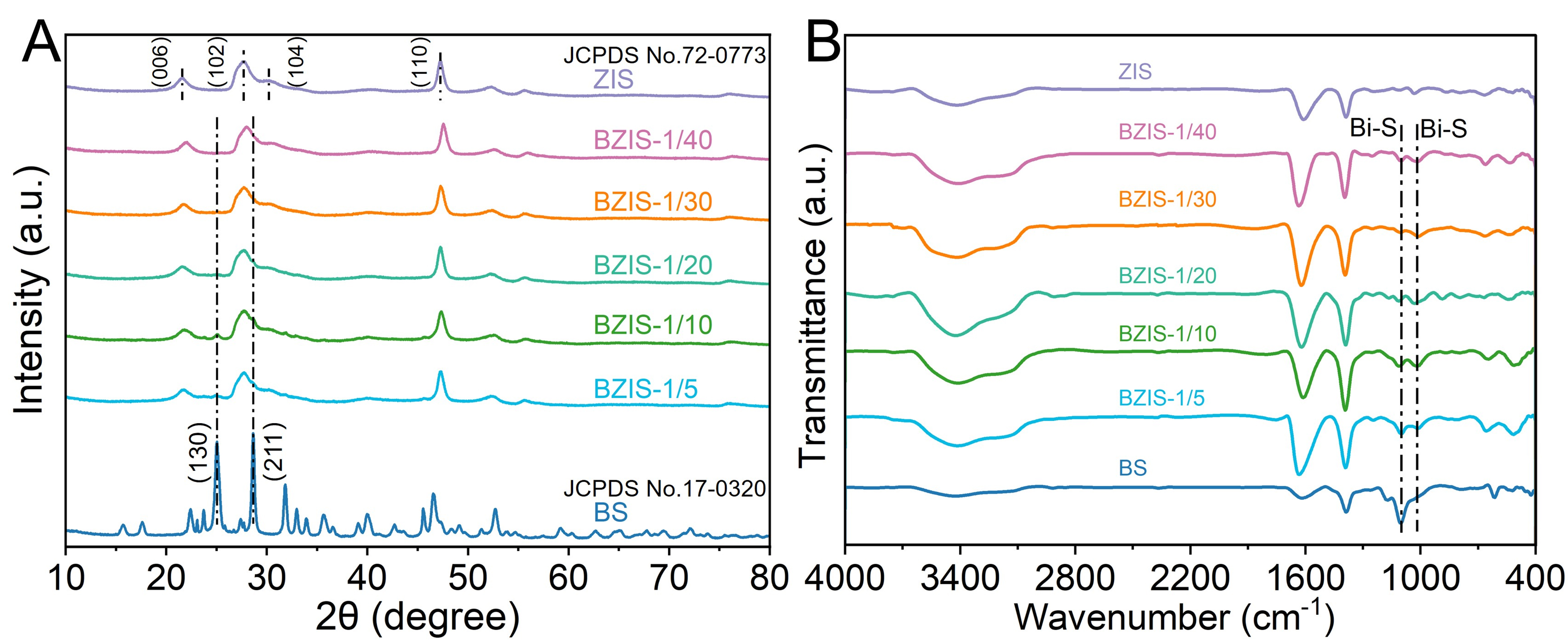
The crystal structures of BS, ZIS, and BZIS heterojunctions were identified by XRD patterns [Figure 2A]. The diffraction peaks of BS and ZIS are indexed to orthorhombic phase Bi2S3 (JCPDS No.17-0320) and hexagonal phase ZIS (JCPDS No.72-0773), respectively[36,37]. For the BZIS heterojunctions, the characteristic diffraction peaks of both BS and ZIS can be observed. The intensities of the diffraction peaks at 24.9° and 28.6° corresponding to the (130) and (211) planes of BS increased with higher BS content, confirming the successful construction of the BZIS heterojunctions. The surface functional groups of the samples were confirmed by FT-IR spectra [Figure 2B]. The peaks located at 1,610 and 3,450 cm-1 can be ascribed to adsorbed water molecules, while the peak at 1,396 cm-1 is attributable to hydroxyl groups[38,39]. The peaks at
Figure 2. (A) XRD patterns and (B) FT-IR spectra of BS, ZIS and BZIS-x. XRD: X-ray diffraction; FT-IR: Fourier transform infrared; BS: Bi2S3; ZIS: ZnIn2S4; BZIS: Bi2S3/ZnIn2S4.
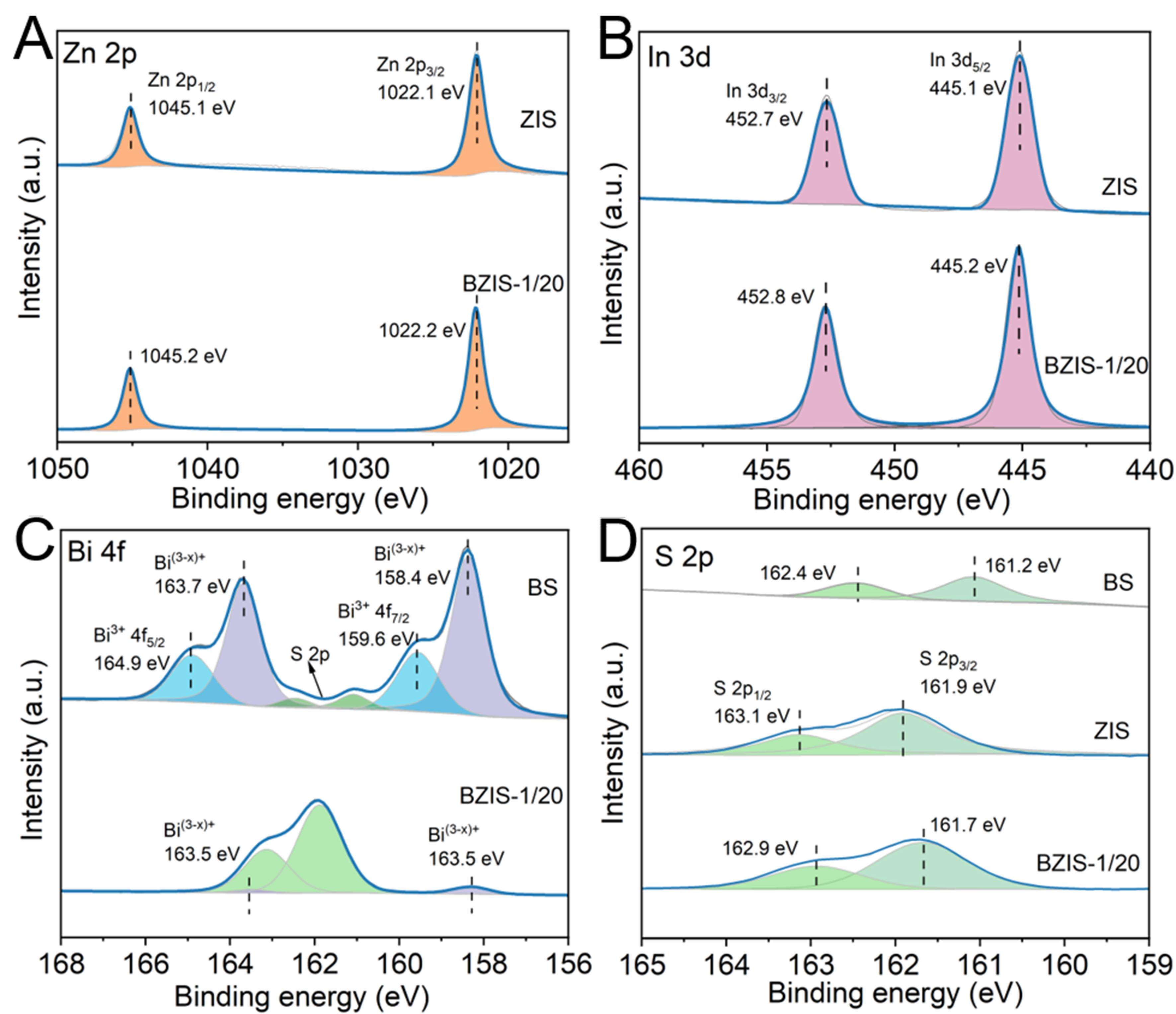
The chemical states of surface elements and the interfacial charge transfer between BS and ZIS were identified by XPS. In the survey XPS spectrum of BZIS-1/20 [Supplementary Figure 2], the peaks corresponding to Zn 2p, In 3d, Bi 4f and S 2p can be observed, confirming the presence of these elements in BZIS. As depicted in Figure 3A, two peaks at 1,045.1 and 1,022.1 eV appear in the Zn 2p spectrum of ZIS, which are attributed to Zn 2p1/2 and Zn 2p3/2, respectively. From the In 3d spectra in Figure 3B, the peaks at 452.7 and 445.1 eV for ZIS can be indexed to In 3d3/2 and In 3d5/2, respectively. For the analysis of XPS spectra, the change in binding energy of a peak is negatively correlated with the change in electron cloud density. A peak shift toward higher binding energy indicates a decrease in electron cloud density, while a shift toward lower binding energy signifies an increase in electron cloud density, according to which the direction of electron flow can be obtained. Compared with pure ZIS, the peaks of Zn 2p and In 3d of BZIS-1/20 are shifted to higher binding energy by 0.1 eV, indicating the decreased electron cloud density in the ZIS phase. Figure 3C shows the Bi 4f XPS spectra of BS and BZIS-1/20. The peaks at 164.9 and 159.6 eV are assigned to Bi3+ 4f5/2 and Bi3+ 4f7/2, respectively, while the two additional peaks at 163.7 and 158.4 eV are attributed to Bi(3-x)+[41,42]. The presence of low-valence Bi is accompanied by the existence of sulfur vacancies in BS[43]. Compared to BS, all Bi elements in BZIS-1/20 are low-valent Bi(3-x)+, indicating the existence of sulfur defects in the heterojunction. Moreover, the peaks in the Bi 4f spectrum of BZIS-1/20 shift towards lower binding energy, suggesting an increase in the electron cloud density in the BS phase. These results reveal that an interfacial electron transfer from ZIS to BS occurs when they contact each other. Figure 3D shows the S 2p XPS spectra of the samples. The peaks at 162.4 and 161.2 eV in the spectrum of BS correspond to S 2p1/2 and S 2p3/2, respectively, while in the ZIS spectrum these peaks are located at 162.9 and 161.7 eV. The deviation between the S 2p binding energies for BS and ZIS is due to the different chemical environments of the S element in the two materials[44]. Notably, the S 2p peaks of the BZIS-1/20 heterojunction are positively shifted relative to those of BS but negatively shifted relative to those of ZIS, suggesting electron redistribution between ZIS and BS through an S “bridge”, thereby confirming the formation of an S “bridge” at the heterojunction interface[45].
Figure 3. XPS spectra of BS, ZIS and BZIS-1/20: (A) Zn 2p, (B) In 3d, (C) Bi 4f and (D) S 2p. XPS: X-ray photoelectron spectroscopy; BS: Bi2S3; ZIS: ZnIn2S4; BZIS: Bi2S3/ZnIn2S4.
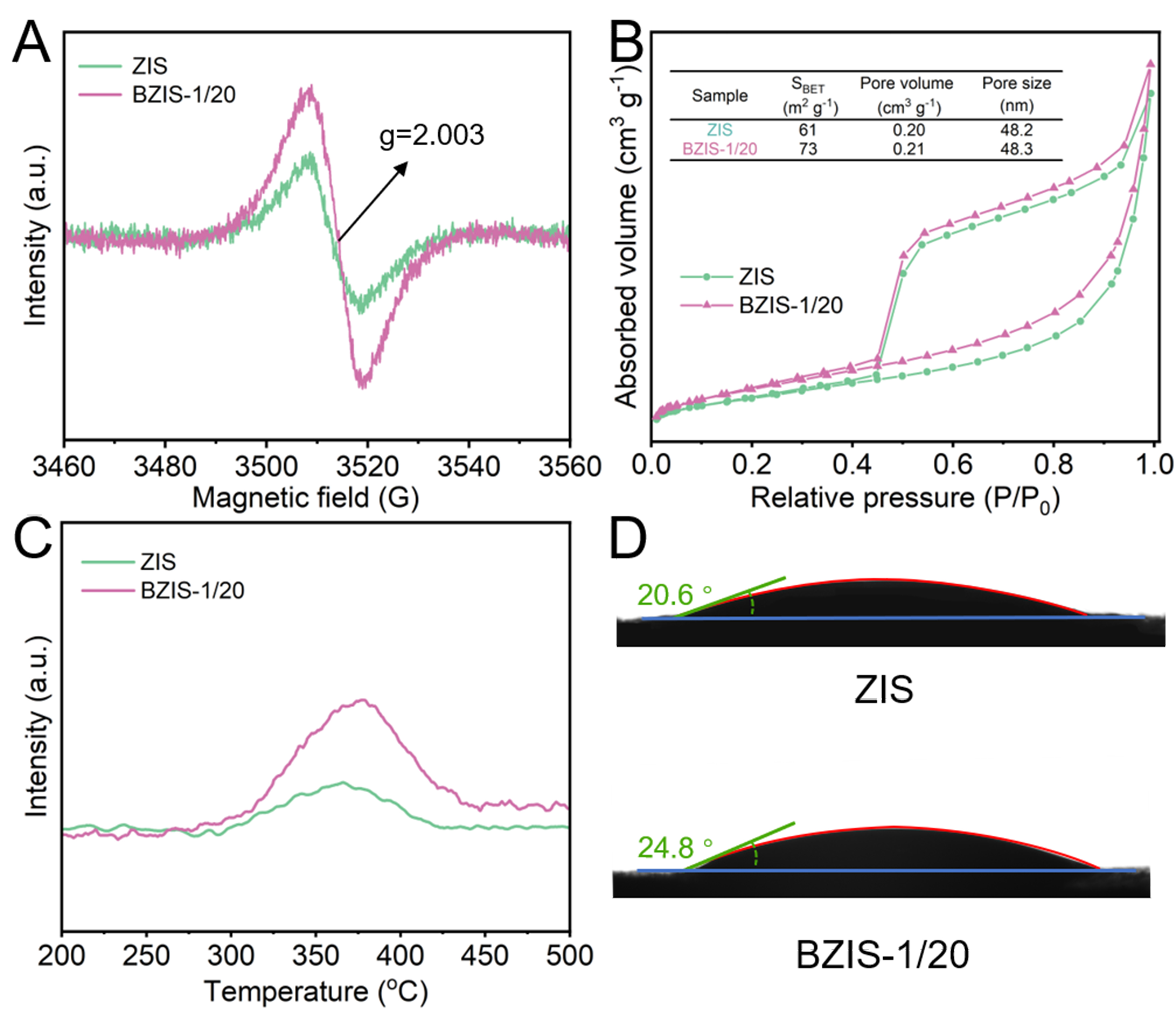
The defect states in ZIS and BZIS-1/20 were studied by the EPR spectra. As shown in Figure 4A, there is a peak at g = 2.003 for both ZIS and BZIS-1/20, which can be attributed to sulfur defects[46]. Besides, the signal of BZIS-1/20 is stronger than that of ZIS, indicating a higher concentration of sulfur defects in BZIS-1/20 compared to ZIS. As reported in the literature[47-49], defects play a pivotal role in enhancing the adsorption of oxygen molecules on the catalyst surface. The N2 physical adsorption tests [Figure 4B] reveal that BZIS-1/20 possesses a higher specific surface area compared to ZIS, which will provide more surface active sites for ORR. O2-TPD profiles of ZIS and BZIS-1/20 [Figure 4C] demonstrate the enhanced O2 adsorption capacity of BZIS-1/20 compared to ZIS. Since the ORR occurs in water, the hydrophilicity/hydrophobicity of the catalysts may affect the reaction performance. The water contact angle can be used to characterize the hydrophilicity/hydrophobicity of a material surface[50]. Typically, the smaller the water contact angle, the stronger the hydrophilicity of the material surface. As shown in Figure 4D, the water contact angles of ZIS and BZIS-1/20 are 20.6° and 24.8°, respectively, indicating the enhanced hydrophobicity of BZIS-1/20 compared to ZIS. The improvement of the hydrophobicity for BZIS-1/20 can create a microscopic three-phase interface, which will boost the capture and diffusion of O2 molecules, and thus promote the ORR[51,52].
Photocatalytic performance for H2O2 production
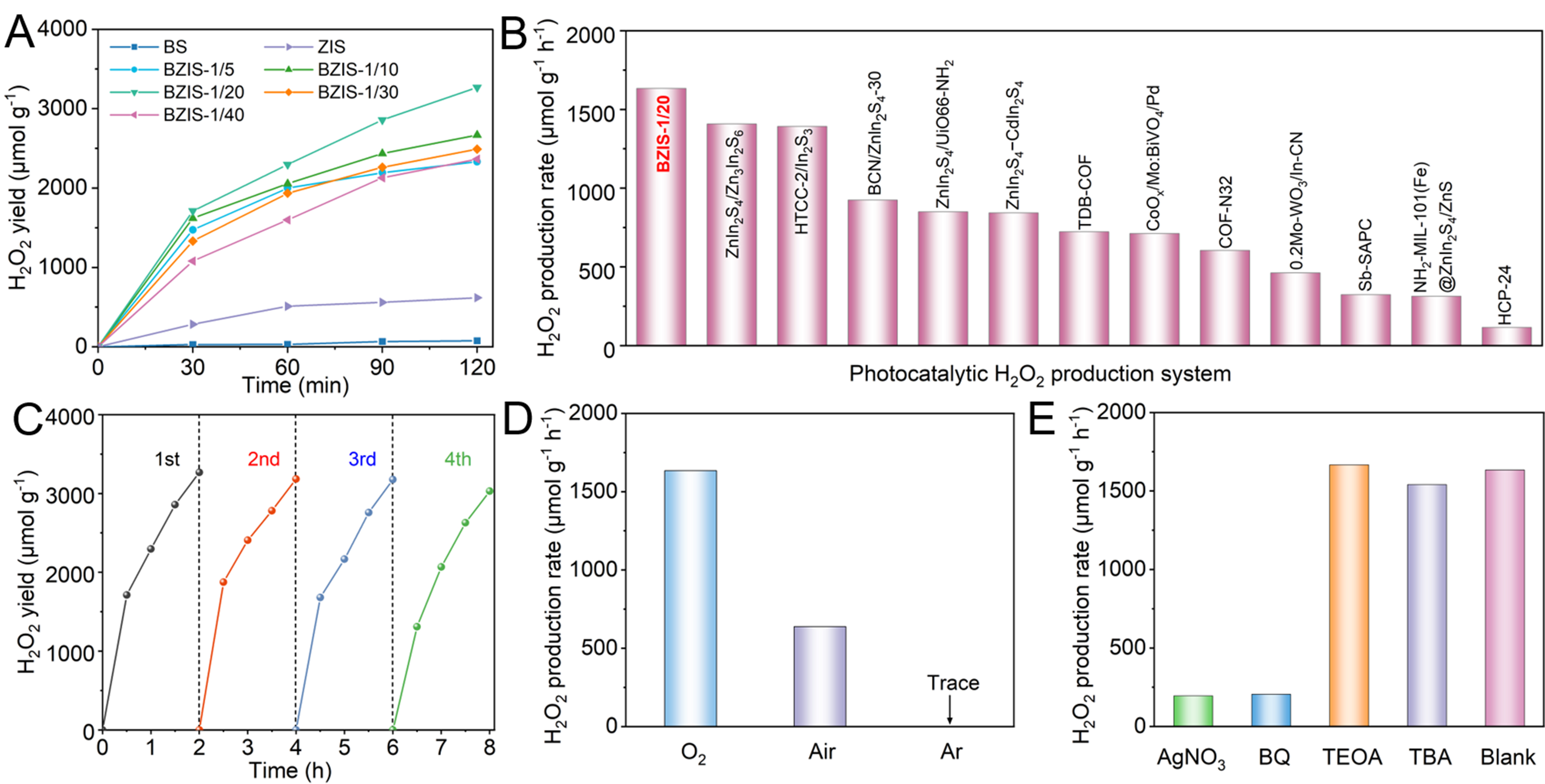
The performance of ZIS, BS and BZIS photocatalysts to produce H2O2 was evaluated under visible light irradiation (λ > 420 nm) in pure water. As shown in Figure 5A, the H2O2 production rates of BS and ZIS without a sacrificial agent were 38 and 309 μmol·g-1·h-1, respectively. The BZIS heterojunctions showed much higher H2O2 yields compared to ZIS and BS. As the BS content increases, the H2O2 yield exhibits an initial rise followed by a decline, among which BZIS-1/20 shows the optimal performance. The H2O2 production rate of BZIS-1/20 reaches 1,634 μmol·g-1·h-1, which is 5.3 times that of ZIS and 43.0 times that of BS. The performance surpasses most of the reported photocatalysts in the literature [Figure 5B and Supplementary Table 1]. The cycling performance of BZIS-1/20 was evaluated. As shown in Figure 5C, there is no obvious decay of H2O2 yield after four cycles. The crystal structure of the photocatalyst is maintained after the reaction from the XRD pattern [Supplementary Figure 3], demonstrating the good structural stability and robust cycling performance of the photocatalyst.
Figure 5. (A) Time-dependent H2O2 yields over ZIS, BS and BZIS heterojunctions; (B) Comparison of the H2O2 production performance of BZIS-1/20 in pure water with other reported photocatalysts; (C) Cycling performance of BZIS-1/20 for H2O2 production; (D) H2O2 yields of BZIS-1/20 under different atmospheres; and (E) H2O2 yields of BZIS-1/20 in the presence of different scavengers. ZIS: ZnIn2S4; BS: Bi2S3; BZIS: Bi2S3/ZnIn2S4.
The H2O2 production performance of BZIS-1/20 was also tested under different gas bubbling conditions [Figure 5D]. It can be seen that the H2O2 yield sharply declines when O2 is replaced by air as feed gas and almost no H2O2 is produced under Ar conditions. This suggests that the production of H2O2 comes exclusively from the ORR pathway. The pathway of H2O2 generation over BZIS-1/20 was further demonstrated by radical quenching experiments, in which AgNO3, BQ, TEOA and TBA were used to capture e-, ·O2-, h+ and ·OH, respectively[53]. As can be seen from Figure 5E, the performance of BZIS-1/20 declines significantly after the addition of AgNO3 or BQ, indicating that H2O2 is mainly produced via the ORR pathway and that ·O2- is an important intermediate in H2O2 synthesis. By contrast, the H2O2 yield has no significant change after adding TEOA or TBA, ruling out the presence of WOR pathway. These results demonstrate that H2O2 production over BZIS-1/20 dominantly follows a two-step single-electron ORR pathway.
Light absorption and charge separation
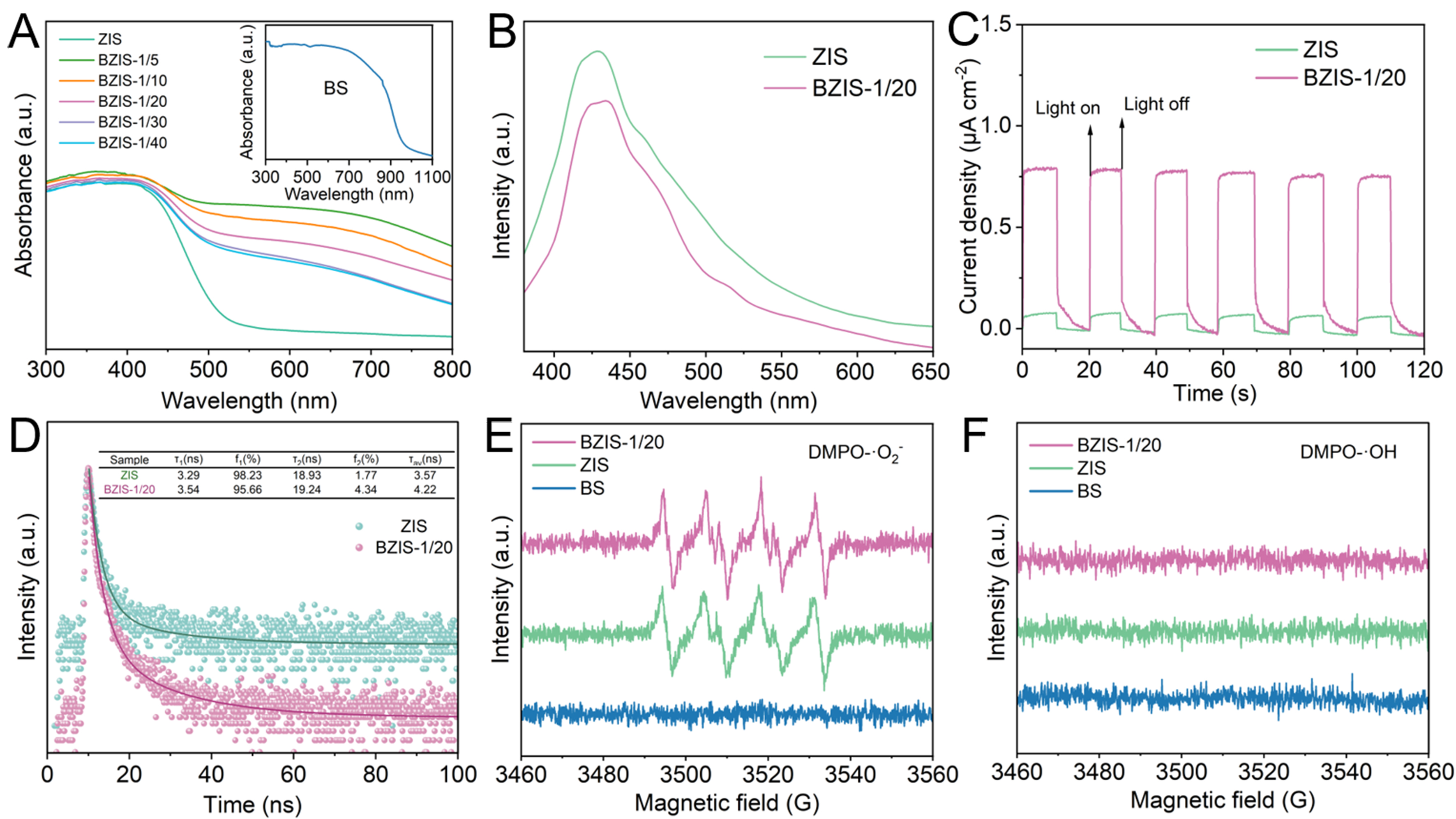
Figure 6A presents the UV–Vis diffuse reflectance spectra of the photocatalysts. Pure ZIS exhibits an absorbance edge at 430 nm, while pure BS shows a broad adsorption in the range of 300-900 nm. After introducing BS to ZIS, the absorption in the 430-800 nm range was markedly enhanced, suggesting that the construction of the heterojunction significantly improves visible-light absorption. The separation efficiency of photogenerated carriers was investigated by PL spectra and transient photocurrent response. As shown in Figure 6B, the lower PL peak intensity of BZIS-1/20 compared to ZIS reveals suppressed radiative recombination and improved carrier separation efficiency due to the formation of heterojunction, consistent with the much higher photocurrent density of BZIS-1/20 [Figure 6C].
Figure 6. (A) UV–Vis diffuse reflectance spectra of BS, ZIS and BZIS heterojunctions; (B) PL spectra, (C) transient photocurrent response and (D) tr-PL spectra of ZIS and BZIS-1/20; Spin-trapping EPR spectra recorded for (E) ·O2- and (F) ·OH under illumination for BS, ZIS and BZIS-1/20. UV–Vis: Ultraviolet–visible; BS: Bi2S3; ZIS: ZnIn2S4; BZIS: Bi2S3/ZnIn2S4; PL: photoluminescence; tr-PL: time-resolved photoluminescence; EPR: electron paramagnetic resonance.
The carrier decay dynamics was analyzed by tr-PL spectra. As shown in Figure 6D, the decay curves were fitted with biexponential kinetic models. Generally, the short lifetime (τ1) corresponds to the radiative recombination process, while the long lifetime (τ2) signifies the non-radiative energy transfer process[54]. Compared with ZIS, both τ1 and τ2 and the average lifetime (τav) for BZIS-1/20 are prolonged. The extended charge lifetime indicates more efficient charge separation, which will provide more opportunities for the photogenerated carriers to participate in the photocatalytic reaction[55,56].
Charge transfer route and photocatalytic mechanism for H2O2 production
In order to confirm the reaction intermediates in the photocatalytic process and the charge transfer pathways of BZIS heterojunction, the spin-trapping EPR spectra were conducted. Aqueous and methanol solutions of 5,5-dimethyl-1-pyrroline-N-oxide (DMPO) were used as spin-trapping reagents for hydroxyl radicals (·OH) and superoxide radicals (·O2-), respectively[57-59]. As shown in Figure 6E and F, the typical quadruple peak signal of DMPO-·O2- is clearly observed, while no signal of DMPO-·OH can be observed for ZIS. This suggests that the conduction band (CB) potential of ZIS is more negative than that of O2/·O2-
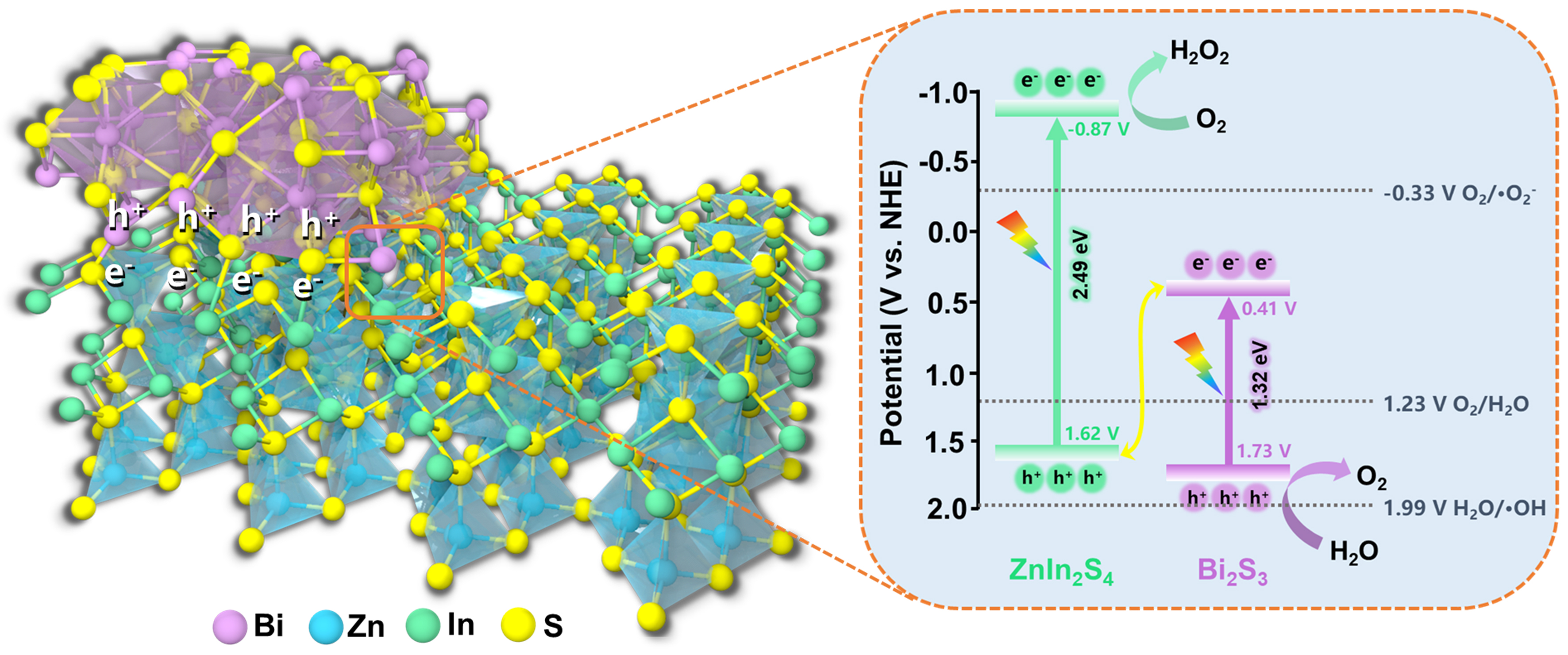
The band structure alignments of the BZIS-1/20 heterojunction and the photocatalytic mechanism were investigated. The band gaps of ZIS and BS are estimated to be 2.49 and 1.32 eV, respectively, according to the Tauc plots [Supplementary Figure 4]. Supplementary Figure 5 depicts the Mott-Schottky plots of BS and ZIS, respectively. The positive slopes of the plots indicate that both ZIS and BS are n-type semiconductors[60], whose CB position is generally 0.1 V more negative than the flat-band potential (Efb). By extrapolating the tangents of the plots at different frequencies to intersect the x-axis, the Efb values for BS and ZIS are obtained to be 0.31 and -0.97 V, respectively (vs. Ag/AgCl, pH = 7). Through the conversion relationship ENHE/V = EAg/AgCl/V + 0.197 V, the CB positions for BS and ZIS are determined to be 0.41 and
The band structure alignments of BZIS-1/20 and the photocatalytic mechanism for H2O2 production are illustrated in Figure 7. BS and ZIS exhibit staggered band structures, indicating possible interfacial charge transfer pathway through either II-scheme or Z-scheme mechanism. If an II-scheme heterojunction is formed, the photogenerated electrons will transfer from the CB of ZIS to that of BS. Since the CB of BS has a more positive potential than that of O2/·O2-, the heterojunction would not generate ·O2- radicals, which contradicts the enhanced EPR signal of DMPO-·O2- detected in the methanol solution containing BZIS-1/20. Therefore, the transfer of photogenerated carriers between BS and ZIS follows the Z-scheme mechanism, which enables the heterojunction to possess stronger redox capabilities.
Figure 7. Schematic illustration of charge transfer route and photocatalytic H2O2 production mechanism of the BZIS-1/20 heterojunction. BZIS: Bi2S3/ZnIn2S4.
The photocatalytic mechanism for producing H2O2 over BZIS-1/20 heterojunction is thus proposed. Under the illumination of visible light, both ZIS and BS are excited and the electrons in their VB leap into the CB. The photogenerated electrons in the CB of BS are recombined with the holes in the VB of ZIS through the S “bridge” at the heterojunction interface. The electrons accumulated in the CB of ZIS transfer to O2 adsorbed on the surface of ZIS to produce H2O2 through two-step single-electron ORR (O2 + e- → ·O2-, ·O2- + 2H+ + e- → H2O2). The holes accumulated in the VB of BS oxidize water to generate O2 through 4e- WOR pathway
CONCLUSIONS
A Z-scheme Bi2S3/ZnIn2S4 heterojunction (BZIS-1/20) was constructed by a one-step hydrothermal method for efficient production of H2O2 in pure water under visible light. The increased specific surface area and sulfur defects as well as enhanced surface hydrophobicity boost oxygen capture capacity via construction of heterojunction. Meanwhile, the intimate interface connected by the S “bridge” efficiently improves the separation and migration of photogenerated carriers. This study highlights the great potential of metal sulfides in the photosynthesis of H2O2 and also provides guidance for the design of novel heterojunction photocatalysts for H2O2 production.
DECLARATIONS
Authors’ contributions
Investigation, data curation, writing - original draft: Zhou, S.
Investigation: Wu, J.
Writing - review and editing: Song, C.
Conceptualization, supervision, writing - review and editing: Li, K.; Guo, X.
Availability of data and materials
The data and materials supporting the findings of this study are available within the article and its Supplementary Materials.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (22278056, 22478058) and the Fundamental Research Funds for the Central Universities (DUT22LAB602).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Tian, Y.; Deng, D.; Xu, L.; et al. Strategies for sustainable production of hydrogen peroxide via oxygen reduction reaction: from catalyst design to device setup. Nanomicro. Lett. 2023, 15, 122.
2. Hayat, A.; Ajmal, Z.; Alzahrani, A. Y. A.; et al. The photocatalytic H2O2 production: design strategies, photocatalyst advancements, environmental applications and future prospects. Coord. Chem. Rev. 2025, 522, 216218.
3. Campos-Martin, J. M.; Blanco-Brieva, G.; Fierro, J. L. Hydrogen peroxide synthesis: an outlook beyond the anthraquinone process. Angew. Chem. Int. Ed. Engl. 2006, 45, 6962-84.
4. Shiraishi, Y.; Kanazawa, S.; Kofuji, Y.; et al. Sunlight-driven hydrogen peroxide production from water and molecular oxygen by metal-free photocatalysts. Angew. Chem. Int. Ed. Engl. 2014, 53, 13454-9.
5. Zheng, D.; Wu, Y.; Yang, X.; Wang, S.; Fang, Y. Developing polymeric carbon nitrides for photocatalytic H2O2 production. ChemSusChem 2024, 17, e202400528.
6. Yang, S.; Liu, S.; Li, H.; et al. Boosting oxygen mass transfer for efficient H2O2 generation via 2e--ORR: a state-of-the-art overview. Electrochim. Acta. 2024, 479, 143889.
7. Sun, X.; Yang, J.; Zeng, X.; et al. Pairing oxygen reduction and water oxidation for dual-pathway H2O2 production. Angew. Chem. Int. Ed. Engl. 2024, 63, e202414417.
8. Xu, X.; Sui, Y.; Chen, W.; et al. The photocatalytic H2O2 production by metal-free photocatalysts under visible-light irradiation. Appl. Catal. B. Environ. 2024, 341, 123271.
9. Pei, Z.; Li, Y.; Fan, G.; et al. Low-coordinated conductive ZnCu metal-organic frameworks for highly selective H2O2 electrosynthesis. Small 2024, 20, e2403808.
10. He, H.; Liu, S.; Liu, Y.; et al. Review and perspectives on carbon-based electrocatalysts for the production of H2O2 via two-electron oxygen reduction. Green. Chem. 2023, 25, 9501-42.
11. Chen, X.; Niu, F.; Ma, T.; Li, Q.; Wang, S.; Shen, S. Anchoring group regulation in semiconductor/molecular complex hybrid photoelectrode for photoelectrochemical water splitting. Smart. Mol. 2025, 3, e20240056.
12. Yang, F.; Feng, C.; Zuo, S.; et al. Photocatalytic H2O2 production with >30% quantum efficiency via monovalent copper dynamics. J. Am. Chem. Soc. 2025, 147, 17112-20.
13. Li, H.; Zhang, Y.; Li, J.; et al. Boosting H2O2 evolution of CdS via constructing a ternary photocatalyst with earth-abundant halloysite nanotubes and NiS co-catalyst. Chin. J. Catal. 2025, 69, 111-22.
14. Lei, Z.; You, W.; Liu, M.; et al. Photocatalytic water reduction under visible light on a novel ZnIn2S4 catalyst synthesized by hydrothermal method. Chem. Commun. 2003, 2142-3.
15. Zhang, G.; Wu, H.; Chen, D.; et al. A mini-review on ZnIn2S4-based photocatalysts for energy and environmental application. Green. Energy. Environ. 2022, 7, 176-204.
16. Wu, X.; Chen, G.; Li, L.; Wang, J.; Wang, G. ZnIn2S4-based S-scheme heterojunction photocatalyst. J. Mater. Sci. Technol. 2023, 167, 184-204.
17. Li, X.; Wu, H.; Yin, S.; et al. Up-conversion effect boosted NIR-driven photocatalytic solar fuel generation of NaYF4: Yb, Er decorated ZnIn2S4 flowers with rich Zn vacancies. Chem. Synth. 2025, 5, 29.
18. Hu, J.; Yang, T.; Chen, J.; Yang, X.; Qu, J.; Cai, Y. Efficient solar-driven H2O2 synthesis in-situ and sustainable activation to purify water via cascade reaction on ZnIn2S4-based heterojunction. Chem. Eng. J. 2022, 430, 133039.
19. Hu, Y.; Ma, X.; Fan, G.; Yang, L.; Li, F. Tailoring the d-band center enables La doped ZnIn2S4 to be active for boosting photocatalytic activation of oxygen and degradation of antibiotics in wastewater. Inorg. Chem. Front. 2024, 11, 4387-400.
20. Peng, H.; Yang, H.; Han, J.; et al. Defective ZnIn2S4 nanosheets for visible-light and sacrificial-agent-free H2O2 Photosynthesis via O2/H2O Redox. J. Am. Chem. Soc. 2023, 145, 27757-66.
21. Liu, H.; Yan, M.; Wang, Z.; et al. Constructing boosted charge separation for efficient H2O2 production and pollutant degradation by highly defective ZnIn2S4/carbon doped boron nitride. J. Environ. Chem. Eng. 2024, 12, 114251.
22. He, Y.; Rao, H.; Song, K.; et al. 3D hierarchical ZnIn2S4 nanosheets with rich Zn vacancies boosting photocatalytic CO2 reduction. Adv. Funct. Mater. 2019, 29, 1905153.
23. Zhang, N.; Liu, M.; Wang, Y.; et al. NH2-MIL-101(Fe)@ZnIn2S4/ZnS heterojunction nanoreactors for efficient photocatalytic-Fenton performance via in-situ H2O2 evolution. Mater. Today. Energy. 2023, 38, 101419.
24. Liu, Y.; Wu, Y.; Liu, Y.; et al. Synergistic interfacial reconstruction and surface polarization in a compact ZnIn2S4–CdIn2S4 heterojunction for enhanced photocatalytic H2O2 Production. ACS. Catal. 2024, 14, 7726-35.
25. Che, Y.; Wang, K.; Wang, C.; Weng, B.; Chen, S.; Meng, S. Lattice match-enabled Zn3In2S6@CdS S-scheme heterojunction with S covalent bond bridge for simultaneous H2O2 photosynthesis and H2 production. J. Mater. Sci. Technol. 2026, 243, 228-36.
26. Yang, J.; Zhu, X.; Yu, Q.; et al. Multidimensional In2O3/In2S3 heterojunction with lattice distortion for CO2 photoconversion. Chin. J. Catal. 2022, 43, 1286-94.
27. Tian, N.; Hu, C.; Wang, J.; Zhang, Y.; Ma, T.; Huang, H. Layered bismuth-based photocatalysts. Coord. Chem. Rev. 2022, 463, 214515.
28. Song, S.; Xing, Z.; Zhao, H.; Li, Z.; Zhou, W. Recent advances in bismuth-based photocatalysts: environment and energy applications. Green. Energy. Environ. 2023, 8, 1232-64.
29. He, R.; Xu, D.; Cheng, B.; Yu, J.; Ho, W. Review on nanoscale Bi-based photocatalysts. Nanoscale. Horiz. 2018, 3, 464-504.
30. Rosaiah, P.; Harikrishnan, L.; Radhalayam, D.; et al. Hierarchical construction of ZnBi2O4 anchored on flower-like Bi2WO6 heterojunction photocatalyst for removal of alizarin red S. Surf. Interfaces. 2025, 58, 105777.
31. Di, J.; Zhu, C.; Ji, M.; et al. Defect-rich Bi12O17Cl2 nanotubes self-accelerating charge separation for boosting photocatalytic CO2 reduction. Angew. Chem. Int. Ed. Engl. 2018, 57, 14847-51.
32. Chawla, A.; Sudhaik, A.; Sonu,
33. Wu, X.; Zhang, Q.; Su, C. Bi2MoO6/Bi2S3 S-scheme heterojunction for efficient photocatalytic oxygen evolution. FlatChem 2021, 27, 100244.
34. Abdel-Moniem, S. M.; El-Liethy, M. A.; Ibrahim, H. S.; Ali, M. E. M. Innovative green/non-toxic Bi2S3@g-C3N4 nanosheets for dark antimicrobial activity and photocatalytic depollution: turnover assessment. Ecotoxicol. Environ. Saf. 2021, 226, 112808.
35. Jing, H.; Xu, G.; Yao, B.; et al. Sulfur vacancy-enriched rhombohedral ZnIn2S4 nanosheets for highly efficient photocatalytic overall water splitting under visible light irradiation. ACS. Appl. Energy. Mater. 2022, 5, 10187-95.
36. Shen, S.; Li, X.; Zhou, Y.; et al. Novel BiOBr/Bi2S3 high-low junction prepared by molten salt method for boosting photocatalytic degradation and H2O2 production. J. Mater. Sci. Technol. 2023, 155, 148-59.
37. Wang, P.; Shen, Z.; Xia, Y.; et al. Atomic insights for optimum and excess doping in photocatalysis: a case study of few-layer Cu-ZnIn2S4. Adv. Funct. Mater. 2019, 29, 1807013.
38. Li, Y.; Yu, B.; Hu, Z.; Wang, H. Construction of direct Z-scheme SnS2@ZnIn2S4@kaolinite heterostructure photocatalyst for efficient photocatalytic degradation of tetracycline hydrochloride. Chem. Eng. J. 2022, 429, 132105.
39. Ye, L.; Fu, J.; Xu, Z.; Yuan, R.; Li, Z. Facile one-pot solvothermal method to synthesize sheet-on-sheet reduced graphene oxide (RGO)/ZnIn2S4 nanocomposites with superior photocatalytic performance. ACS. Appl. Mater. Interfaces. 2014, 6, 3483-90.
40. Uddin, I.; Abzal, S. M.; Kalyan, K.; et al. Starch-assisted synthesis of Bi2S3 nanoparticles for enhanced dielectric and antibacterial applications. ACS. Omega. 2022, 7, 42438-45.
41. Miodyńska, M.; Mikolajczyk, A.; Bajorowicz, B.; et al. Urchin-like TiO2 structures decorated with lanthanide-doped Bi2S3 quantum dots to boost hydrogen photogeneration performance. Appl. Catal. B. Environ. 2020, 272, 118962.
42. Wang, Y.; Wang, Y.; Meng, Y.; et al. A highly efficient visible-light-activated photocatalyst based on bismuth- and sulfur-codoped TiO2. J. Phys. Chem. C. 2008, 112, 6620-6.
43. Jin, Z.; Li, J.; Liu, D.; et al. Effective promotion of spacial charge separation of dual S-scheme (1D/2D/0D) WO3@ZnIn2S4/Bi2S3 heterojunctions for enhanced photocatalytic performance under visible light. Sep. Purif. Technol. 2022, 284, 120207.
44. Hu, J.; Li, B.; Li, X.; et al. Lattice match-enabled covalent heterointerfaces with built-in electric field for efficient hydrogen peroxide photosynthesis. Adv. Mater. 2024, 36, e2412070.
45. Dai, D.; Qiu, J.; Xia, G.; et al. Metal-organic framework templated Z-scheme ZnIn2S4/Bi2S3 hierarchical heterojunction for photocatalytic H2O2 production from wastewater. Small 2024, 20, e2403268.
46. Wang, X.; Wang, X.; Huang, J.; Li, S.; Meng, A.; Li, Z. Interfacial chemical bond and internal electric field modulated Z-scheme Sv-ZnIn2S4/MoSe2 photocatalyst for efficient hydrogen evolution. Nat. Commun. 2021, 12, 4112.
47. Zhao, B.; Shang, C.; Qi, N.; Chen, Z.; Chen, Z. Stability of defects in monolayer MoS2 and their interaction with O2 molecule: a first-principles study. Appl. Surf. Sci. 2017, 412, 385-93.
48. Sun, G.; Li, F.; Wu, T.; et al. O2 adsorption associated with sulfur vacancies on MoS2 microspheres. Inorg. Chem. 2019, 58, 2169-76.
49. Li, Y. F.; Aschauer, U.; Chen, J.; Selloni, A. Adsorption and reactions of O2 on anatase TiO2. Acc. Chem. Res. 2014, 47, 3361-8.
50. Law, K. Water–surface interactions and definitions for hydrophilicity, hydrophobicity and superhydrophobicity. Pure. Appl. Chem. 2015, 87, 759-65.
51. Li, X.; Zheng, Q.; Wang, X.; et al. Introduction of electron-deficient unit in resorcinol-formaldehyde resin to construct donor–acceptor conjugated polymer for enhancing photocatalytic H2O2 production. J. Mater. Chem. A. 2024, 12, 8420-8.
52. He, M.; Peng, X.; Wu, S.; et al. Gas/solid/liquid triphase interface of carbon nitride for efficient photocatalytic H2O2 production. Inorg. Chem. Front. 2025, 12, 3237-45.
53. Lin, S.; Wang, Q.; Huang, H.; Zhang, Y. Piezocatalytic and photocatalytic hydrogen peroxide evolution of sulfide solid solution nano-branches from pure water and air. Small 2022, 18, e2200914.
54. Wu, Y.; Yang, Y.; Gu, M.; et al. 1D/0D heterostructured ZnIn2S4@ZnO S-scheme photocatalysts for improved H2O2 preparation. Chin. J. Catal. 2023, 53, 123-33.
55. Prusty, D.; Mansingh, S.; Priyadarshini, N.; Parida, K. Unraveling the photocatalytic efficiency of quinary alloyed QDs for H2O2 production and antibiotic degradation with detail kinetic and influencing factor study. Surf. Interfaces. 2023, 39, 102999.
56. Yang, J.; Bie, C. Ultrafast S-scheme interfacial electron transport enhances CO2 photoreduction. Chem. Synth. 2024, 4, 68.
57. Tie, W.; Du, Z.; Yue, H.; et al. Self-assembly of carbon nanotube/graphitic-like flake/BiOBr nanocomposite with 1D/2D/3D heterojunctions for enhanced photocatalytic activity. J. Colloid. Interface. Sci. 2020, 579, 862-71.
58. Xia, P.; Cao, S.; Zhu, B.; et al. Designing a 0D/2D S-scheme heterojunction over polymeric carbon nitride for visible-light photocatalytic inactivation of bacteria. Angew. Chem. Int. Ed. Engl. 2020, 59, 5218-25.
59. Tie, W.; Bhattacharyya, S. S.; Ma, T.; et al. Improving photoexcited carrier separation through Z-scheme W18O49/BiOBr heterostructure coupling carbon quantum dots for efficient photoelectric response and tetracycline photodegradation. Carbon 2025, 231, 119707.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].