Coal tar pitch-derived nitrogen-doped carbon hosting iron for enhanced ammonia electrosynthesis
Abstract
Renewable electricity powered N2 electroreduction provides a clean strategy for sustainable NH3 production, of which the fabrication of electrocatalysts with excellent performance, stability, and cost-effectiveness is vital for its real applications. Herein, we for the first time confined Fe species to the coal tar pitch derived nitrogen-doped porous carbon (denoted as Fe2O3/FeNC) by pyrolyzing a uniform mixture of medium temperature coal tar pitch, FeCl3·6H2O, urea and NaCl. The obtained Fe2O3/FeNC exhibits an excellent N2 electroreduction activity in neutral media, evidenced by an NH3 yield of
Keywords
INTRODUCTION
Ammonia (NH3) is important in both chemical industries and carbon-free energy storage, and its large-scale production still rests on the energy-intensive Haber-Bosch (H-B) technique. This technique has shortcomings such as high energy consumption and CO2 emission[1-3]. Therefore, it is imperative to explore sustainable and eco-friendly NH3 synthesis techniques to alleviate the energy shortage, global climate change and environmental crisis.
The electrosynthesis of NH3 from N2 using electricity from renewable energy sources has been considered as the most potential substitute to the H-B technique[4,5]. Nevertheless, the NH3 yield rate and Faradaic efficiency (FE) of this strategy are still far below the practical targets of Department of Energy (DOE) mainly due to the extraordinary inertness of N2 molecule and the competing hydrogen evolution reaction (HER)[6-8]. Electrocatalysts play the most significant role in addressing the above issues[9-11]. As such, designing highly efficient and cost-effective electrocatalysts with both HER restriction capacity and outstanding N2 adsorption/activation capacity is of vital importance for the industrial application of NH3 electrosynthesis.
Up to now, extensive studies have been done in this field and a series of metal-based electrocatalysts, such as noble metals[12-14], main group metals[15,16], and transition metals[3,17], have been explored. Among these, iron (Fe)-based materials exhibit great potential as catalysts for large-scale N2 electroreduction mainly due to the intrinsic excellent affinity for N2, abundant reserves and low toxicity[1,18,19]. From the thermodynamic and kinetic view of point, favorable Fe-based N2 reduction electrocatalysts can be obtained by maximizing exposed Fe active sites and preciously tuning local environment around active sites[20-22]. For example, Zhao and co-workers anchored Fe single-atom catalysts (SACs) on lignocellulose-derived carbon, which afforded an NH3 yield (32.1 μg·h-1·mgcat-1)[23]. Li and co-workers immobilized iron atoms on the N and O co-doped porous carbon matrix (FeSA-NO-C) to obtain a novel electrocatalyst that exhibited an NH3 yield of
Coal tar pitch, a by-product of coking, is suitable as a precursor for preparing carbon supported SACs, clusters and nanoparticles due to its merits of source abundance, low price and precursor synthesis free[28-30]. Li et al. adopted coal tar pitch as the precursor to synthesize NiO Clusters modified Ni-N-C SACs and used it as catalyst for CO2 electroreduction[31]. The obtained NiO/Ni-N-C demonstrates an ultra-high FE of 96.5% towards CO production across a broad range of overpotentials due to the electron delocalization effect facilitated by the NiO clusters at the active sites. Coal tar pitch possesses diverse heteroatoms (e.g., N, S and O) that are applicable for anchoring metal active sites to carbon matrix via forming coordination bonds between metal atoms and heteroatoms[32-34]. In addition, these heteroatoms modulate the charge density of metal active sites for providing a favorable environment for N2 adsorption, activation and hydrogenation, thus enhancing the N2 electroreduction activity. However, the design and synthesis of metal-based catalysts from coal tar pitch for the N2 electroreduction and corresponding mechanism investigation have been barely reported.
Herein, we originally anchored Fe species to the coal tar pitch-derived N-doped porous carbon (Fe2O3/FeNC) through ball-milling and pyrolysis by using coal tar pitch, FeCl3∙6H2O, urea and NaCl as precursors and template, respectively. The merits of the Fe2O3/FeNC electrocatalyst, including highly dispersed Fe active sites, effective adsorption and activation of N2 enabled by N dopants, as well as the extraordinary conductivity of porous carbon matrix, generate an NH3 yield of 38.17 ± 0.88 µg·h-1·mgcat-1
EXPERIMENTAL
Preparation of Fe2O3/FeNC
Fe2O3/FeNC was prepared according to the literature[20,35,36]. Firstly, 0.50 g medium temperature coal tar pitch, 10.00 g NaCl, 0.18 g FeCl3·6H2O and 1.50 g urea were added into the grinding jar and grounded for
Proton exchange membrane pretreatment
The Nafion 117 membrane was protonated before use by first treating in H2O2 (5 wt%) solution at 80 °C for
Preparation of the working electrode
Typically, 5 mg of Fe2O3/FeNC or NC was dispersed in a mixture of 400 μL ethanol, 600 μL ultrapure water and 20 μL 5% Nafion solution by sonication to form a homogeneous ink. Then, 20 µL of the as-prepared catalyst ink was deposited onto the carbon cloth and vacuum dried. The catalyst loading is calculated by measuring the mass difference of the carbon cloth before and after deposition.
Electrochemical measurements
All electrochemical experiments were carried out on a CHI660E (Chenhua Instrument Co., Ltd., Shanghai, China) electrochemical workstation under ambient conditions. In the three-electrode system, carbon cloth supported Fe2O3/FeNC was used as the working electrode, Ag/AgCl (3.5 M KCl) and graphite rod were used as the reference electrode and the counter electrode, respectively. The electrochemical tests were carried out in a H-type gas-tight cell, of which the anode and cathode were filled with 70 mL electrolyte and separated by Nafion 117 membrane.
Before the electrochemical tests, high-purity N2 (> 99.999%) was used to purge the electrolyte for 30 minutes. During the tests, a continuous flow of this high-purity N2 was maintained in the cathodic compartment. Chronoamperometry tests were carried out in the N2-saturated 0.1 M Na2SO4 electrolyte for a duration of 2 h at various potentials. In this work, all potentials mentioned are converted to reversible hydrogen electrode (RHE) by ERHE = EAg/AgCl + 0.205 + 0.0591 pH[2].
Detection of ammonia and hydrazine
Determination of NH3
The NRR of producing NH3 was determined using UV-vis spectrophotometry, the standard concentration-absorbance curve was first calibrated by the ammonia chloride solution with different concentrations. After the NRR test, 4 mL electrolyte solution at different potentials were removed from the cathode chamber, 4 mL of 1.0 mol·L-1 KOH solution containing 5 wt% C6H5Na3O and 5 wt% C7H6O3Na was added. Then adding 2 mL of 0.05 mol·L-1 NaClO and 0.4 mL of 1 wt% C5FeN6Na2O. Keeping the mixed solution in dark 1 h, and the absorbance measurement at λ = 655 nm was determined. The NH3 yield over the catalyst investigated can be calculated by using calibrated stand concentration-absorbance curve.
Determination of N2H4
A mixture of p-C9H11NO (5.99 g), ethanol (300 mL) and 0.1 M HCl (30 mL) was used as a coloring reagent of N2H4 detection. Specifically, 5 mL of the electrolyte was removed from the electrochemical reaction vessel and 5 mL of the prepared chromogenic reagent was added and stirred at room temperature for 15 min. Subsequently, UV-vis spectroscopy was performed. The absorbance of N2H4 was measured at 455 nm and the yields of N2H4 were estimated using a standard curve. The fitting curve provided in Figure. S6b for the N2H4 assay exhibits an excellent linear relationship between absorbance and N2H4 concentration.
Calculation of NH3 yield and FE
The yield rate of NH3 (υNH3) was calculated using the following equation[2]:
The FE for NRR was defined as the proportion of the electric charges used for synthesizing NH3 among the total charges passing through the electrodes during NRR electrolysis. Assuming three electrons were needed to produce one NH3 molecule, the FE could be calculated as follows[2]:
where
RESULTS AND DISCUSSION
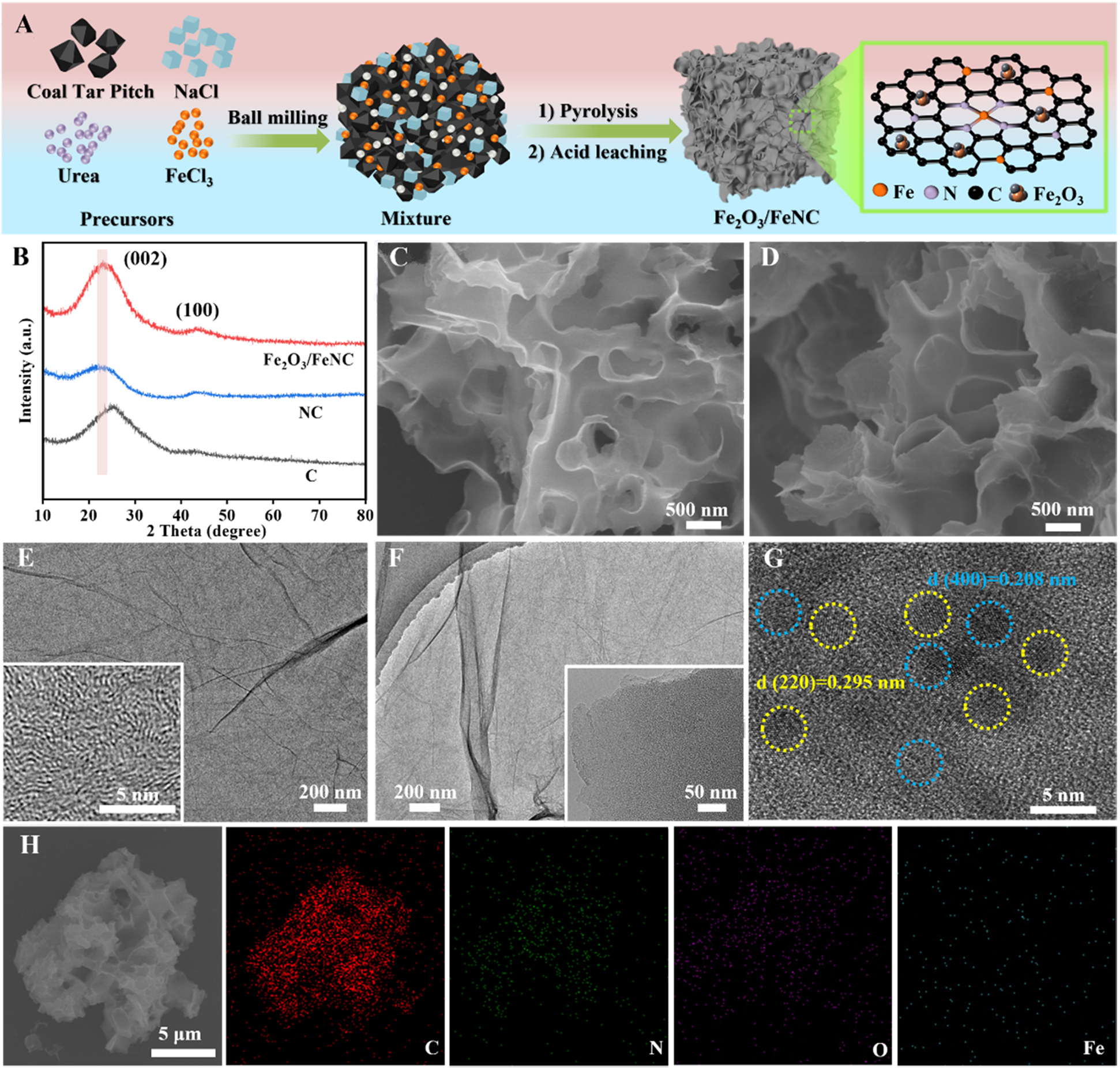
The synthetic process of Fe2O3/FeNC is displayed in Figure 1A. Typically, medium temperature coal tar pitch, FeCl3·6H2O and NaCl with a mass ratio of 0.5:0.18:10 were used as carbon source, iron source and the template, respectively, which were evenly mixed by ball-milling. Then, the mixture was pyrolyzed under the protection of ultra-high argon in a tube furnace, during which ultra-thin carbon nanosheets were formed with the help of the shear force provided by melt NaCl; at the same time, some Fe atoms coordinated with the N atoms in the carbon substrate to form Fe–N bonds; other Fe atoms are confined on the surface of carbon nanosheets to form Fe clusters. Subsequently, the mixture was soaked with dilute hydrochloric acid to remove the Fe clusters and other soluble salts. Finally, the Fe2O3/FeNC was obtained after washing with purified water and vacuum drying. This strategy is an epoch-making technique for the large-scale production of carbon supported metal-based nanocatalysts for electrocatalysis, due to its simple operation, cost-effective, high yield, as well as the readily available of non-artificially synthesized precursors, which dramatically reduce the energy consumption and the environmental pollution. The N-doped carbon, denoted as NC, was also synthesized via this strategy without Fe source for investigating the synergistic effect of Fe species. Based on the X-ray photoelectron spectroscopy (XPS) and Inductively Coupled Plasma Optical Emission Spectroscopy (ICP-OES) analysis, the Fe content in Fe2O3/FeNC is 1.38%.
Figure 1. (A) Schematic of the synthesis of Fe2O3/FeNC; (B) XRD patterns of Fe2O3/FeNC, NC and C. SEM images of (C) NC and (D) Fe2O3/FeNC. TEM images of (E) NC and (F) Fe2O3/FeNC (inset: corresponding HRTEM image); (G) HRTEM image of Fe2O3/FeNC; (H) SEM and corresponding element mapping images of Fe2O3/FeNC. XRD: X-ray diffraction; SEM: scanning electron microscope; TEM: transmission electron microscopy; HRTEM: high-resolution TEM.
Characterizations of Fe2O3/FeNC
The crystal structures of the Fe2O3/FeNC and NC were investigated using X-ray diffraction (XRD). The XRD patterns of both Fe2O3/FeNC and NC exhibit two distinct broad peaks at ~26° and ~43°, corresponding to the (002) and (100) crystallographic planes of defective carbon, respectively[37] [Figure 1B]. Compared to that of NC, no characteristic peaks corresponding to Fe2O3 or Fe are detected in the XRD pattern of Fe2O3/FeNC, mainly due to the low loading content and the high dispersion of these Fe species[38]. Obviously, the introduction of N or/and Fe species enlarges the interlayer distance of the carbon substrate, as evidenced by the peak shift of the (002) plane to lower angle in both Fe2O3/FeNC and NC compared to that of C sample[39]. The scanning electron microscope (SEM) images of NC and Fe2O3/FeNC
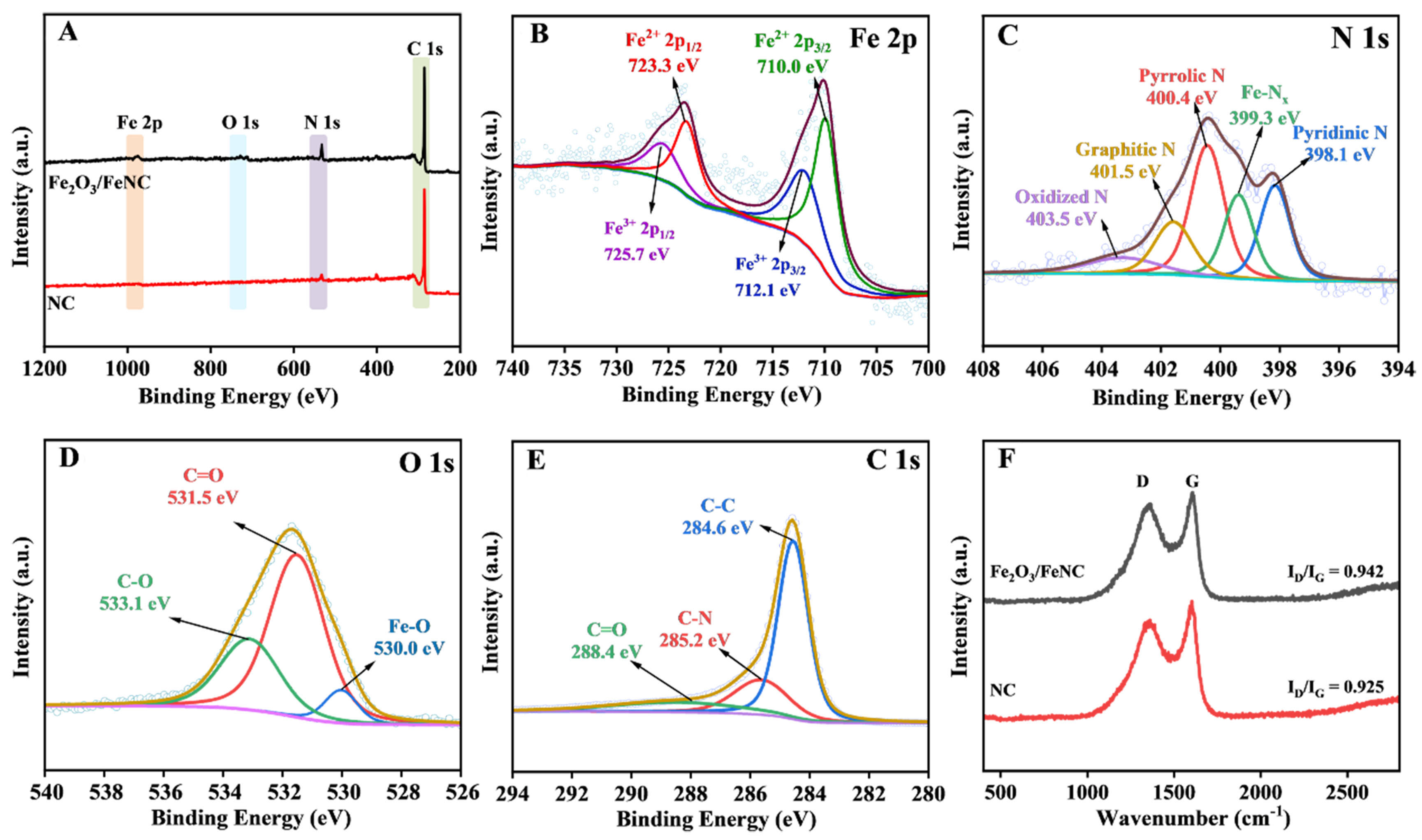
The XPS spectra in Figure 2A confirm the existence of N, C and O elements in both Fe2O3/FeNC and NC. The additional Fe signal demonstrates the successful anchoring of Fe on NC. Moreover, it reveals that the Fe2O3/FeNC contains 1.38 at% Fe, 3.9 at% N, 6.8 at% O and 88.55 at% C, showing good agreement with the corresponding energy-dispersive X-ray spectroscopy (EDS) measurements. Figure 2B displays the XPS spectrum of Fe2O3/FeNC in Fe 2p region, showing two main peaks assigned to Fe2+ 2p1/2 (723.3 eV) and Fe2+ 2p3/2 (710.0 eV)[42,43,44]. Additionally, the other typical peaks at 725.7 and 712.1 eV correspond to the 2p1/2 and 2p3/2 of Fe3+[42,43]. The N 1s XPS spectra of both Fe2O3/FeNC and NC [Figure 2C and Supplementary Figure 2] exhibit four distinct deconvoluted peaks at 398.1 eV (pyridinic N), 400.4 eV (pyrrolic N), 401.5 eV (graphitic N) and 403.5 eV (oxidized N), respectively[45,46]. These pyridinic N and pyrrolic N can facilitate nitrogen reduction reaction (NRR) process[46]. In the N 1s XPS spectrum of Fe2O3/FeNC, the characteristic peak at 399.3 eV corresponding to Fe-Nx indicates that some of Fe atoms are incorporated into the carbon substrate via the Fe–N bonds[47], which is beneficial to prevent the agglomeration and deactivation of the Fe active sites, thereby enhancing the stability of the Fe2O3/FeNC to some extent. The O 1s spectrum of the
Figure 2. (A) Survey XPS spectra of Fe2O3/FeNC and NC. The XPS spectra of Fe2O3/FeNC in (B) Fe 2p, (C) N 1s, (D) O 1s, and (E) C 1s regions; (F) Raman spectra of Fe2O3/FeNC and NC. XPS: X-ray photoelectron spectroscopy.
Raman spectroscopy was performed to analyze the carbonization and graphitization characteristics of
Electrocatalytic N2 reduction performance of the Fe2O3/FeNC catalyst
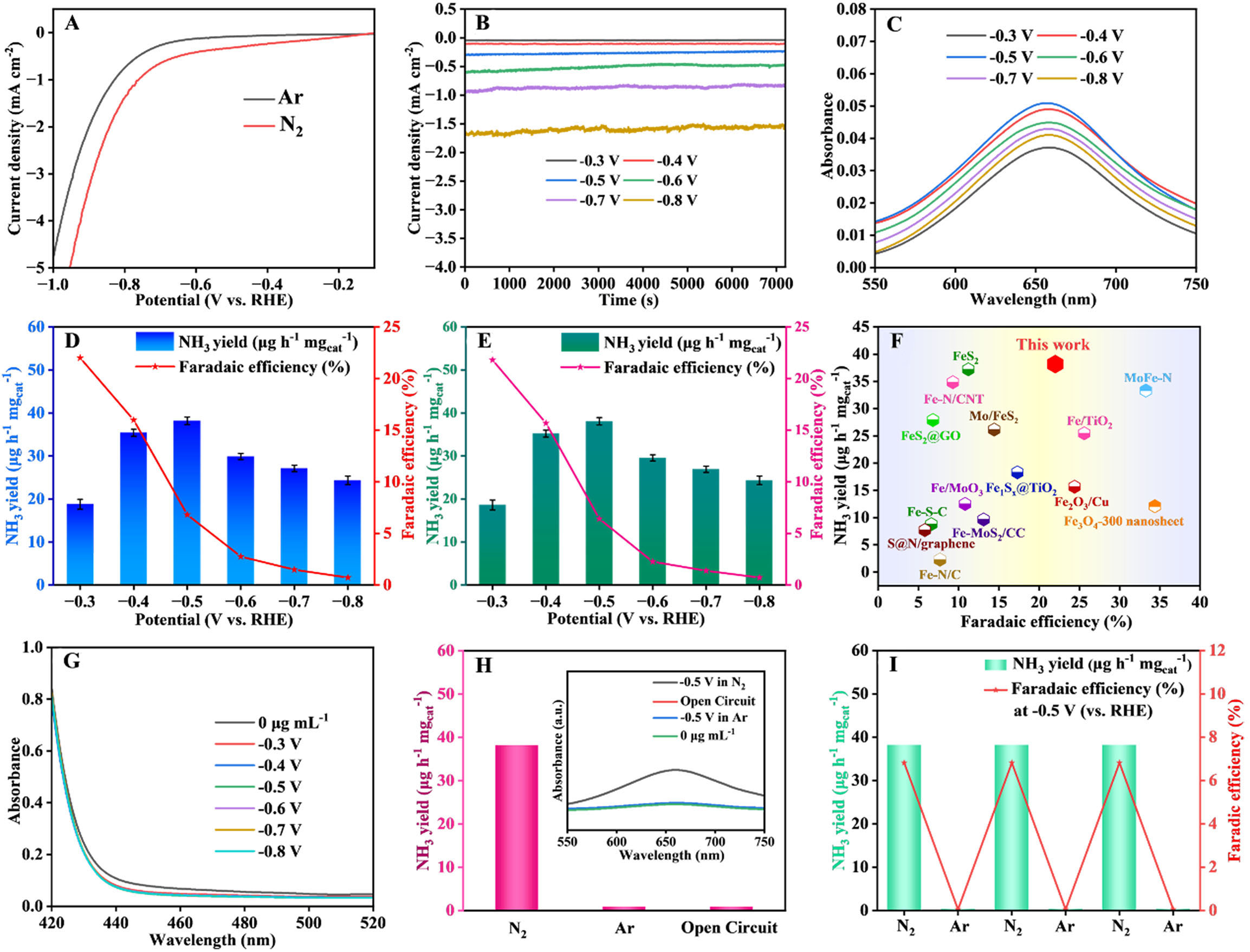
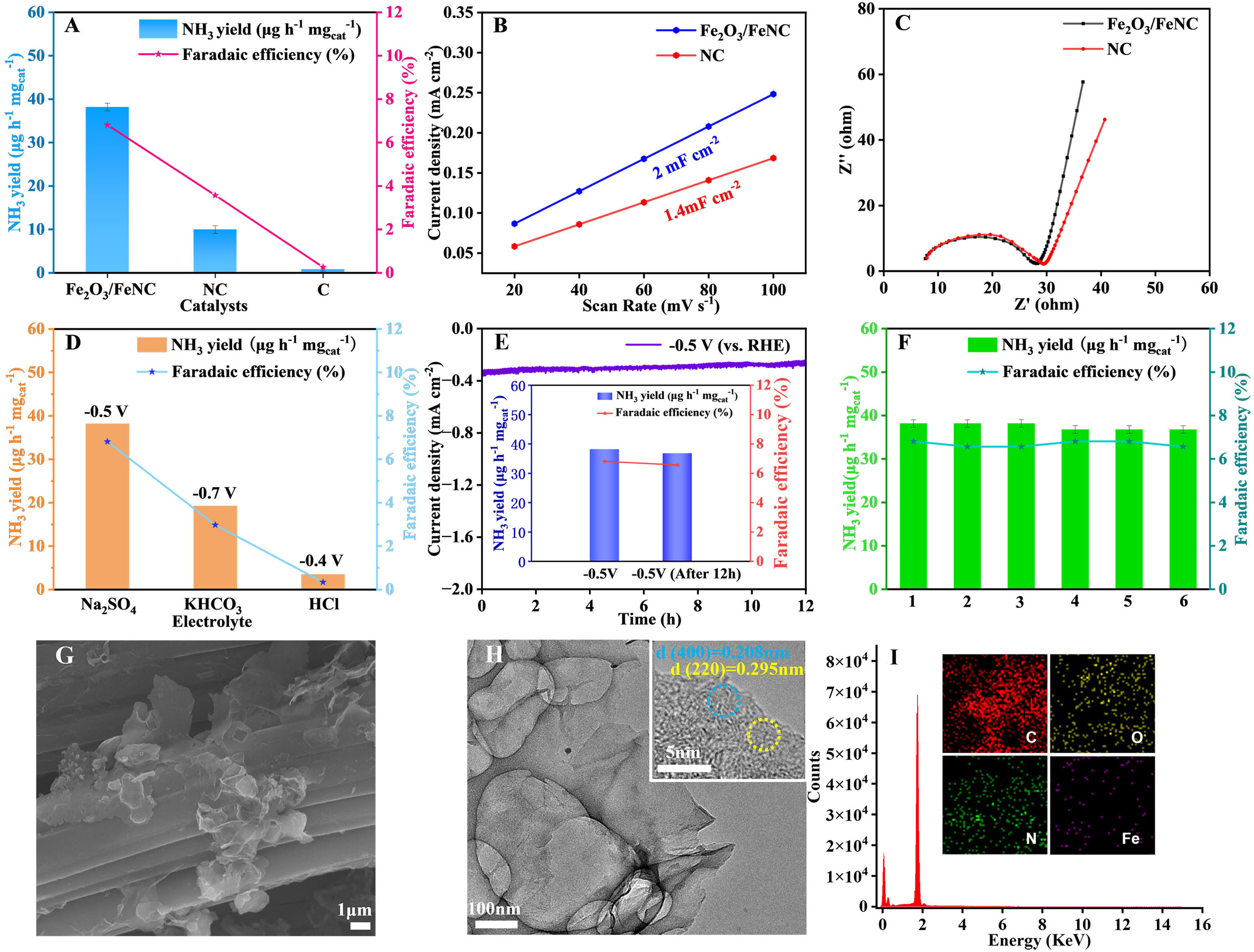
Initially, the electrocatalytic activity of the as-prepared Fe2O3/FeNC catalyst in the N2 reduction process was evaluated by linear sweep voltammetry (LSV). In Figure 3A, the current density in the presence of N2 is higher than that of Ar under the same condition when the overpotential exceeds -0.3 V (vs. RHE), indicating the N2 electroreduction reaction takes place over the Fe2O3/FeNC. Then, chronoamperometry (CA) tests were conducted in 0.1 M Na2SO4 solution at the potentials ranging from -0.3 to -0.8 V (vs. RHE). As demonstrated in Figure 3B, the stable current densities for each 2 h suggest excellent electrochemical stability of Fe2O3/FeNC throughout the N2 electroreduction process at all selected potentials. The ultraviolet-visible (UV-vis) absorption spectra of standard NH4Cl solution in 0.1 M Na2SO4 stained with indophenol blue reagent and corresponding calibration curve are presented in Supplementary Figure 5A and B. The NH3 yields and corresponding Faradaic efficiencies (FEs) were determined from UV-vis absorption spectra [Figure 3C] of electrolytes collected after 2 h of N2 electroreduction catalyzed by Fe2O3/FeNC at selected potentials. The Fe2O3/FeNC demonstrates satisfactory NH3 yields at a wide potential ranging from -0.3 to -0.8 V (vs. RHE) [Figure 3D]. The quantitative determination of NH3 through the indophenol blue method was validated using an ammonia-selective electrode, as shown in Figure 3E. The optimal NH3 yield of
Figure 3. (A) LSV curves for Fe2O3/FeNC in N2- and Ar-saturated 0.1 M Na2SO4; (B) Chronoamperometric curves for Fe2O3/FeNC at selected potentials; (C) the UV-vis absorption spectra of the obtained electrolytes, and corresponding NH3 yields and FEs determined by (D) indophenol blue method and (E) ammonia-selective electrode method (the error line represents the standard deviation); (F) Comparison of electrocatalytic N2 reduction performance with reported catalysts; (G) UV-vis absorption spectra of the electrolytes after 2 h electrolysis at selected potentials; (H) NH3 yields of control tests with the inset showing corresponding UV-vis absorption spectra; (I) NH3 yields and FEs for alternating cycles in Ar- and N2-saturated 0.1 M Na2SO4, respectively. LSV: Linear sweep voltammetry; UV-vis: ultraviolet-visible; FEs: Faradaic efficiencies.
For determining whether the detected NH3 is from N2 electroreduction catalyzed by Fe2O3/FeNC or from the external contamination, we conducted comparative experiments as shown in Figure 3H. Negligible NH3 is detected when the electrolysis experiment is carried out either at -0.5 V (vs. RHE) in the absence of N2 feedstock or at open-circuit potential (OCP) in N2-saturated 0.1 M Na2SO4 for 2 h, confirming that the produced NH3 comes from N2 electroreduction instead of external contamination. In addition, the distinct NH3 yields and FEs obtained at -0.5 V (vs. RHE) in 0.1 M Na2SO4 by alternately using N2 and Ar as feed gas further excludes the influence of nitrogen source from both experimental environment and the Fe2O3/FeNC catalyst on the detection of produced NH3 [Figure 3I].
To evaluate the effects of Fe2O3 and coal tar pitch derived N-doped carbon substrate on the N2 electroreduction performance of Fe2O3/FeNC, the electrocatalytic performances of NC and commercial graphite carbon were investigated under the same condition. As shown in Figure 4A, the commercial graphite carbon exhibits negligible NH3 yield because of lacking efficient active sites for N2 electroreduction. The NC presents its optimal NH3 yield of 9.95 μg·h-1·mgcat-1, which is mainly attributed to the adsorption and activation of N2 provided by N dopants. However, this NH3 yield is only 26.07% of that of Fe2O3/FeNC
Figure 4. (A) NH3 yields and FEs of different catalysts obtained in 0.1 M Na2SO4; (B) Cdl and (C) Nyquist plots of Fe2O3/FeNC and NC; (D) Optimal NH3 yields and FEs of Fe2O3/FeNC obtained in different electrolytes; (E) Time-dependent current density curve of
The as-prepared Fe2O3/FeNC exhibits improved electrocatalytic performances for N2 reduction mainly for the following reasons. On the one hand, the Fe active centers possess both adequate d-orbital electrons and unoccupied d orbitals, which efficiently adsorb and activate the N2 molecules for endowing Fe2O3/FeNC with high N2 electroreduction activity. On the other hand, the cross-linked nanosheets assembled 3D porous structure of Fe2O3/FeNC offers large surface area for supporting Fe species and exposing abundant Fe active sites for N2 electroreduction. Figure 4B presents the double-layer capacitance (Cdl) calculated from cyclic voltammetry measurements [Supplementary Figure 7], of which the Cdl of Fe2O3/FeNC (2 mF·cm-2) is higher than that of NC (1.4 mF·cm-2), showing that Fe2O3/FeNC provides more active sites than NC. In addition, the NC substrate with excellent conductivity ensures the facile electron transfer and speeds up the reaction rate as evidenced by the electrochemical impedance spectroscopy (EIS) tests. As shown in Figure 4C, both the
The electrocatalytic N2 reduction activities of Fe2O3/FeNC in 0.1 M KHCO3 and 0.1 M HCl electrolytes were also investigated at compariable experimental conditions. As shown in Figure 4D, the Fe2O3/FeNC delivers an NH3 yield of 19.25 μg·h-1·mgcat-1 with a low FE of only 2.98% at -0.7 V (vs. RHE) in alkaline media. The sharp decrease in N2 reduction activities is partly due to the limited proton concentration that restricts the protonation of the adsorbed N2 species. In acid media, the unsatisfactory NH3 yield of 3.46 μg·h-1·mgcat-1 with a FE of 0.34% is mainly attributed to the competitive HER that occupies the catalytic active sites and hinders the N2 reduction reaction.
Stability of Fe2O3/FeNC
The electrochemical stability is a vital factor in evaluating the practical use potential of electrocatalysts. As such, long-term electrolysis was carried out at -0.5 V (vs. RHE). As depicted in Figure 4E, the stable current density of the 12-hour continuous electrolysis indicates the excellent durability of Fe2O3/FeNC. The Fe2O3/FeNC electrocatalyst after continuous electrolysis still obtains a high NH3 yield of 36.78 μg·h-1·mgcat-1 with a FE 6.57% at -0.5 V (vs. RHE) as shown in the inset of Figure 4E. The slight attenuation of NH3 yields and FEs before and after 12-hour electrolysis further demonstrate Fe2O3/FeNC can keep high electrochemical activity for at least 12 h. In addition, the NH3 yields and FEs fluctuate slightly during 6 consecutive cycles
CONCLUSIONS
In summary, we prepared an Fe species decorated N-doped porous carbon (Fe2O3/FeNC) via a facile hard template method and pyrolysis treatment by using NaCl as the template and coal tar pitch as carbon source, which not only achieved a high-performance catalyst for N2 electroreduction to NH3 but also enabled the high-value utilization of coal tar pitch. The Fe2O3/FeNC catalyst demonstrates outstanding selectivity and activity for NH3 production, achieving an NH3 yield of 38.17 ± 0.88 μg·h-1·mgcat-1 (-0.5 V vs. RHE) and a FE of 22.01% (-0.3 V vs. RHE) in neutral electrolyte. This remarkable performance mainly originates from three synergistic effects: the abundant highly dispersed Fe species providing sufficient active sites for N2 reduction, the N dopants enhancing the N2 activation through electronic structure modulation, and the hierarchical porous carbon matrix promoting the electron and mass transport. This work highlights the strategy of hosting metallic oxides and metal atoms on coal tar pitch-derived N-doped porous carbon in the mass synthesis of stable, high-performance catalysts towards N2 electroreduction and other electrocatalysis.
DECLARATIONS
Authors’ contributions
Conceptualized and supervised the project: Sun, Y.; Ma, T.
Synthesized the catalysts and performed the electrochemical tests: Fang, Y.; Zhao, Z.
Performed sample characterizations and data analysis: Wang, Y.; Fu, Y.; Fang, Y.; Zhao, Z.; Liu, J.; Yu, J.; Li, H.
Co-wrote the manuscript: Sun, Y.; Zhang, W.; Ma, T.; Qiu, J.
All the authors discussed the results and revised the manuscript.
Availability of data and materials
The raw data supporting the findings of this study are available within this Article and its Supplementary Materials. Further data is available from the corresponding authors upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (Nos. U24B20198, 22308139, 52071171), the Australian Research Council (ARC) through Future Fellowship (FT210100298), Discovery Project (DP220100603), Linkage Project (LP210200504, LP220100088, LP230200897) and the Industrial Transformation Research Hub (IH240100009) schemes, the Australian Government through the Cooperative Research Centres Projects (CRCPXIII000077), the Australian Renewable Energy Agency (ARENA) as part of ARENA's Transformative Research Accelerating Commercialisation Program (TM021), European Commission's Australia-Spain Network for Innovation and Research Excellence (AuSpire), the Foundation of State Key Laboratory of Clean and Efficient Coal Utilization, Taiyuan University of Technology (MJNYSKL202301), the Fundamental Research Funds for Public Universities in Liaoning (LJ232410140033).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Fu, X.; Xu, A.; Pedersen, J. B.; et al. Phenol as proton shuttle and buffer for lithium-mediated ammonia electrosynthesis. Nat. Commun. 2024, 15, 2417.
2. Jiao, F.; Xu, B. Electrochemical ammonia synthesis and ammonia fuel cells. Adv. Mater. 2019, 31, e1805173.
3. Wang, Z.; Guan, Z.; Su, B. Proton shuttle: a key for efficient ammonia electrosynthesis. Chem. Synth. 2025, 5, 20.
4. Dai, T. Y.; Wang, T. H.; Wen, Z.; Jiang, Q. Recent progress on computation-guided catalyst design for highly efficient nitrogen reduction reaction. Adv. Funct. Mate. 2024, 34, 2400773.
5. Li, S.; Wang, Y.; Du, Y.; et al. P-block metal-based electrocatalysts for nitrogen reduction to ammonia: a minireview. Small 2023, 19, e2206776.
6. Singh, A. N.; Anand, R.; Zafari, M.; Ha, M.; Kim, K. S. Progress in single/multi atoms and 2D-nanomaterials for electro/photocatalytic nitrogen reduction: experimental, computational and machine leaning developments. Adv. Energy. Mater. 2024, 14, 2304106.
7. Fu, X.; Zhang, J.; Kang, Y. Recent advances and challenges of electrochemical ammonia synthesis. Chem. Catal. 2022, 2, 2590-613.
8. Wu, J.; Wang, S.; Ji, R.; et al. In situ characterization techniques for electrochemical nitrogen reduction reaction. ACS. Nano. 2024, 18, 20934-56.
9. Sun, Y.; Ma, T.; Wang, H.; Qiu, J. Solvent engineering enables long-term continuous lithium-mediated ammonia synthesis. Chem 2024, 10, 1641-3.
10. Pang, Y.; Su, C.; Jia, G.; Xu, L.; Shao, Z. Emerging two-dimensional nanomaterials for electrochemical nitrogen reduction. Chem. Soc. Rev. 2021, 50, 12744-87.
11. Takashima, T.; Mochida, T.; Irie, H. Electrochemical nitrogen reduction to ammonia using mesoporous iron oxide with abundant oxygen vacancies. Sustain. Energy. Fuels. 2023, 7, 2740-8.
12. Yang, D.; Li, J.; Xiao, M.; Liu, C.; Xing, W.; Zhu, J. Atomically dispersed metal catalysts towards nitrogen reduction for ammonia: from homogeneous to heterogeneous. Chem. Eng. J. 2023, 468, 143776.
13. Li, X.; Wang, X.; Guo, A.; Luo, W.; Yang, L.; Yang, W. Enhancing electrocatalytic nitrogen fixation over core-shell P-Sb2S3/MoS2 heterojunction by vacancy and interface modulation. J. Colloid. Interface. Sci. 2025, 678, 1143-52.
14. Long, X.; Huang, F.; Yao, Z.; et al. Advancements in electrocatalytic nitrogen reduction: a comprehensive review of single-atom catalysts for sustainable ammonia synthesis. Small 2024, 20, e2400551.
15. Xu, R.; Cao, S.; Bo, T.; Mu, N.; Liu, Y.; Zhou, W. Electrochemical nitrogen reduction reaction on anchored SnS2 nanosheets with TM2 dimers. J. Colloid. Interface. Sci. 2024, 660, 290-301.
16. Majumder, M.; Saini, H.; Dědek, I.; et al. Rational design of graphene derivatives for electrochemical reduction of nitrogen to ammonia. ACS. Nano. 2021, 15, 17275-98.
17. Li, X.; Hai, G.; Liu, J.; et al. Bio-inspired NiCoP/CoMoP/Co(Mo3Se4)4@C/NF multi-heterojunction nanoflowers:effective catalytic nitrogen reduction by driving electron transfer. Appl. Catal. B-Environ. 2022, 314, 121531.
18. Li, S.; Zhou, Y.; Fu, X.; et al. Long-term continuous ammonia electrosynthesis. Nature 2024, 629, 92-7.
19. Dai, X.; Du, Z. Y.; Sun, Y.; et al. Enhancing green ammonia electrosynthesis through tuning Sn vacancies in Sn-based MXene/MAX hybrids. Nano-Micro. Lett. 2024, 16, 89.
20. Yu, Y.; Lv, Z.; Liu, Z.; et al. Activation of Ga liquid catalyst with continuously exposed active sites for electrocatalytic C−N coupling. Angew. Chem. Int. Ed. 2024, 63, e202402236.
21. Yao, Z.; Liu, S.; Liu, H.; et al. Pre-adsorbed H-assisted N2 activation on single-atom cadmium-O5 decorated In2O3 for efficient NH3 electrosynthesis. Adv. Funct. Mater. 2023, 33, 2209843.
22. Wu, T.; Melander, M. M.; Honkala, K. Coadsorption of NRR and HER intermediates determines the performance of Ru-N4 toward electrocatalytic N2 reduction. ACS. Catal. 2022, 12, 2505-12.
23. Zhang, S.; Jin, M.; Shi, T.; et al. Electrocatalytically active Fe-(O-C2)4 single-atom sites for efficient reduction of nitrogen to ammonia. Angew. Chem. Int. Ed. 2020, 59, 13423-9.
24. Li, Y.; Li, J.; Huang, J.; et al. Boosting electroreduction kinetics of nitrogen to ammonia via tuning electron distribution of single-atomic iron sites. Angew. Chem. Int. Ed. 2021, 60, 9078-85.
25. Ye, Z.; Jiang, Y.; Li, L.; Wu, F.; Chen, R. MOF-related electrocatalysts for sulfur reduction/evolution reactions: composition modulation, structure design, and mechanism research. eScience 2023, 3, 100107.
26. Zhao, S.; Pan, Y. Coordination structures design of single-atom catalysts for enhanced hydrogen evolution reaction. Sustain. Chem. Energy. Mater. 2025, 1, 100003.
27. Han, C.; Zhang, S.; Zhang, H.; et al. Metal-support interaction in single-atom electrocatalysts: a perspective of metal oxide supports. eScience 2024, 4, 100269.
28. Li, Y.; Liu, Y.; Liu, X.; et al. Fe-doped SnO2 nanosheet for ambient electrocatalytic nitrogen reduction reaction. Nano. Res. 2022, 15, 6026-35.
29. Huang, S.; Zhang, M.; Liu, Y. Preparation and NRR application of transition metal nanosheets on carbon nanofiber membranes. J. Phys:. Conf. Ser. 2021, 1948, 012222.
30. Zhao, H.; Yuan, Z. Progress and perspectives for solar-driven water electrolysis to produce green hydrogen. Adv. Energy. Mater. 2023, 13, 2300254.
31. Li, H.; Gan, K.; Li, R.; et al. Highly dispersed NiO clusters induced electron delocalization of Ni-N-C catalysts for enhanced CO2 electroreduction. Adv. Funct. Materials. 2023, 33, 2208622.
32. Li, P.; Fu, W.; Zhuang, P.; et al. Amorphous Sn/crystalline SnS2 nanosheets via in situ electrochemical reduction methodology for highly efficient ambient N2 fixation. Small 2019, 15, e1902535.
33. Xiong, W.; Guo, Z.; Zhao, S.; Wang, Q.; Xu, Q.; Wang, X. Facile, cost-effective plasma synthesis of self-supportive FeSx on Fe foam for efficient electrochemical reduction of N2 under ambient conditions. J. Mater. Chem. A. 2019, 7, 19977-83.
34. Niu, L.; Wang, D.; Xu, K.; et al. Tuning the performance of nitrogen reduction reaction by balancing the reactivity of N2 and the desorption of NH3. Nano. Res. 2021, 14, 4093-9.
35. Huang, C.; Shang, L.; Han, P.; et al. Electrochemical N2 fixation by Cu-modified iron oxide dendrites. J. Colloid. Interface. Sci. 2019, 552, 312-8.
36. Jin, F.; Yin, H.; Feng, R.; et al. Charge transfer and vacancy engineering of Fe2O3 nanoparticle catalysts for highly selective N2 reduction towards NH3 synthesis. J. Colloid. Interface. Sci. 2023, 647, 354-63.
37. Jiang, Y.; Xiao, N.; Song, X.; et al. Coal tar pitch derived sp2 configuration-dominated vacancy-rich carbon with expand interlayer spacing for low-voltage, durable, and fast potassium storage. Adv. Funct. Mater. 2024, 34, 2316207.
38. Xiang, X.; Wang, Z.; Shi, X.; Fan, M.; Sun, X. Ammonia synthesis from electrocatalytic N2 reduction under ambient conditions by Fe2O3 nanorods. ChemCatChem 2018, 10, 4530-5.
39. Tao, Y.; Wu, D.; Wang, T.; Ren, P.; Jia, D. A universal method of preparing high yield and N riched porous carbon by carbonizing coal tar pitch in air for supercapacitor. J. Power. Sources. 2023, 573, 233114.
40. Murphy, E.; Sun, B.; Rüscher, M.; et al. Synergizing Fe2O3 nanoparticles on single atom Fe-N-C for nitrate reduction to ammonia at industrial current densities. Adv. Mater. 2024, 36, e2401133.
41. Liu, J.; Liu, Y.; Nan, B.; et al. A two-in-one strategy to simultaneously boost the site density and turnover frequency of Fe-N-C oxygen reduction catalysts. Angew. Chem. Int. Ed. 2025, 64, e202425196.
42. Wu, T.; Zhu, X.; Xing, Z.; et al. Greatly improving electrochemical N2 reduction over TiO2 nanoparticles by iron doping. Angew. Chem. Int. Ed. 2019, 58, 18449-53.
43. Zhao, Y.; Tian, Z.; Wang, W.; Deng, X.; Tseng, J.; Wang, G. Size-dependent activity of Fe-N-doped mesoporous carbon nanoparticles towards oxygen reduction reaction. Green. Carbon. 2024, 2, 221-30.
44. Wang, X.; Qiu, S.; Feng, J.; et al. Confined Fe-Cu clusters as sub-nanometer reactors for efficiently regulating the electrochemical nitrogen reduction reaction. Adv. Mater. 2020, 32, e2004382.
45. Wang, C.; Hu, X.; Hu, X.; et al. Typical transition metal single-atom catalysts with a metal-pyridine N structure for efficient CO2 electroreduction. Appl. Catal. B-Environ. 2021, 296, 120331.
46. Kang, D.; Zhang, C.; Wang, X.; et al. Efficient atomically dispersed Fe catalysts with robust three-phase interface for stable seawater-based zinc-air batteries. Green. Carbon. 2025, 3, 1-10.
47. Wang, G.; Sun, X.; Bai, J.; Han, L. Preparation of Fe-C nanofiber composites by metal organic complex and potential application in supercapacitors. J. Mater. Sci. Mater. Electron. 2019, 30, 4665-75.
48. Wu, C.; Pei, Z.; Lv, M.; Huang, D.; Wang, Y.; Yuan, S. Polypyrrole-coated low-crystallinity iron oxide grown on carbon cloth enabling enhanced electrochemical supercapacitor performance. Molecules 2023, 28, 434.
49. Ying, H.; Chen, T.; Zhang, C.; Bi, J.; Li, Z.; Hao, J. Regeneration of porous Fe3O4 nanosheets from deep eutectic solvent for high-performance electrocatalytic nitrogen reduction. J. Colloid. Interface. Sci. 2021, 602, 64-72.
50. Zhang, T.; Qian, C.; Guo, P.; et al. A novel reduced graphene oxide-attapulgite (RGO-ATP) supported Fe2O3 catalyst for heterogeneous fenton-like oxidation of ciprofloxacin: degradation mechanism and pathway. Catalysts 2020, 10, 189.
51. Jiang, S.; Jiang, Z.; Li, C.; et al. Natural pitch-derived carbon networks induced lattice strain engineering in nickel-based heterostructures enables efficient anodes for sodium-ion batteries. Small 2025, 21, e2408011.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].