Triglyceride-Glucose-Waist-to-Height Ratio links to aging and mortality in cardiovascular-kidney-metabolic syndrome
Abstract
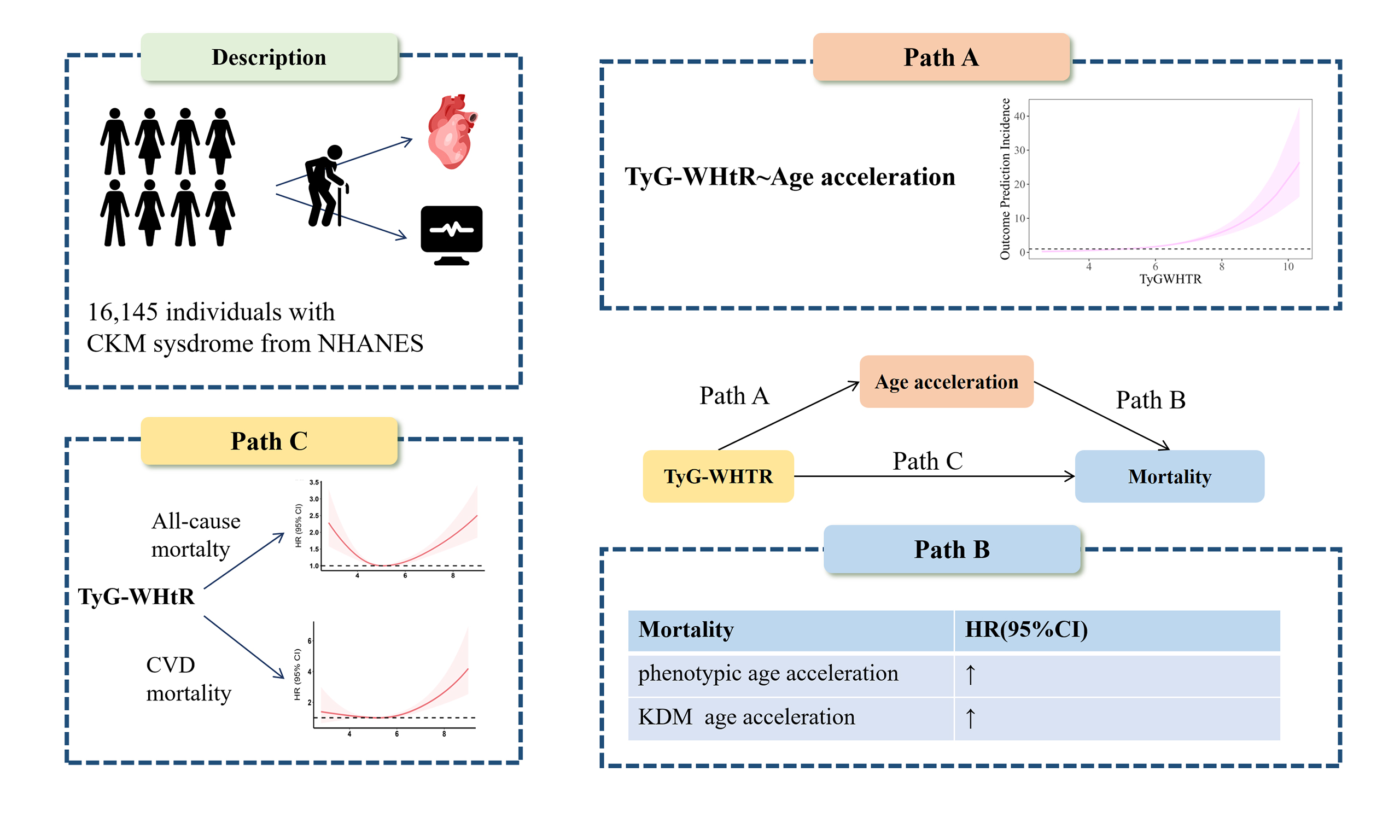
Aim: Cardiovascular-kidney-metabolic (CKM) syndrome accelerates aging and increases mortality. We evaluated whether Triglyceride-Glucose-Waist-to-Height Ratio (TyG-WHtR), a marker of insulin resistance, predicts mortality in CKM and whether age acceleration mediates this association.
Methods: This investigation enrolled 16,145 individuals diagnosed with CKM syndrome. The relationships between TyG-WHtR and mortality were examined using multivariable Cox proportional hazards models, threshold effect analysis, and restricted cubic splines. Besides, the mediating role of age acceleration was investigated through mediation analysis. Risk within different populations was assessed using interaction tests and subgroup analysis.
Results: In multivariate Cox proportional hazards analyses, TyG-WHtR showed a positive association with mortality outcomes. Per one standard deviation (1-SD) increase, the hazard of cardiovascular death was 18% higher (hazard ratio (HR): 1.180; 95% confidence interval (CI): 1.08-1.29), while all-cause mortality risk rose by 8.6% (HR: 1.086, 95%CI: 1.01-1.17). Both cardiovascular and all-cause mortality showed a strong U-shaped association with TyG-WHtR. Mediation analysis revealed that PhenoAge acceleration and Klemera-Doubal Method age acceleration mediated 19.7% and 15.8% of the association between TyG-WHtR and all-cause mortality, respectively, and 20.7% and 16.8% of the association between TyG-WHtR and cardiovascular mortality, respectively.
Conclusion: TyG-WHtR is associated with mortality in patients with CKM syndrome, partly through accelerated aging. It is positively associated with age acceleration and exhibits a U-shaped relationship with mortality. Targeting the metabolic-aging crosstalk may help reduce mortality in CKM patients.
Keywords
INTRODUCTION
The Cardiovascular-Kidney-Metabolic (CKM) syndrome is a pan-vascular disease arising from the interrelated presence of metabolic risk factors, cardiovascular disease (CVD), and chronic kidney disease (CKD)[1]. As a pan-vascular condition, it promotes multi-organ dysfunction and accelerates the aging process, thereby amplifying the risk of adverse cardiovascular outcomes and imposing significant burden on global healthcare systems and economies[1,2]. Therefore, analyzing the mortality risk factors in CKM syndrome from the perspective of metabolic-aging interactions is essential for risk classification, intervention, and improved prognosis.
The gradual reduction in system integrity and physiological function over time is known as aging, which increases susceptibility to illnesses and increases mortality risk[3]. Age acceleration, which is measured by the gap between biological age and chronological age (CA), is a reliable indicator of illness and premature death[4]. The use of Klemera-Doubal Method age acceleration (KDM-AA) and PhenoAge acceleration (PAA) indices provides well-validated measures of aging, allowing for a comprehensive assessment of age acceleration and offering precise estimates of the aging process[5].
One of the main factors for vascular and multi-organ aging has been identified as insulin resistance (IR). It not only accelerates cellular senescence and disrupts tissue homeostasis through interconnected molecular pathways but also contributes to myocardial metabolic abnormalities, cardiac remodeling, vascular aging, and the progression of atherosclerosis[6]. Moreover, IR increases susceptibility to metabolic disorders, thereby fostering the development of pan-vascular diseases[7]. Additionally, evidence suggests that combining the Triglyceride-Glucose (TyG) index with obesity-related measures, such as the Triglyceride-Glucose-Waist Height Ratio (TyG-WHtR), provides superior prediction of patient survival and improved assessment of IR and CVD risk[5,8]. To ascertain how age acceleration mediates the association between TyG-WHtR and mortality risk in the CKM cohort, more research is necessary.
Therefore, considering the importance of vascular aging, this study examines the association between TyG-WHtR, age acceleration, and mortality using data from the National Health and Nutrition Examination Survey (NHANES).
METHODS
Study population
NHANES is a thorough, representative investigation aimed at collecting health-related information from the American population that is not residing in institutions. Data collection included structured in-home interviews, comprehensive health evaluations conducted at mobile examination centers, and laboratory testing of biological samples. NHANES was approved by the Research Ethics Review Board of the National Center for Health Statistics (NCHS), Centers for Disease Control and Prevention (CDC). Each subject provided informed written consent prior to inclusion in NHANES. We examined NHANES data spanning 1999 to 2018, focusing on participants with CKM at enrollment. The exclusion criteria were as follows: (1) no diagnosis of CKM syndrome; (2) pregnancy; (3) insufficient data for TyG-WHtR or aging; (4) lack of survival data. The selection procedure for the 16,145 patients with CKM syndrome is shown in Supplementary Figure 1. In addition, we performed further validation in the UK Biobank. The UK Biobank was approved by the North West Haydock Research Ethics Committee (REC reference: 21/NW/0157).
Definitions of TyG-WHtR index
The TyG-WHtR index was calculated as TyG × [waist circumference (cm)/height (cm)]. Following a 9-h fast, fasting lipid and glucose levels were assessed. Participants’ height and waist circumference were measured by trained professionals at a mobile examination center.
Definition of biological age acceleration
KDM-AA and PAA were calculated as two measures of age-related acceleration.
Following the method of Levine et al.[9], biomarkers including alkaline phosphatase, albumin, creatinine, glucose, C-reactive protein (CRP), white blood cell count, mean cell volume, red blood cell distribution width, lymphocyte percentage and CA were used to assess PhenoAge (PA) and KDM. The biological ages were constructed using the R package BioAge (https://github.com/dayoonkwon/BioAge)[9,10]. Age acceleration is defined as a binary variable when either KDM or PAA exceeds CA[11]. Supplementary Table 1 lists the specific procedures for determining biological ages and age acceleration.
Definition of CKM
The stage of CKM syndrome was determined according to established criteria[12,13]. Participants were categorized as follows: Stage 0, no identifiable CKM-related risk factors; Stage 1, presence of obesity or prediabetes; Stage 2, presence of CKD or other metabolic diseases; Stage 3, subclinical CVD equivalents as defined by the American Heart Association’s (AHA) PREVENT (Predicting Risk of cardiovascular disease EVENTs) equations; and Stage 4, clinically manifest CVD.
The estimated glomerular filtration rate (eGFR), which was determined by applying the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation and the urine albumin/creatinine ratio, served as the foundation for the CKD classification system[14].
The detailed methods for categorizing CKM are provided in Supplementary Table 2.
Covariates
Covariate selection was guided by prior research and statistical techniques, guaranteeing that variables that might have an effect on the result were carefully taken into account. From the NHANES survey data, we collected information on demographic and socioeconomic indicators (participant age, gender, race, education, and Poverty Income Ratio (PIR)), together with smoking status, alcohol-drinking behaviors, prior disease history, and medication use. Additionally, NHANES evaluated various clinical laboratory biomarkers, such as total cholesterol (TC), fasting insulin, fasting plasma glucose (FPG), triglycerides (TG), high-density lipoprotein (HDL), low-density lipoprotein (LDL), and CRP. Hypertension was defined as meeting at least one of the following criteria: (1) self-reported hypertension; (2) three consecutive blood pressure readings with a systolic blood pressure of ≥ 140 mmHg and/or a diastolic blood pressure of ≥ 90 mmHg; or (3) current usage of antihypertensive medication[15]. Self-reported diagnosis of diabetes, usage of insulin or oral hypoglycemic drugs, hemoglobin A1c (HbA1c) levels of ≥ 6.5% or FPG levels of ≥ 126 mg/dL were among the criteria used by the American Diabetes Association to diagnose diabetes[16].
Mortality data
The study outcomes focused on cardiovascular and all-cause mortality in the CKM population. While cardiovascular mortality referred to fatalities from ICD-10 (10th Revision of the International Classification of Diseases) codes I00-I09, I11, I13, I20-I51, and I60-I69, all-cause mortality indicated death from any cause[17]. NHANES mortality data were linked to the National Death Index using probabilistic record linkage, with deaths ascertained through December 31, 2019.
Statistical analysis
To address unequal sampling probabilities and nonresponse, NHANES-recommended fasting subsample weights were applied to all participants. Participants were categorized into TyG-WHtR index tertiles to compare the baseline attributes. Categorical variables were described using weighted frequency percentages, whereas continuous variables were described using weighted averages and standard errors.
We used multivariate Cox proportional hazards models to examine the association between the TyG-WHtR index and mortality, adjusting for confounders. Covariates were selected based on statistical criteria and prior studies, and those with a variance inflation factor > 10 were excluded to avoid multicollinearity. Hazard ratio (HR) was calculated for three models: Model 1 is a baseline model without any adjustments. Demographic factors such as age, gender, race, PIR, marital status, and educational attainment were taken into account while adjusting Model 2. Model 3 was built on Model 2 by including additional variables such as smoking, alcohol use, body mass index (BMI), blood pressure, medication use, and family history of CVD and diabetes. To investigate possible non-linear associations between IR indices and mortality outcome, restricted cubic splines (RCS) were applied. Once non-linearity was confirmed, thresholds were estimated using the method of maximum likelihood estimation. Two-piecewise Cox proportional hazards models were then built in order to assess the correlation between IR indicators and mortality. Also, we examined the association between biological age acceleration and TyG-WHtR and between age-related acceleration and mortality.
We used mediation analysis to determine whether biological age acceleration mediated the relationship between TyG-WHtR and mortality. The study used 1,000 bootstrap samples to estimate the size of the indirect path effect, the fraction of the mediation effect, and its P-value.
Finally, we performed interaction tests and stratified analyses by ethnicity, BMI, gender, and age. To strengthen the validity of our findings, participants who died during the two-year follow-up were excluded.
Data interpolation was not performed because the PREVENT equation does not apply to interpolated data[13]. The threshold for statistical significance was set at 0.05 (P < 0.05). All statistical analyses were conducted using R software (version 4.4.1).
RESULTS
Baseline characteristics of study participants
A total of 16,145 participants were included in our study, and their TyG-WHtR index was used to categorize them into three groups [Table 1]. Participants with higher TyG-WHtR values were disproportionately non-Hispanic White, married, or cohabiting, and also had higher prevalence rates of hypertension, diabetes, and a family history of CVD. They exhibited more adverse metabolic profiles, with lower LDL levels and markedly higher HbA1c, CRP, TC, and TG. Mortality rates and age acceleration also increased with higher TyG-WHtR values.
Weighted participant characteristics stratified by categories of the TyG-WHtR index
| Characteristics | Overall | TyG-WHtR T1 | TyG-WHtR T2 | TyG-WHtR T3 | P-value |
| Age, years, mean (SD) | 46.75 ± 16.62 | 40.74± 15.65 | 49.10 ± 16.26 | 51.94 ± 15.84 | < 0.001 |
| Sex, (%) | < 0.001 | ||||
| Male | 49.47 | 46.78 | 55.78 | 46.03 | |
| Female | 50.53 | 53.22 | 44.23 | 53.97 | |
| Race, (%) | < 0.001 | ||||
| Mexican American | 7.99 | 5.64 | 9.25 | 9.64 | |
| Other Hispanic | 5.26 | 4.49 | 5.97 | 5.48 | |
| Non-Hispanic white | 69.86 | 70.58 | 68.57 | 70.35 | |
| Non-Hispanic black | 10.36 | 11.59 | 9.74 | 9.46 | |
| Other race | 6.52 | 7.70 | 6.46 | 5.06 | |
| Marital status, (%) | < 0.001 | ||||
| Not married | 34.43 | 38.09 | 31.01 | 33.47 | |
| Married/living with partner | 65.57 | 61.91 | 68.99 | 66.54 | |
| Education, (%) | < 0.001 | ||||
| Lower than high school | 17.08 | 13.45 | 18.05 | 20.72 | |
| High school | 25.23 | 21.81 | 26.80 | 27.92 | |
| Higher than high school | 57.69 | 64.74 | 55.15 | 51.37 | |
| Poverty income ratio, (%) | < 0.001 | ||||
| ≤ 1.30 | 19.11 | 17.44 | 18.23 | 22.21 | |
| 1.3 to ≤ 3.5 | 37.32 | 34.98 | 37.71 | 39.90 | |
| > 3.5 | 43.57 | 47.58 | 44.06 | 37.89 | |
| BMI, (kg/m2) | 28.56 ± 6.49 | 23.45 ± 3.06 | 28.45 ± 3.46 | 35.28 ± 6.21 | < 0.001 |
| Smoking status, (%) | < 0.001 | ||||
| Nonsmoker | 52.35 | 55.45 | 51.96 | 48.77 | |
| Former smoker | 25.91 | 20.31 | 26.72 | 32.24 | |
| Current smoker | 21.74 | 24.24 | 21.32 | 18.99 | |
| Drinking status, (%) | 0.009 | ||||
| Nondrinker | 25.94 | 20.90 | 25.72 | 33.36 | |
| Light to moderate drinker | 10.21 | 9.50 | 11.75 | 9.43 | |
| Heavy drinker | 63.85 | 69.60 | 62.53 | 57.22 | |
| Hypertension, (%) | 35.96 | 18.33 | 38.17 | 56.28 | < 0.001 |
| Diabetes, (%) | 11.77 | 2.31 | 8.84 | 27.18 | < 0.001 |
| Antihypertensive agents, (%) | 21.21 | 8.16 | 21.27 | 37.97 | < 0.001 |
| Hypoglycemic agents, (%) | 7.36 | 1.32 | 5.29 | 17.17 | < 0.001 |
| Antihyperlipidemic agents, (%) | 44.27 | 27.20 | 45.47 | 55.22 | < 0.001 |
| family history of CVD, (%) | 14.35 | 12.04 | 13.12 | 18.70 | < 0.001 |
| SBP, mmHg, mean (SD) | 121.95 ± 17.36 | 116.14 ± 15.38 | 123.57 ± 17.20 | 127.67 ± 17.65 | < 0.001 |
| DBP, mmHg, mean (SD) | 70.68 ± 12.12 | 68.74 ± 10.76 | 71.34 ± 12.28 | 72.46 ± 13.19 | < 0.001 |
| HbA1c, % | 5.51 ± 0.74 | 5.24 ± 0.42 | 5.49 ± 0.61 | 5.90 ± 0.99 | < 0.001 |
| eGFR, mL/min/1.73 m2 | 95.41 ± 38.58 | 100.15 ± 38.11 | 90.45 ± 37.55 | 94.74 ± 39.55 | < 0.001 |
| Creatinine, mg/dL | 0.86 ± 0.21 | 0.84 ± 0.19 | 0.87 ± 0.22 | 0.86 ± 0.23 | < 0.001 |
| CRP, mg/dL | 1.22 ± 3.04 | 0.57 ± 1.82 | 1.16 ± 2.72 | 2.11 ± 4.19 | < 0.001 |
| TC, mg/dL | 195.80 ± 40.51 | 185.71 ± 36.08 | 201.65 ± 41.12 | 202.39 ± 42.59 | < 0.001 |
| TG, mg/dL | 130.28 ± 100.12 | 82.61 ± 39.20 | 130.96 ± 68.04 | 191.041 ± 142.32 | < 0.001 |
| HDL, mg/dL | 53.66 ± 16.17 | 60.66 ± 16.68 | 52.13 ± 15.22 | 46.32 ± 12.39 | < 0.001 |
| LDL,mg/dL | 116.56 ± 34.99 | 108.55 ± 32.05 | 123.42 ± 35.91 | 119.62 ± 35.58 | < 0.001 |
| TyG-WHtR | 5.02 ± 1.01 | 4.03± 0.40 | 5.06 ± 0.25 | 6.26 ± 0.64 | < 0.001 |
| PhenoAge | 43.68 ± 17.36 | 36.09 ± 15.86 | 46.12 ± 16.56 | 50.82 ± 16.24 | < 0.001 |
| PAA,% | 22.73 | 12.07 | 21.55 | 37.76 | < 0.001 |
| KDM | 41.70 ± 18.56 | 32.68 ± 15.68 | 43.35 ± 17.40 | 51.54 ± 17.69 | < 0.001 |
| KDM-AA,% | 34.60 | 25.44 | 33.19 | 47.95 | < 0.001 |
| All-cause mortality, (%) | 11.28 | 6.90 | 10.03 | 12.87 | < 0.001 |
| Cardiovascular mortality, (%) | 3.33 | 1.94 | 2.91 | 3.74 | < 0.001 |
Relationship between TyG-WHtR and survival outcomes
Results from Model 3 showed that elevated TyG-WHtR was linked to higher hazards of both mortality when TyG-WHtR was presented as an ongoing variable (per standard deviation (SD)). For every one standard deviation (1-SD) increase in TyG-WHtR, the risk of cardiovascular death rose by 18% (HR: 1.180, 95% confidence interval (CI): 1.08-1.29) and the risk of all-cause death climbed by 8.6% (HR: 1.086, 95%CI: 1.01-1.17) [Supplementary Table 3]. These findings show that, even after thorough adjustment for demographic, clinical, and treatment characteristics, TyG-WHtR, a marker of IR, is independently and substantially linked with mortality risk in people with CKM.
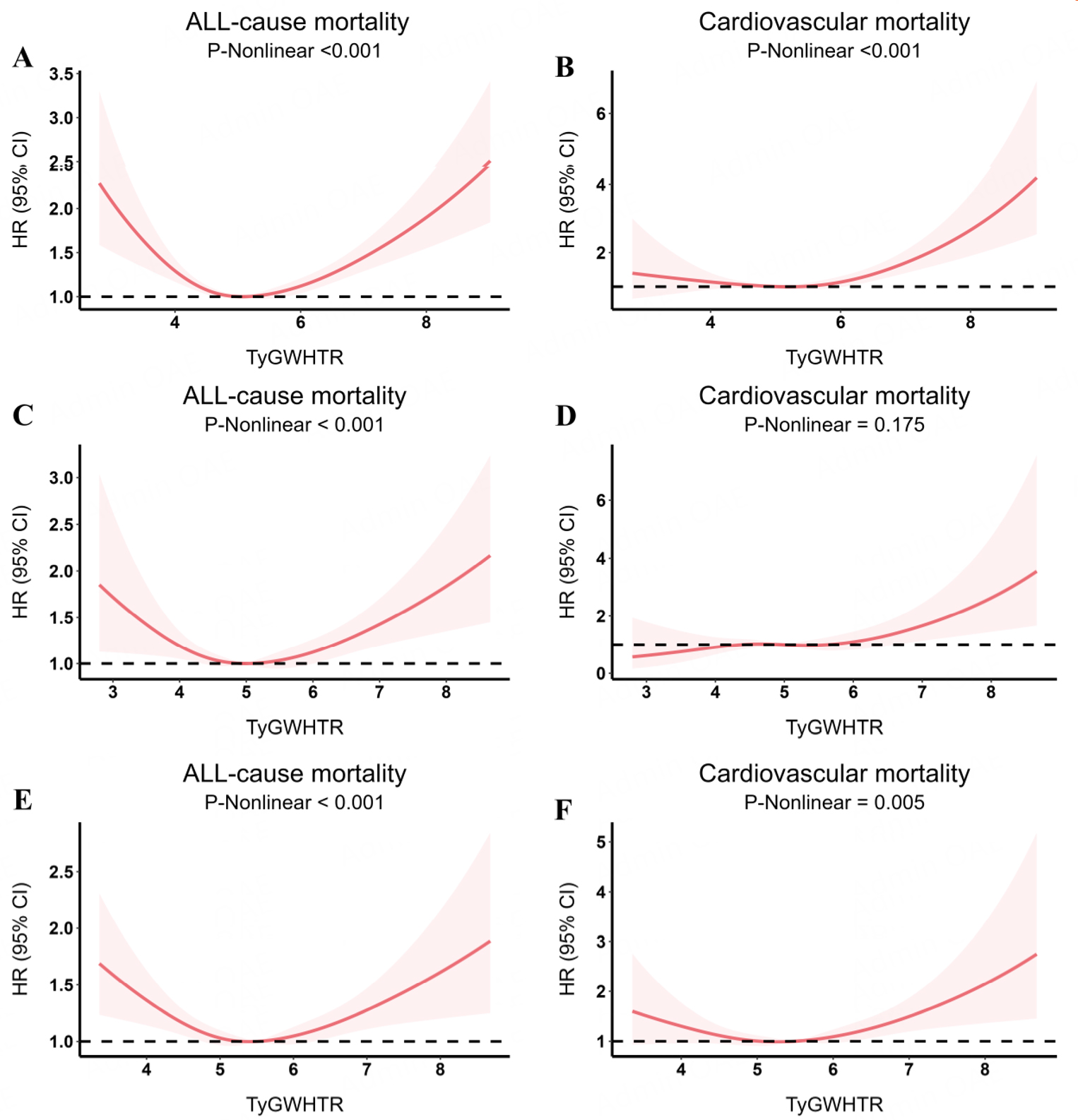
RCS analysis was conducted to explore this relationship further. Substantial nonlinear correlations between TyG-WHtR and both mortality outcomes were found after controlling for all variables
Figure 1. Dose-response relationships between TyG-WHtR and mortality in CKM, evaluated using restricted cubic spline models. Solid curves indicate multivariable-adjusted HRs, and dashed curves show 95%CIs. (A and B) all-cause and cardiovascular mortality in all CKM patients; (C and D) all-cause and cardiovascular mortality in non-advanced CKM; (E and F) all-cause and cardiovascular mortality in advanced CKM. Models were adjusted for demographics (age, sex, race), socioeconomic factors (education, marital status, PIR), lifestyle behaviors (smoking, alcohol), and clinical characteristics (BMI, hypertension, diabetes, medication use, family history of cardiovascular disease).
Table 2 shows the non-linear relationships between the TyG-WHtR index and mortality, analyzed using both a standard and two-piecewise Cox regression model. The inflection points for TyG-WHtR were identified at 6.17 for cardiovascular mortality and 5.04 for all-cause mortality (P for Log-likelihood ratio < 0.001). When TyG-WHtR was under 5.04, the risk of all-cause death decreased by 23.1% for every unit rise in TyG-WHtR (HR: 0.769, 95%CI: 0.68-0.86). Conversely, with every unit increase in TyG-WHtR levels above 5.04, the probability of dying from any cause rose by 19.9% (HR: 1.199, 95%CI: 1.12-1.28). Additionally, above 6.17, cardiovascular death risk rose by 65.3% per unit increase in TyG-WHtR (HR: 1.653, 95%CI: 1.25-2.19)[3]. In participants aged < 60 years, beyond the inflection point of 6.81, per 1-unit higher TyG-WHtR, the hazard of all-cause death was significantly greater (HR: 2.010, 95%CI: 1.47-2.75); beyond 3.66, cardiovascular mortality risk was also markedly elevated (HR: 1.598, 95%CI: 1.22-2.09) [Supplementary Table 4]. In aged ≥ 60 years, TyG-WHtR < 4.90 was negatively correlated with mortality from all causes (HR: 0.702, 95%CI: 0.60-0.82), while values ≥ 4.90 conferred increased risk (HR: 1.17, 95%CI: 1.09-1.25); cardiovascular mortality rose significantly beyond the inflection point of 5.96 (HR: 1.607, 95%CI: 1.32-1.95) [Supplementary Table 5]. These findings suggest that age-specific TyG-WHtR thresholds should help identify high-risk people more precisely and guide focused preventative measures.
Threshold effect analysis of TyG-WHtR index on all-cause and cardiovascular mortality
| TyG-WHtR index | Adjusted HR (95%Cl), P-value |
| All-cause mortality | |
| Fitting by the standard linear model | 1.046 (0.998-1.098) 0.069 |
| Fitting by the two-piecewise linear model | |
| Inflection point | 5.04 |
| TyG-WHtR index < 5.04 | 0.769 (0.685-0.863) < 0.001 |
| TyG-WHtR index ≥ 5.04 | 1.199 (1.122-1.281) < 0.001 |
| P for Log-likelihood ratio | < 0.001 |
| Cardiovascular mortality | |
| Fitting by the standard linear model | 1.220 (1.079-1.380) 0.002 |
| Fitting by the two-piecewise linear model | |
| Inflection point | 6.17 |
| TyG-WHtR index < 6.17 | 1.000 (0.846-1.181) 0.997 |
| TyG-WHtR index ≥ 6.17 | 1.653 (1.246-2.195) < 0.001 |
| P for Log-likelihood ratio | < 0.001 |
Supplementary Table 6 shows the diagnostic efficacy of TyG-WHtR for mortality. TyG-WHtR demonstrated good predictive ability for both all-cause mortality (area under curve (AUC): 0.873, 95%CI: 0.87-0.88) and cardiovascular mortality (AUC: 0.856, 95%CI: 0.84-0.87). Also, the sensitivity and specificity of TyG-WHtR for predicting mortality were both greater than 0.70. This demonstrates its robust discriminatory ability and clinical utility as a reliable prognostic marker.
Associations between TyG-WHtR and aging acceleration
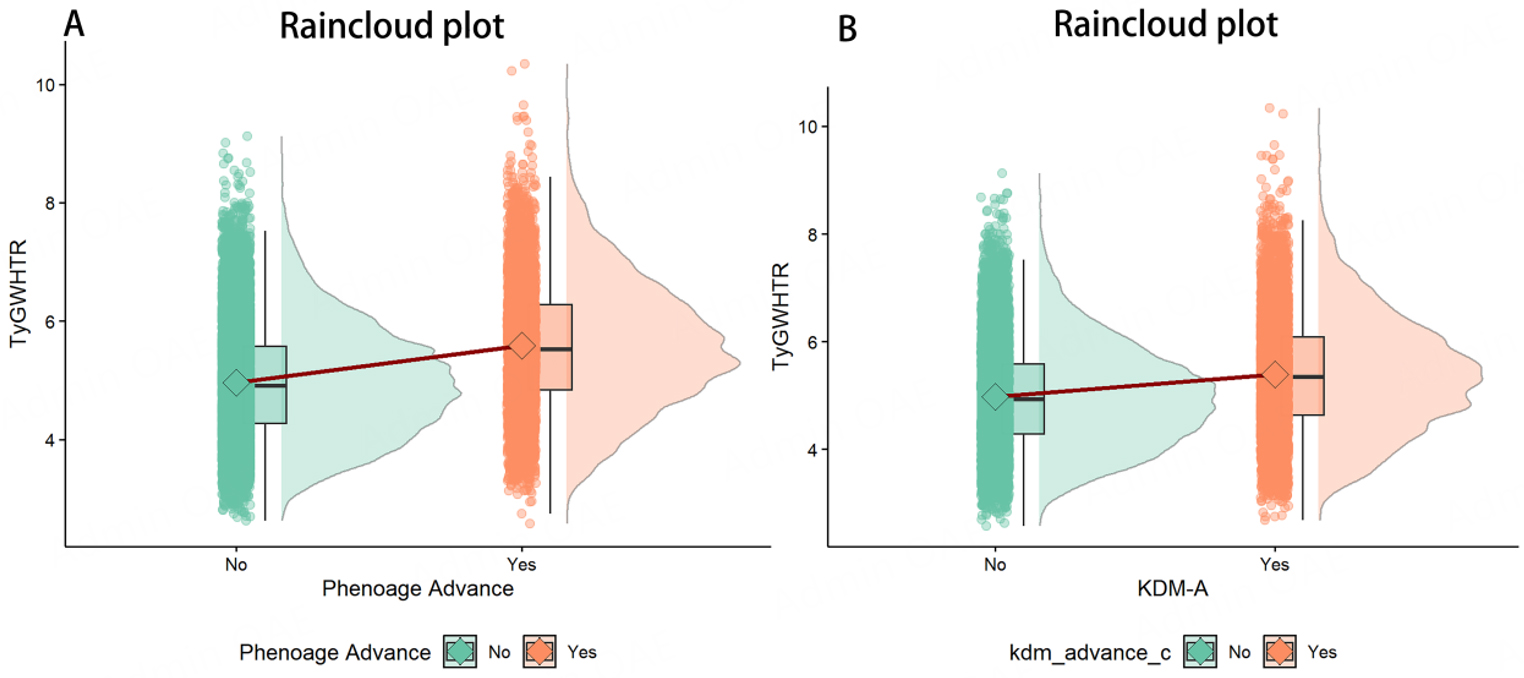
Figure 2 uses a raincloud plot to show how TyG-WHtR and aging acceleration are related. In Model 3, TyG-WHtR demonstrated a strong correlation with both PAA (OR: 1.093, 95%CI: 1.08-1.11) and KDM-AA (OR: 1.126, 95%CI: 1.12-1.14) [Supplementary Table 7]. The relationships between TyG-WHtR and both PAA and KDM-AA remained statistically significant across all regression models when TyG-WHtR was divided into tertiles. Individuals in the highest TyG-WHtR group experienced an 18.9% increase in PAA (OR: 1.189, 95%CI: 1.17-1.21) and 24.4% increase in KDM-AA (OR: 1.244, 95%CI: 1.22-1.27) in contrast to individuals in the group with the lowest TyG-WHtR [Supplementary Table 7]. This finding shows that TyG-WHtR is closely and independently associated with aging.
Associations between aging acceleration and all-cause/cardiovascular mortality
Both all-cause and CVD mortality are significantly correlated with PAA and KDM-AA in Model 3, as shown in Supplementary Tables 8 and 9. Individuals with PAA had a 63.2% higher risk of dying from all causes (HR: 1.632, 95%CI: 1.46-1.82) and an 83.2% higher chance of dying from CVD (HR: 1.832,
Age acceleration as a mediator of the association between TyG-WHtR and mortality
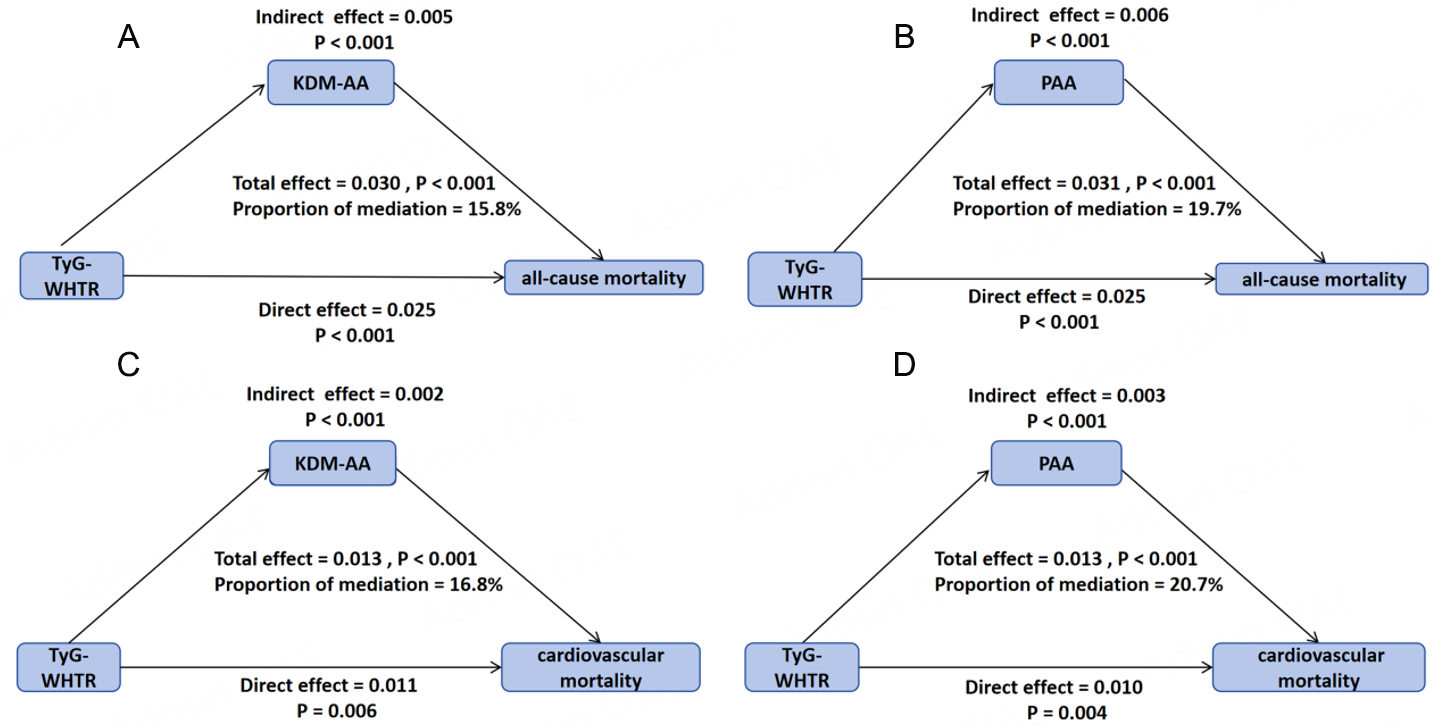
This study examined the potential regulatory effects of KDM-AA and PAA on the association between TyG-WHtR and mortality using mediation analysis [Figure 3]. KDM-AA and PAA mediated 15.8% and 19.7% of the association between TyG-WHtR and all-cause mortality [Supplementary Table 10], and 16.8% and 20.7% of the association between TyG-WHtR and cardiovascular mortality [Supplementary Table 11]. Biological aging can partially explain the link between TyG-WHtR and mortality, underscoring the critical role of aging acceleration in mediating the adverse effects of metabolic dysfunction.
Figure 3. Mediation models evaluating KDM age acceleration and phenotypic age acceleration as mediators of the association between TyG-WHtR and CKM-related mortality (all-cause and cardiovascular). (A and C) models with KDM age acceleration as mediator; (B and D) models with phenotypic age acceleration as mediator.
Subgroup analysis and sensitivity analyses
Subgroup analyses were performed using age, gender, race, BMI, after controlling for variables. According to the subgroup analyses, participants aged 60 years or younger showed a stronger association with increased mortality risk (all-cause mortality: HR: 1.621, 95%CI: 1.48-1.77, P < 0.001; cardiovascular mortality:
To ensure the robustness of our results, we validated the associations between TyG-WHtR, mortality, and accelerated aging in the UK Biobank, and the findings were consistent with those from NHANES [Supplementary Tables 14-17].
We excluded participants who died during the two-year follow-up to reduce the risk of reverse causation. Because the findings of the sensitivity analysis were consistent with those of the primary analysis, the study’s conclusions were reinforced and gained greater validity [Supplementary Table 18].
DISCUSSION
This study demonstrates that TyG-WHtR is strongly associated with cardiovascular and all-cause mortality in CKM patients, and that this relationship is partly mediated by accelerated biological aging. By incorporating KDM-AA and PAA as biomarkers of aging, our work provides new evidence of the vicious cycle linking metabolic dysfunction, vascular aging, and mortality, highlighting the role of IR in shaping long-term outcomes.
WHtR, a key marker of visceral adiposity, is superior to BMI in screening for cardiometabolic diseases[18,19]. Compared with the TyG index, TyG-WHtR captures both metabolic dysfunction and pan-vascular risk, providing stronger predictive value for mortality and CVD in the overall population, elderly individuals, and patients with fatty liver disease associated with metabolic dysfunction[20-23]. Consistent with these findings, our study verified that TyG-WHtR demonstrates strong nonlinear associations with death across CKM stages 0-4. Specifically, we observed U-shaped associations with inflection points at 5.04 (all-cause) and 6.17 (cardiovascular). Above these thresholds, elevated TyG-WHtR indicates severe IR and central obesity, driving lipotoxicity, inflammation, endothelial dysfunction, and myocardial energy dysregulation, highlighting the importance of maintaining optimal TyG-WHtR[24-28].
The relationship between accelerated aging and metabolic disorders has been well documented. The Melbourne Collaborative Cohort Study revealed that 87.5% of individuals with diabetes exhibited signs of aging during a 12-year follow-up, 10% higher than those without diabetes[29]. Among people with type 2 diabetes mellitus (T2DM) and coronary heart disease, the risk of accelerated aging increases by 789% for each one-unit increase in the TyG index[30]. Additionally, the negative correlation between TyG and the anti-aging protein Klotho supports the link between metabolic dysfunction and biological aging[31]. This study’s findings are consistent with those of previous large-scale studies. To fully understand these mechanistic relationships and identify potential intervention targets to slow accelerated aging associated with metabolic diseases, further research is needed. Meanwhile, our findings show that age acceleration is positively associated with mortality, supporting the fundamental notion that an organisms’ aging processes are reflected in its epigenetic clocks[32].
Mechanistically, the elevated TyG-WHtR reflects a dual metabolic burden of IR and central obesity, which accelerates systemic and vascular aging through multiple interconnected pathways. This metabolic dysfunction induces endothelial dysfunction, macrophage polarization, and chronic inflammation, releasing cytokines such as tumor necrosis factor-α and interleukin-6, which activate the protein kinase B/IκB kinase/nuclear factor kappa-light-chain-enhancer of activated B cells pathway, ultimately driving the senescence-associated secretory phenotype[30,33]. Concurrently, IR and visceral adiposity synergistically trigger mitochondrial dysfunction, excessive reactive oxygen species accumulation, oxidative stress, telomere attrition, and reduced telomerase activity, thereby exacerbating cellular senescence and increasing mortality risk[10,34,35]. In addition, epigenetic dysregulation plays a crucial role, as IR alters chromatin states and impairs DNA repair mechanisms, irreversibly activating pro-aging gene programs[36]. In the end, these mechanisms converge on the “seven pillars of aging”, accelerating multi-organ decline and mortality[37,38].
Subgroup analysis showed that TyG-WHtR had a stronger impact in individuals under 60, likely due to genetic susceptibility, or early onset of renal, cardiovascular, and metabolic disorders[39-41]. Both men and women should pay close attention to metabolic abnormalities and their potential health risks. Unhealthy lifestyle factors, including smoking, alcohol consumption, and obesity, further accelerate vascular aging and exacerbate the risks of atherosclerosis and mortality[42]. Younger patients may also overlook symptoms, stressing the importance of early case-finding and risk management to improve long-term outcomes.
Among the study’s strengths are its population-representative sample, prospective design, and relatively long follow-up, which provide robust support for the conclusions. Importantly, this study shows that TyG-WHtR is associated with accelerated aging in CKM patients, which partly mediates its link to mortality, highlighting a cycle of metabolic dysfunction-vascular aging-death and offering new insights for risk stratification and intervention.
Limitations
Nevertheless, several limitations should be noted. The observational design of NHANES precludes causal inference, and biological age was measured only at baseline, preventing assessment of whether dynamic changes in TyG-WHtR can reverse age acceleration. In addition, even after adjustment, residual confounding from unmeasured factors, such as genetic predisposition or socioeconomic status, may still exist. Despite these limitations, our findings provide valuable insights into how IR drives pan-vascular aging and accelerates mortality in CKM populations.
Future research should prioritize validating the predictive performance of TyG-WHtR in large-scale cohorts and establishing population-specific cut-points for early risk stratification and precision intervention. Longitudinal studies are needed to assess whether improvements in TyG-WHtR can decelerate aging processes and reduce mortality risk. Additionally, intervention strategies should evaluate comprehensive management approaches targeting the dual metabolic burden reflected by TyG-WHtR. Pharmacological interventions may include metformin, glucagon-like peptide-1 receptor agonists, or sodium-glucose cotransporter-2 inhibitors to improve insulin sensitivity, combined with statins or fibrates to optimize lipid metabolism[43]. Lifestyle modifications such as Mediterranean diet and combined aerobic-resistance training are equally important for reducing visceral adiposity. Integrating TyG-WHtR into existing risk assessment frameworks and developing individualized monitoring protocols will be critical for clinical implementation. Cross-population validation studies are essential to establish generalizable guidelines and reduce the burden of metabolic dysfunction-driven vascular aging and mortality.
Conclusion
TyG-WHtR and cardiovascular and all-cause mortality in CKM patients were found to have a strong U-shaped relationship. Accelerated aging mediated this relationship. Therefore, by using appropriate therapies, keeping TyG-WHtR within an ideal range may help slow aging and lower the risk of death.
DECLARATIONS
Acknowledgments
We thank all participants for contributing their data.
Authors’ contributions
Conceived the study and provided overall supervision: Liu S, Hu H
Developed the methodology and conducted the statistical analyses: Li X, Zhang H, Zhang J
Curated the data and supported the interpretation of results: Chen M, Shi J, Sun L
Contributed to the investigation and provided technical support: Yao H, Li C
Performed validation and quality control: Zheng X
Prepared the figures and tables: Zhang J
Drafted the manuscript: Li X, Zhang H
All authors reviewed and revised the manuscript critically and approved the final version.
Availability of data and materials
The datasets used and/or analyzed during the current study were publicly available. This data can be found here: https://www.cdc.gov/nchs/nhanes/index.htm. Raw UK Biobank data are protected under data privacy regulations and therefore not publicly available. Qualified researchers may apply for access through the UK Biobank Access Management System (https://ams.ukbiobank.ac.uk/ams/).
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (Grant No. 82300904), the National Natural Science Foundation of China-Joint Fund Major Program (Grant No. U25D9022), and the Clinical Medical Research Transformation Project of Anhui Province (Grant Nos. 202204295107020021, 202204295107020026). The study was also supported by the 2023 Diabetes Young and Middle-aged Physicians Research Project, funded by the Bethune Foundation (Grant No. 2023KY623). The funders had no role in the design or conduct of the study, the collection, analysis, or interpretation of data, or the preparation, review, or approval of the manuscript.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
NHANES has been approved by the Research Ethics Review Board in the NCHS, CDC. The UK Biobank was approved by the North West Haydock Research Ethics Committee (REC reference: 21/NW/0157). All participants provided written informed consent. Analyses were performed under UK Biobank application number 148816.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Ndumele CE, Rangaswami J, Chow SL, et al. Cardiovascular-kidney-metabolic health: a presidential advisory from the American heart association. Circulation. 2023;148:1606-35.
2. Ndumele CE, Neeland IJ, Tuttle KR, et al. A synopsis of the evidence for the science and clinical management of cardiovascular-kidney-metabolic (CKM) syndrome: a scientific statement from the american heart association. Circulation. 2023;148:1636-64.
3. Yang Q, Zhu X, Zhang L, Luo F. Dyslipidemia and aging: the non-linear association between atherogenic index of plasma (AIP) and aging acceleration. Cardiovasc Diabetol. 2025;24:181.
4. Li X, Cao X, Zhang J, et al. Accelerated aging mediates the associations of unhealthy lifestyles with cardiovascular disease, cancer, and mortality. J Am Geriatr Soc. 2024;72:181-93.
5. Hu L, Li J, Tang Z, et al. How does biological age acceleration mediate the associations of obesity with cardiovascular disease? Evidence from international multi-cohort studies. Cardiovasc Diabetol. 2025;24:209.
6. Zhang Z, He X, Sun Y, Li J, Sun J. Type 2 diabetes mellitus: a metabolic model of accelerated aging - multi-organ mechanisms and intervention approaches. Aging Dis. 2025;17:1399-422.
7. Zhang P, Mo D, Zeng W, Dai H. Association between triglyceride-glucose related indices and all-cause and cardiovascular mortality among the population with cardiovascular-kidney-metabolic syndrome stage 0-3: a cohort study. Cardiovasc Diabetol. 2025;24:92.
8. Kityo A, Lee SA. Association of cardiometabolic factors and insulin resistance surrogates with mortality in participants from the Korean Genome and Epidemiology Study. Lipids Health Dis. 2023;22:210.
9. Levine ME, Lu AT, Quach A, et al. An epigenetic biomarker of aging for lifespan and healthspan. Aging. 2018;10:573-91.
10. Pan LY, Jin L. Association between triglyceride glucose index and biological aging in U.S. adults: National Health and Nutrition Examination Survey. Cardiovasc Diabetol. 2025;24:100.
11. Ou Y, Wang Z, Yuan Y, et al. Epigenetic age acceleration and rheumatoid arthritis: an NHANES-based analysis and survival prediction models. Clin Epigenet. 2025;17:111.
12. Aggarwal R, Ostrominski JW, Vaduganathan M. Prevalence of cardiovascular-kidney-metabolic syndrome stages in US adults, 2011-2020. JAMA. 2024;331:1858-60.
13. Zhu R, Wang R, He J, et al. Prevalence of cardiovascular-kidney-metabolic syndrome stages by social determinants of health. JAMA Netw Open. 2024;7:e2445309.
14. Boutouyrie P, Chowienczyk P, Humphrey JD, Mitchell GF. Arterial stiffness and cardiovascular risk in hypertension. Circ Res. 2021;128:864-86.
15. Wei Z, Nie G, Li J, Zhang J, Liu H, Ma M. Association of triglyceride-glucose index and its related parameters with functional disability: evidence from the China Health and Retirement Longitudinal Study. Front Aging Neurosci. 2025;17:1488124.
16. Zhang Q, Xiao S, Jiao X, Shen Y. The triglyceride-glucose index is a predictor for cardiovascular and all-cause mortality in CVD patients with diabetes or pre-diabetes: evidence from NHANES 2001-2018. Cardiovasc Diabetol. 2023;22:279.
17. Sun Y, Liu B, Snetselaar LG, et al. Association of fried food consumption with all cause, cardiovascular, and cancer mortality: prospective cohort study. BMJ. 2019;364:k5420.
18. Ashwell M, Gibson S. Waist-to-height ratio as an indicator of 'early health risk': simpler and more predictive than using a 'matrix' based on BMI and waist circumference. BMJ Open. 2016;6:e010159.
19. Ashwell M, Gunn P, Gibson S. Waist-to-height ratio is a better screening tool than waist circumference and BMI for adult cardiometabolic risk factors: systematic review and meta-analysis. Obes Rev. 2012;13:275-86.
20. Li S, An L, Fu Z, Zhang W, Liu H. Association between triglyceride-glucose related indices and all-cause and cause-specific mortality in the general population: a cohort study. Cardiovasc Diabetol. 2024;23:286.
21. Ren Q, Huang Y, Liu Q, Chu T, Li G, Wu Z. Association between triglyceride glucose-waist height ratio index and cardiovascular disease in middle-aged and older Chinese individuals: a nationwide cohort study. Cardiovasc Diabetol. 2024;23:247.
22. Zhang Y, Wu J, Li T, Qu Y, Wang Y. Association of triglyceride-glucose related indices with mortality among individuals with MASLD combined with prediabetes or diabetes. Cardiovasc Diabetol. 2025;24:52.
23. Dang K, Wang X, Hu J, et al. The association between triglyceride-glucose index and its combination with obesity indicators and cardiovascular disease: NHANES 2003-2018. Cardiovasc Diabetol. 2024;23:8.
24. Las Heras N, Lahera V. Relevance of mitochondrial dysfunction in heart disease associated with insulin resistance conditions. Pflugers Arch. 2022;474:21-31.
25. Laakso M. Cardiovascular disease in type 2 diabetes from population to man to mechanisms: the Kelly West Award Lecture 2008. Diabetes Care. 2010;33:442-9.
26. Gallo G, Savoia C. New insights into endothelial dysfunction in cardiometabolic diseases: potential mechanisms and clinical implications. Int J Mol Sci. 2024:25.
27. Li R, Lau WB, Ma XL. Adiponectin resistance and vascular dysfunction in the hyperlipidemic state. Acta Pharmacol Sin. 2010;31:1258-66.
28. Qiu J, Li J, Xu S, et al. Triglyceride glucose-weight-adjusted waist index as a cardiovascular mortality predictor: incremental value beyond the establishment of TyG-related indices. Cardiovasc Diabetol. 2025;24:306.
29. Hodge AM, Flicker L, O'Dea K, English DR, Giles GG. Diabetes and ageing in the melbourne collaborative cohort study (MCCS). Diabetes Res Clin Pract. 2013;100:398-403.
30. Huang Z, Liu N, Chen S, Chen Z, Wang P. Factors influencing accelerated aging in patients with type 2 diabetes mellitus and coronary heart disease. Front Endocrinol. 2024;15:1416234.
31. Zhang Y, Song K, Yao Z. The association between the triglyceride-glucose index and serum anti-aging protein α- Klotho: a population-based study. Diabetol Metab Syndr. 2024;16:259.
32. Oblak L, van der Zaag J, Higgins-Chen AT, Levine ME, Boks MP. A systematic review of biological, social and environmental factors associated with epigenetic clock acceleration. Ageing Res Rev. 2021;69:101348.
33. Zhang Y, Tu J, Li Y, et al. Inflammation macrophages contribute to cardiac homeostasis. Cardiology Plus. 2023;8:6-17.
34. Lin J, Epel E. Stress and telomere shortening: insights from cellular mechanisms. Ageing Res Rev. 2022;73:101507.
35. Park MH, Kim DH, Lee EK, et al. Age-related inflammation and insulin resistance: a review of their intricate interdependency. Arch Pharmacal Res. 2014;37:1507-14.
36. Zhao Z, Liang Y. Association between triglyceride-glucose index and phenotypic age acceleration: a cross-sectional study based on NHANES database. Front Physiol. 2025;16:1548690.
37. Yan Z, Pu X, Cai Y, Chang X, Liu Z, Liu R. Biological aging traits mediate the association between cardiovascular health levels and all-cause and cardiovascular mortality among adults in the U.S. without cardiovascular disease. Biogerontology. 2025;26:40.
38. Goh J, Wong E, Soh J, Maier AB, Kennedy BK. Targeting the molecular & cellular pillars of human aging with exercise. FEBS J. 2023;290:649-68.
39. Magliano DJ, Sacre JW, Harding JL, Gregg EW, Zimmet PZ, Shaw JE. Young-onset type 2 diabetes mellitus - implications for morbidity and mortality. Nat Rev Endocrinol. 2020;16:321-31.
40. Wang C, Yuan Y, Zheng M, et al. Association of age of onset of hypertension with cardiovascular diseases and mortality. J Am Coll Cardiol. 2020;75:2921-30.
41. Vivante A, Hildebrandt F. Exploring the genetic basis of early-onset chronic kidney disease. Nat Rev Nephrol. 2016;12:133-46.
42. Tromp J, Paniagua SMA, Lau ES, et al. Age dependent associations of risk factors with heart failure: pooled population based cohort study. BMJ. 2021;372:n461.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].