Recent advances in understanding the mechanisms of zeolite crystallization: a review
Abstract
Crystalline zeolites are highly ordered inorganic microporous materials widely used in catalysis, ion exchange, gas separation and biomass conversion. The investigation of crystal growth pathways and mechanisms is needed to enhance the performance of zeolites. However, understanding these mechanisms is highly challenging, as zeolites usually grow in a closed autoclave, which is known as a “black box”. This review provides a comprehensive overview of the progress made in understanding the crystallization mechanism of zeolites in recent years and summarizes the application of advanced characterization in studying these issues. Emphasis is placed on the host-guest interaction and crystallization kinetics during the zeolite crystallization process.
Keywords
INTRODUCTION
Zeolites, as crystalline inorganic framework materials with various applications (catalysis, ion exchange, gas separation, biomass conversion, etc.), exhibit excellent multifunctionality in chemical modification[1]. Typically, zeolites are inorganic frameworks composed of TO4 (T = Si, Al and other framework atoms)[2]. Their crystallization mechanism significantly affects their structures, which in turn affect their properties and performances[3].
In 1756, Swedish mineralogist Axel Crönstedt named zeolites based on the phenomenon observed during calcination. In 1932, McBain proposed the concept of molecular sieve by studying the sieving effect of different molecules[4]. In 1948, Barrer synthesized zeolite P and zeolite Q, which did not exist in nature under hydrothermal conditions, which was the earliest artificial zeolite[5]. Subsequently, a series of zeolites without natural counterparts were synthesized. By the 1960s, zeolite Y was first applied in the fluidized catalytic cracking (FCC) process, greatly improving the conversion rate of raw materials, gasoline yield, and reducing the generation of coking[6]. In 1961, organic templates were added to the zeolite synthesis systems. With the assistance of organic template agents, the framework Si/Al ratio of zeolite A was significantly improved[7]. Afterward, a series of zeolites with high Si/Al ratios, such as ZSM-5 (MFI type) and Beta (BEA type), was successfully synthesized and widely used in industrial catalysis[8]. With the increase of the framework Si/Al ratio of ZSM-5 zeolite, pure silicon framework MFI zeolite was finally synthesized, named silicalite-1. In 1983, titanium was introduced into the framework of silicalite-1, forming titanium silicate zeolite (TS-1) with an MFI framework topological structure. TS-1 is an important catalyst for selective epoxidation of olefins and oxidative desulfurization (ODS) reactions[9]. In 1982, another novel family of zeolites, aluminum phosphate zeolite AlPO-n, was successfully synthesized (n is the code)[10]. The framework of aluminum phosphate zeolites consists of alternating AlO4 and PO4 tetrahedra, with no P–O–P bonds, and is electrically neutral[11]. After long-term development, three main improvements have been made. First, numerous aluminum phosphate microporous compounds with anionic frameworks have been developed[12]. Second, many other phosphate framework materials have been introduced, such as zinc phosphate, gallium phosphate, titanium phosphate, etc.[13]. Thirdly, various heteroatom-substituted aluminum phosphates, such as SAPO-n and Co-AlPO-n series zeolites, have been successfully synthesized. Since the synthesis of the first extra-large micropore zeolite in 1988 (VPI-5, VFI topology), a total of 27 structures of extra-large micropore zeolites have been included in the International Zeolite Association (IZA) database. However, their stability limits their industrial applications. Recently, progress has been made in the synthesis of extra-large pore zeolites, with a series of extra-large pore zeolites exhibiting high stability and low framework density synthesized through different strategies. These include aluminum silicate ZEO-1 (JZO topology)[14], ZEO-3 (JZT topology)[15] synthesized by condensation of chain zeolite precursors, and ZEO-5 (HZF topology)[16] synthesized by layer connection. Due to its high stability and extra-large pore structure, ZEO-1 can be used for adsorption separation, ZEO-3 for volatile organic compound emission reduction and recovery, and ZEO-5 for olefin epoxidation reaction after Ti substitution.
Zeolites are usually synthesized under hydrothermal conditions. Researchers have pioneered the artificial hydrothermal synthesis of zeolites by simulating their natural formation environment. Hydrothermal synthesis is the classic and most widely used method for synthesizing zeolite crystals. Generally, raw materials such as aluminum, silicon, phosphorus, or other heteroatom-containing materials, along with inorganic/organic templates, are mixed with water, placed in a high-pressure vessel, and crystallized at 100-230 °C[17]. This method is easy to operate and suitable for large-scale synthesis. In addition to hydrothermal synthesis, zeolite synthesis methods include solvothermal synthesis[18,19], dry gel synthesis[20], ultrasonic-assisted synthesis[21,22], microwave-assisted synthesis[23,24], solvent-free grinding[25], fluoride ion-assisted synthesis[26], ionic liquid thermal synthesis[27,28], oil bath method[29], dry rolling method[30] and phase transfer synthesis[31]. Although each of these methods has its advantages, they require special equipment, which limits their application. This also makes hydrothermal synthesis still the most classic and commonly used synthesis method.
As of now, a total of 256 zeolite topological structures have been certified by the IZA and assigned a framework type code (FTC) consisting of three letters. The fundamental structural unit of the zeolite framework is the TO4 tetrahedron. These tetrahedra form a three-dimensional zeolite framework by sharing O atomic vertices, where the TO4 tetrahedron is referred to as the primary building unit[32].
To provide a more intuitive understanding of the framework structure of zeolites, Smith[33] proposed the concept of secondary building units (SBUs) in 1988. SBUs are multivariate cyclic structures constructed by primary building units through shared vertex O atoms and different bridging methods. These SBUs can include 3-membered rings, 4-membered rings (4Rs), 5-membered rings (5Rs), 6-membered rings (6Rs), double-6-membered rings (D6R), and more. Currently, 23 types of SBUs are included in the IZA database[34]. It is important for researchers to note that the SBU is only a theoretical structural building unit used to describe the framework structure of zeolites more intuitively, rather than representing actual species in the crystallization process of zeolites.
For zeolites, the crystallization mechanism dictates their structure, which in turn determines their application properties. Therefore, in-depth research on the crystallization mechanism of zeolites and subsequent design before synthesis is of great significance for the development and application of high-performance zeolites in catalysts, adsorbents, and exchange media. However, the crystallization process of zeolites is a very complex process with multiple equilibrium reactions, especially the complex and harsh conditions of hydrothermal reactions. In the high-temperature and high-pressure synthesis system, there may be solutions, gel, amorphous solids, metal and non-metallic ions, crystals, twins and composite crystals, or organic compounds (usually templates or solvents). The crystallization of zeolites occurs in a “black box”. So far, the crystallization mechanism of zeolites has not been well understood at the molecular level.
Here, we have summarized recent work on the zeolite crystallization mechanism, including advanced characterization, host-guest interaction in the zeolite synthesis system and the crystallization kinetics of zeolite. At present, research on crystallization mechanisms mainly focuses on the interaction between the host and guest. Researchers have studied the role of template agents and other additives in zeolite synthesis systems through various characterization methods, and attempted to propose the zeolite crystallization process based on experimental phenomena, pointing out the driving force of zeolite crystallization. This provides not only a summary for relevant researchers, but also some references for researchers engaged in zeolite synthesis and modification.
ZEOLITE CRYSTALLIZATION AND CHARACTERIZATION
The study of the crystallization mechanism and crystal growth pathways of zeolites can provide a deeper understanding of the formation process of zeolites and provide a theoretical basis for regulating the specific performance of zeolites and synthesizing new zeolites.
Crystallization mechanism and crystal growth pathways of zeolites
By studying the crystallization mechanism of zeolites, the essence of the crystallization process of zeolites can be understood, which provides theoretical guidance for controlling their physical properties and synthesizing new zeolite materials. Since Barrer et al. first proposed the hydrothermal synthesis mechanism of zeolites in 1959[35], researchers have never stopped exploring this aspect[36]. It should be noted that due to the diversity of zeolite materials and synthesis conditions, their crystallization mechanisms also vary. Even for the same zeolite, its crystallization mechanism may vary depending on changes in the synthesis system and conditions. The crystallization mechanism can be divided into solution-mediated transport mechanism and solid hydrogel transformation mechanism based on whether the liquid phase components directly participate in crystallization.
Solution-mediated transport mechanism
In 1966, Kerr first proposed a solution-mediated transport mechanism[37]. They believed that zeolite crystals nucleated and grew from the gel-liquid phase. The gel was generated under crystallization conditions and gradually dissolved into the liquid phase, and the dissolved silicate ions and aluminate ions further polymerized to gradually generate zeolite crystals. Later, Zhdanov further elaborated on the solution-mediated transport mechanism and proved it through experiments[38]. He proposed that the initial aluminosilicate gel containing simple SBUs was generated first after the raw materials were mixed. In 1977, Angell and Flank conducted a detailed study on the crystallization process of zeolite A and concluded that the crystallization process of zeolite A conformed to the solution-mediated transport mechanism[39]. The author believed that liquid chromatography could be divided into the following three steps. First: the generation of initial aluminosilicate gel. After the raw materials were mixed, the silicate ion and aluminate ion polymerized to generate the primary gel. The system composition was basically stable within a certain time. Second: the generation of secondary aluminosilicate gel. After heating the primary gel, the secondary gel with a compact structure was obtained. At this point, the increase in solid phase content and the change in liquid phase composition indicated that this transformation was achieved through a local redissolution precipitation process. Third: crystallization of secondary gel. As crystallization progresses, the crystallinity of the solid phase increased, and compositional changes occurred in both solid and liquid phases. Experiments showed that crystallization was achieved through dissolution and repolymerization. Xu and Liu verified the solution-mediated transport mechanism by exchanging the liquid phases of two different L zeolite synthesis systems during the crystallization induction period[40]. In the early 1980s, Ueda et al. directly synthesized zeolites such as analcime, mordenite (MOR), faujasite, erionite, and offretite from a homogeneous liquid-phase crystallization system[41]. Tian et al. found that the elemental composition in both solid and liquid phases gradually changed during the SAPO-34 synthesis through SAPO-5 transformation, revealing that the above zeolite transformation process follows the mechanism of liquid phase transformation[42]. These experiments all support the solution-mediated transport mechanism.
Solid hydrogel transformation mechanism
In 1968, Flanigen and Breck put forward the solid hydrogel transformation mechanism[43]: the initial silicon aluminum gel is generated after the mixing process of raw materials, and the zeolite crystal is gradually formed by depolymerization and rearrangement under the condition of hydrothermal crystallization. In the crystallization process, the solid phase of gel does not dissolve, and the species in the liquid phase do not directly participate in the nucleation and growth of zeolites. In 1989, Xu et al. successfully synthesized ZSM-5 and ZSM-35 zeolites in non-aqueous solutions[44]. The results showed that the Si/Al ratio remained unchanged in the solid phase after crystallization, and silicate or aluminate ions did not exist in the liquid phase, strongly supporting the solid hydrogel transformation mechanism. Furthermore, in the process of synthesizing zeolites using the solvent-free method, no additional liquid water was added, except for a small amount introduced with the raw materials and reagents. This was also a typical example of the solid hydrogel transformation mechanism[45].
Shao et al. pretreated the synthesis system of TS-1 and prepared hierarchical porous TS-1 using an extremely viscous solid gel. The authors studied changes in the synthesis system and the process of zeolite nucleation and growth. They concluded that the synthesis of hierarchical pore TS-1 follows the solid hydrogel transformation mechanism [Figure 1][46]. Similarly, Li et al. synthesized hierarchical porous ZSM-5 using silica spheres as raw material. Research has shown that silica spheres are not only nutrients, but also guide the formation of macroscopic/microscopic pore structures, consistent with solid hydrogel transformation mechanism[47].
Figure 1. Illustration of the proposed formation mechanism of the single-crystalline hierarchical zeolites[46]. Copyright © 2021 Elsevier Inc. All rights reserved.
Researchers have found that, during the crystallization process of zeolites, some phenomena cannot be well explained by a single solid hydrogel transformation or solution-mediated transport mechanism.
Equilibration mechanism
In 1981, Derouane et al. proposed the biphasic transformation mechanism[48]. This viewpoint suggested that both solid-phase and solution-mediated transformation coexisted during the crystallization process of zeolites, and they could occur separately in two crystallization reaction systems or simultaneously in one system. Iton et al. discovered through small-angle neutron scattering that different silicon sources led to various conversion mechanisms during the synthesis of ZSM-5[49] and proposed that the crystallization process was a combination of multiple reaction mechanisms. Under varying crystallization conditions, even the same zeolite would follow different mechanisms.
Besides the issue of which phase crystallization occurs, the growth of crystals has become the next concern for researchers. There are two theories describing the mechanism of zeolite growth: (1) the classical theory of crystallization based on spontaneous nucleation and growth from the addition of silicate molecules; and (2) the non-classical theory of crystal nucleation and growth involving the direct aggregation and attachment of nanoparticles. The schematic diagram of two crystallization pathways is shown in Figure 2[50].
Figure 2. Schematic representation of classical and non-classical crystallization. (A) Classical crystallization pathway; (B) oriented attachment of primary nanoparticles forming an iso-oriented crystal upon fusing; (C) mesocrystal formation via self-assembly of primary nanoparticles covered with organics[50]. Copyright © 2005 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Classical routes usually involve spontaneous nucleation and are derived by the addition of monomers (atoms or molecules) from the growth solution to kink, step, and terrace sites on crystal surfaces, resulting in crystals with smooth surfaces[51]. Non-classical processes encompass the addition and attachment of precursors, which range from oligomers to primary particles or even fully developed nanoparticles, comprising disorder-to-order transformation processes, leading to the formation of crystals with an irregular morphology, rough crystal surfaces, and mesoscopic structures[52,53]. To determine which mechanism the zeolite crystallization process follows or to enable theoretical innovation, a detailed characterization of the synthesis system and its products is required. With the progress of analytical science, multiple advanced characterization methods are now available.
Characterization of zeolite crystallization process
Ex-situ characterization
In most cases, the ex-situ method is used to characterize the crystallization mechanism, which has the characteristics of convenience and low instrument requirements, bringing good operability. The combination of multiple methods can comprehensively characterize the products or intermediate at each stage of crystallization and thus infer the complete crystallization process.
X-ray diffraction (XRD) is the most important tool for studying zeolites. The crystal planes of zeolites act as gratings to diffract X-rays, allowing the distinction of the crystal planes through diffraction peaks. These characteristic peaks are then used to determine the topological structure of the zeolite. When characterization conditions are consistent, the XRD spectrum can be used to calculate the relative crystallinity of zeolites, providing a guarantee for studying crystallization kinetics. From the relative crystallinity obtained via XRD, it is found that almost all hydrothermal synthesized zeolites follow the Avrami equation during their crystallization process. This process consists of induction periods, crystal growth periods, and completion periods[54]. Based on this, the zeolite crystallization process can be understood as an auto-catalytic reaction, where, upon heating, the raw material generates an active center for crystallization, and subsequent crystallization occurs around this active center.
Apart from XRD, scanning electron microscopy (SEM) and transmission electron microscopy (TEM) are important fundamental characterization methods for materials. The material states of various crystallization stages can be visually characterized with SEM, including raw materials, amorphous precursors, and crystallized zeolites. TEM provides local structural information of small species. High-resolution TEM (HRTEM) typically offers a resolution below 1 Å, enabling the direct observation of material defects, structures, and other local structural information at the atomic scale, as well as simultaneous electron diffraction analysis. TEM can also be used as evidence to distinguish between classical and non-classical pathways. Meanwhile, field emission SEM with energy dispersive X-ray (FE-SEM-EDX) spectroscopy is an effective method for visually characterizing the surface element distribution (content and uniformity) of zeolites through electron microscopy[55]. In summary, electron microscopy can track the entire process from the initial raw material to the complete zeolite, intuitively describe the nucleation and growth of zeolite, and provide element distribution information for relevant stages. It plays an important role in characterizing the crystallization mechanism of zeolites.
Fourier transform infrared spectroscopy (FT-IR) and ultraviolet (UV)-Raman techniques can be applied to the study of zeolite synthesis and provide structural information of intermediate species during the crystallization process. In particular, UV-Raman can effectively identify the SBU structures in zeolite and zeolite synthesis systems. It is worth noting that UV Raman spectroscopy is also frequently used in the study of zeolite transformation to elucidate the decomposition or retention of certain structures[56].
Solid-state nuclear magnetic resonance (SS NMR) is an effective technique for studying local structural changes and the chemical environments of atoms. As an analysis method that does not destroy samples, nuclear magnetic resonance (NMR) is an indispensable means to analyze the structure of zeolites. Ex-situ NMR can maximize the use of commercial instruments, thus having the characteristics of convenient testing and reproducible results. Meanwhile, multiple pulse sequences [e.g., one-pulse, cross-polarization (cp), rotational-echo double-resonance (REDOR)] and 2D spectra [heteronuclear correlation (HETCOR), multiple-quantum (MQ) magic angle spinning (MAS), double quantum-single quantum (DQ-SQ), heteronuclear multiple-quantum correlation (HMQC)] can reveal the chemical environment of various atoms in zeolite and reveal their interactions[57].
Dynamic nuclear polarization (DNP) is a branch of NMR spectroscopy that utilizes the electron nucleus double resonance principle to excite free electron transitions in microwaves, polarizing the spin energy level distribution of related nuclei. This can significantly enhance the sensitivity of NMR methods and provide information on microscopic electronic structures. Berkson et al. investigated the crystallization process of siliceous MFI zeolite and investigated the meta-structured MFI zeolite nanosheets using DNP-enhanced 2D J mediated 29Si{29Si} NMR experimental [Figure 3] proving that the mesostructured MFI zeolite nanoplates formed from non-topological layered sites, which clearly established the coefficient con activities of transforming intermediate nanolayered site motifs and crystallizing mesostructured MFI zeolite nanosheets[58]. Although the experiments have shown that DNP technology significantly improves detection sensitivity and obtains structural information, the limited number of instruments and high experimental difficulty currently make the research in this area insufficient.
Figure 3. Solid-state 2D DNP-enhanced J-mediated (through-bond) 29Si{29Si} correlation spectrum of the intermediate product of crystallizing MFI zeolite[58]. Copyright © 2017 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim. DNP: Dynamic nuclear polarization.
Although the exome representation methods have the aforementioned advantages, they cannot avoid the possible changes that may occur during the material annealing or quenching stage, which may lead to the obtained results not reflecting the true state of reaction crystallization. Meanwhile, certain characterization and sample preparation processes inevitably influence the physicochemical properties of intermediates in the crystallization, even after the completion of zeolite crystallization. As analytical science advances, various in-situ characterizations have been applied to the study of zeolite crystallization processes.
In-situ characterization
To address the above-mentioned challenges and make the relevant characterization more accurate in reflecting the system and intermediate products during the crystallization process, various in-situ methods have been used. This includes in-situ XRD, in-situ NMR, in-situ infrared (IR) spectroscopy, and in-situ Raman spectroscopy, among others. Among these, in-situ NMR is considered the most important and promising tool for researching the crystallization mechanism of zeolites.
Due to the particularity of NMR instruments and the effectiveness of NMR characterization, some researchers were committed to in-situ NMR characterization of reaction systems. Due to the unique nature of hydrothermal systems, instruments were required to meet the requirements of the “three resistances” - high-temperature resistance, high-pressure resistance, and alkali corrosion resistance. For this purpose, they designed and manufactured some instruments or equipment accessories, and successfully characterized the synthesis system by in-situ NMR. Since the first in-situ characterization of zeolite A in 1996[59], relevant work has been mainly on materials with low silicon-aluminum ratios and low-temperature crystallization. In addition, some atomic nuclei have lower natural abundance and weaker signals, requiring longer sampling times, which contradicts the original intention of in-situ characterization - time resolution. Balancing time resolution and spectral resolution is also a major challenge in in-situ characterization. Characterizing high-temperature, high-pressure, and highly corrosive systems remains a challenge in relevant research.
With the advancement of materials science and the development of nuclear magnetic instruments, in-situ instruments used in zeolite synthesis processes are gradually developing; some special devices for in-situ NMR have been manufactured and successfully characterized for the zeolite crystallization process[59-63] [Figure 4]. The basic trends of the development are as follows: (1) From specialized probes to commercial probes, the requirements for instruments are reduced; (2) From liquid probes to solid probes, the magic angle rotation improves the spectral resolution; (3) Increasing the synthesis temperature from low to high expands the range of materials that can be characterized; (4) Just as an increase in temperature leads to a rise in pressure, transitioning from low to high pressure also expands the range of materials that can be characterized; (5) From natural to enriched isotopes, some nuclei, such as 29Si and 13C, have lower natural abundance and can only provide extremely low signals within a limited sampling time. Isotope labeling can greatly improve their signal-to-noise ratio and save sampling time[64]. Although in-situ characterization of hydrothermal synthesis processes has been achieved, more scenes of in-situ NMR application are dry gel conversion (DGC). This is because the pressure of DGC is lower, reducing the risk of instrument damage. The application of in-situ SS NMR technology in the study of zeolite crystallization mechanism still requires significant effort.
Figure 4. Sample tube for in-situ NMR characterization of crystallization process. (A) The first MAS NMR cell sused for in situ studies of hydrothermal synthesis of zeolite sreported by Shi et al. Copyright © 1996, American Chemical Society[59]; (B) Quartz glass ampoules by Liu et al. Copyright © 2009 Elsevier Inc. All rights reserved[60]; (C) HPHT cell for liquid state NMR. HPHT MAS NMR cells suitable for in situ NMR studies of zeolite synthesis under hydrothermal conditions. Copyright © 2000 Wiley[61]; (D) PEEK insert for Bruker 7 mm MAS rotor. Copyright © 2006 Elsevier Inc[62]; and (E) 7.5 mm ZrO2 rotor for Chemagnetic/Varian MAS probe. Copyright © 2016, American Chemical Society[63]. NMR: Nuclear magnetic resonance; MAS: magic angle spinning; HPHT: high pressure high temperature; PEEK: polyether ether ketone.
In addition to in-situ NMR, in-situ IR and in-situ Raman spectroscopy have also been used to characterize the crystallization of zeolites. In situ IR can observe the changes in functional groups in synthetic systems and is used to describe the process of changes in the bonding relationships of atoms. In situ Raman can observe the Raman vibration in the system, which is mainly used to describe the formation of SBUs and frameworks. Due to the equipment limitations, synthetic systems with a crystallization temperature around 100 °C are often chosen for in-situ IR and in-situ Raman characterization. Xue et al. monitored the crystallization of LTA zeolite from an organic silicon source using in-situ IR spectroscopy. The in-situ IR analysis revealed the appearance of Si-OH, and based on this, the authors proposed a crystallization mechanism[65]. Fan et al. monitored the crystallization of Zeolite X[66] and AlPO-5[67] using in-situ Raman, described the changes in Al-P bonding relationships and the structure of organic templates, and proposed the crystallization mechanism of AlPO-5. In addition to hydrothermal systems, this method is also applicable to the dry gel method[68] and the synthesis of metal-organic frameworks (MOFs) in solvothermal systems[69].
As technology advances, some other characterization methods have been extended into the field of zeolite synthesis. For example, conductivity testing [alternating current (AC) impedance spectroscopy] is commonly used for characterizing battery electrolytes[70] and photo density waves are used for studying material particle size[71]. These methods provide another perspective for studying the zeolite crystallization process. All the above experiments demonstrate that applying some other characterization methods to zeolite synthesis systems can reflect certain laws in zeolite synthesis and can serve as a supplement to XRS, SEM, TEM, and NMR.
Combination of in-situ and ex-situ
Ex-situ and in-situ characterizations each offer distinct advantages. By preparing samples according to experimental requirements and combining both methods, researchers can leverage the convenience and excellent signal characteristics of ex-situ experiments, along with the time resolution and realistic environmental conditions provided by in-situ experiments. This integrated approach is a key aspect of future research on zeolite crystallization mechanisms.
Paula et al. combined in-situ XRD and in-situ NMR to investigate the interzeolite conversion (IZC) process of VPI-5. Under humid conditions, water promoted the breaking of old Al–O–P bonds and the formation of new Al–O–P bonds, leading to the conversion of VPI-5 zeolite into AlPO-8. This process was difficult to achieve in dry or high temperatures (> 550 °C). Experiments showed that at 100 °C, for the AlPO-8 signal, the time evolution was interpreted using the Avrami equation. Similarly, the authors investigated the water adsorption behavior of MOF materials MIL-53 and MIL-53-ADP. The process of water adsorption and action on the framework is different in MIL-53 and MIL-53-ADP. The authors believed that the complex mechanism would not be accessible without the real time insights into the structure transition obtained by in-situ analytical techniques[72]. The above experiments have demonstrated that in-situ characterization is suitable not only for the study of zeolite synthesis/crystallization systems, but also for the study of MOF material properties.
It should be noted that the characterizations listed in this article are commonly used. Additionally, there are other characterization methods that have not been mentioned in this communication. However, it should be noted that the sample may undergo changes during the characterization process, resulting in signals that cannot truly reflect the situation of the synthetic system. For example, some characterizations involving energy input (such as in-situ UV Raman spectroscopy), change in gravity conditions (such as in-situ NMR), and so on. These factors should be considered when analyzing results obtained through these methods. In summary, combining these characterization methods can comprehensively characterize the zeolite crystallization and provide a comprehensive understanding of the substances in the system, especially the study of the host-guest interactions in the system, which can effectively promote the research process of crystallization mechanism.
HOST-GUEST INTERACTIONS AND TEMPLATE ACTIONS IN ZEOLITE CRYSTALLIZATION
As mentioned above, in the process of artificial zeolite synthesis, some substances have been found to change the synthesis gel, especially providing a certain template for the formation of the zeolite framework. Using these substances, many new structures have been successfully synthesized. Therefore, these substances are referred to as template or structural directing agents. Beyond their role in structural orientation, further research has revealed their complex roles in the synthesis process, such as affecting the liquid phase structure and promoting crystallization[73]. However, due to convention, they are still commonly referred to as template or structure-directing agents. Common structural directing agents include metal cations, quaternary ammoniums, organic amines, organic hydroxyl compounds, inorganic cations, anions represented by fluoride ions, and some metal complexes. In recent years, some surfactants and amino acids have also been used to regulate the crystallization process of zeolites, but their main role is to change the morphology of zeolites or generate hierarchical pore structures. It is evident that they interact with the precursors of the zeolite framework (including silicon, aluminum, phosphorus species, SBUs formed by these elements, and even amorphous structures generated during the crystallization) during the synthesis process[74]. Similar to the host-guest interaction in fully crystallized zeolites, we refer to the zeolite framework and its precursors as the host, and template agents and other substances that affect crystallization as the guest, to elucidate the host-guest interaction during the crystallization process. The following summarizes the relevant work in recent years.
Metal cations
Silcoaluminate zeolites are generally synthesized in alkaline systems, where high alkalinity is beneficial for dissolving silicon species in raw materials or accelerating the decomposition of organic materials. In this way, when adding inorganic bases, it is inevitable to introduce metal cations. Those commonly used include alkali metal ions Li+, K+, Na+, and alkaline earth metal ions Ca2+ and Ba2+[75]. Overall, metal cations play a role in regulating the pH of the synthesis system, guiding the structure, and balancing the charge of the framework.
As is well known, the difference between zeolites and amorphous aluminosilicates lies in the fact that zeolites have a regular framework and pore structure, and metal ions can balance the charge of framework acid sites. Wang et al. investigated the crystallization of ZSM-57. It was observed with TEM that the use of different inorganic cations could lead to the formation of different twins. By regulating the growth route, the size of twin single-crystal chips was adjusted, revealing the reasons for the formation of morphological differences. Experiments have shown that the use of different inorganic cations leads to different crystallization mechanisms. In the K+ system, the classical solution-mediated pathway dominates the whole crystallization process while the classical solution-mediated path in the initial stage and the subsequent non-classical solid-mediated path are identified in the Na+ system[76].
Metal cations commonly used as inorganic template agents, such as Na+, have the characteristics of high natural abundance and strong nuclear magnetic response. Researchers can easily track changes in the chemical environment during the crystallization process, providing a powerful tool for studying the crystallization mechanism of zeolites. Yamada et al. studied the differences between the precursors of FAU zeolite and glass with a similar composition. Regarding the crystallization behavior of FAU, NMR analyses revealed that the 23Na environment changed slightly due to a gradient in order during the crystallization and an increased Na–O bond distance [Figure 5]. Furthermore, based on pair distribution function (PDF) patterns, the fraction of small-membered rings (e.g., 4Rs) was found to be higher in the zeolite precursor. The authors believed that Löwenstein’s rule, which prohibited the formation of Al–O–Al bonding, was also substantially applicable to the structure of the zeolite precursor[77].
Figure 5. 23Na MQ-MAS-NMR of (A) glass and zeolites with different crystallization times, (B) 3 h, (C) 10 h and (D) 24 h[77]. Copyright © 2018 Elsevier Inc. All rights reserved. MQ: Multiple-quantum; MAS: magic angle spinning; NMR: nuclear magnetic resonance.
In addition to the type of cation, the amount of cation also influences the zeolite crystallization process. Chawla et al. used the FE-SEM-EDX method to investigate MER, MFI, MOR, TON, and LTL zeolites. The intermediate states during the crystallization process were characterized. The authors monitored the initiation of nucleation, completion of crystallization, and presence of impurities using FE-SEM-EDX. Ex-situ experiments have demonstrated that sufficient differences in chemical composition (such as alkali metal content) between amorphous precursors and crystals can be used to (i) track the early stages of zeolite crystallization, (ii) locate its occurrence, and (iii) distinguish residual amorphous materials in the crystallization domain[78].
The above experiments indicate that different metal ions play different roles in complex zeolite synthesis systems and interlayer conversion processes. Their chemical environment becomes irregular as synthesis progresses, and during the zeolite conversion process, some framework aluminum atoms are considered important active centers and driving forces.
Inorganic anions
In hydrothermal synthesis systems, inorganic cations serve multiple roles. However, previous research on the role of anions is not sufficient and mainly focuses on hydroxide and fluoride ions. In recent years, research on anions has begun to expand to more inorganic anions.
Sun et al. studied the effect of inorganic anions in the S-1 zeolite synthesis system. The sodium salts of the Hofmeister series promoted the crystallization of silicalite-1, in the order of Na2SO4 > NaF > NaCl > NaBr > NaI > NaSCN. The research proved with liquid 1H-NMR that the ions were located around the template with a distance to the N atom of TPA+. The authors believed that the anions affected the release of water molecules by activating the hydration spheres around the TPA+ cation to a different degree, thus regulating zeolitic crystallization[79].
Moreover, they introduced halogen anions into the synthesis system of MOR and studied their influence on the crystallization process. Theoretical calculations [Figure 6] showed that iodine ions could accelerate the formation of Si–O–Si bonds and hinder the formation of Si–O–Al. Experimental results showed that this strategy indeed improved the silicon-aluminum ratio of MOR, verifying the theoretical calculation results. The authors conducted other experiments to prove that this rule was also applicable to the crystallization of NaY and ZSM-5, and the obtained zeolite had an improved silicon-aluminum ratio[80]. This is an application of the mechanism described above. In parallel, Wang et al. conducted a similar work in the synthesis system of ZSM-12 zeolite and obtained identical conclusions. The obtained zeolite, as a precious metal carrier, exhibited excellent performance in the hydrogenation isomerization reaction of n-dodecane[81].
Figure 6. Gibbs free energy surfaces of (1)-(4) at 453 K and 1 atm obtained by DFT theoretical calculation[80]. Copyright © The Royal Society of Chemistry 2022. DFT: Density functional theory.
The above experiment proves that inorganic anions influence the zeolite synthesis system through their interaction with organic cation templates. This interaction increases the rate of Si–O–Si bond formation while reducing the rate of Si–O–Al bond formation, thereby enhancing the Si/Al ratio of the obtained zeolite. This effect is primarily due to the charge density and degree of charge non-uniformity in the zeolite synthesis system.
Organic amines and quaternary ammoniums
Starting from the early 1960s, organic compounds mainly composed of amines have been used as templates for zeolite synthesis. Organic molecules or quaternary ammonium and quaternary phosphine cations have more possibilities in terms of size, shape, and functional groups compared with spherical inorganic cations[82]. Organic chains have complexity, flexibility, and selectivity, making the synthesis system of zeolites complex. So far, except for some zeolites synthesized with metal cations as structural directing agents, most zeolites have been synthesized with organic compounds as template agents.
The use of organic templates can adjust the pH of the synthesis system and synthesize the zeolites that cannot be synthesized with metal ions as templates. When the topological structure is the same, the use of organic template agents can improve the silicon-aluminum ratio of the synthesized zeolite. Because of the charge of amines and ammonium, they interact with the framework and can balance the framework charge and affect the product framework charge density. In addition, to obtain a single phase, some other molecules can act as growth inhibitors to inhibit the formation process of certain phases in mixed-phase synthesis systems. However, the use of organic compounds can cause wastewater and exhaust gas problems, making template-free synthesis an important research direction.
The general understanding of the role of organic templates in structure orientation during the hydrothermal process is that, under the heating of gel/dry gel, the raw material species and/or their solvates surround the template[83], followed by the nucleation and growth of the zeolites. Due to differences in the structure and polarity of organic matter, gel concentration, the initial silicon, aluminum and/or phosphorus species, the hydrolysis rate, and the crystallization temperature, the interaction between organic matter and raw materials varies. This variation leads to differences in the structure and properties of the synthesized zeolites, providing a theoretical basis for regulating zeolite properties by controlling the synthesis process.
Nishitoba et al. investigated the process of synthesizing CHA-type silicoaluminate zeolites using FAU and LTL zeolites as raw materials and trimethyladamantyl ammonium hydroxide and tetraethyl ammonium hydroxide as organic templates. The authors conducted detailed monitoring of crystallization behavior, and the experiment proved that organic structure-directing agents (OSDA) greatly affected the composition of the final products, and that the raw materials strongly influenced the Al distribution in the final products. The results obtained from this study clearly showed that the raw materials for the CHA-type zeolite would be optimized depending on its application. The methanol to olefins (MTO) performance of the obtained zeolite improved, compared to traditional CHA zeolite[84].
In-situ SS NMR is an effective characterization method for the zeolite crystallization process. Ivanova et al. monitored the crystallization of Beta under in-situ conditions using NMR and enhanced the signal of silicon species and template agents using 29Si and 13C isotopes. The experiment proved that the solution-mediated transport and solid hydrogel transformation mechanisms were followed in the two synthetic systems, respectively. Similarly, NMR also demonstrated the different structural orientation behaviors of the template in the two reaction pathways. As shown in Figure 7A, using in-situ13C MAS NMR, the authors proved that there were methyl TEA+ cations in the pores of BEA zeolite, TEA+ interacting with silicon species gel and TEA+ clusters in the reaction gel system. This indicated that in the crystallization process, the template performed multiple functions simultaneously, such as combining with the raw materials, balancing the zeolite framework charge and stabilizing the gel. The synergy of these effects demonstrated the complexity of the synthesis system and crystallization process. The results indicate that MAS NMR can serve as a superior tool for monitoring the hydrothermal synthesis of various solids (including zeolites, zeolite types, mesoporous materials, MOFs, etc.), and can also be used to design new excellent materials for different applications[85]. Similarly, Zhao et al. used in-situ NMR technology to characterize the changes in
Figure 7. (A) Representation of the mechanism of zeolite BEA crystallization[85]. Copyright © 2017 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim; (B) Schematic representation of the proposed mechanism for formation of the AlPO-11 framework[86]. Copyright © 2023 The Authors. Published by American Chemical Society.
In-situ NMR can characterize the gel system and conduct in-situ hydrothermal synthesis; however, due to the above reasons, especially the limitations of high-temperature resistance, high-pressure resistance and alkali corrosion resistance of the instrument, the current in-situ NMR characterization still focuses on the crystallization of DGC. Alahakoon et al. studied the crystallization mechanism of the DGC AlPO-11 using a commercially available 5 mm high pressure high temperature (HPHT) rotor. NMR tracked the chemical environment changes of Al and P atoms, and characterized the evolution of the framework structure. 1H → 13C cross-polarization (CP) tracked the role of organic structure directing agents, and 1H was used to reveal the effect of water content on crystallization kinetics [Figure 7B]. Experiments showed that the interaction between water and structure-directing agents (SDA) played an important role in the zeolite crystallization process, which was difficult to achieve through ex-situ characterization. The authors believed that combining the ex-situ and in-situ results would provide a clearer, more comprehensive, and less ambiguous picture of the formation of molecular sites[86].
In addition to in-situ NMR, Raman spectroscopy and simulated annealing can also be used to study the host-guest interactions during zeolite synthesis. Yuan et al. studied the crystallization mechanism of SAPO-34 synthesized with the hydrothermal method using TEA as a template. They first detected 4R and 8-membered rings (8R, 8MR) through Raman spectroscopy, while 6R appeared slightly later, indicating that the formation of cha cages occurred first. d6r units developed during the cage combination connection process[87]. Yan et al. synthesized four CHA silicoaluminophosphate zeolites (SAPO-34/44) using different OSDAs and studied the host-guest interactions using XRD and simulated annealing[88].
The above experiments prove that the interaction between raw materials and organic templates in gel is of positive significance, especially for the adjustment of aluminum distribution in zeolite. However, recent research has shown that organic template agents not only interact with raw materials and/or their solvates, but also have interactions with metal ions. In some systems, even interactions between template agents exist. These effects result in significant differences in the physicochemical properties of the synthesized zeolite.
Park et al. investigated the synthesis process of Al-CON and B-CON zeolites under hydrothermal conditions. The experiment indicated that the CON nucleus contained Al(SiO)4 specifications and the crystal growth was processed by incorporating B atoms into the CON structure together with the reorganization of the tetrahedral boron silicate specifications. The authors believed that although the details on the reactions between liquid and the solid phases during the crystallization could not be fully understood, they could discriminate the different crystallization stages in the solid phase[89]. Meanwhile, the authors found that during the crystallization of CON, the amount of Na+ in the solid gradually decreased over time, while the quantity of organic template agents gradually increased, which was reflected in the results of thermogravimetric analysis and inductively coupled plasma atomic emission spectroscopy (ICP-AES) [Table 1]. This showed the different interactions between metal ions and organic compounds at varying stages of crystallization. Similarly, Sada et al. synthesized FAU zeolite at 180 °C using TPA+ cations and crystal seeds and studied the crystallization mechanism. Experiments showed that seed crystals and Na+ played a critical role in the FAU secondary nucleus after the solution of seeds, and TPA+ stabilized the FAU framework through the formation of an organic-organic complex during crystal growth[90].
Physicochemical properties of samples[89]
| Samples | Weight loss in TGAa (wt%) | Na/(Al + B)b Molar ratio | Si/Alb | Si/Bb |
| AG | 8.4 | 1.5 | 310 | 615 |
| CON_24 | 10.6 | 1.1 | 300 | 130 |
| CON_48 | 10.8 | 0.9 | 280 | 130 |
| CON_72 | 10.9 | 0.9 | 290 | 130 |
| CON_96 | 10.0 | 0.8 | 280 | 125 |
| CON_120 | 11.2 | 0.7 | 290 | 90 |
| CON_132 | 14.6 | 0.6 | 300 | 60 |
| CON_144 | 16.2 | 0.4 | 300 | 45 |
| CON_168 | 17.5 | 0.2 | 300 | 20 |
Some organic compounds can alter the crystal growth pathway of zeolites. Kumar et al. used scanning probe microscopy to in-situ monitor the crystallization of zeolte LTA with a spatiotemporal resolution that captured dynamic processes in real time. The experiment showed a new crystal growth pathway, which involved the formation of gel-like islands from supersaturated solutions compared to (aluminum) silica molds. The author believed that LTA crystallization occurred through multiple pathways[91]. Later, the authors replaced Na+ with TMA+, leading to the non-classical growth pathways of LTA zeolite. They provided unprecedented insights into the surface growth of zeolite with molecular addition using a time-resolved microscope, which directly captured the occlusion of silica nanoparticles and emphasized the universal role of defects in zeolite crystallization[92]. These results demonstrated the synergistic effect of metal cations and organic compounds in the synthesis of certain zeolites, providing new insights into the role of template agents.
Recent studies have shown that the interactions between template agents themselves can also affect the crystallization process of zeolites, mainly through hydrogen bonding. Liu et al. investigated the synthesis process of SSZ-13 and ITQ-1 zeolites using 2D 1H DQ-SQ. Experiments demonstrated specific spatial correlations between the TMAda+ cations and the SiO-H-OSi hydrogen bonds in the frameworks of SSZ-13 and ITQ-1, indicating that the electronic interactions between the TMAda+ cations and the negative charge centers in the framework oriented the crystallization process. The authors also studied SAPO-11 and SAPO-34. Two-dimensional 1H DQ-SQ confirmed that two organic amines were H-bonded in each unit cell. It could be suggested that the hydrogen bonds between organic amines in the channels were the driving force for the formation of zeolites[93]. This result indicated that in studying the crystallization process of zeolites, the interaction between the template and the framework atoms and the interaction between the template itself should be considered. Afterward, they used the same method to study S-1 zeolite, and the experiment proved that the SiO…H-OSi hydrogen bonds between lamellae were formed and gradually transformed into the in-cage ones during the crystallization process. The SiO…H-OSi hydrogen bond between lamellae acted as a connector to assemble the silicate species together to generate the zeolite framework[94].
In conclusion, organic templates exist in various forms in gel, such as existing independently, interacting with raw materials, forming cluster molecules in gel, interacting through hydrogen bonds, and existing in formed zeolite channels. Experimental results have shown that there are various host-guest interactions related to template agents in zeolite synthesis systems. These interactions between the host and guest lead to the complexity of the synthesis system, affecting the structural orientation of the template agent. The crystallization process of zeolite is a comprehensive effect of these factors, and in-depth research on the host-guest interactions remains an important aspect of future research on the zeolite crystallization mechanism.
Other organic additives
In addition to organic amines and quaternary ammonium template agents with structural directing effects, many organic molecules, while unable to act as structure-directing agents, can modify the crystal growth process of zeolites. These molecules, known as crystal growth regulators, can adjust the pore properties of zeolites and influence the atomic distribution within their frameworks. Specifically, some form complexes with transition metals, facilitating their incorporation into the zeolite framework. Others form micelles in the gel to become hierarchical pore-forming agents. Certain surfactants alter the aggregation state of the gel, thus changing the crystallization mechanism of the zeolite. In summary, adding organic compounds to zeolite systems is an effective means of regulating zeolite performance.
Some substances decompose, producing compounds that affect the synthesis system and alter the crystal growth pathway of zeolites. Qin et al. added arginine to the synthesis system of S-1 zeolite, switching the dominant mode of growth from non-classical to classical pathways. The experiment proved that arginine decomposed into guanine in the synthesis system, inhibiting non-classical pathways[95].
In the above experiment, arginine switches the crystal growth pathways of S-1 from non-classical pathways to classical pathways. Similarly, other substances can switch the crystal growth path from classical to non-classical pathways. Zhang et al. added 1,3,5-benzenetricarboxylic acid (H3BTC) to the synthesis system of TS-1 to regulate the distribution of titanium species. H3BTC converted the precursor gel to the solid-liquid mixed state, leading to the transformation of the traditional liquid phase mechanism to solid-liquid combined mechanism to form TS-1 crystals. The performance observed in the oxidation of 1-hexene was objectively improved[96]. After that, they changed the crystal growth pathways of TS-1 zeolite from classical to non-classical pathways by adding polyacrylamide (PAM) to the TS-1 zeolite synthesis system. They also demonstrated the diffraction pattern of zeolite precursors under the non-classical pathways through TEM. At the same time, The PAM-regulated TS-1 zeolite exhibited enhanced catalytic performance in oxidative reactions[55]. Afterward, they replaced polymer PAM with acrylamide (AM) monomers and other similar substances, regulating the distribution of framework titanium species while adjusting the zeolite particle size. The authors attributed this to the interaction between monomers and titanium species and the self-polymerization of AM. The obtained zeolites exhibited excellent performance in ODS reaction[97]. Overall, these studies underscore the complexity of elucidating the role of modifiers in zeolite synthesis. Experiments have shown that organic additives can alter both the crystallization mechanism and the crystal growth pathways.
Some cationic polymers, such as polydimethyldiallyl ammonium chloride (PDDA) and polyhexamethylene biguanide hydrochloride (PHMB), can also regulate the growth of zeolites. Unlike quaternary ammonium salts, their organic parts are so large that they cannot function as structural directing agents. In the synthesis system, they mainly act as surfactants, affecting the crystallization process of zeolites by adjusting the colloid chemistry of the synthesis system. In 2022, Dai et al. discovered that PDDA can accelerate the crystallization of SSZ-13 and adjust the particle size by several orders of magnitude. Light attention techniques demonstrated that polymers in the synthetic system accelerated the aggregation of amorphous substances, accelerated nucleation, and thus altered the particle size of zeolites. A lower polymer concentration can achieve high-speed synthesis of SSZ-13 at a very low cost[98]. In 2024, Ren et al. synthesized TS-1 with a short B-axis by adding PDDA to the synthesis system. Experiments had shown that PDDA significantly increased the titanium content on the surface of zeolites, boosted the amount of hexacoordinated titanium, and reduced the formation of anatase. The obtained zeolites exhibit excellent performance in ODS[99]. Research had shown that PHMB exhibits strong coordination with aluminum species in the synthetic system, resulting in the presence of hexacoordinated Al3+ species in the micropores of SAPO-11 zeolite and regulating the crystal orientation of the zeolite to form plate-like SAPO-11[100]. A similar effect was observed in MFI (ZSM-5, S-1, and TS-1), where PHMB allowed more Al and Ti to enter the framework, adjusting the crystal orientation to form plate-like MFI zeolite[101]. Even in the template-free synthesis of the ZSM-22 system, PHMB, as the only organic compound, created a hierarchical pore structure and adjusted the aluminum distribution of the zeolite, reducing the concentration of Brønsted acid[102].
Unlike the additives mentioned above, some additives can accelerate zeolite nucleation by interacting with template agents. Yang et al. added N-methyl-2-pyrrolidone (NMP) to the synthesis system to shorten the crystallization time of ZSM-11. It was found that NMP could facilitate the bonding of TBA+ with aluminosilicate by breaking the hydration layer and accelerating the nucleation process. The authors studied the effects of NMP content, silicon-aluminum ratio, and synthesis temperature on crystallization and proposed an acceleration mechanism of NMP. To verify the accuracy of this mechanism, the authors repeated the experiment using N-ethyl-2-pyrrolidone (NEP) as a replacement for NMP. The observed ZSM-11 exhibited similar hydraulic stability to conventional ZSM-11[103].
In a word, organic additives play multiple roles in the synthesis system of zeolite. The way they influence the crystallization mechanism and crystal growth pathway depends on their form in the synthesis system and their role in the composition of the gel. However, whether acting independently or synergistically, these additives are widely used to regulate the synthesis process of zeolites in order to control the properties of the synthesized materials. Therefore, in-depth research on the crystallization mechanism and zeolite crystallization pathway under the presence of organic additives, especially understanding their form and mechanism of action, is of great significance for the directional design of zeolites with high application performance.
Free radicals
After discussing the impact of organic additives, the next topic of discussion is free radicals, discovered in the past decade, that have specific effects on silicon species during the crystallization process. These radicals are a type of substance with a single electron and active chemical properties and can be introduced into the synthesis system of zeolite by using UV irradiation, free radical initiators, and Fenton reagents. The introduced free radicals can be easily characterized using electron paramagnetic resonance (EPR) spectroscopy. Experimental results have shown that in zeolite synthesis systems, free radicals can accelerate the breaking of old Si–O–Si bonds and the formation of new ones, significantly shortening the induction period of zeolite crystallization[104]. This principle has been applied in aluminosilicate zeolites, pure silica zeolites, ordered mesoporous materials, and silicon aluminum phosphate zeolites.
Specifically, in 2016, Feng et al. found that hydroxyl radicals, generated through UV irradiation and Fenton reagents, could accelerate the breaking of old silicon oxide silicon bonds and the generation of new ones. Guided by this strategy, they accelerated the synthesis of a series of silicon aluminum zeolites, including NaA, NaX, NaZ-21, and silicalite-1[104,105]. Later, Wang et al. added hydrogen peroxide as a free radical initiator to the synthesis system of zeolite Y, successfully increasing the silicon-aluminum ratio of zeolite Y without the use of organic templates, and as shown in Figure 8, the signal of free radicals can be detected by EPR[106].
Figure 8. EPR spectrum of initial synthesis gel containing spin-trapping agent DMPO in the synthesis system[104]. Copyright © 2016, American Association for the Advancement of Science. EPR: Electron paramagnetic resonance; DMPO: 5,5-dimethyl-1-pyrroline N-oxide.
Some researchers have introduced this conclusion into the synthesis system of silicon aluminum phosphate zeolites. Zhang et al. added potassium persulfate to the synthesis system of SAPO-18 zeolite, greatly accelerating its crystallization. Experimental results showed that extremely low concentrations of potassium persulfate (nK2S2O8:nAl2O3 ≈ 10-3) could accelerate the crystallization of SAPO-18, and the introduction of free radicals could enhance the acidity of the zeolite. The MTO performance of the obtained samples was improved[107]. Zhou et al. recognized that silicon species acted as the crystallization centers of SAPO-34 zeolite when free radical initiators were added to the synthesis system. Experimental results showed that free radical initiators had the optimal concentration and could effectively shorten the crystallization induction period of SAPO-34 zeolite[108], which confirmed previous conclusions[109].
In summary, whether in the silica-alumina zeolite or the phosphate silica-alumina zeolite system, free radical initiators accelerate the activity of silicon species in the system and increase the crystallization rate of the zeolite. Under the assistance of free radicals, high crystallinity zeolites can be produced within the same duration. There are various ways to introduce free radicals into zeolite systems, and the operation is simple and convenient. This method has great potential for expansion and application.
Host-guest interaction in IZC process
When the raw material used for synthesizing zeolite is zeolite with a different topological structure, this method is called the IZC method. This involves the fracturing of the topological structure of the old zeolite and the formation of new topological structures in the zeolites. Typically, the IZC process can be divided into the following four stages: (I) incongruent source dissolution; (II) an equilibrium or induction phase with steady compositions; (III) crystallization toward a crystalline material; and finally, (IV) maturation of fully crystalline materials. During this process, the smallest building units formed by the decomposition of old zeolites and their role in the formation of new zeolites have always been a focus. Additionally, research has focused on the host-guest interactions in the IZC process, such as how old zeolites decompose under the influence of new template agents, and how the “structural fragments” formed after decomposition recombine to form new zeolites.
Undoubtedly, in the direct crystallization process, “non framework” metal ions act as templates and charge balancers. However, in the IZC process, Al species serve as the driving force in IZC synthesis, with possible effects including charge, dissolution, concentration, and mobility. Devos et al. investigated the IZC of FAU to MFI. For comparison, they also studied equivalent crystallization systems without Na+ and compared the differences between the IZC process and the SSZ-13 zeolite crystallization process. The authors believed that the key to this study was temporal synthesis profiles while probing the distribution and evolution of proximal acid sites with divalent cation capacity measurements. The author detected changes in local charges, distinguishing between Al-loving and Al-averse systems, enabling a new degree of control over the acidity and ion-exchange properties of zeolites[110]. This provides an important reference for us to understand the role of metal ions in the conversion process of zeolite.
In addition to metal cations, water is also an important factor in the IZC process. Zhang et al. investigated the IZC process from FAU to CHA and MFI. They monitored the transformation process of SBUs using UV Raman spectroscopy. Experiments have shown that D6R is an important intermediate. For the IZC from FAU to CHA, D6R is directly converted, while for the conversion from FAU to MFI, it is first decomposed into single 6Rs (S6Rs), which is then assembled into the MFI framework. Meanwhile, 5Rs are only observed in the MFI framework formation, suggesting that the SBUs in the transformation of FAU to MFI are S6Rs compared to 5Rs[111]. Later, they also studied the transformation process of FAU, MFI, and
In the above IZC process, zeolite decomposes in the synthesis system and is retained as SBU or larger units. However, recent research suggests that the IZC process does not necessarily follow this exact pattern. Li
Figure 9. Preparation and structure of ZEO-4A, ZEO-4B and ZEO-5[16]. Copyright © 2024, The Author(s), under exclusive licence to Springer Nature Limited.
These two examples utilize the host-guest interaction between guest molecules and zeolite frameworks to synthesize new structures and raise new questions about the conversion process between zeolites. Specifically, it is necessary to consider whether the zeolite structure of the raw material needs to be completely separated during the zeolite conversion process. Additionally, what are the largest structural units that can be retained during zeolite conversion and how are they preserved? Addressing these questions will be the focus of future research on the IZC process. Based on Chen’s strategy, the development of more extra-large pore zeolite structures is anticipated in the future.
CRYSTALLIZATION KINETICS OF ZEOLITES
The crystallization of zeolite under hydrothermal conditions follows a general pattern of crystal growth and exploring the zeolite crystallization from the perspective of macroscopic statistical laws has certain theoretical and practical significance. The study of zeolite crystallization kinetics primarily focuses on depicting the crystallization curve.
Dynamics research based on macroscopic phenomena
Generally speaking, the crystallization curve of zeolite shows an “S” shape. During the early stages of crystallization, there is a considerable period during which crystals cannot be detected by XRD, known as the induction period. After crystallization begins, the crystallization rate gradually increases, which is a self-catalytic process. As the process continues, the rate gradually slows down, and then the crystallization curve tends to flatten out. With ongoing research, the Avrami equation has been proposed to describe the crystallization process of crystals.
Chen et al. synthesized ZSM-5 zeolite using the leached illite clay by organic template-free and solvent-free methods. The authors investigated its crystallization behavior and kinetics at different temperatures. According to calculations, the apparent activation energies for nucleation and growth were 61.0 and 70.9 kJ/mol, respectively[113]. Similarly, the crystallization process of NaA zeolite according to solid-state theory also follows the Avrami equation, and in-situ Raman spectroscopy revealed the transformation process of SBUs during the crystallization process. Experiments showed that mesoscale intermediates were formed in the aging stage of gel, primarily with a double quaternary ring and beta cage structure. The synthesized zeolite had excellent exchange performance[114].
Although some researchers have also fitted the Avrami equation based on relative crystallinity, their understanding of the crystallization process varies. Corregidor et al. investigated the crystallization kinetics of ZSM-5 synthesized under hydrothermal conditions at different temperatures using natural perlite as a silicon aluminum source. The relative crystallinity of ZSM-5 zeolite was obtained by using XRD and the crystallization curve was plotted. The results indicated that the crystallization curve conformed to the nonlinear Avrami equation, and the authors calculated kinetic constants (k), Introduction periods (t0), and Avrami’s components (n). Based on this, A five-step crystallization mechanism has been proposed
Figure 10. Crystallization curves of ZSM-5 zeolite at different temperatures and the schematic diagram of crystallization mechanism[54]. Reprinted (adapted) with permission from Corregidor P, Acosta D, Destéfanis H. Kinetic study of seed-assisted crystallization of ZSM‑5 zeolite in an OSDA-free system using a natural aluminosilicate as starting source. Ind Eng Chem Res 2018;57:13713-13720. Copyright © 2018 American Chemical Society.
Apart from XRD, the conductivity of zeolite synthesis systems can also be used to characterize the crystallization kinetics of zeolites. Pellens et al. designed a device for in-situ measurement of conductivity and characterized the precursor single-phase hydrogenated silica ionic liquids used for the synthesis of gismondine (GIS) zeolites. The testing of conductivity could characterize the crystallization kinetics of zeolites. The authors proposed a crystallization model to explain the results based on a surface growth mechanism, which derived the experiment indicates zeolite crystallization from highly ionic media proceeds via a multi-step mechanism, involving an initial reversible surface condensation of a growth unit, followed by incorporation of that unit into the growing crystal. A new crystallization model based on surface growth mechanism was proposed to explain the experimental results. The experimental results show that the crystallization of zeolite in high ion media is carried out by a multi-step mechanism[70].
In addition to conductivity, in-situ kinetic characterization of zeolite synthesis systems also includes photon density waves (PDW). Häne et al. used PDWs to in-situ monitor the crystallization process of zeolite A. The onset and end of the crystallization process could be detected online and in real time. The reduced scattering coefficient, being a measure for particle number, size, and morphology, provided distinct process information, including the formation of ammonium particles and their transfer into crystalline zeolite structures. During the crystallization of zeolite A, the system exhibited maximum light scattering at a specific moment, indicating the formation of zeolite A crystals. Following this, the reduced scattering coefficient increased rapidly and eventually stabilized a constant value. The authors suggested that real-time determination of zeolite synthesis endpoint using PDW spectroscopy could reduce synthesis time, improve spatiotemporal yield, and eliminate the need for time-consuming in-process sampling. Finally, they investigated the applicability of PDW spectra under more stringent chemical and physical conditions by monitoring the synthesis of zeolite L[71].
In the above discussion, researchers have studied the macroscopic dynamics of the crystallization mechanism. Beyond the traditional nonlinear Avrami equation, new insights have emerged. In addition to conductivity and PDW, advanced characterization methods from other fields are being introduced into the study of zeolite crystallization mechanisms, further deepening researchers’ understanding. Recently, attention has shifted toward exploring the relationship between macro dynamics and microstructure, with researchers seeking to uncover the connection between these aspects and identify the true driving force behind the crystallization process.
Microscopic driving force of crystallization
The zeolite synthesis system is complex, and the driving force may vary between different systems. However, there is a consensus among researchers that the driving force is related to the microstructure of the zeolite synthesis system. This may involve uneven element distribution within the zeolite precursor or the formation of SBU structures during the early stage of crystallization. Li et al. investigated the nucleation/crystallization process of nanoscale ZSM-5 crystals with aluminum partitioning using HRTEM and EDX. They discovered that amorphous particles with aluminum-rich edges were formed in the initial stage of zeolite growth, with sizes between 40 and 80 nm. Afterward, the nanoparticle crystallized into structures with an amorphous core and crystal shell. Experiments showed that nucleation occurred preferentially at the edges of particles. Subsequently, the rapid propagation of the lattice towards the core resulted in particles with a fully crystalline structure. The high aluminum content at the edges of the particles seemed to be the driving force for the nucleation and growth of ZSM-5 zeolite[116].
The uneven distribution of aluminum above explains well the driving force behind the crystallization of MFI zeolite under non-classical pathways. However, other researchers propose that the crystallization driving force of ZSM-5 comes from the uneven distribution of hydroxyl groups. Zhang et al. extracted protozeolite (PZ) from the growth solution before the crystallization of MFI. These amorphous particles, approximately 8-10 nm in size, were rich in Si-OH, exhibited short-range ordering, and possessed microporous properties. The researchers then subjected PZ to ball milling and stamping treatment to produce integrated PZ (IPZ). Subsequently, under hydrothermal conditions, they synthesized ZSM-5 zeolite using PZ and IPZ as the sole raw materials and TPA as the template agent, thereby avoiding classical crystallization pathways. The authors characterized PZ, IPZ, and the resulting ZSM-5 using SEM, TEM, and SS NMR techniques. The results revealed that the crystallization process of ZSM-5 synthesized from PZ and IPZ followed non-classical pathways, with the microparticles composing ZSM-5 exhibiting diffraction patterns [Figure 11]. Ball-milling caused a change in the chemical environment of silicon hydroxyl groups. The authors believe that the oriented coalescence of the IPZ is achieved through condensation of high-density silanols, leading to anisotropic rates of non-classical growth. This finding is not only meaningful for studying non-classical crystallization pathways, but also establishes a new method for synthesizing zeolite nanosheets via silanol-engineered non-classical pathways[117].
Figure 11. Proposed evolution process of MFI-type zeolite nanosheets[117]. Reprinted (adapted) with permission from Zhang Q, Li J, Wang X, et al. Silanol-Engineered Nonclassical Growth of Zeolite Nanosheets from Oriented Attachment of Amorphous Protozeolite Nanoparticles. J Am Chem Soc 2023;145:21231-21241. Copyright © 2023 American Chemical Society.
For a long time, the formation process of SAPOs has been described as the incorporation of silicon atoms into the aluminum phosphate framework through isomorphic substitution through SM2 and SM3 mechanisms. However, this explanation fails to account for certain phenomena: for some topological structures, AlPO is easy to synthesize while SAPO is challenging to synthesize (such as AFN). Conversely, for other topological structures, SAPO is readily synthesized, but AlPO proves difficult (CHA). Therefore, even for the same topology, the crystallization mechanisms of AlPO/SAPO are likely different. Zhou et al. identified silicon species as the crystallization centers of SAPO-34 zeolite by adding free radical initiators to the synthesis system. Experimental results have shown that free radical initiators have the optimal concentration and can effectively shorten the crystallization induction period of SAPO-34 zeolite[108], which confirms previous conclusions[109]. Later, they verified these findings by adding polymers to the synthesis system, changing the gel concentration, and using the two-step crystallization process to characterize the early synthesis system using liquid NMR[118]. Finally, they applied these insights to synthesize SAPO-34/SAPO-14 composite zeolites through a seed-assisted method[119]. The resulting zeolites exhibited MTO performance. However, Zhou et al. have yet to address a problem: what are the composition and structure of the crystallization center of SAPO-34?
A few years later, this issue was resolved. Zhang et al. designed a top-down synthesis system for SAPO-34 zeolite and conducted extensive investigations. Using Fourier transform ion cyclotron resonance mass spectroscopy (FT-ICR-MS), they characterize the liquid precursors. Their experiments demonstrated that the crystallization centers of SAPO-34 are 4Rs and 6Rs with silicon aluminum structures[120]. After that, they successfully synthesized SAPO-34 zeolite in the SAPO-34 synthesis system using a seed-assisted method with SAPO-18, SAPO-35, and SAPO-56 as seeds. The results indicate that D6R units are key features for a highly effective SAPO seed, potentially providing small fragments containing intact or broken d6r units with Si-O-Al domains after seed dissolution. These fragments then serve as the nucleus for the crystallization of SAPO-34. The obtained zeolite exhibits excellent catalytic performance in the MTO reaction. The authors believe this conclusion provides a theoretical basis for the synthesis of SAPO-34 molecular sieves by the seed-assisted method[121].
Research in both early and recent literature primarily focuses on AlPO-5 and SAPO-34. AlPO-5 is considered the simplest aluminum phosphate zeolite, while SAPO-34 is the most important catalyst for the MTO reaction. The MTO catalytic performance of SAPO-34 is significantly influenced by its structure, and the crystallization mechanism is the factor determining the structure of SAPO-34 zeolite.
Factors affecting crystallization kinetics
Based on numerous examples, the factors that currently influence the crystallization mechanism and crystal growth pathways of zeolites can be summarized as follows: the concentration of the synthesis gel system, silicon content, raw material specifications, crystallization temperature, and even the order in which raw materials are added. The following summarizes the effects of these factors on the zeolite crystallization process.
Concentration of synthesis gel system
The concentration of the synthesis gel is a key factor affecting the crystallization mechanism of zeolites, as it directly influences the concentration of crystallization raw materials. This, in turn, affects the process of raw material polymerization, zeolite nucleation, and crystal growth. However, the impact of gel concentration varies across different synthesis systems. Zhang et al. synthesized nanosized ZSM-5 zeolite by reducing the amount of solvent in the ZSM-5 synthesis system. Experiments showed that concentrated gel could effectively reduce the apparent activation energy of zeolite nucleation, accelerate the nucleation rate of zeolite, and thus decrease the crystal size of zeolite. The ZSM-5 zeolite obtained by this strategy exhibited excellent MTO performance, with a catalytic lifetime even longer than the nano ZSM-5 synthesized by traditional hydrothermal methods[122].
Silicon content
Similar to gel concentration, the silicon content in some systems also affects the crystallization mechanism of zeolite. The increase in silicon content is essentially a result of increased gel concentration in the system. This has led to changes in the nucleation and growth. Jonscher et al. investigated the crystallization of ZSM-5, focusing on the correlation between the Si/Al ratio and zeolite morphology. They characterized zeolites with different Si/Al ratios and crystallization times. The results showed that a slow dissolution recrystallization process has been changed into a fast solid-state transformation with increasing Si/Al ratio. After that, small agglomerates began to assemble into bigger spherical particles [Figure 12A]. As a result, the performance of these two zeolites differs in methanol conversion experiment, which verifies that different crystallization mechanisms can lead to various zeolite structures, thereby exhibiting distinct catalytic performance[123].
Figure 12. (A) Scheme of the mechanistic reason for the interdependency between Si/Al ratio and zeolite morphology of ZSM-5[123]. Copyright © 2021 The Authors. ChemCatChem published by Wiley-VCH GmbH; (B) Schematic illustration of the crystallization pathways of TS-1 zeolite through classical and non-classical processes[125]. Copyright © The Royal Society of Chemistry 2022; (C) Elemental mapping images of EU-45[127]. Copyright © 2022 Elsevier Inc. All rights reserved.
Raw material species
Apart from the influence of organic/inorganic templates discussed above, raw material species here refer to the source of the zeolite framework. In the synthesis system, the reactivity of these raw materials significantly affects the crystallization mechanism of zeolites, mainly due to their varying strengths and order in which they interact with other species in the reaction system. At present, the precursors used for zeolite synthesis mainly include: (1) natural minerals, such as pseudo-boehmite, diatomaceous earth, coal gangue, etc.; (2) Inorganic substances in industrial production, such as silica powder, various aluminum containing inorganic salts, phosphoric acid, phosphite and their salts, etc.; (3) Organic compounds, such as orthosilicate esters (different alkyl groups), aluminum isopropoxide (different alcohols, acids), organic phosphorus, etc.; (4) Zeolite precursors or amorphous aluminosilicates, such as PZ and IPZ; (5) Zeolites with other topological structures, also known as the IZC process.
Chen et al. proposed that the reactivity of reactants and the dynamics of condensation reaction could influence the formation of short-range ordered structural units in amorphous media, and thus greatly affected the nucleation rate of zeolite by comparing the structural evolution of aluminosilicate species in dense phase hydrogel systems when different silicon sources were used. The research results indicate that when white carbon black gradually reacts with NaAlO2 solution at 80 °C, amorphous solids containing a large number of aluminum rich quaternary, and silicon rich hexagonal rings can be generated. The interaction between these rings promotes the construction of the structural building units of zeolite X. In contrast, when sodium silicate solution is mixed with sodium aluminate solution, complex aluminosilicate species will immediately form due to strong condensation reactions. In addition, when the reaction mixture is subjected to hydrothermal treatment at 80 °C, this complex species becomes more stable, significantly hindering the crystallization process. On the contrary, aging the reaction mixture at ambient temperature is beneficial for the formation of short-range ordered structural units in amorphous media, thereby increasing the nucleation rate of zeolite[124].
Bai et al. regulated the growth pathways of TS-1 by controlling the hydrolysis rate of the titanium source in the synthesis system. They monitored the evolution process of zeolite precursors using dynamic light scattering (DLS) techniques. During the crystallization process, the particle size of the precursor gradually increased, and the particle size of the precursor reflected the evolution process information of soluble monomers/oligomers. The authors believed that this could indicate changes in the microstructure. The different microstructures led to various growth pathways of TS-1 [Figure 12B], and TS-1 zeolite crystallized according to non-classical pathways had better performance in olefin epoxidation reaction. This work has provided a reference for regulating the crystallization pathway of zeolites[125].
Crystallization temperature
The crystallization temperature is another important factor affecting the crystallization mechanism. Species in the zeolite synthesis system exhibit varying reaction activities at different temperatures, which may lead to distinct crystallization mechanisms. In a certain sense, the commonly used low-temperature synthesis, two-step/multi-step crystallization methods in the synthesis process are all based on this principle. Houlleberghs et al. synthesized zeolite W (MER topology) with the hydrothermal method, and studied the crystallization mechanism at different temperatures using atomic force microscopy. Zeolite W crystals observed in a highly supersaturated solution at 90 °C through nucleation and spread growth, where synthesis at 175 °C results in prolonged, Spiral growth zeolite W particles. The authors believed that supersaturation conditions determined the nucleation rate, the prevailing crystal growth mechanism, and resulting crystal morphology[126].
Wei et al. investigated the formation mechanism of different precursors produced by hydrolysis of ethyl orthosilicate at different temperatures and EUO zeolites produced by introducing aluminum sources at different times. Experiments proved that the hydrolysis temperature of tetraethylorthosilicate (TEOS) was closely related to the nucleation rate of EUO. When gel was hydrolyzed at low temperatures, the evolution from wormlike precursor to bulk precursor was accelerated. In addition, when a uniform gel with uniform Si-Al distribution was formed, the introduction of sodium aluminate could also promote nucleation. When the gel environment was uneven, the transformation from amorphous to crystalline involved two steps. First, there was *MRE due to the presence of silicon-rich regions [Figure 12C], and then EUO zeolite was formed through the crystallization process. The authors believed that since many zeolite syntheses involved the formation of metastable precursors with heterogeneous composition, the selection of gel preparation procedures in this study could be extended to other related materials[127].
Order of raw material addition
In addition to the factors mentioned above, the order in which raw materials are added can also influence the formation process of zeolites, thereby affecting their microstructure and properties. This may be due to the way the order of addition affects the solvation process of the raw materials, which in turn affects their activity and the interactions between them, resulting in different crystallization processes.
Bok et al. changed the feeding order of the BEA zeolite synthesis system, and studied the differences in the composition, structure, and subsequent crystallization of the two gel systems, the initial addition of silicon sources formed aluminum-rich gel, and nucleation occurred in the liquid phase. The initial addition of an aluminum source formed amorphous hydrogel, and the formation of SBU occurred in the solid phase. It has been demonstrated that to obtain zeolite Beta crystals without admixtures of other phases, it is necessary to have not only a high concentration of TEA+ cations, but also a high concentration of aluminum in the reaction mixture[128]. Similarly, Shen et al. obtained two ZSM-5 samples by only changing the mixing order of Si and Al sources. The two not only have differences in crystal morphology, but also have clear differences in inner structure. The authors posited that the process began with biased nucleation within the Si-rich aluminosilicate amorphous precursor, which was followed by multistage crystal growth within a heterogeneous feedstock. This ultimately led to the formation of Al-zoning and distinct structural features within the ZSM-5 zeolite crystals. Experimental results have shown that zeolites with different structural properties exhibit high cracking activity in n-hexane cracking reactions, while demonstrating varying activity, stability, and product selectivity in ethylene dehydrogenation aromatization (EDA) reactions. This is a typical example reflecting the mechanism-structure-performance relationship in the synthesis process of zeolites[129].
Etching and recrystallization
Although in the dynamics section, we mainly discuss crystallization kinetics, it should be noted that under certain specific conditions, the crystallization and dissolution of zeolite happen simultaneously. Chen et al. synthesized silicalite-1 in advance and treated it with a mixed solution of TPAOH and aluminum isopropoxide under hydrothermal conditions. Etching and recrystallization occurred during this process, ultimately resulting in hierarchical single crystalline ZSM-5 nanocages. The authors proposed an anisotropic-kinetics transformation strategy through the study of numerous samples. They used the concentration of TPAOH in the solution to adjust the rate difference between Etching and recrystallization, and synthesized ZSM-5 with adjustable Si/Al ratio and morphology. This method can also be used to synthesize B-MFI and Ga-MFI nanocages. The authors believe that this approach can construct zeolites with complex nanocage structures, and the obtained samples exhibit excellent performance in MTH reactions[130].
Here, we take MFI (ZSM-5/Silicalite-1/TS-1) as an example and list the factors that affect crystallization kinetics, as shown in Table 2. However, it should be noted that the synthesis systems and crystallization conditions of these works are not the same.
Factors affecting the crystallization kinetics of MFI zeolite
| Factors | Infuluence | Ref. |
| Uneven distribution of species | The uneven distribution of Al or silanol will significantly accelerate the crystallization of ZSM-5 | [116,117] |
| Concentration of synthesis gel system | Reduce the apparent activation energy nucleation, accelerate the nucleation rate and reduce the crystal size | [122] |
| Silicon content | Slow dissolution recrystallization process has been changed into a fast solid-state-transformation with the increase of Si/Al ratio | [123] |
| Raw material species | The hydrolysis rate of organic titanium source regulates the crystal growth pathways of TS-1 | [125] |
| Crystallization temperature | Adjust the nucleation rate and reduce the crystal size | [131] |
| Order of raw material addition | Affects the chemical composition of crystal nuclei and the growth mechanism | [129] |
| Polymer | PHMB affect the framework and changed the crystal morphology | [101] |
From the extensive research mentioned above, we can draw the following conclusion: Several factors influence the crystallization kinetics of zeolites, which can be summarized as follows: (1) Activity effects, such as gel concentration, silicon species concentration and crystallization temperature; (2) The impact of species, including raw material species and their addition process. These factors affect the crystallization kinetics of zeolite, which in turn influence the structure and properties of the synthesized zeolite. Studying these factors can provide valuable insights for designing zeolites before synthesis, allowing for the adjustment of their structure and function.
CONCLUSION AND OUTLOOK
In this article, we have summarized recent research on the crystallization mechanism of zeolites, categorizing and reviewing relevant literature based on characterization methods, host-guest interaction and crystallization kinetics. These studies focus on advanced characterization, in-situ and ex-situ characterization, integrating recent achievements in spectroscopy, electromagnetics, and optics to track the nucleation and growth processes of zeolites. This includes the conversion of raw materials, the formation of SBU and the assembly of zeolite layers. On this basis, we describe the specific crystallization mechanism and crystal growth pathways of the material. We also explore the host-guest interactions between template agents and zeolite framework atoms in the synthesis process, discuss the macroscopic crystallization kinetics of zeolites, and examine possible crystallization driving forces in the synthesis system, along with the relationship between the two.
Currently, researchers have distinguished the crystallization mechanisms and crystal growth processes of zeolites, analyzing the factors that affect zeolite crystallization in specific systems. Some studies have even applied these mechanisms to improve the application performance of zeolites. Despite the depth of research, there are still unresolved issues and potential misunderstandings regarding the crystallization mechanism of zeolites. For example, although abundant evidence supports the significant role of silicon in the crystallization process of silicon-aluminum phosphate zeolites, and in some cases, silicon may even be the crystallization center, some researchers continue to adhere to the outdated, potentially problematic belief that the aluminum phosphate framework forms first and then undergoes silicon isomorphic substitution. This explanation persists despite the lack of experimental evidence. Furthermore, in some synthetic systems, changes in parameters (such as concentration or additives) can alter the crystallization mechanism of zeolites, a fact often overlooked, leading to the use of the same theory to explain different crystallization processes.
With advancements in analytical and materials science, in-situ monitoring of hydrothermal processes has become feasible. However, the development of in-situ characterization instruments with enhanced resistance to high temperatures, high pressures, and alkali corrosion remains a key focus for future research. Additionally, as mentioned earlier, instruments from other research fields have been applied to characterize the zeolite synthesis process. This integration highlights the need for interdisciplinary studies to clarify the relationship between the physicochemical properties of zeolite synthesis systems and the crystallization process.
In-situ characterization of zeolite synthesis systems is crucial, but also presents challenges that can affect the synthesis process. These challenges include, but are not limited to, issues related to energy input, electron-guided flow, and rotational centrifugal forces. For example, when using in-situ NMR technology, the centrifugal force generated by rotation may affect the zeolite crystallization process, potentially slowing down the crystallization of B-MFI. Although in-situ characterization technology has made significant progress, achieving dialectical unity between the principles and results of in-situ and ex-situ characterization remains a challenge for researchers. These methods are not only applicable to zeolite systems but also can be extended to MOF/COF synthesis systems or other phase change systems, such as the crystallization of organic compounds and polymer formation.
The interaction between host and guest during the synthesis of zeolites is another important research area in the crystallization process. Currently, research on host-guest interactions mainly focuses on the interactions between ions (metal cations and quaternary ammonium cations) and zeolite framework atoms/SBUs. However, when two types of template agents are present in the synthesis system, the situation becomes complicated. In this case, it is essential to study the interaction between the template agents to understand the connection and differences between the actual structure and theoretical calculation results, ultimately offering a more accurate description of the crystallization process. Moreover, further research is needed on the host-guest interactions during the IZC process, including how the precursor zeolite decomposes under the influence of the guest and re-polymerizes to complete IZC. Additionally, leveraging host-guest interactions could provide a pathway for synthesizing extra-large microporous zeolites with new topological structures.
In terms of zeolite crystallization kinetics, there is general agreement on the basic fact that the crystallization curve consists of three stages in macroscopic kinetics studies. Although there may be slight differences in the specific descriptions of these stages, they are all based on XRD analysis. Additionally, conductivity and PDW methods have been used to study zeolite crystallization kinetics, providing further insights into the process. At the micro level, understanding the driving force of crystallization lies in identifying specific species and their uneven distribution, to gain a deeper understanding of the crystallization process. Once the crystallization mechanism of zeolites is fully understood, a major challenge remains: synthesizing zeolites with special catalytic, adsorption, and separation properties by adjusting the synthesis system. The future direction will focus on reducing raw material costs, achieving greener synthesis methods, minimizing instrument requirements, and scaling up production for industrial applications based on the crystallization mechanism of the required zeolites.
DECLARATIONS
Authors’ contributions
Article conceptualization and writing: Zhou, Y.
Article conceptualization and revisions: Xu, S.
Administrative support: Wei, Y., Liu, Z.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (22241801, 22022202, 22032005, 22288101, 21991090, 21991092, 21991093) and Dalian Outstanding Young Scientist Foundation (2021RJ01).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Peng, P.; Gao, X. H.; Yan, Z. F.; Mintova, S. Diffusion and catalyst efficiency in hierarchical zeolite catalysts. Natl. Sci. Rev. 2020, 7, 1726-42.
2. Loewenstein, W. The distribution of aluminum in the tetrahedra of silicates and aluminates. Am. Mineral. 1954, 39, 92-6. https://pubs.geoscienceworld.org/msa/ammin/article-abstract/39/1-2/92/539331/The-distribution-of-aluminum-in-the-tetrahedra-of. (accessed 2025-01-22)
3. Li, Y.; Shi, C.; Li, L.; et al. Unraveling templated-regulated distribution of isolated SiO4 tetrahedra in silicoaluminophosphate zeolites with high-throughput computations. Natl. Sci. Rev. 2022, 9, nwac094.
5. Barrer, R. M. Synthesis of a zeolitic mineral with chabazite-like sorptive properties. J. Chem. Soc.1948, 127-32.
6. Vogt, E. T.; Weckhuysen, B. M. Fluid catalytic cracking: recent developments on the grand old lady of zeolite catalysis. Chem. Soc. Rev. 2015, 44, 7342-70.
7. Barrer, R. M.; Denny, P. J. 202. Hydrothermal chemistry of the silicates. Part X. A partial study of the field CaO–Al2O3–SiO2–H2O. J. Chem. Soc.1961, 982-1000.
8. Kokotailo, G. T.; Chu, P.; Lawton, S. L.; Meier, W. M. Synthesis and structure of synthetic zeolite ZSM-11. Nature 1978, 275, 119-20.
9. Gordon, C. P.; Engler, H.; Tragl, A. S.; et al. Efficient epoxidation over dinuclear sites in titanium silicalite-1. Nature 2020, 586, 708-13.
10. Wilson, S. T.; Lok, B. M.; Messina, C. A.; Cannan, T. R.; Flanigen, E. M. Aluminophosphate molecular sieves: a new class of microporous crystalline inorganic solids. J. Am. Chem. Soc. 1982, 104, 1146-7.
11. Bennett, J.; Dytrych, W.; Pluth, J.; Richardson, J.; Smith, J. Structural features of aluminophosphate materials with. Zeolites 1986, 6, 349-60.
12. Yu, J.; Xu, R. Rich structure chemistry in the aluminophosphate family. Acc. Chem. Res. 2003, 36, 481-90.
13. Cheetham, A. K.; Férey, G.; Loiseau, T. Open-framework inorganic materials. Angew. Chem. Int. Ed. 1999, 38, 3268-92.
14. Lin, Q. F.; Gao, Z. R.; Lin, C.; et al. A stable aluminosilicate zeolite with intersecting three-dimensional extra-large pores. Science 2021, 374, 1605-8.
15. Li, J.; Gao, Z. R.; Lin, Q. F.; et al. A 3D extra-large-pore zeolite enabled by 1D-to-3D topotactic condensation of a chain silicate. Science 2023, 379, 283-7.
16. Gao, Z. R.; Yu, H.; Chen, F. J.; et al. Interchain-expanded extra-large-pore zeolites. Nature 2024, 628, 99-103.
17. Zhang, W.; Zhang, P.; Liu, K.; et al. Synthesis and crystallization mechanisms of zeolites: a comprehensive review. Int. J. Appl. Ceram. Technol. 2026, 23, e70169.
18. Fan, D.; Tian, P.; Su, X.; et al. Aminothermal synthesis of CHA-type SAPO molecular sieves and their catalytic performance in methanol to olefins (MTO) reaction. J. Mater. Chem. A. 2013, 1, 14206.
19. Fan, D.; Tian, P.; Xu, S.; et al. A novel solvothermal approach to synthesize SAPO molecular sieves using organic amines as the solvent and template. J. Mater. Chem. 2012, 22, 6568.
20. Askari, S.; Sedighi, Z.; Halladj, R. Rapid synthesis of SAPO-34 nanocatalyst by dry gel conversion method templated with morphline: investigating the effects of experimental parameters. Micropor. Mesopor. Mat. 2014, 197, 229-36.
21. Askari, S.; Halladj, R.; Sohrabi, M. Methanol conversion to light olefins over sonochemically prepared SAPO-34 nanocatalyst. Micropor. Mesopor. Mat. 2012, 163, 334-42.
22. Askari, S.; Halladj, R. Ultrasonic pretreatment for hydrothermal synthesis of SAPO-34 nanocrystals. Ultrason. Sonochem. 2012, 19, 554-9.
23. Yang, G.; Wei, Y.; Xu, S.; et al. Nanosize-enhanced lifetime of SAPO-34 catalysts in methanol-to-olefin reactions. J. Phys. Chem. C. 2013, 117, 8214-22.
24. Lin, S.; Li, J.; Sharma, R. P.; Yu, J.; Xu, R. Fabrication of SAPO-34 crystals with different morphologies by microwave heating. Top. Catal. 2010, 53, 1304-10.
25. Sun, Y.; Ma, S.; Du, Y.; et al. Solvent-free preparation of nanosized sulfated zirconia with Brønsted acidic sites from a simple calcination. J. Phys. Chem. B. 2005, 109, 2567-72.
26. Xi, D.; Sun, Q.; Xu, J.; et al. In situ growth-etching approach to the preparation of hierarchically macroporous zeolites with high MTO catalytic activity and selectivity. J. Mater. Chem. A. 2014, 2, 17994-8004.
27. Li, X.; Li, K.; Ma, H.; Xu, R.; Tao, S.; Tian, Z. Ionothermal synthesis of a CHA-type aluminophosphate molecular sieve membrane and its formation mechanism. Micropor. Mesopor. Mat. 2015, 217, 54-62.
28. Xu, Y.; Tian, Z.; Wang, S.; et al. Microwave-enhanced ionothermal synthesis of aluminophosphate molecular sieves. Angew. Chem. Int. Ed. Engl. 2006, 118, 4069-74.
29. Sun, Q.; Wang, N.; Guo, G.; Yu, J. Ultrafast synthesis of nano-sized zeolite SAPO-34 with excellent MTO catalytic performance. Chem. Commun. 2015, 51, 16397-400.
30. Alam, S. F.; Kim, M.; Kim, Y. J.; et al. A new seeding method, dry rolling applied to synthesize SAPO-34 zeolite membrane for nitrogen/methane separation. J. Membr. Sci. 2020, 602, 117825.
31. Ding, H.; Ding, J.; Liu, W.; et al. A phase-transfer crystallization pathway to synthesize ultrasmall silicoaluminophosphate for enhanced catalytic conversion of dimethylether-to-olefin. CrystEngComm 2019, 21, 577-82.
32. Cejka, J.; Zilkova, N. Synthesis and structure of zeolites. Chem. Listy. 2000, 94, 278-87. http://www-.chemicke-listy.cz/ojs3/index.php/chemicke-listy/article/view/2507. (accessed 2026-04-27)
33. Smith, J. V. Topochemistry of zeolites and related materials. 1. Topology and geometry. Chem. Rev. 1988, 88, 149-82.
34. IZA. Database of Zeolite Structures. Secondary Building Units. https://europe.iza-structure.org/IZA-SC/SBUList.html. (accessed 2025-01-22).
35. Barrer, R. M.; Baynham, J. W.; Bultitude, F. W.; Meier, W. M. 36. Hydrothermal chemistry of the silicates. Part VIII. Low-temperature crystal growth of aluminosilicates, and of some gallium and germanium analogues. J. Chem. Soc.1959, 195-208.
36. Mallette, A. J.; Shilpa, K.; Rimer, J. D. The current understanding of mechanistic pathways in zeolite crystallization. Chem. Rev. 2024, 124, 3416-93.
37. Kerr, G. T. Chemistry of crystalline aluminosilicates. I. Factors affecting the formation of zeolite A. J. Phys. Chem. 1966, 70, 1047-50.
38. Zhdanov, S. P. Some problems of zeolite crystallization. In: Flanigen EM, Sand LB, editors. Molecular sieve zeolites - I. Washington: American Chemical Society; 1974. pp. 20-43.
39. Angell, C. L.; Flank, W. H. Mechanism of zeolite A synthesis. In: Katzer JR, editor. Molecular sieves - II. Washington: American Chemical Society; 1977. pp. 194-206.
40. Xu, R.; Liu, X. The mechanism of the formation and the crystal growth of molecular-sieve-zeolite. IX. The liquid phase mechanism of the formation of zeolite K-L. Acta. Chim. Sin. 1984, 42, 227-32. https://sioc-journal.cn/Jwk_hxxb/CN/Y1984/V42/I3/227. (accessed 2025-01-22)
41. Ueda, S.; Murata, H.; Koizumi, M.; Nishimura, H. Crystallization of mordenite from aqueous solutions. Am. Mineral. 1980, 65, 1012-9. http://www.minsocam.org/ammin/AM65/AM65_1012.pdf. (accessed 2025-01-22)
42. Tian, P.; Su, X.; Wang, Y.; et al. Phase-transformation synthesis of SAPO-34 and a novel SAPO molecular sieve with RHO framework type from a SAPO-5 precursor. Chem. Mater. 2011, 23, 1406-13.
43. Breck, D. W.; Flanigen, E. Synthesis and properties of Union Carbide zeolites L, X and Y. Mol. Sieves. 1968:47-60. https://scholar.google.com/scholar_lookup?&title=Synthesis%20and%20properties%20of%20Union%20Carbide%20zeolites%20L%2C%20X%2C%20and%20Y&publication_year=1968&author=Breck%2CD.W.&author=Flanigen%2CE.M.#d=gs_cit&t=1737550892811&u=%2Fscholar%3Fq%3Dinfo%3ArYOIpYTbC_YJ%3Ascholar.google.com%2F%26output%3Dcite%26scirp%3D0%26hl%3Dzh-CN. (accessed 2025-01-22)
44. Wenyang, X.; Jianquan, L.; Wenyuan, L.; Huiming, Z.; Bingchang, L. Nonaqueous synthesis of ZSM-35 and ZSM-5. Zeolites 1989, 9, 468-73.
45. Wu, Q.; Meng, X.; Gao, X.; Xiao, F. S. Solvent-free synthesis of zeolites: mechanism and utility. Acc. Chem. Res. 2018, 51, 1396-403.
46. Shao, Y.; Wang, H.; Liu, X.; et al. Single-crystalline hierarchically-porous TS-1 zeolite catalysts via a solid-phase transformation mechanism. Micropor. Mesopor. Mat. 2021, 313, 110828.
47. Li, S.; Yang, H.; Wang, S.; Dong, M.; Wang, J.; Fan, W. Facile synthesis of hierarchical macro/microporous ZSM-5 zeolite with high catalytic stability in methanol to olefins. Micropor. Mesopor. Mat. 2022, 329, 111538.
48. Derouane, E. G.; Determmerie, S.; Gabelica, Z.; Blom, N. Synthesis and characterization of ZSM-5 type zeolites I. physico-chemical properties of precursors and intermediates. Appl. Catal. 1981, 1, 201-24.
49. Iton, L. E.; Trouw, F.; Brun, T. O.; Epperson, J. E.; White, J. W.; Henderson, S. J. Small-angle neutron-scattering studies of the template-mediated crystallization of ZSM-5-type zeolite. Langmuir 1992, 8, 1045-8.
50. Wohlrab, S.; Pinna, N.; Antonietti, M.; Cölfen, H. Polymer-induced alignment of DL-alanine nanocrystals to crystalline mesostructures. Chemistry 2005, 11, 2903-13.
51. Karthika, S.; Radhakrishnan, T. K.; Kalaichelvi, P. A review of classical and nonclassical nucleation theories. Cryst. Growth. Des. 2016, 16, 6663-81.
52. Niederberger, M.; Cölfen, H. Oriented attachment and mesocrystals: non-classical crystallization mechanisms based on nanoparticle assembly. Phys. Chem. Chem. Phys. 2006, 8, 3271-87.
53. Jehannin, M.; Rao, A.; Cölfen, H. New horizons of nonclassical crystallization. J. Am. Chem. Soc. 2019, 141, 10120-36.
54. Corregidor, P. F.; Acosta, D. E.; Destéfanis, H. A. Kinetic study of seed-assisted crystallization of ZSM-5 zeolite in an OSDA-free system using a natural aluminosilicate as starting source. Ind. Eng. Chem. Res. 2018, 57, 13713-20.
55. Zhang, J.; Bai, R.; Zhou, Y.; et al. Impact of a polymer modifier on directing the non-classical crystallization pathway of TS-1 zeolite: accelerating nucleation and enriching active sites. Chem. Sci. 2022, 13, 13006-14.
56. Li, P.; Xiong, G.; Liu, L.; Wang, L. Investigation on the effect of zeolite precursor on the formation process of MCM-41containing zeolite Y building units. Spectrochim. Acta A Mol. Biomol. Spectrosc.2013, 107, 218-26.
57. Zheng, M.; Chu, Y.; Wang, Q.; Wang, Y.; Xu, J.; Deng, F. Advanced solid-state NMR spectroscopy and its applications in zeolite chemistry. Prog. Nucl. Magn. Reson. Spectrosc.2024, 140, 1-41.
58. Berkson, Z. J.; Messinger, R. J.; Na, K.; Seo, Y.; Ryoo, R.; Chmelka, B. F. Non-topotactic transformation of silicate nanolayers into mesostructured MFI zeolite frameworks during crystallization. Angew. Chem. Int. Ed. Engl. 2017, 56, 5164-9.
59. Shi, J.; Anderson, M. W.; Carr, S. W. Direct observation of zeolite a synthesis by in situ solid-state NMR. Chem. Mater. 1996, 8, 369-75.
60. Romanova, E.; Scheffler, F.; Freude, D. Crystallization of zeolite MFI under supergravity, studied in situ by 11B MAS NMR spectroscopy. Micropor. Mesopor. Mat. 2009, 126, 268-71.
61. Gerardin, C.; Haouas, M.; Lorentz, C.; Taulelle, F. NMR quantification in hydrothermalin situ syntheses. Magn. Reson. Chem. 2000, 38, 429-35.
62. Deuchande, T.; Breton, O.; Haedelt, J.; Hughes, E. Design and performance of a high pressure insert for use in a standard magic angle spinning NMR probe. J. Magn. Reson. 2006, 183, 178-82.
63. Zhao, Z.; Xu, S.; Hu, M. Y.; Bao, X.; Hu, J. Z. In situ high temperature high pressure MAS NMR study on the crystallization of AlPO4-5. J. Phys. Chem. C. 2016, 120, 1701-8.
64. Ivanova, I. I.; Kolyagin, Y. G. Application of multinuclear MAS NMR for the in situ monitoring of hydrothermal synthesis of zeolites. Chemistry 2021, 27, 14143-67.
65. Xue, Z.; Ma, J.; Hao, W.; et al. Synthesis and characterization of ordered mesoporous zeolite LTA with high ion exchange ability. J. Mater. Chem. 2012, 22, 2532-8.
66. Fan, F.; Feng, Z.; Li, G.; Sun, K.; Ying, P.; Li, C. In situ UV Raman spectroscopic studies on the synthesis mechanism of zeolite X. Chemistry 2008, 14, 5125-9.
67. Fan, F.; Feng, Z.; Sun, K.; et al. In situ UV Raman spectroscopic study on the synthesis mechanism of AlPO-5. Angew. Chem. Int. Ed. Engl. 2009, 48, 8743-7.
68. Honda, S.; Matsuda, Y.; Nakashima, K.; Daiko, Y.; Hashimoto, S.; Iwamoto, Y. In situ Raman spectroscopy analysis of local structure in dry gel for zeolite beta synthesis. Int. J. Appl. Ceram. Technol. 2023, 20, 1081-9.
69. Embrechts, H.; Kriesten, M.; Hoffmann, K.; Peukert, W.; Hartmann, M.; Distaso, M. Elucidation of the formation mechanism of metal–organic frameworks via in-situ Raman and FTIR spectroscopy under solvothermal conditions. J. Phys. Chem. C. 2018, 122, 12267-78.
70. Pellens, N.; Doppelhammer, N.; Asselman, K.; et al. A zeolite crystallisation model confirmed by in situ observation. Faraday. Discuss. 2022, 235, 162-82.
71. Häne, J.; Brühwiler, D.; Ecker, A.; Hass, R. Real-time inline monitoring of zeolite synthesis by photon density wave spectroscopy. Micropor. Mesopor. Mat. 2019, 288, 109580.
72. Paula, C.; Wisser, D.; Rangus, M.; et al. Phase transformations in porous materials studied by in situ solid-state NMR spectroscopy and in situ X-ray diffraction. J. Phys. Chem. C. 2020, 124, 19136-45.
73. Zhou, Y.; Liao, Y.; Lou, C.; et al. Dynamic migration of framework Al atoms during the crystallization of MOR zeolite. J. Energy. Chem. 2026, 117, 657-68.
74. Zhang, Q.; Mayoral, A.; Terasaki, O.; et al. Amino acid-assisted construction of single-crystalline hierarchical nanozeolites via oriented-aggregation and intraparticle ripening. J. Am. Chem. Soc. 2019, 141, 3772-6.
75. Asselman, K.; Kirschhock, C.; Breynaert, E. Illuminating the black box: a perspective on zeolite crystallization in inorganic media. Acc. Chem. Res. 2023, 56, 2391-402.
76. Wang, L.; Yan, N.; Liu, X.; et al. Unraveling the twin and tunability of the crystal domain sizes in the medium-pore zeolite ZSM-57 by electron crystallography. Chemistry 2019, 25, 1029-36.
77. Yamada, H.; Sukenaga, S.; Ohara, K.; et al. Comparative study of aluminosilicate glass and zeolite precursors in terms of Na environment and network structure. Micropor. Mesopor. Mat. 2018, 271, 33-40.
78. Chawla, A.; Linares, N.; Li, R.; García-Martínez, J.; Rimer, J. D. Tracking zeolite crystallization by elemental mapping. Chem. Mater. 2020, 32, 3278-87.
79. Sun, C.; Liu, Z.; Wang, S.; et al. Anionic tuning of zeolite crystallization. CCS. Chem. 2021, 3, 189-98.
80. Sun, C.; Chen, W.; Wang, J.; et al. An anion-promoted increase of the SiO2/Al2O3 ratio of zeolites. Inorg. Chem. Front. 2022, 9, 1293-9.
81. Wang, S.; Wang, C.; Liu, H.; et al. Acceleration effect of sodium halide on zeolite crystallization: ZSM-12 as a case study. Micropor. Mesopor. Mat. 2022, 331, 111652.
82. Mintova, S.; Gilson, J.; Valtchev, V. Advances in nanosized zeolites. Nanoscale 2013, 5, 6693-703.
83. Sheng, N.; Chu, Y.; Xin, S.; et al. Insights of the crystallization process of molecular sieve AlPO4-5 prepared by solvent-free synthesis. J. Am. Chem. Soc. 2016, 138, 6171-6.
84. Nishitoba, T.; Nozaki, T.; Park, S.; et al. CHA-type zeolite prepared by interzeolite conversion method using FAU and LTL-type zeolite: effect of the raw materials on the crystallization mechanism, and physicochemical and catalytic properties. Catalysts 2020, 10, 1204.
85. Ivanova, I. I.; Kolyagin, Y. G.; Kasyanov, I. A.; Yakimov, A. V.; Bok, T. O.; Zarubin, D. N. Time-resolved in situ MAS NMR monitoring of the nucleation and growth of zeolite BEA catalysts under hydrothermal conditions. Angew. Chem. Int. Ed. Engl. 2017, 56, 15344-7.
86. Alahakoon, S. H.; Willans, M. J.; Huang, Y. In situ multinuclear magic-angle spinning NMR: monitoring crystallization of molecular sieve AlPO4-11 in real time. JACS. Au. 2023, 3, 1670-83.
87. Yuan, D.; Xing, A.; Miao, P.; et al. Assembly of sub-crystals on the macroscale and construction of composite building units on the microscale for SAPO-34. Chem. Asian. J. 2018, 13, 3063-72.
88. Yan, N.; Xu, H.; Zhang, W.; et al. Probing locations of organic structure-directing agents (OSDAs) and host-guest interactions in CHA-type SAPO-34/44. Micropor. Mesopor. Mat. 2018, 264, 55-9.
89. Park, S.; Onozuka, H.; Tsutsuminai, S.; Kondo, J. N.; Yokoi, T. Insight into the crystallization mechanism of the CON-type zeolite. Micropor. Mesopor. Mat. 2020, 302, 110213.
90. Sada, Y.; Miyagi, S.; Iyoki, K.; et al. Biased Al distribution of high-silica FAU-type zeolite synthesized by fast manner at high temperature. Micropor. Mesopor. Mat. 2022, 344, 112196.
91. Kumar, M.; Choudhary, M. K.; Rimer, J. D. Transient modes of zeolite surface growth from 3D gel-like islands to 2D single layers. Nat. Commun. 2018, 9, 2129.
92. Choudhary, M. K.; Jain, R.; Rimer, J. D. In situ imaging of two-dimensional surface growth reveals the prevalence and role of defects in zeolite crystallization. Proc. Natl. Acad. Sci. U. S. A. 2020, 117, 28632-9.
93. Liu, F.; Wang, B.; Liu, X. Structure-directing roles of organic molecules in the formation of aluminosilicate and aluminophosphate molecular sieves revealed by 2D 1H DQ-SQ NMR spectroscopy. Chemistry 2021, 27, 1955-60.
94. Liu, X.; Wang, Q.; Wang, C.; Xu, J.; Deng, F. Hydrogen-bond induced crystallization of silicalite-1 zeolite as revealed by solid-state NMR spectroscopy. Acta. Phys. Chim. Sin. 2020, 36, 1905035.
95. Qin, W.; Agarwal, A.; Choudhary, M. K.; Palmer, J. C.; Rimer, J. D. Molecular modifiers suppress nonclassical pathways of zeolite crystallization. Chem. Mater. 2019, 31, 3228-38.
96. Zhang, J.; Shi, H.; Song, Y.; Xu, W.; Meng, X.; Li, J. High-efficiency synthesis of enhanced-titanium and anatase-free TS-1 zeolite by using a crystallization modifier. Inorg. Chem. Front. 2021, 8, 3077-84.
97. Zhang, J.; Bai, R.; Feng, Z.; Li, J. Amide-assisted synthesis of TS-1 zeolites with active Ti(OH2)2(OH)2(OSi)2 sites toward efficient oxidative desulfurization. Appl. Catal. B. Environ. 2024, 342, 123339.
98. Dai, H.; Claret, J.; Kunkes, E. L.; et al. Accelerating the crystallization of zeolite SSZ-13 with polyamines. Angew. Chem. Int. Ed. Engl. 2022, 61, e202117742.
99. Ren, T.; Wang, Y.; Wang, L.; Liang, L.; Kong, X.; Wang, H. Controllable synthesis of titanium silicon molecular zeolite nanosheet with short b-axis thickness and application in oxidative desulfurization. Nanomaterials 2024, 14, 953.
100. Zhang, F.; Liu, Y.; Sun, Q.; et al. Design and preparation of efficient hydroisomerization catalysts by the formation of stable SAPO-11 molecular sieve nanosheets with 10-20 nm thickness and partially blocked acidic sites. Chem. Commun. 2017, 53, 4942-5.
101. Shang, Z.; Chen, Y.; Zhang, L.; Zhu, X.; Wang, X.; Shi, C. Plate-like MFI crystal growth achieved by guanidine compounds. Inorg. Chem. Front. 2022, 9, 2097-103.
102. Xiong, S.; Sun, J.; Li, H.; Wang, W.; Wu, W. The synthesis of hierarchical ZSM-22 zeolite with only the PHMB template for hydroisomerization of n-hexadecane. Micropor. Mesopor. Mat. 2024, 365, 112895.
103. Yang, C.; Zhao, D.; Chu, W.; et al. N-methyl-2-pyrrolidone-promoted crystallization of MEL zeolite and its acceleration mechanism. J. Mater. Chem. A. 2020, 8, 26139-49.
104. Feng, G.; Cheng, P.; Yan, W.; et al. Accelerated crystallization of zeolites via hydroxyl free radicals. Science 2016, 351, 1188-91.
105. Cheng, P.; Feng, G.; Sun, C.; et al. An efficient synthetic route to accelerate zeolite synthesis via radicals. Inorg. Chem. Front. 2018, 5, 2106-10.
106. Wang, J.; Liu, P.; Boronat, M.; et al. Organic-free synthesis of zeolite Y with high Si/Al ratios: combined strategy of in situ hydroxyl radical assistance and post-synthesis treatment. Angew. Chem. Int. Ed. Engl. 2020, 59, 17225-8.
107. Zhang, Y.; Li, Y.; Zhao, L.; et al. Rapid synthesis of SAPO-18 molecular sieve and its MTO performance. Chem. Ind. Eng. Prog. 2018, 37, 1815-22.
108. Zhou, Y.; Shen, X.; Li, J. Crystallization and MTO performance of SAPO-34 zeolite under the influence of hydroxyl radicals. Inorg. Chem. Commun. 2019, 107, 107462.
109. Tan, J.; Liu, Z.; Bao, X.; et al. Crystallization and Si incorporation mechanisms of SAPO-34. Micropor. Mesopor. Mat. 2002, 53, 97-108.
110. Devos, J.; Robijns, S.; Van, G. C.; Khalil, I.; Dusselier, M. Interzeolite conversion and the role of aluminum: toward generic principles of acid site genesis and distributions in ZSM-5 and SSZ-13. Chem. Mater. 2021, 33, 2516-31.
111. Zhang, J.; Chu, Y.; Liu, X.; et al. Interzeolite transformation from FAU to CHA and MFI zeolites monitored by UV Raman spectroscopy. Chin. J. Catal. 2019, 40, 1854-9.
112. Zhang, J.; Chu, Y.; Deng, F.; Feng, Z.; Meng, X.; Xiao, F. Evolution of D6R units in the interzeolite transformation from FAU, MFI or *BEA into AEI: transfer or reassembly? Inorg. Chem. Front. 2020, 7, 2204-11.
113. Chen, S.; Guan, D.; Zhang, Y.; Wang, Z.; Jiang, N. Composition and kinetic study on template- and solvent-free synthesis of ZSM-5 using leached illite clay. Micropor. Mesopor. Mat. 2019, 285, 170-7.
114. Chen, X.; Wang, Y.; Wang, C.; et al. Synthesis of NaA zeolite via the mesoscale reorganization of submolten salt depolymerized kaolin: a mechanistic study. Chem. Eng. J. 2023, 454, 140243.
115. Wang, X.; Li, R.; Yu, C.; Liu, Y. Study on the reconstruction in the crystallization process of mordenite. Micropor. Mesopor. Mat. 2021, 311, 110665.
116. Li, T.; Krumeich, F.; van, B. J. A. Where does the zeolite ZSM-5 nucleation and growth start? The effect of aluminum. Cryst. Growth. Des. 2019, 19, 2548-51.
117. Zhang, Q.; Li, J.; Wang, X.; et al. Silanol-engineered nonclassical growth of zeolite nanosheets from oriented attachment of amorphous protozeolite nanoparticles. J. Am. Chem. Soc. 2023, 145, 21231-41.
118. Zhou, Y.; Shi, H.; Wang, B.; Chen, G.; Yi, J.; Li, J. The synthesis of SAPO-34 zeolite for an improved MTO performance: tuning the particle size and an insight into the formation mechanism. Inorg. Chem. Front. 2021, 8, 2315-22.
119. Zhou, Y.; Zhang, J.; Ma, W.; et al. Small pore SAPO-14-based zeolites with improved propylene selectivity in the methanol to olefins process. Inorg. Chem. Front. 2022, 9, 1752-60.
120. Zhang, X.; Yang, M.; Wang, L.; et al. Recognizing the minimum structural units driving the crystallization of SAPO-34 in a top-down process. Chemistry 2023, 29, e202203886.
121. Zhang, X.; Yang, M.; Wang, Y.; et al. Insights on the seed selection criteria of SAPO-34 synthesis: structural units and their chemical microenvironment. Inorg. Chem. Front. 2023, 10, 3874-83.
122. Zhang, C.; Wu, Q.; Lei, C.; et al. An efficient, rapid, and non-centrifugation synthesis of nanosized zeolites by accelerating the nucleation rate. J. Mater. Chem. A. 2018, 6, 21156-61.
123. Jonscher, C.; Seifert, M.; Kretzschmar, N.; et al. Origin of morphology change and effect of crystallization time and Si/Al ratio during synthesis of zeolite ZSM-5. ChemCatChem 2022, 14, e202101248.
124. Chen, C. T.; Iyoki, K.; Hu, P.; et al. Reaction kinetics regulated formation of short-range order in an amorphous matrix during zeolite crystallization. J. Am. Chem. Soc. 2021, 143, 10986-97.
125. Bai, R.; Song, Y.; Lätsch, L.; et al. Switching between classical/nonclassical crystallization pathways of TS-1 zeolite: implication on titanium distribution and catalysis. Chem. Sci. 2022, 13, 10868-77.
126. Houlleberghs, M.; Breynaert, E.; Asselman, K.; et al. Evolution of the crystal growth mechanism of zeolite W (MER) with temperature. Micropor. Mesopor. Mat. 2019, 274, 379-84.
127. Wei, P.; Liu, W.; Li, J.; et al. Synthesis and crystallization mechanism of EUO zeolite. Micropor. Mesopor. Mat. 2022, 337, 111911.
128. Bok, T. O.; Andriako, E. P.; Knyazeva, E. E.; Ivanova, I. I. Influence of synthesis conditions on the crystallization mechanism and properties of BEA zeolite. Pet. Chem. 2019, 59, 354-61.
129. Shen, Y.; Zhang, S.; Qin, Z.; et al. Effect of mixing order of Si and Al sources on the inner architecture and catalytic performance of ZSM-5 zeolites. ACS. Catal. 2024, 14, 3766-77.
130. Chen, G.; Li, J.; Wang, S.; et al. Construction of single-crystalline hierarchical ZSM-5 with Open Nanoarchitectures Via Anisotropic-Kinetics Transformation For The Methanol-To-Hydrocarbons Reaction. Angew. Chem. Int. Ed. Engl. 2022, 61, e202200677.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data





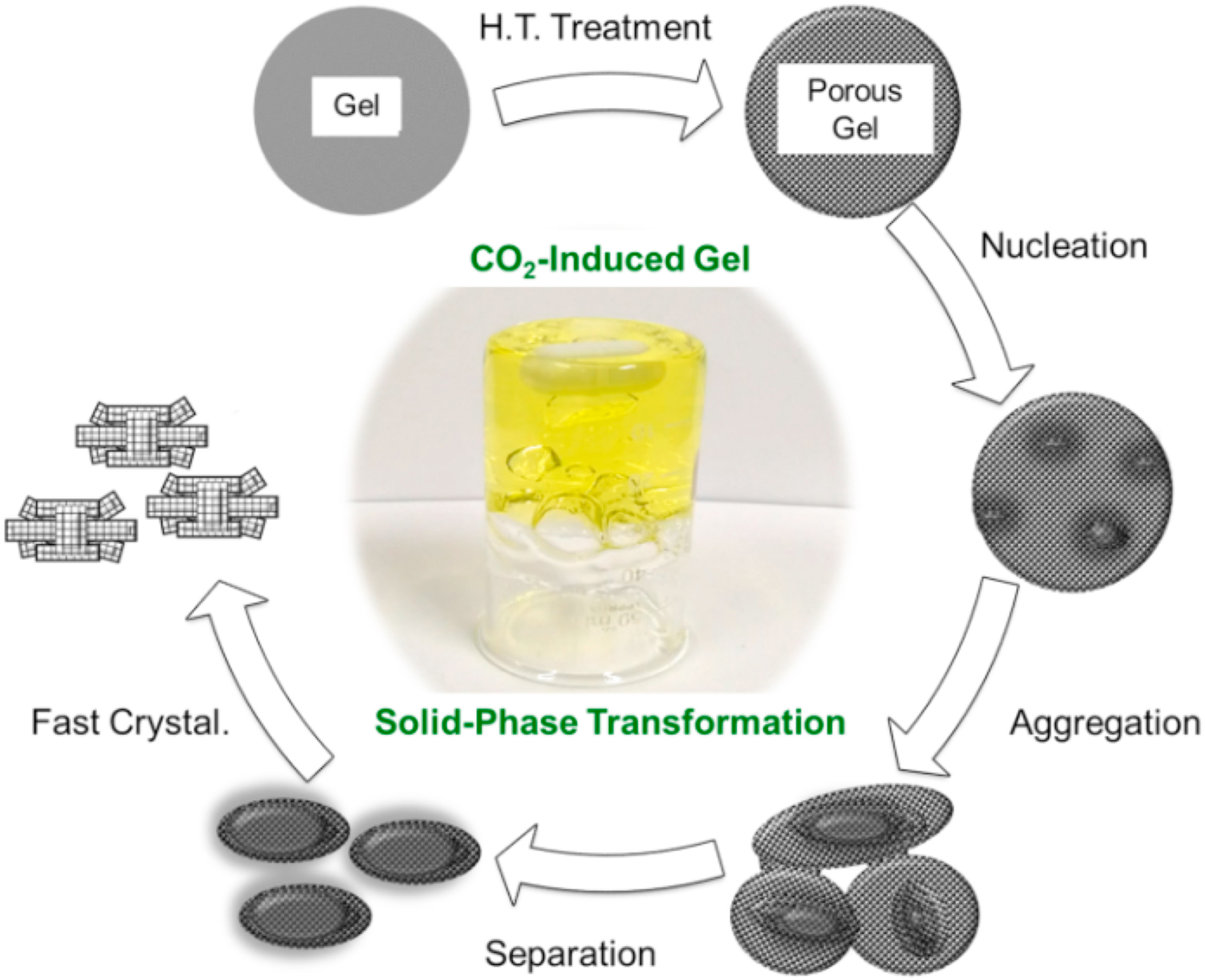
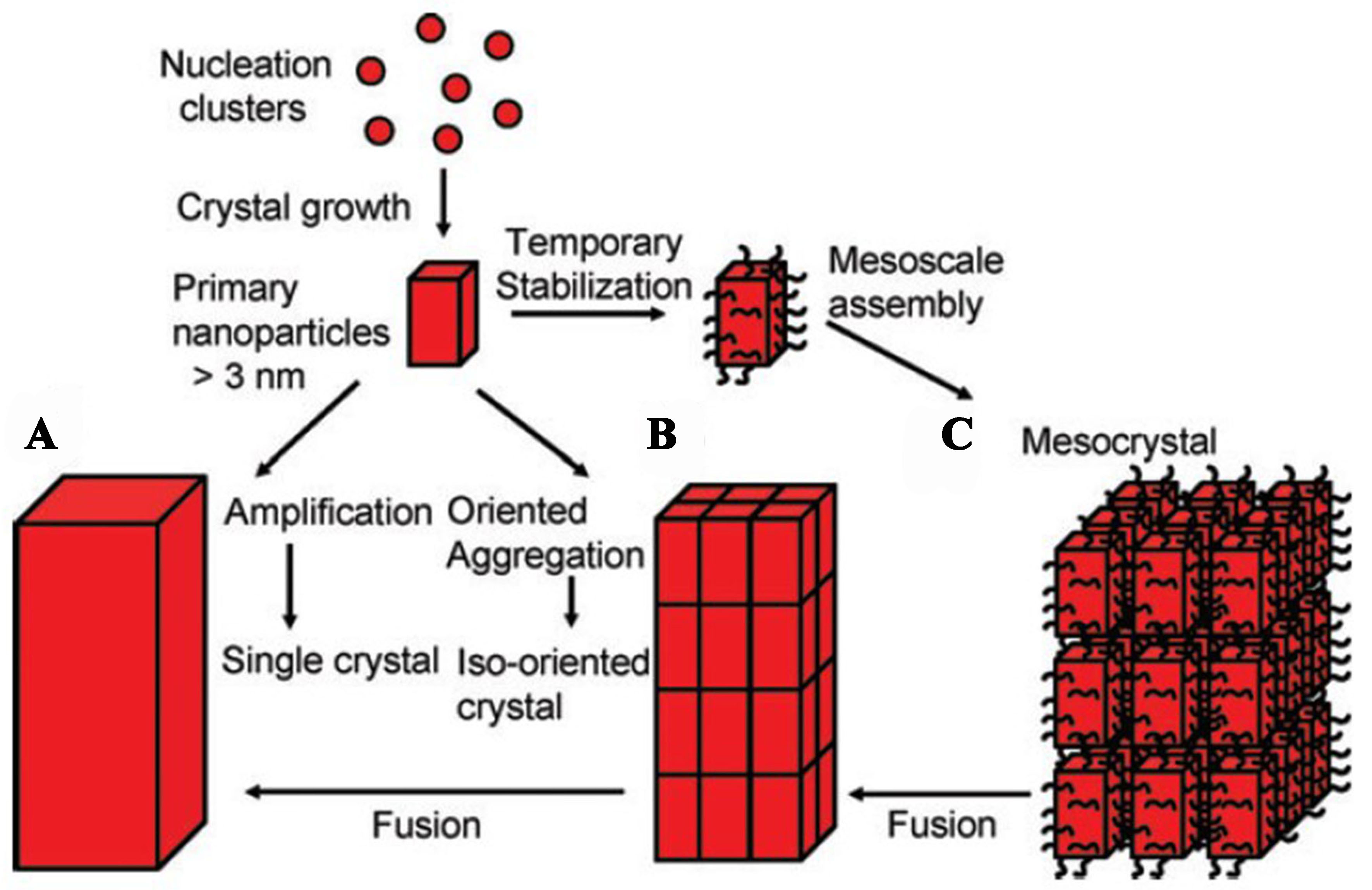
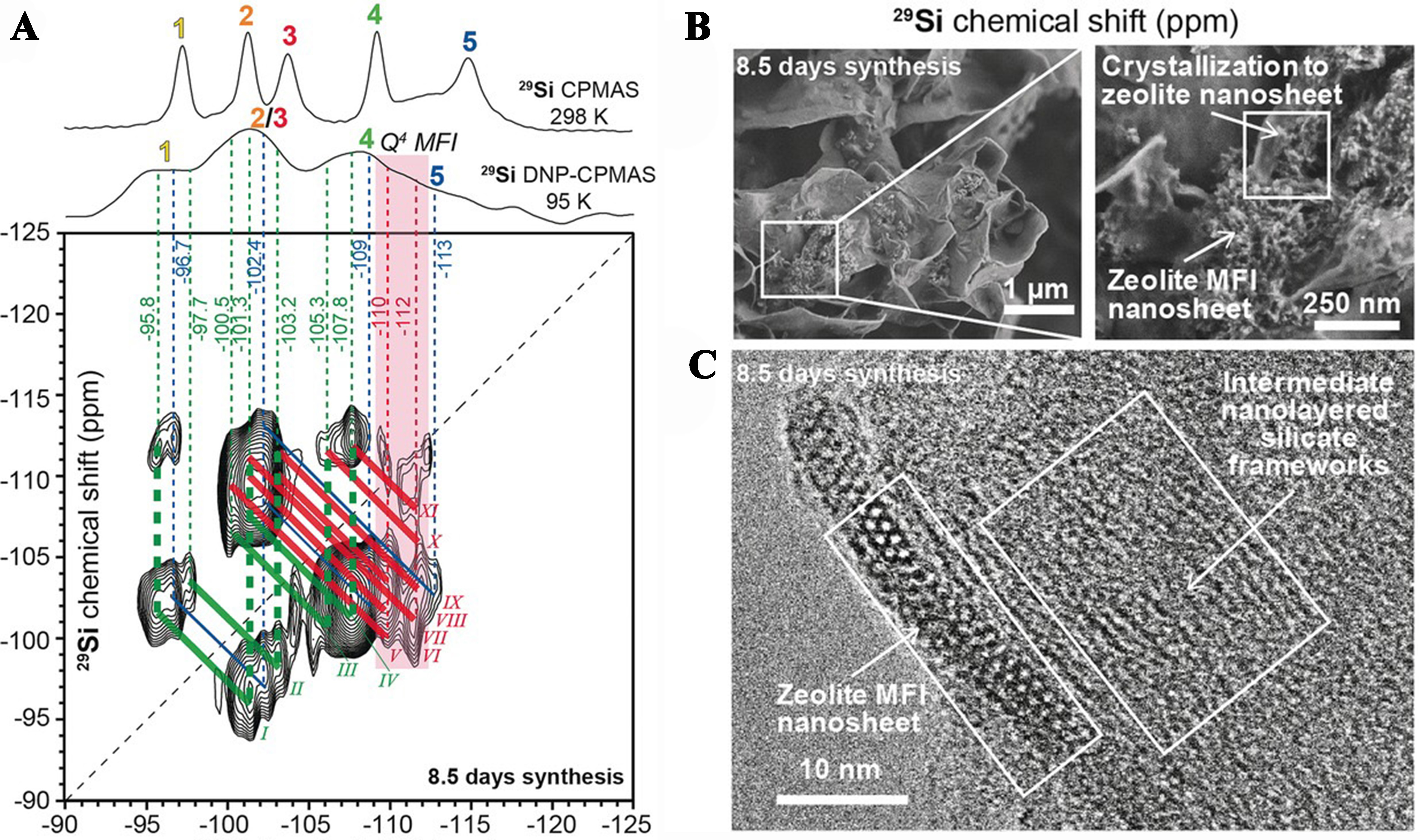
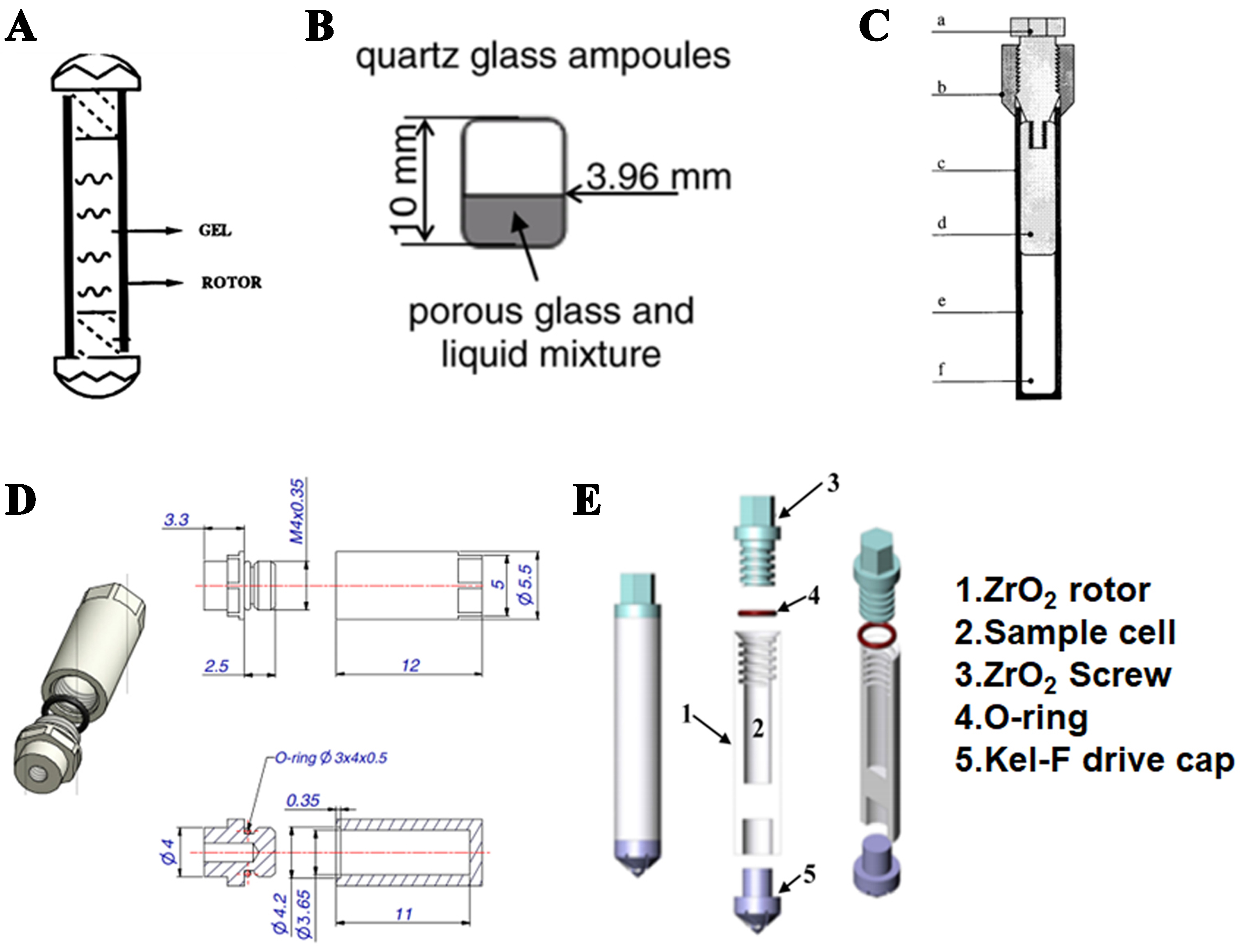
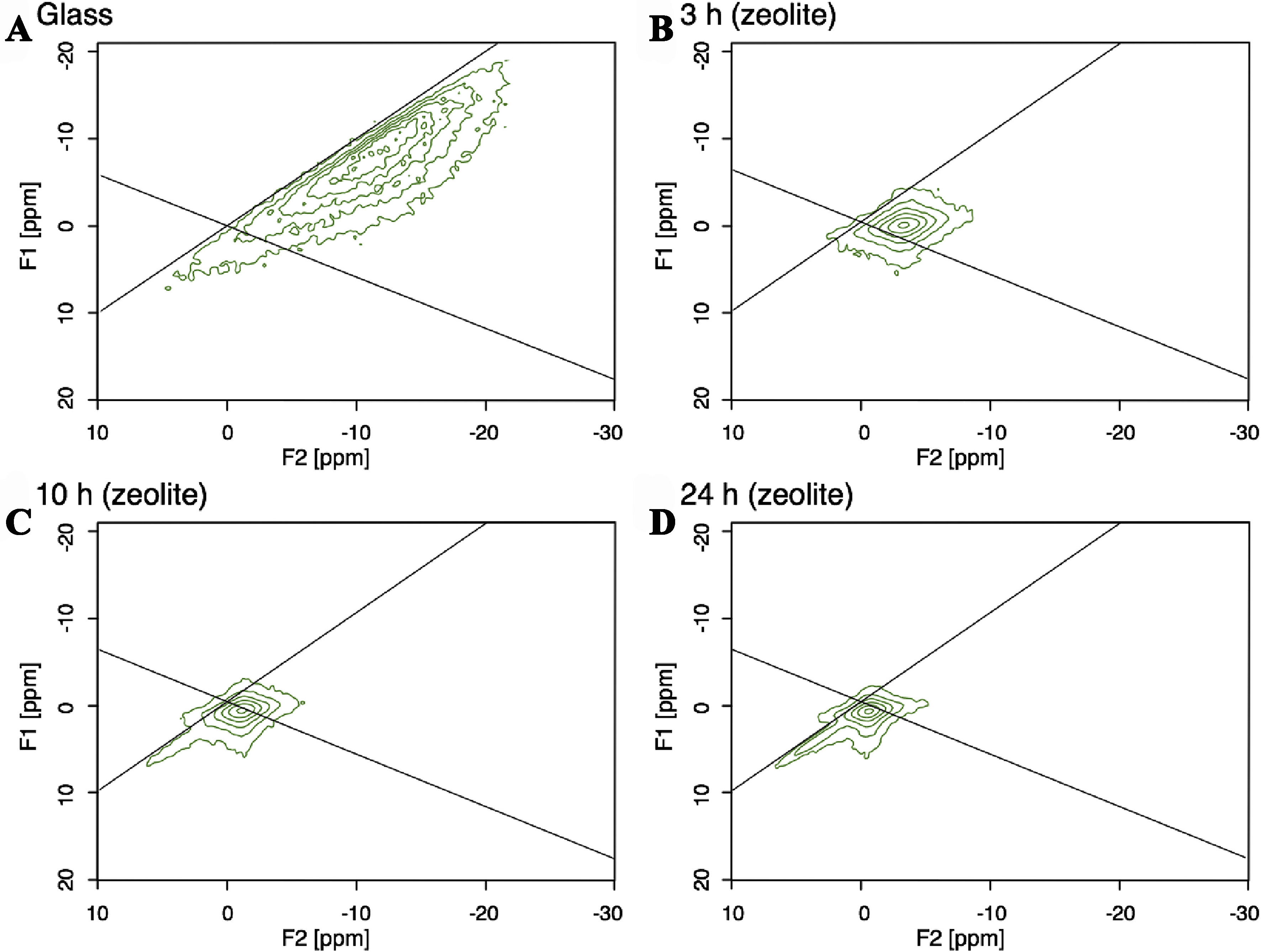
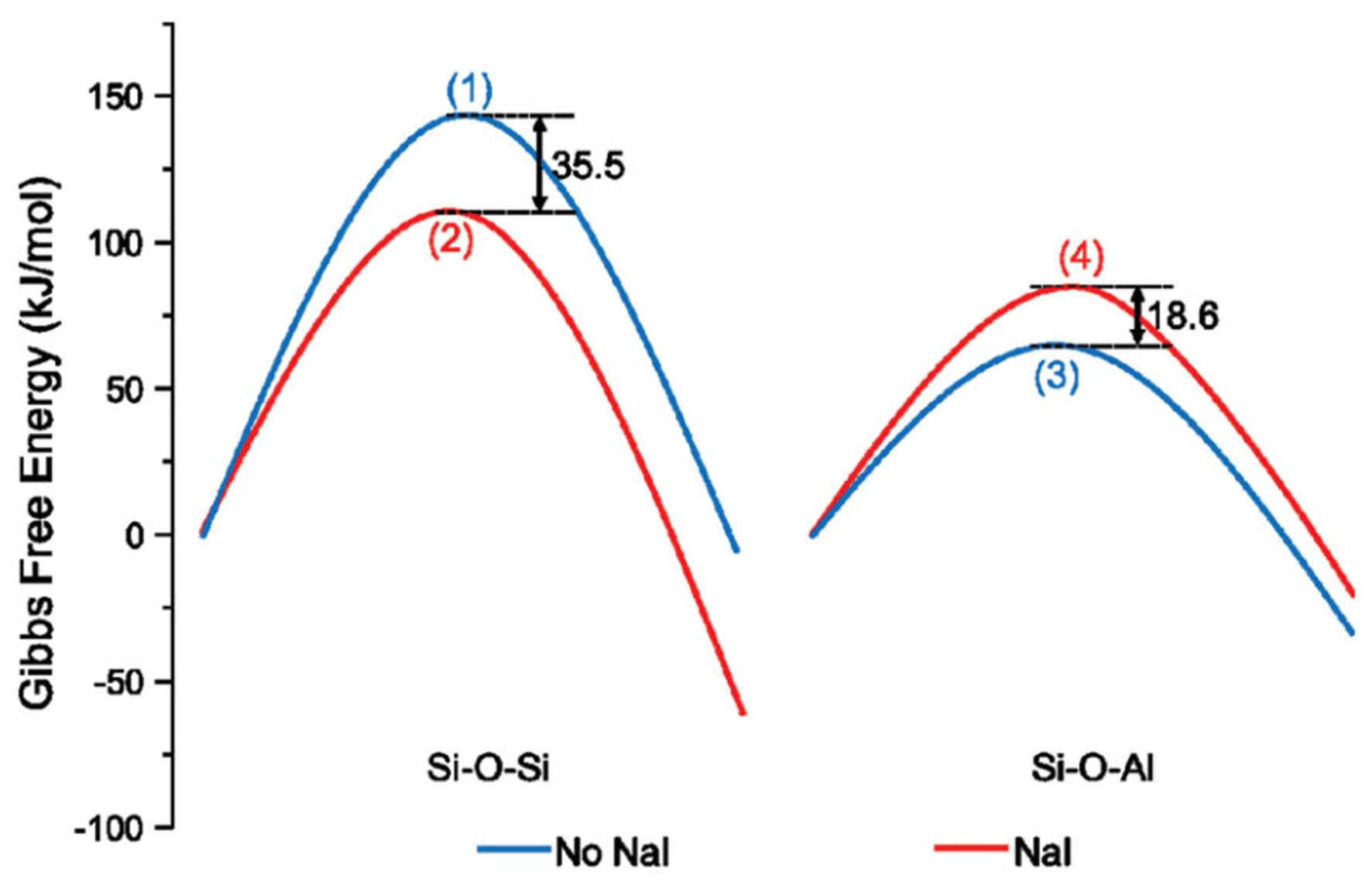
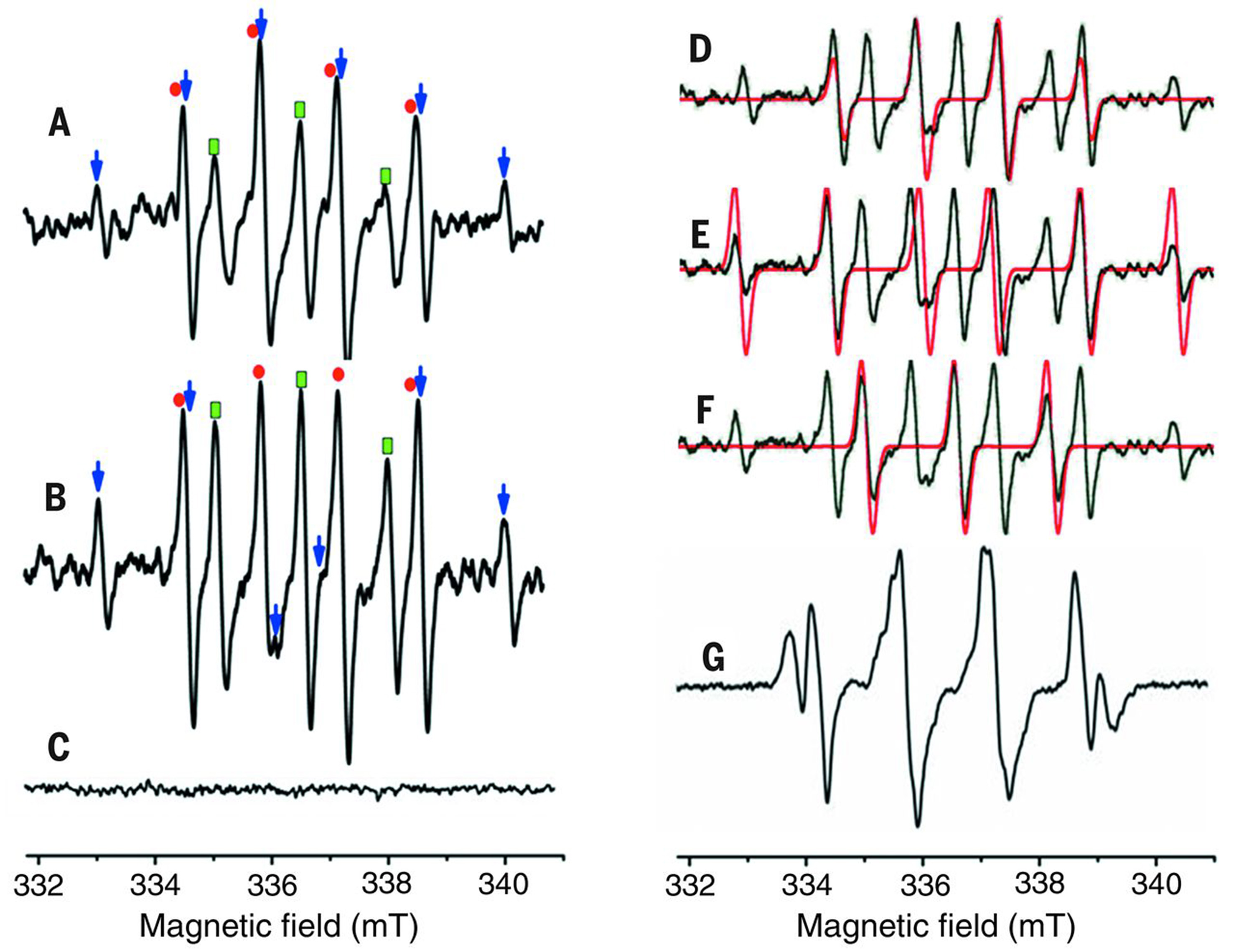
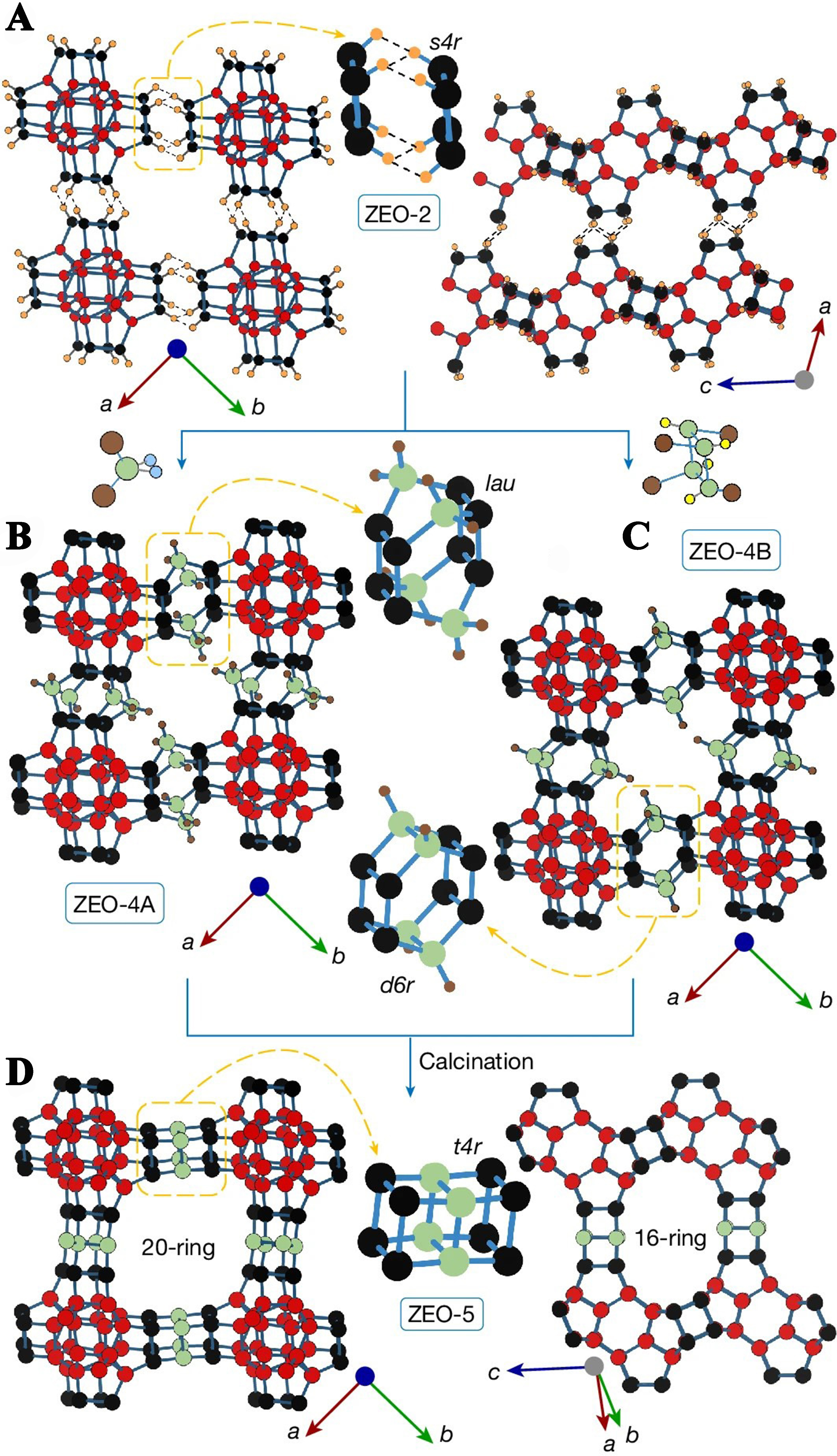
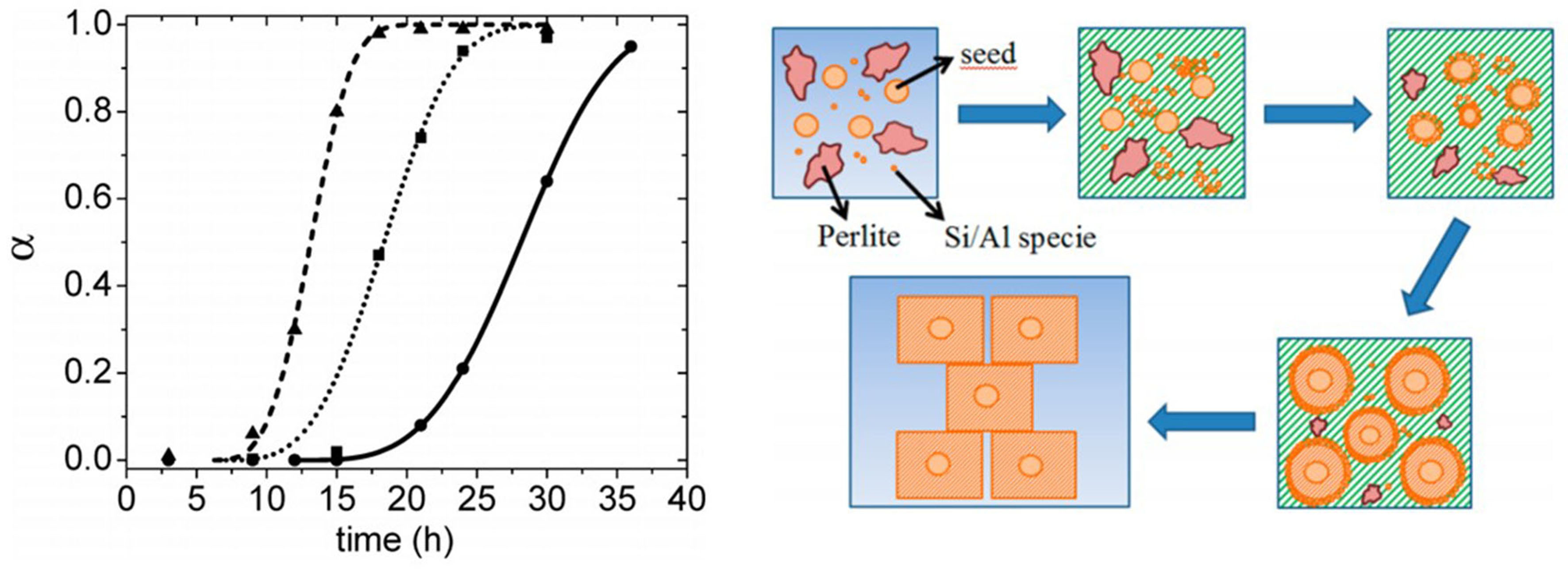
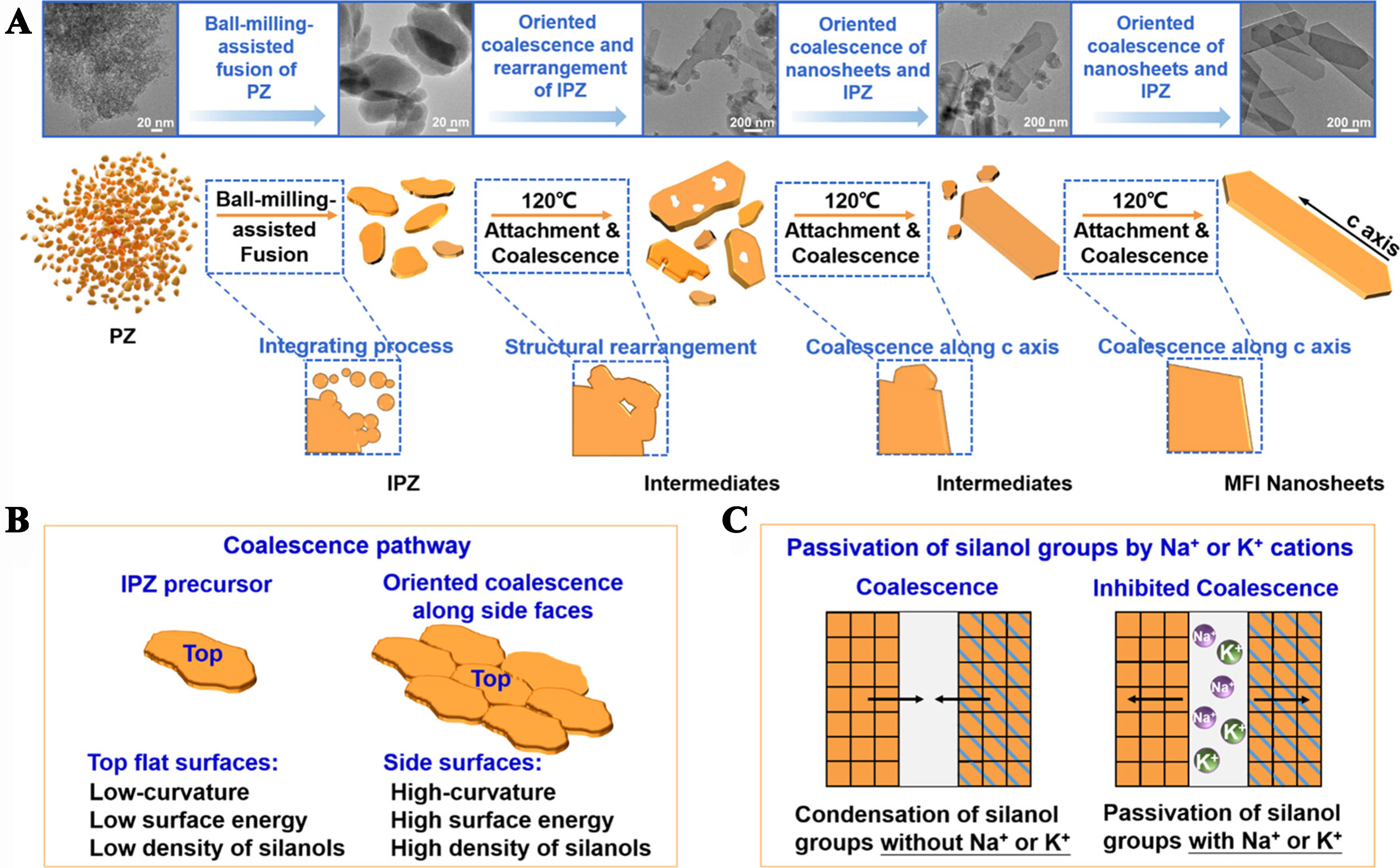
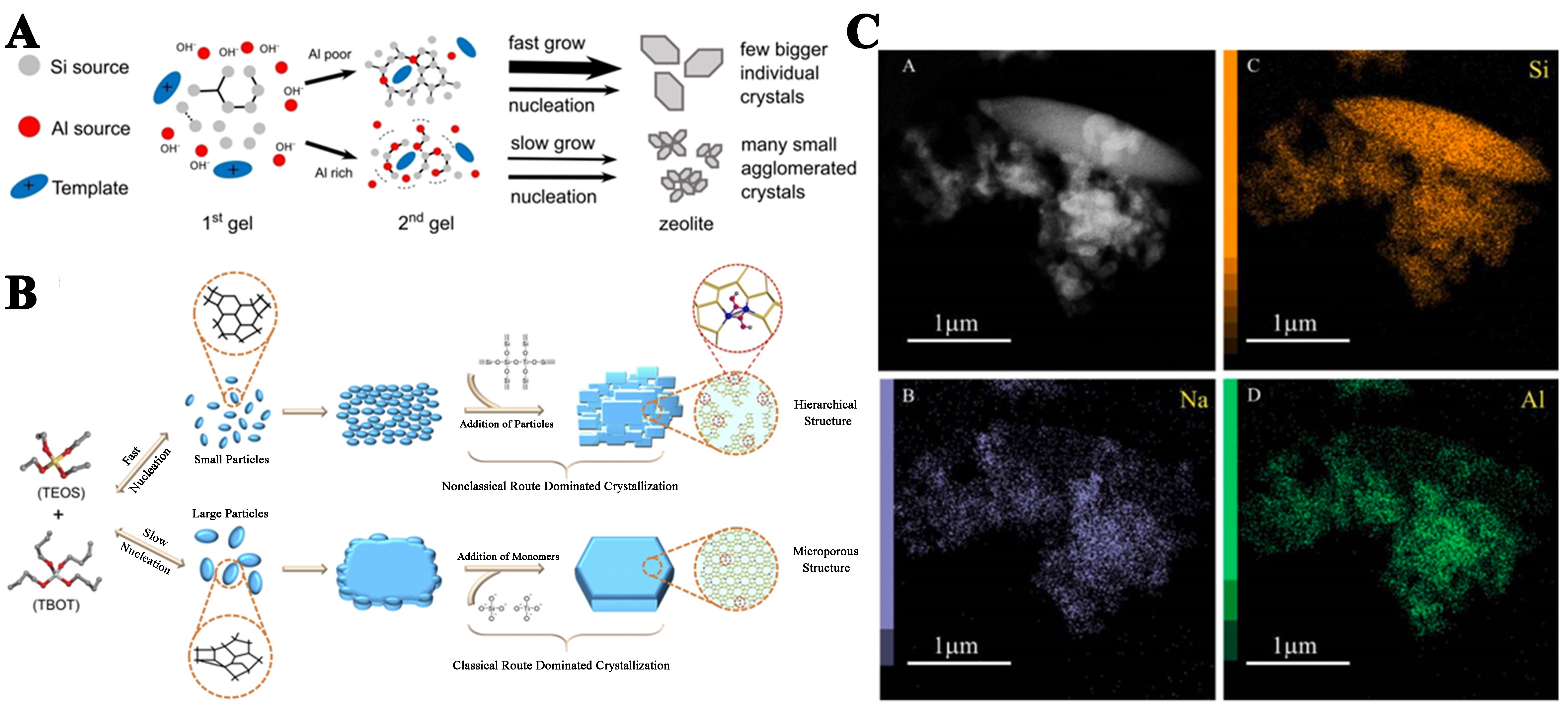













Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].