Construction of triazine/heptazine carbon nitride homojunction for photocatalytic reduction of high-concentration 4-nitrophenol
Abstract
Homogeneous carbon nitride (C3N4) photocatalysts with their suitable electronic structure, environmental benignity, and outstanding chemical stability properties have been exhibiting excellent performances in various fields. However, their mechanism is still relatively unclear, and there is limited research on treating high-concentration water pollutants. This work presents a crystallized carbon nitride homojunction (HCN) catalyst originating from triazine-based C3N4 and bulk phenazine-based C3N4 (BCN) via one-step calcination. As a result, the optimized HCN first demonstrates significantly enhanced photocatalytic reduction efficiency (≈1.44 min-1) towards 150 mg/L high-concentration 4-nitrophenol (4-NP) under visible light irradiation, up to two-fold better than BCN. The concentration and pH were meticulously adjusted, accompanied by a thorough exploration of the underlying mechanisms. Subsequently, comprehensive verification was conducted to confirm the existence of a type-II homojunction and the establishment of an interfacial electric field in HCN, which serve as potent facilitators for the separation and migration of photo-excited carriers. This novel study provides a comprehensive understanding of homojunction catalyst design and advances the development of efficient metal-free photocatalysts for degrading high-concentration pollutants.
Keywords
INTRODUCTION
Nowadays, freshwater contamination due to organic molecules from dye and textile industries has seriously threatened the human body and become an increasingly urgent global issue[1]. A variety of methods have been proposed for wastewater treatment, such as membrane filtration, flocculation, coagulation, adsorption, and reduction[2,3]. Unfortunately, a majority of these methods generally require high temperatures and pressurized hydrogen, involve noble metal-based catalysts, and quickly produce secondary pollution[4,5]. In this regard, photocatalysis is considered a potential alternative since it can utilize the readily available light energy to trigger organic molecule conversion into hydrocarbon products or fuels under mild conditions[6,7].
As a highly prevalent organic contaminant in wastewater, 4-nitrophenol (4-NP) has garnered significant attention owing to its concerning properties, including potential carcinogenic, mutagenic, and bactericidal effects, posing a serious threat to environmental and human health[8,9]. However, its corresponding reduction product, 4-aminophenol (4-AP), is an irreplaceable raw material in the chemical industry to produce polymers, pharmaceuticals, and agrochemicals[3,10]. Numerous efforts have been devoted to the photocatalytic reduction of 4-NP since semiconductor materials facilitate the absorption of solar energy, which excites photoelectrons to reduce 4-NP to 4-AP[11]. For instance, Wu et al. anchored noble metal (Ag, Au) nanoparticles on SnNb2O6 nanosheets and effectively enhanced the photocatalytic activity of 4-NP conversion by improving visible-light absorption in the noble metal-semiconductor composites[12]. Liu et al. synthesized nitrogen-doped graphene quantum dots (N-GQDs), which exhibited excellent catalytic activity for the conversion of 4-NP to 4-AP under near-infrared (NIR) light irradiation due to their superb upconverted photoluminescence (PL) and photothermal conversion ability[13]. However, current endeavors are primarily conducted under low-concentration 4-NP conditions below 20 mg/L, while the photocatalytic reduction of high-concentration 4-NP relies on the involvement of precious metals. This is mainly ascribed to the inferior photocatalytic performance due to unsatisfactory material design, such as excessive defects, mismatched interfaces, rapid carrier recombination, and slow electronic transmission of semiconductor photocatalysts[14-18]. Hence, the rational design and synthesis of effective metal-free photocatalysts with excellent degradation capability for high-concentration pollutants is of importance.
Graphene carbon nitride (g-C3N4), as an emerging nonmetallic semiconductor photocatalyst, has attracted widespread scientific attention[19,20]. Nevertheless, the severe recombination of photogenerated carriers and low active sites dramatically limits its application in the photocatalytic reduction of 4-NP[21,22]. The construction of type-II or Z-scheme C3N4-based heterojunctions with appropriate energy levels to establish a built-in electric field (BIEF) is commonly employed to enhance carrier separation[23-25]. However, achieving precise structural manipulation from the molecular level remains challenging due to the typical preparation of heterojunction materials through bulk phase integration[26]. Consequently, the fabrication of homojunction is the direct and straightforward route to enhance the charge separation without introducing heteroatoms[27,28]. Many efforts have been adopted to construct the g-C3N4-based homojunction[29-32]. For example, Das et al. have showcased an effective hierarchical g-C3N4 homojunction photocatalyst to achieve superior photocatalytic degradation efficiency of antibiotic pollutants in water[33]. Zeng et al. initially reported the formation of a crystalline carbon nitride phase junction, where triazine-based C3N4 (TCN) is vertically aligned on tri-s-TCN[34], demonstrating a significantly enhanced hydrogen evolution rate (HER) under visible light irradiation. So far, the crystallized carbon nitride homojunction (HCN) has emerged as an exemplary photocatalyst for reducing high-concentration pollutants due to its exceptional crystallinity, optimal alignment at the homojunction interface, and efficient charge separation. Nevertheless, the exploration on photocatalytic reduction of high-concentration 4-NP remains limited, and revealing the electron-transfer mechanism at the homojunction interface still needs to be reached.
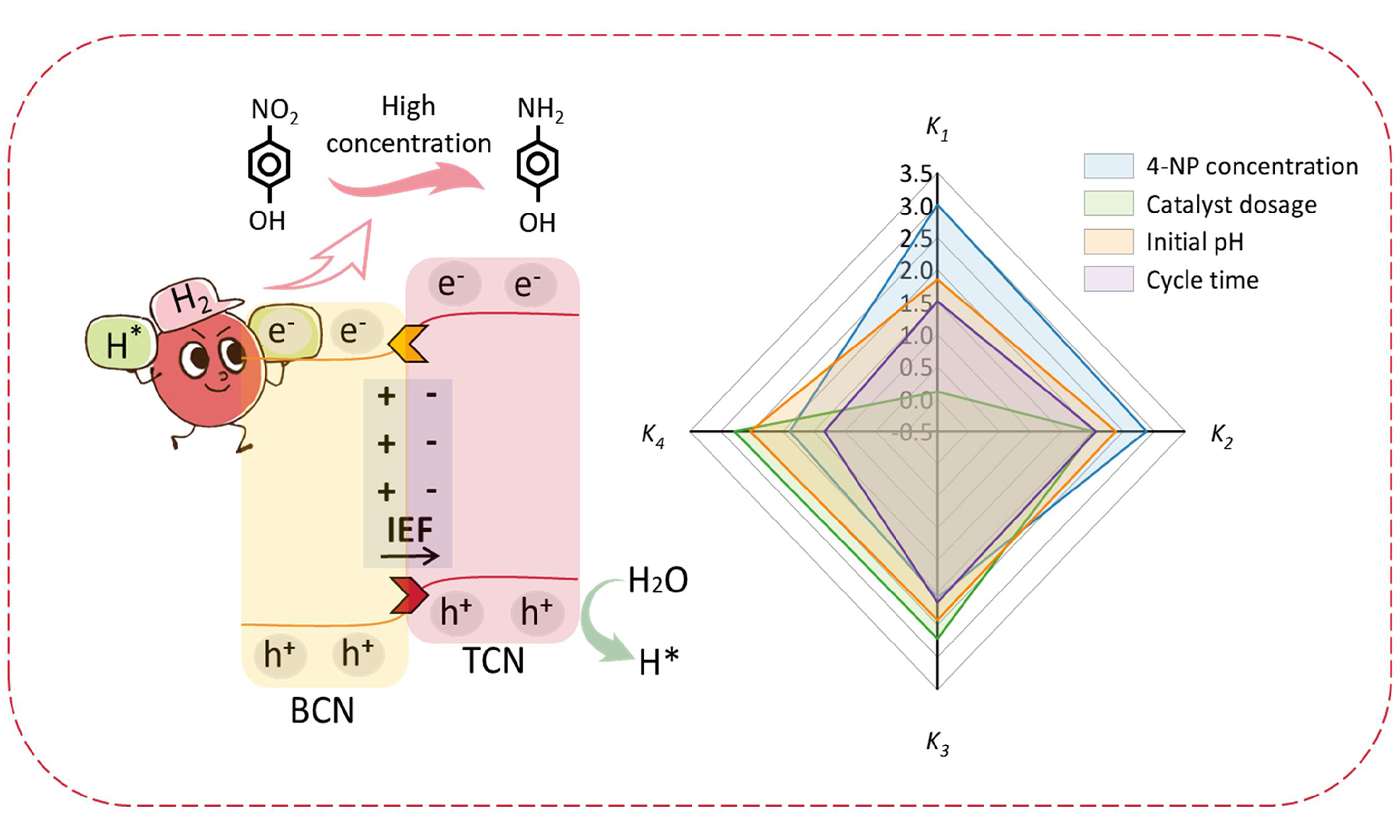
Herein, we present a type of HCN catalyst via a one-step calcination process. Such HCN comprises triazine/heptazine with high crystallinity and a blend of rod and sheet morphology. Due to the introduction of the triazine phase in HCN, such homojunction shows satisfactory photocatalytic activity and stability for reducing high-concentration 4-NP to 4-AP, including a high-rate constant (1.44 min-1) and the cycle stability up to four times. Interestingly, 4-NP with high concentrations of up to 200 mg/L can be rapidly and entirely reduced into 4-AP using the HCN catalyst, which is far better than reported C3N4-based photocatalysts. Furthermore, the separation/migration of the photo-excited carriers and band structures in HCN and bulk phenazine-based C3N4 (BCN) were studied, elaborating on a type-II homojunction photocatalyst with BIEF that was ingeniously constructed by utilizing two distinct carbon nitrides with a significant disparity in work function. Moreover, such photocatalytic reduction of 4-NP at different concentrations, catalyst amounts, and pH values were further investigated, showing that metal-free HCN boasts many applications.
EXPERIMENTAL
Chemicals
Melamine (C3H6N6) was purchased from the Tianjin Damao Chemical Reagent Factory. Additionally, 4-NP and sodium borohydride (NaBH4) were obtained from Shanghai Macklin Biochemical Technology Co., Ltd. All chemicals were purchased and used without any further purification.
Synthesis of BCN and HCN
Typically, 5.0 g melamine was calcinated at 500 °C for 2 h at a rate of 5 °C/min in a muffle furnace. The product was then prepared using the method described above and finally recorded as HCN. Similarly, 8.0 g of melamine was calcinated in a muffle furnace at a rate of 12 °C/min, reaching a temperature of 500 °C and maintained for 4 h. The resultant yellow solids were subsequently obtained and pulverized into powder. Then, these solids were washed with deionized water and ethanol repeatedly and dried under vacuum at
Structural characterizations
The microstructures were characterized by a scanning electron microscopy (SEM, Regulus 8100 with voltage 10 kV) and high-resolution transmission electron microscopy (HRTEM, FEI Tecnai F20 S-Twin). The crystal structures and elemental compositions were studied with an X-ray diffractometer (XRD, PANalytical Empyrean) operated with Cu Kα source by scanning at angles of 2θ = 5°-80°. Chemical valence states were characterized by X-ray photoelectron spectroscopy (XPS, Thermo ESCALAB 250XI). The specific surface areas and pore structures of materials were measured by the Brunauer-Emmett-Teller (BET, Micromeritics 3-Flex) under N2 adsorption-desorption isotherms. An electron paramagnetic resonance spectrometer (EPR, Bruker EMXplus) was used to monitor the radical trapping at room temperature with excitation at 420 nm. The Fourier transform infrared spectroscopy (FT-IR, Bruker VERTEX 70 V) was employed to identify the molecular structures. The Ultraviolet-visible spectrophotometry (UV-Vis, Shimadzu UV-2600) was employed to determine the absorbance of liquid samples.
Photocatalytic performance
The photocatalytic activities of samples were evaluated by reducing 4-NP aqueous solution (150 mg·L-1) under irradiation of a 300 W xenon lamp equipped with a 420 nm cutoff filter. Typically, 10 mg of photocatalyst was added to 30 mL of the above 4-NP solution, including 2.0 mL of 0.5 mol/L NaBH4. Before irradiation, the suspension was stirred for 30 min in the dark to establish the absorption-desorption equilibrium between the surface of the photocatalysts and 4-NP. A photochemical reaction apparatus (BL-GHX-V, Shanghai Bilon Instrument Co., Ltd.) was used to stir and irradiate samples. The reaction temperature was maintained at 25 °C with a stirring speed of 800 r/min, and the stirring duration for different samples was selected according to their reaction rates. About 3.0 mL aliquot dispersions were sampled during given time intervals, followed by filtration to remove the catalysts. A Shimadzu UV-2600 spectrophotometer was used to analyze the filtrates, and the characteristic absorption peak of the 4-NP reduction at 400 nm was used to determine the extent of its reduction. Considering that the initial concentration of NaBH4 greatly exceeds that of 4-NP, the photoreduction reaction can be described by the pseudo-first-order kinetics[12]. The catalytic performance can be evaluated by the apparent rate constant k, which is determined using
where C0 and Ct represent the 4-NP concentration before and after the visible light irradiation, and t is the irradiation time.
Photoelectrochemical measurements
PL spectra (JSX-3400R) were employed to assess the recombination ability of carriers at an excitation wavelength of 360 nm. Electrochemical impedance spectroscopy (EIS), transient photocurrent response
RESULTS AND DISCUSSION
Design and synthesis of carbon nitride homojunction
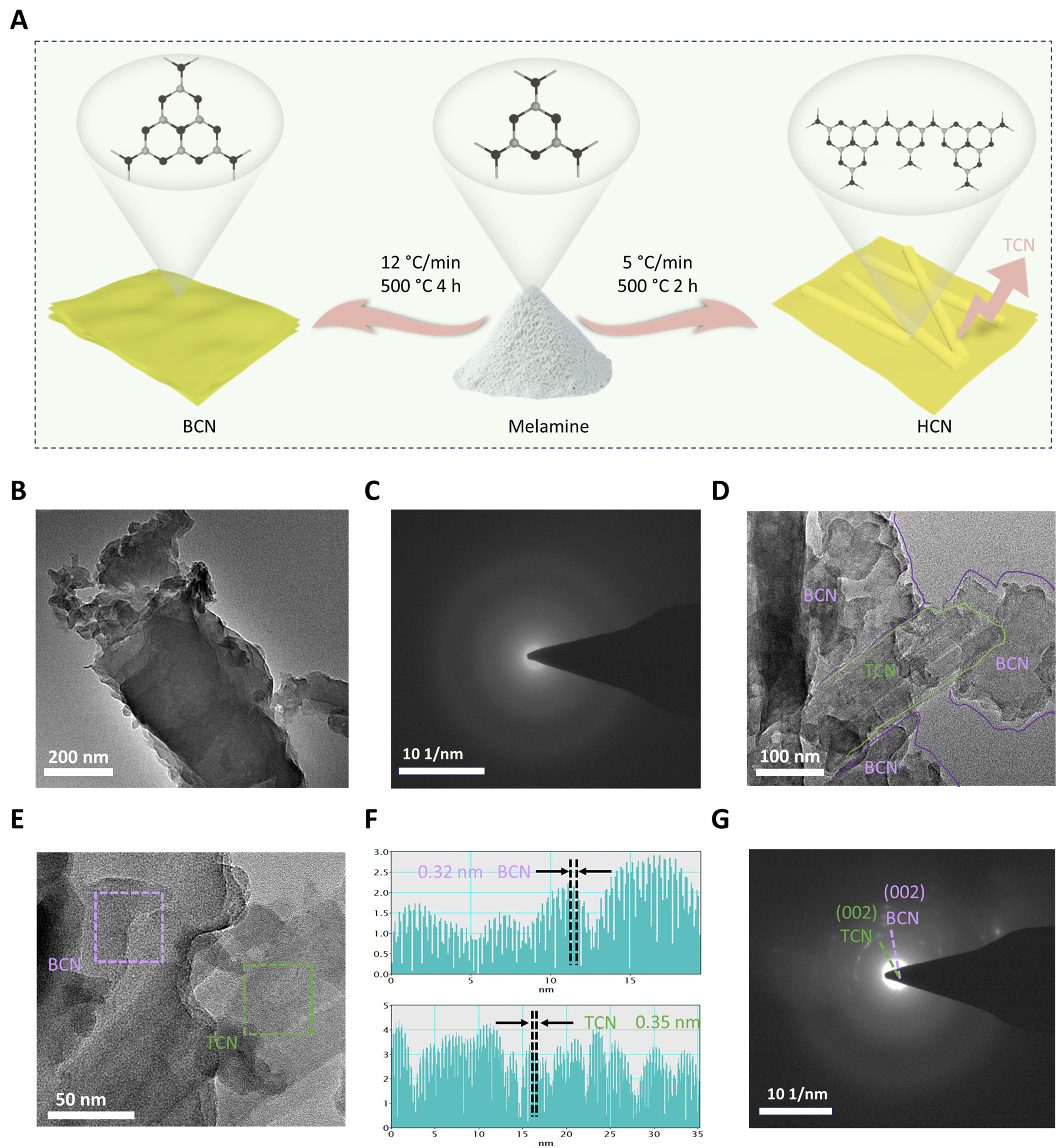
Melamine was used as a precursor, and two different types of graphite carbon nitride were obtained by adjusting the reaction time and heating rate for a fixed calcination temperature at 500 °C. In the prepared carbon nitride, ensuring the uniformity of heptazine units is challenging; typically, triazine units persist in the scaffold during the thermal polymerization, leading to the in-plane being less unified[35]. As shown in Figure 1A, rapid thermodynamic polymerization yields bulk BCN, while a slow thermal process forms HCN consisting of triazine and phenazine groups.
Figure 1. Synthesis and characterizations. (A) Schematic illustration for the synthetic process of the HCN and BCN; (B and C) TEM image and SAED pattern of the BCN after calcined melamine at 500 °C for 4 h; (D and E) TEM images of the HCN after calcined melamine at 500 °C for 2 h; (F) lattice profile, and (G) SAED pattern of TCN and BCN. HCN: Crystallized carbon nitride homojunction; BCN: bulk phenazine-based C3N4; TEM: transmission electron microscopy; SAED: selected area electron diffraction; TCN: triazine-based C3N4.
As seen from the scanning electron microscopy (SEM) images in Supplementary Figure 1, both BCN and HCN exhibit the typical layered structures, except bundles of rod-shaped particles appear on the layer in HCN. As observed in the transmission electron microscopy (TEM) images [Figure 1B], the synthesized BCN shows a stacked-layer structure attributed to weak interlayer Van der Waals bonding and the strong covalent C–N bonds in each layer[36]. The amorphous halo in the selected area electron diffraction (SAED) pattern reveals the low crystallinity of BCN [Figure 1C]. In contrast, the HCN presents two morphologies, including rod-like particles and much thinner layers [Figure 1D and E]. Furthermore, the obtained lattice profiles [Figure 1F] of rod-shaped particles and thin sheets are measured as 0.35 and 0.33 nm, corresponding to the (002) planes of TCN and BCN, respectively. In Figure 1G, the diffraction points belonging to the (002) planes of TCN and BCN can also be identified from the SAED pattern[18].
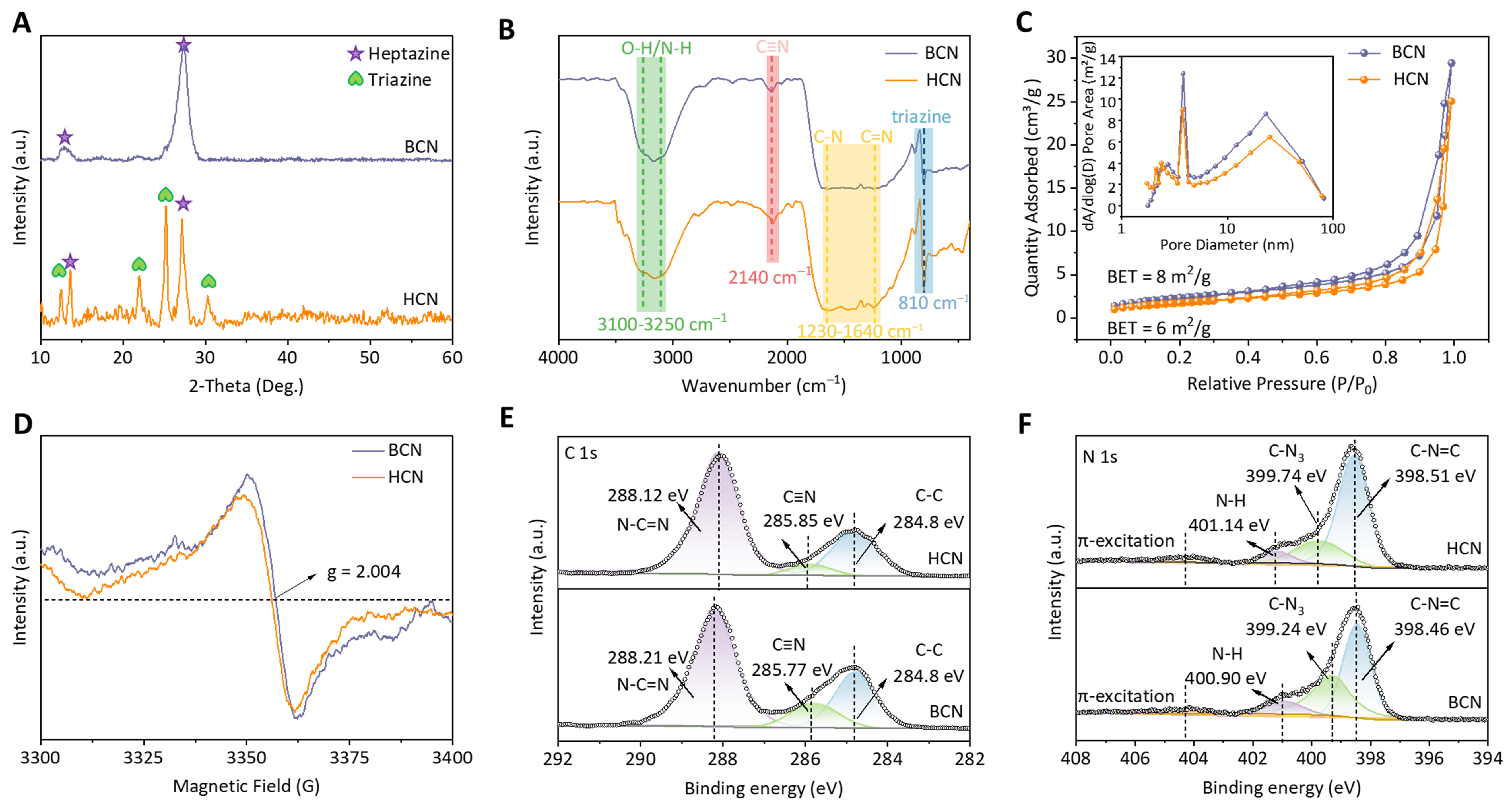
X-ray diffraction (XRD) patterns in Figure 2A show that the HCN and BCN possess two typical diffraction peaks located at 13° and 27°, consistent with the interplanar packing (100) and π-π interlayer stacking (002) of heptazine structure[37]. Notably, apart from the two prominent peaks of heptazine units, four diffraction peaks at 14°, 21°, 25°, and 30° are also observed over HCN, matching with (100), (110), (002), and (102) planes of triazine units, respectively, suggesting that the HCN consists of a combination of homogeneous triazine and heptazine. The crystallinity of BCN calculated by XRD is 92%, which is larger than that of HCN (53%). The above results further prove that HCN consists of a combination of homogeneous triazine and heptazine. From the FTIR spectra in Figure 2B, distinct peaks corresponding to the vibrational modes of
Figure 2. Physicochemical characterizations. (A) XRD patterns; (B) FT-IR spectra; (C) N2 sorption isotherms and pore size distributions; (D) ESR spectra of the HCN and BCN. High-resolution XPS spectra of (E) C 1s, (F) N 1s of the HCN and BCN. XRD: X-ray diffraction; FT-IR: Fourier transform infrared spectroscopy; ESR: electron spin resonance spectra; XPS: X-ray photoelectron spectroscopy; HCN: crystallized carbon nitride homojunction; BCN: bulk phenazine-based C3N4.
In the N2 adsorption-desorption curves of Figure 2C, all samples displayed type IV adsorption isotherms accompanied by H3-type hysteresis loops, suggesting the existence of mesoporous pores, which may be ascribed to the re-release of volatile gases from the thermal polymerization process. The measured specific surface areas for BCN and HCN are calculated to be 8 and 6 m2·g-1, respectively, which may be caused by the smooth lamellar structure. In addition, relevant experiments involving electron spin resonance spectra (ESR) were performed to investigate the spin state of unpaired electrons. It is observed that the BCN exhibits a higher peak intensity than the HCN, providing evidence for a more significant number of defects in BCN [Figure 2D][43]. Compared with the structure of BCN, the lower porosity and fewer edge defects in HCN result from the pore size distribution in the inset of Figure 2C. As shown from the survey XPS spectra [Supplementary Figure 2], the C and N peaks dominate in these two samples. In the C 1s spectra [Figure 2E], three peaks at 284.80, 285.77, and 288.21 eV are observed in the BCN, assigned to adventitious hydrocarbons (C–C), cyano group (C≡N), and N–C=N of the heptazine unit, respectively. The high-resolution N 1s spectra of BCN [Figure 2F] exhibit four peaks at 398.46 eV (sp2-coordinated nitrogen, C–N=C), 399.24 eV (tertiary nitrogen, C–N3), 400.90 eV (terminal N–Hx group), and 404.02 eV (charge effect or π excitations). Notably, the peaks for sp2-coordinated N–C=N and C–N=C are enhanced in HCN due to the improved crystallinity and reduced defects compared to BCN. Besides, the main peak of N 1s moves to higher binding energy after introducing TCN in BCN, indicating the shift of local electrons from heptazine units (BCN) to triazine neighbors (TCN).
Photocatalytic reduction 4-NP
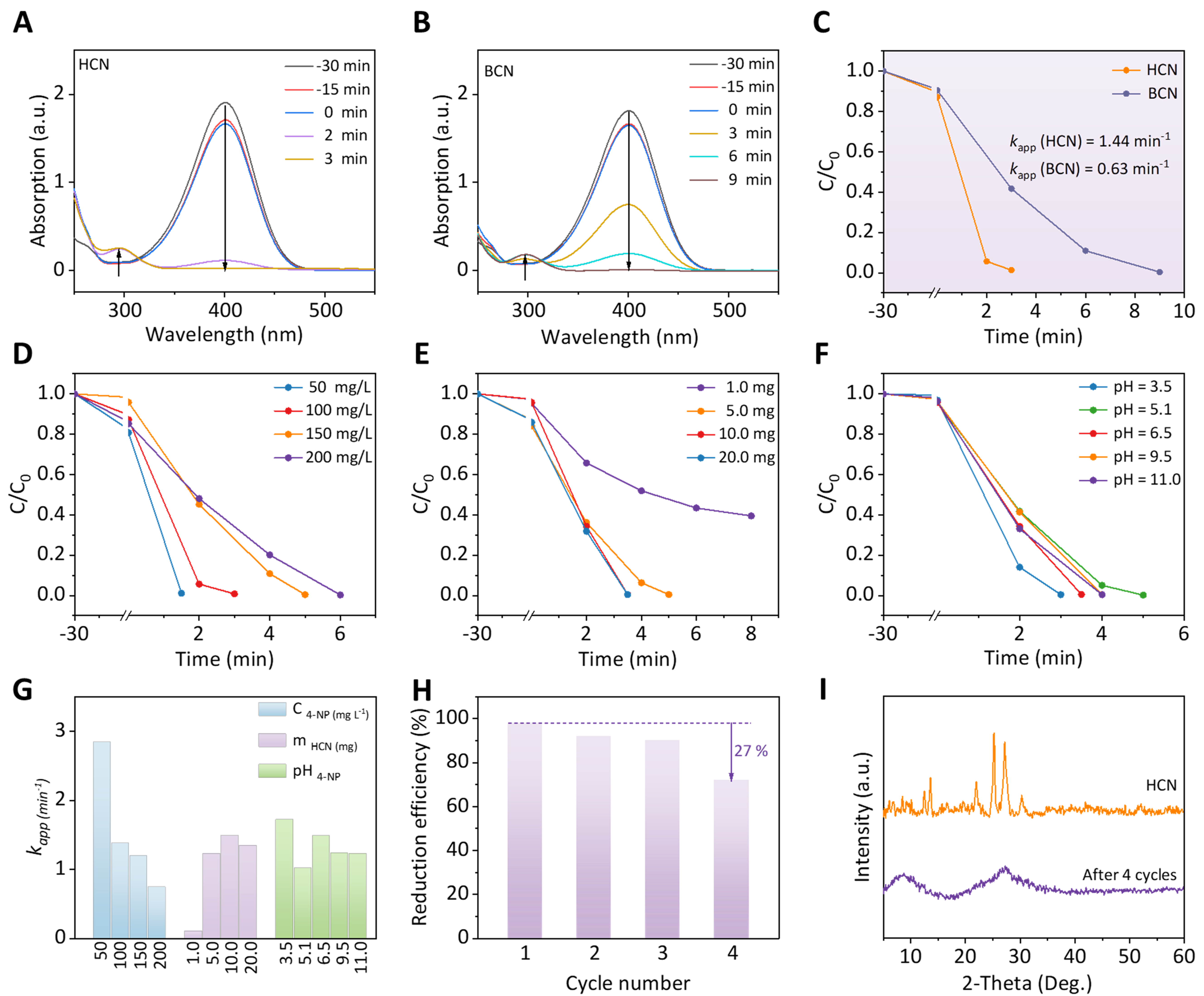
To exemplify the potential use, the catalytic properties of HCN and BCN samples were evaluated by the reduction of 4-NP to 4-AP with NaBH4. As shown in Supplementary Figure 3A, the UV-Vis peak of 4-NP in pure water is located at 317 nm, whereas it shifts to 400 nm upon the addition of NaBH4. Accordingly, the color of the 4-NP solution changes from light yellow to bright yellow, attributed to the formation of nitrophenolate ions in an alkaline solution[44]. In Supplementary Figure 3B and C, the absorption peak of 4-NP remains unchanged without catalysts or NaBH4, demonstrating that the catalyst is active under NaBH4 to reduce 4-NP. As indicated in Figure 3A-C, the conversion rate of 4-NP with no HCN or BCN is only 11.2% or 8.7% in the dark after 30 min, even with the addition of catalyst and NaBH4, indicating that illumination is necessary. Although both BCN and HCN can ultimately reduce 4-NP (150 mg/L), the reduction rate of 4-NP by HCN sharply increased to 1.44 min-1, compared to the efficiency (k = 0.63 min-1) of BCN, indicating that the incorporation of TCN into BCN can considerably improve the catalytic activity.
Figure 3. Photocatalytic measurements. UV-Vis absorption spectra for 4-NP reduction with (A) HCN and (B) BCN; (C) Photocatalytic reduction plots of 4-NP for HCN and BCN under visible light; Photocatalytic reduction plots of 4-NP with HCN under visible light at (D) different concentrations, (E) catalyst amounts, and (F) different pH values; (G) Summary of Kapp values under different reaction conditions; (H) Recyclability of HCN as a photocatalyst in the reduction of 4-NP under visible light irradiation; (I) XRD comparison of the HCN catalyst before and after four cycles. UV-Vis: Ultraviolet-visible; 4-NP: 4-nitrophenol; HCN: crystallized carbon nitride homojunction; BCN: bulk phenazine-based C3N4; XRD: X-ray diffraction.
Meanwhile, the effects of the concentration of 4-NP substrate, the amount of catalyst added, and the pH of the reaction on the photocatalytic properties were further investigated. As shown in Figure 3D and Supplementary Figure 4, the HCN catalyst can achieve complete conversion of 4-NP to 4-AP within a wide concentration range, but the degradation rate slightly decreases with increasing concentration of 4-NP. Treating 200 mg/L of 4-NP only takes about 7 min, which is better than many metal-free catalysts and comparable to some precious metal-based catalysts, as shown in Supplementary Table 1. Under different dosages of catalyst [Figure 3E and Supplementary Figure 5] and pH conditions [Figure 3F and Supplementary Figure 6], HCN catalyst with a smaller amount (10.0 mg) can achieve an excellent photocatalytic reduction of high-concentration of 4-NP (150 mg/L) over a more comprehensive pH range (3.5-11.0). Apparent rate constants (k) for photocatalytic reduction of 4-NP with HCN under visible light at different concentrations, catalyst amounts, and other pH values can be shown in Figure 3G and Supplementary Table 2. Therefore, the metal-free HCN boasts a vast array of applications at various concentrations, catalyst amounts, and pH values.
Moreover, the reusability of the HCN catalyst was explored, and the results are shown in Supplementary Figure 7 and Figure 3H. After the fourth experiment, the reduction efficiency of HCN only decreased by 27% within 6 min, demonstrating that HCN has good reusability. As can be seen from the XRD pattern in Figure 3I, the crystallinity of the recycled HCN sample is significantly reduced, and the peak of TCN is difficult to observe. Compared to the original morphology in Figure 1D, irregular two-dimensional layers without rod-like particles and amorphous halo patterns are shown in the TEM and SAED images
Photoelectrochemical properties
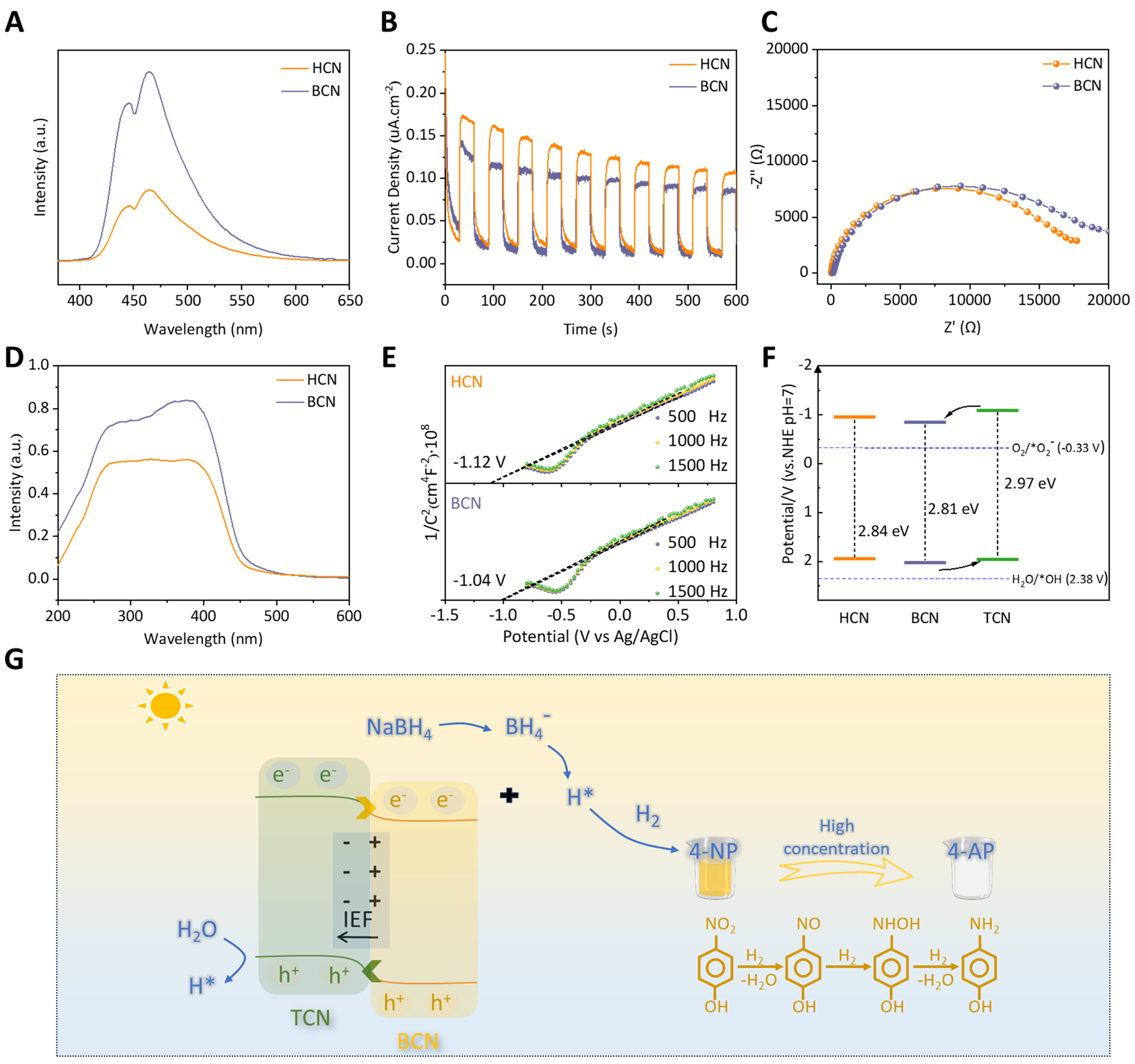
To reveal the mechanistic details of the elevated photocatalytic activity, a series of photoelectrochemical measurements were undertaken to gain insight into the separation and transfer of charge carriers. PL studies [Figure 4A] show that the HCN sample has a quenching emission relative to the BCN in 446 and 464 nm, indicating the inhibited recombination of photogenerated electron-hole pairs due to the formation of homojunction structure[45]. Figure 4B shows the transient photocurrent response of HCN and BCN under intermittent visible light irradiation. There is an increased transient photocurrent response of HCN compared to that of the single BCN, suggesting the accelerated separation efficiency of the charge carriers and improved photocatalytic activity for the HCN. As shown in Figure 4C, the Nyquist diagram of HCN shows a smaller semicircle than bare BCN, confirming the homojunction’s superior photogenerated charge-transfer dynamics and interfacial charge separation[46]. Furthermore, we employed time-resolved surface photovoltage (TPV) measurement in Supplementary Figure 9; the HCN exhibits an average lifetime of
Figure 4. Photoelectrochemical properties and mechanism. (A) PL spectra, (B) Transient photocurrent density, (C) EIS curves, (D) UV-Vis spectra, and (E) M-S plots of the BCN and HCN; (F) Band gap structure of BCN, TCN, and HCN; (G) The mechanism illustration for photoreduction of 4-NP using the type-II HCN homojunction photocatalyst. PL: Photoluminescence; EIS: electrochemical impedance spectroscopy; UV-Vis: ultraviolet-visible; M-S: Mott-Schottky; BCN: bulk phenazine-based C3N4; HCN: crystallized carbon nitride homojunction; TCN: triazine-based C3N4; 4-NP: 4-nitrophenol.
and the prolonged lifespan of charge carriers illustrates the enhanced possibility of their participation in photocatalytic reactions before recombination[47-49].
In Figure 4D, the BCN and HCN have strong absorption peaks owing to the π → π* electronic transition in the visible light range. As seen from the Tauc plots shown in Supplementary Figure 10, the HCN and BCN band gaps are calculated to be 2.84 and 2.81 eV, respectively. Moreover, the MS plots of HCN and BCN have positive slopes [Figure 4E], corresponding to n-type nature[50]. Conduction band energy (ECB) values of HCN and BCN are found to be -1.12 and -1.04 V, relative to Ag|AgCl. These values could be converted to
Photocatalytic mechanism
Based on the structural measurements, the homojunction HCN is formed by layered BCN loaded with rod-like TCN. The mechanism for photoreduction of 4-NP using the type II HCN homojunction photocatalyst is shown in Figure 4G. Under visible light irradiation, due to IEF from BCN to TCN, electrons (e-) in the CB of TCN rapidly migrate to the CB of BCN, while holes (h+) in the VB of BCN shift to the TCN. The formation of type-II homojunction significantly enhances the separation of photogenerated electron-hole pairs. Firstly, 4-NP and NaBH4 are all adsorbed on the surface of catalysts. Subsequently, NaBH4 is converted to BH4- in an aqueous solution, a hydrolysis reaction generates active hydrogen (H*) and then combines with the electron (e-) to generate H2. The H2 produced here is released onto the catalyst’s surface and further, a reduction reaction occurs with 4-NP[53]. In addition, the hole (h+) is transferred from the valence band of BCN to TCN to oxidize H2O to form H*. Consequently, these abundant active H* species derived from NaBH4 and H2O within the homojunction are crucial in reducing highly concentrated 4-NP molecules into 4-AP[13,54].
Implications of the structure design
The dynamics of photogenerated charge carriers are regarded as pivotal in attaining photocatalysis with remarkable efficiency. One effective strategy for enhancing photocatalytic efficiency involves creating an IEF, which reduces lattice mismatch and increases photogenerated charge density. An n-n type homojunction photocatalyst with IEF was ingeniously constructed by utilizing two distinct carbon nitrides with a significant disparity in work function. Incorporating TCN into BCN induces a redistribution of charges, resulting in the formation of BIEF at the interface. Consequently, the band alignment of TCN exhibited downward bending while that of BCN displayed upward bending. Under illumination, a BIEF facilitates the transfer and separation of photogenerated holes (h+) from BCN to TCN, while simultaneously promoting the migration of photogenerated electrons (e-) from TCN to BCN. On the one hand, this facilitates electronic transfer at the interface, precisely regulating the charge density surrounding active sites and optimizing the adsorption and activation of 4-NP. On the other hand, the resulting space charge region can expedite reaction kinetics and alleviate high recombination rates and low photocatalytic efficiency. However, the charge transfer mechanism underlying homogeneous junction formation remains enigmatic and necessitates further exploration. Experimental and theoretical results have substantiated that the work function difference between TCN and BCN makes it possible to implement II-scheme charge separation driven by an IEF at the interface. The successful establishment of a type-II homojunction has achieved the dual benefits of circumventing impurity introduction and lattice mismatch, thus simultaneously enhancing the efficiency of photogenerated charge carrier separation and transfer.
CONCLUSIONS
In summary, a novel HCN homojunction formed by triazine-based TCN and phenazine-based BCN is synthesized through a facile one-step calcination procedure. Through a series of characterization and density functional theory (DFT) calculations, establishing a type-II homojunction and generating an IEF serve as effective measures to enhance the spatial segregation and proficient migration of photo-induced charge carriers. As a result, the prepared HCN has exhibited a high-rate constant (1.44 min-1) up to two-fold better than BCN at a high 4-NP concentration of 150 mg/L and good reusability for four cycles. The excellent photocatalytic performance can primarily account for the limited defects, good matched interface, efficient charge separation, and fast electronic transmission in the homojunction structure. Additionally, the metal-free HCN boasts a vast array of applications at different concentrations, catalyst amounts, and pH values. This study offers valuable insights into the design of advanced, metal-free photocatalytic platforms for sustainable light-driven applications, especially in high-concentration wastewater treatment.
DECLARATIONS
Authors’ contributions
Writing-original draft, characterization, data curation, formal analysis: Tong, H.
Data curation, formal analysis: Zhang, J.
Editing: Wang, W.
Resources: Yang, L.; Zhang, Z.
Investigation: Jia, Q.
Discussed and revised the manuscript and provided administrative support: Lan, K.; Chai, Z.
Availability of data and materials
The raw data supporting the findings of this study are available within this Article and its Supplementary Materials. Further data is available from the corresponding authors upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (22066018, 22205118, and 22375108), Young Talents of Science and Technology of Inner Mongolia (NJYT23036 and NJYT23037), Major Basic Research and Open Project of Inner Mongolia (30500-515330303), “Junma” Program of Inner Mongolia University, Grassland Talent Program of Inner Mongolia, and Natural Science Foundation of Inner Mongolia (2023JQ06 and 2024LHMS02001). We also thank the Electron Microscopy Centre of Inner Mongolia University for the microscopy and microanalysis of our specimens.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. He, C.; Liu, Z.; Wu, J.; et al. Future global urban water scarcity and potential solutions. Nat. Commun. 2021, 12, 4667.
2. Long, C.; Jiang, Z.; Shangguan, J.; Qing, T.; Zhang, P.; Feng, B. Applications of carbon dots in environmental pollution control: a review. Chem. Eng. J. 2021, 406, 126848.
3. Chishti, A. N.; Ma, Z.; Liu, Y.; et al. Synthesis of highly efficient and magnetically separable Fe3O4@C-TiO2-Ag catalyst for the reduction of organic dyes and 4-nitrophenol. Colloid. Surfaces. A. 2021, 631, 127694.
4. Gao, D.; Zhang, X.; Dai, X.; et al. Morphology-selective synthesis of active and durable gold catalysts with high catalytic performance in the reduction of 4-nitrophenol. Nano. Res. 2016, 9, 3099-115.
5. Narayanan, R.; El-Sayed, M. A. Effect of colloidal catalysis on the nanoparticle size distribution: dendrimer−Pd vs PVP−Pd nanoparticles catalyzing the Suzuki coupling reaction. J. Phys. Chem. B. 2004, 108, 8572-80.
6. Cheruvathoor Poulose, A.; Zoppellaro, G.; Konidakis, I.; et al. Fast and selective reduction of nitroarenes under visible light with an earth-abundant plasmonic photocatalyst. Nat. Nanotechnol. 2022, 17, 485-92.
7. Liao, G.; Tao, X.; Fang, B. An innovative synthesis strategy for high-efficiency and defects-switchable-hydrogenated TiO2 photocatalysts. Matter 2022, 5, 377-9.
8. Sarkar, S.; Balisetty, L.; Shanbogh, P. P.; Peter, S. C. Effect of ordered and disordered phases of unsupported Ag3In nanoparticles on the catalytic reduction of p-nitrophenol. J. Catal. 2014, 318, 143-50.
9. Sahiner, N.; Ozay, H.; Ozay, O.; Aktas, N. A soft hydrogel reactor for cobalt nanoparticle preparation and use in the reduction of nitrophenols. Appl. Catal. B. Environ. 2010, 101, 137-43.
10. Huang, C.; Ye, W.; Liu, Q.; Qiu, X. Dispersed Cu2O octahedrons on h-BN nanosheets for p-nitrophenol reduction. ACS. Appl. Mater. Interfaces. 2014, 6, 14469-76.
11. Hu, H.; Xin, J. H.; Hu, H.; Wang, X. Structural and mechanistic understanding of an active and durable graphene carbocatalyst for reduction of 4-nitrophenol at room temperature. Nano. Res. 2015, 8, 3992-4006.
12. Wu, J.; Wang, J.; Wang, T.; et al. Photocatalytic reduction of p-nitrophenol over plasmonic M (M = Ag, Au)/SnNb2O6 nanosheets. Appl. Surf. Sci. 2019, 466, 342-51.
13. Liu, H.; Wang, H.; Qian, Y.; et al. Nitrogen-doped graphene quantum dots as metal-free photocatalysts for near-infrared enhanced reduction of 4-nitrophenol. ACS. Appl. Nano. Mater. 2019, 2, 7043-50.
14. She, X.; Xu, H.; Xu, Y.; et al. Exfoliated graphene-like carbon nitride in organic solvents: enhanced photocatalytic activity and highly selective and sensitive sensor for the detection of trace amounts of Cu2+. J. Mater. Chem. A. 2014, 2, 2563.
15. Dong, G.; Zhao, K.; Zhang, L. Carbon self-doping induced high electronic conductivity and photoreactivity of g-C3N4. Chem. Commun. 2012, 48, 6178-80.
16. Liang, Q.; Li, Z.; Huang, Z.; Kang, F.; Yang, Q. Holey graphitic carbon nitride nanosheets with carbon vacancies for highly improved photocatalytic hydrogen production. Adv. Funct. Mater. 2015, 25, 6885-92.
17. Vinu, A. Two-dimensional hexagonally-ordered mesoporous carbon nitrides with tunable pore diameter, surface area and nitrogen content. Adv. Funct. Mater. 2008, 18, 816-27.
18. Li, F.; Yue, X.; Liao, Y.; Qiao, L.; Lv, K.; Xiang, Q. Understanding the unique S-scheme charge migration in triazine/heptazine crystalline carbon nitride homojunction. Nat. Commun. 2023, 14, 3901.
19. Li, Y.; Gao, C.; Long, R.; Xiong, Y. Photocatalyst design based on two-dimensional materials. Mater. Today. Chem. 2019, 11, 197-216.
20. Wang, Y.; Liu, L.; Ma, T.; Zhang, Y.; Huang, H. 2D graphitic carbon nitride for energy conversion and storage. Adv. Funct. Mater. 2021, 31, 2102540.
21. Li, A.; Cao, Q.; Zhou, G.; et al. Three-phase photocatalysis for the enhanced selectivity and activity of CO2 reduction on a hydrophobic surface. Angew. Chem. Int. Ed. Engl. 2019, 58, 14549-55.
22. Ong, W. J.; Tan, L. L.; Ng, Y. H.; Yong, S. T.; Chai, S. P. Graphitic carbon nitride (g-C3N4)-based photocatalysts for artificial photosynthesis and environmental remediation: are we a step closer to achieving sustainability? Chem. Rev. 2016, 116, 7159-329.
23. Pan, Z.; Zhang, G.; Wang, X. Polymeric carbon nitride/reduced graphene oxide/Fe2O3: all-solid-state Z-scheme system for photocatalytic overall water splitting. Angew. Chem. Int. Ed. Engl. 2019, 58, 7102-6.
24. Xu, Q.; Zhang, L.; Cheng, B.; Fan, J.; Yu, J. S-scheme heterojunction photocatalyst. Chem 2020, 6, 1543-59.
25. Zhang, Y.; Cao, Q.; Meng, A.; et al. Molecular heptazine-triazine junction over carbon nitride frameworks for artificial photosynthesis of hydrogen peroxide. Adv. Mater. 2023, 35, e2306831.
26. Shin, J.; Eo, J. S.; Jeon, T.; Lee, T.; Wang, G. Advances of various heterogeneous structure types in molecular junction systems and their charge transport properties. Adv. Sci. 2022, 9, e2202399.
27. Deng, Q.; Li, H.; Ba, G.; Huo, T.; Hou, W. The pivotal role of defects in fabrication of polymeric carbon nitride homojunctions with enhanced photocatalytic hydrogen evolution. J. Colloid. Interface. Sci. 2021, 586, 748-57.
28. Yang, X.; Chen, Z.; Zhao, W.; et al. Recent advances in photodegradation of antibiotic residues in water. Chem. Eng. J. 2021, 405, 126806.
29. He, B.; Feng, M.; Chen, X.; Cui, Y.; Zhao, D.; Sun, J. Fabrication of potassium ion decorated 1D/2D g-C3N4/g-C3N4 homojunction enabled by dual-ions synergistic strategy for enhanced photocatalytic activity towards degradation of organic pollutants. Appl. Surf. Sci. 2022, 575, 151695.
30. Ma, P.; Zhang, X.; Wang, C.; et al. Band alignment of homojunction by anchoring CN quantum dots on g-C3N4 (0D/2D) enhance photocatalytic hydrogen peroxide evolution. Appl. Catal. B. Environ. 2022, 300, 120736.
31. Zheng, Y.; Liu, Y.; Guo, X.; et al. In-situ construction of morphology-controllable 0D/1D g-C3N4 homojunction with enhanced photocatalytic activity. Appl. Surf. Sci. 2021, 563, 150317.
32. Luo, M.; Jiang, G.; Yu, M.; et al. Constructing crystalline homophase carbon nitride S-scheme heterojunctions for efficient photocatalytic hydrogen evolution. J. Mater. Sci. Technol. 2023, 161, 220-32.
33. Das, S.; Ng, L. S.; Chong, C.; et al. Effective interfacing of surface homojunctions on chemically identical g-C3N4 for efficient visible-light photocatalysis without sacrificial agents. Small 2024, 20, e2400780.
34. Zeng, Z.; Yu, H.; Quan, X.; Chen, S.; Zhang, S. Structuring phase junction between tri-s-triazine and triazine crystalline C3N4 for efficient photocatalytic hydrogen evolution. Appl. Catal. B. Environ. 2018, 227, 153-60.
35. Zhao, G.; Liu, G.; Pang, H.; et al. Improved photocatalytic H2 evolution over G-carbon nitride with enhanced in-plane ordering. Small 2016, 12, 6160-6.
36. Lan, Y.; Li, Z.; Li, D.; Yan, G.; Yang, Z.; Guo, S. Graphitic carbon nitride synthesized at different temperatures for enhanced visible-light photodegradation of 2-naphthol. Appl. Surf. Sci. 2019, 467-8, 411-22.
37. Wang, X.; Blechert, S.; Antonietti, M. Polymeric graphitic carbon nitride for heterogeneous photocatalysis. ACS. Catal. 2012, 2, 1596-606.
38. Mo, Z.; She, X.; Li, Y.; et al. Synthesis of g-C3N4 at different temperatures for superior visible/UV photocatalytic performance and photoelectrochemical sensing of MB solution. RSC. Adv. 2015, 5, 101552-62.
39. Yu, H.; Shi, R.; Zhao, Y.; et al. Alkali-assisted synthesis of nitrogen deficient graphitic carbon nitride with tunable band structures for efficient visible-light-driven hydrogen evolution. Adv. Mater. 2017, 29, 1605148.
40. Lin, L.; Ou, H.; Zhang, Y.; Wang, X. Tri-s-triazine-based crystalline graphitic carbon nitrides for highly efficient hydrogen evolution photocatalysis. ACS. Catal. 2016, 6, 3921-31.
41. Hu, E.; Chen, Q.; Gao, Q.; et al. Cyano-functionalized graphitic carbon nitride with adsorption and photoreduction isosite achieving efficient uranium extraction from seawater. Adv. Funct. Mater. 2024, 34, 2312215.
42. Cao, Y.; Zhang, Z.; Long, J.; et al. Vacuum heat-treatment of carbon nitride for enhancing photocatalytic hydrogen evolution. J. Mater. Chem. A. 2014, 2, 17797-807.
43. Sun, H.; Shi, Y.; Shi, W.; Guo, F. High-crystalline/amorphous g-C3N4 S-scheme homojunction for boosted photocatalytic H2 production in water/simulated seawater: interfacial charge transfer and mechanism insight. Appl. Surf. Sci. 2022, 593, 153281.
44. Padhiari, S.; Tripathy, M.; Hota, G. Nitrogen-doped reduced graphene oxide covalently coupled with graphitic carbon nitride/sulfur-doped graphitic carbon nitride heterojunction nanocatalysts for photoreduction and degradation of 4-nitrophenol. ACS. Appl. Nano. Mater. 2021, 4, 7145-61.
45. Cheng, L.; Zhang, D.; Liao, Y.; Fan, J.; Xiang, Q. Structural engineering of 3D hierarchical Cd0.8Zn0.2S for selective photocatalytic CO2 reduction. Chin. J. Catal. 2021, 42, 131-40.
46. Yan, A.; Shi, X.; Huang, F.; Fujitsuka, M.; Majima, T. Efficient photocatalytic H2 evolution using NiS/ZnIn2S4 heterostructures with enhanced charge separation and interfacial charge transfer. Appl. Catal. B. Environ. 2019, 250, 163-70.
47. Nie, H.; Liu, Y.; Li, Y.; et al. In-situ transient photovoltage study on interface electron transfer regulation of carbon dots/NiCo2O4 photocatalyst for the enhanced overall water splitting activity. Nano. Res. 2022, 15, 1786-95.
48. Li, F.; Liu, Y.; Chen, Q.; et al. Transient photovoltage study of the kinetics and synergy of electron/hole co-extraction in MoS2/Ag-In-Zn-S/carbon dot photocatalysts for promoted hydrogen production. Chem. Eng. J. 2022, 439, 135759.
49. Chen, Q.; Liu, Y.; Mao, B.; et al. Carbon-dot-mediated highly efficient visible-driven photocatalytic hydrogen evolution coupled with organic oxidation. Adv. Funct. Mater. 2023, 33, 2305318.
50. Zhu, D.; Zhou, Q. Nitrogen doped g-C3N4 with the extremely narrow band gap for excellent photocatalytic activities under visible light. Appl. Catal. B. Environ. 2021, 281, 119474.
51. Zhang, Z.; Yang, X.; Hedhili, M. N.; Ahmed, E.; Shi, L.; Wang, P. Microwave-assisted self-doping of TiO2 photonic crystals for efficient photoelectrochemical water splitting. ACS. Appl. Mater. Interfaces. 2014, 6, 691-6.
52. Wu, J.; Gao, X.; Chen, Z. Elucidating the construction and modulation of built-in electric field in the oxygen evolution reaction. Chem. Eng. J. 2024, 492, 152241.
53. Ma, J.; Deng, H.; Zhang, Z.; et al. Facile synthesis of Ag3PO4/PPy/PANI ternary composites for efficient catalytic reduction of 4-nitrophenol and 2-nitroaniline. Colloid. Surf. A. 2022, 632, 127774.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].