fig8
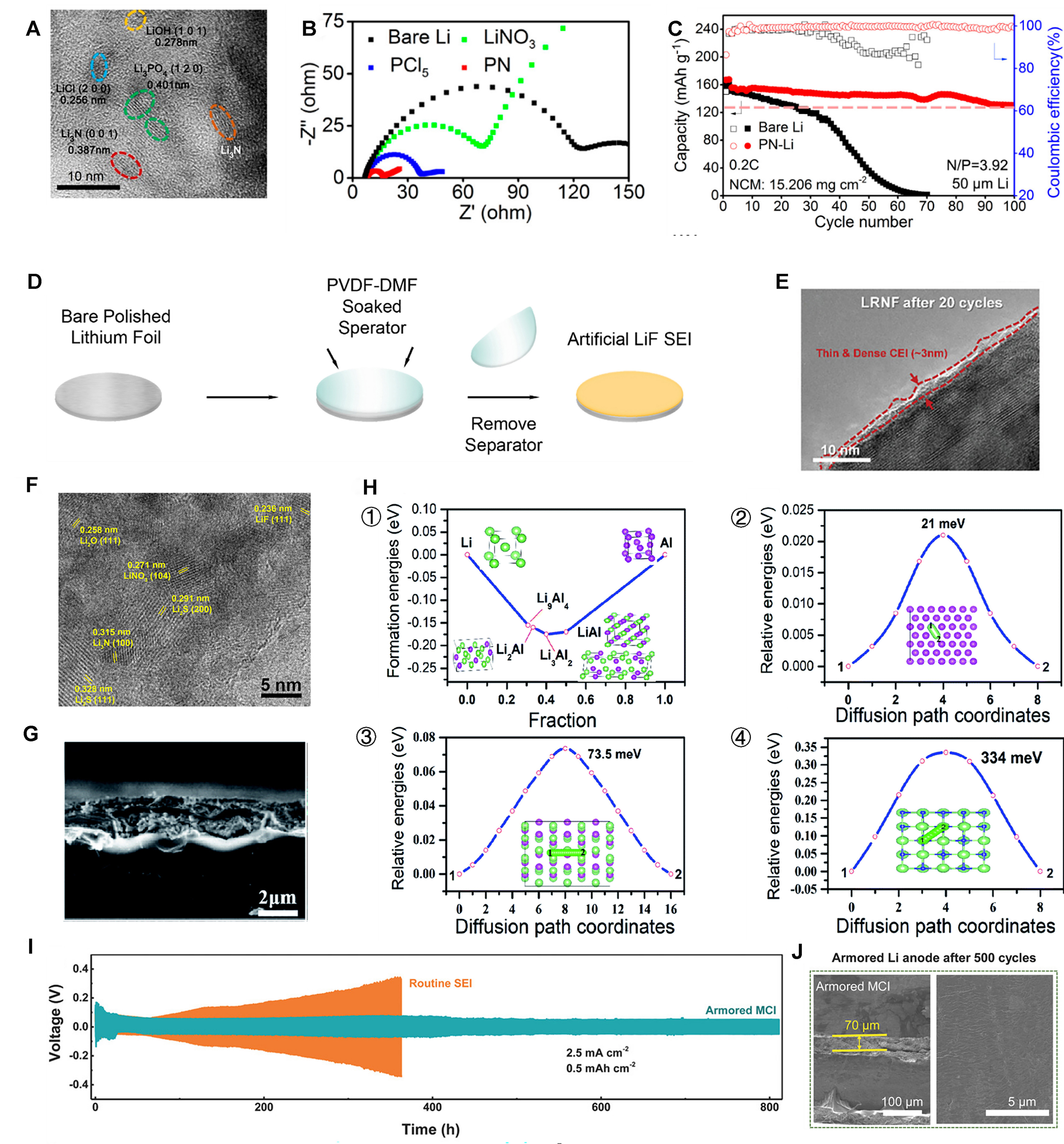
Figure 8. (A) HR-TEM image of PN interface; (B) Electrochemical impedance spectra of the Li anode treated by different materials; (C) Long-term cycle performance of Li/NCM523 full cells with mass loadings of 9.9 and 15.206 mg·cm-2[109]. Copyright 2020, ACS; (D) Illustration of the process for fabricating LiF-coated Li[113]. Copyright 2019, Elsevier; (E) HR-TEM images of LRNF after 20 cycles[114]. Copyright 2023, Wiley‐VCH; (F) A HR-TEM image of the sulfurized SEI[120]. Copyright 2017, Elsevier; (G) Cross-sectional view of SEM images of the lithium anode AlF3@Li; (H) (1) DFT calculated formation energies of the Li-Al binary alloys; energy variations of Li ion diffusion on (2) Al (111), (3) Li9Al4 (100) and (4) LiF (100) surfaces, respectively[121]. Copyright 2020, RSC; (I) Long-term stability of symmetric Li | Li cells at a large current density of 2.5 mA cm-2; (J) Cycled cross-section and top-view SEM images for armored MCI SEI-protected lithium-metal anodes[122]. Copyright 2018, Wiley-VCH. ACS: American Chemical Society; DFT: density functional theory; HR-TEM: high-resolution transmission electron microscopy; LiF: lithium fluoride; NCM: nickel cobalt manganese; LRNF: gradient fluorine-doped lithium-rich layered oxide cathode material; MCI: composite mixed ionic/electronic conductor interphase; SEI: solid electrolyte interphase; PN: phosphating-nitriding; RSC: Royal Society of Chemistry; SEI: solid electrolyte interphase; SEM: scanning electron microscopy.








