Impact of obstructive sleep apnea risk profile assessed by the Berlin Questionnaire on atrial fibrillation recurrence after catheter ablation: a 16-year follow-up
Abstract
Aim: To evaluate the long-term impact of obstructive sleep apnea (OSA) risk profile on atrial fibrillation (AF) recurrence after catheter ablation in patients with paroxysmal AF.
Methods: This prospective study enrolled 161 patients with paroxysmal AF undergoing initial ablation. Patients were stratified by the Berlin Questionnaire (BQ) into high-risk
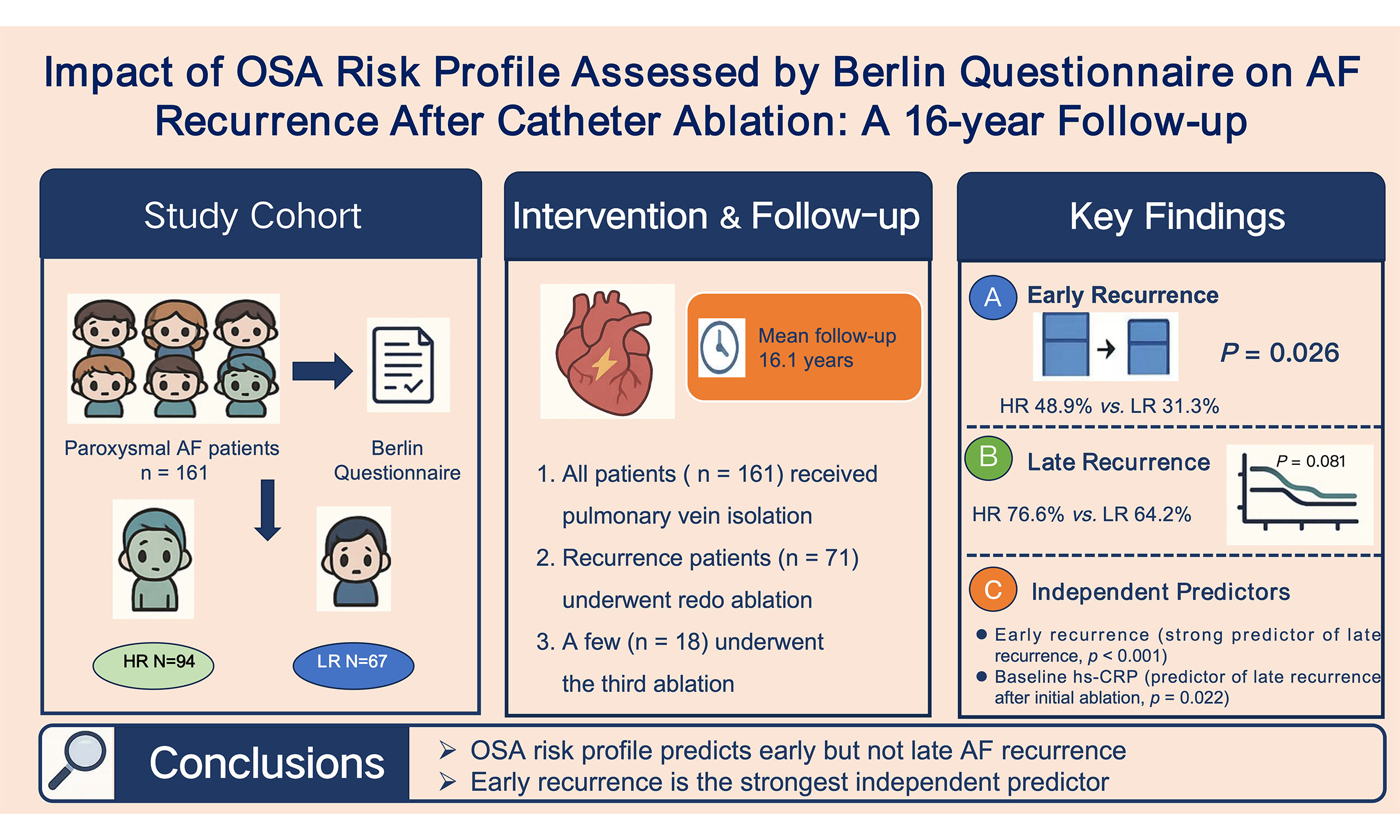
Results: After 16.1 ± 0.4 years, multivariable models revealed that a BQ-defined high OSA risk was independently associated with early recurrence (odds ratio [OR] 1.99, 95% confidence interval [CI] 1.02-3.86, P = 0.043), whereas it was not independently associated with late recurrence after the initial procedure (hazard ratio [HR] 1.05, 95%CI: 0.70-1.57, P = 0.810). Cox regression identified early recurrence (HR 2.95, 95%CI: 1.99-4.39, P < 0.001) and baseline high-sensitivity C-reactive protein (hs-CRP; HR 1.02, 95%CI: 1.00-1.03, P = 0.022) as independent predictors of late recurrence after the initial procedure. Following redo ablation, early recurrence remained a strong independent predictor (HR 5.93, 95%CI: 2.13-16.50, P < 0.001).
Conclusions: The BQ-defined high OSA risk was an independent predictor of early recurrence; however, it was not significantly associated with late recurrence after AF ablation. Early recurrence was the strongest predictor of long-term outcome, highlighting the importance of management during the post-procedural blanking period.
Keywords
INTRODUCTION
Atrial fibrillation (AF) is the most prevalent persistent arrhythmia in clinical settings. Approximately 37.574 million cases of AF have been reported globally, representing 0.51% of the worldwide population, and this number has increased by 33% over the past two decades[1]. In China, the prevalence of AF is at 1.8%, which increases with advancing age[2]. Catheter ablation is a well-established therapy for AF and is more effective than antiarrhythmic drugs in maintaining sinus rhythm[3]. Obstructive sleep apnea (OSA) is a recognized independent risk factor for AF[4,5], driven by a cascade of interconnected pathophysiological mechanisms. These include intermittent hypoxia and hypercapnia, which promote systemic inflammation and oxidative stress; exaggerated negative intrathoracic pressure swings, leading to mechanical atrial stretch; and resultant autonomic nervous system imbalance with sympathetic surges. Collectively, these processes contribute to atrial electrical and structural remodeling, creating a substrate conducive to the initiation and perpetuation of AF[6]. Studies have suggested that patients with OSA may be at higher risk for AF recurrence after catheter ablation than those without OSA[7,8]. However, other reports have found no significant association between OSA and ablation outcomes[9,10], highlighting the ongoing controversy in this area. Our prior study enrolled a cohort of 178 patients with paroxysmal AF, all of whom underwent circumferential pulmonary vein ablation (CPVA). Over a mean follow-up of 344 days, we found that the OSA risk profile assessed by the Berlin Questionnaire (BQ) did not affect late recurrence of AF[11]. To address the limitation of the relatively short follow-up in our previous study, we extended the follow-up duration for this cohort to more than 16 years, aiming to delve deeper into the enduring effects of this OSA risk profile on the recurrence and subsequent redo ablation in patients with paroxysmal AF after pulmonary vein isolation.
METHODS
Study subjects
This study initially enrolled 178 patients with paroxysmal AF who underwent first-time circumferential pulmonary vein radiofrequency catheter ablation at Beijing Anzhen Hospital, Capital Medical University, between January and November 2007. Paroxysmal AF was defined in accordance with contemporary guidelines as self-terminating episodes lasting less than seven days[12]. The exclusion criteria were as follows: (1) presence of atrial or left atrial appendage thrombi; (2) current use of continuous positive airway pressure (CPAP) therapy for OSA. The study was approved by the Institutional Review Board prior to patient participation.
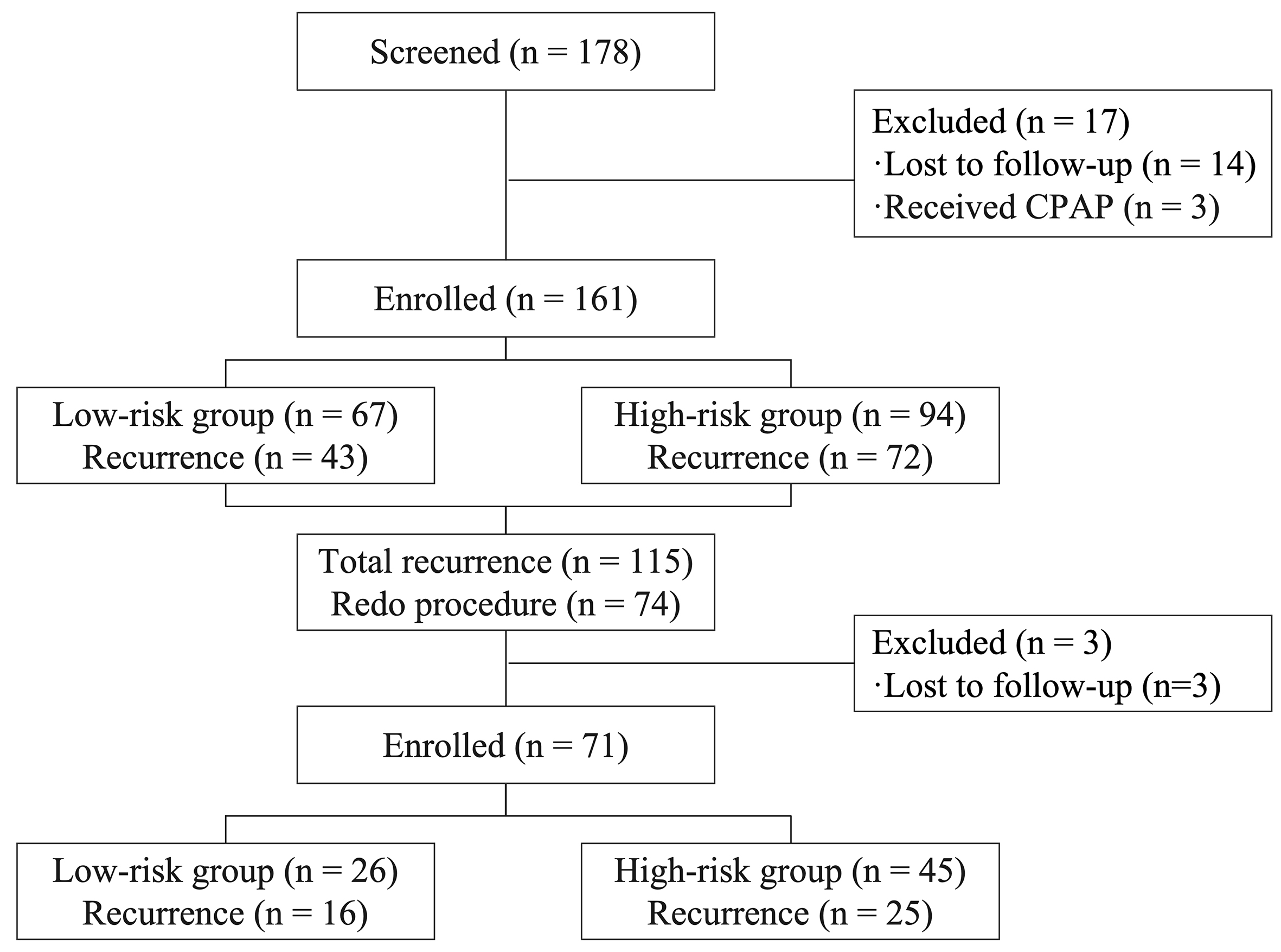
During a median follow-up period of 16.1 ± 0.4 years through June 2023, 14 patients were lost to follow-up due to invalid contact information, and 3 received CPAP treatment, resulting in 161 patients completing long-term follow-up. The study flowchart details participant enrollment and attrition [Figure 1].
OSA risk profile evaluation
This study employed the BQ for OSA screening to ensure continuity with our prior cohort study and because it was the standard tool used at our center at the time of patient enrollment (2007)[11]. Although validated screening tools such as STOP-BANG (Snoring, Tiredness, Observed apnea, high blood Pressure, Body mass index, Age, Neck circumference, and Gender) have been widely used, the BQ provided a consistent baseline assessment for this long-term follow-up. Designed to target the primary symptoms of OSA, the questionnaire encompasses three evaluative dimensions: snoring, sleepiness, and the presence of hypertension or obesity. Patients, together with their bed partners, completed the questionnaire to assess OSA risk after hospitalization and prior to the procedure. The study cohort was stratified into OSA high-risk (HR) and low-risk (LR) groups based on the BQ. High-risk OSA designation applies to patients with two or more of the following criteria: (1) persistent symptoms occurring more than three times per week in at least two snoring-related questions; (2) persistent daytime somnolence (> 3 times/week) and/or somnolence while driving; and (3) diagnosed with hypertension or a body mass index (BMI) > 30 kg/m2. Patients not meeting these criteria were classified as low risk for OSA.
In addition, we administered the Epworth Sleepiness Scale (ESS) questionnaire to all participants and selected 30 patients for polysomnography (PSG) to validate the accuracy of the screening tool in the target population. The apnea-hypopnea index (AHI) refers to the mean number of apnea and hypopnea episodes occurring per hour during sleep. Based on the sleep-monitoring criteria outlined in the International Consensus Statement, OSA was diagnosed when the AHI was ≥ 5 events per hour[13].
Electrophysiological study, mapping, and ablation
Prior to the procedure, all patients underwent preoperative transthoracic echocardiography to measure relevant parameters (left atrial diameter was measured in the parasternal long-axis view at end-systole), followed by transesophageal echocardiography to exclude intracardiac thrombi. Antiarrhythmic drugs (except amiodarone) were discontinued for at least five half-lives before ablation. Written informed consent was obtained from all enrolled patients before procedure. The procedure was conducted under fasting conditions, with the patients in an awake and sedated state. A coronary sinus electrode was inserted through the left femoral vein puncture. The initial dose of unfractionated heparin was 6000 IU, with additional heparin administered to maintain the activated clotting time within the range of 300-350 s after transseptal puncture[12]. A non-force-sensing 3.5 mm mapping ablation catheter (Navi-Star ThermoCool, Biosense-Webster, USA) was used with the CARTO XP system to create a three-dimensional electroanatomic map of the left atrium. Ablation parameters included a power setting of 35 W, a preset temperature of 43 °C, and an infusion rate of 17 mL/min for cold saline perfusion. Continuous ablation was applied around the antral region of each pulmonary vein to achieve encirclement and electrical isolation of the ipsilateral veins. Isolation was confirmed using a Lasso electrode (Biosense-Webster, USA). In the event of a typical atrial flutter during the procedure, the tricuspid isthmus responsible for the tachycardia was identified and ablated.
In cases of AF recurrence, a redo catheter ablation may be undertaken three months after the initial ablation. During the redo ablation, a Lasso electrode (Biosense-Webster, USA) was used to assess electrical conduction recovery in all pulmonary veins. The ablation endpoints were pulmonary vein isolation and the absence of inducible atrial tachycardia during rapid atrial pacing. For patients experiencing atrial flutter or atrial tachycardia, mapping and entrainment techniques were used to analyze the underlying mechanism, and the critical isthmus was targeted. Additional ablation lines, including the mitral isthmus, tricuspid isthmus, and left atrial roof, were created at the operator's discretion. Superior vena cava (SVC) isolation and fragmented potential ablation could also be performed. The endpoint of linear ablation was to achieve bidirectional blocks across the ablation lines.
Follow-up
Post-procedure, all patients were prescribed oral antiarrhythmic drugs unless contraindicated or not tolerated. If tachyarrhythmia was absent after three months, antiarrhythmic drugs were discontinued. Anticoagulants were administered for a minimum of three months, and the long-term anticoagulation strategy was determined based on the risks of stroke and bleeding. Follow-up assessments included standard 12-lead electrocardiogram (ECG) and 24-h Holter examination at one, two, three, six, and 12 months post-ablation, followed by assessments every six months thereafter. Trained and experienced personnel conducted follow-up visits, using methods such as telephone consultations and outpatient visits. If palpitations or other symptoms indicative of arrhythmia occurred during the follow-up period, patients were advised to undergo an ECG promptly at a local hospital. The patients were censored at the time of AF recurrence. At the final follow-up visit in June 2023, 14 patients had been lost to follow-up.
Definition of early recurrence and late recurrence
Recurrent AF was diagnosed when an episode of atrial tachyarrhythmia (atrial tachycardia, atrial flutter, or AF) lasting ≥ 30 s in duration was documented after the 3-month blanking period[12]. Recurrence occurring within the first three months was defined as early recurrence, whereas recurrence manifesting after the initial three months was termed late recurrence. AF recurrence was confirmed on ECG during follow-up.
Statistical analysis
Statistical analyses were conducted with SPSS 26.0 and R 4.5.1. Normally distributed continuous variables are expressed as mean ± standard deviation (SD) and were compared using the independent samples t-test, while non-normally distributed data are presented as median (interquartile range, IQR) and compared using the Mann-Whitney U test. Categorical variables are expressed as frequencies or percentages, and were compared using the χ2 test or Fisher's exact test. The impact of OSA risk profile assessed by BQ on late AF recurrence was analyzed using Kaplan-Meier estimation, with the log-rank test used for comparison. Logistic regression analysis was employed to assess the risk factors associated with early recurrence. Association with late recurrence was evaluated using both univariate and multivariate Cox regression analyses. Variables identified in a univariate Cox regression analysis with P-values < 0.15, along with other pertinent risk factors (e.g., OSA risk profile) influencing AF recurrence, were included in the multivariate Cox regression analysis. All probability values were two-sided, and statistical significance was set at P < 0.05. Variance inflation factors (VIFs, with a VIF <5 indicating no substantial collinearity) and sensitivity analyses (under extreme assumptions regarding outcomes in patients lost to follow-up) were used to assess robustness. Subgroup analyses were performed according to BMI, age, sex, left atrial diameter, and high-sensitivity C-reactive protein (hs-CRP) level.
RESULTS
Validation of the BQ
In the initial phase of this cohort study, we validated the BQ using PSG in a subset of 30 participants. Comparative analysis demonstrated that the questionnaire exhibited 100.0% sensitivity, 30.0% specificity, 74.1% positive predictive value, and 100% negative predictive value for detecting OSA. Notably, the AHI differed significantly between the high- and low-risk groups as stratified by the questionnaire (28.5 ± 22.7 vs. 2.2 ± 1.4, P = 0.001). In addition, dedicated ESS assessments within this AF cohort validated symptom-aligned risk stratification: HR patients exhibited significantly higher ESS scores (7.2 ± 4.4 vs.
Characteristics of the high- and low-risk groups
Among the 161 patients, 94 (58.4%) were classified as the HR group, while 67 (41.6%) were classified as the LR group. No significant differences were observed between the two groups in sex, age, AF duration, diabetes, coronary heart disease, stroke, or other baseline factors. However, compared to the LR group, the HR group exhibited higher BMI, neck circumference, abdominal circumference, hypertension, left atrial diameter, and the use of angiotensin-converting enzyme inhibitors or angiotensin receptor blocker medications [Table 1].
Characteristics of high-risk and low-risk groups
| Category | LR (n = 67) | HR (n = 94) | P |
| Age (years) | 56.16 ± 11.83 | 57.84 ± 11.40 | 0.367 |
| Males, n (%) | 44 (65.7%) | 65 (69.1%) | 0.642 |
| AF duration (years) | 5.00 (2.00-9.00) | 5.50 (2.00-10.00) | 0.608 |
| DM, n (%) | 5 (7.5%) | 14 (14.9%) | 0.15 |
| Hypertension, n (%) | 15 (22.4%) | 62 (66.0%) | < 0.001* |
| Heart failure, n (%) | 4 (6.0%) | 3 (3.2%) | 0.645 |
| CAD, n (%) | 4 (6.0%) | 8 (8.5%) | 0.545 |
| Stroke, n (%) | 1 (1.5%) | 6 (6.4%) | 0.178 |
| BMI (kg/m2) | 24.72 ± 2.97 | 26.08 ± 3.25 | 0.008* |
| LAD (mm) | 35.99 ± 5.08 | 38.02 ± 5.82 | 0.023* |
| LVEDD (mm) | 48.92 ± 5.17 | 48.37 ± 4.65 | 0.485 |
| LVESD (mm) | 32.07 ± 4.87 | 31.43 ± 4.85 | 0.409 |
| Ejection fraction (%) | 63.37 ± 6.59 | 63.63 ± 6.79 | 0.805 |
| Neck circumference (cm) | 37.97 ± 3.16 | 40.11 ± 3.58 | < 0.001* |
| Abdominal circumference (cm) | 90.15 ± 9.22 | 93.90 ± 10.13 | 0.017* |
| Antiarrhythmic drugs | 0.746 | ||
| No medication, n (%) | 22 (32.8%) | 26 (27.7%) | |
| Class Ⅰ, n (%) | 10 (14.9%) | 12 (12.8%) | |
| Amiodarone, n (%) | 35 (52.2%) | 54 (57.4%) | |
| Sotalol, n (%) | 0 (0) | 2 (2.1%) | |
| β-blockers, n (%) | 16 (23.9%) | 28 (29.8%) | 0.407 |
| ACEI/ARB, n (%) | 11 (16.4%) | 43 (45.7%) | < 0.001* |
| Statins, n (%) | 9 (14.3%) | 14 (15.4%) | 0.851 |
| hs-CRP (mg/L) | 2.00 (0.70-4.50) | 1.95 (0.78-4.50) | 0.940 |
| Procedure time (min) | 159.70 ± 40.04 | 166.98 ± 38.05 | 0.244 |
| X-ray time (min) | 31.00 ± 7.46 | 32.60 ± 7.24 | 0.175 |
| PVI, n (%) | 66 (98.5%) | 91 (96.8%) | 0.866 |
| Early recurrence, n (%) | 21 (31.3%) | 46 (48.9%) | 0.026* |
BQ-defined OSA risk and recurrence of AF after the initial ablation
After the initial ablation, early recurrence occurred in 48.9% (46/94) of patients in the HR group, compared with 31.3% (21/67) in the LR group (P = 0.026). In multivariate logistic regression analysis, OSA risk profile assessed by BQ (odds ratio [OR] 1.99, 95% confidence interval [CI] 1.02-3.86; P = 0.043) was identified as the only independent risk factor for early AF recurrence [Table 2].
Predictors of early AF recurrence after the initial ablation
| Variable | Univariate analysis | Multivariate analysis | ||||
| OR | 95%CI | P | OR | 95%CI | P | |
| Age | 0.99 | 0.94-1.02 | 0.493 | - | - | - |
| Sex | 0.68 | 0.35-1.32 | 0.250 | - | - | - |
| OSA risk profile | 2.10 | 1.09-4.04 | 0.027* | 1.99 | 1.02-3.86 | 0.043* |
| AF duration | 1.03 | 0.96-1.10 | 0.269 | - | - | - |
| CAD | 0.44 | 0.12-1.07 | 0.240 | - | - | - |
| Stroke | 2.22 | 0.43-11.33 | 0.328 | - | - | - |
| Heart failure | 1.93 | 0.42-8.90 | 0.401 | - | - | - |
| DM | 2.71 | 1.01-7.31 | 0.049* | 2.47 | 0.90-6.74 | 0.079 |
| Hypertension | 1.22 | 0.65-2.29 | 0.531 | - | - | - |
| hs-CRP | 1.02 | 0.98-1.05 | 0.368 | - | - | - |
| BMI | 1.05 | 0.94-1.17 | 0.372 | - | - | - |
| LAD | 1.05 | 0.99-1.11 | 0.203 | - | - | - |
| LVEDD | 0.98 | 0.92-1.04 | 0.504 | - | - | - |
| LVESD | 1.01 | 0.95-1.08 | 0.669 | - | - | - |
| Ejection fraction | 0.97 | 0.92-1.02 | 0.199 | - | - | - |
| Procedure time | 1.00 | 0.99-1.00 | 0.287 | - | - | - |
| Neck circumference | 1.02 | 0.93-1.12 | 0.660 | - | - | - |
| Abdominal circumference | 1.02 | 0.99-1.05 | 0.307 | - | - | - |
| ACEI/ARB | 1.06 | 0.55-2.06 | 0.860 | - | - | - |
| Statins | 0.46 | 0.18-1.17 | 0.102 | - | - | - |
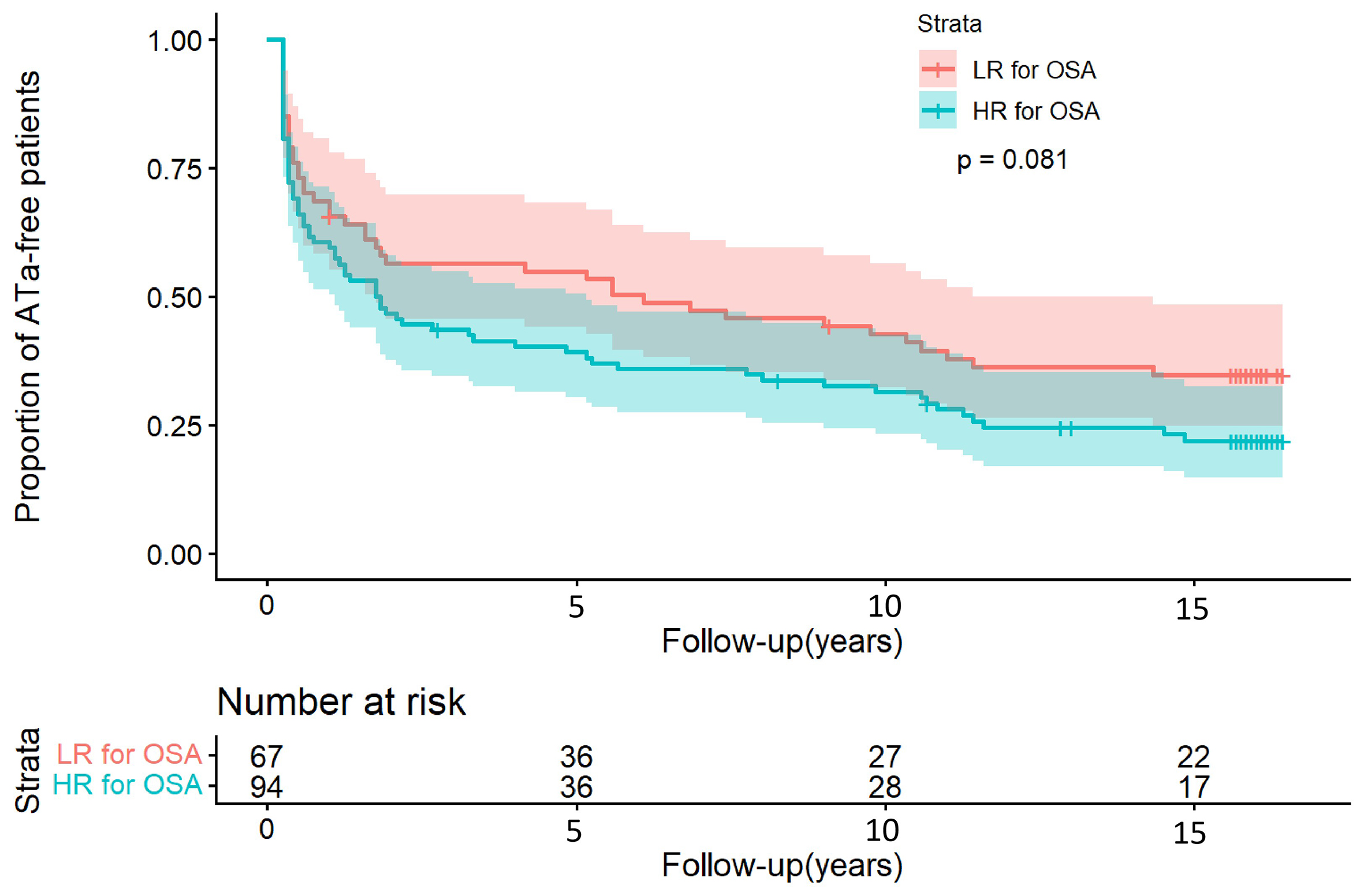
Over a mean follow-up duration of 16.1 ± 0.4 years, the late recurrence rate was 76.6% (72/94) in the HR group and 64.2% (43/67) in the LR group (P = 0.086). Among patients who experienced early recurrence, 89.6% progressed to late recurrence, with no significant difference between the groups (HR group 89.1% vs. LR group 90.4%, P = 1.000). The Kaplan-Meier curve indicated that this OSA risk profile was not significantly associated with late AF recurrence (log-rank P = 0.081, Figure 2). Univariate Cox regression analysis identified early recurrence [Hazard Ratio (HR) 3.08, 95%CI: 2.11-4.49, P < 0.001], baseline hs-CRP (HR 1.02, 95%CI: 1.00-1.04, P = 0.019), BMI (HR 1.05, 95%CI: 1.00-1.10, P = 0.122) and left atrial diameter (HR 1.04, 95%CI: 1.01-1.08, P = 0.025) as risk factors for late AF recurrence. Subsequent inclusion of these factors and OSA risk profile assessed by BQ in the multivariate Cox regression analysis revealed that early recurrence (HR 2.95, 95%CI: 1.99-4.39, P < 0.001) and baseline hs-CRP (HR 1.02, 95%CI: 1.00-1.03,
Figure 2. Kaplan-Meier analysis in atrial tachyarrhythmia (ATa) recurrence between the HR and LR groups after initial ablation (76.6% vs. 64.2%; log-rank P = 0.081). Time zero corresponds to the date of catheter ablation. The shaded areas around the curves represent the 95% confidence intervals.
Predictors of late AF recurrence after the initial ablation
| Variable | Univariate analysis | Multivariate analysis | ||||
| HR | 95%CI | P | HR | 95%CI | P | |
| Age | 0.99 | 0.98-1.01 | 0.29 | - | - | - |
| Sex | 0.99 | 0.67-1.46 | 0.94 | - | - | - |
| OSA risk profile | 1.38 | 0.95-2.02 | 0.092 | 1.05 | 0.70-1.57 | 0.810 |
| AF duration | 1.01 | 0.98-1.04 | 0.422 | - | - | - |
| PVI | 1.27 | 0.40-4.01 | 0.679 | - | - | - |
| Hypertension | 1.15 | 0.97-1.65 | 0.469 | - | - | - |
| DM | 1.05 | 0.59-1.87 | 0.868 | - | - | - |
| Heart failure | 1.33 | 0.46-2.78 | 0.786 | - | - | - |
| CAD | 1.48 | 0.77-2.85 | 0.236 | - | - | - |
| Stroke | 0.60 | 0.22-1.63 | 0.317 | - | - | - |
| Early recurrence | 3.08 | 2.11-4.49 | < 0.001* | 2.95 | 1.99-4.39 | < 0.001* |
| hs-CRP | 1.02 | 1.00-1.04 | 0.019* | 1.02 | 1.00-1.03 | 0.022* |
| LAD | 1.04 | 1.01-1.08 | 0.025* | 1.01 | 0.97-1.05 | 0.413 |
| LVEDD | 0.98 | 0.95-1.02 | 0.406 | - | - | - |
| LVESD | 1.01 | 0.97-1.05 | 0.758 | - | - | - |
| Procedure time | 1.00 | 0.10-1.00 | 0.773 | - | - | - |
| X-ray time | 1.00 | 0.98-1.03 | 0.925 | - | - | - |
| BMI | 1.05 | 1.00-1.10 | 0.122* | 1.02 | 0.96-1.08 | 0.564 |
| Neck circumference | 1.03 | 0.97-1.08 | 0.339 | - | - | - |
| Abdominal circumference | 1.01 | 0.99-1.03 | 0.296 | - | - | - |
| ACEI/ARB | 1.08 | 0.74-1.58 | 0.689 | - | - | - |
| Statins | 1.00 | 0.61-1.64 | 0.999 | - | - | - |
Supplementary and sensitivity analyses
Collinearity diagnostics confirmed the absence of substantial multicollinearity among the predictors. The VIFs were as follows: OSA risk profile, 1.39; early recurrence, 1.05; hs-CRP, 1.03; left atrial diameter, 1.30; BMI, 2.34; neck circumference, 2.18; abdominal circumference, 3.49; and hypertension, 1.25. All VIF values were well below the threshold of 5. To address potential bias due to patients lost to follow-up, we performed sensitivity analyses under extreme assumptions [Supplementary Table 1]. We constructed two extreme scenarios: in Scenario A (most optimistic), all lost patients were assumed to have no late recurrence; in Scenario B (most pessimistic), all lost patients were assumed to have experienced late recurrence. Multivariable Cox models were re-run under both scenarios. In both scenarios, the association between BQ-defined high OSA risk and late recurrence remained non-significant (all P > 0.05), whereas early recurrence and hs-CRP persisted as significant predictors. These results confirm the robustness of our primary findings. Furthermore, comparisons of baseline characteristics revealed that the recurrence and non-recurrence groups were well-balanced, except for the expected differences in OSA risk profile and early recurrence rate (all other P > 0.05; Supplementary Table 2). Subgroup analyses stratified by BMI, age, left atrial diameter, hypertension status, and other clinical factors revealed no significant association between the BQ-defined OSA risk profile and late recurrence in any subgroup (all P > 0.05; Supplementary Table 3).
Procedural findings during the redo ablation
Of the 74 patients who underwent redo ablation, 71 (HR group, 45 vs. LR group, 26) were included in the subsequent analysis after exclusion of three patients lost to follow-up, with an average age of 55.2 years [male 47 (66.2%)]. The rate of pulmonary vein reconnection did not differ significantly between the HR and LR groups (95.6% [43/45] vs. 88.5% [23/26], P = 0.458). All patients with reconnection underwent successful repeat isolation. Empirical linear ablation and SVC isolation were performed in both groups. In the HR group, 36 patients had linear ablation (mitral isthmus, n = 13; tricuspid isthmus, n = 27; left atrial roof, n = 8), and 6 underwent SVC isolation. The corresponding numbers in the LR group were 18 (8, 11, 4) and 7, respectively. Bidirectional mitral isthmus block was achieved via coronary sinus ablation in 5 HR and 4 LR patients, but was unsuccessful in 2 HR cases. Among the few patients without pulmonary vein reconnection (HR group 2 vs. LR group 3), recurrence was attributable to atrial flutter mechanisms (mitral/tricuspid isthmus-dependent, or left atrial appendage base-related), which were successfully treated with targeted linear ablation.
Eighteen patients (HR group 10 vs. LR group 8) underwent a third ablation. Pulmonary vein reconnection rates during the third procedure were also similar between groups (70.0% vs. 62.5%, P = 1.000). In the minority without reconnection, alternative mechanisms (e.g., mitral isthmus flutter, focal atrial tachycardia) were identified and ablated.
BQ-defined OSA risk and late AF recurrence after redo ablation
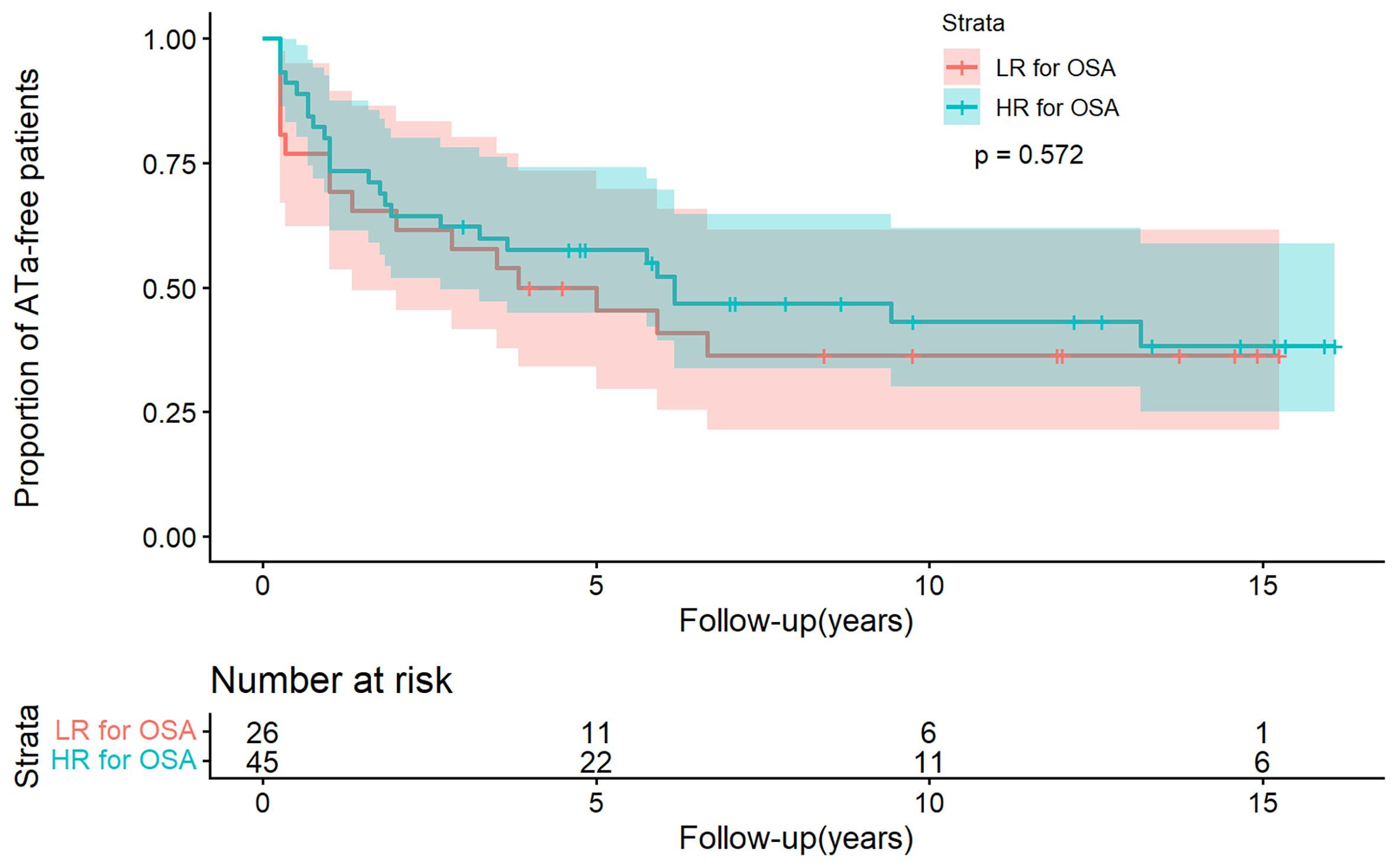
Among 71 patients who underwent redo ablation, the incidence of early recurrence was 6.7% (3/45) in the HR group and 19.2% (5/26) in the LR group (P = 0.221). Similarly, late recurrence rates did not differ significantly between groups (55.6% [25/45] vs. 61.5% [16/26], P = 0.623). Kaplan-Meier analysis confirmed that the OSA risk profile assessed by BQ had no significant effect on late recurrence (log-rank P = 0.572; Figure 3). On multivariate Cox regression, early recurrence following redo ablation remained an independent predictor of late recurrence (HR = 5.93, 95%CI: 2.13-16.50, P < 0.001), while this OSA risk profile showed no independent association with it (HR = 1.05, 95%CI: 0.50-2.20, P = 0.892; Table 4). Finally, BQ-defined OSA risk still had no impact on late AF recurrence between the two groups (HR group 60% vs. LR group 50%, P = 1.000) in the third ablation.
Figure 3. Kaplan-Meier analysis in atrial tachyarrhythmia (ATa) recurrence between the HR and LR groups after redo ablation (55.6% vs. 61.5%; log-rank P = 0.572). Time zero corresponds to the date of catheter ablation. The shaded areas around the curves represent the 95% confidence intervals.
Predictors of late AF recurrence after redo ablation
| Variable | Univariate analysis | multivariate analysis | ||||
| HR | 95%CI | P | HR | 95%CI | P | |
| Age | 1.01 | 0.98-1.03 | 0.654 | - | - | - |
| Gender | 1.21 | 0.62-2.38 | 0.577 | - | - | - |
| OSA risk profile | 0.84 | 0.45-1.57 | 0.58 | 1.05 | 0.50-2.20 | 0.892 |
| Early recurrence of redo ablation | 5.90 | 2.59-13.45 | < 0.001* | 5.93 | 2.13-16.50 | < 0.001* |
| Early recurrence of initial ablation | 2.09 | 1.09-3.99 | 0.026* | 1.35 | 0.65-2.81 | 0.426 |
| CAD | 0.75 | 0.29-1.91 | 0.547 | - | - | - |
| Stroke | 0.94 | 0.13-6.86 | 0.95 | - | - | - |
| Heart failure | 0.54 | 0.13-2.24 | 0.394 | - | - | - |
| DM | 2.51 | 1.18-5.33 | 0.017* | 2.29 | 0.93-5.66 | 0.073 |
| Hypertension | 0.71 | 0.38-1.31 | 0.274 | - | - | - |
| hs-CRP | 1.01 | 1.00-1.02 | 0.142 | 1.00 | 0.99-1.02 | 0.666 |
| BMI | 1.05 | 0.94-1.17 | 0.372 | - | - | - |
| LAD | 1.02 | 0.97-1.08 | 0.417 | - | - | - |
| LVEDD | 1.01 | 0.96-1.07 | 0.653 | - | - | - |
| LVESD | 1.03 | 0.98-1.09 | 0.203 | - | - | - |
| Ejection fraction | 1.21 | 0.64-2.29 | 0.549 | - | - | - |
| ACEI/ARB | 1.26 | 0.67-2.36 | 0.476 | - | - | - |
| Statins | 0.59 | 0.23-1.50 | 0.268 | - | - | - |
BQ-defined OSA risk and prognosis
In this study, AF recurrence rates at three, five, ten, and 16 years were 44.8%, 54.7%, 55.9%, and 71.4%, respectively, with no significant differences between the two groups at any time point (all P > 0.05). Notably, most recurrences occurred within the first three years. Among patients who remained recurrence-free at three years, subsequent late recurrence was not associated with OSA risk profile assessed by BQ (HR group 50.0% [20/40] vs. LR group 38.5% [14/37], P = 0.291). Among patients with recurrence, 12 (7.5%) progressed to persistent AF after initial ablation (LR group 3/43 vs. HR group 9/72, P = 0.534), and 11 additional cases (6.8%) developed persistent AF following multiple ablations (HR group 3/43 vs. LR group 8/72, P = 0.688). Regarding mortality, 23 deaths occurred in the cohort (nine patients died without prior recurrence), with no significant difference in all-cause mortality between the HR and LR groups (13.8% vs. 14.9%, P = 0.845). Causes of death included cardiogenic events (heart failure: 4, myocardial infarction: 1, sudden death: 1), cerebral hemorrhage (1), non-cardiogenic/non-neurogenic causes (13), and unknown etiologies (3).
DISCUSSION
Main findings
The major findings of this study could be summarized as follows: (i) In this long-term follow-up study, the OSA risk profile assessed by BQ was an independent predictor of early recurrence after catheter ablation for paroxysmal AF, but was not independently associated with late recurrence following either the initial or subsequent procedures; (ii) Early recurrence was an independent risk factor for late recurrence following both the first and redo ablation procedures; (iii) A higher baseline hs-CRP level independently predicted late recurrence after the initial ablation, while the predictive value for late recurrence after the redo catheter ablation for AF was limited.
Demonstration of the Berlin questionnaire on OSA diagnosis
The methods used for structured screening and testing OSA vary greatly. Clinical practice currently lacks standardized protocols for both screening AF patients for OSA and implementing subsequent OSA therapy[14]. As reported in prior research[15], the BQ displayed a sensitivity of 86%, specificity of 77%, positive predictive value of 89%, and a likelihood ratio of 3.79, underscoring its diagnostic utility and high accuracy in unselected populations. However, the BQ did not perform as well in the AF population as in the general population[16]. Daytime sleepiness was minimally present in patients with AF[17]. As shown in our earlier investigations[11] and benchmarked against PSG, the diagnostic performance for OSA in this cohort was 100% sensitivity, 30% specificity, 74.1% positive predictive value, and 100% negative predictive value. However, this performance was not validated in an external population.
Impact of BQ-defined OSA risk on AF late recurrence
In contrast to some previous studies[7,8,18,19], we found no significant association between OSA risk profile assessed by BQ and late recurrence following either initial or redo catheter ablation for AF. Several factors may account for this discrepancy. First, differences in follow-up duration may influence the conclusions. Our study had a follow-up period of up to 16 years, which is significantly longer than most prior studies
Phased impact of BQ-defined OSA risk stratification
Early recurrence is a key independent risk factor for late recurrence[29,30]. In our ultra-long-term cohort, we confirmed this association: nearly 90% of early recurrence cases progressed to late recurrence, confirming its key role in driving long-term outcomes. However, the BQ-defined OSA risk predicted early recurrence but not late recurrence.
During the blanking period following ablation, OSA promotes early recurrence through autonomic remodeling characterized by sympathetic overactivation and by exacerbating systemic inflammation and oxidative stress via intermittent hypoxia and intrathoracic pressure swings[6]. These factors collectively lead to atrial electrical instability. The occurrence of early recurrence holds a dual significance: it is both a consequence of atrial instability during the blanking period and a persistent pathological stimulus. It perpetuates atrial destabilization through the classic “AF begets AF” vicious cycle[31], thereby interfering with the normal healing of ablation lesions. Gottlieb et al. suggested that the ideal outcome post-ablation is the formation of a mature, rigid, and electrically inert collagen scar within the blanking period[32]. Such a scar provides lasting antiarrhythmic protection primarily by reducing local stretch, which underlies the mechanism of “delayed cure”[32]. Conversely, persistent early recurrence signals a dysregulated healing environment marked by ongoing autonomic imbalance and inflammation. This dysregulation may result in incomplete or aberrant repair, preventing the formation of a uniform, dense protective scar. Instead, it favors the development of a structurally heterogeneous and conductively disordered fibrotic substrate. Characterized by areas of slow conduction and conduction block, this substrate creates a highly favorable milieu for re-entrant arrhythmias[29]. Therefore, a potential continuum from this OSA risk profile to long-term AF recurrence may exist: OSA-related acute perturbations (e.g., autonomic imbalance) and chronic inflammation jointly contribute to early recurrence and adverse atrial healing, ultimately leading to the formation of a fibrotic substrate that facilitates AF maintenance. This study indicates that this risk profile primarily affects the initial link in this chain (predicting early recurrence), while late recurrence is influenced by multiple factors, most importantly the occurrence of early recurrence itself.
Notably, this predictive value was not observed in the redo ablation. The BQ-defined OSA risk showed no significant predictive value for early recurrence among patients who underwent redo ablation. We believe the primary reason is the limited sample size in the redo-ablation subgroup. This was particularly true for early recurrence events, which were very few (HR group 3 vs. LR group 5), making it difficult to detect a potential association. Furthermore, patients undergoing redo ablation represent a highly selected cohort with prior recurrence. Their atrial substrate may have been altered by the initial and redo ablations, which could also diminish the discriminatory power of the baseline OSA risk assessment. Therefore, the predictive value of the OSA risk profile assessed by BQ for redo ablation requires clarification through future large-scale studies.
Impact of hs-CRP on late AF recurrence
Beyond the well-recognized acute inflammatory response triggered by the ablation procedure itself, our findings highlight that pre-existing, chronic systemic inflammation, as reflected by elevated baseline hs-CRP, is an independent risk factor for late recurrence after the initial ablation. However, the predictive capacity for late recurrence after the redo ablation is limited. Previous studies have identified baseline hs-CRP as a biomarker indicative of AF recurrence following catheter ablation[33]. As a pivotal non-specific inflammatory marker, hs-CRP reflects the degree of systemic inflammation levels. Inflammation plays a substantial role in the mechanisms underpinning AF recurrence post-ablation, disrupting calcium homeostasis and connexin function. This disruption leads to changes in atrial electrophysiology and structural substrate, thereby augmenting vulnerability to AF triggers[34,35]. Additionally, during the chronic course of the disease, inflammation promotes fibroblast activation and proliferation through activation of macrophages and the secretion of various chemotactic factors, thereby mediating electrical and anatomical remodeling that promotes the initiation and maintenance of AF[36]. Therefore, in our initial 1-year follow-up analysis, hs-CRP did not demonstrate predictive value for AF recurrence, possibly because the follow-up period was relatively short and the remodeling effects of inflammation on the heart had not yet manifested. Moreover, the heightened inflammatory state in individuals with recurrent AF at baseline, coupled with a relatively modest sample size, may explain the absence of statistically significant differences in hs-CRP levels for late AF recurrence following redo ablation.
Clinical context of the findings
This study characterized the association between OSA risk profile assessed by BQ and outcomes after AF ablation in a cohort from which CPAP users were excluded. Consequently, our findings provide insights into the risk trajectory of untreated OSA. Whether CPAP can improve the AF ablation outcome requires independent research.
Limitations
The limitations of this study include: (1) The investigation is confined to a single center, and the sample size is relatively modest. The sample size was estimated in our previous study. However, this association should be explored in multicenter studies with a larger sample size; (2) Although PSG serves as the diagnostic tool for OSA, its prolonged testing duration may not align with the clinical demands of diagnosing OSA patients with AF. The BQ, a validated OSA screening tool, has demonstrated high accuracy in previous studies. However, BQ performance in patients with AF was not as good as in the general population; (3) The study exclusively enrolled paroxysmal AF patients. While this homogeneity strengthens internal validity, results may not generalize to populations with persistent AF; (4) The 16-year follow-up may involve unmeasured confounders. The OSA risk profile assessed by BQ was evaluated only at baseline, and new-onset or progressive OSA after enrollment—despite exclusion of CPAP users—remains uncaptured, potentially leading to residual confounding; (5) The study was conducted during a period when contact-force-guided catheter ablation was not widespread, potentially limiting its applicability to the current landscape where force-sensing catheter ablation for AF is more prevalent; (6) During this ultra-long-term follow-up, limitations of the follow-up protocol may have led to under-detection of asymptomatic AF, which could potentially affect the accurate estimation of between-group differences in late recurrence rates; (7) The redo-ablation analysis was limited by a relatively modest sample size, which reduced the statistical power of these subgroup comparisons. Therefore, the findings should be interpreted with caution and are considered exploratory.
Conclusion
This long-term study shows that the BQ-defined high OSA risk profile predicts early recurrence after AF ablation, but its association with late recurrence did not reach statistical significance. Baseline hs-CRP was an independent risk factor for late recurrence after the initial ablation; however, its predictive value is limited following a redo ablation. Furthermore, early recurrence is the strongest predictor of long-term outcome. These findings suggest that the BQ-defined OSA risk can be assessed before ablation to identify patients who may benefit from intensified follow-up within the first 3 months post-procedure. For long-term management, whether early recurrence occurs provides more meaningful guidance than assessing the OSA risk profile alone.
DECLARATIONS
Author contributions
Concept and design: Tang RB, Dong JZ, Ma CS
Material preparation, data collection, and analysis: Wu ZY, Long DY, Yu RH, Sang CH, Jiang CX, Wang W, Zhao X, Li CY, Jia CQ, Guo XY, Ning M, Feng L, Lv WH, Li YK, Wang XS, Liu XY, Du ZH
First draft of the manuscript: Wu ZY
Critical revision of the manuscript: Tang RB.
All authors read and approved the final manuscript.
Availability of data and materials
Raw data are available upon reasonable request to the corresponding author.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This study was supported by the National Natural Science Foundation of China (82570373); Zhong Nanshan Medical Foundation of Guangdong Province (ZNSA-2020017); and Beijing Xinlian Zhicheng Cardiovascular Health Public Welfare Foundation.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
The study was approved by the Institutional Review Board of Beijing Anzhen Hospital, Capital Medical University (Approval No. 2025314X). Due to the retrospective, observational nature of this long-term follow-up study and its designation as minimal-risk research, the ethics committee waived the requirement for written informed consent from participants.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Lippi G, Sanchis-Gomar F, Cervellin G. Global epidemiology of atrial fibrillation: an increasing epidemic and public health challenge. Int J Stroke 2021;16:217-21.
2. Du X, Guo L, Xia S, et al. Atrial fibrillation prevalence, awareness and management in a nationwide survey of adults in China. Heart 2021;107:535-41.
3. Chen Q, Xiong X, Li L, Gui C. Efficacy and safety of different forms of ablation therapies and antiarrhythmic drugs in atrial fibrillation: a systematic review and network meta-analysis of randomized clinical trials. J Thorac Dis 2025;17:1323-34.
4. Sánchez-de-la-Torre M, Lee CH, Barbé F. Obstructive sleep apnea and atrial fibrillation: we need to go step by step. J Clin Sleep Med 2021;17:869-70.
5. Linz D, Linz B, Hohl M, Böhm M. Atrial arrhythmogenesis in obstructive sleep apnea: therapeutic implications. Sleep Med Rev 2016;26:87-94.
6. Goudis CA, Ketikoglou DG. Obstructive sleep and atrial fibrillation: Pathophysiological mechanisms and therapeutic implications. Int J Cardiol 2017;230:293-300.
7. Szymanski FM, Filipiak KJ, Platek AE, et al. Presence and severity of obstructive sleep apnea and remote outcomes of atrial fibrillation ablations - a long-term prospective, cross-sectional cohort study. Sleep Breath 2015;19:849-56.
8. Chilukuri K, Dalal D, Marine JE, et al. Predictive value of obstructive sleep apnoea assessed by the Berlin Questionnaire for outcomes after the catheter ablation of atrial fibrillation. Europace 2009;11:896-901.
9. Zhai WX, Zhang TJ, Jia YH, et al. Effect of sleep apnea hypopnea syndro me on the prognosis of atrial fibrillation after radiofrequency catheter ablation. Chinese Circ J 2022;37:1144-9. (in Chinese).
10. Lee CJ, Kim TH, Park S, Pak HN. Obstructive sleep apnea is closely related to cardiovascular risk factors, but not to clinical recurrence of atrial fibrillation after catheter ablation: an analysis of atrial fibrillation patients. Pulse 2018;6:103-11.
11. Tang RB, Dong JZ, Liu XP, et al. Obstructive sleep apnoea risk profile and the risk of recurrence of atrial fibrillation after catheter ablation. Europace 2009;11:100-5.
12. Joglar JA, Chung MK, Armbruster AL, et al. 2023 ACC/AHA/ACCP/HRS guideline for the diagnosis and management of atrial fibrillation: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 2024;149:e1-156.
13. Chang JL, Goldberg AN, Alt JA, et al. International consensus statement on obstructive sleep apnea. Int Forum Allergy Rhinol 2023;13:1061-482.
14. Desteghe L, Hendriks JML, Heidbuchel H, Potpara TS, Lee GA, Linz D. Obstructive sleep apnoea testing and management in atrial fibrillation patients: a joint survey by the European Heart Rhythm Association (EHRA) and the Association of Cardiovascular Nurses and Allied Professions (ACNAP). Europace 2021;23:1677-84.
15. Netzer NC, Stoohs RA, Netzer CM, Clark K, Strohl KP. Using the Berlin questionnaire to identify patients at risk for the sleep apnea syndrome. Ann Intern Med 1999;131:485-91.
16. Desteghe L, Hendriks JML, McEvoy RD, et al. The why, when and how to test for obstructive sleep apnea in patients with atrial fibrillation. Clin Res Cardiol 2018;107:617-31.
17. Kadhim K, Middeldorp ME, Elliott AD, et al. Self-reported daytime sleepiness and sleep-disordered breathing in patients with atrial fibrillation: SNOozE-AF. Can J Cardiol 2019;35:1457-64.
18. de Heide J, Kock-Cordeiro DBM, Bhagwandien RE, et al. Impact of undiagnosed obstructive sleep apnea on atrial fibrillation recurrence following catheter ablation (OSA-AF study). Int J Cardiol Heart Vasc 2022;40:101014.
19. Matiello M, Nadal M, Tamborero D, et al. Low efficacy of atrial fibrillation ablation in severe obstructive sleep apnoea patients. Europace 2010;12:1084-9.
20. Zheng N, Fu Y, Xue F, Xu M, Ling L, Jiang T. Which ablation strategy is the most effective for treating persistent atrial fibrillation? A systematic review and Bayesian network meta-analysis of randomized controlled trials. Heart Rhythm 2025;22:e60-73.
21. Pranata R, Henrina J, Yonas E, et al. BMI and atrial fibrillation recurrence post catheter ablation: a dose-response meta-analysis. Eur J Clin Investig 2021;51:e13499.
22. Nalliah CJ, Wong GR, Lee G, et al. Sleep apnoea has a dose-dependent effect on atrial remodelling in paroxysmal but not persistent atrial fibrillation: a high-density mapping study. Europace 2021;23:691-700.
23. Ghias M, Scherlag BJ, Lu Z, et al. The role of ganglionated plexi in apnea-related atrial fibrillation. J Am Coll Cardiol 2009;54:2075-83.
24. Choi EK, Zhao Y, Everett TH 4th, Chen PS. Ganglionated plexi as neuromodulation targets for atrial fibrillation. J Cardiovasc Electrophysiol 2017;28:1485-91.
25. Yu L, Scherlag BJ, Sha Y, et al. Interactions between atrial electrical remodeling and autonomic remodeling: how to break the vicious cycle. Heart Rhythm 2012;9:804-9.
26. Schlögl S, Schlögl KS, Bengel P, et al. Contact force sensing manual catheter versus remote magnetic navigation ablation of atrial fibrillation: a single-center comparison. Heart Vessels 2024;39:427-37.
27. Alhuarrat MAD, Flomenbaum D, Schiavone M, et al. Ablation index-guided radiofrequency ablation for atrial fibrillation: an updated meta-analysis on its procedural efficacy and safety profiles. J Cardiovasc Electrophysiol 2025;36:2597-607.
28. Kaddoura R, Abushanab D, Asaad N. Pulsed-field ablation versus radiofrequency or cryoballoon thermal ablation in atrial fibrillation: a systematic review and meta-analysis. Postgrad Med J 2025;101:690-703.
29. De Becker B, El Haddad M, De Smet M, et al. Early atrial fibrillation recurrence post catheter ablation: Analysis from insertable cardiac monitor in the era of optimized radiofrequency ablation. Heart Rhythm 2024;21:521-9.
30. Kim YG, Boo KY, Choi JI, et al. Early recurrence is reliable predictor of late recurrence after radiofrequency catheter ablation of atrial fibrillation. JACC Clin Electrophysiol 2021;7:343-51.
31. Wijffels MC, Kirchhof CJ, Dorland R, Allessie MA. Atrial fibrillation begets atrial fibrillation. A study in awake chronically instrumented goats. Circulation 1995;92:1954-68.
32. Gottlieb LA, Dekker LRC, Coronel R. The blinding period following ablation therapy for atrial fibrillation: proarrhythmic and antiarrhythmic pathophysiological mechanisms. JACC Clin Electrophysiol 2021;7:416-30.
33. Carballo D, Noble S, Carballo S, et al. Biomarkers and arrhythmia recurrence following radiofrequency ablation of atrial fibrillation. J Int Med Res 2018;46:5183-94.
34. Korodi S, Toganel R, Benedek T, et al. Impact of inflammation-mediated myocardial fibrosis on the risk of recurrence after successful ablation of atrial fibrillation - the FIBRO-RISK study: protocol for a non-randomized clinical trial. Medicine 2019;98:e14504.
35. Hu YF, Chen YJ, Lin YJ, Chen SA. Inflammation and the pathogenesis of atrial fibrillation. Nat Rev Cardiol 2015;12:230-43.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].