Big data and AI for precision panvascular aging management and healthy longevity
Abstract
Panvascular aging-related diseases, including coronary artery disease, ischemic stroke, and peripheral artery disease, are leading global causes of death and disability, yet their management remains fragmented. Emerging technologies offer solutions to this challenge. Big data integration across imaging, multi-omics, wearables, and environmental exposures provides opportunities for cross-organ insights but faces issues of heterogeneity and privacy. Artificial intelligence enables early detection and refined risk prediction by recognizing subtle vascular changes and integrating biomarkers, though adoption is limited by interpretability and bias. Foundation models, through cross-modal learning, offer a unifying framework for mechanism discovery, personalized management, and digital twin applications. By linking technological innovation with clinical practice, these approaches can transform panvascular aging management and promote healthy longevity. Importantly, translating these innovations into policy and practice will be essential for advancing equitable vascular health and achieving population-level impact.
Keywords
INTRODUCTION
Panvascular aging-related diseases, such as coronary artery disease (CAD), ischemic stroke, and peripheral artery disease (PAD), are leading causes of mortality and long-term disability worldwide[1]. Global statistics consistently show the rising burden of cardiovascular and cerebrovascular conditions, yet the more pressing issue remains their fragmented management[2]. Clinical care for CAD, stroke, and PAD has traditionally been siloed within cardiology, neurology, and vascular surgery, respectively. This specialty-based separation emphasizes local lesions while overlooking the systemic and interconnected nature of vascular aging[3].
This compartmentalization often leads to missed opportunities for early detection and coordinated intervention. Chronic inflammation, metabolic dysregulation, and cellular senescence represent convergent mechanisms that cut across vascular territories, yet advances in one domain rarely inform strategies in another[4]. Currently, there is a lack of cross-system and cross-disciplinary strategies for prevention and intervention, leaving clinical practice unable to fully address the systemic challenges of vascular aging.
Emerging technologies - including big data, artificial intelligence (AI), and foundation models - provide a unique opportunity to overcome this fragmentation[5]. By integrating heterogeneous data sources and uncovering shared mechanisms, they offer the potential to reframe vascular aging from a set of isolated conditions into a unified target for precision prevention and holistic management.
BIG DATA AS THE FOUNDATION FOR PANVASCULAR AGING RESEARCH
The study of panvascular aging increasingly depends on the integration of multimodal data streams. Advanced imaging modalities such as magnetic resonance imaging (MRI), computed tomography (CT), and positron emission tomography (PET) provide structural and functional insights into vascular remodeling, cerebral white matter integrity, and atherosclerotic plaque burden[6]. At the molecular level, large-scale profiling of the genome, transcriptome, proteome, and metabolome reveals age-related alterations in signaling pathways and metabolic networks[7]. These biological data are complemented by longitudinal clinical records, wearable devices that capture blood pressure, heart rhythm, or physical activity in real time, and environmental datasets that characterize exposures such as air pollution and temperature[8].
The importance of big data lies not simply in the accumulation of large datasets, but in their capacity to reveal cross-organ and cross-population patterns of vascular aging that remain invisible in traditional single-center studies. For example, analyses from the UK Biobank have identified sex-specific trajectories of carotid atherosclerosis and brain white matter changes, linking cardiovascular and cerebrovascular aging in a way that would be difficult to capture without harmonized, population-level data[9]. Similarly, initiatives such as the Alzheimer’s Disease Neuroimaging Initiative (ADNI) and the China Kadoorie Biobank demonstrate how integrating imaging, genetics, and longitudinal follow-up enables the discovery of novel biomarkers that inform risk prediction across vascular territories[10,11].
Despite this promise, significant challenges remain. Data are often collected under heterogeneous protocols and stored in isolated repositories, limiting reproducibility and large-scale validation[6,12]. Ethical and privacy concerns add further barriers, particularly for genetic information and continuously collected wearable data[13]. These obstacles underscore the importance of developing interoperable data platforms and federated learning frameworks that enable secure data sharing without compromising patient privacy.
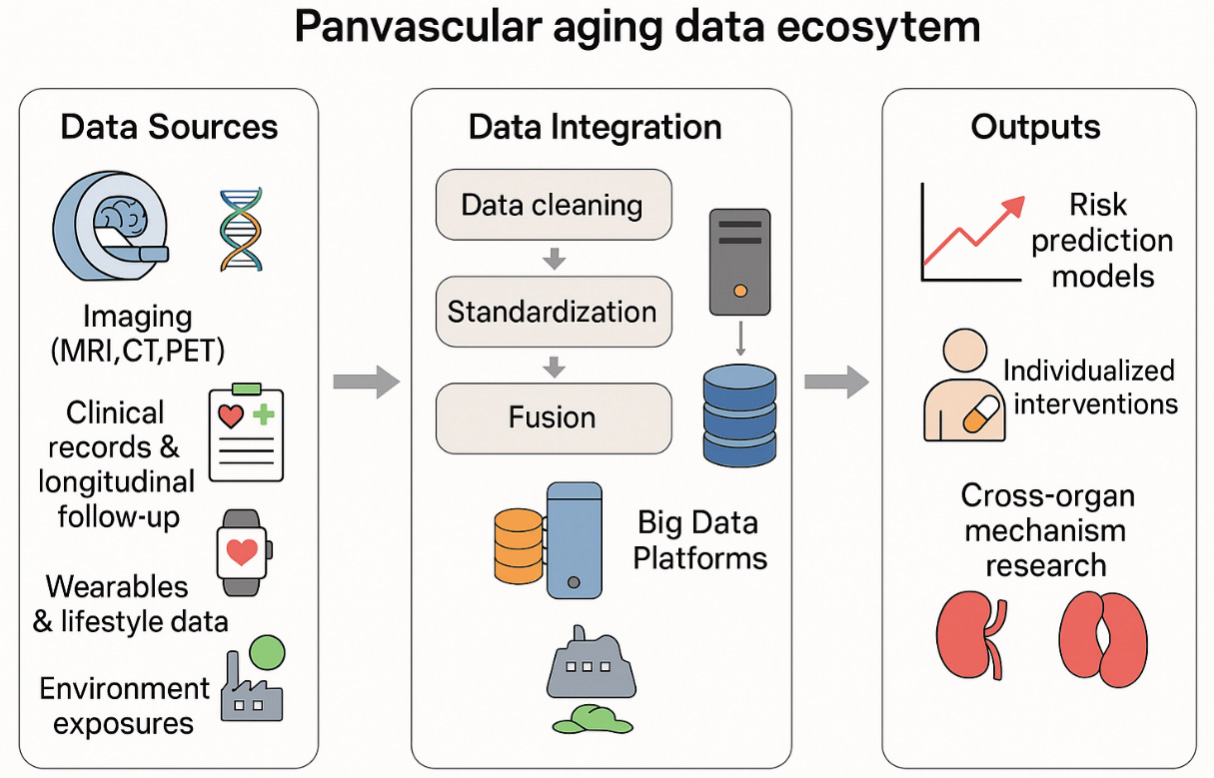
By establishing big data as the foundation of panvascular aging research, the field can integrate multimodal information, encompass large-scale populations, and uncover cross-organ mechanisms. This approach enables a shift from fragmented, organ-specific paradigms toward a more comprehensive and equitable understanding of vascular health across populations[8]. As illustrated in Figure 1, multi-source inputs - such as imaging, multi-omics, clinical follow-up, wearable monitoring, and environmental exposures - can be standardized and integrated into big data platforms. This integration enables risk prediction, individualized interventions, and the discovery of cross-organ mechanisms.
Figure 1. Panvascular aging data ecosystem: a multi-source integration and application framework. Data sources (left) include imaging (MRI, CT, PET), multi-omics (genomics, proteomics, metabolomics), clinical records & longitudinal follow-up, wearables & lifestyle data, and environmental exposures. The integration layer (center) performs data cleaning, standardization, and multimodal fusion into Big Data Platforms. Outputs (right) comprise risk prediction models, individualized interventions, and cross-organ mechanism research. Arrows indicate the direction of data flow and hierarchical aggregation. MRI: Magnetic resonance imaging; CT: computed tomography; PET: positron emission tomography.
ARTIFICIAL INTELLIGENCE FOR EARLY DETECTION AND RISK PREDICTION
AI is emerging as a transformative tool in vascular medicine, particularly for the early detection of age-related vascular changes[14]. Imaging-based AI algorithms have shown remarkable capacity to identify subtle features of vascular aging that may be overlooked by conventional assessments. For instance, deep learning systems can detect micro-plaques within coronary arteries or white matter hyperintensities in the brain, both of which serve as early indicators of systemic vascular decline[15,16]. By recognizing these subclinical signatures, AI enables earlier intervention and creates opportunities to prevent irreversible damage.
Beyond imaging, AI excels in integrating diverse data types to build robust risk prediction models. Traditional cardiovascular risk scores rely on a limited set of variables, such as blood pressure, cholesterol, and smoking history. In contrast, machine learning approaches can combine imaging biomarkers, genomic data, proteomic signatures, lifestyle factors, and longitudinal clinical records to generate individualized risk profiles[17]. Such models can refine patient stratification, distinguish between rapid and slow progressors, and predict adverse outcomes with greater precision than conventional tools.
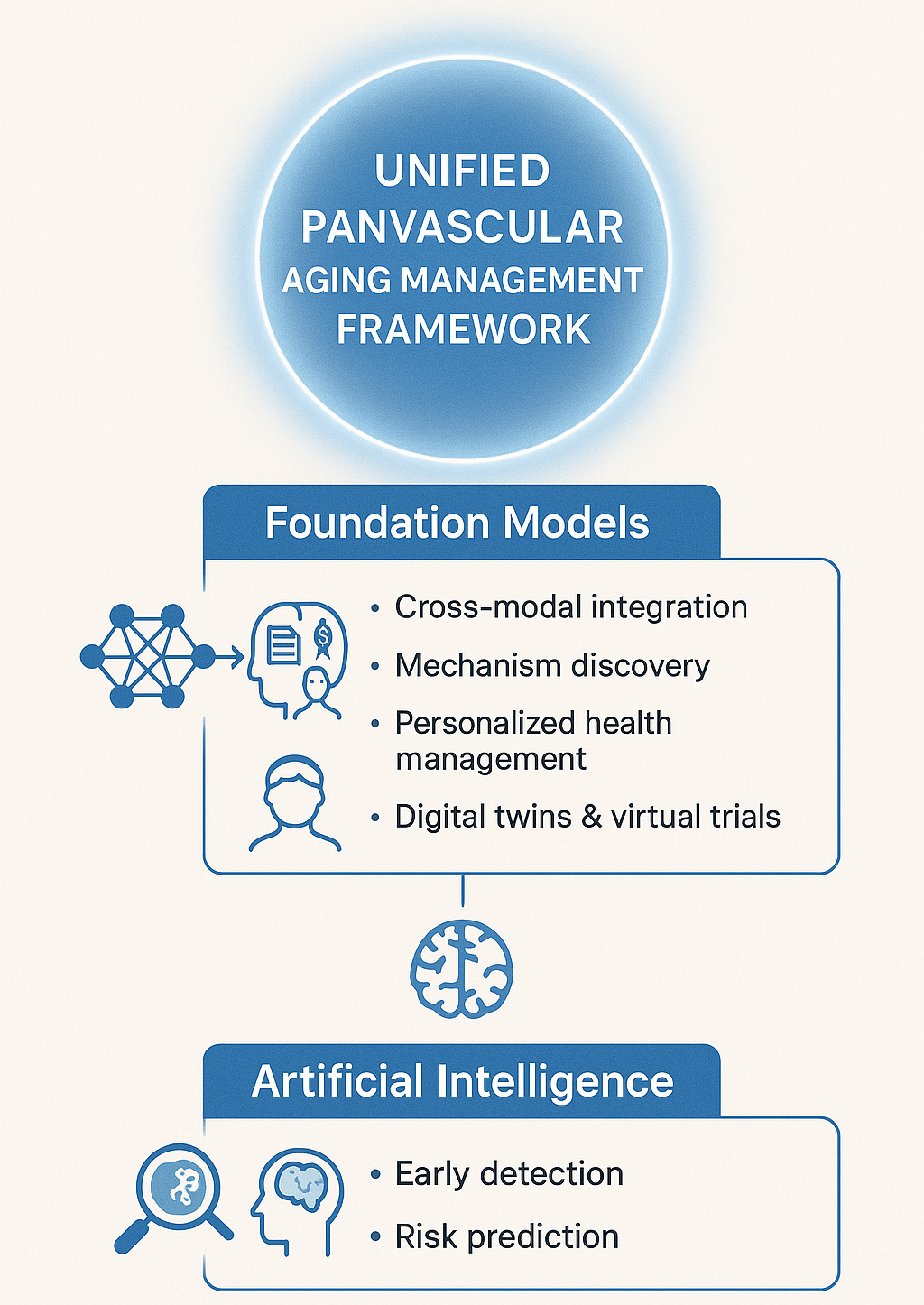
Nevertheless, several barriers remain. A central concern is the limited interpretability of many algorithms, which undermines clinician trust and slows adoption in practice. In addition, biases arising from underrepresentation of women, ethnic minorities, or low-resource populations threaten the fairness and generalizability of AI applications. Recent progress in explainable AI (XAI) methods, fairness-aware model design, and federated learning frameworks provides a pathway to address these challenges. These approaches offer more transparent, equitable, and clinically reliable tools for vascular aging research and management[14]. Building on these foundations, Figure 2 illustrates the progressive roles of AI and foundation models: AI algorithms enable early detection of subclinical vascular alterations and individualized risk prediction, while foundation models extend these capabilities through cross-modal integration, mechanism discovery, personalized health management, and digital twin applications, ultimately converging into a unified panvascular aging management framework.
Figure 2. Artificial intelligence and foundation models driving unified panvascular aging management. Artificial intelligence (AI) enables early detection of subclinical vascular alterations, such as micro-plaques and white matter hyperintensities, and provides individualized risk prediction models.
BIG MODELS TO UNIFY SYSTEMIC VASCULAR AGING MANAGEMENT
Foundation models represent the next frontier in applying AI to vascular medicine[18-20]. Unlike conventional AI systems trained for narrow tasks, these models are capable of cross-modal learning, simultaneously integrating text, imaging, and multi-omics data. Such capacity allows them to generate a “universal language” of vascular aging, bridging cardiovascular, cerebrovascular, and peripheral vascular research that has traditionally evolved in isolation.
Several breakthroughs can be anticipated from this paradigm. First, foundation models are well-positioned to uncover convergent mechanisms that cut across organ systems, including chronic inflammation, dysregulated nicotinamide adenine dinucleotide (NAD+) metabolism, and cellular senescence pathways[21]. Second, by leveraging longitudinal and individualized data, these models could inform personalized health management strategies, predicting trajectories of healthspan versus disease span[22]. Third, foundation models may serve as the backbone for digital twin frameworks - virtual representations of patients that allow simulation of disease progression, testing of therapeutic strategies, optimization of treatment pathways, and even in-silico clinical trial design[18].
Importantly, early initiatives have already provided proof of concept. For instance, the European Union (EU) Horizon Digital Twin projects are developing cardiovascular and cerebrovascular twins to simulate disease dynamics and evaluate preventive strategies. Meanwhile, pilot programs in heart failure management demonstrate the feasibility of tailoring therapy through patient-specific virtual models[23-25]. In parallel, domain-specific language models such as BioGPT (Biomedical Generative Pretrained Transformer) and Med-PaLM (Medical Pathways Language Model) show strong performance in processing biomedical literature and clinical narratives, highlighting the potential of foundation models to integrate knowledge across domains[26,27].
Nonetheless, challenges remain substantial. Effective deployment will require close collaboration between clinicians, data scientists, and public health experts to ensure clinical relevance and equity of access. Ethical and regulatory concerns - such as data privacy, algorithmic bias, and accountability - must be carefully addressed. Regulatory agencies including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) are actively exploring frameworks for AI and foundation model oversight, focusing on transparency, continuous monitoring, and risk-benefit assessment[28]. Without such governance, there is a risk that foundation models could amplify disparities or propagate errors on a large scale.
In sum, foundation models offer transformative opportunities to integrate knowledge, reveal shared mechanisms, and support individualized management of panvascular aging. Their successful adoption will depend not only on technical advances but also on robust ethical safeguards and adaptive regulatory frameworks, ensuring that these tools contribute meaningfully to both scientific discovery and the promotion of healthy longevity.
FUTURE DIRECTIONS
Looking ahead, several priorities should guide the field. First, the establishment of multicenter shared databases and standardized frameworks is essential to ensure data quality, interoperability, and inclusivity across populations. Second, advancing the interpretability and clinical validation of AI models will be crucial to building trust and ensuring safe, equitable adoption in practice. Third, foundation models should be leveraged to probe cross-organ mechanisms and to design interventions aimed not only at preventing disease but also at extending healthspan.
CONCLUSION
Big data, AI, and foundation models collectively represent a critical turning point for panvascular aging research and management. By addressing the limitations of fragmented, specialty-based approaches, these technologies provide the tools to unify mechanistic insights, enhance early risk prediction, and foster integrative clinical translation. Ultimately, the promise of these technologies lies in their capacity to bridge innovation with medical practice. By moving beyond the traditional focus on survival to embrace the goal of healthy longevity, panvascular aging research can help shift the paradigm from simply prolonging life to enhancing the quality of life in aging populations.
DECLARATIONS
Authors’ contributions
Contributed to conceptualization, literature review, and drafting of the manuscript: Geng C
Contributed to conceptualization, critical revision of the manuscript for important intellectual content, supervision, and final approval of the version to be published: Tang Y
All authors read and approved the final manuscript.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
Not applicable.
Conflicts of interest
Tang Y is a Guest Editor of the Special Topic “Panvascular Aging”. Tang Y was not involved in any steps of editorial processing, notably including reviewer selection, manuscript handling, or decision-making. The other authors declare that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Zhou X, Yu L, Zhao Y, Ge J. Panvascular medicine: an emerging discipline focusing on atherosclerotic diseases. Eur Heart J. 2022;43:4528-31.
2. Guo W, Yi X. Advancements and future prospects in the study of panvascular disease. Clin Hemorheol Microcirc. 2025;89:198-204.
3. Hu Y, Zhao Y, Dai N, Lu H, Ge J. Unwavering excellence: how to be a competent cardiovascular doctor in “panvascular medicine +”. Innovation. 2023;4:100489.
4. Hu Y, Zhao Y, Li P, Lu H, Li H, Ge J. Hypoxia and panvascular diseases: exploring the role of hypoxia-inducible factors in vascular smooth muscle cells under panvascular pathologies. Sci Bull. 2023;68:1954-74.
5. Hu Y, Zhou Y, Dai N, et al. Enhancing panvascular medicine: unveiling the nexus of pan-cardio-oncology and expanding therapeutic frontiers. Sci Bull. 2025;70:798-800.
6. Yousefi B, Khansari M, Trask R, et al. Measuring subtle HD data representation and multimodal imaging phenotype embedding for precision medicine. IEEE Trans Instrum Meas. 2025;74:4503911.
7. Nam Y, Kim J, Jung SH, et al. Harnessing artificial intelligence in multimodal omics data integration: paving the path for the next frontier in precision medicine. Annu Rev Biomed Data Sci. 2024;7:225-50.
8. Ran X, Shi J, Chen Y, Jiang K. Multimodal neuroimage data fusion based on multikernel learning in personalized medicine. Front Pharmacol. 2022;13:947657.
9. Glynn P, Greenland P. Contributions of the UK biobank high impact papers in the era of precision medicine. Eur J Epidemiol. 2020;35:5-10.
10. Arai H, Okamura N, Furukawa K, Kudo Y. Geriatric medicine, Japanese Alzheimer’s disease neuroimaging initiative and biomarker development. Tohoku J Exp Med. 2010;221:87-95.
11. Huang R, Kartsonaki C, Turnbull I, et al. Incidence and mortality rates of 14 site-specific infectious diseases in 10 diverse areas of China: findings from China Kadoorie Biobank, 2006-2018. Int J Infect Dis. 2024;147:107169.
12. Braunack-Mayer A, Carolan L, Street J, Ha T, Fabrianesi B, Carter S. Ethical issues in big data: a qualitative study comparing responses in the health and higher education sectors. PLoS ONE. 2023;18:e0282285.
13. Al-Ani A, Rayyan A, Maswadeh A, et al. Evaluating the understanding of the ethical and moral challenges of big data and AI among Jordanian medical students, physicians in training, and senior practitioners: a cross-sectional study. BMC Med Ethics. 2024;25:18.
14. Rijken L, Zwetsloot S, Smorenburg S, et al. Developing trustworthy artificial intelligence models to predict vascular disease progression: the VASCUL-AID-RETRO study protocol. J Endovasc Ther. 2025:15266028251313963.
15. Sinitca AM, Lyanova AI, Kaplun DI, et al. Microscopy image dataset for deep learning-based quantitative assessment of pulmonary vascular changes. Sci Data. 2024;11:635.
16. Lee GY, Choi YH, Kim D, et al. Deep learning analysis of white matter hyperintensity and its association with comprehensive vascular factors in two large general populations. J Imaging Inform Med. 2025;38:2761-78.
17. Skovbo JS, Obel LM, Lindholt JS. Artificial intelligence in vascular surgery: bridging technology and clinical practice. J Vasc Surg. 2025;81:1232-3.
18. Ranisch R, Haltaufderheide J. Foundation models in medicine are a social experiment: time for an ethical framework. NPJ Digit Med. 2025;8:525.
19. Khan W, Leem S, See KB, Wong JK, Zhang S, Fang R. A comprehensive survey of foundation models in medicine. IEEE Rev Biomed Eng. 2026;19:283-304.
20. Alsaedi S, Gao X, Gojobori T. Beyond digital twins: the role of foundation models in enhancing the interpretability of multiomics modalities in precision medicine. FEBS Open Bio. 2025;15:1192-208.
21. Silva-Rodríguez J, Dolz J, Ben Ayed I. Towards foundation models and few-shot parameter-efficient fine-tuning for volumetric organ segmentation. Med Image Anal. 2025;103:103596.
22. Ramprasad P, Pai N, Pan W. Enhancing personalized gene expression prediction from DNA sequences using genomic foundation models. HGG Adv. 2024;5:100347.
23. Laleci Erturkmen GB, Yuksel M, Sarigul B, Lilja M, Chen R, Arvanitis TN. Personalised care plan management utilizing guideline-driven clinical decision support systems. Stud Health Technol Inform. 2018;247:750-754.
24. Harrisson S, Myers H, Wynne-Jones G, et al. Clinical and cost-effectiveness of a personalised guided consultation versus usual physiotherapy care in people presenting with shoulder pain: a protocol for the PANDA-S cluster randomised controlled trial and process evaluation. BMJ Open. 2025;15:e100501.
25. Guan G, Michel K, Corke C, Ranmuthugala G. A clinical prediction model for personalised emergency department discharge decisions for residential care facility residents post-fall. J Pers Med. 2025;15::332.
26. Yuan Z, Zhang S, Zhang H, Xie P, Jia Y. Optimized drug-drug interaction extraction with BioGPT and focal loss-based attention. IEEE J Biomed Health Inform. 2025;29:4560-70.
27. Luo R, Sun L, Xia Y, et al. BioGPT: generative pre-trained transformer for biomedical text generation and mining. Brief Bioinform. 2022;23::bbac409.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].