Systematic review and meta-analysis on safety and effectiveness of postoperative radiotherapy after ear keloid excision
Abstract
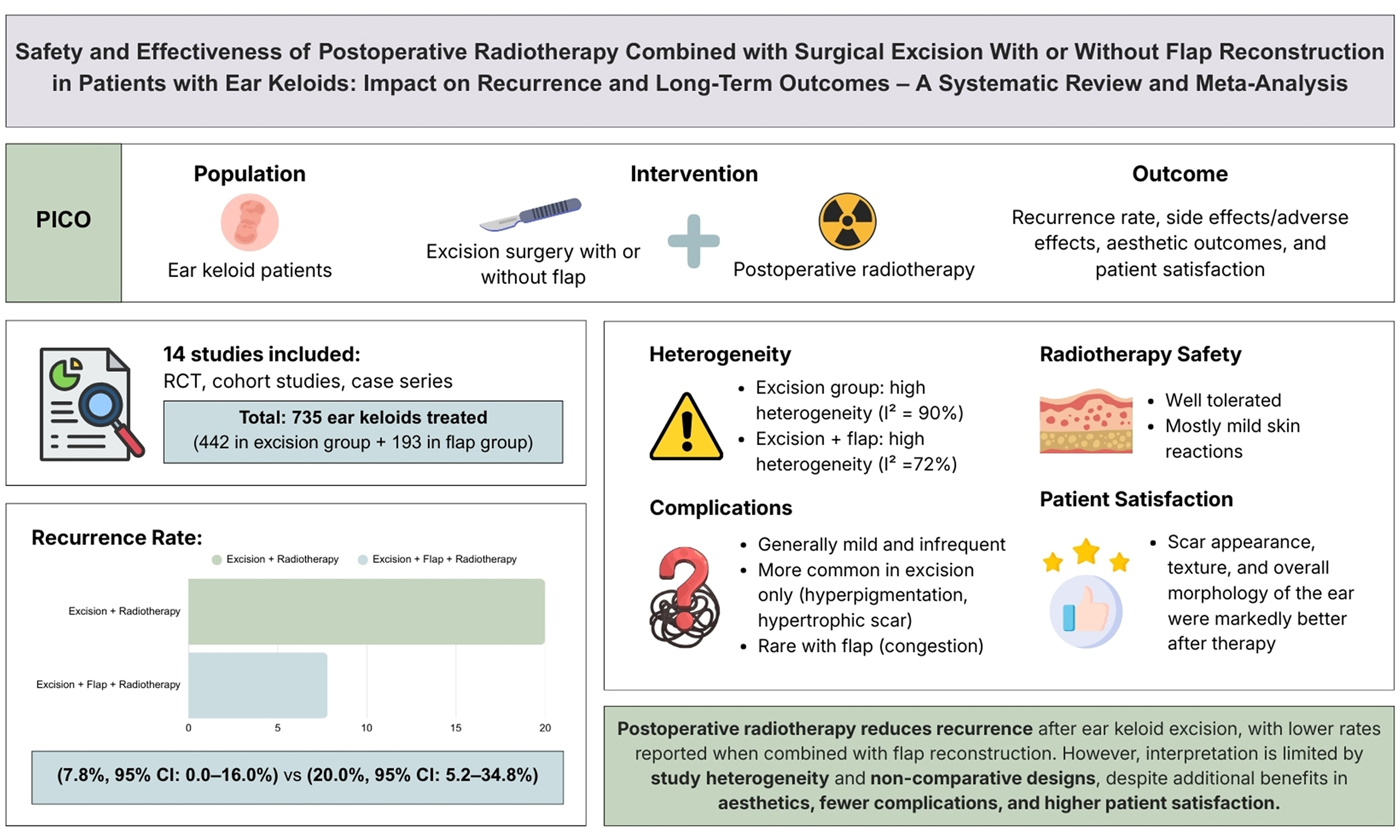
Aim: This systematic review and meta-analysis aims to evaluate the efficacy and safety of postoperative radiotherapy following surgical excision, with or without reconstruction by flap, for patients with ear keloids.
Methods: A comprehensive literature search was performed in PubMed, Cochrane Library, ScienceDirect, and EBSCOHost through August 2025. We included randomized controlled trials, cohort studies, and case series involving patients with ear keloids treated with surgical excision and postoperative radiotherapy, with or without flap reconstruction. Data of recurrence, complications, aesthetic outcomes, and patient satisfaction were abstracted and analyzed.
Results: Fourteen studies were included based on the criteria, with 735 treated keloids in total. Pooled recurrence proportions were 7.8% [95% confidence interval (CI): 0.0%-16.0%] in studies reporting excision with flap reconstruction plus postoperative radiotherapy and 20.0% (95%CI: 5.2%-34.8%) in studies reporting excision plus postoperative radiotherapy without flap reconstruction. Reported adverse effects were generally mild, most commonly pigmentary change and hypertrophic scarring; flap-related events were uncommon. However, long-term oncologic risk could not be reliably assessed, as most included studies had limited follow-up and were not designed to detect rare late events. Cosmetic outcomes and patient satisfaction were frequently reported as favorable across the subset of studies that assessed these outcomes.
Conclusion: Postoperative radiotherapy is commonly reported as an effective adjuvant after excision of auricular keloids, with relatively low recurrence and generally mild short-term adverse effects. Cosmetic outcomes and patient satisfaction were frequently favorable, although interpretation is limited by substantial heterogeneity and predominantly non-comparative observational evidence, underscoring the need for well-designed comparative studies with standardized outcome reporting.
Keywords
INTRODUCTION
Keloids are a challenging fibroproliferative scar disorder with high recurrence rates despite treatment. They are abnormal fibroproliferative scars that develop after surgery, injury, or minor skin irritation. While some external stimuli can evoke the development of keloids, there are patients with genetic predispositions to developing keloids, and in some instances, keloids may also occur spontaneously without an identifiable trigger[1,2].
A range of therapeutic strategies has been investigated to manage keloids, including topical treatments such as corticosteroid injections, intralesional chemotherapy, cryotherapy, and laser therapy[3]. Nevertheless, surgical excision remains the primary treatment approach, particularly for primary auricular keloids. Although surgical excision may be successful in excising lesions, recurrence rates remain high and are often reported to exceed 50%. Surgical method choice is critical in reducing rates of recurrence and preserving morphology of the ear. Complete excision is largely favored for minor auricular keloids; nevertheless, application in larger keloids is restricted by increased wound tension and risk of distortion of auricular structure. Core excision (i.e., removal of the fibrotic core while preserving the overlying epidermis and papillary dermis) may reduce contour deformity risk[4]. For the management of earlobe keloids, excision with primary closure is generally successful, although for simple direct linear closure of helical keloids it is generally not feasible or results in severe deformity of ear shape[5]. In such cases, reconstruction through the application of flaps has helped to redistribute stress at the periphery of the wound and consequently diminish the risk of recurrence[6].
Postoperative radiotherapy has emerged as a valuable adjunct to surgical excision in the treatment of keloids[7]. Radiotherapy alone is associated with high recurrence; however, adjuvant radiotherapy after excision has been associated with improved outcomes[8]. Reported efficacy rates for this combined approach range from 28% to 98%; however, the variability in results reflects a lack of standardized treatment protocols in prior studies. Numerous studies have evaluated the effectiveness of combined treatment modalities, of which results from some studies reflect superb success and limited recurrence rates, thus suggesting its effectiveness as a treatment approach[9].
The aim of this systematic review is to analyze the efficacy and safety of postoperative radiotherapy plus surgical excision, with/without reconstruction by flap, for the treatment of ear keloids. Specifically, this review assesses the effects of these treatment modalities on recurrence rates and late clinical outcomes and provides a combined analysis of their clinical value.
METHODS
The systematic review adheres to the guidelines established by the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA)[10]. The protocol for this systematic review has been registered with the International Prospective Register of Systematic Reviews (PROSPERO), under the registration number CRD420251015501.
Search strategy and study selection
A comprehensive literature search was conducted via four databases: PubMed (https://pubmed.ncbi.nlm.nih.gov/), Cochrane (https://www.cochranelibrary.com/), ScienceDirect (https://www.sciencedirect.com/), and EBSCOHost (https://www.ebsco.com/), up to August 2025. In addition, references of potentially relevant articles were carefully searched to reveal any additional relevant studies. After removing duplicates, authors independently screened titles and abstracts; disagreements were resolved by consensus. Study selection was also guided by the PICO (Population, Intervention, Comparison, and Outcome) approach, encompassing the following aspects: (1) Population: Ear keloid patients; (2) Intervention/exposure: surgical excision with postoperative radiotherapy, with or without flap reconstruction; (3) Comparison: not applicable; (4) Outcomes: Recurrence rate, side effects/adverse effects, aesthetic outcomes, and patient satisfaction. Studies were excluded according to various criteria such as unavailability of full articles, classification of experimental studies, availability only as abstracts, conference articles, non-English articles, and incomplete at the time of the search. To reflect contemporary reporting standards and modern radiotherapy practice, we restricted eligibility to studies published within the last 10 years (January 2015-August 2025). This restriction was applied to publication date (not to patient enrollment period), acknowledging that some included cohorts began enrollment earlier to allow sufficient follow-up.
Data extraction
Data extraction was conducted according to suggestions from the Cochrane Handbook for Systematic Reviews of Interventions. Any differences in the data extraction process were resolved by consensus. Google Sheets and Microsoft Excel facilitated the data extraction process. The extracted data from relevant articles included the following: data from the research (authors, year of publication, level of evidence, country, length of the study, design of the study, and period of follow-up); participant characteristics (number of subjects, gender of participants, participant’s age, location of the keloid, size of the keloid, and etiology of the keloid); intervention details (type of flaps, type of radiotherapy, and prescription regimen); and outcomes (recurrence rate, side effects/adverse effects, and patients’ satisfaction). The level of evidence for each article was assessed according to the Oxford Centre for Evidence-Based Medicine criteria[11].
Quality assessment
An extensive assessment of each study was performed utilizing the appropriate evaluation tool relevant to its study design. Three authors independently appraised the quality of each study employing the Cochrane Risk of Bias 2 (RoB 2) tool for randomized clinical trials[12], the Newcastle Ottawa Scale for cohort studies[13], and the Joanna Briggs Institute (JBI) for case series studies[14]. In addition, the certainty of evidence was assessed using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach for all key outcomes, considering risk of bias, inconsistency, indirectness, imprecision, and publication bias. Any discrepancies encountered were resolved through consensus.
Statistical analysis
All statistical analyses were performed using R version 2023.06.2.561. Proportional meta-analysis was done to estimate the pooled recurrence rates with their corresponding 95% confidence intervals (CI). Fixed- or random-effects models were used, depending on the degree of heterogeneity. Heterogeneity was assessed using the I2 statistic; values of 25%, 50%, and 75% were interpreted as low, moderate, and high heterogeneity, respectively. Funnel plot was graphically inspected to investigate potential publication bias. Subgroup analyses were performed where sufficient data existed, according to intervention type (excision vs. reconstruction by flap) and radiotherapy regimen. A P value < 0.05 was considered statistically significant.
RESULTS
Search results and included studies
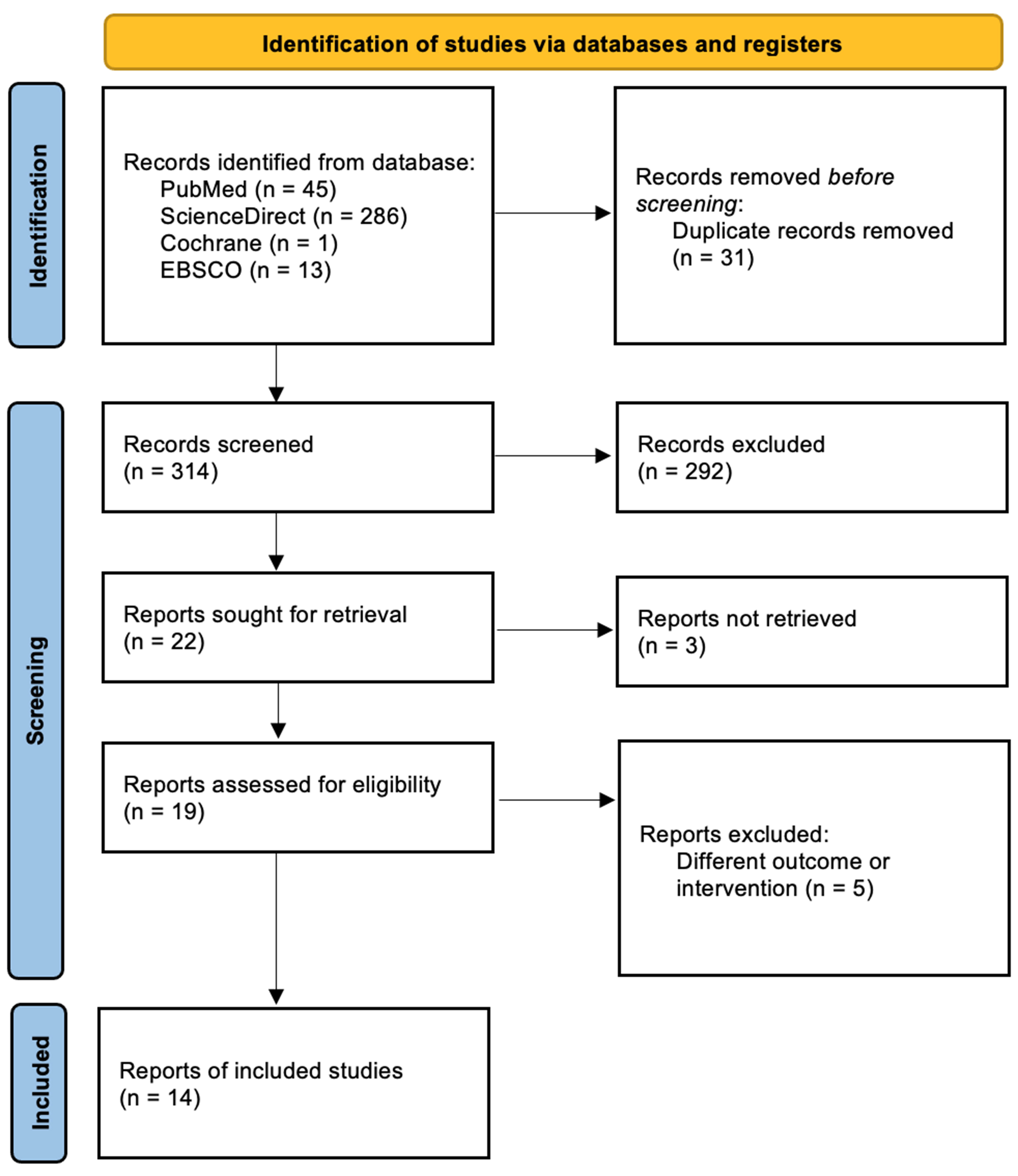
A comprehensive literature search was conducted across four electronic databases, PubMed, ScienceDirect, Cochrane, and EBSCOHost, using the keywords “ear keloid” or “auricular keloid” combined with “radiotherapy”, “radiation therapy”, or “postoperative radiotherapy”, and “excision”. The literature search was confined to publications in the English language and was restricted to studies published between January 2015 and August 2025. This search yielded 45 articles in PubMed, 286 articles in ScienceDirect, 1 article in Cochrane, and 13 articles in EBSCOHost. A total of 345 articles were identified through the initial database search. Following the removal of duplicates, 314 articles underwent title and abstract screening. Of these, 19 articles were assessed for full-text eligibility, and ultimately, 14 studies were included in this systematic review and meta-analysis. The study designs varied, comprising one randomized controlled trial (RCT), three case series, and multiple cohort studies. Figure 1 illustrates the progression of the literature search process.
Participants
The included studies encompassed a diverse patient population with varying demographic distributions. Sample sizes ranged from as few as two participants in a case series to larger cohorts exceeding 200 participants. The excision group included a total of 442 keloids, while the flap group included 193. The majority of studies reported a higher prevalence of keloids among females, with ear piercing being the most commonly identified etiological factor. The mean age of participants varied across studies, with most falling within the range of 20 to 40 years. The anatomical location of ear keloids predominantly included the lobule and helix, although some studies also evaluated keloids affecting the scapha and conchal regions. Detailed characteristics of each included study are presented in Table 1.
Study characteristics and population
| Author (year) | LoE | Duration of study | Country | Design | Follow-up (months) | Population | ||||||
| Subject (n) | Sex [n (%)] | Age | Location (L/R) | Number of keloid | Site | Etiology [n (%)] | ||||||
| Excision | ||||||||||||
| Kim et al. (2015)[15] | 2b | May 2000-February 2011 | South Korea | Retrospective cohort | 49.25 | 16 | Not mentioned | 26.69 ± 3.51 | 9/7 | 16 | Not mentioned | Not mentioned |
| Shen et al. (2015)[16] | 2b | February 1998-January 2012 | China | Retrospective cohort | 40 | Not mentioned | Not mentioned | 29 (5-80) | Not mentioned | 239 | Lobule | Not mentioned |
| Lee and Park (2015)[17] | 2b | January 2006-January 2012 | South Korea | Prospective cohort | 27.4 | Not mentioned | Not mentioned | Not mentioned | Not mentioned | 28 | Lobule: 22 (78.6%) Helix: 6 (21.4%) | Piercing: 24 (85.7%) Trauma: not specified Surgical scar: not specified Acne: not specified |
| Khalid et al. (2018)[9] | 1b | May 2014-January 2015 | Pakistan | RCT | 19 | 30 | Male: 9 (30%) Female: 21 (70%) | 32.73 ± 7.09 years | 20/10 | 30 | Lobule only: 17 (56.67%) Lobule and helix: 7 (23.3%) | Related to ear piercing [20 (66.67%)] Blunt trauma: not specified Sharp trauma: not specified |
| Renz et al. (2018)[18] | 2b | April 2009-January 2017 | United States | Retrospective cohort | 40 | 86 | Not mentioned | Not mentioned | Not mentioned | Not mentioned | Not mentioned | Not mentioned |
| Liu et al. (2019)[19] | 2b | January 2011-July 2017 | China | Retrospective cohort | 26 (14-93) | 23 | Male: 8 (35%) Female: 15 (65%) | 28 (17-63) | 16/14 | 30 | Lobule: 30 (100%) | Ear piercing: 21 (70%) Acne: 6 (20%) Trauma: 3 (10%) |
| Son et al. (2020)[20] | 2b | Not mentioned | Mexico | Case series | 12 | 3 | Male: 2 (66.7%) Female: 1 (33.3%) | 43 ± 18.2 | 1/2 | 3 | Lobule | Not mentioned |
| Hwang et al. (2022)[21] | 2b | January 2015-July 2020 | China | Retrospective cohort | 29 (12-77) | Not mentioned | Not mentioned | Not mentioned | Not mentioned | 92 | Lobule: 26 (28.3%) Helix, scapha, conch: 66 (71.7%) | Not mentioned |
| Franzetti et al. (2024)[22] | 2b | 2004-2020 | Italy | Retrospective cohort | 64 (25-103) | 18 | Not mentioned | Not mentioned | Not mentioned | 18 | Not mentioned | Not mentioned |
| Flap | ||||||||||||
| Ogawa et al. (2015)[4] | 2b | 2006-2012 | Japan | Retrospective cohort | 18 months | 57 | Female: (91.2%) | Not mentioned | Not mentioned | 63 | Not mentioned | Ear piercing: (95.2%) Trauma: (4.8%) |
| Maeda et al. (2019)[6] | 4 | Not mentioned | Japan | Case series | 80.7 months | 2 | Female: 2 (100%) | 20 | 0/2 | 2 | Helix: 2 (100%) | Not mentioned |
| Ahmednaji et al. (2023)[23] | 2b | May 2001-January 2018 | China | Retrospective cohort | 32 months | 41 | Female: 36 (87.8%) Male: 5 (12.2%) | 28.1 | Not mentioned | 44 | Not mentioned | Ear piercing: 36 (87.8%) Unknown: 5 (12.2%) |
| Li et al. (2024)[24] | 2b | January 2019-December 2021 | China | Prospective cohort | 24 months | 96 | Male: 28 (29.17%) Female: 68 (70.83%) | 18-24: 28 (29.17%) 25-30: 47 (48.96%) 31-35: 21 (21.87%) | 54/42 | 96 | Earlobe: 49 (51.4%) Helix: 47 (48.96%) | Not mentioned |
| Park et al. (2024)[25] | 2b | May 2021-March 2023 | South Korea | Retrospective cohort | 12 months | 25 | Male: 0 (0%) Female: 25 (100%) | 26.68 ± 7.74 | 12/13 | 25 | Not mentioned | Piercing: 22 (88%) Surgery: 3 (12%) |
Outcomes: recurrence rate
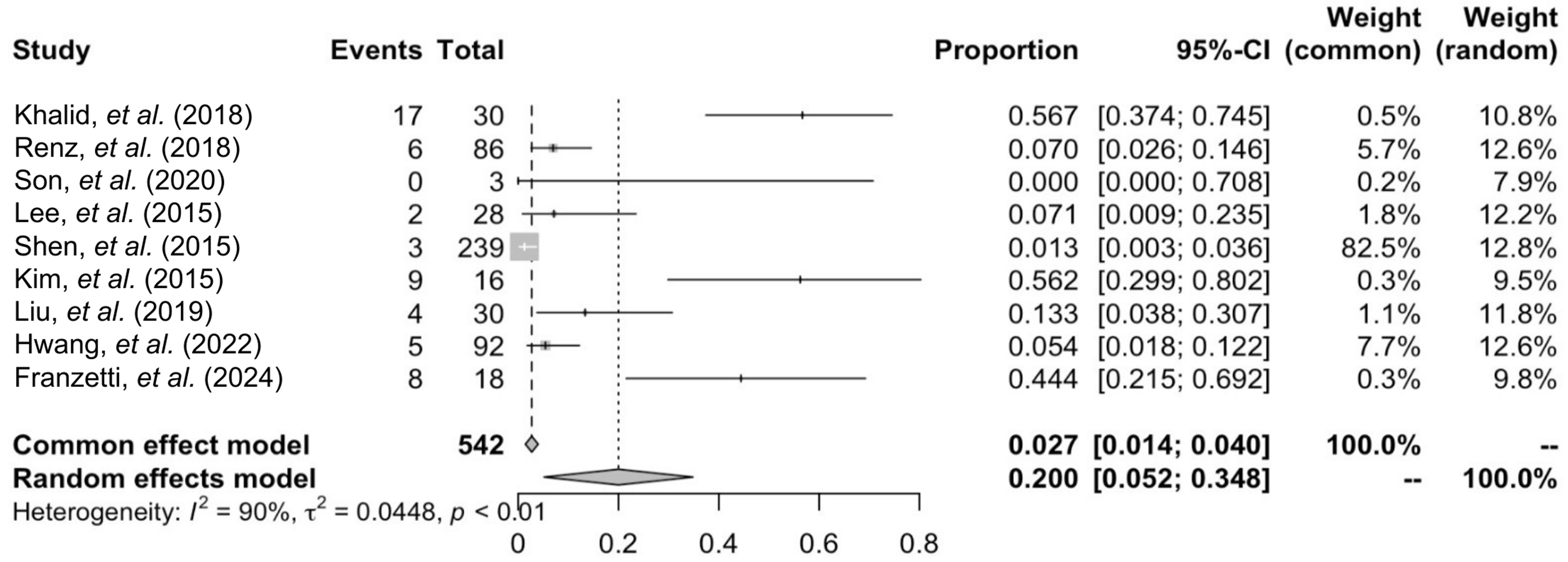
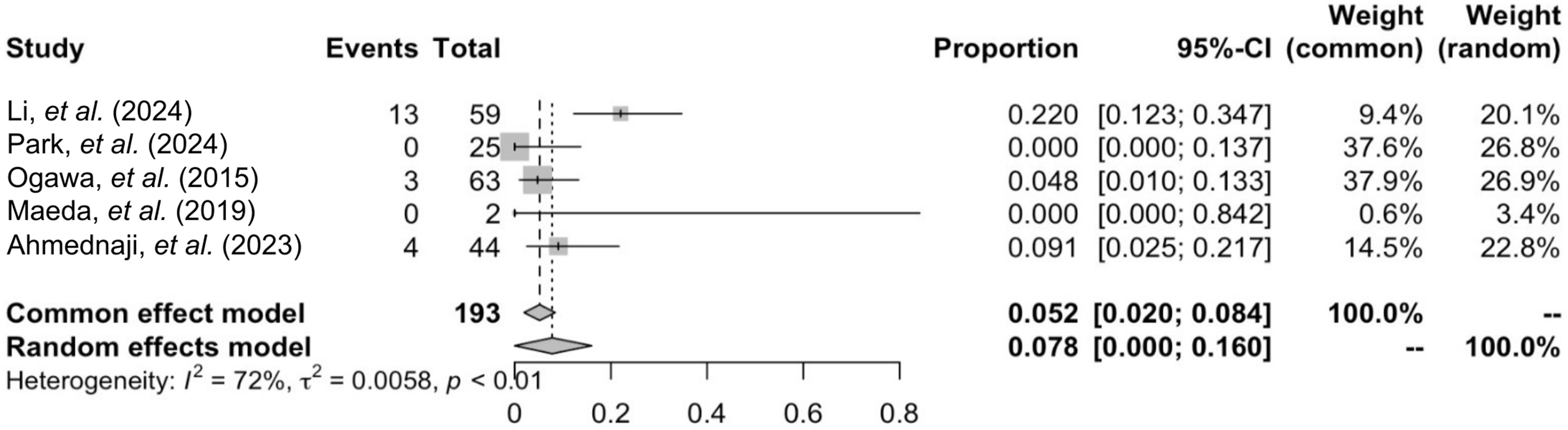
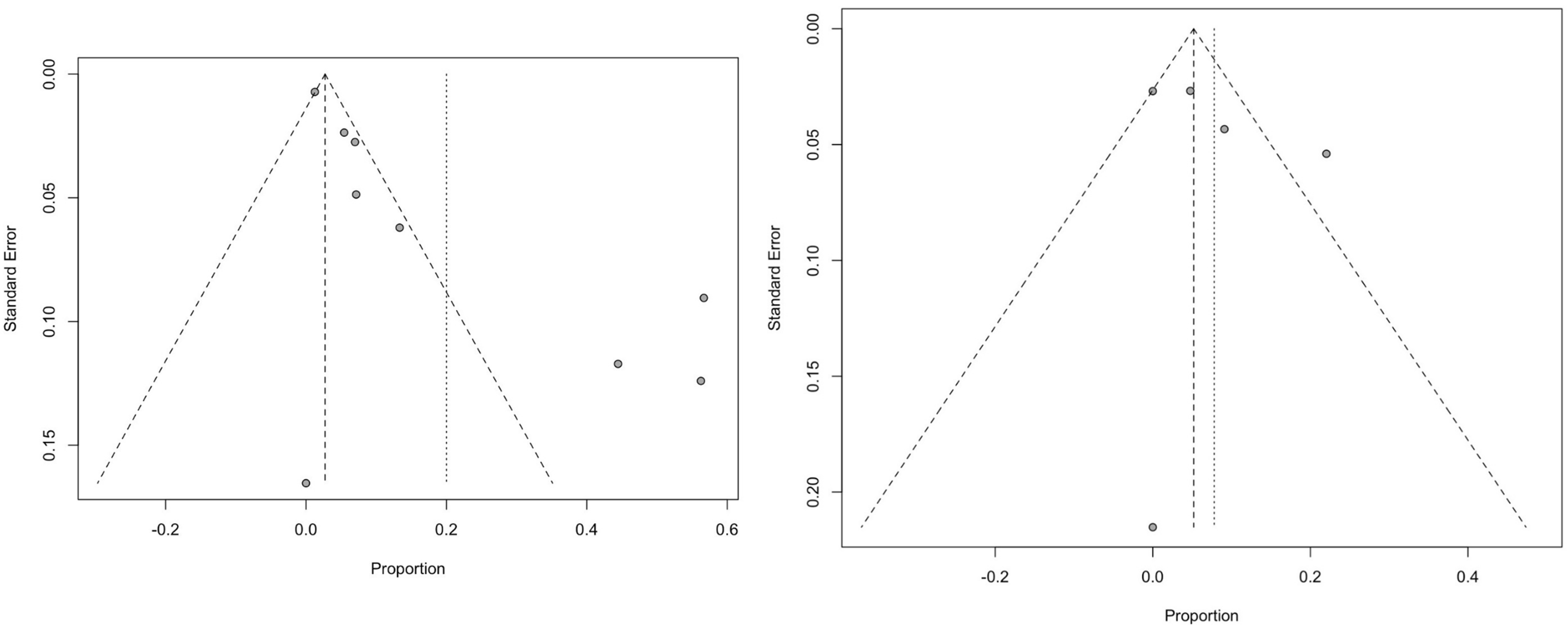
Two proportional meta-analyses [Figures 2 and 3] summarized pooled recurrence proportions for studies reporting excision with postoperative radiotherapy and studies reporting excision with flap reconstruction plus postoperative radiotherapy. The pooled recurrence proportion was 0.200 (95%CI: 0.052-0.348) for excision plus radiotherapy and 0.078 (95%CI: 0.000-0.160) for flap plus radiotherapy, both under random-effects models. Between-study heterogeneity was substantial in both analyses (I2 = 90% and 72%, respectively). Funnel plot inspection suggested asymmetry, particularly for the excision plus radiotherapy group [Figure 4].
Figure 4. The funnel plot illustrates the standard error in relation to the proportion for studies evaluating excision interventions (left) and flap interventions (right).
As an additional descriptive summary, crude recurrence proportions were calculated across studies with extractable numerators and denominators. For electron-beam radiotherapy, 38 recurrences/553 keloids (6.9%) were reported; for non-electron modalities, 27/203 (13.3%). When grouped by total prescribed dose, regimens ≥ 15 gray (Gy) reported 15/426 (3.5%), whereas regimens < 15 Gy reported 25/73 (34.2%); other dose groupings were heterogeneous (2/28, 7.1%) or unclear/not reported (23/229, 10.0%). When grouped by reported timing of initiation, radiotherapy started ≤ 24 h reported 38/225 (16.9%), 24-72 h reported 3/65 (4.6%), > 72 h reported 3/239 (1.3%), and unclear/unreported timing reported 21/227 (9.3%).
Safety outcomes: complications or side effects
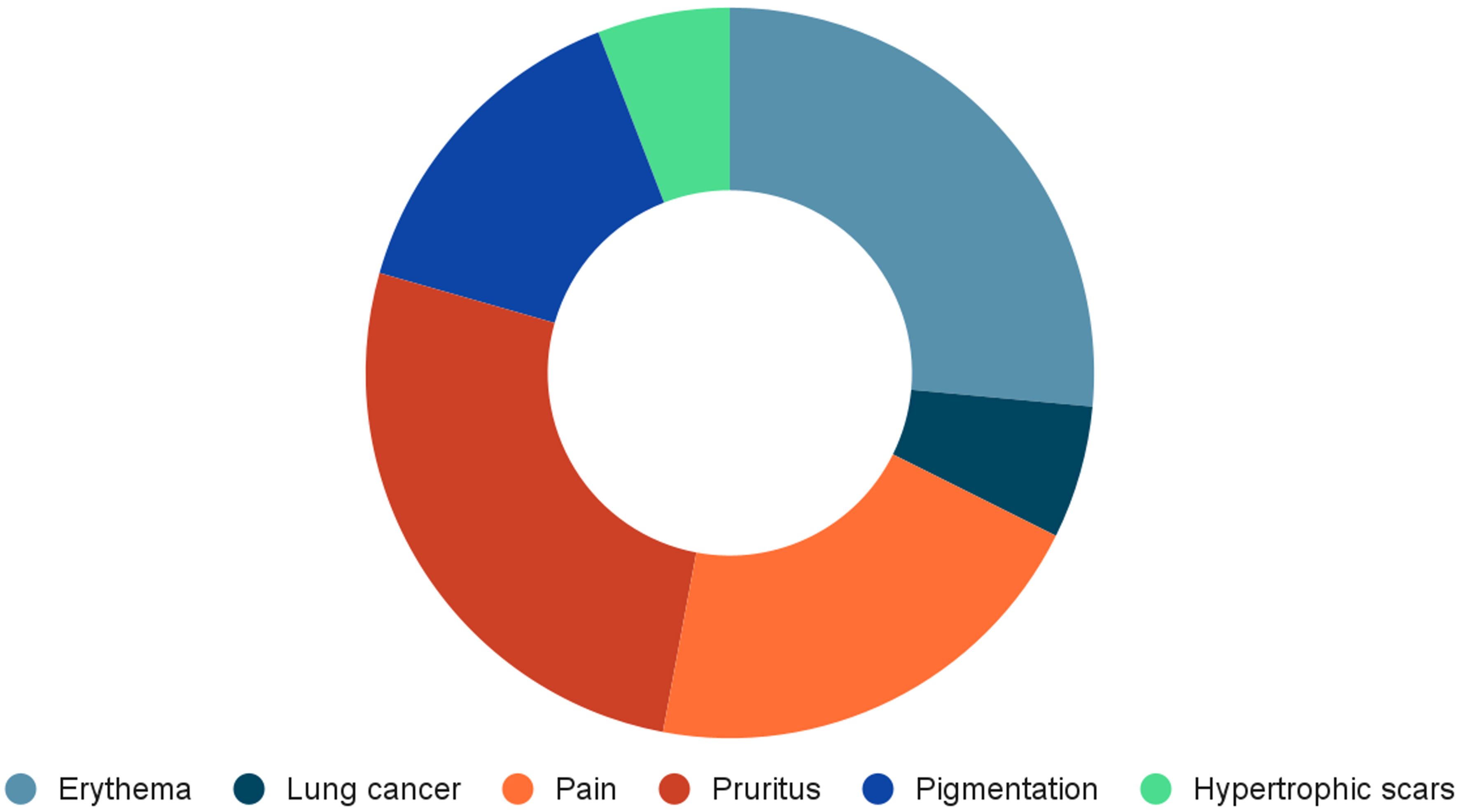
Postoperative adverse effects and complications are summarized in Figure 5. Overall, reported events were relatively infrequent and generally mild across the included studies, suggesting that the interventions were well tolerated in most series. Acute radiotherapy-related effects were most commonly limited to transient skin reactions, including mild erythema and redness, which were typically self-limiting and did not require additional intervention. Late non-malignant complications were variably reported and primarily included skin hyperpigmentation and hypertrophic scarring. These events were more frequently described in excision-based series. In flap-based reports, flap-related complications were uncommon, with a single case of postoperative flap congestion documented. Reporting of long-term and oncologic outcomes was limited. One cohort study described a case of lung carcinoma during follow-up, while most other studies did not systematically report malignancy-related outcomes or were not designed to detect rare late events.
Outcomes: patient satisfaction and aesthetic outcome
High patient satisfaction further supported the effectiveness of combined excision and radiotherapy. Some of the studies investigated cosmetic outcomes by valid scoring of scar outcomes such as Vancouver Scar Scale (VSS), Patient and Observer Scar Assessment Scale (POSAS), and Visual Analog Scale (VAS)[23-25]. Scar appearance, texture, and overall morphology of the ear were markedly better after therapy, according to patients. Detailed outcome measures for individual studies are presented in Table 2, while a consolidated summary of treatment characteristics and variables across all included studies is provided in Supplementary Table 1.
Study intervention and outcomes
| Author (year) | Intervention | Outcome | ||||
| Type | Prescription schedule | Flap | Recurrence rate | Complication | Satisfaction level/Aesthetic outcome | |
| Excision | ||||||
| Kim et al. (2015)[15] | Electron beam | · RT modality: 6-9 MeV electron beam · Dose: 1,500 cGy · Fractions: 3 · Timing: Within 24 h · Treatment duration: 4-5 days | Not applicable | Itching sensations: 5 (32.3%) Pain: 4 (25%) | · Hyperpigmentation · Hypopigmentation · Delayed wound healing · Chondritis · Ulceration (0%) · Cancer (0%) | Satisfaction level of patients: 4.06 ± 1.12 |
| Shen et al. (2015)[16] | Electron beam | · RT modality: 6-7 MeV external beam · Dose: 18 Gy · Fractions: 2 · Timing: 1 week apart | 3 (1.26%) | Hyperpigmentation | Not mentioned | |
| Lee and Park (2015)[17] | Electron beam | · RT modality: electron beam · Energy: 6 MeV · Total dose (Gy): 12-15 · Dose per fraction (Gy): 3-4 · Anatomical site: earlobe and helix · 12 Gy was administered to 9 earlobe lesions and 2 helix lesions; 15 Gy was administered to 13 earlobe lesions and 4 helix lesions | 2 (7.14%) Radiation dose administered to the 2 recurred lesions were 12 Gy | · Mild skin erythema: 2 (7.14%) · Transient hyperpigmentation: 0 (0%) | Not mentioned | |
| Khalid et al. (2018)[9] | Electron beam | · Total dose (Gy): 10 · Fractions: 2 · Dose per fraction (Gy): 5 · Timing: Within 24 h post-excision (two consecutive days) | 17 (56.67%) | Skin redness: 3 (10%) | Not mentioned | |
| Renz et al. (2018)[18] | Linear accelerator-based external beam radiation | · 37 keloids were treated to 20 Gy in 5 fractions and 49 keloids were treated to 12 to 16 Gy in 3 to 4 fractions | 6 (7%) | Lung cancer: 1 | Not mentioned | |
| Liu and Yuan (2019)[19] | Electron beam | · RT modality: Electron beam · Energy: 6 MeV · Total dose (Gy): 15 · Fractions: 3 · Dose per fraction (Gy): 5 · Timing: Initiated on the day of surgery · Treatment duration: 3 days (shortened to 2 days since 2015) | 4 (13.3%) | · Peeling and slight hypertrophic scars: 1 (4.34%) · Mild hyperpigmentation: 3 (13%) · Delayed wound healing, skin ulceration, infections: 0% | Not mentioned | |
| Son et al. (2020)[20] | Sensus SRT-100 irradiator | · 50 kV with 8Gy ± 1Gy single dose | 0% | Mild hyperpigmentation and pruritus | Not mentioned | |
| Hwang et al. (2022)[21] | Electron beam | · RT modality: electron beam · Energy: 6-12 MeV · Total dose (Gy): 15-18 · Fractions: 3 · Dose per fraction (Gy): 5-6 · Regimen: hypofractionated | 5 (5.4%) | N/A | 70.2% of patients declared being either very satisfied (44.7%) or satisfied (25.5%) | |
| Franzetti et al. (2024)[22] | High-dose rate interventional radiotherapy (HDR-IRT) | · Total dose (Gy): 12 · Fractions: 4 · Dose per fraction (Gy): 3 · Schedule: twice daily (BID) | 8 (44.4%) (HR = 6.03, 95%CI: 1.71-21.3, P = 0.005) | Not mentioned | Not mentioned | |
| Flap | ||||||
| Ogawa et al. (2015)[4] | Electron beam | · RT modality: electron beam · Energy: 4 MeV · Total dose (Gy): 15 · Fractions: 3 · Dose per fraction (Gy): 5 · Timing: within 3 days after surgery · Treatment duration: 3 days | N/A | 3 (4.8%) | Wound dehiscence, pigmentation (0%) | Not mentioned |
| Maeda et al. (2019)[6] | Electron beam | · Total dose (Gy): 20 · Fractions: 4 · Dose per fraction (Gy): 5 · Timing: postoperative days 3-4 | Chondrocutaneous bilateral advancement flap | 0% | Not mentioned | Not mentioned |
| Ahmednaji et al. (2023)[23] | Electron beam | · The wound was irradiated for 3 consecutive days (within 24 h after surgery), with a dose of 7 Gy/4 mSv per session. The total radiation dose was 21 Gy | Tongue flap | 4 [9% (mild recurrence)] | Not mentioned | Vancouver Scar Scale and Visual Analog Scale have shown a significant decrease after treatment with a P value of less than 0.05 (Wilcoxon signed-rank test) |
| Li et al. (2024)[24] | Superficial radiotherapy | · First treatment within 24 h: energy 100 kV, dose 4 Gy · Daily dose of 4 Gy on three consecutive days | Cross-flap | 3 (13.5%) | · Infection · Wound dehiscence · Erythema, pigmentation · Secondary malignancies (0%) | PSAS score: 12 (11-14) OSAS score: 10 (10-12) Aesthetic score: 4 (3-4) |
| Park (2024)[25] | Electron beam | · RT modality: electron beam · Energy: 6 MeV · Total dose (Gy): 9.5-10 · Fractions: 1 · Dose per fraction (Gy): 9.5-10 · Timing: within 24 h after surgery | Omega keystone flap | 0% | Postoperative flap congestion: 1 (4%) | POSAS score (pain, itchiness, color, stiffness, thickness, irregularity, vascularity, pigmentation, thickness, relief) significantly improved (P < 0.001) |
Critical appraisal
The studies underwent a critical appraisal in accordance with their respective study designs. Table 3 presents the critical evaluation of the RCT. The key evaluation components for the RoB-2 tools are described in detail in Supplementary Table 2. In addition, Tables 4 and 5 offer detailed critical assessments of the cohort study and the case series studies, respectively. Overall, the included studies demonstrated acceptable methodological quality when assessed using design-appropriate risk-of-bias tools. However, non-randomized studies raised some concerns regarding potential selection and confounding biases, inherent to observational designs. Most cohort studies were rated as having good methodological quality, with minor deductions primarily related to incomplete follow-up. Case series studies generally showed low risk of bias within their respective assessment frameworks, although inconsistent reporting of site- or clinic-level demographic characteristics was noted, which may limit interpretability. Taken together, while the overall risk of bias was considered acceptable, the predominance of non-randomized designs warrants cautious interpretation of the findings. Using the GRADE framework, the certainty of evidence was rated as very low for recurrence outcomes in both treatment groups, driven by the predominance of observational designs, substantial heterogeneity, and imprecision. The certainty of evidence was also rated for complications/side effects and for patient satisfaction/aesthetic outcomes; however, these outcomes were variably defined and inconsistently reported across studies, with limited comparability of measurement instruments and time points, resulting in low-very-low certainty ratings. The detailed GRADE assessments are provided in Supplementary Table 3.
Critical appraisal of randomized controlled trials
| Author (year) | Bias arising from the randomisation process | Bias due to deviations from intended interventions | Bias in classification of interventions | Bias due to missing data | Bias due to measurement of outcomes | Bias in selection of the reported result |
| Khalid et al. (2018)[9] | Low risk | Some concerns | Low risk | Low risk | Some concerns | Some concerns |
Critical appraisal of cohort studies
| Author (year) | S1 | S2 | S3 | S4 | C1 | O1 | O2 | O3 | Total | Quality |
| Kim et al. (2015)[15] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | 8 | Good | |
| Shen et al. (2015)[16] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | 8 | Good | |
| Lee and Park (2015)[17] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | 8 | Good | |
| Ogawa et al. (2015)[4] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | 8 | Good | |
| Renz et al. (2018)[18] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9 | Good |
| Liu and Yuan (2019)[19] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | 8 | Good | |
| Hwang et al. (2022)[21] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | 8 | Good | |
| Ahmednaji et al. (2023)[23] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9 | Good |
| Li et al. (2024)[24] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9 | Good |
| Park (2024)[25] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | ★ | 9 | Good |
| Franzetti et al. (2024)[22] | ★ | ★ | ★ | ★ | ★★ | ★ | ★ | 8 | Good |
Critical appraisal of case series studies
| Question | Maeda et al. (2019)[6] | Son et al. (2020)[20] |
| Were there clear criteria for inclusion in the case series? | Yes | Yes |
| Was the condition measured in a standard, reliable way for all participants included in the case series? | Yes | Yes |
| Were valid methods used for identification of the condition for all participants included in the case series? | Yes | Yes |
| Did the case series have consecutive inclusion of participants? | Unclear | Unclear |
| Did the case series have complete inclusion of participants? | Yes | No |
| Was there clear reporting of the demographics of the participants in the study? | No | Yes |
| Was there clear reporting of the clinical information of the participants? | Yes | Yes |
| Were the outcomes or follow-up results of cases clearly reported? | Yes | Yes |
| Was there clear reporting of the presenting sites’/clinics’ demographic information? | No | No |
| Was statistical analysis appropriate? | Not applicable | Yes |
DISCUSSION
Keloids are defined as raised scars that extend beyond the margin of the original wound, often arising from minor tissue injuries or a genetic predisposition. Histologically, keloids are marked by an excessive aggregation of fibroblasts and type I collagen in the inflammatory reticular dermis. As a result, the balance between collagen deposition and collagen lysis, which is essential in normal wound healing, is not achieved[26]. In their review on the epidemiology of keloids, Huang et al. reported that the average incidence is estimated to be 5%-10% among individuals of African populations, 0%-0.1% in Asian populations, and less than 0.1% in other regions[27].
Keloids can develop on any part of the body, with the most commonly affected areas including the anterior chest, earlobes, upper chest, and cheeks. In contrast, they are rarely found on the eyelids, cornea, palms, mucous membranes, genitalia, and soles[28]. Bayat et al. reported that 42.2% of patients had keloids in multiple anatomical locations, with the earlobe being the most commonly affected site, accounting for 24% of cases. Ear piercing was the most frequent triggering factor, and earlobe keloids typically appeared at an average age of 17.1 years. Ear keloids often cause significant stress for patients due to their visible cosmetic impact, and pose challenges for surgeons because of the complexity of treatment and high risk of recurrence[29].
Management of ear keloids typically involves a combination of nonsurgical and surgical modalities. Various methodologies have been utilized to treat ear keloids, such as surgical excision, intralesional corticosteroids, use of radiotherapy, laser, cryotherapy, or combination therapies. Injection of intralesional corticosteroids is usually utilized for small-sized ear keloids, while large keloids can be surgically excised. If the keloid has a narrow pedicle, complete excision with primary closure can usually be performed. If, however, the stalk is broader, then a conservative approach is advised, with the goal of reducing scar bulk rather than achieving complete excision. Excising large auricular keloids with wide bases can produce large defects that cannot be primarily closed. Tensioning the wound edges can deform the ear. When primary closure cannot be performed and there is a risk of ear deformity due to improper closure, reconstruction with flaps can be utilized. Anatomical subsite within the auricle may act as an important effect modifier in surgical planning and outcomes. Earlobe keloids may often be managed with wedge excision and primary closure, whereas keloids involving cartilaginous auricular subunits (such as the helix or scapha) more frequently require local flap reconstruction to prevent contour deformity and excessive wound tension[7,30,31]. Types of flaps that can be utilized to reconstruct the ear for keloids include tongue flap, fillet flap, cross flap, and keystone flap[32].
Surgical excision alone has been reported to have a high recurrence rate, ranging from 45% to 100%[33]. To reduce this risk, adjuvant radiotherapy may be considered as part of postoperative management. First described by De Beurmann and Gougerot in 1906, radiotherapy has since been widely used as an adjunct to surgical excision. It has demonstrated a success rate of 67% to 98%, with recurrence rates below 10%, significantly lower than excision alone[34]. Postoperative radiotherapy has been established as one of the most effective strategies for preventing keloid recurrence after surgical excision. Its mechanism involves the suppression of fibroblast proliferation, neovascularization, and inflammatory mediators, key elements responsible for the abnormal wound healing characteristic of keloids. Ionizing radiation inhibits the expression of growth factors such as transforming growth factor beta (TGF-β) and platelet-derived growth factor (PDGF), which play central roles in promoting fibroblast activity and collagen deposition. By targeting rapidly proliferating cells at the wound site, especially immature fibroblasts and inflammatory cells, radiotherapy disrupts the cycle of excessive scar formation and helps stabilize the wound healing process before abnormal fibrosis can reinitiate[7,17,35].
Excision methods for keloids vary in technique, resulting in different biological effects on wound healing. A more recent method of excision, keloid intralesional excision (KILE) was introduced and presented a distinct surgical strategy compared to conventional extralesional excision. KILE removes the fibrotic core selectively while preserving the overlying epidermis and dermis. Therefore, it maintained the vascular integrity, reducing wound tension, and potentially modulating the fibroproliferative wound-healing response. A recent meta-analysis by Lawera et al. of 608 patients showed the anti-recurrence effect of KILE with a pooled recurrence rate of approximately 13% following KILE without postoperative radiotherapy, which is substantially lower than recurrence rates reported for excision alone[36]. These findings showed that KILE itself functions as an active recurrence-reducing intervention rather than a variant of excision. Although the meta-analysis by Lawera et al. was not incorporated into the present quantitative analysis due to the differences in study scope and interventions, its findings highlight the influence of the surgical technique of keloid excision on recurrence outcome[36].
There is consensus that the biologically effective dose (BED) for optimal keloid control should exceed
The timing of radiotherapy initiation post-excision is crucial for maximizing its efficacy. Hintz et al. suggested that delaying radiotherapy after excision might benefit keloid treatment by enhancing radiosensitization during the proliferative phase of cell growth, though immediate treatment is effective mainly for earlobe keloids[39]. Conversely, studies show starting radiotherapy within 24 h post-surgery can reduce recurrence by 10% to 23% by inhibiting fibroblast proliferation, leading to the common practice of early initiation to minimize the chance of recurrence. As a result, radiotherapy is now commonly initiated within 24 h after surgical excision[16,40,41].
However, the approach to postoperative radiotherapy for treating keloids has shifted from a generic 15 Gy in three fractions to individualized regimens. Initially, the generic protocol had revealed over 35%-40% recurrence rates, particularly at high-risk areas such as the scapula and chest front. To minimize that, in 2003, they instituted a protocol of dose boosts for higher recurrence sites to 20 Gy over four days and reduced doses for less recurrence sites (e.g., the earlobe) to 10 Gy over two days. That lowered recurrence rates significantly. Subsequent changes in 2013 refined the treatment by lessening sessions: higher recurrence sites received 18 Gy over three days, the earlobe 8 Gy as a single-day dose, and all others 15 Gy over two days. Those changes maintained the treatment at an efficacy level, reaching less than 10% for total recurrence, emphasizing the need for individualization of treatment based on localization of keloids and recurrence risk[7].
In circumstances where adjuvant radiotherapy is not aligned with patient-specific factors or local resource constraints, multiple non-radiation options remain part of contemporary keloid management, including silicone/pressure-based therapies, intralesional agents (most commonly corticosteroids, with adjuvant options such as 5-fluorouracil, bleomycin, or verapamil), cryotherapy/cryosurgery, and laser-based approaches. Evidence syntheses and practice guidelines generally support intralesional corticosteroids as a core modality, with combination regimens (e.g., corticosteroid plus 5-fluorouracil) frequently reported to improve response in refractory lesions and reduce recurrence when used as adjuvant therapy. Laser modalities (e.g., pulsed-dye laser or ablative lasers) are commonly used as adjuncts, either to improve vascularity/erythema and scar texture or to enhance drug penetration when combined with intralesional or topical therapies. Cryotherapy has also been described as an effective option and is recommended in some guidelines particularly in combination with intralesional corticosteroids. Nevertheless, the comparative effectiveness of these alternatives vs. adjuvant radiotherapy for auricular keloids remains heterogeneous across the literature, so selection should be individualized based on lesion characteristics, prior recurrence, anatomic subunit, access to modalities, and patient preference within shared decision-making[3,42,43].
The systematic review and meta-analysis evaluated recurrence rates following surgical excision of auricular keloids with and without reconstruction by flaps in combination with postoperative radiotherapy. In this review, the pooled recurrence proportion was 7.8% (95%CI: 0.0%-16.0%) in studies of excision with flap reconstruction plus postoperative radiotherapy and 20.0% (95%CI: 5.2%-34.8% in studies of excision plus postoperative radiotherapy without flap. Differences in the anatomical distribution of lesions across studies may partially confound the apparent benefit observed in the flap subgroup. The excision group also demonstrated higher heterogeneity (I2 = 90%) than the flap subgroup (I2 = 72%), suggesting greater variability among studies evaluating excision-based approaches. This heterogeneity may be attributable to variations in surgical techniques, patient selection, radiotherapy protocols, and follow-up duration. Accordingly, technique-based comparisons should be interpreted cautiously, particularly in the absence of direct comparative designs[23-25]. Descriptive subgroup summaries show variability in reported recurrence by modality, dose schedules, and treatment timing. Electron-beam radiotherapy was the most frequently reported modality, while non-electron techniques were less commonly represented and heterogeneous. Across the included evidence base, radiotherapy regimens and reporting practices varied substantially, and key clinical factors that may drive treatment selection (e.g., lesion complexity, prior recurrence, auricular subunit, and wound tension) were inconsistently captured. As a result, the subgroup patterns should be interpreted as study-level signals rather than comparative effects, reinforcing the need for standardized reporting of radiotherapy parameters and recurrence definitions in future controlled studies.
The included studies reported minimal and generally manageable complications. Reported adverse effects in the excision group included hyperpigmentation and hypertrophic scarring, while in flap-based reports, a single instance of postoperative flap congestion was described. Reconstructive flap techniques are used to achieve stable wound closure and redistribute wound tension, which may influence postoperative healing and scar behavior. Radiotherapy was generally well tolerated in the majority of studies, with only mild skin reactions reported in selected cases. However, safety reporting in the included literature primarily reflects short-term toxicity and selected late benign effects, with heterogeneous definitions and follow-up duration across studies. While acute reactions were generally mild in reported series, the available evidence base is not designed to quantify rare late outcomes. In particular, radiation-associated malignancy risk cannot be reliably estimated from these studies due to limited follow-up, inconsistent oncologic outcome reporting, and the rarity of such events. Although one study reported a case of lung carcinoma following radiotherapy, no such events were observed in other included studies.
Postoperative radiotherapy following excision surgery may offer advantages in reducing recurrence and improving patient outcomes. Early administration of radiotherapy, particularly within 24 h after surgery, has been suggested to interfere with the early inflammatory cascade involved in keloid pathogenesis. When combined with flap reconstruction, which redistributes mechanical tension away from wound edges, this approach may address both biological and biomechanical contributors to keloid recurrence. Patients treated with flap reconstruction have also reported less skin irritation and discoloration, which may reflect improved tissue perfusion and vascularity within the flap, potentially enhancing radiation tolerance[24,44]. Consequently, this combined approach not only mitigates the risk of recurrence but also promotes improved wound healing and aesthetic satisfaction. However, interpretation of these findings should consider the very low certainty of evidence for recurrence outcomes as assessed by the GRADE framework. Although pooled analyses suggest lower recurrence following flap reconstruction with adjuvant radiotherapy, the predominance of non-randomized studies, substantial heterogeneity, and limited adjustment for confounding preclude definitive causal inference. Accordingly, these results should be interpreted as hypothesis-generating and warrant confirmation in well-designed comparative studies.
Strengths and limitations
A key strength of this review lies in the inclusion of diverse study designs encompassing multiple populations, anatomical sites, and surgical approaches, which provides a broad overview of contemporary clinical practice in auricular keloid management. A comprehensive and up-to-date search strategy was applied across multiple databases with rigorous quality appraisal, and methodological consistency was enhanced by restricting the quantitative synthesis to radiation-based modalities only. By synthesizing data from a large cumulative sample of 735 keloids, this analysis offers a comprehensive assessment of recurrence rates, complications, and patient-reported outcomes, thereby enhancing the external validity of the findings. The use of random-effects models appropriately accounted for clinical and methodological heterogeneity, yielding conservative pooled estimates that better reflect real-world variability. Additionally, the inclusion of both excision-based and flap-based interventions allows for a nuanced evaluation of surgical strategies when combined with postoperative radiotherapy.
Nevertheless, several limitations should be acknowledged. Anatomical location (earlobe vs. cartilaginous auricle) was inconsistently reported across studies, and subgroup sample sizes were small, precluding robust location-stratified analyses and potentially confounding comparisons between excision-based and flap-based approaches. In addition, most primary studies reported outcomes per keloid rather than per patient, and patient-level data were unavailable to adjust for potential within-patient clustering when multiple lesions originated from the same individual; consequently, variance may be underestimated. Substantial variability in radiotherapy dose, fractionation, and timing of initiation, together with incomplete reporting, also limited meaningful exploratory analyses based on BED or treatment timing. Moreover, stratified analyses by radiotherapy modality, biologically effective dose BED, timing of initiation, primary versus recurrent lesions, and auricular subunits were not feasible because these variables were inconsistently reported across studies. Recurrence outcomes were also rarely reported in a comparable manner, limiting subgroup pooling or meta-regression and increasing the risk of underpowered analyses and misclassification. Restricting eligibility by publication year may also have limited the inclusion of older cohorts with very long-term follow-up, which is particularly relevant when considering rare oncologic outcomes after radiotherapy. Moreover, the absence of internal non-radiotherapy control groups and the inclusion of heterogeneous excision techniques limited causal attribution of recurrence reduction to radiotherapy alone. Consistent with the GRADE assessment for recurrence outcomes, the certainty of evidence was rated as very low, reflecting the predominance of observational designs, substantial heterogeneity, imprecision, and variable reporting of recurrence definitions, alongside limited availability of patient-level data. These factors may introduce residual confounding and reduce the precision and generalizability of the pooled estimates. Moreover, GRADE ratings for secondary outcomes (complications/side effects and patient-reported satisfaction/cosmesis) were limited by variable outcome definitions, inconsistent reporting, heterogeneous measurement instruments, and sparse extractable data across studies; therefore, certainty ratings should be interpreted cautiously.
Conclusion
This systematic review and meta-analysis indicate that, across the included literature, postoperative radiotherapy following excision of auricular keloids is associated with relatively low reported recurrence, generally mild short-term adverse effects, and frequently favorable cosmetic outcomes and patient satisfaction. However, interpretation is limited by substantial between-study heterogeneity, predominantly non-comparative observational designs, and limited long-term safety data. Future prospective, well-designed studies with standardized treatment protocols, comprehensive reporting of radiotherapy parameters, and extended follow-up are needed to better define comparative effectiveness and long-term outcomes.
DECLARATIONS
Acknowledgments
We acknowledge the use of Canva (Canva Pty Ltd., Sydney, Australia) for the design of the graphical abstract.
Authors’ contributions
Contributed to conception and design, data acquisition, analysis, and interpretation: Putri NM, Tunjung N
Drafted and revised the manuscript: Putri NM, Tunjung N
Supervised and managed the project: Putri NM, Tunjung N
Contributed to data acquisition and investigation: Sanjaya IS, Oklia S
Assisted in manuscript writing and review, editing, and proofreading: Sanjaya IS, Oklia S
All authors approved the final version.
All authors agreed to be accountable for all aspects of the work.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tools GPT-5 (OpenAI; released August 7, 2025) and Grammarly (version 1.2.183; released August 6, 2025) were used solely for language editing and grammar checking. These tools did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
This research is funded by the Indonesian Endowment Fund for Education (LPDP) on behalf of the Indonesian Ministry of Higher Education, Science and Technology and managed under the EQUITY Program (Contract No. 4302/B3/DT.03.08/2025 and 573/PKS/R/UI/2025).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Betarbet U, Blalock TW. Keloids: a review of etiology, prevention, and treatment. J Clin Aesthet Dermatol. 2020;13:33-43.
2. Berman B, Maderal A, Raphael B. Keloids and hypertrophic scars: pathophysiology, classification, and treatment. Dermatol Surg. 2017;43:S3-18.
3. Walsh LA, Wu E, Pontes D, et al. Keloid treatments: an evidence-based systematic review of recent advances. Syst Rev. 2023;12:42.
4. Ogawa R, Akaishi S, Dohi T, Kuribayashi S, Miyashita T, Hyakusoku H. Analysis of the surgical treatments of 63 keloids on the cartilaginous part of the auricle: effectiveness of the core excision method. Plast Reconstr Surg. 2015;135:868-75.
5. Park TH, Seo SW, Kim JK, Chang CH. Earlobe keloids: classification according to gross morphology determines proper surgical approach. Dermatol Surg. 2012;38:406-12.
6. Maeda T, Hayashi T, Murao N, Yamamoto Y. Chondrocutaneous bilateral advancement flap with postoperative radiation therapy for a helical rim keloid. Aesthetic Plast Surg. 2019;43:658-62.
7. Ogawa R, Tosa M, Dohi T, Akaishi S, Kuribayashi S. Surgical excision and postoperative radiotherapy for keloids. Scars Burn Heal. 2019;5:2059513119891113.
8. Ogawa R, Huang C, Akaishi S, et al. Analysis of surgical treatments for earlobe keloids: analysis of 174 lesions in 145 patients. Plast Reconstr Surg. 2013;132:818e-25.
9. Khalid FA, Farooq UK, Saleem M, et al. The efficacy of excision followed by intralesional 5-fluorouracil and triamcinolone acetonide versus excision followed by radiotherapy in the treatment of ear keloids: a randomized control trial. Burns. 2018;44:1489-95.
10. Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
11. CEBM. Oxford centre for evidence-based medicine: levels of evidence (March 2009). Available from: https://www.cebm.ox.ac.uk/resources/levels-of-evidence/oxford-centre-for-evidence-based-medicine-levels-of-evidence-march-2009. [Last accessed on 12 Mar 2026].
12. Sterne JAC, Savović J, Page MJ, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898.
13. The Ottawa Hospital. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Available from: https://ohri.ca/en/who-we-are/core-facilities-and-platforms/ottawa-methods-centre/newcastle-ottawa-scale. [Last accessed on 12 Mar 2026].
14. Munn Z, Barker TH, Moola S, et al. Methodological quality of case series studies: an introduction to the JBI critical appraisal tool. JBI Evid Synth. 2020;18:2127-33.
15. Kim K, Son D, Kim J. Radiation therapy following total keloidectomy: a retrospective study over 11 years. Arch Plast Surg. 2015;42:588-95.
16. Shen J, Lian X, Sun Y, et al. Hypofractionated electron-beam radiation therapy for keloids: retrospective study of 568 cases with 834 lesions. J Radiat Res. 2015;56:811-7.
17. Lee SY, Park J. Postoperative electron beam radiotherapy for keloids: treatment outcome and factors associated with occurrence and recurrence. Ann Dermatol. 2015;27:53-8.
18. Renz P, Hasan S, Gresswell S, Hajjar RT, Trombetta M, Fontanesi J. Dose effect in adjuvant radiation therapy for the treatment of resected keloids. Int J Radiat Oncol Biol Phys. 2018;102:149-54.
19. Liu CL, Yuan ZY. Retrospective study of immediate postoperative electron radiotherapy for therapy-resistant earlobe keloids. Arch Dermatol Res. 2019;311:469-75.
20. Son Y, Phillips EON, Price KM, et al. Treatment of keloids with a single dose of low-energy superficial X-ray radiation to prevent recurrence after surgical excision: an in vitro and in vivo study. J Am Acad Dermatol. 2020;83:1304-14.
21. Hwang NH, Lee NK, Chae JH, Park SH, Yoon ES. The efficacy of CT-based conformal electron beam radiation therapy after keloid excision. Dermatol Surg. 2022;48:435-40.
22. Franzetti J, Durante S, Mastroleo F, et al. Post-operative KEloids iRradiation (POKER): does the surgery/high-dose interventional radiotherapy association make a winning hand? Radiol Med. 2024;129:328-34.
23. Ahmednaji NM, Wu XY, Wang YX, Chen XD. Surgical core excision with tongue flap closure in combination with electron beam radiotherapy in the treatment of ear keloids. Dermatol Surg. 2023;49:S58-63.
24. Li Q, Chen Z, Zou X, et al. Effectiveness of postoperative superficial radiotherapy following the keloid-cross-flap surgery for auricular keloid: a prospective cohort study. Radiother Oncol. 2024;199:110425.
25. Park TH. Outcomes of omega variant keystone flap in conjunction with single fraction radiotherapy for helical keloids: a comprehensive one-year follow-up study. J Plast Reconstr Aesthet Surg. 2024;90:359-66.
26. Chung KC. Grabb and Smith’s plastic surgery. 9th ed. Lippincott Williams & Wilkins Publisher: Philadelphia; 2014. Available from: https://www.wolterskluwer.com/en/solutions/ovid/grabb-and-smiths-plastic-surgery-3467. [Last accessed on 12 Mar 2026].
27. Huang C, Wu Z, Du Y, Ogawa R. The epidemiology of keloids. In: Téot L, Mustoe TA, Middelkoop E, Gauglitz GG, Editors. Textbook on scar management: state of the art management and emerging technologies. Cham (CH): Springer; 2020.
28. Gauglitz GG, Korting HC, Pavicic T, Ruzicka T, Jeschke MG. Hypertrophic scarring and keloids: pathomechanisms and current and emerging treatment strategies. Mol Med. 2011;17:113-25.
29. Bayat A, Arscott G, Ollier WE, McGrouther DA, Ferguson MW. Keloid disease: clinical relevance of single versus multiple site scars. Br J Plast Surg. 2005;58:28-37.
30. Kwek JWM, Lee TS, Loh ICY. Helical rim advancement - a technique to avoid keloid recurrence. JPRAS Open. 2019;19:61-6.
31. Park TH. Aesthetic reconstruction of auricular keloids with a novel hemi-keystone flap. Aesthetic Plast Surg. 2022;46:2807-13.
32. Lee JH, Lee BH, Chang J. Surgical treatment of keloid scars on the ear: the usefulness of the fillet flap. J Wound Manag Res. 2024;20:63-8.
33. McGoldrick RB, Theodorakopoulou E, Azzopardi EA, Murison M. Lasers and ancillary treatments for scar management Part 2: keloid, hypertrophic, pigmented and acne scars. Scars Burn Heal. 2017;3:2059513116689805.
34. De Beurmann, Gougerot H. Cheloides des muqueuses. Ann Derm Syph. 1906;7:151-2. Available from: https://commons.wikimedia.org/wiki/File:Annales_de_dermatologie_et_de_syphiligraphie_(IA_annalesdedermat00syphgoog).pdf. [Last accessed on 17 Mar 2026].
35. Lee JW, Seol KH. Adjuvant radiotherapy after surgical excision in keloids. Medicina. 2021;57:730.
36. Lawera NG, Madzia J, Casey LC, et al. Keloid intralesional excision reduces recurrence: a meta-analytic study of the available literature on 608 keloids. Plast Reconstr Surg Glob Open. 2024;12:e5652.
37. Kal HB, Veen RE. Biologically effective doses of postoperative radiotherapy in the prevention of keloids. Dose-effect relationship. Strahlenther Onkol. 2005;181:717-23.
38. Flickinger JC. A radiobiological analysis of multicenter data for postoperative keloid radiotherapy. Int J Radiat Oncol Biol Phys. 2011;79:1164-70.
40. van Leeuwen MC, Stokmans SC, Bulstra AE, et al. Surgical excision with adjuvant irradiation for treatment of keloid scars: a systematic review. Plast Reconstr Surg Glob Open. 2015;3:e440.
41. Jiang P, Baumann R, Dunst J, et al. Perioperative interstitial high-dose-rate brachytherapy for the treatment of recurrent keloids: feasibility and early results. Int J Radiat Oncol Biol Phys. 2016;94:532-6.
42. Kim SW. Management of keloid scars: noninvasive and invasive treatments. Arch Plast Surg. 2021;48:149-57.
43. Gold MH, Berman B, Clementoni MT, Gauglitz GG, Nahai F, Murcia C. Updated international clinical recommendations on scar management: part 1--evaluating the evidence. Dermatol Surg. 2014;40:817-24.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].