Enhanced hydrogen evolution reaction performance of manganese-doped vanadium disulfide nanosheet across full pH spectrum and in simulated seawater
Abstract
Hydrogen production technologies that can efficiently operate across the entire pH range hold great promise for diverse application scenarios, especially in seawater electrolysis. In this work, we report a facile colloidal-chemistry synthesis of manganese-doped vanadium disulfide (Mn-VS2) monolayer nanosheets with superior hydrogen evolution reaction performance. The incorporation of manganese lowers the formation energy of sulfur vacancies and modifies electronic states, thereby creating additional catalytic sites and enhancing reaction kinetics. Benefiting from these features, Mn-VS2 achieves an overpotential of 40 mV at -10 mA·cm-2 in simulated seawater and maintains excellent operational stability for over 100 h. These findings underscore the potential of Mn-VS2 as a robust electrocatalyst for direct seawater electrolysis, providing a viable avenue for exploring sustainable hydrogen production in practical marine environments.
Keywords
INTRODUCTION
The quest for sustainable and clean energy solutions has intensified global focus on hydrogen production via water electrolysis, where hydrogen evolution reaction (HER) plays a pivotal role[1-5]. This fundamental water-splitting reaction necessitates catalysts that are both efficient and robust[6,7], capable of operating in a range of aqueous environments[8]. There is a particular emphasis on developing catalysts that can function effectively across the entire pH spectrum and in simulated seawater conditions[9-11], to address the specific challenges these settings present. Despite significant advances in the field, crafting catalysts that are cost-effective, highly active, and durable remains a substantial challenge, especially for applications in such varied aqueous conditions[12-14].
As research intensifies in the development of sustainable catalysts for HER, the exceptional catalytic efficiency of platinum (Pt) group metals serves as a critical reference point[15-17]. However, due to the high cost and limited availability of these noble metals, there is a pressing need to explore more accessible alternatives[18,19]. This search has steered scientific interest towards non-noble metal catalysts, particularly transition metal dichalcogenides (TMDs), known for their promising electrocatalytic properties[20-22]. Among TMDs, materials such as MoS2, WS2, and vanadium disulfide (VS2) have garnered significant attention due to their outstanding catalytic efficiencies and structural flexibility. The cost-effectiveness and abundance of TMDs make them an attractive choice for sustainable energy applications, as their unique two-dimensional structures provide numerous active sites that enhance catalytic performance. Recent advancements, such as the development of 2D/2D NiAl-based layered double hydroxide (LDH)/CoNi-based metal-organic framework (MOF) nanohybrid catalysts for elevated symmetric supercapacitor performance and H2O2 production under simulated solar light, further underscore the potential of layered 2D materials in catalysis[23]. These innovative approaches demonstrate the continuous evolution in electrocatalyst design, where hybrid structures and material engineering play pivotal roles in boosting catalytic performance, positioning TMDs such as VS2 as highly promising candidates for sustainable energy solutions.
VS2 has emerged as a particularly promising candidate within TMDs due to its unique structural and electronic features[24-28]. Its graphene-like layered structure not only facilitates high electronic conductivity, as suggested by its metallic nature in theoretical studies[29,30], but also hosts numerous edge sites that are active for catalysis[31,32], making it a compelling alternative to traditional noble metal catalysts in electrocatalytic applications. Recent studies have demonstrated that VS2 can significantly enhance HER when combined with other materials such as MoS2 or when defect-rich structures are used, improving both efficiency and multifunctionality. Additionally, VS2 self-assembled into layered microstructures has shown promise for water and urea decomposition reactions, further proving its versatility in electrocatalysis[33,34]. Particularly, the monolayer form of VS2 exemplifies its potential by offering an abundance of active sites and exceptional in-plane conductivity, enhancing its suitability for HER[35,36]. However, the potential of VS2 has not been fully realized due to inherent challenges related to its vertical conductivity in multi-layer or bulk forms, which limits the overall catalytic efficiency. Addressing these challenges involves structural optimization to enhance the exposure of catalytically active sites and improve electrical conductivity, both of which are critical for efficient catalytic reactions[37,38]. While several TMD materials have demonstrated catalytic promise, VS2 is underexplored compared to MoS2 and WS2, particularly in the context of seawater electrolysis. This research aims to address that gap.
To address the intrinsic limitations of VS2 in HER applications, researchers have turned to doping as a strategy to enhance its electrochemical properties[39]. Doping introduces foreign atoms into the crystal lattice of VS2, which can significantly alter its electronic structure and catalytic activity[40,41]. Mn is specifically chosen for doping due to its ability to introduce new electronic states within the band structure of VS2. These new states can facilitate improved electron transfer processes, crucial for the dynamics of HER. Additionally, Mn incorporation into VS2 leads to structural distortions that are beneficial for catalysis. These distortions can create more favorable sites for hydrogen adsorption and desorption, which are essential steps in the catalytic cycle of HER[42]. By adjusting the electronic properties and geometric configuration of VS2, Mn doping enhances both the reactivity and stability of the catalyst, thereby addressing some of the key challenges faced by the pristine material in electrocatalytic applications[43-45]. Unlike traditional doping strategies used for MoS2 and WS2, Mn doping in VS2 significantly modifies the sulfur vacancy formation energy and enhances its stability in seawater conditions, which sets this study apart from previous works.
In this study, manganese-doped vanadium disulfide (Mn-VS2) nanosheets were synthesized via the colloidal chemistry method to rigorously evaluate their electrocatalytic HER activities across a comprehensive pH spectrum and in simulated seawater conditions. The synthesized catalyst demonstrated notably low overpotentials in varied electrolytes, confirming its superior performance. The integration of Mn substantially enhanced the HER performance of VS2, as evidenced by the remarkably low overpotentials required to achieve a current density of -10 mA·cm-2 in 1 M KOH, 1 M phosphate buffered saline (PBS), and 0.5 M H2SO4 solutions, which were 41, 55, and 37 mV, respectively. Particularly in simulated seawater,
EXPERIMENTAL
The Mn-VS2 catalyst was synthesized using a colloid chemistry method (experimental details are provided in the Supplementary Materials). In summary, sulfur powder was dissolved in oleylamine and heated under stirring. At the same time, VO(acac)2 and MnCl2 were dissolved in oleylamine, ensuring thorough mixing. The mixture was then heated under a nitrogen atmosphere; the sulfur solution was injected into the preheated VO(acac)2 and MnCl2 solutions, and the temperature was raised to 330 °C and maintained for
RESULTS AND DISCUSSION
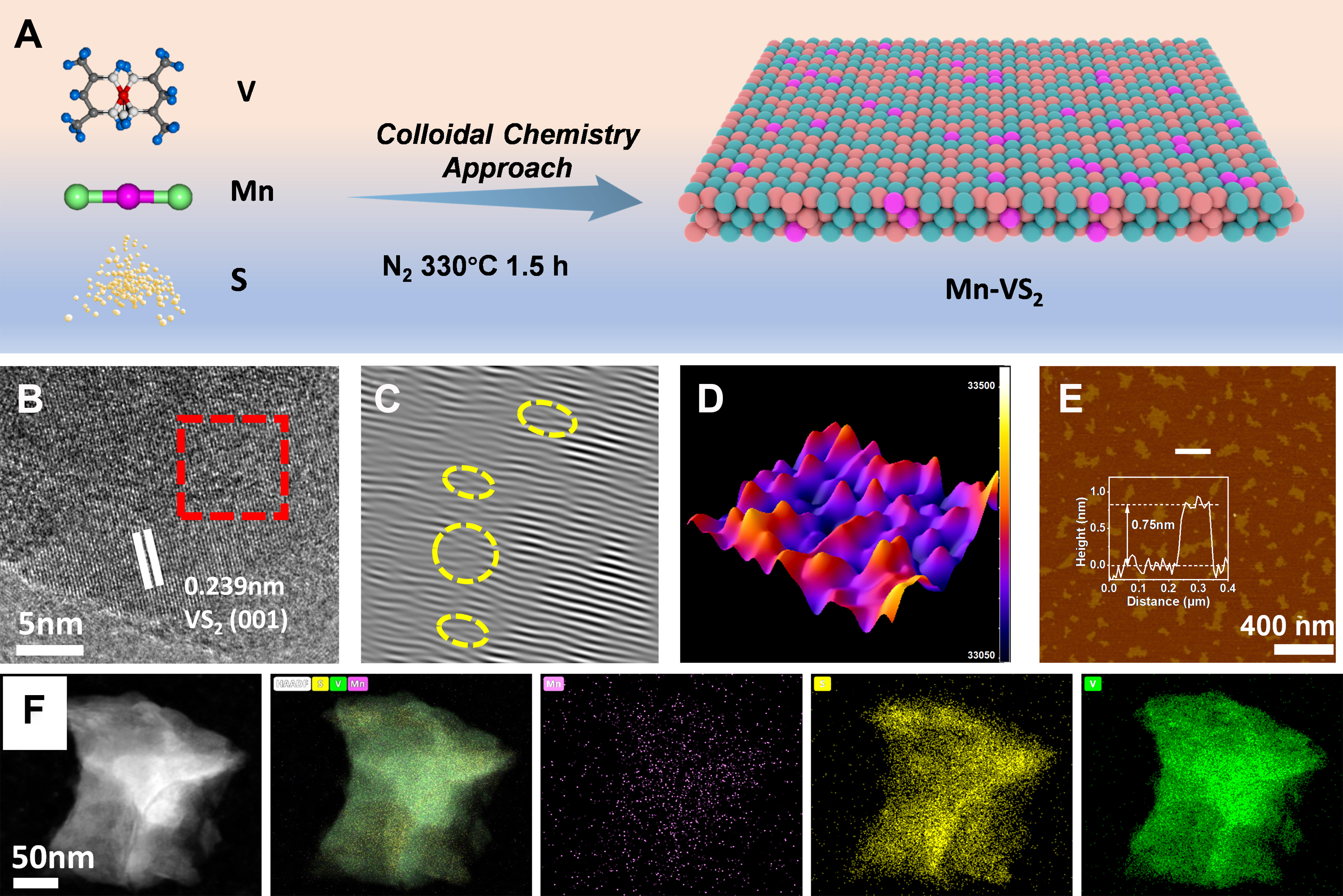
Mn-VS2 nanosheets were synthesized using a colloidal chemistry approach, as depicted in Figure 1A. Initially, a precursor sol containing V, S, and Mn was prepared. This sol gradually transformed into a gel with a defined shape and structure. During the synthesis phase, to prevent oxidation, the material was heated to 330 °C under a nitrogen atmosphere and maintained at this temperature for 1.5 h to ensure complete conversion of the precursor into Mn-VS2[24,27,46].
Figure 1. (A) Synthetic process route for Mn-VS2; (B) HRTEM image of Mn-VS2; (C) IFFT analysis of Mn-VS2 derived from (B); (D) 3D AOGF mappings from (B); (E) AFM image of Mn-VS2; (F) EDS mapping spectra of various elements in Mn-VS2. HRTEM: High-resolution transmission electron microscopy; IFFT: inverse fast fourier transform; AOGF: Atom-Overlapping Gaussian Function; AFM: atomic force microscopy; EDS: energy dispersive spectroscopy.
As shown in Supplementary Figure 1, the as-synthesized Mn-VS2 exhibits a sheet-like morphology, evident from the scanning electron microscopy (SEM) images taken at different magnifications. Large-scale transmission electron microscopy (TEM) images further confirm the nanosheet structure of Mn-VS2, complementing the SEM observations [Supplementary Figure 2]. High-resolution TEM (HRTEM) images of Mn-VS2 are shown in Figure 1B, displaying clear lattice fringes. The measured lattice spacing of 0.239 nm corresponds to the (001) planes of VS2. Figure 1C, which is a magnified view of the area marked in red in Figure 1B, includes a fast fourier transform (FFT) analysis commonly utilized in HRTEM studies to highlight defects within the crystal structure. In these images, defects are circled in yellow, revealing the microstructural impacts of Mn doping, which are markedly distinct from the HRTEM images of undoped VS2 [Supplementary Figure 3]. Such microstructural doping likely contributes to the enhanced electrocatalytic HER performance of Mn-VS2. Additionally, Figure 1D presents the 3D Atom-Overlapping Gaussian Function (AOGF) mappings for Mn-VS2, employing a Gaussian-function fitting to visualize the spatial distribution and intensity of atomic sites. Significant color variations in the AOGF plots highlight the presence of defects, as changes in atomic density and arrangement alter the Gaussian overlap, thereby indicating the presence of structural defects. Conversely, the AOGF plot for undoped VS2, as shown in Supplementary Figure 4, exhibits a uniform color distribution, suggesting more intact structural integrity. To further investigate the structure of Mn-VS2, atomic force microscopy (AFM) characterization was conducted, as depicted in Figure 1E. AFM is a sophisticated technique utilized for assessing surface roughness, morphology, and thickness of materials. In the image, varying color blocks represent different heights, effectively illustrating the surface topography and thickness variations of the sample. The inset, an AFM-derived height profile, reveals that the nanosheets possess a thickness of approximately 0.75 nm, indicating that the Mn-VS2 exists in a monolayer structure. Monolayer structures are known for their high specific surface area, unique electronic properties, and high surface activity, all contributing to their exceptional catalytic performance in reactions. It is worth mentioning that the monolayer configuration of Mn-VS2 offers a high density of exposed active sites, which maximizes the material’s surface area and facilitates efficient interaction with reactants. Additionally, the exceptional in-plane conductivity of the monolayer structure ensures rapid electron transport, a key factor for enhancing the efficiency of reactions such as hydrogen evolution. The reduced thickness of the monolayer minimizes electron scattering, further improving the material’s conductivity. However, in multi-layer or bulk configurations, the limited vertical conductivity between stacked layers can restrict electron flow, thereby reducing the overall catalytic efficiency. Figure 1F shows Energy-dispersive X-ray spectroscopy (EDS) mapping, which provides a clear visualization of the homogeneous distribution of Mn, V, and S elements within the composite material. This mapping is crucial for a detailed analysis of the elemental composition of the material, further supporting the understanding of its enhanced catalytic properties.
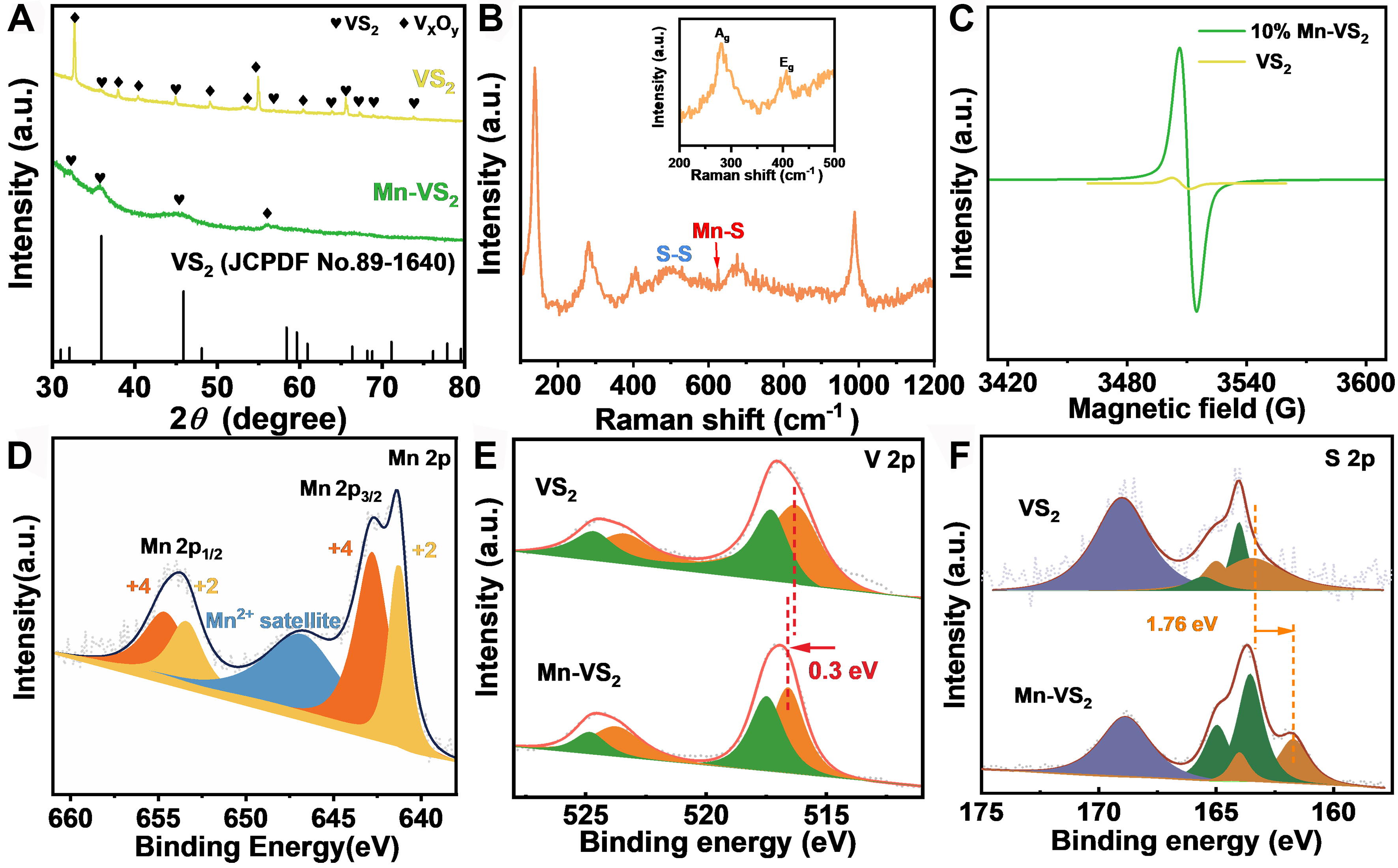
As shown in Figure 2A, the X-ray diffraction (XRD) patterns of both Mn-VS2 and pristine VS2 correspond to the hexagonal phase of VS2 (JCPDF No. 89-1640), with primary reflections at (100) 32.10°, (011) 35.68°, and (012) 45.23°. Pristine VS2 exhibits sharp and intense diffraction peaks, indicating a highly ordered structure and excellent crystallinity. In contrast, the Mn-VS2 sample displays noticeably broadened peaks and slight shifts in peak positions, which reflect a reduction in grain size and an increase in lattice strain. These changes suggest that Mn doping introduces lattice distortions and local defects, thereby decreasing the overall crystallinity. Additionally, the incorporation of Mn not only alters the lattice constants of VS2 but also induces local structural distortions and stress, increasing the density of defect sites and the active surface area. This structural modification is beneficial for enhancing the electrocatalytic hydrogen evolution performance. Figure 2B presents the Raman spectroscopy analysis of Mn-VS2. The principal Raman peaks identified in the spectrum can be attributed to the characteristic vibrational modes of VS2. The marked
Figure 2. (A) XRD patterns of VS2 and Mn-VS2; (B) Raman spectrum of Mn-VS2; (C) EPR spectra of VS2 and Mn-VS2; XPS spectrum of (D) Mn 2p in Mn-VS2 (E) V 2p, (F) S 2p in VS2 and Mn-VS2. XRD: X-ray diffraction; EPR: electron paramagnetic resonance; XPS: X-ray photoelectron spectroscopy.
To realize the commercial potential of HER catalysts, the development of highly active and enduringly stable catalysts has become imperative. Achieving this is challenging, particularly under alkaline or neutral conditions where the paucity of protons leads to slower water dissociation steps, thus impeding the rate of hydrogen evolution. Despite the expansion of HER research to encompass the entire pH spectrum in recent years, developing superior electrocatalysts that perform efficiently across all pH conditions remains a formidable task.
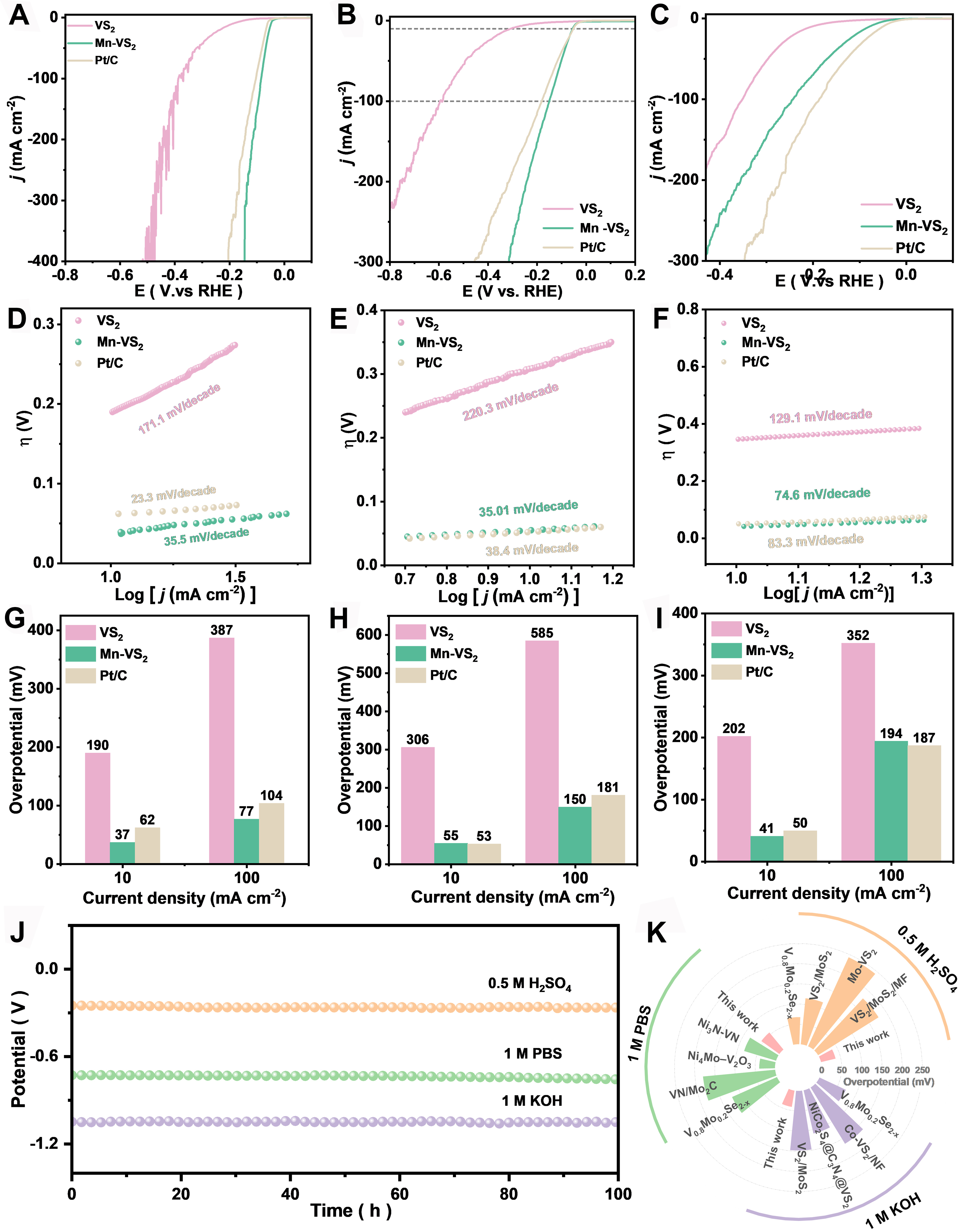
Electrochemical tests conducted on VS2, Mn-VS2 and Pt/C with varying doping levels, using a three-electrode setup across different pH environments, have undergone 95% iR compensation. The polarization curves derived [Figure 3A-C] indicate that Mn-VS2 exhibits the lowest overpotentials required to achieve a current density of -10 mA·cm-2 in 1 M KOH, 1 M PBS, and 0.5 M H2SO4 solutions at 41, 55, and 37 mV, respectively, significantly outperforming pure VS2 (202 mV in 1 M KOH, 306 mV in 1 M PBS, and 190 mV in 0.5 M H2SO4). The Tafel slopes, derived from the linear regions of the polarization curves [Figure 3D-F], reveal that Mn-VS2 exhibits significantly improved HER kinetics compared to pristine VS2 in all tested electrolytes: specifically, Mn-VS2 shows slopes of 74.69, 35.01, and 35.5 mV·dec-1 in 1 M KOH, 1 M PBS, and 0.5 M H2SO4, respectively, whereas VS2 displays relatively higher slopes of 129, 220.31, and
Figure 3. HER performance of VS2 and Mn-VS2 with varying doping levels (iR compensation at 95%). (A-C) HER polarization curves; (D-F) Tafel slopes; (G-I) Overpotentials at current densities of -10 mA·cm-2 and -100 mA·cm-2; (J) Stability at a current density of -10 mA·cm-2. Measurements were conducted in 0.5 M H2SO4 (A, D, G), 1 M PBS (B, E, H), and 1 M KOH (C, F, I) aqueous solutions; (K) Comparison of the overpotential (current density of -10 mA·cm-2) of Mn-VS2 with other recently reported vanadium-based electrocatalysts in 0.5 M H2SO4, 1 M PBS, and 1 M KOH. HER: Hydrogen evolution reaction.
Electrochemical assessments were conducted in an alkaline environment, where the electrochemical Cdl was derived from CV at different scan rates, as illustrated in Supplementary Figure 14. Mn-VS2 exhibited a notably higher Cdl compared to undoped VS2, indicating a larger electrochemically active surface area. This enhancement in active surface area is instrumental for improving the kinetics of HER. Further electrochemical impedance spectroscopy (EIS), as shown in Supplementary Figure 15, demonstrated that Mn-VS2 exhibits a significantly reduced charge transfer resistance (Rct) compared to pristine VS2. The lower Rct of Mn-VS2 highlights the critical role of manganese doping in enhancing charge transport efficiency. Doping introduces additional electronic states into the band structure of VS2, facilitating faster electron transfer and reducing the energy barriers for charge transfer at the electrode-electrolyte interface. This reduction in Rct is essential for minimizing voltage losses at high current densities, thereby accelerating the HER process[56,57]. Moreover, the wetting properties of Mn-VS2 were evaluated through contact angle measurements, as depicted in Supplementary Figure 16. Mn-VS2 exhibited improved wettability with a lower contact angle relative to VS2. This enhancement in hydrophilicity can be attributed to the lattice defects induced by Mn doping. Such defects often create unsaturated chemical bonds or regions of increased chemical activity, which can attract and retain water molecules, thereby augmenting the hydrophilicity of the material. The increased wettability of Mn-VS2, likely contributing to its superior electrocatalytic performance in HER, underscores the importance of surface properties in catalytic efficiency.
Given the global scarcity of freshwater resources, harnessing the abundant seawater resources on Earth is crucial for advancing the commercialization of water electrolysis technologies and meeting the escalating energy demands. However, the complex composition of natural seawater poses additional challenges to direct seawater electrolysis. For instance, chloride ions can compete with the oxygen evolution reaction (OER) at the anode, thereby reducing the overall efficiency of seawater electrolysis. Furthermore, catalyst degradation due to chloride-induced corrosion and poisoning by insoluble precipitates (such as dust, colloids, and bacteria) can diminish both the catalytic activity and stability of the catalyst.
In this study, the electrocatalytic water-splitting performance of Mn-VS2 was evaluated in an alkaline simulated seawater environment (1 M KOH + 0.5 M NaCl). For comparative analysis, HER activities of undoped VS2 and Mn-VS2 with different doping ratios were also tested under the same conditions. Mn-VS2 exhibited superior electrochemical performance, as demonstrated by its low overpotential of 40 mV at a current density of -10 mA·cm-2 [Supplementary Figure 17A], which is comparable to its catalytic performance in pure alkaline solutions. Additionally, as demonstrated in Supplementary Figure 17B, the lower Tafel slope of Mn-VS2, recorded at 39.34 mV·dec-1, further confirms its accelerated HER kinetics in simulated seawater. This value is comparable to other advanced electrocatalysts designed for HER in simulated seawater, such as Pt-based catalysts (41 mV·dec-1)[46] and Ru-doped Ni0.85Se nanosheets
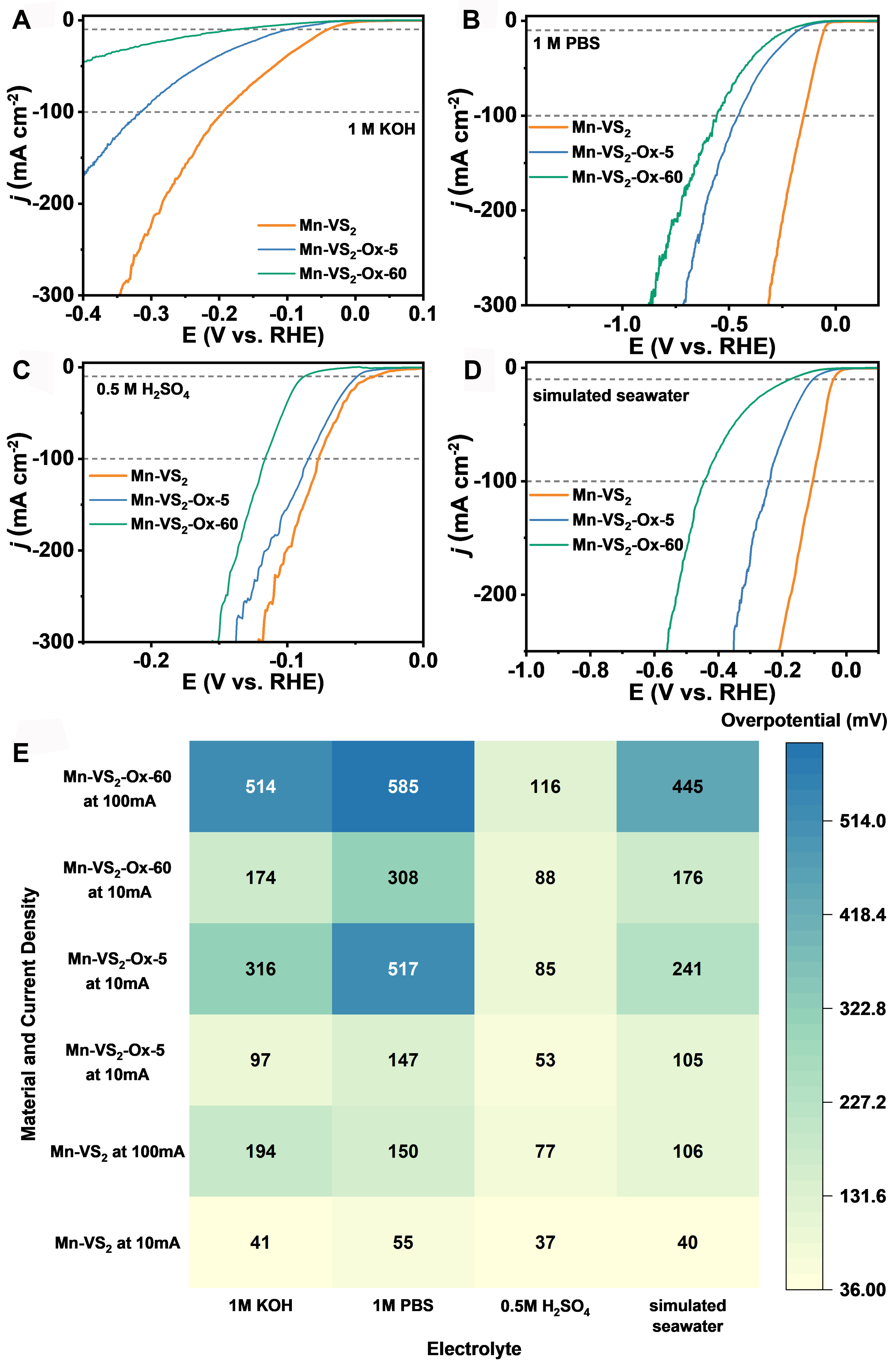
The long-term stability of the catalysts in simulated seawater was assessed using chronoamperometric methods, and as depicted in Supplementary Figure 17D, after 100 h of stability testing, the electrocatalytic performance of Mn-VS2 showed negligible degradation, demonstrating exceptional stability. Collectively, these experimental results confirm that Mn-VS2 not only exhibits excellent catalytic performance across the entire pH spectrum and in simulated seawater but also maintains outstanding stability, highlighting its significant potential for practical applications. To further elucidate the role of sulfur in Mn-VS2, the material was subjected to calcination in an air atmosphere for 5 min to partially remove sulfur, yielding a product referred to as Mn-VS2-Ox-5. This modified sample underwent a series of characterizations and electrochemical tests. The XRD peaks presented in Supplementary Figure 18 align with the standard diffraction data for VS2 (JCPDF No. 89-1640) and V2O5 (JCPDF No. 89-0612), indicating that while the VS2 structure was retained post-calcination, partial transformation to V2O5 occurred.
XPS full spectrum analysis of Mn-VS2-Ox-5, shown in Supplementary Figure 19A, confirms the presence of Mn, V, S, and O. Supplementary Figure 19B displays the XPS spectra of vanadium in both Mn-VS2 and Mn-VS2-Ox-5. For Mn-VS2-Ox-5, the V 2p peaks predominantly suggest a V4+ oxidation state, indicating an increase in the oxidative state of vanadium due to the calcination process, which typically involves reactions between vanadium and atmospheric oxygen, reducing the divalent vanadium content relative to Mn-VS2. The XPS spectrum of Mn in Supplementary Figure 19C reveals the Mn 2p3/2 peak split into two parts, representing the valence states of Mn2+ and Mn4+. In Mn-VS2-Ox-5, these peaks also display the Mn2+ and
Figure 4 displays the electrocatalytic HER performance of Mn-VS2 and its calcined derivatives,
Figure 4. Polarization curves of Mn-VS2, Mn-VS2-Ox-5 and Mn-VS2-Ox-60 measured in (A) 0.5 M H2SO4, (B) 1 M PBS, (C) 1 M KOH, and (D) simulated seawater with iR compensation at 95%; (E) Overpotentials of Mn-VS2, Mn-VS2-Ox-5 and Mn-VS2-Ox-60 at current densities of -10 and -100 mA·cm-2 in different environments.
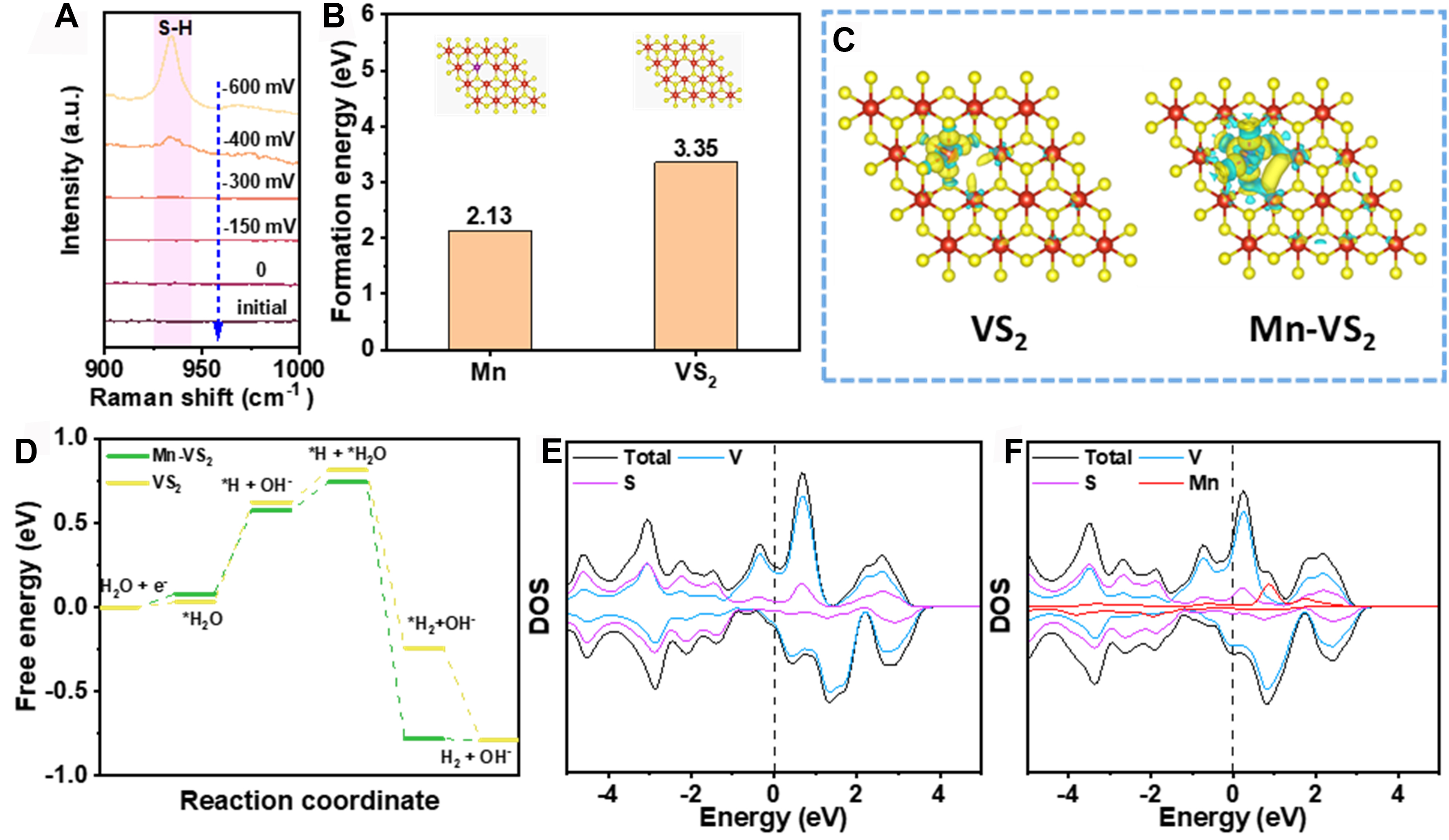
To further investigate the mechanism of the HER in Mn-VS2, in situ Raman spectroscopy was performed, as shown in Figure 5A. The tests covered a potential range from 0 to -600 mV relative to the Ag/AgCl reference electrode, encompassing both non-Faradaic and HER-active regions. Notably, at a potential of -300 mV, a new Raman peak at 934 cm-1 was observed, which intensified with further increases in cathodic potential. Previous studies suggest that this Raman peak likely originates from the stretching vibration of the S–H bond[59]. The enhancement of this S–H vibrational mode indicates the possible formation of an intermediate during the electrocatalytic process, where the increased potential facilitates the adsorption of hydrogen atoms or protons at the electrode surface, subsequently forming S–H bonds with sulfur atoms. The presence of S–H bonds indicates that sulfur atoms act as active sites in the HER catalyzed by Mn-VS2. To better understand the type of defects, calculations were performed on the formation energy of sulfur vacancies. As displayed in Figure 5B, the energy required to form a sulfur vacancy in VS2 is 3.35 eV; however, with the introduction of Mn, the formation energy significantly decreases to 2.13 eV. This result implies that Mn doping substantially reduces the formation energy of sulfur vacancies, making their formation more feasible. This finding is consistent with the results from EPR studies shown in Figure 2C, further illustrating that Mn doping promotes the generation of sulfur vacancies.
Figure 5. (A) In situ Raman spectra of Mn-VS2 at various potentials in 1 M KOH solution; (B) Formation energy of sulfur vacancies in VS2 and Mn-VS2; (C) Differential charge density maps of VS2 and Mn-VS2; (D) Free energy diagrams of VS2 and Mn-VS2 during the HER reaction; (E) DOS diagram of VS2 and (F) Mn-VS2. DOS: Density of states; HER: hydrogen evolution reaction.
Figure 5C displays the differential charge density maps for VS2 and Mn-VS2. In these maps, regions of increased charge density are depicted in yellow, while areas of decreased charge density appear in blue. In the VS2 model shown on the left, the charge distribution is relatively uniform, with no significant areas of charge accumulation or loss, indicating an even electron distribution among the atoms with slight charge accumulation at sulfur vacancies. In contrast, the Mn-VS2 model on the right exhibits distinct yellow regions around sulfur vacancies, indicating substantial charge accumulation due to the introduction of Mn. Such electron rearrangement is often associated with electrocatalytic activity, as HER typically requires electron density to facilitate the adsorption of reactants. This charge accumulation at sulfur vacancies suggests enhanced adsorption capabilities and catalytic activity at these sites. Combined with electrochemical test results and in situ Raman spectroscopy, these observations further suggest that sulfur acts as an active center for HER. The redistribution of the electron cloud around sulfur due to sulfur vacancies likely increases the electron affinity of sulfur for adsorbates such as water molecules or hydrogen atoms, facilitating the catalytic process. In alkaline conditions, the water dissociation process, or Volmer step, often presents a high reaction barrier and is the rate-determining step in HER. Analysis from Figure 5D indicates that the introduction of Mn significantly lowers the water dissociation barrier in
The impact of Mn doping on the electronic properties of sulfur vacancies in VS2 was further explored through changes in the density of states. As revealed in Figure 5E and F, the introduction of Mn leads to an accumulation of electrons at sulfur vacancies, which in turn restructures the electronic configuration of the material, particularly near the Fermi level, where a noticeable change in the density of states is observed. Adjusting the electronic structure is crucial for enhancing the material’s catalytic activity, thus making sulfur vacancies a key factor in optimizing electron transport. These sulfur vacancies not only reduce electron density in adjacent regions but may also enhance the material’s adsorption capabilities for electrons and protons.
CONCLUSIONS
In conclusion, this study has successfully synthesized Mn-VS2 using a colloidal chemistry approach, systematically examining their HER performance across the full pH spectrum and in simulated seawater environments. This research thoroughly elucidated the catalytic mechanisms at sulfur sites, highlighting them as active sites for HER. The uniform doping of Mn within the VS2 nanostructures was achieved by finely tuning the colloidal chemistry reaction conditions. The Mn-VS2 catalysts demonstrated remarkably low overpotentials of 41, 55, and 37 mV to reach a current density of -10 mA·cm-2 in 1 M KOH, 1 M PBS, and 0.5 M H2SO4 solutions, respectively. Particularly noteworthy is the performance of the catalyst in simulated seawater, where it achieved the same current density at an overpotential of just 40 mV, showcasing its potential as an effective catalyst for direct seawater electrolysis. In situ Raman spectroscopy and analyses assessing the impact of partial sulfur removal have confirmed the pivotal role of sulfur in the HER process. The introduction of Mn was found to increase the number of sulfur vacancies, thereby affecting the electron cloud density around the active sulfur sites and enhancing the adsorption of reaction intermediates. This study provides substantial evidence supporting the development of Mn-VS2 as an effective catalyst for HER, offering valuable insights for future strategies aimed at designing and improving HER electrocatalysts.
DECLARATIONS
Authors’ contributions
Conceptualized and supervised the project: Wang, R.; Cao, L.; Dong, B.
Synthesized the catalysts and performed the electrochemical tests: Wang, R.
Performed sample characterizations and data analysis: Wang, R.; Wan, L.
Co-wrote the manuscript: Wang, R.; Wan, L.; Liu, X.; Dong, B.
Reviewed the paper: Wang, R.; Wan, L.; Cao, L.; Dong, B.
All the authors discussed the results and revised the manuscript.
Availability of data and materials
The raw data supporting the findings of this study are available within this Article and its Supplementary Materials. Further data is available from the corresponding authors upon reasonable request.
Financial support and sponsorship
This research was supported by the Shandong Provincial of China Natural Science Foundation (ZR2022MB003), the Key Research and Development Program of Shandong Province (2020ZLYS04), and the Research Project of Henan Ruilong New Material Technology Co., Ltd (Heng20240014). All organizations have made equivalent contributions.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Zhai, P.; Xia, M.; Wu, Y.; et al. Engineering single-atomic ruthenium catalytic sites on defective nickel-iron layered double hydroxide for overall water splitting. Nat. Commun. 2021, 12, 4587.
2. Zheng, Y.; Jiao, Y.; Li, L. H.; et al. Toward design of synergistically active carbon-based catalysts for electrocatalytic hydrogen evolution. ACS. Nano. 2014, 8, 5290-6.
3. Zhou, Z.; Pei, Z.; Wei, L.; Zhao, S.; Jian, X.; Chen, Y. Electrocatalytic hydrogen evolution under neutral pH conditions: current understandings, recent advances, and future prospects. Energy. Environ. Sci. 2020, 13, 3185-206.
4. Lang, C.; Jia, Y.; Yan, X.; Ouyang, L.; Zhu, M.; Yao, X. Molecular chemisorption: a new conceptual paradigm for hydrogen storage. Chem. Synth. 2022, 2, 1.
5. Wang, Y.; Chen, Y.; Yun, Y.; Hong, X.; Huang, Y.; Ji, H. CoMoP hole transfer layer functionally enhances efficiency and stability of BiVO4 based photoanode for solar water splitting. Appl. Catal. B-Environ. 2024, 358, 124375.
6. McCrory, C. C.; Uyeda, C.; Peters, J. C. Electrocatalytic hydrogen evolution in acidic water with molecular cobalt tetraazamacrocycles. J. Am. Chem. Soc. 2012, 134, 3164-70.
7. Chen, Y.; Li, X.; Yang, H.; Huang, Y. Systematic constructing FeOCl/BiVO4 hetero-interfacial hybrid photoanodes for efficient photoelectrochemical water splitting. Small 2024, 20, e2402406.
8. Wu, H.; Lu, S.; Yang, B. Carbon-dot-enhanced electrocatalytic hydrogen evolution. Acc. Mater. Res. 2022, 3, 319-30.
9. Yang, F.; Zhao, Y.; Du, Y.; et al. A monodisperse Rh2 P-based electrocatalyst for highly efficient and pH-universal hydrogen evolution reaction. Adv. Energy. Mater. 2018, 8, 1703489.
10. Callejas, J. F.; McEnaney, J. M.; Read, C. G.; et al. Electrocatalytic and photocatalytic hydrogen production from acidic and neutral-pH aqueous solutions using iron phosphide nanoparticles. ACS. Nano. 2014, 8, 11101-7.
11. He, Y.; Hu, Y.; Zhu, Z.; et al. High-performance multidimensional-structured N-doped nickel modulated Mo2N/FeOxNy bifunctional electrocatalysts for efficient alkaline seawater splitting. Chem. Eng. J. 2024, 489, 151348.
12. Meng, L.; Shang, S.; Liu, S.; et al. Cooperative effects between NiMo alloy enable highly efficient for all-pH-value and alkaline seawater hydrogen evolution. Appl. Catal. B-Environ. 2024, 358, 124388.
13. Wang, L.; Cao, J.; Cheng, X.; et al. ZIF-derived carbon nanoarchitecture as a bifunctional pH-universal electrocatalyst for energy-efficient hydrogen evolution. ACS. Sustainable. Chem. Eng. 2019, 7, 10044-51.
14. Zhang, X.; Gao, B.; Rao, R.; et al. Defects materials of institut lavoisier-125(Ti) materials enhanced photocatalytic activity for toluene and chlorobenzene mixtures degradation: mechanism study. J. Colloid. Interface. Sci. 2024, 660, 423-39.
15. Jiao, Y.; Zheng, Y.; Davey, K.; et al. Activity origin and catalyst design principles for electrocatalytic hydrogen evolution on heteroatom-doped graphene. Nature. Energy. 2016, 1, 16130.
16. Trasatti, S. Electrocatalysis of Hydrogen Evolution: Progress in Cathode Activation. In Advances in Electrochemical Science and Engineering; Gerischer, H., Tobias, C. W., Eds.; Wiley, 1991; pp 1-85.
17. Lu, Q.; Yu, Y.; Ma, Q.; Chen, B.; Zhang, H. 2D transition-metal-dichalcogenide-nanosheet-based composites for photocatalytic and electrocatalytic hydrogen evolution reactions. Adv. Mater. 2016, 28, 1917-33.
18. Guo, F.; Macdonald, T. J.; Sobrido, A. J.; Liu, L.; Feng, J.; He, G. Recent advances in ultralow-Pt-loading electrocatalysts for the efficient hydrogen evolution. Adv. Sci. 2023, 10, e2301098.
19. Li, C.; Baek, J. B. Recent advances in noble metal (Pt, Ru, and Ir)-based electrocatalysts for efficient hydrogen evolution reaction. ACS. Omega. 2020, 5, 31-40.
20. Xia, L.; Wang, F.; Pan, K.; et al. Dual CoxSy-modified tungsten disulfide double-heterojunction electrocatalyst for efficient hydrogen evolution in all-pH media. ACS. Appl. Mater. Interfaces. 2023, 15, 11765-76.
21. Sun, L.; Xu, H.; Cheng, Z.; et al. A heterostructured WS2/WSe2 catalyst by heterojunction engineering towards boosting hydrogen evolution reaction. Chem. Eng. J. 2022, 443, 136348.
22. Wang, R.; Guo, Z.; Tan, X.; et al. Atmosphere plasma treatment and Co heteroatoms doping on basal plane of colloidal 2D VSe2 nanosheets for enhanced hydrogen evolution. Int. J. Hydrogen. Energy. 2021, 46, 32425-34.
23. Husain, A.; Lee, D.; Danish, M.; et al. Elevated symmetric supercapacitor performance and simulated solar light-functioning H2O2 production using single-step fabricated 2D/2D NiAl-based LDH/CoNi-based MOF nanohybrid. Surf. Interfaces. 2024, 51, 104749.
24. Guo, Z.; Yang, L.; Wang, W.; Cao, L.; Dong, B. Ultrathin VS2 nanoplate with in-plane and out-of-plane defects for an electrochemical supercapacitor with ultrahigh specific capacitance. J. Mater. Chem. A. 2018, 6, 14681-8.
25. Kim, H. J.; Choi, B. K.; Lee, I. H.; et al. Electronic structure and charge-density wave transition in monolayer VS2. Current. Applied. Physics. 2021, 30, 8-13.
26. Luo, N.; Si, C.; Duan, W. Structural and electronic phase transitions in ferromagnetic monolayer VS2 induced by charge doping, Phys Rev B 2017;95:205432.
27. Wang, R.; Chen, Q.; Liu, X.; Hu, Y.; Cao, L.; Dong, B. Synergistic effects of dual-doping with Ni and Ru in monolayer VS2 nanosheet: unleashing enhanced performance for acidic HER through defects and strain. Small 2024, 20, e2311217.
28. Wang, R.; Wan, L.; Liu, X.; Cao, L.; Hu, Y.; Dong, B. Anchoring Pt nanoparticle onto monolayer VS2 nanosheets boost efficient acidic hydrogen evolution. Int. J. Hydrogen. Energy. 2024, 74, 384-91.
29. Feng, J.; Sun, X.; Wu, C.; et al. Metallic few-layered VS2 ultrathin nanosheets: high two-dimensional conductivity for in-plane supercapacitors. J. Am. Chem. Soc. 2011, 133, 17832-8.
30. Cui, Y.; Fan, W.; Liu, X.; Ren, J.; Gao, Y. Electronic conductivity of two-dimensional VS2 monolayers: a first principles study. Comput. Mater. Sci. 2021, 200, 110767.
31. Zhang, J.; Zhang, C.; Wang, Z.; et al. Synergistic interlayer and defect engineering in VS2 nanosheets toward efficient electrocatalytic hydrogen evolution reaction. Small 2018, 14, 1703098.
32. Zhang, H.; Liu, L.; Lau, W. Dimension-dependent phase transition and magnetic properties of VS2. J. Mater. Chem. A. 2013, 1, 10821.
33. Du, C.; Liang, D.; Shang, M.; et al. In situ engineering MoS2 NDs/VS2 lamellar heterostructure for enhanced electrocatalytic hydrogen evolution. ACS. Sustainable. Chem. Eng. 2018, 6, 15471-9.
34. Guo, T.; Song, Y.; Sun, Z.; et al. Bio-templated formation of defect-abundant VS2 as a bifunctional material toward high-performance hydrogen evolution reactions and lithium-sulfur batteries. J. Energy. Chem. 2020, 42, 34-42.
35. van, E. C.; Berges, J.; Hall, J.; et al. A full gap above the Fermi level: the charge density wave of monolayer VS2. Nat. Commun. 2021, 12, 6837.
36. Liang, H.; Shi, H.; Zhang, D.; et al. Solution growth of vertical VS2 nanoplate arrays for electrocatalytic hydrogen evolution. Chem. Mater. 2016, 28, 5587-91.
37. Su, J.; Wang, M.; Li, Y.; et al. Sub-millimeter-scale monolayer p-type H-phase VS2. Adv. Funct. Mater. 2020, 30, 2000240.
38. Peng, X.; Chen, L.; Liu, Y.; et al. Strain engineering of two-dimensional materials for energy storage and conversion applications. Chem. Synth. 2023, 3, 47.
39. Wang, T.; Zhang, X.; Yu, X.; et al. Modulating the electronic structure of VS2via Ru decoration for an efficient pH-universal electrocatalytic hydrogen evolution reaction. Nanoscale 2024, 16, 11250-61.
40. Dong, S.; Dong, Y.; Liu, B.; et al. Guiding transition metal-doped hollow cerium tandem nanozymes with elaborately regulated multi-enzymatic activities for intensive chemodynamic therapy. Adv. Mater. 2022, 34, e2107054.
41. Yang, Z.; Hu, C.; Zhang, Q.; et al. Bulk-phase reconstruction enables robust zinc metal anodes for aqueous zinc-ion batteries. Angew. Chem. Int. Ed. Engl. 2023, 62, e202308017.
42. Kuraganti, V.; Jain, A.; Bar-Ziv, R.; Ramasubramaniam, A.; Bar-Sadan, M. Manganese doping of MoSe2 promotes active defect sites for hydrogen evolution. ACS. Appl. Mater. Interfaces. 2019, 11, 25155-62.
43. Wang, L.; Gao, W.; Chen, X.; et al. Charge-modulated VS2 monolayer for effective hydrogen evolution reaction. J. Phys. Chem. C. 2021, 125, 12004-11.
44. Zhao, R.; Zhu, Y.; Zhou, J.; et al. Dual glutathione depletion enhanced enzyme catalytic activity for hyperthermia assisted tumor therapy on semi-metallic VSe2/Mn-CS. ACS. Nano. 2022, 16, 10904-17.
45. Zhang, W.; Chen, X.; Zhang, J.; et al. Exposure of active edge structure for electrochemical H2 evolution from VS2/MWCNTs hybrid catalysts. Int. J. Hydrogen. Energy. 2018, 43, 22949-54.
46. Bao, D.; Huang, L.; Gao, Y.; Davey, K.; Zheng, Y.; Qiao, S. Z. Dynamic creation of a local acid-like environment for hydrogen evolution reaction in natural seawater. J. Am. Chem. Soc. 2024, 146, 34711-9.
47. Xu, R.; Huang, J.; Cao, L.; et al. VS2 microflowers with in situ embedded few-layer MoS2 nanobelts for enhanced hydrogen evolution reaction at high current density. J. Electrochem. Soc. 2020, 167, 026508.
48. He, W.; Zheng, X.; Peng, J.; Dong, H.; Wang, J.; Zhao, W. Mo-dopant-strengthened basal-plane activity in VS2 for accelerating hydrogen evolution reaction. Chem. Eng. J. 2020, 396, 125227.
49. Xu, J.; Zhu, Y.; Yu, B.; Fang, C.; Zhang, J. Metallic 1T-VS2 nanosheets featuring V2+ self-doping and mesopores towards an efficient hydrogen evolution reaction. Inorg. Chem. Front. 2019, 6, 3510-7.
50. Feng, T.; Ouyang, C.; Zhan, Z.; Lei, T.; Yin, P. Cobalt doping VS2 on nickel foam as a high efficient electrocatalyst for hydrogen evolution reaction. Int. J. Hydrogen. Energy. 2022, 47, 10646-53.
51. Bolar, S.; Samanta, P.; Jang, W.; Yang, C.; Murmu, N. C.; Kuila, T. Regulating the metal concentration for selective tuning of VS2/MoS2 heterostructures toward hydrogen evolution reaction in acidic and alkaline media. ACS. Appl. Energy. Mater. 2022, 5, 10086-97.
52. Zhang, J.; Li, J.; Huang, H.; et al. Spatial relation controllable Di-defects synergy boosts electrocatalytic hydrogen evolution reaction over VSe2 nanoflakes in all pH electrolytes. Small 2022, 18, e2204557.
53. Feng, L.; Li, S.; He, D.; et al. Heterostructured VN/Mo2C nanoparticles as highly efficient pH-universal electrocatalysts toward the hydrogen evolution reaction. ACS. Sustainable. Chem. Eng. 2021, 9, 15202-11.
54. Xie, Z.; Wang, W.; Ding, D.; et al. Accelerating hydrogen evolution at neutral pH by destabilization of water with a conducting oxophilic metal oxide. J. Mater. Chem. A. 2020, 8, 12169-76.
55. Ni, Y.; Ma, X.; Wang, S.; et al. Heterostructured nickel/vanadium nitrides composites for efficient electrocatalytic hydrogen evolution in neutral medium. J. Power. Sources. 2022, 521, 230934.
56. Chhowalla, M.; Shin, H. S.; Eda, G.; Li, L. J.; Loh, K. P.; Zhang, H. The chemistry of two-dimensional layered transition metal dichalcogenide nanosheets. Nat. Chem. 2013, 5, 263-75.
57. Voiry, D.; Yamaguchi, H.; Li, J.; et al. Enhanced catalytic activity in strained chemically exfoliated WS2 nanosheets for hydrogen evolution. Nat. Mater. 2013, 12, 850-5.
58. Xu, P.; Wang, H.; Guo, S.; et al. Atomic Ru-doped Ni0.85Se nanosheets as efficient electrocatalysts toward simulated seawater hydrogen evolution. Energy. Fuels. 2023, 37, 18204-9.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].